Human Dental Pulp Stem Cell Osteogenic Differentiation Seeded on Equine Bone Block with Graphene and Melatonin
Abstract
1. Introduction
2. Materials and Methods
2.1. Ammonia Graphene-Oxide (G-N) Equine Bone Block Coating
2.2. hDPSCs Extraction and Cell Cultures
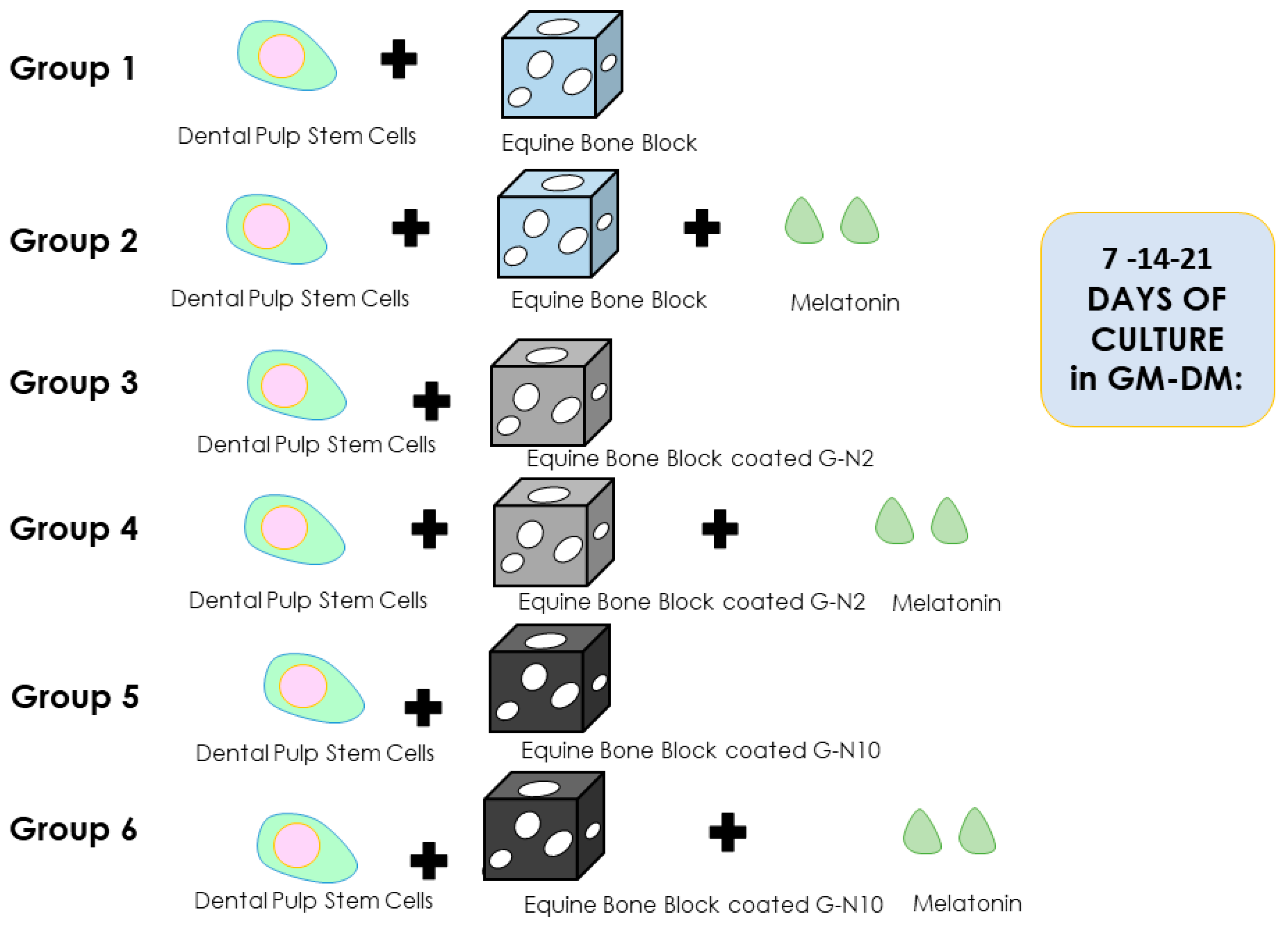
2.3. Experimental Study Design
- Group 1: hDPSCs + equine bone block (Ctrl);
- Group 2: hDPSCs + equine bone block + melatonin (Ctrl + Mel);
- Group 3: hDPSCs + equine bone block G-N-coated 2 μg/mL (G-N2);
- Group 4: hDPSCs + equine bone block G-N-coated 2 μg/mL + melatonin (G-N2 + Mel);
- Group 5: hDPSCs + equine bone block G-N-coated 10 μg/mL (G-N10);
- Group 6: hDPSCs + equine bone block G-N-coated 10 μg/mL + melatonin (G-N10 + Mel).
2.4. Live/Dead Test
2.5. RNA Isolation and Quantification
2.6. Quantitative Real-Time PCR for MicroRNAs Expression
- -
- has-miR-133a (UUUGGUCCCCUUCAACCAGCUG; #002246);
- -
- has-miR-133b (UUUGGUCCCCUUCAACCAGCUA; #002247);
- -
- hsa-miR-135a-5p (UAUGGCUUUUUAUUCCUAUGUGA; #000460);
- -
- hsa-miR-206 (UGGAAUGUAAGGAAGUGUGUGG; #000510);
- -
- hsa-miR- 29b (CUGGUUUCACAUGGUGGCUUAG; # 002166);
- -
- hsa-miR-let-7b (UGAGGUAGUAGGUUGUGUGGUU; #002619);
- -
- hsa-miR-16-5p (UAGCAGCACGUAAAUAUUGGCG; # 000391).
2.7. Gene Expression Analysis
2.8. Osteocalcin Levels Measurement
2.9. Histological Evaluations
2.10. Statistical Analysis
3. Results
3.1. Chemico-Physical Characterization of Ammonia Graphene-Oxide-Coated Collagenated Bone Block
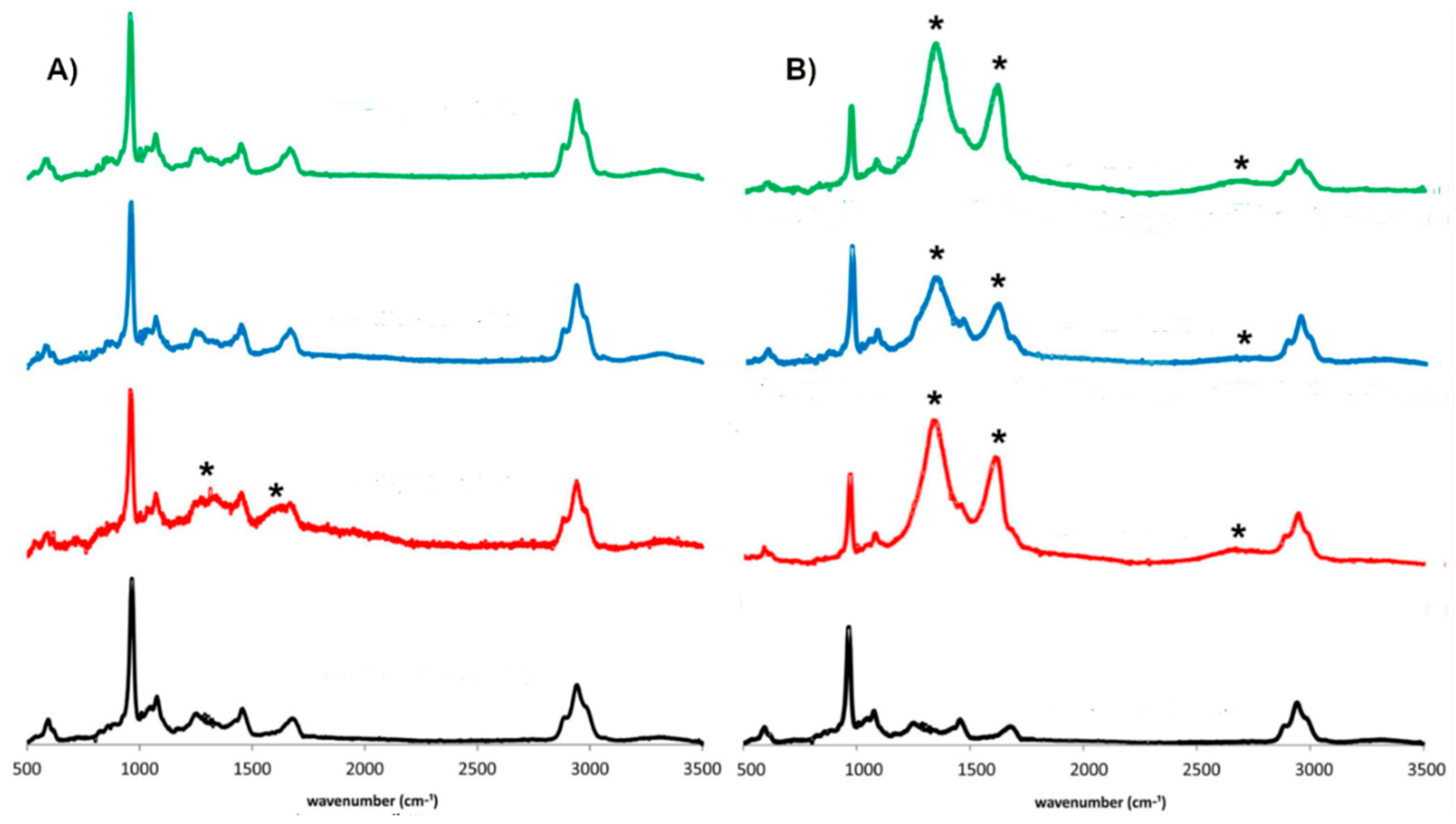
3.2. UV-VIS Spectroscopy
3.3. Raman Spectroscopy
3.4. TGA Analysis
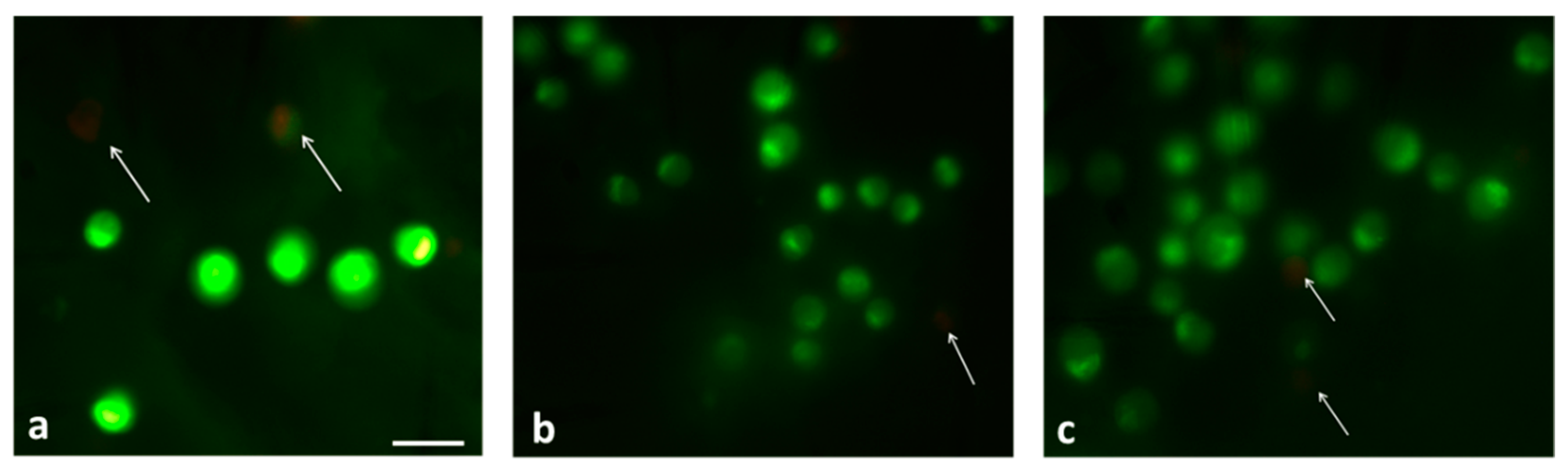
3.5. Cell Viability Assessment on Blocks
3.6. Gene Expression
3.7. MicroRNAs Expression
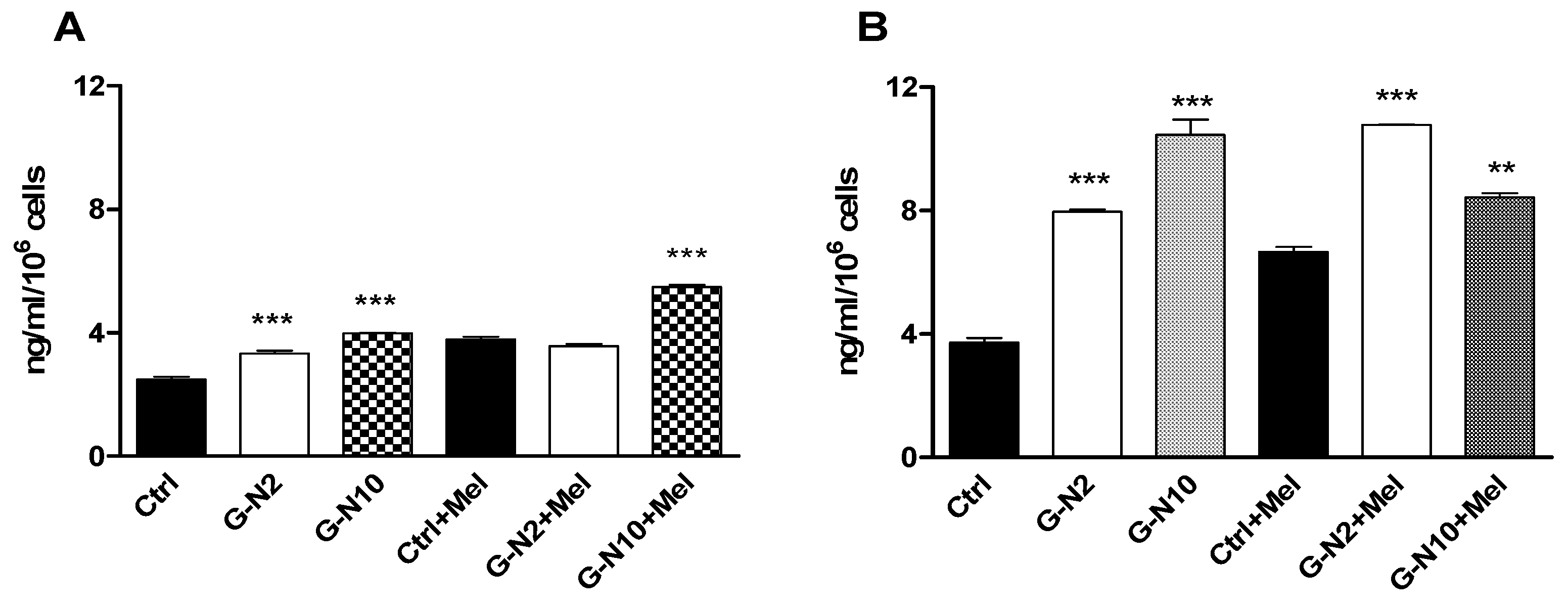
3.8. Osteocalcin Assay
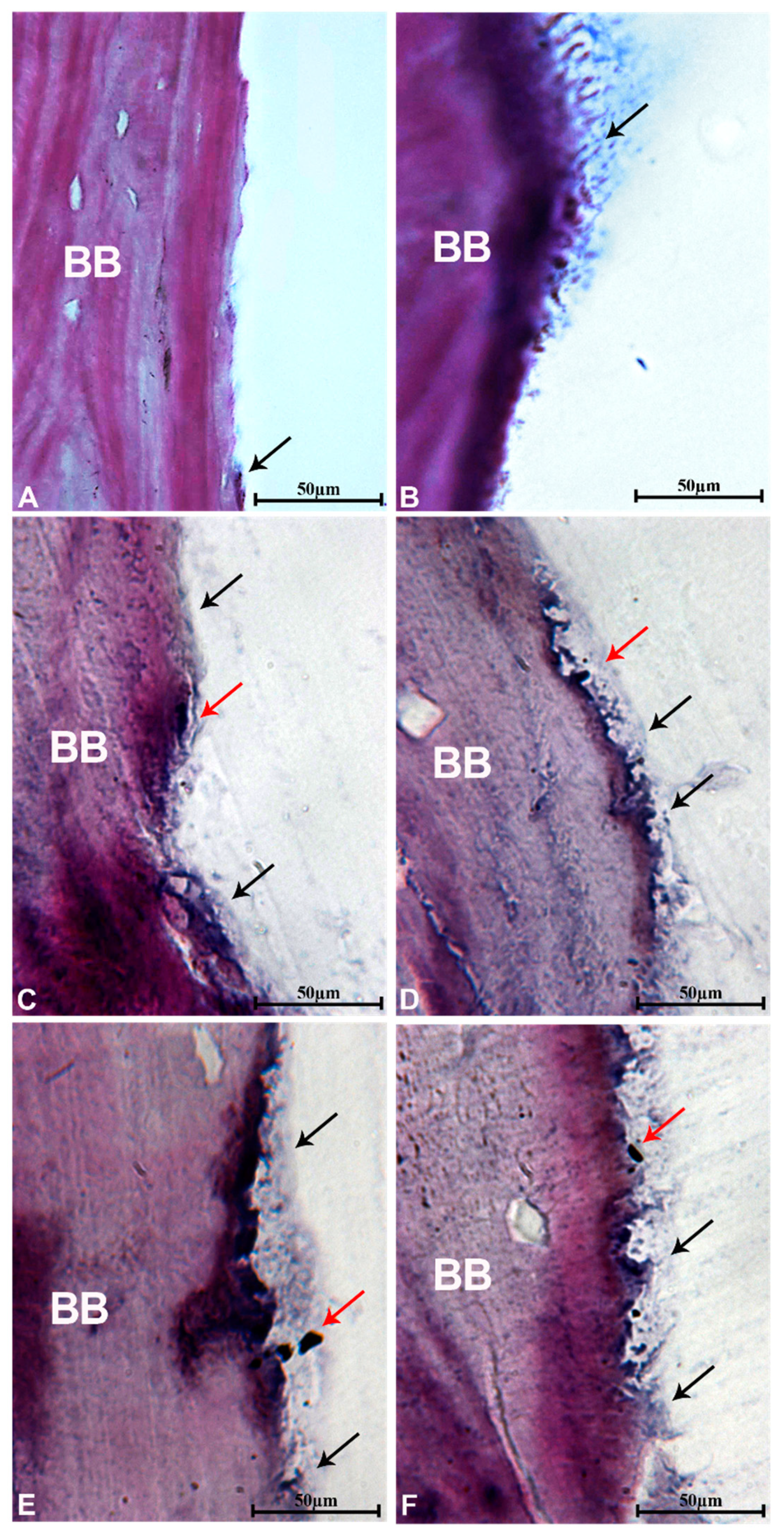
3.9. Histological Evaluation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Drosse, I.; Volkmer, E.; Capanna, R.; Biase, P.D.; Mutschler, W.; Schieker, M. Tissue Engineering for Bone Defect Healing: An Update on a Multi-Component Approach. Injury 2008, 39, S9–S20. [Google Scholar] [CrossRef]
- Cassetta, M.; Perrotti, V.; Calasso, S.; Piattelli, A.; Sinjari, B.; Iezzi, G. Bone Formation in Sinus Augmentation Procedures Using Autologous Bone, Porcine Bone, and a 50:50 Mixture: A Human Clinical and Histological Evaluation at 2 Months. Clin. Oral Implant Res. 2015, 26, 1180–1184. [Google Scholar] [CrossRef]
- Becker, W.; Urist, M.; Becker, B.E.; Jackson, W.; Party, D.A.; Bartold, M.; Vincenzzi, G.; De Georges, D.; Niederwanger, M. Clinical and Histologic Observations of Sites Implanted with Intraoral Autologous Bone Grafts or Allografts. 15 Human Case Reports. J. Periodontol. 1996, 67, 1025–1033. [Google Scholar] [CrossRef]
- Albrektsson, T.; Johansson, C. Osteoinduction, Osteoconduction and Osseointegration. Eur. Spine J. 2001, 10, S96–S101. [Google Scholar]
- Sculean, A.; Nikolidakis, D.; Nikou, G.; Ivanovic, A.; Chapple, I.L.C.; Stavropoulos, A. Biomaterials for Promoting Periodontal Regeneration in Human Intrabony Defects: A Systematic Review. Periodontology 2000 2015, 68, 182–216. [Google Scholar] [CrossRef]
- Kim, Y.-K.; Yun, P.-Y.; Kim, S.-G.; Lim, S.-C. Analysis of the Healing Process in Sinus Bone Grafting Using Various Grafting Materials. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2009, 107, 204–211. [Google Scholar] [CrossRef] [PubMed]
- Jakoi, A.M.; Iorio, J.A.; Cahill, P.J. Autologous Bone Graft Harvesting: A Review of Grafts and Surgical Techniques. Musculoskelet. Surg. 2015, 99, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Iaculli, F.; Di Filippo, E.S.; Piattelli, A.; Mancinelli, R.; Fulle, S. Dental Pulp Stem Cells Grown on Dental Implant Titanium Surfaces: An in Vitro Evaluation of Differentiation and MicroRNAs Expression. J. Biomed. Mater. Res. Part B Appl. Biomater. 2017, 105, 953–965. [Google Scholar] [CrossRef] [PubMed]
- Motamedian, S.R.; Tabatabaei, F.S.; Akhlaghi, F.; Torshabi, M.; Gholamin, P.; Khojasteh, A. Response of Dental Pulp Stem Cells to Synthetic, Allograft, and Xenograft Bone Scaffolds. Int. J. Periodontics Restor. Dent. 2017, 37, 49–59. [Google Scholar] [CrossRef]
- Graziano, A.; d’Aquino, R.; Laino, G.; Papaccio, G. Dental Pulp Stem Cells: A Promising Tool for Bone Regeneration. Stem Cell Rev. 2008, 4, 21–26. [Google Scholar] [CrossRef]
- Del Fabbro, M.; Testori, T.; Francetti, L.; Weinstein, R. Systematic Review of Survival Rates for Implants Placed in the Grafted Maxillary Sinus. J. Prosthet. Dent. 2005, 94, 266. [Google Scholar] [CrossRef]
- Tumedei, M.; Savadori, P.; Del Fabbro, M. Synthetic Blocks for Bone Regeneration: A Systematic Review and Meta-Analysis. Int. J. Mol. Sci. 2019, 20, 4221. [Google Scholar] [CrossRef] [PubMed]
- Scarano, A.; Degidi, M.; Iezzi, G.; Pecora, G.; Piattelli, M.; Orsini, G.; Caputi, S.; Perrotti, V.; Mangano, C.; Piattelli, A. Maxillary Sinus Augmentation with Different Biomaterials: A Comparative Histologic and Histomorphometric Study in Man. Implant Dent. 2006, 15, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Hasan, A.; Byambaa, B.; Morshed, M.; Cheikh, M.I.; Shakoor, R.A.; Mustafy, T.; Marei, H.E. Advances in Osteobiologic Materials for Bone Substitutes. J. Tissue Eng. Regen. Med. 2018, 12, 1448–1468. [Google Scholar] [CrossRef] [PubMed]
- Tetè, S.; Zizzari, V.L.; Vinci, R.; Zara, S.; Di Tore, U.; Manica, M.; Cataldi, A.; Mortellaro, C.; Piattelli, A.; Gherlone, E. Equine and Porcine Bone Substitutes in Maxillary Sinus Augmentation: A Histological and Immunohistochemical Analysis of VEGF Expression. J. Craniofac. Surg. 2014, 25, 835–839. [Google Scholar] [CrossRef]
- Felice, P.; Piana, L.; Checchi, L.; Corvino, V.; Nannmark, U.; Piattelli, M. Vertical Ridge Augmentation of an Atrophic Posterior Mandible with an Inlay Technique and Cancellous Equine Bone Block: A Case Report. Int. J. Periodontics Restor. Dent. 2013, 33, 159–166. [Google Scholar] [CrossRef]
- Barone, A.; Toti, P.; Menchini-Fabris, G.-B.; Felice, P.; Marchionni, S.; Covani, U. Early Volumetric Changes after Vertical Augmentation of the Atrophic Posterior Mandible with Interpositional Block Graft versus Onlay Bone Graft: A Retrospective Radiological Study. J. Cranio-Maxillofac. Surg. 2017, 45, 1438–1447. [Google Scholar] [CrossRef]
- Orsini, G.; Scarano, A.; Piattelli, M.; Piccirilli, M.; Caputi, S.; Piattelli, A. Histologic and Ultrastructural Analysis of Regenerated Bone in Maxillary Sinus Augmentation Using a Porcine Bone–Derived Biomaterial. J. Periodontol. 2006, 77, 1984–1990. [Google Scholar] [CrossRef]
- Scarano, A.; Carinci, F.; Assenza, B.; Piattelli, M.; Murmura, G.; Piattelli, A. Vertical Ridge Augmentation of Atrophic Posterior Mandible Using an Inlay Technique with a Xenograft without Miniscrews and Miniplates: Case Series: Vertical Ridge Augmentation of Atrophic Posterior Mandible. Clin. Oral Implant. Res. 2011, 22, 1125–1130. [Google Scholar] [CrossRef]
- Tumedei, M.; Mancinelli, R.; Di Filippo, E.S.; Marrone, M.; Iezzi, G.; Piattelli, A.; Fulle, S. Osteogenic Potential of Human Dental Pulp Stem Cells Co-Cultured with Equine Bone Substitute Combined with Melatonin. Int. J. Periodontics Restor. Dent. 2021. accepted. [Google Scholar]
- Roth, J.A.; Kim, B.-G.; Lin, W.-L.; Cho, M.-I. Melatonin Promotes Osteoblast Differentiation and Bone Formation. J. Biol. Chem. 1999, 274, 22041–22047. [Google Scholar] [CrossRef] [PubMed]
- Allegra, M.; Reiter, R.J.; Tan, D.-X.; Gentile, C.; Tesoriere, L.; Livrea, M.A. The Chemistry of Melatonin’s Interaction with Reactive Species: Melatonin, a Radical Scavenger. J. Pineal Res. 2003, 34, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sethi, S.; Radio, N.M.; Kotlarczyk, M.P.; Chen, C.-T.; Wei, Y.-H.; Jockers, R.; Witt-Enderby, P.A. Determination of the Minimal Melatonin Exposure Required to Induce Osteoblast Differentiation from Human Mesenchymal Stem Cells and These Effects on Downstream Signaling Pathways: Chronic Melatonin Induces Osteoblast Formation. J. Pineal Res. 2010, 49, 222–238. [Google Scholar] [CrossRef]
- Gómez-Moreno, G.; Aguilar-Salvatierra, A.; Boquete-Castro, A.; Guardia, J.; Piattelli, A.; Perrotti, V.; Delgado-Ruiz, R.A.; Calvo-Guirado, J.L. Outcomes of Topical Applications of Melatonin in Implant Dentistry: A Systematic Review. Implant Dent. 2015, 24, 25–30. [Google Scholar] [CrossRef]
- Calvo-Guirado, J.L.; Gómez-Moreno, G.; Barone, A.; Cutando, A.; Alcaraz-Baños, M.; Chiva, F.; López-Marí, L.; Guardia, J. Melatonin plus Porcine Bone on Discrete Calcium Deposit Implant Surface Stimulates Osteointegration in Dental Implants. J. Pineal Res. 2009, 47, 164–172. [Google Scholar] [CrossRef]
- Maria, S.; Witt-Enderby, P.A. Melatonin Effects on Bone: Potential Use for the Prevention and Treatment for Osteopenia, Osteoporosis, and Periodontal Disease and for Use in Bone-Grafting Procedures. J. Pineal Res. 2014, 56, 115–125. [Google Scholar] [CrossRef] [PubMed]
- De Marco, P.; Zara, S.; De Colli, M.; Radunovic, M.; Lazović, V.; Ettorre, V.; Di Crescenzo, A.; Piattelli, A.; Cataldi, A.; Fontana, A. Graphene Oxide Improves the Biocompatibility of Collagen Membranes in an in Vitro Model of Human Primary Gingival Fibroblasts. Biomed. Mater. 2017, 12, 055005. [Google Scholar] [CrossRef] [PubMed]
- Guazzo, R.; Gardin, C.; Bellin, G.; Sbricoli, L.; Ferroni, L.; Ludovichetti, F.S.; Piattelli, A.; Antoniac, I.; Bressan, E.; Zavan, B. Graphene-Based Nanomaterials for Tissue Engineering in the Dental Field. Nanomaterials 2018, 8, 349. [Google Scholar] [CrossRef] [PubMed]
- Radunovic, M.; De Colli, M.; De Marco, P.; Di Nisio, C.; Fontana, A.; Piattelli, A.; Cataldi, A.; Zara, S. Graphene Oxide Enrichment of Collagen Membranes Improves DPSCs Differentiation and Controls Inflammation Occurrence. J. Biomed. Mater. Res. A 2017, 105, 2312–2320. [Google Scholar] [CrossRef] [PubMed]
- Tahriri, M.; Del Monico, M.; Moghanian, A.; Tavakkoli Yaraki, M.; Torres, R.; Yadegari, A.; Tayebi, L. Graphene and Its Derivatives: Opportunities and Challenges in Dentistry. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 102, 171–185. [Google Scholar] [CrossRef]
- Jing, D.; Hao, J.; Shen, Y.; Tang, G.; Li, M.-L.; Huang, S.-H.; Zhao, Z.-H. The Role of MicroRNAs in Bone Remodeling. Int. J. Oral Sci. 2015, 7, 131–143. [Google Scholar] [CrossRef]
- Harada, S.; Rodan, G.A. Control of Osteoblast Function and Regulation of Bone Mass. Nature 2003, 423, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Lian, J.B.; Stein, G.S.; Javed, A.; van Wijnen, A.J.; Stein, J.L.; Montecino, M.; Hassan, M.Q.; Gaur, T.; Lengner, C.J.; Young, D.W. Networks and Hubs for the Transcriptional Control of Osteoblastogenesis. Rev. Endocr. Metab. Disord. 2006, 7, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Shookhoof, J.M.; Gallicano, G.A. The Emerging Role of microRNAs in Adult Stem Cells; Stem Cell Biology and Regenerative Medicine; Phinney, D.G., Ed.; Springer: Cham, Switzerland, 2011. [Google Scholar]
- Li, Z.; Hassan, M.Q.; Volinia, S.; van Wijnen, A.J.; Stein, J.L.; Croce, C.M.; Lian, J.B.; Stein, G.S. A MicroRNA Signature for a BMP2-Induced Osteoblast Lineage Commitment Program. Proc. Natl. Acad. Sci. USA 2008, 105, 13906–13911. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xie, R.L.; Croce, C.M.; Stein, J.L.; Lian, J.B.; van Wijnen, A.J.; Stein, G.S. A Program of MicroRNAs Controls Osteogenic Lineage Progression by Targeting Transcription Factor Runx2. Proc. Natl. Acad. Sci. USA 2011, 108, 9863–9868. [Google Scholar] [CrossRef]
- Sharma, R.; Kapusetti, G.; Bhong, S.Y.; Roy, P.; Singh, S.K.; Singh, S.; Balavigneswaran, C.K.; Mahato, K.K.; Ray, B.; Maiti, P.; et al. Osteoconductive Amine-Functionalized Graphene–Poly(Methyl Methacrylate) Bone Cement Composite with Controlled Exothermic Polymerization. Bioconjug. Chem. 2017, 28, 2254–2265. [Google Scholar] [CrossRef] [PubMed]
- Boaretti, M.; Castellani, F.; Merli, M.; Lucidi, C.; Lleo, M.M. Presence of Multiple Bacterial Markers in Clinical Samples Might Be Useful for Presumptive Diagnosis of Infection in Cirrhotic Patients with Culture-Negative Reports. Eur. J. Clin. Microbiol. Infect. Dis. 2016, 35, 433–441. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Berney, M.; Hammes, F.; Bosshard, F.; Weilenmann, H.-U.; Egli, T. Assessment and Interpretation of Bacterial Viability by Using the LIVE/DEAD BacLight Kit in Combination with Flow Cytometry. Appl. Environ. Microbiol. 2007, 73, 3283–3290. [Google Scholar] [CrossRef] [PubMed]
- Ettorre, V.; De Marco, P.; Zara, S.; Perrotti, V.; Scarano, A.; Di Crescenzo, A.; Petrini, M.; Hadad, C.; Bosco, D.; Zavan, B.; et al. In Vitro and in Vivo Characterization of Graphene Oxide Coated Porcine Bone Granules. Carbon 2016, 103, 291–298. [Google Scholar] [CrossRef]
- Zhao, Q.; Chen, D.; Li, Y.; Zhang, G.; Zhang, F.; Fan, X. Rhodium Complex Immobilized on Graphene Oxide as an Efficient and Recyclable Catalyst for Hydrogenation of Cyclohexene. Nanoscale 2013, 5, 882–885. [Google Scholar] [CrossRef]
- Kozielski, M.; Buchwald, T.; Szybowicz, M.; Błaszczak, Z.; Piotrowski, A.; Ciesielczyk, B. Determination of Composition and Structure of Spongy Bone Tissue in Human Head of Femur by Raman Spectral Mapping. J. Mater. Sci. Mater. Med. 2011, 22, 1653–1661. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, A.C.; Meyer, J.C.; Scardaci, V.; Casiraghi, C.; Lazzeri, M.; Mauri, F.; Piscanec, S.; Jiang, D.; Novoselov, K.S.; Roth, S.; et al. Raman Spectrum of Graphene and Graphene Layers. Phys. Rev. Lett. 2006, 97. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, A.C.; Robertson, J. Interpretation of Raman Spectra of Disordered and Amorphous Carbon. Phys. Rev. B 2000, 61, 14095–14107. [Google Scholar] [CrossRef]
- Ellingham, S.T.D.; Thompson, T.J.U.; Islam, M. Thermogravimetric Analysis of Property Changes and Weight Loss in Incinerated Bone. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2015, 438, 239–244. [Google Scholar] [CrossRef]
- Gangaraju, V.K.; Lin, H. MicroRNAs: Key Regulators of Stem Cells. Nat. Rev. Mol. Cell Biol. 2009, 10, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Inose, H.; Ochi, H.; Kimura, A.; Fujita, K.; Xu, R.; Sato, S.; Iwasaki, M.; Sunamura, S.; Takeuchi, Y.; Fukumoto, S.; et al. A MicroRNA Regulatory Mechanism of Osteoblast Differentiation. Proc. Natl. Acad. Sci. USA 2009, 106, 20794–20799. [Google Scholar] [CrossRef]
- Li, Z.; Hassan, M.Q.; Jafferji, M.; Aqeilan, R.I.; Garzon, R.; Croce, C.M.; van Wijnen, A.J.; Stein, J.L.; Stein, G.S.; Lian, J.B. Biological Functions of MiR-29b Contribute to Positive Regulation of Osteoblast Differentiation. J. Biol. Chem. 2009, 284, 15676–15684. [Google Scholar] [CrossRef]
- Wei, J.; Li, H.; Wang, S.; Li, T.; Fan, J.; Liang, X.; Li, J.; Han, Q.; Zhu, L.; Fan, L.; et al. Let-7 Enhances Osteogenesis and Bone Formation While Repressing Adipogenesis of Human Stromal/Mesenchymal Stem Cells by Regulating HMGA2. Stem Cells Dev. 2014, 23, 1452–1463. [Google Scholar] [CrossRef]
- Piattelli, A.; Scarano, A.; Quaranta, M. High-Precision, Cost-Effective Cutting System for Producing Thin Sections of Oral Tissues Containing Dental Implants. Biomaterials 1997, 18, 577–579. [Google Scholar] [CrossRef]
- Spalazzi, J.P.; Doty, S.B.; Moffat, K.L.; Levine, W.N.; Lu, H.H. Development of Controlled Matrix Heterogeneity on a Triphasic Scaffold for Orthopedic Interface Tissue Engineering. Tissue Eng. 2006, 12, 3497–3508. [Google Scholar] [CrossRef]
- Edgar, L.; McNamara, K.; Wong, T.; Tamburrini, R.; Katari, R.; Orlando, G. Heterogeneity of Scaffold Biomaterials in Tissue Engineering. Materials 2016, 9, 332. [Google Scholar] [CrossRef] [PubMed]
- Aghaloo, T.L.; Tencati, E.; Hadaya, D. Biomimetic Enhancement of Bone Graft Reconstruction. Oral Maxillofac. Surg. Clin. N. Am. 2019, 31, 193–205. [Google Scholar] [CrossRef] [PubMed]
- Scarano, A.; Lorusso, F.; Staiti, G.; Sinjari, B.; Tampieri, A.; Mortellaro, C. Sinus Augmentation with Biomimetic Nanostructured Matrix: Tomographic, Radiological, Histological and Histomorphometrical Results after 6 Months in Humans. Front. Physiol. 2017, 8, 565. [Google Scholar] [CrossRef] [PubMed]
- Hoornaert, A.; Maazouz, Y.; Pastorino, D.; Aparicio, C.; de Pinieux, G.; Fellah, B.H.; Ginebra, M.-P.; Layrolle, P. Vertical Bone Regeneration with Synthetic Biomimetic Calcium Phosphate onto the Calvaria of Rats. Tissue Eng. Part C Methods 2019, 25, 1–11. [Google Scholar] [CrossRef]

| Gene Symbol | Days | Growth Medium | Differentiation Medium | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTRL | G-N2 | G-N10 | CTRL + MEL | G-N2 + MEL | G-N10 + MEL | CTRL | G-N2 | G-N10 | CTRL ù + MEL | G-N2 + MEL | G-N10 + MEL | ||
| SMAD5 | 7 | 6.7 ± 0.092 | 6.9 ± 0.03 | 6.52 ± 0.04 | 6.58 ± 0.007 | 6.2 ± 0.007 *** | 6.42 ± 0.05 | 6.18 ± 0.06 | 6.8 ± 0.014 *** | 6.46 ± 0.07 * ## | 6.01 ± 0.06 | 6.56 ± 0.06 *** | 7.06 ± 0.012 *** |
| 14 | 5.5 ± 0.05 | 6 ± 0.03 | 5.4 ± 0.08 | 6 ± 0.05 | 5.8 ± 0.03 | 5.6 ± 0.1 | 7.4 ± 0.1 | 6.6 ± 0.006 *** | 5.8 ± 0.08 *** | 6.4 ± 0.035 | 6 ± 0.01 *** | 6.15 ± 0.015 *** | |
| 21 | 7.2 ± 0.02 | 6.3 ± 0.1 *** | 6.7 ± 0.04 * ## | 7.2 ± 0.07 | 6.2 ± 0.02 *** | 6 ± 0.04 *** | 8.25 ± 0.03 | 8.4 ± 0.14 | 8 ± 0.05 ** | 8.1 ± 0.06 | 8.4 ± 0.025 ** | 8.3 ± 0.05 | |
| RUNX2 | 7 | 7.1 ± 0.08 | 7.1 ± 0.017 | 6.6 ± 0.08 *** | 6.6 ± 0.13 | 6.2 ± 0.04 *** | 6.55 ± 0.092 | 6.2 ± 0.01 | 6 ± 0.03 | 5.8 ± 0.04 | 6.1 ± 0.6 | 5.7 ± 0.06 | 5.9 ± 0.05 |
| 14 | 5.36 ± 0.015 | 5.6 ± 0.04 ** | 5.26 ± 0.07 | 5.2 ± 0.07 | 5.53 ± 0.036 *** | 4.96 ± 0.035 *** | 8.5 ± 0.06 | 6.1 ± 0.02 *** | 6 ± 0.07 *** | 6.5 ± 0.09 | 5.5 ± 0.07 *** | 5.7 ± 0.1 *** | |
| 21 | 6.5 ± 0.05 | 6 ± 0.0 *** | 6 ± 0.01 *** | 6.7 ± 0.007 | 6.2 ± 0.02 *** | 6 ± 0.04 *** | 7.4 ± 0.075 | 7.6 ± 0.08 | 7.25 ± 0.04 | 7.25 ± 0.007 | 7.4 ± 0.09 | 7.7 ± 0.035 *** | |
| miR Symbol | Days | Growth Medium | Differentiation Medium | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTRL | G-N2 | G-N10 | CTRL + MEL | G-N2 + MEL | G-N10 + MEL | CTRL | G-N2 | G-N10 | CTRL + MEL | G-N2 + MEL | G-N10 + MEL | ||
| miR-133a | 7 | 11.6 ± 0.2 | 12.5 ± 0.4 ** | 12.8 ± 0.1 *** | 13.1 ± 0.1 | 13.1 ± 0.2 | --- | 10.9 ± 0.05 | 11.6 ± 0.7 | 10.6 ± 0.1 | 10.8 ± 0.8 | 9.7 ± 0.3 | 12.3 ± 0.1 * |
| 14 | 11.2 ± 0.2 | 11.5 ± 0.7 | 11.9 ± 1.01 | 11.3 ± 0.7 | 12.1 ± 0.3 | 11.3 ± 0.8 | 9 ± 0.3 | 10.7 ± 0.1 | 11.5 ± 0.5 * | 10 ± 0.2 | 11.7 ± 0.9 | 12.3 ± 0.8 ** | |
| 21 | 11.3 ± 0.2 | 11.5 ± 0.7 | 12.7 ± 0.8 | 11.3 ± 0.7 | 12.5 ± 0.3 | 12.3 ± 0.1 | 12.2 ± 0.05 | 12.2 ± 0.6 | 13.2 ± 0.6 | 11.6 ± 0.4 | 11.9 ± 0.2 | 11.7 ± 0.5 | |
| miR-133b | 7 | 10.1 ± 0.4 | 12.3 ± 0.2 | --- | 11.4 ± 0.8 | 13 ± 0.8 | --- | 11.4 ± 0.05 | 11.9 ± 0.4 | 11.9 ± 0.2 | 11.7 ± 0.6 | 12.4 ± 0.02 | --- |
| 14 | 11 ± 0.4 | 12 ± 0.4 | 12.2 ± 1.3 | 11.2 ± 0.1 | 12.3 ± 0.4 | 12.8 ± 0.8 | 11.1 ± 0.6 | 11.5 ± 0.8 | 11.5 ± 0.8 | 10.8 ± 0.2 | 11.1 ± 0.9 | 12.5 ± 0.8 | |
| 21 | 12.6 ± 0.4 | 12.4 ± 0.4 | 12.8 ± 0.05 | 11.5 ± 0.7 | 12.7 ± 0.2 | 12.6 ± 0.1 | 11.8 ± 0.05 | 12 ± 0.9 | 10.5 ± 0.5 | 10.5 ± 0.3 | 12.2 ± 0.1 * | 14.4 ± 0.4 *** | |
| miR-135a | 7 | 11 ± 0.5 | 12.7 ± 0.1 * | 12.3 ± 0.6 | 11.1 ± 0.1 | 12.4 ± 0.8 | 12.4 ± 0.4 | 11.2 ± 0.05 | 13.4 ± 0.7 *** | 12.2 ± 0.1 | 11.8 ± 0.1 | 10.4 ± 0.4 * | --- |
| 14 | 10.2 ± 0.1 | 10.9 ± 0.7 | 11.03 ± 0.2 | 10.6 ± 1.1 | 10.6 ± 0.6 | 10.8 ± 0.3 | 12 ± 0.1 | --- | --- | 12.2 ± 0.2 | --- | --- | |
| 21 | 12.1 ± 0.5 | 11.7 ± 0.3 | 12.3 ± 0.5 | 12.5 ± 0.6 | 12.2 ± 0.2 | 12.1 ± 0.4 | 10.9 ± 0.5 | 12 ± 1.04 | 12.8 ± 0.6 * | 11.8 ± 0.6 | 12.1 ± 0.5 | 11.3 ± 0.7 | |
| miR-206 | 7 | 10.8 ± 0.8 | 12 ± 0.1 | 11.8 ± 1.1 | 11.4 ± 0.8 | 12 ± 0.7 | 11 ± 0.3 | 11.9 ± 0.6 | 11.9 ± 0.9 | 12.4 ± 0.2 | 11.7 ± 0.8 | 11.2 ± 0.5 | 11.6 ± 0.6 |
| 14 | 9.9 ± 0.2 | 12.1 ± 0.4 *** | 11.5 ± 0.6 ** | 11.7 ± 0.4 | 11.4 ± 0.4 | 12.4 ± 0.3 | 11.5 ± 0.3 | 11.1 ± 0.04 | 11.2 ± 0.4 | 11.6 ± 0.7 | 11 ± 0.8 | 11.9 ± 0.3 | |
| 21 | 12 ± 0.9 | 12.6 ± 0.5 | 11.3 ± 0.8 | 12.5 ± 0.7 | 11.5 ± 0.1 | 11.8 ± 0.3 | 12 ± 0.8 | 11.3 ± 0.5 | 12.3 ± 1.1 | 11.4 ± 0.4 | 11.8 ± 0.2 | 11.2 ± 0.07 | |
| miR-29b | 7 | 3.1 ± 0.2 | 2.7 ± 0.2 | 2.4 ± 0.1 ** | 2.4 ± 0.05 | 3 ± 0.2 ** | 1.9 ± 0.1 * | 4.62 ± 0.02 | 4.99 ± 0.04 ** | 4.85 ± 0.1 | 4.14 ± 0.03 | 4.87 ± 0.02 *** | 3.81 ± 0.08 ** |
| 14 | 2.9 ± 0.2 | 2.5 ± 0.1 | 1.7 ± 0.3 *** | 1.9 ± 0.2 | 1.6 ± 0.1 | 2.1 ± 0.2 | 3 ± 0.3 | 3.4 ± 0.05 | 3.9 ± 0.2 ** | 2.7 ± 0.1 | 2.9 ± 0.1 | 3.3 ± 0.2 * | |
| 21 | 3.3 ± 0.08 | 2.5 ± 0.1 *** | 3.8 ± 0.2 ** | 2.6 ± 0.1 | 2.6 ± 0.1 | 3 ± 0.04 ** | 2.9 ± 0.06 | 2.7 ± 0.04 ** | 2.7 ± 0.09 * | 3 ± 0.06 | 2.9 ± 0.04 | 2.6 ± 0.07 *** | |
| miR-let-7b | 7 | −1.8 ± 0.02 | −3.1 ± 0.3 *** | −3.7 ± 0.2 *** | −1.7 ± 0.1 | −4.6 ± 0.3 *** | −5 ± 0.2 *** | −1.46 ± 0.07 | −1.80 ± 0.03 | −2.03 ± 0.14 ** | −1.51 ± 0.14 | −1.8 ± 0.12 | −1.96 ± 0.06 * |
| 14 | −2.3 ± 2 | − 2.02 ± 0.2 | −2.3 ± 0.3 | −2.5 ± 0.1 | −2.5 ± 0.2 | −3 ± 0.3 | −1.3 ± 0.07 | −1.5 ± 0.08 | −1.6 ± 0.2 | − 2.1 ± 0.08 | − 1.7 ± 0.08 * | −1.6 ± 0.1 ** | |
| 21 | −2.1 ± 0.1 | −2.6 ± 0.1 *** | −1.9 ± 0.1 | −2.1 ± 0.1 | −2.2 ± 0.04 | −2.4 ± 0.08 * | −1.4 ± 0.05 | −1.7 ± 0.07 *** | −0.7 ± 0.06 *** | −1.05 ± 0.08 | −1.04 ± 0.08 | − 1.6 ± 0.05 *** | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mancinelli, R.; Di Filippo, E.S.; Tumedei, M.; Marrone, M.; Fontana, A.; Ettorre, V.; Giordani, S.; Baldrighi, M.; Iezzi, G.; Piattelli, A.; et al. Human Dental Pulp Stem Cell Osteogenic Differentiation Seeded on Equine Bone Block with Graphene and Melatonin. Appl. Sci. 2021, 11, 3218. https://doi.org/10.3390/app11073218
Mancinelli R, Di Filippo ES, Tumedei M, Marrone M, Fontana A, Ettorre V, Giordani S, Baldrighi M, Iezzi G, Piattelli A, et al. Human Dental Pulp Stem Cell Osteogenic Differentiation Seeded on Equine Bone Block with Graphene and Melatonin. Applied Sciences. 2021; 11(7):3218. https://doi.org/10.3390/app11073218
Chicago/Turabian StyleMancinelli, Rosa, Ester Sara Di Filippo, Margherita Tumedei, Mariangela Marrone, Antonella Fontana, Valeria Ettorre, Silvia Giordani, Michele Baldrighi, Giovanna Iezzi, Adriano Piattelli, and et al. 2021. "Human Dental Pulp Stem Cell Osteogenic Differentiation Seeded on Equine Bone Block with Graphene and Melatonin" Applied Sciences 11, no. 7: 3218. https://doi.org/10.3390/app11073218
APA StyleMancinelli, R., Di Filippo, E. S., Tumedei, M., Marrone, M., Fontana, A., Ettorre, V., Giordani, S., Baldrighi, M., Iezzi, G., Piattelli, A., & Fulle, S. (2021). Human Dental Pulp Stem Cell Osteogenic Differentiation Seeded on Equine Bone Block with Graphene and Melatonin. Applied Sciences, 11(7), 3218. https://doi.org/10.3390/app11073218













