High-Entropy Spinel Oxides Produced via Sol-Gel and Electrospinning and Their Evaluation as Anodes in Li-Ion Batteries
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Chemicals
2.2. Syntheses of HESO-Samples
2.2.1. Syntheses by the Sol-Gel Method
2.2.2. Syntheses by Electrospinning
2.3. Characterization
2.4. Electrochemical Characterization
3. Results
3.1. Morphology
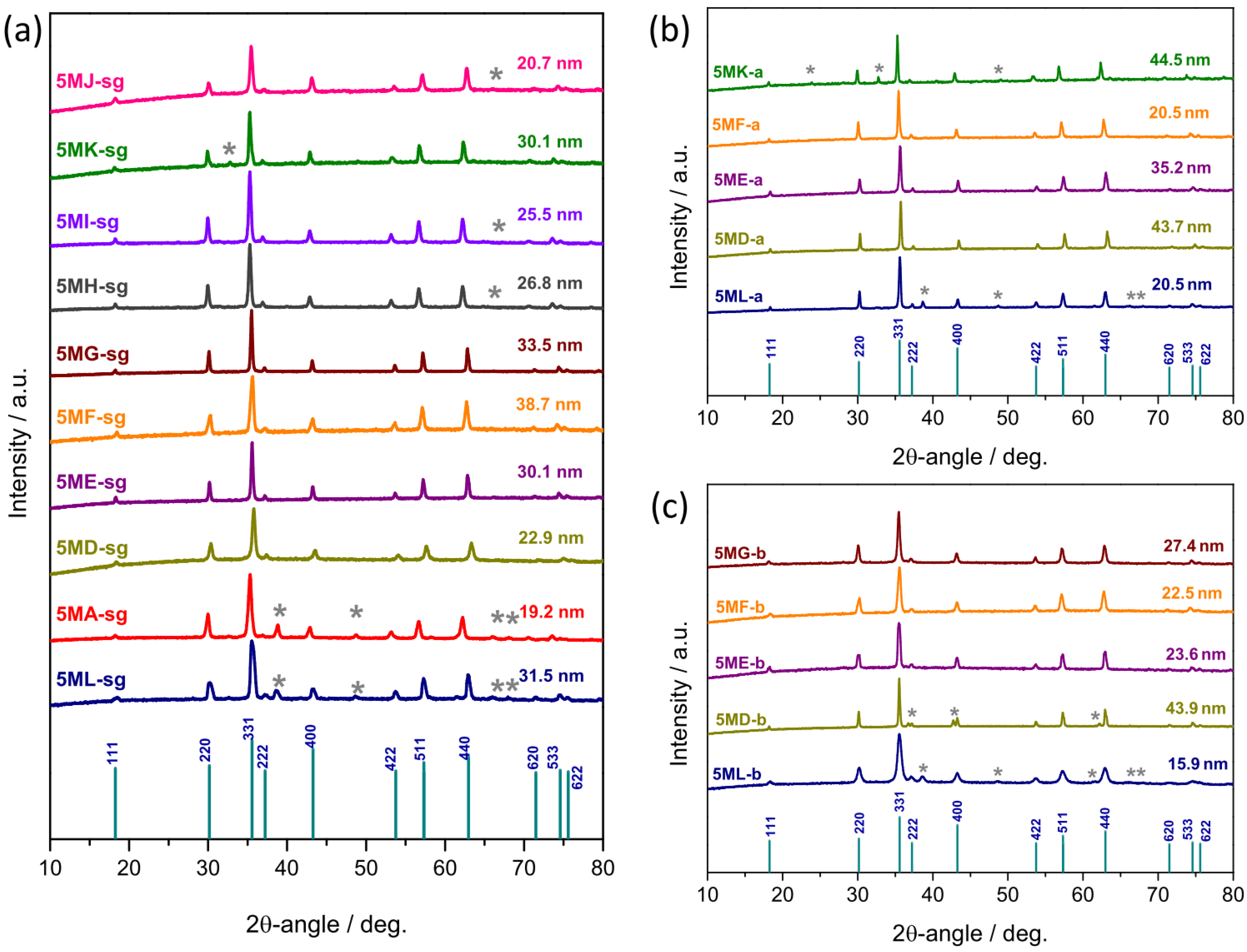
3.2. Phase of the Oxide
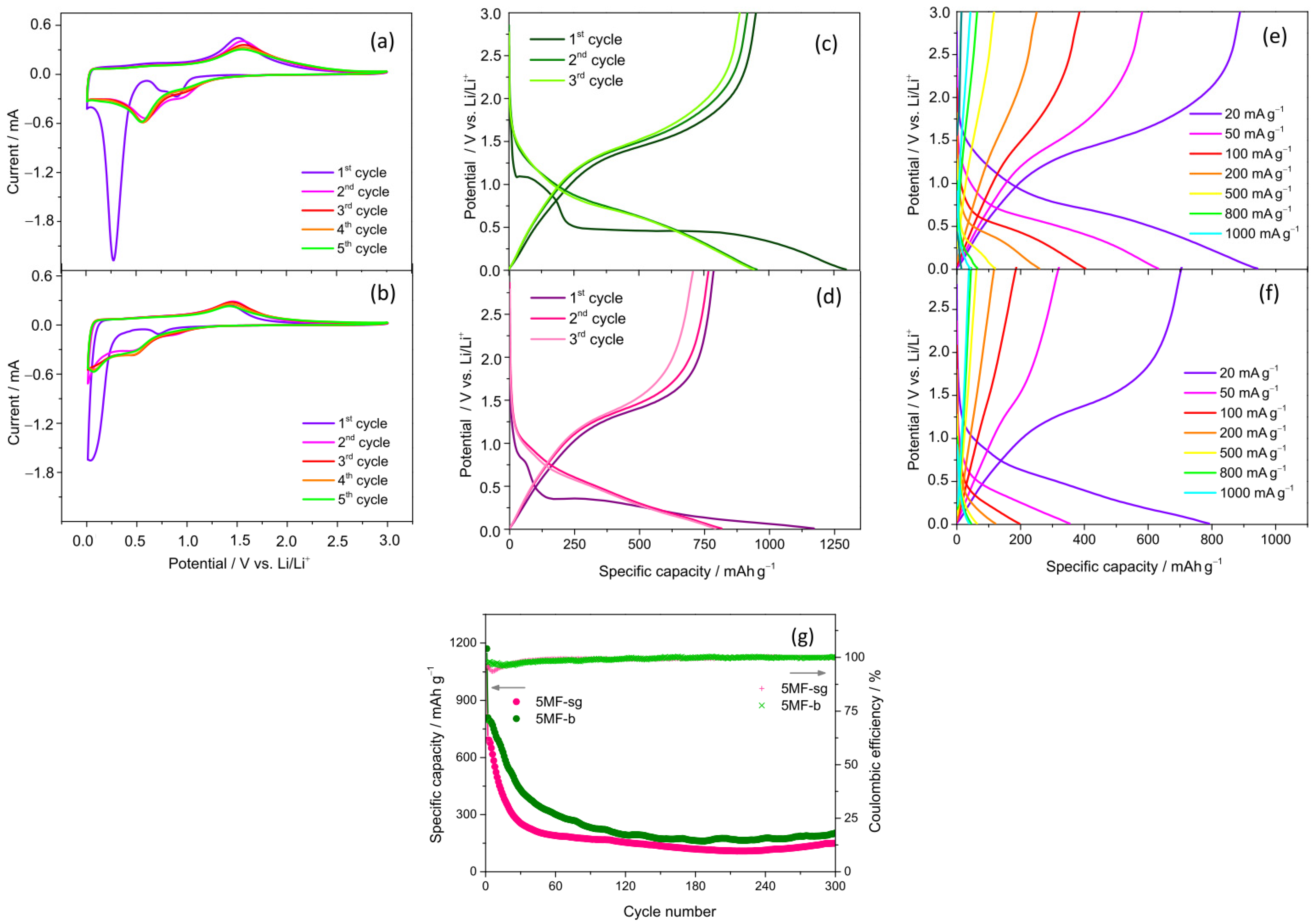
3.3. Electrochemical Properties
3.4. Strategies to Improve the Electrochemical Performance of the Investigated Samples
3.4.1. Incorporating Lithium in the HESO Lattice
3.4.2. Incorporating the Oxide in a Carbonaceous Matrix
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Miracle, D.B.; Senkov, O.N. A critical review of high entropy alloys and related concepts. Acta Mater. 2017, 122, 448–511. [Google Scholar] [CrossRef]
- Yeh, J.W.; Lin, S.J. Breakthrough applications of high-entropy materials. J. Mater. Res. 2018, 33, 3129–3137. [Google Scholar] [CrossRef]
- Musicó, B.L.; Gilbert, D.; Ward, T.Z.; Page, K.; George, E.; Yan, J.; Mandrus, D.; Keppens, V. The emergent field of high entropy oxides: Design, prospects, challenges, and opportunities for tailoring material properties. APL Mater. 2020, 8, 040912. [Google Scholar] [CrossRef]
- Oses, C.; Toher, C.; Curtarolo, S. High-entropy ceramics. Nat. Rev. Mater. 2020, 5, 295–309. [Google Scholar] [CrossRef]
- Braic, V.; Vladescu, A.; Balaceanu, M.; Luculescu, C.R.; Braic, M. Nanostructured multi-element (TiZrNbHfTa)N and (TiZrNbHfTa)C hard coatings. Surf. Coat. Technol. 2012, 211, 117–121. [Google Scholar] [CrossRef]
- Jin, T.; Sang, X.; Unocic, R.R.; Kinch, R.T.; Liu, X.; Hu, J.; Liu, H.; Dai, S. Mechanochemical-assisted synthesis of high-entropy metal nitride via a soft urea strategy. Adv. Mater. 2018, 30, 1707512. [Google Scholar] [CrossRef]
- Castle, E.; Csanádi, T.; Grasso, S.; Dusza, J.; Reecet, M. Processing and properties of high-entropy ultra-high temperature carbides. Sci. Rep. 2018, 8, 8609. [Google Scholar] [CrossRef]
- Zhou, J.; Zhang, J.; Zhang, F.; Niu, B.; Lei, L.; Wang, W. High-entropy carbide: A novel class of multicomponent ceramics. Ceram. Int. 2018, 44, 22014–22018. [Google Scholar] [CrossRef]
- Gild, J.; Zhang, Y.; Harrington, T.; Jiang, S.; Hu, T.; Quinn, M.C.; Mellor, W.M.; Zhou, N.; Vecchio, K.; Luo, J. High-entropy metal diborides: A new class of high-entropy materials and a new type of ultrahigh temperature ceramics. Sci. Rep. 2016, 6, 37946. [Google Scholar] [CrossRef]
- Zhang, R.Z.; Gucci, F.; Zhu, H.; Chen, K.; Reece, M.J. Data-driven design of ecofriendly thermoelectric high-entropy sulfides. Inorg. Chem. 2018, 57, 13027–13033. [Google Scholar] [CrossRef]
- Rost, C.M.; Sachet, E.; Borman, T.; Moballegh, A.; Dickey, E.C.; Hou, D.; Jones, J.L.; Curtarolo, S.; Maria, J.-P. Entropy-stabilized oxides. Nat. Commun. 2015, 6, 8485. [Google Scholar] [CrossRef]
- Sarkar, A.; Wang, Q.; Schiele, A.; Chellali, M.R.; Bhattacharya, S.S.; Wang, D.; Brezesinski, T.; Hahn, H.; Velasco, L.; Breitung, B. High-entropy oxides: Fundamental aspects and electrochemical properties. Adv. Mater. 2019, 31, 1806236. [Google Scholar] [CrossRef]
- Dabrowa, J.; Stygar, M.; Mikuła, A.; Knapik, A.; Mroczka, K.; Tejchman, W.; Danielewski, M.; Martin, M. Synthesis and microstructure of the (Co, Cr, Fe, Mn, Ni)3O4 high entropy oxide characterized by spinel structure. Mater. Lett. 2018, 216, 32–36. [Google Scholar] [CrossRef]
- Mao, A.; Quan, F.; Xiang, H.; Zhang, Z.; Kuramoto, K.; Xia, A. Facile synthesis and ferrimagnetic property of spinel (CoCrFeMnNi)3O4 high-entropy oxide nanocrystalline powder. J. Mol. Struct. 2019, 1194, 11–18. [Google Scholar] [CrossRef]
- Biesuz, M.; Spiridigliozzi, L.; Dell’Agli, G.; Bortolotti, M.; Sglavo, V.M. Synthesis and sintering of (Mg, Co, Ni, Cu, Zn)O entropy-stabilized oxides obtained by wet chemical methods. J. Mater. Sci. 2018, 53, 8074–8085. [Google Scholar] [CrossRef]
- Qiu, N.; Chen, H.; Yang, Z.; Sun, S.; Wang, Y.; Cui, Y. A high entropy oxide (Mg0.2Co0.2Ni0.2Cu0.2Zn0.2O) with superior lithium storage performance. J. Alloy. Compd. 2019, 777, 767–774. [Google Scholar] [CrossRef]
- Sarkar, A.; Breitung, B.; Hahn, H. High entropy oxides: The role of entropy, enthalpy and synergy. Scr. Mater. 2020, 187, 43–48. [Google Scholar] [CrossRef]
- Grzesik, Z.; Smoła, G.; Miszczak, M.; Stygar, M.; Dabrowa, J.; Zajusz, M.; Swierczek, K.; Danielewski, M. Defect structure and transport properties of (Co, Cr, Fe, Mn, Ni)3O4 spinel-structured high entropy oxide. J. Eur. Ceram. Soc. 2020, 40, 835–839. [Google Scholar] [CrossRef]
- Chen, K.; Pei, X.; Tang, L.; Cheng, H.; Li, Z.; Li, C.; Zhang, X.; An, L. A five-component entropy-stabilized fluorite oxide. J. Eur. Ceram. Soc. 2018, 38, 4161–4164. [Google Scholar] [CrossRef]
- Anandkumar, M.; Bhattacharya, S.; Deshpande, A.S. Low temperature synthesis and characterization of single phase multi-component fluorite oxide nanoparticle sols. RSC Adv. 2019, 9, 26825–26830. [Google Scholar] [CrossRef]
- Zhao, C.; Ding, F.; Lu, Y.; Chen, L.; Hu, Y.-S. High-entropy layered oxide cathodes for sodium-ion batteries. Angew. Chem. Int. Ed. 2020, 59, 264–269. [Google Scholar] [CrossRef]
- Teng, Z.; Zhu, L.; Tan, Y.; Zeng, S.; Xia, Y.; Wang, Y.; Zhang, H. Synthesis and structures of high-entropy pyrochlore oxides. J. Eur. Ceram. Soc. 2020, 40, 1639–1643. [Google Scholar] [CrossRef]
- Jiang, S.; Hu, T.; Gild, J.; Zhou, N.; Nie, J.; Qin, M.; Harrington, T.; Vecchio, K.; Luo, J. A new class of high-entropy perovskite oxides. Scr. Mater. 2018, 142, 116–120. [Google Scholar] [CrossRef]
- Sarkar, A.; Djenadic, R.; Wang, D.; Hein, C.; Kautenburger, R.; Clemens, O.; Hahn, H. Rare earth and transition metal based entropy stabilised perovskite type oxides. J. Eur. Ceram. Soc. 2018, 38, 2318–2327. [Google Scholar] [CrossRef]
- Okejiri, F.; Zhang, Z.; Liu, J.; Liu, M.; Yang, S.; Dai, S. Room-temperature synthesis of high-entropy perovskite oxide nanoparticle catalysts through ultrasonication-based method. ChemSusChem 2020, 13, 111–115. [Google Scholar] [CrossRef]
- Bérardan, D.; Franger, S.; Dragoe, D.; Meena, A.K.; Dragoe, N. Colossal dielectric constant in high entropy oxides. Phys. Status Solidi RRL 2016, 10, 328–333. [Google Scholar] [CrossRef]
- Sarkar, A.; Loho, C.; Velasco, L.; Thomas, T.; Bhattacharya, S.S.; Hahn, H.; Djenadic, R. Multicomponent equiatomic rare earth oxides with a narrow band gap and associated praseodymium multivalency. Dalton Trans. 2017, 46, 12167–12176. [Google Scholar] [CrossRef]
- Bérardan, D.; Franger, S.; Meena, A.K.; Dragoe, N. Room temperature lithium superionic conductivity in high entropy oxides. J. Mater. Chem. A 2016, 4, 9536–9541. [Google Scholar] [CrossRef]
- Chen, H.; Fu, J.; Zhang, P.; Peng, H.; Abney, C.W.; Jie, K.; Liu, X.; Chi, M.; Dai, S. Entropy-stabilized metal oxide solid solutions as CO oxidation catalysts with high-temperature stability. J. Mater. Chem. A 2018, 6, 11129–11133. [Google Scholar] [CrossRef]
- Gao, Y.; Liu, Y.; Yu, H.; Zou, D. High-entropy oxides for catalysis: Status and perspectives. Appl. Catal. A Gen. 2022, 631, 118478. [Google Scholar] [CrossRef]
- Sarkar, A.; Velasco, L.; Wang, D.; Wang, Q.; Talasila, G.; de Biasi, L.; Kübel, C.; Brezesinski, T.; Bhattacharya, S.S.; Hahn, H.; et al. High entropy oxides for reversible energy storage. Nat. Commun. 2018, 9, 3400. [Google Scholar] [CrossRef]
- Chen, T.-Y.; Wang, S.-Y.; Kuo, C.-H.; Huang, S.-C.; Lin, M.-H.; Li, C.-H.; Chen, H.-Y.T.; Wang, C.-C.; Liao, Y.-F.; Lin, C.-C.; et al. In operando synchrotron X-ray studies of a novel spinel (Ni0.2Co0.2Mn0.2Fe0.2Ti0.2)3O4 high-entropy oxide for energy storage applications. J. Mater. Chem. A 2020, 8, 21756–21770. [Google Scholar] [CrossRef]
- Wang, S.-Y.; Chen, T.-Y.; Kuo, C.-H.; Lin, C.-C.; Huang, S.-C.; Lin, M.-H.; Wang, C.-C.; Chen, H.-Y. Operando synchrotron transmission X-ray microscopy study on (Mg, Co, Ni, Cu, Zn)O high-entropy oxide anodes for lithium-ion batteries. Mater. Chem. Phys. 2021, 274, 125105. [Google Scholar] [CrossRef]
- Guo, H.-X.; Wang, W.-M.; He, C.-Y.; Liu, B.-H.; Yu, D.-M.; Liu, G.; Gao, X.-H. Entropy-assisted high-entropy oxide with a spinel structure toward high-temperature infrared radiation materials. ACS Appl. Mater. Interfaces 2022, 14, 1950–1960. [Google Scholar] [CrossRef]
- Zhang, J.; Yan, J.; Calder, S.; Zheng, Q.; McGuire, M.A.; Abernathy, D.L.; Ren, Y.; Lapidus, S.H.; Page, K.; Zheng, H.; et al. Long-range antiferromagnetic order in a rocksalt high entropy oxide. Chem. Mater. 2019, 31, 3705–3711. [Google Scholar] [CrossRef]
- Triolo, C.; Xu, W.; Petrovičovà, B.; Pinna, N.; Santangelo, S. Evaluation of entropy-stabilised (Mg0.2Co0.2Ni0.2Cu0.2Zn0.2)O oxides produced via solvothermal method or electrospinning as anodes in lithium ion batteries. Adv. Funct. Mater. 2022, 2202892. [Google Scholar] [CrossRef]
- Liu, J.; Ma, C.; Wang, L.; Ren, K.; Ran, H.; Feng, D.; Du, H.; Wang, Y. Single-phase formation mechanism and dielectric properties of sol-gel-derived Ba(Ti0.2Zr0.2Sn0.2Hf0.2Ce0.2)O3 high-entropy ceramics. J. Mater. Sci. Technol. 2022, 130, 103–111. [Google Scholar] [CrossRef]
- Li, Z.; Zhou, F.; Xu, B.; Guo, D. Characterization of novel high-entropy (La0.2Nd0.2Sm0.2Dy0.2Yb0.2)2Zr2O7 electrospun ceramic nanofibers. Ceram. Int. 2022, 48, 12074–12078. [Google Scholar] [CrossRef]
- Xing, Y.; Dan, W.; Fan, Y.; Li, X. Low temperature synthesis of high-entropy (Y0.2Yb0.2Sm0.2Eu0.2Er0.2)2O3 nanofibers by a novel electrospinning method. J. Mater. Sci. Technol. 2022, 103, 215–220. [Google Scholar] [CrossRef]
- Wang, G.; Qin, J.; Feng, Y.; Feng, B.; Yang, S.; Wang, Z.; Zhao, Y.; Wei, J. Sol–gel synthesis of spherical mesoporous high-entropy oxides. ACS Appl. Mater. Interfaces 2020, 12, 45155–45164. [Google Scholar] [CrossRef]
- Santangelo, S. Electrospun nanomaterials for energy applications: Recent advances. Appl. Sci. 2019, 9, 1049. [Google Scholar] [CrossRef]
- Pantò, F.; Fan, Y.; Frontera, P.; Stelitano, S.; Fazio, E.; Patanè, S.; Marelli, M.; Antonucci, P.; Neri, F.; Pinna, N.; et al. Are electrospun carbon/metal oxide composite fibers relevant electrode materials for Li-Ion batteries? J. Electrochem. Soc. 2016, 163, A2930. [Google Scholar] [CrossRef]
- Pantò, F.; Fan, Y.; Stelitano, S.; Fazio, E.; Patanè, S.; Frontera, P.; Antonucci, P.; Pinna, N.; Santangelo, S. Are electrospun fibrous membranes relevant electrode materials for li-ion batteries? The case of the C/Ge/GeO2 composite fibers. Adv. Funct. Mater. 2018, 28, 1800938. [Google Scholar] [CrossRef]
- Han, X.; Russo, P.A.; Goubard-Bretesché, N.; Patanè, S.; Santangelo, S.; Zhang, R.; Pinna, N. Exploiting the condensation reactions of acetophenone to engineer carbon-encapsulated Nb2O5 nanocrystals for high-performance Li and Na energy storage systems. Adv. Energy Mater. 2019, 9, 1902813. [Google Scholar] [CrossRef]
- Wang, D.; Jiang, S.; Duan, C.; Mao, J.; Dong, Y.; Dong, K.; Wang, Z.; Luo, S.; Liu, Y.; Qi, X. Spinel-structured high entropy oxide (FeCoNiCrMn)3O4 as anode towards superior lithium storage performance. J. Alloy. Compd. 2020, 844, 156158. [Google Scholar] [CrossRef]
- Duan, C.; Tian, K.; Li, X.; Wang, D.; Sun, H.; Zheng, R.; Wang, Z.; Liu, Y. New spinel high-entropy oxides (FeCoNiCrMnXLi)3O4 (X = Cu, Mg, Zn) as the anode material for lithium-ion batteries. Ceram. Int. 2021, 47, 32025–32032. [Google Scholar] [CrossRef]
- Sun, Z.; Zhao, Y.; Sun, C.; Ni, Q.; Wang, C.; Jin, H. High entropy spinel-structure oxide for electrochemical application. Chem. Eng. J. 2022, 431, 133448. [Google Scholar] [CrossRef]
- Chen, H.; Qiu, N.; Wu, B.; Yang, Z.; Sun, S.; Wang, Y. A new spinel high-entropy oxide (Mg0.2Ti0.2Zn0.2Cu0.2Fe0.2)3O4 with fast reaction kinetics and excellent stability as an anode material for lithium ion batteries. RSC Adv. 2020, 10, 9736–9744. [Google Scholar] [CrossRef]
- Stygar, M.; Dąbrowa, J.; Moździerz, M.; Zajusz, M.; Skubida, W.; Mroczka, K.; Berent, K.; Swierczek, K.; Danielewski, M. Formation and properties of high entropy oxides in Co-Cr-Fe-Mg-Mn-Ni-O system: Novel (Cr, Fe, Mg, Mn, Ni)3O4 and (Co, Cr Fe,Mg, Mn)3O4 high entropy spinels. J. Eur. Ceram. Soc. 2020, 40, 1644–1650. [Google Scholar] [CrossRef]
- Liang, B.; Ai, Y.; Wang, Y.; Liu, C.; Ouyang, S.; Liu, M. Spinel-type (FeCoCrMnZn)3O4 high-entropy oxide: Facile preparation and supercapacitor performance. Materials 2020, 13, 5798. [Google Scholar] [CrossRef]
- Nguyen, T.X.; Patra, J.; Chang, J.K.; Ting, J.M. High entropy spinel oxide nanoparticles for superior lithiation–delithiation performance. J. Mater. Chem. A 2020, 8, 18963–18973. [Google Scholar] [CrossRef]
- Mao, A.; Xiang, H.Z.; Zhang, Z.G.; Kuramoto, K.; Zhang, H.; Jia, Y. A new class of spinel high-entropy oxides with controllable magnetic properties. J. Magn. Magn. Mater. 2020, 497, 165884. [Google Scholar] [CrossRef]
- Raul, P.K.; Senapati, S.; Sahoo, A.K.; Umlong, I.M.; Devi, R.R.; Thakur, A.J.; Veer, V. CuO nanorods: A potential and efficient adsorbent in water purification. RSC Adv. 2014, 4, 40580–40587. [Google Scholar] [CrossRef]
- Alhumaimess, M.S.; Essawy, A.A.; Kamel, M.M.; Alsohaimi, I.H.; Hassan, H.M.A. Biogenic-mediated synthesis of mesoporous Cu2O/CuO nano-architectures of superior catalytic reductive towards nitroaromatics. Nanomaterials 2020, 10, 781. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.L.; Mukherjee, S.; Nisbet, D.R.; Birbilis, N.; Chen, X.B. In vitro evaluation of biodegradable magnesium alloys containing micro-alloying additions of strontium, with and without zinc. J. Mater Chem. B 2015, 3, 8874–8883. [Google Scholar] [CrossRef]
- Hu, G.; Zhong, M.; Guo, C. Microstructures and high temperature tensile properties of As-Aged Mg-6Zn-1Mn-4Sn-(01, 0.5 and 1.0) Y alloys. Metals 2019, 9, 1. [Google Scholar] [CrossRef]
- Thota, S.; Prasad, B.; Kumar, J. Formation and magnetic behaviour of manganese oxide nanoparticles. J. Mater. Sci. Eng. B 2010, 167, 153–160. [Google Scholar] [CrossRef]
- Sharrouf, M.; Awad, R.; Roumié, M.; Marhaba, S. Structural, Optical and room temperature magnetic study of Mn2O3 nanoparticles. Mater. Sci. Appl. 2015, 6, 850–859. [Google Scholar]
- Qiao, H.; Wei, Z.; Yang, H.; Zhu, L.; Yan, X. Preparation and characterization of NiO nanoparticles by anodic arc plasma Method. J. Nanomater. 2009, 2009, 1–5. [Google Scholar] [CrossRef]
- Hong, S.J.; Mun, H.J.; Kim, B.J.; Kim, Y.S. Characterization of nickel oxide nanoparticles synthesized under low temperature. Micromachines 2021, 12, 1168. [Google Scholar] [CrossRef]
- Deori, K.; Deka, S. Morphology oriented surfactant dependent CoO and reaction time dependent Co3O4 nanocrystals from single synthesis method and their optical and magnetic properties. CrystEngComm 2013, 15, 8465. [Google Scholar] [CrossRef]
- Lutz, H.D.; Müller, B.; Steiner, H.J. Lattice vibration spectra. LIX. Single crystal infrared and Raman studies of spinel type oxides. J. Solid State Chem. 1991, 90, 54–60. [Google Scholar] [CrossRef]
- Kumar, P.R.; Jung, Y.H.; Bharathi, K.K.; Lim, C.H.; Kim, D.K. High capacity and low cost spinel Fe3O4 for the Na-ion battery negative electrode materials. Electrochim. Acta 2014, 146, 503–510. [Google Scholar] [CrossRef]
- Abdellatif, M.H.; Azab, A.A.; Salerno, M. Effect of rare earth doping on the vibrational spectra of spinel Mn-Cr ferrite. Mater. Res. Bull. 2018, 97, 260–264. [Google Scholar] [CrossRef]
- D’Ippolito, V.; Andreozzi, G.B.; Bersani, D.; Lottici, P.P. Raman fingerprint of chromate, aluminate and ferrite spinels. J. Raman Spectrosc. 2015, 46, 1255–1264. [Google Scholar] [CrossRef]
- Rivas-Murias, B.; Salgueirino, V. Thermodynamic CoO–Co3O4 crossover using Raman spectroscopy in magnetic octahedron-shaped nanocrystals. J. Raman Spectrosc. 2017, 48, 837–841. [Google Scholar] [CrossRef]
- Lazarević, Z.; Jovalekić, Č.; Milutinović, A.; Sekulić, D.; Ivanovski, V.N.; Rečnik, A.; Cekic, B.; Romčević, N.Ž. Nanodimensional spinel NiFe2O4 and ZnFe2O4 ferrites prepared by soft mechanochemical synthesis. J. Applied Physics 2013, 113, 187221. [Google Scholar] [CrossRef]
- Supriya, S.; Kumar, S.; Kar, M. Structural and electrical properties of CFO nanoparticle-filled PVA. J. Electron. Mater. 2019, 48, 3612–3623. [Google Scholar] [CrossRef]
- Laguna-Bercero, M.A.; Sanjuán, M.L.; Merino, R.I. Raman spectroscopic study of cation disorder in poly-and single crystals of the nickel aluminate spinel. J. Phys. Condens. Matter 2007, 19, 186217. [Google Scholar] [CrossRef]
- Xiao, B.; Wu, G.; Wang, T.; Wei, Z.; Sui, Y.; Shen, B.; Qi, J.; Wei, F.; Meng, Q.; Ren, Y.; et al. High entropy oxides (FeNiCrMnX)3O4 (X = Zn, Mg) as anode materials for lithium ion batteries. Ceram. Int. 2021, 47, 33972–33977. [Google Scholar] [CrossRef]
- Cao, Z.; Yang, Y.; Qin, J.; He, J.; Su, Z. Co3O4 polyhedron@MnO2 nanotube composite as anode for high-performance lithium-ion batteries. Small 2021, 17, 2008165. [Google Scholar] [CrossRef] [PubMed]
- Ghigna, P.; Airoldi, L.; Fracchia, M.; Callegari, D.; Anselmi-Tamburini, U.; D’angelo, P.; Pianta, N.; Ruffo, R.; Cibin, G.; Oliveira de Souza, D.; et al. Lithiation mechanism in high-entropy oxides as anode materials for Li-ion batteries: An operando XAS study. ACS Appl. Mater. Interfaces 2020, 12, 50344–50354. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Sarkar, A.; Wang, D.; Velasco, L.; Azmi, R.; Bhattacharya, S.S.; Bergfeldt, T.; Duvel, A.; Heitjans, P.; Brezesinski, T.; et al. Multi-anionic and -cationic compounds: New high entropy materials for advanced Li-ion batteries. Energy Environ. Sci. 2019, 12, 2433. [Google Scholar] [CrossRef]
- Lokcu, E.; Toparli, C.; Anik, M. Electrochemical performance of (MgCoNiZn)1−xLixO high-entropy oxides in lithium-ion batteries. ACS Appl. Mater. Interfaces 2020, 12, 23860–23866. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, H.; Kozu, T.; Shima, H.; Funakubo, H.; Uchida, H.; Katoda, T.; Nishida, K. Evaluation of oxygen vacancy in ZnO using Raman spectroscopy. In Proceedings of the 2015 Joint IEEE International Symposium on the Applications of Ferroelectric (ISAF), International Symposium on Integrated Functionalities (ISIF), and Piezoelectric Force Microscopy Workshop (PFM), Singapore, 24–27 May 2015. [Google Scholar] [CrossRef]
- Chapron, D.; Cordero, F.; Fontana, M.D. Characterization on of oxygen vacancies in SrTiO3 by means of inelastic and Raman speectroscopy. J. Appl. Phys. 2019, 126, 154101. [Google Scholar] [CrossRef]
- De Castro Silva, I.; Sigoli, F.A.; Mazali, I.O. Reversible oxygen vacancy generation on pure CeO2 nanorods evaluated by in situ Raman spectroscopy. J. Phys. Chem. C 2017, 121, 12928–12935. [Google Scholar] [CrossRef]
- Wang, B.; Yao, J.; Wang, J.; Chang, A. Spinel-type high-entropy (Co0.2Mn0.2Fe0.2Zn0.2Ti0.2)3O4 oxides constructed from disordered cations and oxygen vacancies. J. Alloy. Compd. 2022, 897, 163188. [Google Scholar] [CrossRef]





| Selected Element Combination | Single/Main Oxide Phase | Sample Codes | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mg | Ti | Cr | Mn | Fe | Co | Ni | Cu | Zn | ||||
| SG | ES | |||||||||||
| PVP | PAN | |||||||||||
| (Mg0.2Ti0.2Fe0.2Cu0.2Zn0.2)3O4 | 5MA-sg | -- | -- | |||||||||
| (Mn0.2Fe0.2Co0.2Ni0.2Zn0.2)3O4 | 5MD-sg | 5MD-a | 5MD-b | |||||||||
| (Cr0.2Mn0.2Fe0.2Co0.2Ni0.2)3O4 | 5ME-sg | 5ME-a | 5ME-b | |||||||||
| (Cr0.2Mn0.2Fe0.2Co0.2Zn0.2)3O4 | 5MF-sg | 5MF-a | 5MF-b | |||||||||
| (Cr0.2Mn0.2Fe0.2Ni0.2Zn0.2)3O4 | 5MG-sg | -- | 5MG-b | |||||||||
| (Ti0.2Mn0.2Fe0.2Cu0.2Zn0.2)3O4 | 5MH-sg | -- | -- | |||||||||
| (Mg0.2Cr0.2Mn0.2Fe0.2Ni0.2)3O4 | 5MI-sg | -- | -- | |||||||||
| (Mg0.2Cr0.2Mn0.2Fe0.2Co0.2)3O4 | 5MJ-sg | -- | -- | |||||||||
| (Ti0.2Mn0.2Fe0.2Co0.2Ni0.2)3O4 | 5MK-sg | 5MK-a | -- | |||||||||
| (Mn0.2Fe0.2Ni0.2Cu0.2Zn0.2)3O4 | 5ML-sg | 5ML-a | 5ML-b | |||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrovičovà, B.; Xu, W.; Musolino, M.G.; Pantò, F.; Patanè, S.; Pinna, N.; Santangelo, S.; Triolo, C. High-Entropy Spinel Oxides Produced via Sol-Gel and Electrospinning and Their Evaluation as Anodes in Li-Ion Batteries. Appl. Sci. 2022, 12, 5965. https://doi.org/10.3390/app12125965
Petrovičovà B, Xu W, Musolino MG, Pantò F, Patanè S, Pinna N, Santangelo S, Triolo C. High-Entropy Spinel Oxides Produced via Sol-Gel and Electrospinning and Their Evaluation as Anodes in Li-Ion Batteries. Applied Sciences. 2022; 12(12):5965. https://doi.org/10.3390/app12125965
Chicago/Turabian StylePetrovičovà, Beatrix, Wenlei Xu, Maria Grazia Musolino, Fabiola Pantò, Salvatore Patanè, Nicola Pinna, Saveria Santangelo, and Claudia Triolo. 2022. "High-Entropy Spinel Oxides Produced via Sol-Gel and Electrospinning and Their Evaluation as Anodes in Li-Ion Batteries" Applied Sciences 12, no. 12: 5965. https://doi.org/10.3390/app12125965
APA StylePetrovičovà, B., Xu, W., Musolino, M. G., Pantò, F., Patanè, S., Pinna, N., Santangelo, S., & Triolo, C. (2022). High-Entropy Spinel Oxides Produced via Sol-Gel and Electrospinning and Their Evaluation as Anodes in Li-Ion Batteries. Applied Sciences, 12(12), 5965. https://doi.org/10.3390/app12125965











