1. Introduction
Considered the most common form of sleep obstruction, obstructive sleep apnea (OSA) is a life-threatening health anomaly that many people suffer from today. It is characterized by repeated pharyngeal airway collapse during sleep [
1], which means that subjects’ breathing frequently pauses while asleep, sometimes for minutes. Bruxism is another symptom that negatively affects sleep quality by significantly harming teeth and damaging temporomandibular joints (TMJ) [
2].
Lack of sleep hinders focus and concentration during the day, causes chest pain upon waking up [
3], causes morning headaches [
4], and is a factor in many psychological symptoms such as depression, anxiety, or excessive irritability [
5], and even death [
6].
Continuous positive airway pressure (CPAP) treatment is the most commonly used method to treat OSA. It has proven to be the most effective way to prevent sleep obstruction because research has shown that subjects with OSA have significantly greater activity reductions in airway muscles than normal people [
7,
8]. A facemask is connected to a machine via a tube and positive airway pressure is mechanically pushed directly into the mouth to keep the airways open (
Figure 1).
However, the problem with this universally accepted method is that it is simply too obstructive. In fact, an in-depth study concerning the effect of CPAP on sleep apnea syndrome revealed that patients showed discontent regarding CPAP because the device that was designed to enhance sleep quality paradoxically hindered sleep because it was too “bothersome” [
9]. This naturally led to insufficient compliance rates; Yetkin et al. [
10] proved that after months of compliance test results, only very severely affected patients adhered to CPAP devices until the end of the study period.
As a result, many alternative devices have been developed, such as the mandibular advancement device (MAD), which increases the airway diameter with soft tissue displacement achieved by mandibular protrusion [
11]. It is a much smaller form of the oral device, compared to CPAP, which resembles the form of a mouthguard commonly used in sports (
Figure 2). This oral device is also known to prevent bruxism in the absence of a CPAP device. A critical problem still persists with MAD in that there is still ongoing debate about the device’s actual effectiveness in treating OSA. Many uncertainties regarding MAD have prevented comparably comfortable devices from becoming more widespread clinical sleep treatment services.
Therefore, this research proposes s-Guard: a multisensor embedded OSA monitoring intraoral appliance device specifically targeted for MAD prototypes. This research presents the unique design and development of the MAD-based model that enables sensors to be embedded and presents practical implementation results among institutional review board (IRB) approved subjects. Since this study targets OSA patients, relevant health information monitoring sensors such as temperature, gyroscope, accelerometer, and SpO2 sensors were embedded. This research is expected to greatly motivate other sensors to be embedded in our proposed model for the application of other disease areas.
2. Related Literature
In the healthcare industry and business sectors, many innovations for global well-being are being developed as biosensor embedded solutions for enhancing health and wellness. The most commonly applied areas are wristband types, such as smartwatches [
13] and ring types [
14]. There are many other variations to monitor the health status of users/patients, but intraoral appliance-embedded healthcare solutions have rarely been addressed in previous research.
For example, Matsuda et al. demonstrated a sensor-embedded oral appliance to monitor the airway [
1]. They fabricated maxillary and mandibular oral appliances from 2 mm thick resin plates using pressure welding. The activity of the left genioglossus (GG) was recorded using two silver ball electrodes attached to the lingual edge of the mandibular oral appliance. Respiratory status and right masseter muscle activity were measured using an airflow sensor and surface electrodes, respectively. Their attempts to apply sensors to oral appliances were impressive, although this solution was only designed for experimental purposes and not for practical purposes.
Jin et al. [
15] developed a flexible surface acoustic wave respiration sensor for monitoring obstructive sleep apnea syndrome. They demonstrated the potential for flexible microsensors for monitoring OSA. Rocher et al. [
16] also developed a smart system for monitoring apnea episodes in domestic environments with a sound sensor. Their proposed system consisted of a sound sensor, a vibrating element, and a microcontroller to process the collected data to detect OSA during sleep.
Some wearable-sensor-related researches targeted animal testing also. Zhang et al. [
17] evaluated live mutton sheep during transportation based on a wearable multisensor system. More up-to-date literature review about the development of sensors are viewable from a review by Nasiri and Khosravani [
18]. They systematically focused on applications of wearable sensors divided into (a) biophysical tracking, (b) biochemical monitoring, and (c) detection of real-time data.
Sekita et al. [
19] developed a measuring system for 3D movement of the denture during function. The system for measuring denture movement consisted of an inertial measurement unit (IMU), a control unit, and a Bluetooth module. The IMU contained a three-axis gyroscope, a three-axis accelerometer, and a three-axis digital compass. The IMU was embedded in a self-curing acrylic resin. An important point of their research was that they successfully embedded several sensors despite the limited space allowed in dentures. They proposed a prototype that may serve as a future diagnostic appliance, such as occlusal examination in a denture, but this was also not developed to a practical level.
On the other hand, the current research proposes a multisensor embedded OSA monitoring intraoral appliance device, specially targeted for MAD prototypes. Compared to the recent research outlined above, our research was fully designed and tested for practical use. In addition, our proposed solution holds the highest number of embedded sensors known in all oral appliance-related studies thus far, which makes this research more unique considering the fact that MAD devices allow even less space tolerance, compared to normal dentures.
3. Overall System
3.1. Overall Service System Application
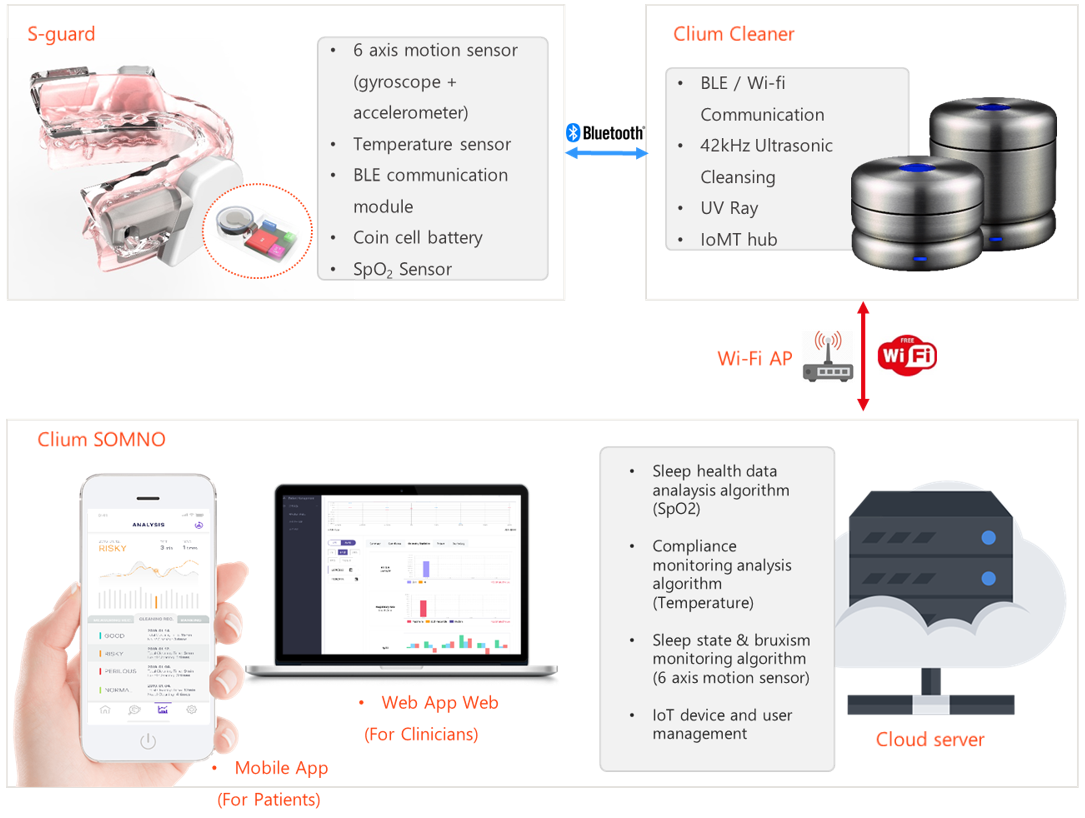
The overall system description of the proposed solution, s-Guard, is depicted in
Figure 3. First, patients who use s-Guard need to be diagnosed with OSA after polysomnography (PSG) testing. Although s-Guard itself provides sleep quality monitoring functions, it is “treatment-centered” rather than “monitoring-” or “diagnosis-centered”.
In our proposed sensible intraoral appliance s-Guard, six-axis motion sensors (composed of a three-axis gyroscope and a two-axis accelerometer) [
20] are embedded to monitor the sleep state and bruxism state. In addition, temperature sensors and SpO
2 sensors were embedded to monitor compliance status (measurement of properly complying to device usage) and OSA status, respectively. Finally, Bluetooth low energy (BLE) [
21,
22] communication module, microcontroller unit (MCU), and coin cell battery were used for the overall chip operation.
Healthcare data sensed from s-Guard are then transmitted to the MAD cleaner (named Clium) via Bluetooth communication. This is because the Bluetooth protocol is a form of near-field communication (NFC) [
23], which basically needs a form of gateway to fully communicate with a server for full-service launch.
Raw data are then transmitted to the server from the cleaner communication hub through a Wi-Fi access point (AP). Other forms of fourth- (4G) or fifth-generation (5G) wireless communication can be applicable.
In the server, a sleep health data analysis algorithm is loaded to analyze the SpO
2 data. A compliance monitoring analysis algorithm was conducted based on temperature sensing data. The sleep state monitoring algorithm analyzes data obtained from six-axis motion sensors (accelerometer and gyroscope). The bruxism status can be additionally analyzed using this function. Servers also manage connected IoT devices and users. The specific design and development features of the sensible intraoral appliance, s-Guard, are explained in
Section 3.2.
Statistical data analyzed from the server can be viewed by the patients through the mobile application. Only basic statistical data that could be easily perceived by no-professional patients are provided. More professional diagnosis-, treatment-, and monitoring-related data are provided to clinical professionals by the web application that is fully accessible through PC.
3.2. Specific Features of s-Guard
3.2.1. Design
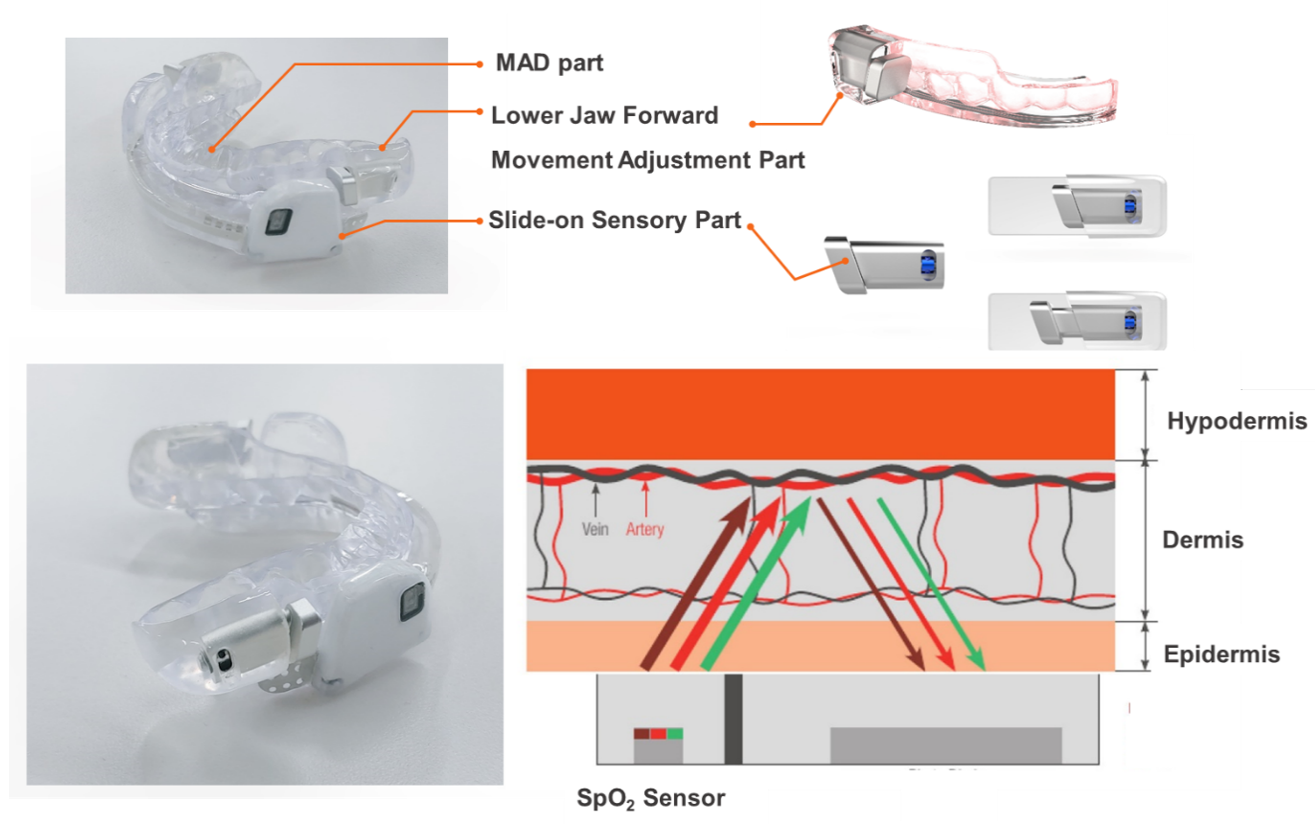
A 3D design model prototype of the proposed sensible intraoral appliance s-Guard is shown in
Figure 4. We used the capacity allowed in the inner cheek part of the MAD to embed sensory parts. The shape of the MAD is subordinate to the designated user.
Figure 4a depicts the slide-on type of the sensory part, and
Figure 4b depicts the screw-in type. A limitation of this model is that this design only supports patients/users older than 19 years old (adults only). This was due to the lack of capacity allowed when applied to younger ages with smaller inner oral sizes. In this paper, the slide-on type of s-Guard is shown.
Figure 5 shows the actual slide-on prototype model of the s-Guard. The MAD part was adjusted to fit the individual patient/user through a dental prescription. The lower jaw forward movement adjustment part provides the patient/user breathing space. All sensors and modules were embedded in the slide-on sensory parts. This part can be attached and detached to replace the power supply battery. SpO
2 is sensed through our solution by reflecting the data obtained from the vein and artery in the dermis part of the gums.
3.2.2. MCU, Communication Module, and Power Supply
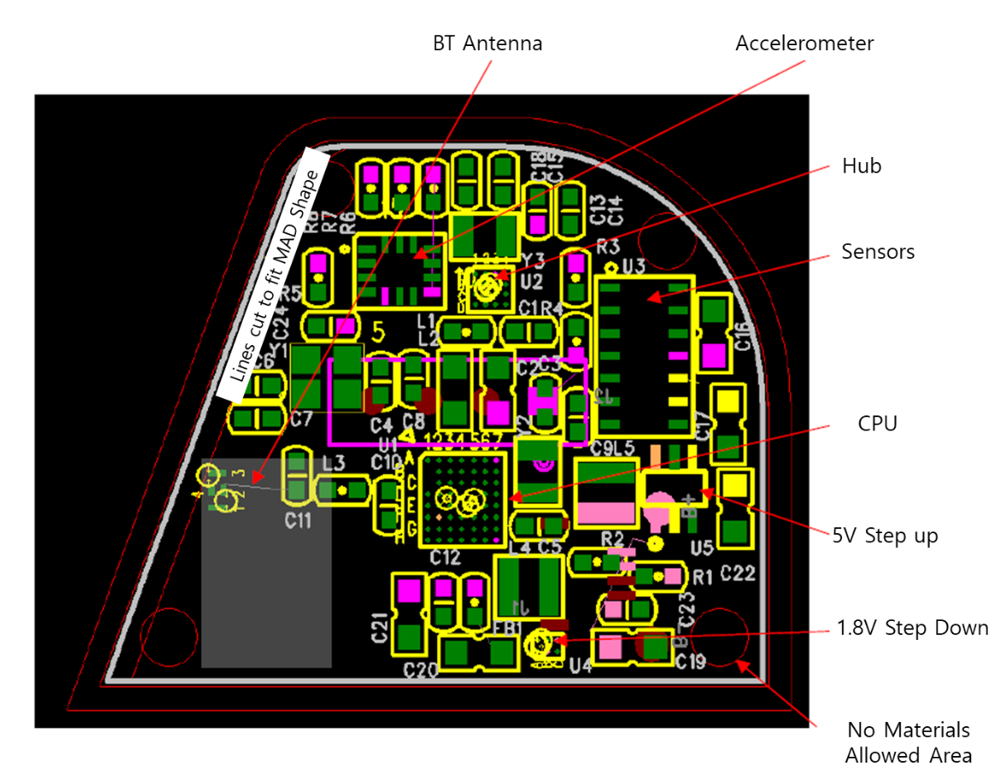
The printed circuit board (PCB) and circuit schematic of s-Guard are shown in
Figure 6. MCU, communication module, power, and other specifications are listed in
Table 1. In summary, important features such as 64 Megahertz (MHz) 32-bit Advanced RISC Machine (ARM) Cortex-M4 was used for MCU, Bluetooth 5/Antenna (ANT)/2.4 Gigahertz (GHz) proprietary was used for communication module, and voltage (V) supply of 1.7 to 3.6 V low dropout (LDO) or direct current/direct current (DC/DC) was used for power.
3.2.3. Sensors
Core embedded sensors related to temperature, six-axis, and SpO
2 are presented in
Table 2. On the board, SpO
2 includes algorithms, quick evaluation and data collection, stream data to mobile apps, schematics, or Gerbers to download, with components of MAX30101 (Optical Module) and MAX32664A (Algorithm MCU). The sampling rate per second (SPS) for the temperature sensor, six-axis sensor, and SpO
2 sensors were 2 SPS, 10 SPS, and 10 SPS, respectively.
4. Results
4.1. Communication Speed Evaluation
The most important features of s-Guard include stable sensors, wireless communication, and seamless monitoring. Necessarily, this can be assessed mainly by two categories: communication speed and communication errors. This section presents the Bluetooth communication speed function evaluation results. The results will be followed by communication error rate performances that represent the stability of the fast communication in the next
Section 4.2. The communication experiment was conducted using the experimental model shown in
Figure 7. s-Guard was connected to our custom-developed MAD cleaner communication gateway (Clium) via Bluetooth. This gateway acts as a communication hub for server/personal computer (PC) connections and can also be used as a sanitized intraoral device storage.
The MAD cleaner communication gateway was then connected to the test PC via a serial connection. Bluetooth communication speed performance was shown in a basic disk operating system (DOS) interface, which was shown in the console command in bits per second (bps). The specifications of the mad cleaner communication gateway and test PC are listed in
Table 3.
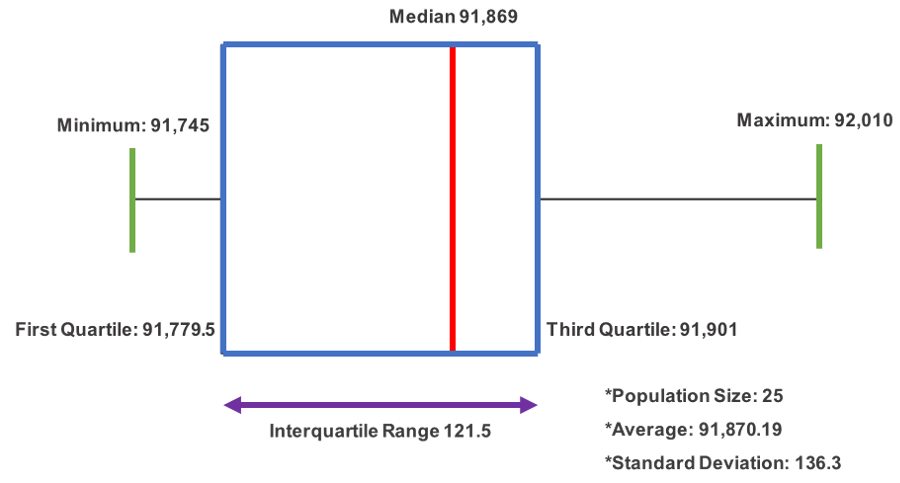
The transmission speed results obtained from volunteer test patients are shown in
Figure 8. The test environment was conducted according to the specifications mentioned in
Section 3.2. All test procedures were approved by the Institutional Review Board (IRB) of Yonsei Severance Hospital (IRB approval number 2-2019-0060). Starting from a set time (time stamp = 0), the transmission speed was measured 26 times (n = 26) with a time interval of 2 s. All results were rounded up to the second decimal place. The results showed an average transmission speed of 91,870.19 bytes per second (standard deviation 136.3), with a population size of 25. The minimum value was 91,745, and the maximum value was 92,010, with a median of 91,869. The first quartile was 91,779.5, and the third quartile was 91,901, with the interquartile range of 121.5.
4.2. Wireless Connection Success Ratio Evaluation
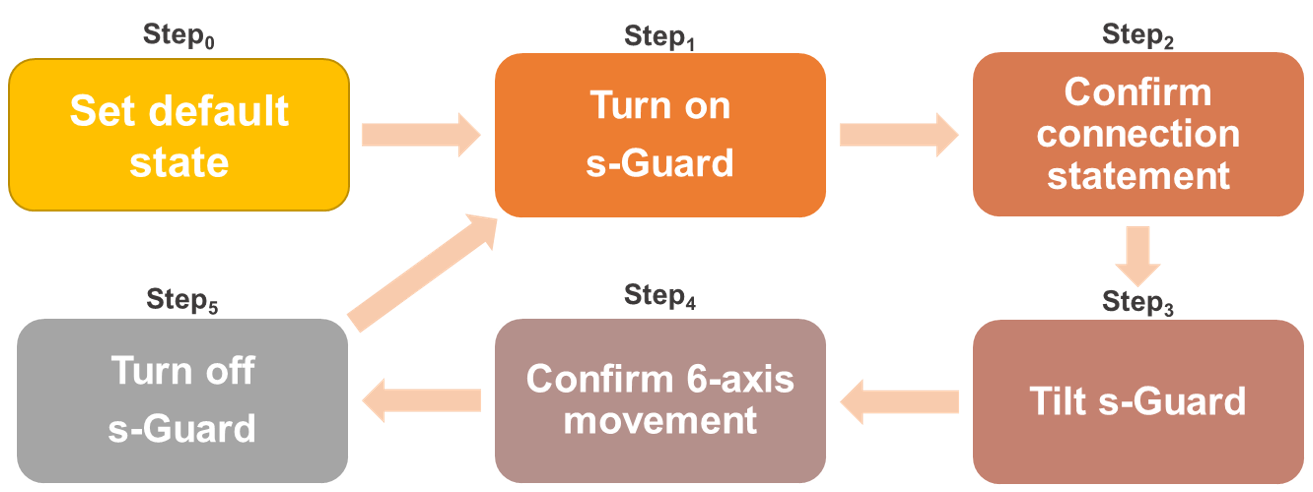
A wireless Bluetooth connection success ratio evaluation was conducted to measure the seamless transmission of the proposed solution in a real-time usage scenario. The test scheme shown in
Figure 9 was conducted under the conditions specified in
Section 4.1. The test cycle was repeated 10 times (n = 10).
First, s-Guard was set to a default state (Step
0). It was switched from off to on (Step
1), which was the start of the first cycle. When pairing is complete, confirm the fixed connection statement “Now connected to the BLE Server” from the screen shown on the test computer (Step
2). Then, the tilt s-Guard is used to check the sensing connection (Step
3) and then check the screen that shows the six-axis values from the test computer again (Step
4). Here, six-axis values refer to combinations of values (2 × 3) calculated from the accelerometer (two-dimensional data) multiplied with gyroscope (x, y, z three-dimensional data). Finally, the s-Guard (Step
5) completes one cycle, and the next cycle begins. When the final Step
5 is completed, complete C
n and start another cycle. The final results indicated a success rate of 100%, as shown in
Table 4.
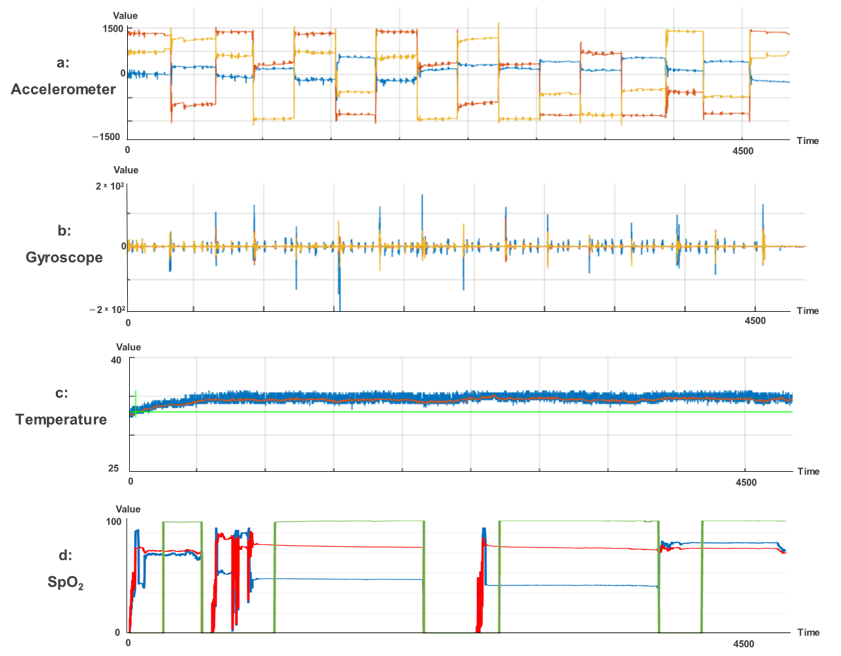
Accordingly, actual physiological signals of six-axis sensors (accelerometer and gyroscope) are shown in
Figure 10.
Figure 10a,b shows the accelerometer raw data and gyroscope data acquired by time. Each of the three colors of red, yellow, and blue in
Figure 10a indicate x
1, y
2, and z
2 three-dimensional axes, respectively. On the other hand, each of the two colors of yellow and blue indicate x
2 and y
2 two-dimensional axes, respectively.
Figure 10c shows the temperature data acquired by s-Guard. The blue line indicates raw data, the red line indicates linear average, and the green line indicates temperature critical level threshold. Lastly,
Figure 10d shows raw data acquired from the SpO
2 sensor from s-Guard. The green, red, and blue lines refer to SpO
2 raw data, heart rate, and infrared raw data, respectively.
4.3. Wireless Data Stream Error Ratio Evaluation
This section presents a wireless data stream error ratio evaluation. The target sample patient’s aggregate sensor data signal was monitored up to a maximum length of 8 h. Data transmission status was measured at several set time intervals—first at 10 s, then at 100 s, 10 min, 1 h, 4 h, and finally, at 8 h. Eight hours was set because it is the most commonly known average suggested sleep time for adults.
According to theoretical calculations, the number of data transmission samples per second should be 250. This theoretical calculation was compared with the actual transmission sample calculation to assess the actual error rate. The measurement results are presented in
Table 5. The results showed the highest error rate of 18.24% at 10 s elapsed time. However, the error rate greatly decreased as time passed. Eventually, the error rate decreased to a minimum of 0.1% when 8 h had fully elapsed.
5. Discussion and Conclusions
This research proposed a multisensor embedded sleep quality health monitoring intraoral appliance device with innovative design and development features. Sleep quality could be improved by preventing bruxism and OSA via real-time monitoring and biofeedback.
s-Guard is fundamentally a wearable sensor. Therefore, assessing sensor stability in terms of wireless communication and seamless monitoring is most important. Necessarily, this can be assessed mainly by two categories: communication speed and communication errors.
The results revealed a wireless data transmission monitoring speed of 91,870.19 bytes per second, a connection success rate of 100%, and a minimum transmission error rate of 0.1%. Considering that previous research mentioned the problems regarding limited bandwidth in wireless health monitoring situations [
30,
31], this performance can be considered highly suitable.
Actual monitoring results showed successful sleep posture monitoring data calculated by the s-Guard algorithm based on sensed six-axis data. s-Guard was capable of monitoring a total of five sleep statuses: unidentified posture (null), prone position, supine position, leftward position, and rightward position. In addition, bruxism anomaly was also successfully detected by s-Guard, specifically monitored according to severity.
The solution proposed herein could contribute significantly to enhancing the sleep quality of modern lives, in which many people suffer from early age OSA and bruxism. OSA patients who have to use CPAP for intensive care may now use a much more compact and convenient method for treatment and management. In addition, this research opened the possibility of embedding sensors other than temperature, six-axis, and SpO2. This is envisioned to be applied in various other fields of information communication technology (ICT) and IoT. In addition, s-Guard can be used in both medicine and everyday healthcare management.
A limitation of this research was that this solution was not suitable for short-term monitoring situations due to the high error rate in short-term sensing. Future research should investigate the reason for this and solve the issue of error rate so that this solution can also be used in short-term diagnosis situations.
Author Contributions
Conceptualization, S.-J.L. and I.-D.J.; methodology, E.-B.K.; software, I.-H.J.; validation, J.-Y.P. and J.-H.H.; formal analysis, S.-J.L.; investigation, E.-B.K. and J.-Y.P.; resources, J.-H.H., and T.-Y.J.; data curation, T.-Y.J.; writing—original draft preparation, S.-J.L., J.-H.H., and T.-Y.J.; writing—review and editing, I.-D.J. and T.-Y.J.; visualization, J.-Y.P.; supervision, T.-Y.J.; project administration, S.-J.L. and I.-D.J.; funding acquisition, S.-J.L., I.-D.J., and T.-Y.J. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded the Ministry of Health & Welfare, Republic of Korea (grant number: HI19C1243), and the National Research Foundation of Korea (grant number: NRF-2020R1l1A1A01053726).
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Review Board (or Ethics Committee) of Yonsei Severance Hospital (IRB approval number 2-2019-0060).
Informed Consent Statement
Patient consent was waived due to nonidentifiable personal information included in this study.
Data Availability Statement
Not applicable.
Acknowledgments
All research results were certified by the Korea Testing Laboratory (KTL). The authors would like to thank the Electronics and Telecommunications Research Institute (ETRI) and Curaum Inc. for their experimental assistance.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Matsuda, M.; Ogawa, T.; Sitalaksmi, R.M.; Miyashita, M.; Ito, T.; Sasaki, K. Effect of mandibular position achieved using an oral appliance on genioglossus activity in healthy adults during sleep. Head Face Med. 2019, 15, 26. [Google Scholar] [CrossRef] [PubMed]
- Braga, S.P.; Fiamengui, L.M.S.P.; da Silveira, V.R.S.; Chaves, H.V.; Furquim, B.D.; Cunha, C.O.; Repeke, C.E.P.; Conti, P.C.R. Insights for temporomandibular disorders management: From psychosocial factors to genetics—A case report. Spec. Care Dent. 2021, 41, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Madam, N.; Mosleh, W.; Punnanithinont, N.; Carmona-Rubio, A.; Said, Z.H.; Sharma, U.C. Pulmonary Arterial Enlargement is Associated with Acute Chest Pain in Patients without Obstructive Coronary Artery Disease. Clin. Med. Insights Circ. Respir. Pulm. Med. 2018, 12. [Google Scholar] [CrossRef] [PubMed]
- Demonte, G.; Santangelo, D.; Roccia, F.; Gambardella, A.; Bono, F. Obstructive sleep apnea (OSA) in headache sufferers with Idiopathic intracranial Hypertension (IIH): A prospective study. Cephalalgia 2019, 39, 296–297. [Google Scholar]
- Park, K.-M.; Kim, S.-Y.; Sung, D.; Kim, H.; Kim, B.-N.; Park, S.; Jung, K.-I.; Park, M.-H. The relationship between risk of obstructive sleep apnea and other sleep problems, depression, and anxiety in adolescents from a community sample. Psychiatry Res. 2019, 280. [Google Scholar] [CrossRef] [PubMed]
- Philip, P.; Taillard, J.; Micoulaud-Franchi, J.-A. Sleep Restriction, Sleep Hygiene, and Driving Safety The Importance of Situational Sleepiness. Sleep Med. Clin. 2019, 14, 407–412. [Google Scholar] [CrossRef]
- Johal, A.; Gill, G.; Ferman, A.; McLaughlin, K. The effect of mandibular advancement appliances on awake upper airway and masticatory muscle activity in patients with obstructive sleep apnoea. Clin. Physiol. Funct. Imaging 2007, 27, 47–53. [Google Scholar] [CrossRef]
- Mezzanotte, W.S.; Tangel, D.J.; White, D.P. Influence of sleep onset on upper-airway muscle activity in apnea patients versus normal controls. Am. J. Respir. Crit. Care Med. 1996, 153, 1880–1887. [Google Scholar] [CrossRef]
- Willman, M.; Igelström, H.; Martin, C.; Åsenlöf, P. Experiences with CPAP treatment in patients with obstructive sleep apnea syndrome and obesity. Adv. Physiother. 2012, 14, 166–174. [Google Scholar] [CrossRef]
- Yetkin, O.; Kunter, E.; Gunen, H. CPAP compliance in patients with obstructive sleep apnea syndrome. Sleep Breath. 2008, 12, 365–367. [Google Scholar] [CrossRef]
- Basyuni, S.; Barabas, M.; Quinnell, T. An update on mandibular advancement devices for the treatment of obstructive sleep apnoea hypopnoea syndrome. J. Thorac. Dis. 2018, 10, S48–S56. [Google Scholar] [CrossRef]
- Chaves, C.M., Jr.; Dal-Fabbro, C.; Machado, M.; Bruin, V.; Bruin, P.; Gurgel, M.; Torgeiro, S.; Cevidanes, L.; Haddad, F.; Bittencourt, L. Use Of Mandibular Advancement Devices For Obstructive Sleep Apnoea Treatment in Adults. Int. Arch. Med. 2017, 10. [Google Scholar] [CrossRef]
- Raja, J.M.; Elsakr, C.; Roman, S.; Cave, B.; Pour-Ghaz, I.; Nanda, A.; Maturana, M.; Khouzam, R.N. Apple Watch, Wearables, and Heart Rhythm: Where do we stand? Ann. Transl. Med. 2019, 7. [Google Scholar] [CrossRef]
- Zhu, K.; Perrault, S.; Chen, T.; Cai, S.; Peiris, R.L. A sense of ice and fire: Exploring thermal feedback with multiple thermoelectric-cooling elements on a smart ring. Int. J. Hum. Comput. Stud. 2019, 130, 234–247. [Google Scholar] [CrossRef]
- Jin, H.; Tao, X.; Dong, S.; Qin, Y.; Yu, L.; Luo, J.; Deen, M.J. Flexible surface acoustic wave respiration sensor for monitoring obstructive sleep apnea syndrome. J. Micromech. Microeng. 2017, 27, 115006. [Google Scholar] [CrossRef]
- Rocher, J.; Parra, L.; Sendra, S.; Lloret, J. Smart System for Monitoring Apnea Episodes in Domestic Environments with Sound Sensor. In International Conference on Advanced Intelligent Systems for Sustainable Development; Springer: Berlin/Heidelberg, Germany, 2018; pp. 205–215. [Google Scholar]
- Zhang, M.; Feng, H.; Luo, H.; Li, Z.; Zhang, X. Comfort and health evaluation of live mutton sheep during the transportation based on wearable multi-sensor system. Comput. Electron. Agric. 2020, 176, 105632. [Google Scholar] [CrossRef]
- Nasiri, S.; Khosravani, M.R. Progress and challenges in fabrication of wearable sensors for health monitoring. Sens. Actuators A Phys. 2020, 312, 112105. [Google Scholar] [CrossRef]
- Sekita, T.; Takeuchi, S.; Minakuchi, S. Measuring system for an attitude angle of a denture using an Inertial Measurement Unit. J. Prosthodont. Res. 2018, 62, 195–199. [Google Scholar] [CrossRef]
- Jarchi, D.; Casson, A.J. Description of a Database Containing Wrist PPG Signals Recorded during Physical Exercise with Both Accelerometer and Gyroscope Measures of Motion. Data 2017, 2, 1. [Google Scholar] [CrossRef]
- Shan, G.; Roh, B. Performance Model for Advanced Neighbor Discovery Process in Bluetooth Low Energy 5.0-Enabled Internet of Things Networks. IEEE Trans. Ind. Electron. 2020, 67, 10965–10974. [Google Scholar] [CrossRef]
- Park, E.; Lee, M.-S.; Kim, H.-S.; Bahk, S. AdaptaBLE: Adaptive control of data rate, transmission power, and connection interval in bluetooth low energy. Comput. Netw. 2020, 181. [Google Scholar] [CrossRef]
- Figueroa Lorenzo, S.; Anorga Benito, J.; Garcia Cardarelli, P.; Alberdi Garaia, J.; Arrizabalaga Juaristi, S. A Comprehensive Review of RFID and Bluetooth Security: Practical Analysis. Technologies 2019, 7, 15. [Google Scholar] [CrossRef]
- Chia, J.; Yang, C. Thermal Management of QFN 48 Package Attached to Different Multi-Layers of Printed Circuit Board Designs. In Proceedings of the International Electronic Packaging Technical Conference and Exhibition, Maui, HI, USA, 6–11 July 2003; Volume 36908, pp. 57–61. [Google Scholar]
- Atmel. AT90USB82 Datasheet. On-chip Analog, On-Chip Debug Interface, and Power-On Reset. ISP Flash and USB Controller AT90USB82, AT90USB162. Available online: https://www.digchip.com/datasheets/parts/datasheet/054/AT90USB162.php (accessed on 4 May 2021).
- Maxim Integrated. MAX30101 Datasheet: High-Sensitivity Pulse Oximeter and Heart-Rate Sensor for Wearable Health. 2020. Available online: https://www.maximintegrated.com/en/products/interface/sensor-interface/MAX30101.html (accessed on 4 May 2021).
- Anceschi, E.; Bonifazi, G.; De Donato, M.C.; Corradini, E.; Ursino, D.; Virgili, L. SaveMeNow. AI: A Machine Learning Based Wearable Device for Fall Detection in a Workplace. In Enabling AI Applications in Data Science; Springer: Berlin/Heidelberg, Germany, 2020; pp. 493–514. [Google Scholar]
- Nordic Semiconductor. nRF52840 Objective Product Specification v0.5.1. 2017. Available online: https://infocenter.nordicsemi.com/pdf/nRF52840_OPS_v0.5.1.pdf (accessed on 6 July 2017).
- Miele, P.; uwaisem, M.A.; Kim, D.-K. Comparative Assessment of Static Analysis Tools for Software Vulnerability. JCP 2018, 13, 1136–1144. [Google Scholar] [CrossRef]
- Lee, S.-J.; Cho, G.-Y.; Lee, T.-R. N-WRETS: Near-Lossless Wireless Real-time Efficient Electroencephalogram Transmission Solution to Support Sleep Disorder Monitoring Platforms. Telemed. e-Health 2019, 25, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Cho, G.-Y.; Lee, G.-Y.; Lee, T.-R. Efficient Real-Time Lossless EMG Data Transmission to Monitor Pre-Term Delivery in a Medical Information System. Appl. Sci. 2017, 7, 366. [Google Scholar] [CrossRef]
| Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).