1. Introduction
In the case of implant treatment, which involves the aesthetic zone of the maxilla, immediate or early loading of an implant is extremely important, not only for functionality, but primarily for aesthetic reasons. For decades, high primary stability has been the key factor in determining the early loading of an implant and the success of a treatment [
1,
2].
Osteointegration has been defined as a direct and functional connection between the bone and an artificial implant [
2]. Primary stability is the grade of engagement of an implant in the bone structure immediately after insertion. It is one of the main conditions for the osteointegration process [
1,
2,
3]. The factors that influence the values of primary stability can be divided into three main groups. The first one is the quantitative and qualitative characteristics of the bone structure into which the implant is inserted [
4]. Another factor is the method of implant bed preparation [
5]. The final factor affecting primary stability are the characteristics of an implant, which include both the macroscopic and microscopic features of the implant surface [
6]. The macroscopic features of an implant relate to aspects such as the dimensions, shape, diameter and thread pitch of the implant. The microscopic features are the overall characteristics of the surface, primarily including the grade of the roughness of the surface [
1,
2,
3,
6].
In the context of osteointegration, much attention has been paid in recent years to the physical and chemical aspects of dental implant surfaces. These include electrochemical potential, surface wettability, thickness of the titanium dioxide (TiO
2) layer, ion adhesion, active peptides, growth factors, and antibiotics. Currently, work is still underway to develop a type of surface that would allow for even faster loading of an implant even when primary stability is low [
7,
8,
9]. One such potential method is the chemical modification of the surface using hydroxyl ion. With such a modification, the titanium surface of an implant has a negative electrochemical potential. The negative potential determines the improvement of the osteointegration process at all stages, starting from better stability of blood clot and adhesion of Ca++ ions in the first hours after the loading of a dental implant, through to a better adhesion of proteins (fibronectin, osteocalcin) and the cells that determine the process of osteointegration (mesenchymal stem cells (MSC)) in the later stages of the process [
10,
11].
In the case of implant treatment within the aesthetic zone, a temporary restoration, usually a removable one, is needed. It causes many difficulties for the patient. Therefore, the methods for shortening the healing period and loading an implant as early as possible are constantly being sought after. The first reports claiming a high rate of success concerning early loading of an implant date back to the 1990s and refer to the Branemark implant system [
9,
10].
For decades, implant loading after six weeks was considered an early loading. However, it seems that the key period for the process of osteointegration and secondary stability is in the third and fourth week. During that time, the process of mineralization of the primary osseous tissue takes place, and thus, the bone tissue that surrounds an implant achieves the mechanical values that enable loading. Therefore, it seems reasonable to load an implant even faster, i.e., after 3–4 weeks of healing [
9,
10,
11,
12,
13,
14,
15].
The main objective of the study was to assess the marginal bone loss and stability of the early loaded Thommen Incell®SPI implants using the single non-splinted screw-retained final chairside-prepared prosthetic restoration. The secondary objective was to evaluate the influence of an implant’s diameter on the mentioned parameters.
2. Materials and Methods
2.1. Inclusion and Exclusion Criteria
In this study, 40 patients aged over 18, partially edentulous within the aesthetic zone, were enrolled. The patients could not have any active periodontal disease or an approximal plague index (API) > 25%. The patients were subjected to clinical and radiological examinations. The minimum alveolar ridge dimension in the lingual–buccal aspect was 6.5–7 mm in the region of interest, so the implant could be placed in the native bone. Furthermore, the bone density in the region of the implant insertion had to be D2 or D3 according to Misch et al. [
16]. Patients were randomly divided into 2 separate groups depending on the implant diameter used (3.5 and 4.0 mm).
- (a)
group 1 (G2; n = 20 patients)—3.5 mm diameter implants were used
- (b)
group 2 (G3; n = 20 patients)—4.0 mm diameter implants were used
The procedures of the guided bone regeneration were not performed neither before nor during the implant placement. Furthermore, at least 3 months for the healing period after extraction were established.
Exclusion criteria were:
2.2. Protocol of the Experiment
The schedule of visits included:
consultation visit: qualification of the patient for the surgery, clinical and radiological examination CBCT (cone-beam computed tomography) (Galileos®D3437, Sirona Dental, Erlangen, Germany), API assessment;
implantation: intraoperative and postoperative RVG (radiovisiography) (Planmeca OY, Helsinki, Finland), torque values, primary stability assessment using Ostell ISQ (Osstell; Integration Diagnostics, Gothenburg, Sweden);
4 weeks after the implantation: assessment of stability with the use of Ostell ISQ, intraoral scan, placement of prosthetic, RVG;
12 months after the surgery: clinical and radiological assessment (RVG and CBCT).
The research was performed in accordance with the conditions of declaration of Helsinki and with the approval of the Local Ethical Committee (229/2019). The personal data protection procedures (GDPR) were complied with. The patients signed two written consents: first, a general consent for the implant treatment, and second, consent for participation in the study.
2.3. Implants
The cylindrical dental implants, Thommen Innicell®SPI Element MC Innicel (Thommen Medical AG, Grenchen, Switzerland) were used for the surgery. The superhydrophilic implant surface was obtained through NaOH conditioning using the Apliquiq system (Thommen Medical AG, Grenchen, Switzerland). The length of the inserted implants ranged from 8 mm to 11 mm and depended on the height of the bone base, while the diameter of the implant was determined by the width of the alveolar processes.
2.4. Surgical Phase
The implant surgery was performed with antibiotic cover, one-shot therapy: 1 dose of clindamycin 600 mg (MIP Pharma, Gdansk, Poland). Infiltration anesthesia was applied using Septanest 1:100,000 (SEPTODONT 58, Saint Maur des Fossés, France) with the Wand STA device (Milestone Scientific, Inc., Roseland, NJ, USA). A diamond drill was used for deepithelialization and a blade (no. 15C) was used for an H-shaped papilla-preservation incision, shifted palatially. Next, each implant was inserted at bone level, according to the procedure provided by the manufacturer. Subsequently, the primary stability was assessed using Ostell ISQ. The measurements were performed three times in the mesiodistal, buccal and palatal, as well as periapical direction measurements with the application of Ostel smartpeg for the Thommen implants. The smallest value was considered to be the cut-off point. Open healing was used with a standard healing screw. The partially deepithelized flap was repositioned and stabilized with 0–5 simple interrupted sutures (Seralene
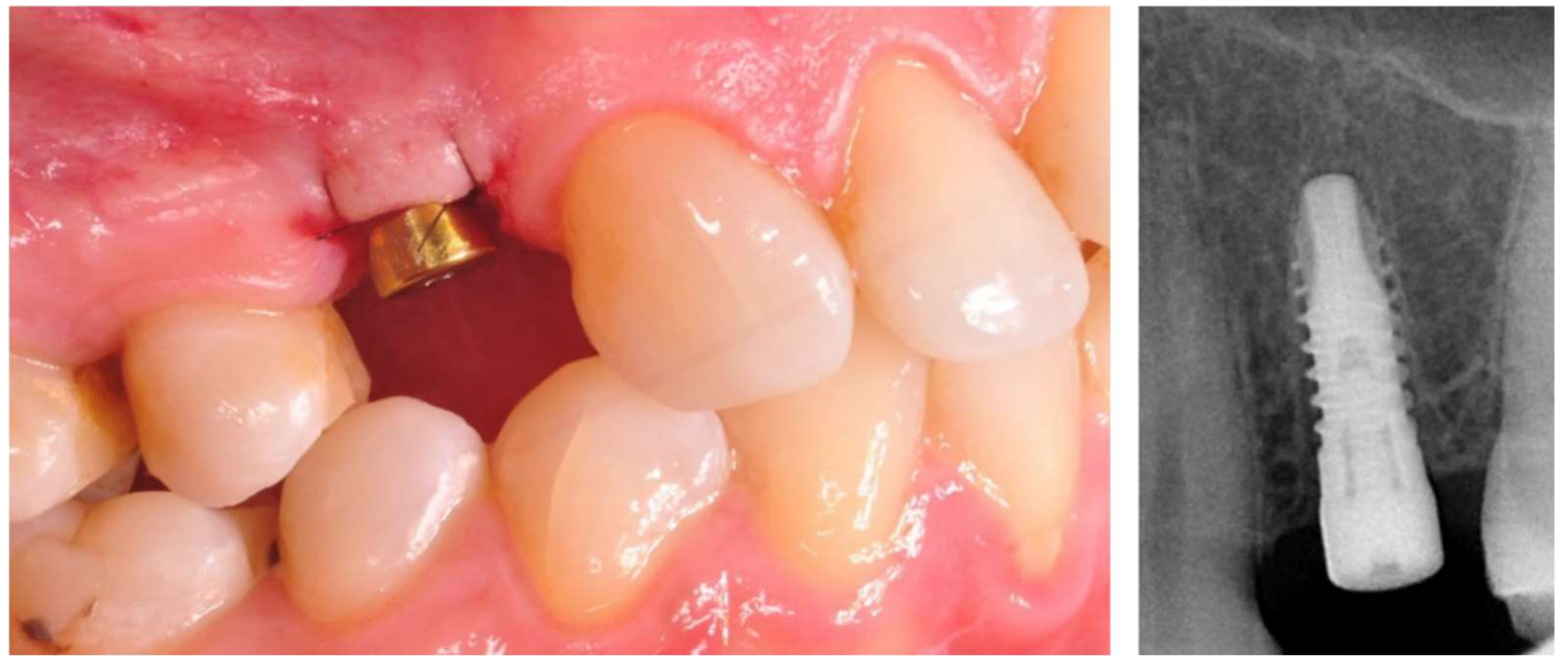
®, Serag Wiessner, Naila, Germany). At the end of the surgery, a RVG image was taken to assess the correctness of the inserted implant (
Figure 1). The X-ray tubehead was aimed at right angles (vertically and horizontally) to both the implant and the sensor. A paralleling device was used for this purpose. The surgeries were performed by three members of the team: M.K., J.H. and A.B. Postoperative recommendations included analgesic and anti-inflammatory treatment with Nimesil (Laboratories Menarini SA, Barcelona, Spain) at 200 mg/per day, and rinsing the oral cavity with Eludril Classic (Pierre Fabre S.A, Paris, France) 3 times a day.
2.5. Prosthetic Phase
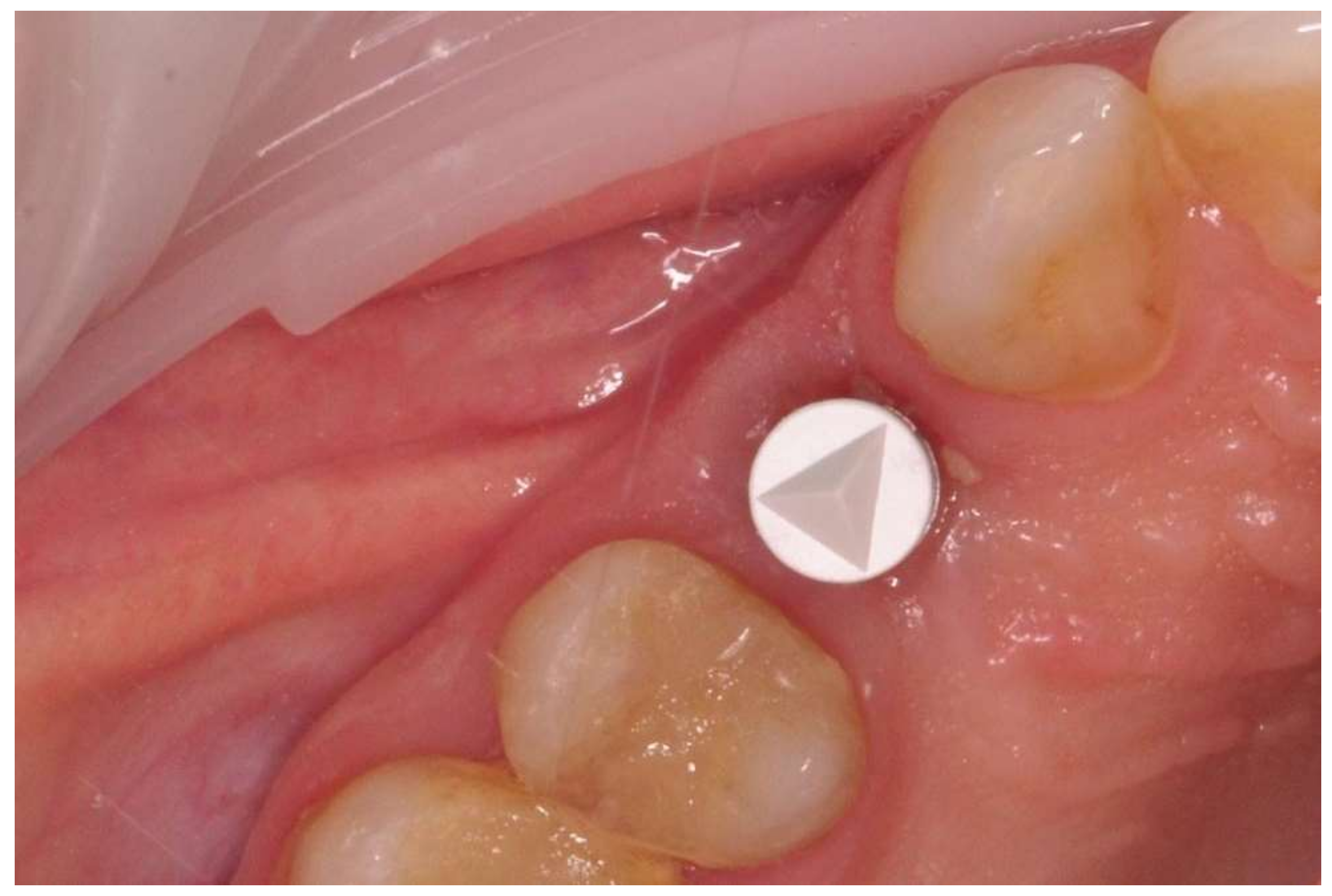
The prosthetic restoration stage started 4 weeks after the implant placement surgery and was prepared in the chairside laboratory by W.G. Patients with no signs of inflammation in the direct vicinity of the implant and with an ISQ (implant’s stability quotient) value of 65 or greater were allowed to participate in the prosthetic protocol. The measurements using the aforementioned device were performed three times in the mesiodistal, buccal and palatal, as well as periapical direction, and the smallest value was considered the cut-off point. Screw-retained implant crowns made of lithium disilicate glass-ceramics, IPS e max CAD LT (Ivoclar Vivadent AG, Schaan, Liechtenstein), were used as the prosthetic restoration materials. After the removal of the healing abutment, the implant bed was cleaned. The scans were taken with an intraoral scanner Sirona Cerec AC Bluecam (DentsplySirona, York, PA, USA) (
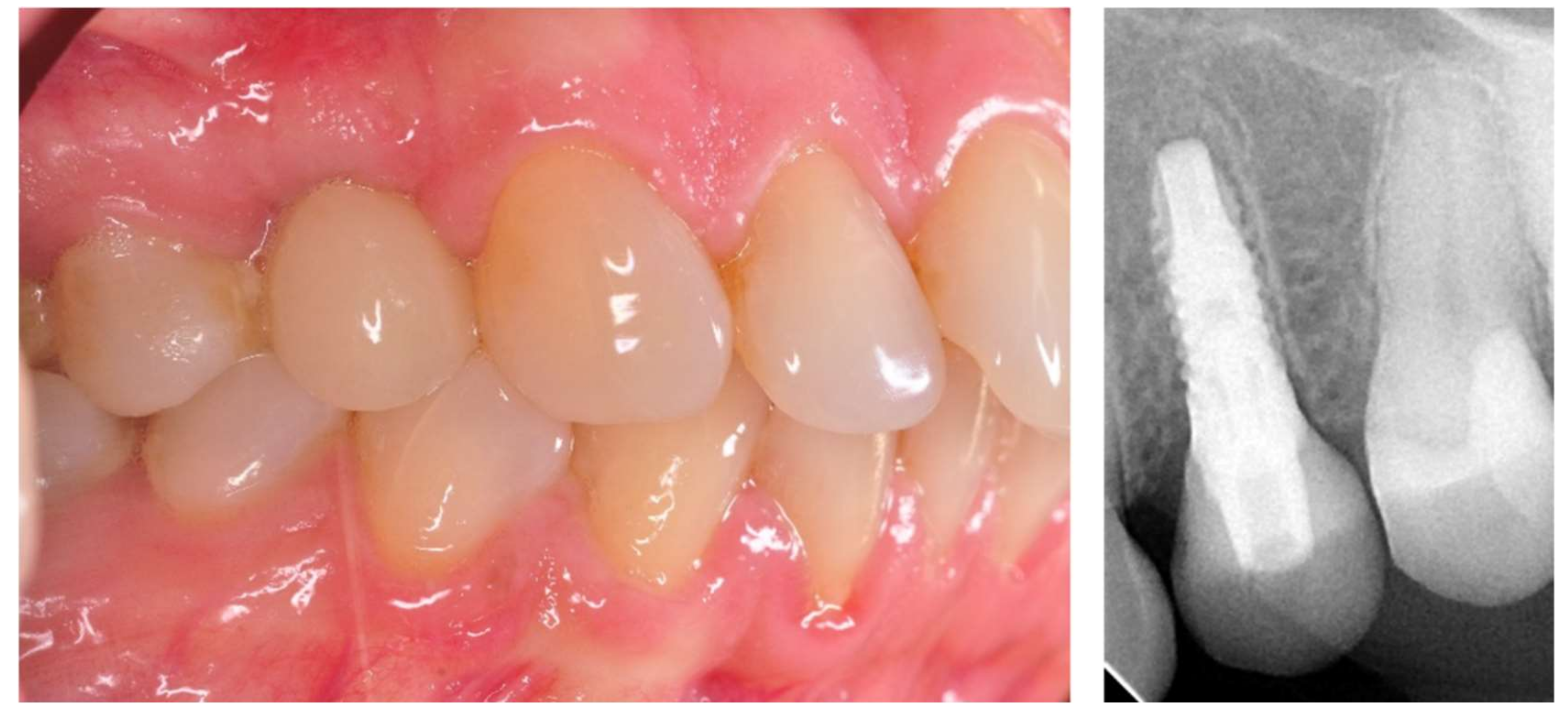
Figure 2). Subsequently, the crown internal surface was etched and then fixed using Multilink Hybrid Abutment cement (Ivoclar Vivadent AG, Schaan, Liechtenstein) on the previously sandblasted titanium base (TiBase) for Sirona Cerec (DentsplySirona, York, PA, USA). The crown was then screwed onto the implant with a force of 25 Ncm. The occluding relations were controlled using articulating paper (Bausch
®, Cologne, Germany) with a thickness of 200, 80, and 8 μm. The hole was filled in with Gradia composite (GC Corporation, Tokyo, Japan) and an RVG image was taken (
Figure 3). The patients were instructed on proper hygiene around the dental implant.
2.6. Assessment of Implant’s Stability
Values of the implant’s stability quotient (ISQ) were obtained immediately after implant placement (primary stability) and after 4 weeks (secondary stability). For every series of resonance frequency analysis (RFA) measurements, the ISQ values were recorded using an Osstell device in three different directions: vertical, buccal and palatal. A transducer (Smartpegs) was attached to the implant, and ISQs ranging from 1 to 100 were recorded. The Osstells were brought into very close contact with the Smartpegs without touching them, until an audible signal confirmed that the measurement had been taken.
2.7. MBL (Marginal Bone Loss) Assessment Using the Radiological Examination
Before surgery and during the 12-month follow-up, CBCT was performed to assess the marginal bone loss (MBL). The MBL was calculated as follows: first, dimensions were calibrated by the known parameters of the implant diameter and length. Starting from the implant shoulder, distances were measured to the mesial and distal points of the bone to implant contact, parallel to the implant axis. All measurements were taken by P.KR, a member of the research group who was not involved directly in the preparation of the implant.
2.8. Statistical Analyses
To answer the research questions, statistical analyses were performed using the IBM SPSS Statistics 25 software (IBM, New York, USA). The software was used to analyze the basic descriptive statistics together with the Shapiro–Wilk test. To examine the differences between two or more groups, a non-parametric equivalent of variance analysis, the Kruskal–Wallis test, was used. Dunn’s test with the Bonferroni correction was chosen for post hoc comparisons. The relationships between continuous variables were examined by calculating Pearson’s linear correlation coefficient. The value of α = 0.05 was assumed as the significance level.
To check the distribution of continuous variables and to study their compliance with a normal distribution, basic descriptive statistics were used, and the Shapiro–Wilk test of normal distribution was performed. For nominal variables, the frequency and the percentage of individual values in the entire observation pool were calculated. The results were presented separately for the three groups. In the first group, all observations were taken into consideration while the second group included only those observations for which an implant diameter = 3.5 mm, and in the third group the implant diameter = 4.0 mm.
4. Discussion
The values for implant stability can be affected by many factors, though not in the same proportions. Sim and Lang investigated the influence of bone density structure and implant length on stability levels, and reported that the length of the implant influences the implant stability only at the time of the surgery and has lesser impact on further stages of osteointegration. Conversely, bone density seems to influence the stability level in a greater way and on all stages of osteointegration [
17]. Subsequently, to standardize the sample population, we selected homogenous bone sites of the frontal aspect of the maxilla as a region of interest.
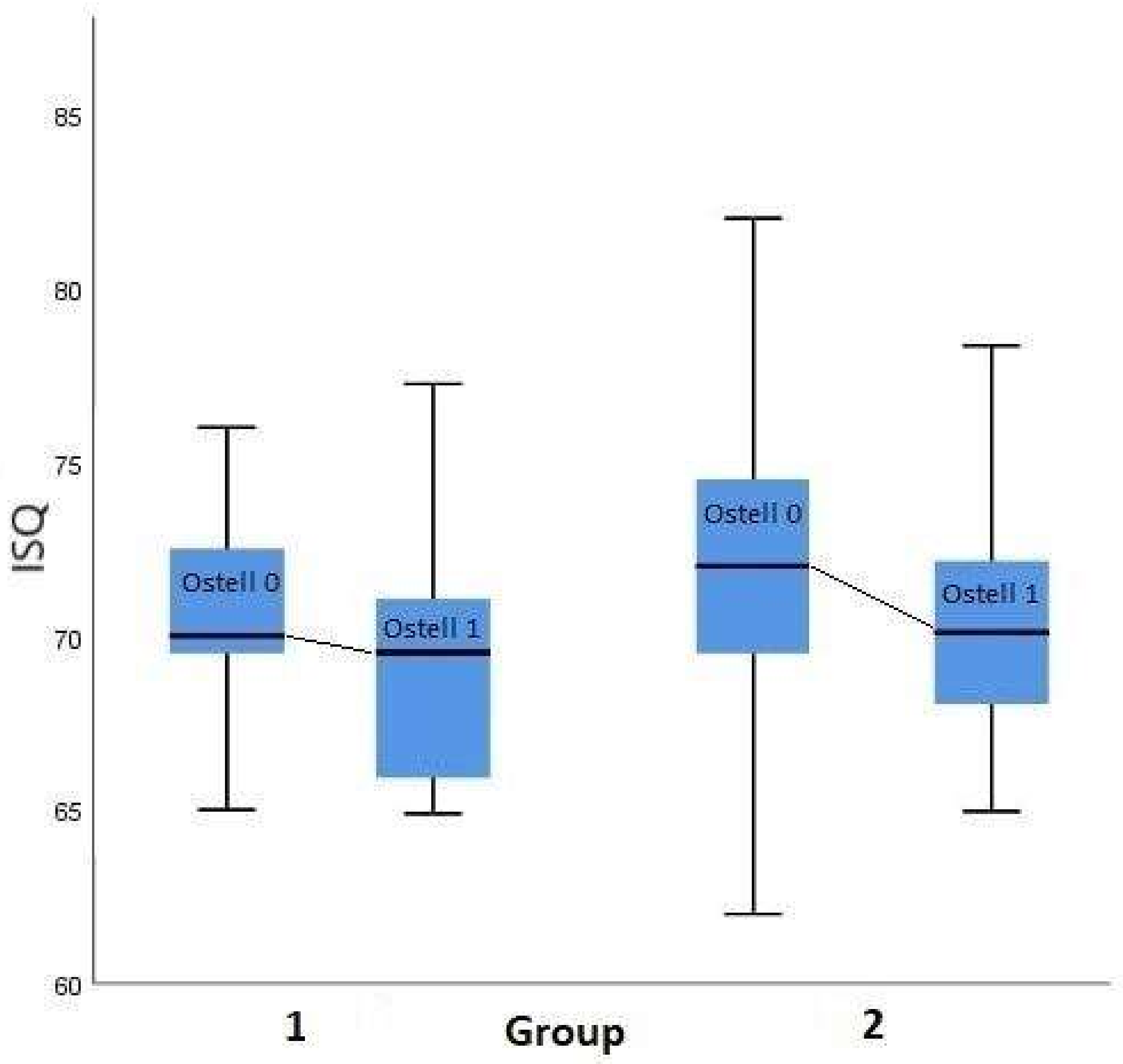
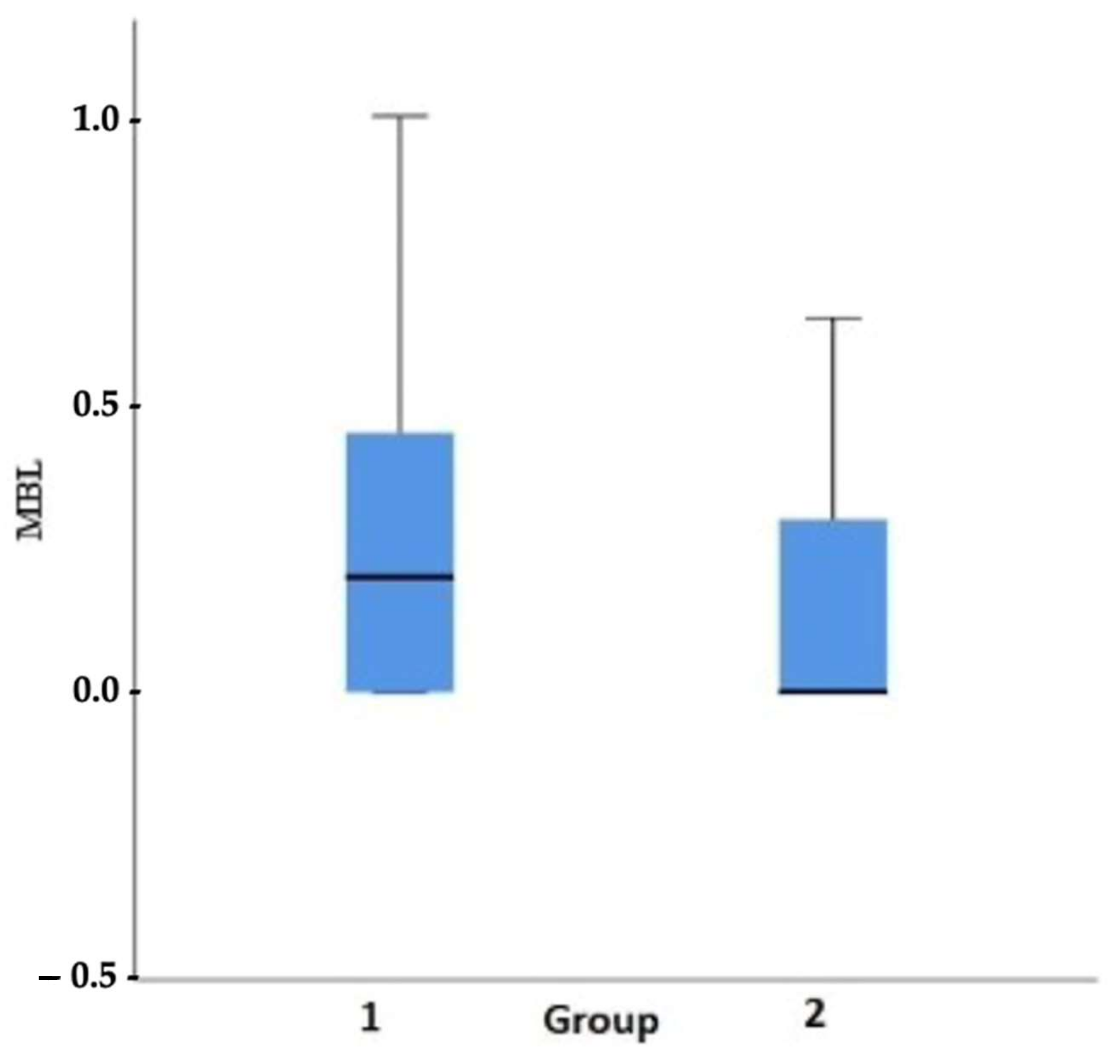
In our study, a decrease in the stability level in the four weeks following the implant insertion was observed (
Figure 4). These results are in accordance with other studies and suggest the existence of bone resorption in direct contact with the implant surface. This behavior is attributed to the time dependency of bone remodeling observed at the initial stage. Therefore, the bone shape and remodeling toward the implant surfaces interfere with the bone-implant contact [
18,
19]. Carmo Filho, in the study on 4.0 and 4.1 mm diameter implants, reported the decline in implant stability at 21 days after surgery for the hydrophilic SLActive implants to 78.8 ± 2.6 ISQ, and to 78.4 ± 3.2 ISQ at 28 days for hydroxyapatite coated implants. However, hydroxyapatite coated implants regain the secondary stability much faster (42 days) when compared to SLActive implants (68 days) [
19].
Regarding the primary stability levels, there is a consensus that ISQ values above 70 are optimal for osteointegration to occur, and they enable the consideration of immediate implant loading. In contrast, the ISQ value of 55 for primary stability is the threshold value for the possibility of leaving the implant in place. Below this value, the implant should be replaced with an implant that enables higher primary stability [
18,
19,
20,
21]. However, there are studies reporting that modification of the implant’s surface in nano-scale allows for successful early loading even in the case of a lower than optimal primary stability level. Östman et al. compared 242 oxidized surface implants, loaded immediately and delayed. Apart from the relatively low primary stability levels (62.9 ± 4.9 ISQ) of immediately loaded implants, the overall success rate was high (99.2%) in that group. Furthermore, the MBL level for immediately loaded implants during 12 months of observation was 0.78 ± 0.9 mm and seems also acceptable [
22].
Subsequent studies addressing this subject demonstrated a correlation between low baseline values of RFA and the potential for implant loss due to lack of osteointegration. Sjöström assessed the primary stability value of 17 implants that were lost within the first year of use. It was found that the average ISQ value in that group was 54.6, whereas the implant group with successful treatments had an average ISQ value of 62.0 [
23]. Other studies reported an average primary stability value of 63.3 ISQ in the group of implants that survived the 12-month follow-up period, whereas, in the group with lost implants, the average primary stability value was 56 ISQ [
24]. Some studies have found a slightly lower (56 ISQ) threshold value for primary stability, which is necessary for osteointegration to occur [
25,
26]. That is why it seems that, apart from the established consensus, this issue is still a current topic and further studies are needed for its full development.
Aragoneses et al. included the implant diameter in the assessment of the levels of secondary stability. The ISQ value measured three months after the insertion of implants with a diameter of 3.7 mm was 69.62, while for implants with larger diameters (4.0 mm and 4.3 mm), that value was 72.02 and 69.67, respectively [
27]. This finding is in agreement with the following study results (
Figure 4 and
Figure 5).
In the following study, hydrophilic surface implants were applied. The hydrophilic surface is regarded to osteointegrate faster than other commonly used types of surfaces, including sand blasted and acid etched. Novelino et al. reported that hydrophilic surface implants gain a stability of 70 ISQ in less than five weeks which means that it is 2.24 times faster than implants treated with sand blasting and acid etching [
20].
The hydrophilic surface implants modified with hydroxyl ion have been assessed for primary and secondary stability only in two other studies to date. Primary stability in the mentioned studies’ plants amounted to 57.3 (±7.4) ISQ and raised to 71.3 (±8.2) ISQ while loading [
28]. The above-mentioned studies mostly focused on measuring the stability of conventionally loaded implants.
An evaluation of the obtained results of MBL shows that they do not deviate from the established norm, according to which MBL should not be greater than 1.5 mm in the first year and then 0.1 mm in each following year. It is important to note that some researchers claim that original remodeling of the alveolar process occurs after implant loading with the aim of restoring biological width [
29,
30,
31,
32,
33]. However, the condition of the surrounding implant soft tissues seems to play a role in this process. Linkevicius et al. reported significantly lower MBL in a one-year observation when the implant surrounding soft tissue was thicker than 2 mm. The average MBL level in those cases was 0.21 mm and is comparable to our results [
29].
The lowest (0.22 ± 0.49 mm) MBL level of early loaded hydrophilic implants with the same, as presented in the following manuscript, observation period (12 months), to date, has been reported by Liaje et al. [
30]. This finding of a low MBL was accompanied by high stability values at all time points (above 71 ISQ) and, similar to the presented study findings, (
Figure 4) wide implants showed statistically higher stability values than narrow implants. On the contrary, much higher MBL levels than those found in our study for hydrophilic implants were reported by Ryu et al. (0.98 ± 0.61 mm) in 13 months of follow-up, and by Hinkle et al. (0.99 ± 0.29) in 12 months of follow-up [
31,
32].
In the literature, there are relatively few studies concerning the effects of early loading and changes in stability in connection with MBL. Olsson et al. were among the first who reported the results of studies concerning 68 early-loaded maxillary implants. The value of the average baseline ISQ parameters was 60.1, whereas the implant survival rate was 93.4% and the MBL level was 1.3 ± 0.6 mm at a 12-month follow-up observation [
33]. Fischer et al. assessed oxidized-surface implants that were loaded for the period ranging from a few days to 16 days. They achieved an overall success rate of 98.1% and the averaged MBL index was 1.1 mm at a 12-month follow-up observation. In contrast, the ISQ index increased from 63.3 (±6.1) to 66.8 (±5.6) after 12 months [
24].
The effect of early loading relative to conventional loading on bone tissue levels (without linking with primary stability levels) was also previously studied in other papers. Degidi et al. assessed immediately loaded implants with a diameter of 3 mm, obtaining the MBL level of 0.85 ± 0.71 mm at a 36-month follow-up observation [
34]. In studies concerning early-loaded implants (three weeks), Grandi proved efficacy comparable to immediate loading (with a loss of one implant in both groups) at a 12-month follow-up observation. However, a higher average MBL was found in the group of implants loaded after three weeks (0.390 ± 0.840 mm) when compared to immediately-loaded implants (0.120 ± 0.230 mm), with no statistically significant differences between those groups [
35]. Other authors report that there is no effect of implant loading time on marginal bone loss. MBL levels in the mentioned studies ranged from 0 to 1.32 mm in longtime observation [
36,
37,
38,
39].
In our previous studies on conventional SLA implants, the average stability level was initially lower (58.67 ± 12.3 ISQ) than in a following study, and raised to 81 ± 5.82 ISQ six months after implant placement and was accompanied by a mean MBL of 0.22 ± 0.46, calculated based on radiographs taken in 36-months of follow-up [
40,41].
The present study has some limitations: firstly, the observation period. We decided to evaluate the MBL level in 12 months. In the literature, there exist studies of such methodology; however, the considerable changes in the bone of the alveolar process can occur in the later stages of implant treatment and further studies are needed to confirm all theses of the present study. Secondly, we decided not to measure the stability levels at the final follow-up after 12 months, as the need to remove the implant restoration would emerge under those conditions and could influence implant preservation.