Tissue Engineering Strategies for Retina Regeneration
Abstract
1. Introduction
2. Cell Types Used for Therapies in the Eye
3. Tissue Engineering of the Retina
4. Biomaterials and Scaffolds Used for Tissue Engineering
4.1. Natural Biomaterials Used as Scaffolds
4.2. Synthetic Biomaterials Used as Scaffolds
4.3. Biohybrid Scaffolds
4.4. Scaffold Free Cell Sheets Using Thermoresponsive Polymers
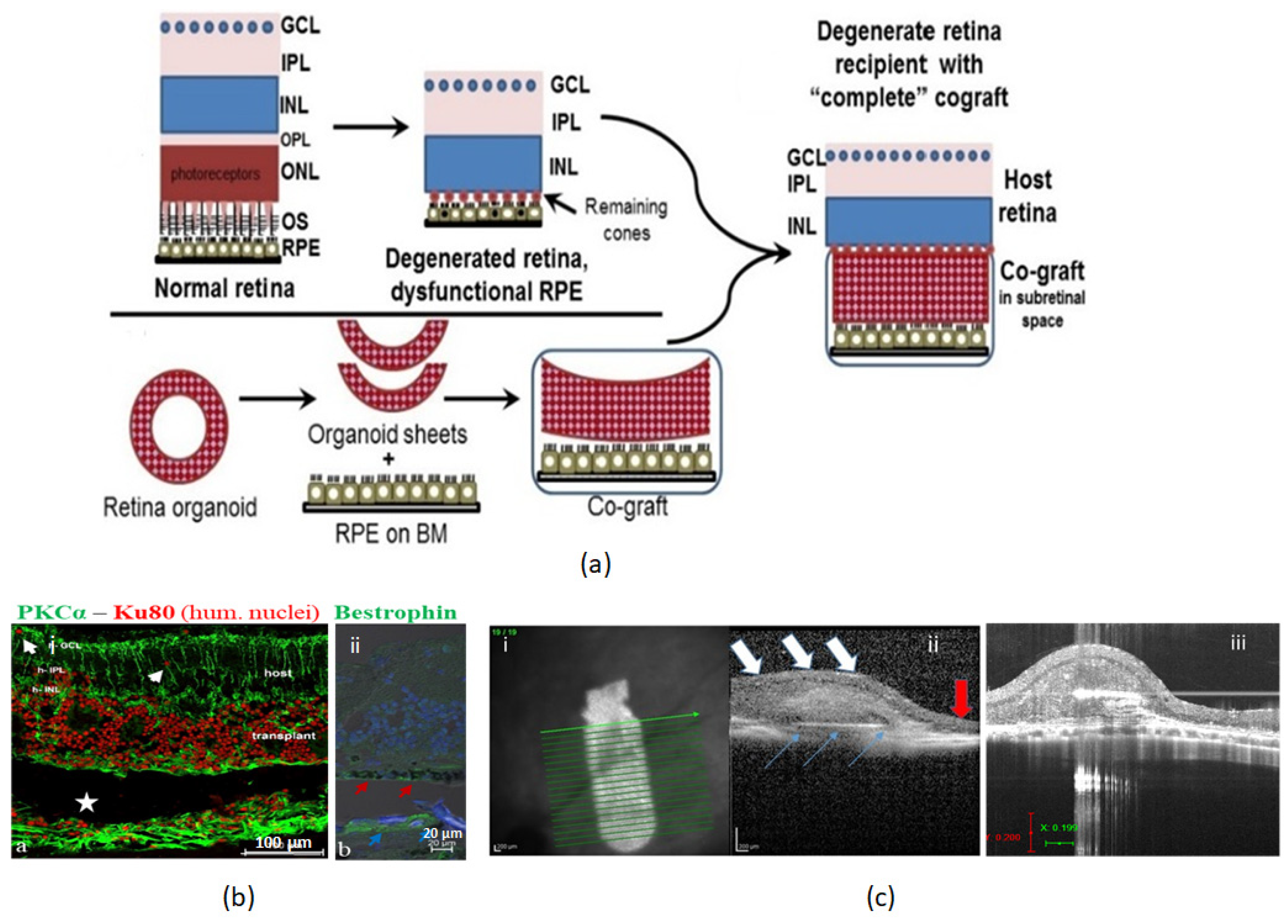
4.5. Co-Graft of RPE and Retinal Organoid
5. Other Complex Tissue Engineering Approaches
6. Current Clinical Trials Using Biomaterial Scaffolds
7. Challenges and Future Directions
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hendrickson, A. Development of Retinal Layers in Prenatal Human Retina. Am. J. Ophthalmol. 2016, 161, 29–35.e1. [Google Scholar] [CrossRef] [PubMed]
- Mannu, G.S. Retinal Phototransduction. Neurosciences 2014, 19, 275–280. [Google Scholar] [PubMed]
- Cunha-Vaz, J.; Bernardes, R.; Lobo, C. Blood-Retinal Barrier. Eur. J. Ophthalmol. 2011, 21 Suppl. 6, S3–S9. [Google Scholar] [CrossRef] [PubMed]
- Paulus, Y.M.; Sodhi, A. Anti-Angiogenic Therapy for Retinal Disease. Handb. Exp. Pharmacol. 2017, 242, 271–307. [Google Scholar] [CrossRef]
- Yue, L.; Wuyyuru, V.; Gonzalez-Calle, A.; Dorn, J.D.; Humayun, M.S. Retina-Electrode Interface Properties and Vision Restoration by Two Generations of Retinal Prostheses in One Patient-One in Each Eye. J. Neural Eng. 2020, 17, 026020. [Google Scholar] [CrossRef] [PubMed]
- Cehajic-Kapetanovic, J.; Xue, K.; Martinez-Fernandez de la Camara, C.; Nanda, A.; Davies, A.; Wood, L.J.; Salvetti, A.P.; Fischer, M.D.; Aylward, J.W.; Barnard, A.R.; et al. Initial Results from a First-in-Human Gene Therapy Trial on X-Linked Retinitis Pigmentosa Caused by Mutations in RPGR. Nat. Med. 2020, 26, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, S.D.; Tan, G.; Hosseini, H.; Nagiel, A. Subretinal Transplantation of Embryonic Stem Cell-Derived Retinal Pigment Epithelium for the Treatment of Macular Degeneration: An Assessment at 4 Years. Investig. Ophthalmol. Vis. Sci. 2016, 57, ORSFc1–ORSFc9. [Google Scholar] [CrossRef]
- Song, W.K.; Park, K.-M.; Kim, H.-J.; Lee, J.H.; Choi, J.; Chong, S.Y.; Shim, S.H.; Del Priore, L.V.; Lanza, R. Treatment of Macular Degeneration Using Embryonic Stem Cell-Derived Retinal Pigment Epithelium: Preliminary Results in Asian Patients. Stem Cell Rep. 2015, 4, 860–872. [Google Scholar] [CrossRef] [PubMed]
- Diniz, B.; Thomas, P.; Thomas, B.; Ribeiro, R.; Hu, Y.; Brant, R.; Ahuja, A.; Zhu, D.; Liu, L.; Koss, M.; et al. Subretinal Implantation of Retinal Pigment Epithelial Cells Derived from Human Embryonic Stem Cells: Improved Survival When Implanted as a Monolayer. Investig. Ophthalmol. Vis. Sci. 2013, 54, 5087–5096. [Google Scholar] [CrossRef] [PubMed]
- West, E.L.; Pearson, R.A.; MacLaren, R.E.; Sowden, J.C.; Ali, R.R. Cell Transplantation Strategies for Retinal Repair. Prog. Brain Res. 2009, 175, 3–21. [Google Scholar] [CrossRef] [PubMed]
- Hsiung, J.; Zhu, D.; Hinton, D.R. Polarized Human Embryonic Stem Cell-Derived Retinal Pigment Epithelial Cell Monolayers Have Higher Resistance to Oxidative Stress-Induced Cell Death than Nonpolarized Cultures. Stem Cells Transl. Med. 2015, 4, 10–20. [Google Scholar] [CrossRef]
- White, C.E.; Olabisi, R.M. Scaffolds for Retinal Pigment Epithelial Cell Transplantation in Age-Related Macular Degeneration. J. Tissue Eng. 2017, 8, 2041731417720841. [Google Scholar] [CrossRef]
- Kashani, A.H.; Lebkowski, J.S.; Rahhal, F.M.; Avery, R.L.; Salehi-Had, H.; Dang, W.; Lin, C.-M.; Mitra, D.; Zhu, D.; Thomas, B.B.; et al. A Bioengineered Retinal Pigment Epithelial Monolayer for Advanced, Dry Age-Related Macular Degeneration. Sci. Transl. Med. 2018, 10. [Google Scholar] [CrossRef]
- Heussen, F.M.A.; Fawzy, N.F.; Joeres, S.; Lux, A.; Maaijwee, K.; Meurs, J.C.; Kirchhof, B.; Joussen, A.M. Autologous Translocation of the Choroid and RPE in Age-Related Macular Degeneration: 1-Year Follow-up in 30 Patients and Recommendations for Patient Selection. Eye 2008, 22, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Lane, C.; Boulton, M.; Marshall, J. Transplantation of Retinal Pigment Epithelium Using a Pars Plana Approach. Eye 1989, 3 (Pt 1), 27–32. [Google Scholar] [CrossRef][Green Version]
- Li, L.X.; Turner, J.E. Inherited Retinal Dystrophy in the RCS Rat: Prevention of Photoreceptor Degeneration by Pigment Epithelial Cell Transplantation. Exp. Eye Res. 1988, 47, 911–917. [Google Scholar] [CrossRef]
- Lopez, R.; Gouras, P.; Kjeldbye, H.; Sullivan, B.; Reppucci, V.; Brittis, M.; Wapner, F.; Goluboff, E. Transplanted Retinal Pigment Epithelium Modifies the Retinal Degeneration in the RCS Rat. Investig. Ophthalmol. Vis. Sci. 1989, 30, 586–588. [Google Scholar]
- Peyman, G.A.; Blinder, K.J.; Paris, C.L.; Alturki, W.; Nelson, N.C.; Desai, U. A Technique for Retinal Pigment Epithelium Transplantation for Age-Related Macular Degeneration Secondary to Extensive Subfoveal Scarring. Ophthalmic Surg. 1991, 22, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Shah, N.; Ishii, M.; Brandon, C.; Ablonczy, Z.; Cai, J.; Liu, Y.; Chou, C.J.; Rohrer, B. Extracellular Vesicle-Mediated Long-Range Communication in Stressed Retinal Pigment Epithelial Cell Monolayers. Biochim. Biophys. Acta Mol. Basis Dis. 2018, 1864, 2610–2622. [Google Scholar] [CrossRef]
- Sheedlo, H.J.; Li, L.; Turner, J.E. Photoreceptor Cell Rescue in the RCS Rat by RPE Transplantation: A Therapeutic Approach in a Model of Inherited Retinal Dystrophy. Prog. Clin. Biol. Res. 1989, 314, 645–658. [Google Scholar]
- Sheedlo, H.J.; Li, L.X.; Turner, J.E. Functional and Structural Characteristics of Photoreceptor Cells Rescued in RPE-Cell Grafted Retinas of RCS Dystrophic Rats. Exp. Eye Res. 1989, 48, 841–854. [Google Scholar] [CrossRef]
- Cao, J.; Murat, C.; An, W.; Yao, X.; Lee, J.; Santulli-Marotto, S.; Harris, I.R.; Inana, G. Human Umbilical Tissue-Derived Cells Rescue Retinal Pigment Epithelium Dysfunction in Retinal Degeneration. STEM CELLS 2016, 34, 367–379. [Google Scholar] [CrossRef]
- Leow, S.; Mok, P.; Mh, H.N.; Ruhaslizan, R.; Halim, W.; Wong, H.; Ng, M.; Idrus, R.; Then, K.Y.; Bastion, M.-L. Subretinal Transplantation with Human Umbilical Cord-Derived Mesenchymal Stem Cells Delays Retinal Degeneration in Royal College of Surgeons Rats. Cytotherapy 2013, 15, S11. [Google Scholar] [CrossRef]
- Park, S.S.; Moisseiev, E.; Bauer, G.; Anderson, J.D.; Grant, M.B.; Zam, A.; Zawadzki, R.J.; Werner, J.S.; Nolta, J.A. Advances in Bone Marrow Stem Cell Therapy for Retinal Dysfunction. Prog. Retin. Eye Res. 2017, 56, 148–165. [Google Scholar] [CrossRef]
- Satarian, L.; Nourinia, R.; Safi, S.; Kanavi, M.R.; Jarughi, N.; Daftarian, N.; Arab, L.; Aghdami, N.; Ahmadieh, H.; Baharvand, H. Intravitreal Injection of Bone Marrow Mesenchymal Stem Cells in Patients with Advanced Retinitis Pigmentosa; a Safety Study. J. Ophthalmic Vis. Res. 2017, 12, 58–64. [Google Scholar] [CrossRef]
- Weiss, J.N.; Levy, S. Stem Cell Ophthalmology Treatment Study: Bone Marrow Derived Stem Cells in the Treatment of Retinitis Pigmentosa. Stem Cell Investig. 2018, 5, 18. [Google Scholar] [CrossRef] [PubMed]
- Oner, A.; Gonen, Z.B.; Sevim, D.G.; Smim Kahraman, N.; Unlu, M. Suprachoroidal Adipose Tissue-Derived Mesenchymal Stem Cell Implantation in Patients with Dry-Type Age-Related Macular Degeneration and Stargardt’s Macular Dystrophy: 6-Month Follow-Up Results of a Phase 2 Study. Cell Reprogram 2018, 20, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.K.; Lu, B.; Saghizadeh, M.; Wang, S. Gene Expression Changes in the Retina Following Subretinal Injection of Human Neural Progenitor Cells into a Rodent Model for Retinal Degeneration. Mol. Vis. 2016, 22, 472–490. [Google Scholar]
- Schraermeyer, U.; Thumann, G.; Luther, T.; Kociok, N.; Arnhold, S.; Kruttwig, K.; Andressen, C.; Addicks, K.; Bartz-Schmidt, K.U. Subretinally Transplanted Embryonic Stem Cells Rescue Photoreceptor Cells from Degeneration in the RCS Rats. Cell Transplant. 2001, 10, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Shin, M.C.; Tezel, T.H.; Kaplan, H.J.; Del Priore, L.V. Use of Iris Pigment Epithelium to Replace Retinal Pigment Epithelium in Age-Related Macular Degeneration: A Gene Expression Analysis. Arch. Ophthalmol. 2006, 124, 1276–1285. [Google Scholar] [CrossRef] [PubMed]
- Schraermeyer, U.; Kayatz, P.; Thumann, G.; Luther, T.T.; Szurman, P.; Kociok, N.; Bartz-Schmidt, K.U. Transplantation of Iris Pigment Epithelium into the Choroid Slows down the Degeneration of Photoreceptors in the RCS Rat. Graefe’s Arch. Clin. Exp. Ophthalmol. 2000, 238, 979–984. [Google Scholar] [CrossRef]
- Talcott, K.E.; Ratnam, K.; Sundquist, S.M.; Lucero, A.S.; Lujan, B.J.; Tao, W.; Porco, T.C.; Roorda, A.; Duncan, J.L. Longitudinal Study of Cone Photoreceptors during Retinal Degeneration and in Response to Ciliary Neurotrophic Factor Treatment. Investig. Ophthalmol. Vis. Sci. 2011, 52, 2219–2226. [Google Scholar] [CrossRef]
- Zhang, K.; Hopkins, J.J.; Heier, J.S.; Birch, D.G.; Halperin, L.S.; Albini, T.A.; Brown, D.M.; Jaffe, G.J.; Tao, W.; Williams, G.A. Ciliary Neurotrophic Factor Delivered by Encapsulated Cell Intraocular Implants for Treatment of Geographic Atrophy in Age-Related Macular Degeneration. Proc. Natl. Acad. Sci. USA 2011, 108, 6241–6245. [Google Scholar] [CrossRef]
- Idelson, M.; Alper, R.; Obolensky, A.; Ben-Shushan, E.; Hemo, I.; Yachimovich-Cohen, N.; Khaner, H.; Smith, Y.; Wiser, O.; Gropp, M.; et al. Directed Differentiation of Human Embryonic Stem Cells into Functional Retinal Pigment Epithelium Cells. Cell Stem Cell 2009, 5, 396–408. [Google Scholar] [CrossRef]
- Klimanskaya, I.; Hipp, J.; Rezai, K.A.; West, M.; Atala, A.; Lanza, R. Derivation and Comparative Assessment of Retinal Pigment Epithelium from Human Embryonic Stem Cells Using Transcriptomics. Cloning Stem Cells 2004, 6, 217–245. [Google Scholar] [CrossRef]
- Foltz, L.P.; Clegg, D.O. Rapid, Directed Differentiation of Retinal Pigment Epithelial Cells from Human Embryonic or Induced Pluripotent Stem Cells. J. Vis. Exp. 2017. [Google Scholar] [CrossRef]
- Shirai, H.; Mandai, M.; Matsushita, K.; Kuwahara, A.; Yonemura, S.; Nakano, T.; Assawachananont, J.; Kimura, T.; Saito, K.; Terasaki, H.; et al. Transplantation of Human Embryonic Stem Cell-Derived Retinal Tissue in Two Primate Models of Retinal Degeneration. Proc. Natl. Acad. Sci. USA 2016, 113, E81–E90. [Google Scholar] [CrossRef]
- Hazim, R.A.; Karumbayaram, S.; Jiang, M.; Dimashkie, A.; Lopes, V.S.; Li, D.; Burgess, B.L.; Vijayaraj, P.; Alva-Ornelas, J.A.; Zack, J.A.; et al. Differentiation of RPE Cells from Integration-Free IPS Cells and Their Cell Biological Characterization. Stem Cell Res. Ther. 2017, 8. [Google Scholar] [CrossRef]
- Buchholz, D.E.; Hikita, S.T.; Rowland, T.J.; Friedrich, A.M.; Hinman, C.R.; Johnson, L.V.; Clegg, D.O. Derivation of Functional Retinal Pigmented Epithelium from Induced Pluripotent Stem Cells. STEM CELLS 2009, 27, 2427–2434. [Google Scholar] [CrossRef]
- Chichagova, V.; Hilgen, G.; Ghareeb, A.; Georgiou, M.; Carter, M.; Sernagor, E.; Lako, M.; Armstrong, L. Human IPSC Differentiation to Retinal Organoids in Response to IGF1 and BMP4 Activation Is Line- and Method-Dependent. Stem Cells 2020, 38, 195–201. [Google Scholar] [CrossRef]
- Zerti, D.; Dorgau, B.; Felemban, M.; Ghareeb, A.E.; Yu, M.; Ding, Y.; Krasnogor, N.; Lako, M. Developing a Simple Method to Enhance the Generation of Cone and Rod Photoreceptors in Pluripotent Stem Cell-derived Retinal Organoids. Stem Cells 2020, 38, 45–51. [Google Scholar] [CrossRef]
- Gu, P.; Harwood, L.J.; Zhang, X.; Wylie, M.; Curry, W.J.; Cogliati, T. Isolation of Retinal Progenitor and Stem Cells from the Porcine Eye. Mol. Vis. 2007, 13, 1045–1057. [Google Scholar]
- Wiley, L.A.; Burnight, E.R.; Songstad, A.E.; Drack, A.V.; Mullins, R.F.; Stone, E.M.; Tucker, B.A. Patient-Specific Induced Pluripotent Stem Cells (IPSCs) for the Study and Treatment of Retinal Degenerative Diseases. Prog. Retin. Eye Res. 2015, 44, 15–35. [Google Scholar] [CrossRef]
- Wu, H.; Li, J.; Mao, X.; Li, G.; Xie, L.; You, Z. Transplantation of Rat Embryonic Stem Cell-Derived Retinal Cells Restores Visual Function in the Royal College of Surgeons Rats. Doc. Ophthalmol. 2018, 137, 71–78. [Google Scholar] [CrossRef]
- Sharma, R.; Khristov, V.; Rising, A.; Jha, B.S.; Dejene, R.; Hotaling, N.; Li, Y.; Stoddard, J.; Stankewicz, C.; Wan, Q.; et al. Clinical-Grade Stem Cell–Derived Retinal Pigment Epithelium Patch Rescues Retinal Degeneration in Rodents and Pigs. Sci. Transl. Med. 2019, 11. [Google Scholar] [CrossRef]
- Carr, A.-J.; Vugler, A.A.; Hikita, S.T.; Lawrence, J.M.; Gias, C.; Chen, L.L.; Buchholz, D.E.; Ahmado, A.; Semo, M.; Smart, M.J.K.; et al. Protective Effects of Human IPS-Derived Retinal Pigment Epithelium Cell Transplantation in the Retinal Dystrophic Rat. PLOS ONE 2009, 4, e8152. [Google Scholar] [CrossRef]
- Wang, L.; Wu, W.; Gu, Q.; Liu, Z.; Li, Q.; Li, Z.; Fang, J.; Liu, W.; Wu, J.; Zhang, Y.; et al. The Effect of Clinical-Grade Retinal Pigment Epithelium Derived from Human Embryonic Stem Cells Using Different Transplantation Strategies. Protein Cell 2019, 10, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Ikelle, L.; Al-Ubaidi, M.R.; Naash, M.I. Pluripotent Stem Cells for the Treatment of Retinal Degeneration: Current Strategies and Future Directions. Front. Cell Dev. Biol. 2020, 8. [Google Scholar] [CrossRef]
- McLelland, B.T.; Lin, B.; Mathur, A.; Aramant, R.B.; Thomas, B.B.; Nistor, G.; Keirstead, H.S.; Seiler, M.J. Transplanted HESC-Derived Retina Organoid Sheets Differentiate, Integrate, and Improve Visual Function in Retinal Degenerate Rats. Investig. Ophthalmol. Vis. Sci. 2018, 59, 2586–2603. [Google Scholar] [CrossRef]
- Westenskow, P.D.; Bucher, F.; Bravo, S.; Kurihara, T.; Feitelberg, D.; Paris, L.P.; Aguilar, E.; Lin, J.H.; Friedlander, M. IPSC-Derived Retinal Pigment Epithelium Allografts Do Not Elicit Detrimental Effects in Rats: A Follow-Up Study. Stem Cells Int. 2016, 2016. [Google Scholar] [CrossRef]
- Lin, B.; McLelland, B.T.; Aramant, R.B.; Thomas, B.B.; Nistor, G.; Keirstead, H.S.; Seiler, M.J. Retina Organoid Transplants Develop Photoreceptors and Improve Visual Function in RCS Rats With RPE Dysfunction. Investig. Ophthalmol. Vis. Sci. 2020, 61, 34. [Google Scholar] [CrossRef] [PubMed]
- Zerti, D.; Hilgen, G.; Dorgau, B.; Collin, J.; Ader, M.; Armstrong, L.; Sernagor, E.; Lako, M. Transplanted Pluripotent Stem Cell-Derived Photoreceptor Precursors Elicit Conventional and Unusual Light Responses in Mice with Advanced Retinal Degeneration. bioRxiv 2020. [Google Scholar] [CrossRef]
- Mandai, M.; Watanabe, A.; Kurimoto, Y.; Hirami, Y.; Morinaga, C.; Daimon, T.; Fujihara, M.; Akimaru, H.; Sakai, N.; Shibata, Y.; et al. Autologous Induced Stem-Cell–Derived Retinal Cells for Macular Degeneration. N. Engl. J. Med. 2017, 376, 1038–1046. [Google Scholar] [CrossRef]
- 1st Autologous IPS Cell Clinical Trial for Vision Loss in the US. Available online: https://ipscell.com/2019/12/1st-autologous-ips-cell-clinical-trial-for-vision-loss-in-the-us/ (accessed on 3 January 2021).
- Zhou, Q. Safety and Efficacy of Subretinal Transplantation of Human Embryonic Stem Cell Derived Retinal Pigment Epitheliums in Treatment of Age-Related Macular Degeneration Diseases. clinicaltrials.gov; 2018. Available online: https://clinicaltrials.gov/ct2/show/NCT03944239 (accessed on 25 February 2021).
- Cyranoski, D. Japanese Man Is First to Receive “Reprogrammed” Stem Cells from Another Person. Available online: https://www.scientificamerican.com/article/japanese-man-is-first-to-receive-reprogrammed-stem-cells-from-another-person1/ (accessed on 3 January 2021).
- Del Cerro, M.; Notter, M.F.; del Cerro, C.; Wiegand, S.J.; Grover, D.A.; Lazar, E. Intraretinal Transplantation for Rod-Cell Replacement in Light-Damaged Retinas. J. Neural Transplant. 1989, 1, 1–10. [Google Scholar] [CrossRef]
- MacLaren, R.E.; Pearson, R.; MacNeil, A.; Douglas, R.; Salt, T.; Akimoto, M.; Swaroop, A.; Sowden, J.; Ali, R. Retinal Repair by Transplantation of Photoreceptor Precursors. Nature 2006, 444, 203–207. [Google Scholar] [CrossRef]
- Bartsch, U.; Oriyakhel, W.; Kenna, P.F.; Linke, S.; Richard, G.; Petrowitz, B.; Humphries, P.; Farrar, G.J.; Ader, M. Retinal Cells Integrate into the Outer Nuclear Layer and Differentiate into Mature Photoreceptors after Subretinal Transplantation into Adult Mice. Exp. Eye Res. 2008, 86, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Pearson, R.A.; Gonzalez-Cordero, A.; West, E.L.; Ribeiro, J.R.; Aghaizu, N.; Goh, D.; Sampson, R.D.; Georgiadis, A.; Waldron, P.V.; Duran, Y.; et al. Donor and Host Photoreceptors Engage in Material Transfer Following Transplantation of Post-Mitotic Photoreceptor Precursors. Nat. Commun. 2016, 7, 13029. [Google Scholar] [CrossRef] [PubMed]
- Santos-Ferreira, T.; Llonch, S.; Borsch, O.; Postel, K.; Haas, J.; Ader, M. Retinal Transplantation of Photoreceptors Results in Donor–Host Cytoplasmic Exchange. Nat. Commun. 2016, 7. [Google Scholar] [CrossRef]
- Singh, M.; Balmer, J.; Barnard, A.R.; Aslam, S.A.; Moralli, D.; Green, C.M.; Barnea-Cramer, A.; Duncan, I.; MacLaren, R.E. Transplanted Photoreceptor Precursors Transfer Proteins to Host Photoreceptors by a Mechanism of Cytoplasmic Fusion. Nat. Commun. 2016, 7, 13537. [Google Scholar] [CrossRef]
- jCyte, Inc. A Prospective, Multicenter, Open-Label, Single-Arm Study of the Safety and Tolerability of a Single, Intravitreal Injection of Human Retinal Progenitor Cells (JCell) in Adult Subject. Available online: https://adisinsight.springer.com/trials/700252853 (accessed on 25 February 2021).
- Wahlin, K.J.; Maruotti, J.A.; Sripathi, S.R.; Ball, J.; Angueyra, J.M.; Kim, C.; Grebe, R.; Li, W.; Jones, B.W.; Zack, D.J. Photoreceptor Outer Segment-like Structures in Long-Term 3D Retinas from Human Pluripotent Stem Cells. Sci. Rep. 2017, 7, 766. [Google Scholar] [CrossRef] [PubMed]
- Fligor, C.M.; Langer, K.B.; Sridhar, A.; Ren, Y.; Shields, P.K.; Edler, M.C.; Ohlemacher, S.K.; Sluch, V.M.; Zack, D.J.; Zhang, C.; et al. Three-Dimensional Retinal Organoids Facilitate the Investigation of Retinal Ganglion Cell Development, Organization and Neurite Outgrowth from Human Pluripotent Stem Cells. Sci. Rep. 2018, 8, 14520. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.-L.; Lei, X.-L.; Han, F.; He, K.-W.; Jin, S.-Q.; Zhang, Y.-Y.; Jin, Z.-B. Patient-Specific Retinal Organoids Recapitulate Disease Features of Late-Onset Retinitis Pigmentosa. Front. Cell Dev. Biol. 2020, 8. [Google Scholar] [CrossRef]
- Akhtar, T.; Xie, H.; Khan, M.I.; Zhao, H.; Bao, J.; Zhang, M.; Xue, T. Accelerated Photoreceptor Differentiation of HiPSC-Derived Retinal Organoids by Contact Co-Culture with Retinal Pigment Epithelium. Stem Cell Res. 2019, 39, 101491. [Google Scholar] [CrossRef]
- Assawachananont, J.; Mandai, M.; Okamoto, S.; Yamada, C.; Eiraku, M.; Yonemura, S.; Sasai, Y.; Takahashi, M. Transplantation of Embryonic and Induced Pluripotent Stem Cell-Derived 3D Retinal Sheets into Retinal Degenerative Mice. Stem Cell Rep. 2014, 2, 662–674. [Google Scholar] [CrossRef]
- Gagliardi, G.; Ben M’Barek, K.; Chaffiol, A.; Slembrouck-Brec, A.; Conart, J.-B.; Nanteau, C.; Rabesandratana, O.; Sahel, J.-A.; Duebel, J.; Orieux, G.; et al. Characterization and Transplantation of CD73-Positive Photoreceptors Isolated from Human IPSC-Derived Retinal Organoids. Stem Cell Rep. 2018, 11, 665–680. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Tang, L.; Zhou, Y. Subretinal Injection: A Review on the Novel Route of Therapeutic Delivery for Vitreoretinal Diseases. ORE 2017, 58, 217–226. [Google Scholar] [CrossRef]
- Ramsden, C.M.; Powner, M.B.; Carr, A.-J.F.; Smart, M.J.K.; da Cruz, L.; Coffey, P.J. Stem Cells in Retinal Regeneration: Past, Present and Future. Development 2013, 140, 2576–2585. [Google Scholar] [CrossRef]
- McLenachan, S.; Hao, E.; Zhang, D.; Zhang, L.; Edel, M.; Chen, F. Bioengineered Bruch’s-like Extracellular Matrix Promotes Retinal Pigment Epithelial Differentiation. Biochem. Biophys. Rep. 2017, 10, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Karwatowski, W.S.; Jeffries, T.E.; Duance, V.C.; Albon, J.; Bailey, A.J.; Easty, D.L. Preparation of Bruch’s Membrane and Analysis of the Age-Related Changes in the Structural Collagens. Br. J. Ophthalmol. 1995, 79, 944–952. [Google Scholar] [CrossRef]
- Del Priore, L.V.; Geng, L.; Tezel, T.H.; Kaplan, H.J. Extracellular Matrix Ligands Promote RPE Attachment to Inner Bruch’s Membrane. Curr. Eye Res. 2002, 25, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Rehman, I.; Mahabadi, N.; Motlagh, M.; Ali, T. Anatomy, Head and Neck, Eye Fovea. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Hotaling, N.A.; Khristov, V.; Wan, Q.; Sharma, R.; Jha, B.S.; Lotfi, M.; Maminishkis, A.; Simon, C.G.; Bharti, K. Nanofiber Scaffold-Based Tissue-Engineered Retinal Pigment Epithelium to Treat Degenerative Eye Diseases. J. Ocul. Pharmacol. Ther. 2016, 32, 272–285. [Google Scholar] [CrossRef] [PubMed]
- Booij, J.C.; Baas, D.C.; Beisekeeva, J.; Gorgels, T.G.M.F.; Bergen, A.A.B. The Dynamic Nature of Bruch’s Membrane. Prog. Retin. Eye Res. 2010, 29, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Thumann, G.; Viethen, A.; Gaebler, A.; Walter, P.; Kaempf, S.; Johnen, S.; Salz, A.K. The in Vitro and in Vivo Behaviour of Retinal Pigment Epithelial Cells Cultured on Ultrathin Collagen Membranes. Biomaterials 2009, 30, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.T.; Lee, C.J.; Bent, S.F.; Fishman, H.A.; Sabelman, E.E. Thin Collagen Film Scaffolds for Retinal Epithelial Cell Culture. Biomaterials 2007, 28, 1486–1494. [Google Scholar] [CrossRef]
- Bhatt, N.S.; Newsome, D.A.; Fenech, T.; Hessburg, T.P.; Diamond, J.G.; Miceli, M.V.; Kratz, K.E.; Oliver, P.D. Experimental Transplantation of Human Retinal Pigment Epithelial Cells on Collagen Substrates. Am. J. Ophthalmol. 1994, 117, 214–221. [Google Scholar] [CrossRef]
- Thumann, G.; Hueber, A.; Dinslage, S.; Schaefer, F.; Yasukawa, T.; Kirchhof, B.; Yafai, Y.; Eichler, W.; Bringmann, A.; Wiedemann, P. Characteristics of Iris and Retinal Pigment Epithelial Cells Cultured on Collagen Type I Membranes. Curr. Eye Res. 2006, 31, 241–249. [Google Scholar] [CrossRef]
- Hsiue, G.-H.; Lai, J.-Y.; Lin, P.-K. Absorbable Sandwich-like Membrane for Retinal-Sheet Transplantation. J. Biomed. Mater. Res. 2002, 61, 19–25. [Google Scholar] [CrossRef]
- Lai, J.-Y.; Li, Y.-T. Evaluation of Cross-Linked Gelatin Membranes as Delivery Carriers for Retinal Sheets. Mater. Sci. and Engi. C 2010, 30, 677–685. [Google Scholar] [CrossRef]
- Jeong, S.M.; Kim, E.Y.; Hwang, J.H.; Lee, G.Y.; Cho, S.J.; Bae, J.Y.; Song, J.E.; Yoon, K.H.; Joo, C.-K.; Lee, D.; et al. A Study on Proliferation and Behavior of Retinal Pigment Epithelial Cells on Purified Alginate Films. Int. J. Stem Cells 2011, 4, 105–112. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Heidari, R.; Soheili, Z.-S.; Samiei, S.; Ahmadieh, H.; Davari, M.; Nazemroaya, F.; Bagheri, A.; Deezagi, A. Alginate as a Cell Culture Substrate for Growth and Differentiation of Human Retinal Pigment Epithelial Cells. Appl. Biochem. Biotechnol. 2015, 175, 2399–2412. [Google Scholar] [CrossRef] [PubMed]
- Hunt, N.C.; Hallam, D.; Karimi, A.; Mellough, C.B.; Chen, J.; Steel, D.H.W.; Lako, M. 3D Culture of Human Pluripotent Stem Cells in RGD-Alginate Hydrogel Improves Retinal Tissue Development. Acta Biomater. 2017, 49, 329–343. [Google Scholar] [CrossRef] [PubMed]
- Tran, S.H.; Wilson, C.G.; Seib, F.P. A Review of the Emerging Role of Silk for the Treatment of the Eye. Pharm. Res. 2018, 35. [Google Scholar] [CrossRef]
- Shadforth, A.M.A.; George, K.A.; Kwan, A.S.; Chirila, T.V.; Harkin, D.G. The Cultivation of Human Retinal Pigment Epithelial Cells on Bombyx Mori Silk Fibroin. Biomaterials 2012, 33, 4110–4117. [Google Scholar] [CrossRef]
- Kundu, J.; Michaelson, A.; Talbot, K.; Baranov, P.; Young, M.; Carrier, R. Decellularized Retinal Matrix: Natural Platforms for Human Retinal Progenitor Cell Culture. Acta Biomater. 2015, 31. [Google Scholar] [CrossRef] [PubMed]
- Lassota, N.; Kiilgaard, J.F.; Prause, J.U.; la Cour, M. Correlation between Clinical and Histological Features in a Pig Model of Choroidal Neovascularization. Graefe’s Arch. Clin. Exp. Ophthalmol. 2005, 244, 394–398. [Google Scholar] [CrossRef]
- Pan, Z.; Ding, J. Poly(Lactide- Co -Glycolide) Porous Scaffolds for Tissue Engineering and Regenerative Medicine. Interface Focus. 2012, 2, 366–377. [Google Scholar] [CrossRef] [PubMed]
- Sabir, M.I.; Xu, X.; Li, L. A Review on Biodegradable Polymeric Materials for Bone Tissue Engineering Applications. J. Mater. Sci. 2009, 44, 5713–5724. [Google Scholar] [CrossRef]
- Bernards, D.A.; Bhisitkul, R.B.; Wynn, P.; Steedman, M.R.; Lee, O.-T.; Wong, F.; Thoongsuwan, S.; Desai, T.A. Ocular Biocompatibility and Structural Integrity of Micro- and Nanostructured Poly(Caprolactone) Films. J. Ocul. Pharmacol. Ther. 2013, 29, 249–257. [Google Scholar] [CrossRef]
- Sorkio, A.; Haimi, S.; Verdoold, V.; Juuti-Uusitalo, K.; Grijpma, D.; Skottman, H. Poly(Trimethylene Carbonate) as an Elastic Biodegradable Film for Human Embryonic Stem Cell-Derived Retinal Pigment Epithelial Cells. J. Tissue Eng. Regen. Med. 2017, 11, 3134–3144. [Google Scholar] [CrossRef]
- Tao, S.; Young, C.; Redenti, S.; Zhang, Y.; Klassen, H.; Desai, T.; Young, M.J. Survival, Migration and Differentiation of Retinal Progenitor Cells Transplanted on Micro-Machined Poly(Methyl Methacrylate) Scaffolds to the Subretinal Space. Lab Chip 2007, 7, 695–701. [Google Scholar] [CrossRef]
- Redenti, S.; Tao, S.; Yang, J.; Gu, P.; Klassen, H.; Saigal, S.; Desai, T.; Young, M.J. Retinal Tissue Engineering Using Mouse Retinal Progenitor Cells and a Novel Biodegradable, Thin-Film Poly(e-Caprolactone) Nanowire Scaffold. J. Ocul. Biol. Dis. Infor. 2008, 1, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Neeley, W.L.; Redenti, S.; Klassen, H.; Tao, S.; Desai, T.; Young, M.J.; Langer, R. A Microfabricated Scaffold for Retinal Progenitor Cell Grafting. Biomaterials 2008, 29, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Lavik, E.B.; Klassen, H.; Warfvinge, K.; Langer, R.; Young, M.J. Fabrication of Degradable Polymer Scaffolds to Direct the Integration and Differentiation of Retinal Progenitors. Biomaterials 2005, 26, 3187–3196. [Google Scholar] [CrossRef]
- Ho, A.C.; Humayun, M.S.; Dorn, J.D.; da Cruz, L.; Dagnelie, G.; Handa, J.; Barale, P.-O.; Sahel, J.-A.; Stanga, P.E.; Hafezi, F.; et al. Long-Term Results from an Epiretinal Prosthesis to Restore Sight to the Blind. Ophthalmology 2015, 122, 1547–1554. [Google Scholar] [CrossRef]
- Thomas, B.B.; Zhu, D.; Zhang, L.; Thomas, P.B.; Hu, Y.; Nazari, H.; Stefanini, F.; Falabella, P.; Clegg, D.O.; Hinton, D.R.; et al. Survival and Functionality of HESC-Derived Retinal Pigment Epithelium Cells Cultured as a Monolayer on Polymer Substrates Transplanted in RCS Rats. Investig. Ophthalmol. Vis. Sci. 2016, 57, 2877–2887. [Google Scholar] [CrossRef]
- Kashani, A.H.; Uang, J.; Mert, M.; Rahhal, F.; Chan, C.; Avery, R.L.; Dugel, P.; Chen, S.; Lebkowski, J.; Clegg, D.O.; et al. Surgical Method for Implantation of a Biosynthetic Retinal Pigment Epithelium Monolayer for Geographic Atrophy: Experience from a Phase 1/2a Study. Oph. Retina 2020, 4, 264–273. [Google Scholar] [CrossRef] [PubMed]
- Lu, B.; Zhu, D.; Hinton, D.; Humayun, M.S.; Tai, Y.-C. Mesh-Supported Submicron Parylene-C Membranes for Culturing Retinal Pigment Epithelial Cells. Biomed. Microdevices 2012, 14, 659–667. [Google Scholar] [CrossRef]
- Koss, M.J.; Falabella, P.; Stefanini, F.R.; Pfister, M.; Thomas, B.B.; Kashani, A.H.; Brant, R.; Zhu, D.; Clegg, D.O.; Hinton, D.R.; et al. Subretinal Implantation of a Monolayer of Human Embryonic Stem Cell-Derived Retinal Pigment Epithelium: A Feasibility and Safety Study in Yucatán Minipigs. Graefe’s Arch. Clin. Exp. Ophthalmol. 2016, 254, 1553–1565. [Google Scholar] [CrossRef] [PubMed]
- Calejo, M.T.; Ilmarinen, T.; Jongprasitkul, H.; Skottman, H.; Kellomäki, M. Honeycomb Porous Films as Permeable Scaffold Materials for Human Embryonic Stem Cell-Derived Retinal Pigment Epithelium. J. Biomed. Mater. Res. A 2016, 104, 1646–1656. [Google Scholar] [CrossRef]
- Peng, C.-H.; Chuang, J.-H.; Wang, M.-L.; Jhan, Y.-Y.; Chien, K.-H.; Chung, Y.-C.; Hung, K.-H.; Chang, C.-C.; Lee, C.-K.; Tseng, W.-L.; et al. Laminin Modification Subretinal Bio-Scaffold Remodels Retinal Pigment Epithelium-Driven Microenvironment in Vitro and in Vivo. Oncotarget 2016, 7, 64631–64648. [Google Scholar] [CrossRef] [PubMed]
- Rahmani, S.; Tabandeh, F.; Faghihi, S.; Amoabediny, G.; Shakibaie, M.; Noorani, B.; Yazdian, F. Fabrication and Characterization of Poly(ε-Caprolactone)/Gelatin Nanofibrous Scaffolds for Retinal Tissue Engineering. Int. J. Polym. Mater. Polym. Biomater. 2018, 67, 27–35. [Google Scholar] [CrossRef]
- Warnke, P.H.; Alamein, M.; Skabo, S.; Stephens, S.; Bourke, R.; Heiner, P.; Liu, Q. Primordium of an Artificial Bruch’s Membrane Made of Nanofibers for Engineering of Retinal Pigment Epithelium Cell Monolayers. Acta Biomater. 2013, 9, 9414–9422. [Google Scholar] [CrossRef] [PubMed]
- White, C.; DiStefano, T.; Olabisi, R. The Influence of Substrate Modulus on Retinal Pigment Epithelial Cells. J. Biomed. Mater. Res. A 2017, 105, 1260–1266. [Google Scholar] [CrossRef] [PubMed]
- Baranov, P.; Regatieri, C.; Melo, G.; Clissold, H.; Young, M. Synthetic Peptide-Acrylate Surface for Self-Renewal of Human Retinal Progenitor Cells. Tissue Eng. Part C Methods 2013, 19, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.; Yao, H.; Weber, J.L.; Melkoumian, Z.K.; Ye, K. A Synthetic, Xeno-Free Peptide Surface for Expansion and Directed Differentiation of Human Induced Pluripotent Stem Cells. PLOS ONE 2012, 7, e50880. [Google Scholar] [CrossRef] [PubMed]
- Thomson, H.A.J.; Treharne, A.J.; Walker, P.; Grossel, M.C.; Lotery, A.J. Optimisation of Polymer Scaffolds for Retinal Pigment Epithelium (RPE) Cell Transplantation. Br. J. Ophthalmol. 2011, 95, 563–568. [Google Scholar] [CrossRef]
- Zhang, D.; Ni, N.; Chen, J.; Yao, Q.; Shen, B.; Zhang, Y.; Zhu, M.; Wang, Z.; Ruan, J.; Wang, J.; et al. Electrospun SF/PLCL Nanofibrous Membrane: A Potential Scaffold for Retinal Progenitor Cell Proliferation and Differentiation. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef]
- Chen, H.; Fan, X.; Xia, J.; Chen, P.; Zhou, X.; Huang, J.; Yu, J.; Gu, P. Electrospun Chitosan-Graft-Poly (ɛ-Caprolactone)/Poly (ɛ-Caprolactone) Nanofibrous Scaffolds for Retinal Tissue Engineering. Int. J. Nanomed. 2011, 6, 453–461. [Google Scholar] [CrossRef]
- Steedman, M.R.; Tao, S.L.; Klassen, H.; Desai, T.A. Enhanced Differentiation of Retinal Progenitor Cells Using Microfabricated Topographical Cues. Biomed. Microdevices 2010, 12, 363–369. [Google Scholar] [CrossRef]
- Mokhtarinia, K.; Nourbakhsh, M.S.; Masaeli, E.; Entezam, M.; Karamali, F.; Nasr-Esfahani, M.H. Switchable Phase Transition Behavior of Thermoresponsive Substrates for Cell Sheet Engineering. J. Polym. Sci. Part B Polym. Phys. 2018, 56, 1567–1576. [Google Scholar] [CrossRef]
- Kushida, A.; Yamato, M.; Konno, C.; Kikuchi, A.; Sakurai, Y.; Okano, T. Decrease in Culture Temperature Releases Monolayer Endothelial Cell Sheets Together with Deposited Fibronectin Matrix from Temperature-Responsive Culture Surfaces. J. Biomed. Mater. Res. 1999, 45, 355–362. [Google Scholar] [CrossRef]
- Kubota, A.; Nishida, K.; Yamato, M.; Yang, J.; Kikuchi, A.; Okano, T.; Tano, Y. Transplantable Retinal Pigment Epithelial Cell Sheets for Tissue Engineering. Biomaterials 2006, 27, 3639–3644. [Google Scholar] [CrossRef]
- Haraguchi, Y.; Shimizu, T.; Yamato, M.; Okano, T. Scaffold-Free Tissue Engineering Using Cell Sheet Technology. RSC Adv. 2012, 2, 2184–2190. [Google Scholar] [CrossRef]
- Singh, R.K.; Nasonkin, I.O. Limitations and Promise of Retinal Tissue From Human Pluripotent Stem Cells for Developing Therapies of Blindness. Front. Cell. Neurosci. 2020, 14. [Google Scholar] [CrossRef] [PubMed]
- Tan, E.Y.S.; Sing, S.L.; Yeong, W.Y. 60 - Scaffolds for retinal repairs. In Handbook of Tissue Engineering Scaffolds: Volume Two; Mozafari, M., Sefat, F., Atala, A., Eds.; Woodhead Publishing Series in Biomaterials; Woodhead Publishing: Cambridge, UK, 2019; pp. 673–691. ISBN 978-0-08-102561-1. [Google Scholar]
- Bhuthalingam, R.; Lim, P.Q.; Irvine, S.A.; Agrawal, A.; Mhaisalkar, P.S.; An, J.; Chua, C.K.; Venkatraman, S. A Novel 3D Printing Method for Cell Alignment and Differentiation. Int. J. Bioprinting 2015, 1. [Google Scholar] [CrossRef]
- Shi, P.; Edgar, T.; Yeong, W.Y.; Laude, A. Three-Dimensional (3D) Bioprinting of Retina Equivalent for Ocular Research. Int. J. Bioprinting 2017, 3. [Google Scholar] [CrossRef] [PubMed]
- Masaeli, E.; Forster, V.; Picaud, S.; Karamali, F.; Nasr-Esfahani, M.H.; Marquette, C. Tissue Engineering of Retina through High Resolution 3-Dimensional Inkjet Bioprinting. Biofabrication 2020, 12, 025006. [Google Scholar] [CrossRef]
- da Cruz, L.; Fynes, K.; Georgiadis, O.; Kerby, J.; Luo, Y.H.; Ahmado, A.; Vernon, A.; Daniels, J.T.; Nommiste, B.; Hasan, S.M.; et al. Phase 1 Clinical Study of an Embryonic Stem Cell-Derived Retinal Pigment Epithelium Patch in Age-Related Macular Degeneration. Nat. Biotechnol. 2018, 36, 328–337. [Google Scholar] [CrossRef]
- Fernandes, R.A.B.; Stefanini, F.R.; Falabella, P.; Koss, M.J.; Wells, T.; Diniz, B.; Ribeiro, R.; Schor, P.; Maia, M.; Penha, F.M.; et al. Development of a New Tissue Injector for Subretinal Transplantation of Human Embryonic Stem Cell Derived Retinal Pigmented Epithelium. Int. J. Retin. Vitr. 2017, 3, 41. [Google Scholar] [CrossRef]
- Stanzel, B.V.; Liu, Z.; Brinken, R.; Braun, N.; Holz, F.G.; Eter, N. Subretinal Delivery of Ultrathin Rigid-Elastic Cell Carriers Using a Metallic Shooter Instrument and Biodegradable Hydrogel Encapsulation. Investig. Ophthalmol. Vis. Sci. 2012, 53, 490–500. [Google Scholar] [CrossRef] [PubMed]
- Thackaberry, E.A.; Farman, C.; Zhong, F.; Lorget, F.; Staflin, K.; Cercillieux, A.; Miller, P.E.; Schuetz, C.; Chang, D.; Famili, A.; et al. Evaluation of the Toxicity of Intravitreally Injected PLGA Microspheres and Rods in Monkeys and Rabbits: Effects of Depot Size on Inflammatory Response. Investig. Ophthalmol. Vis. Sci. 2017, 58, 4274–4285. [Google Scholar] [CrossRef] [PubMed]
- Xian, B.; Huang, B. The Immune Response of Stem Cells in Subretinal Transplantation. Stem Cell Res. Ther. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
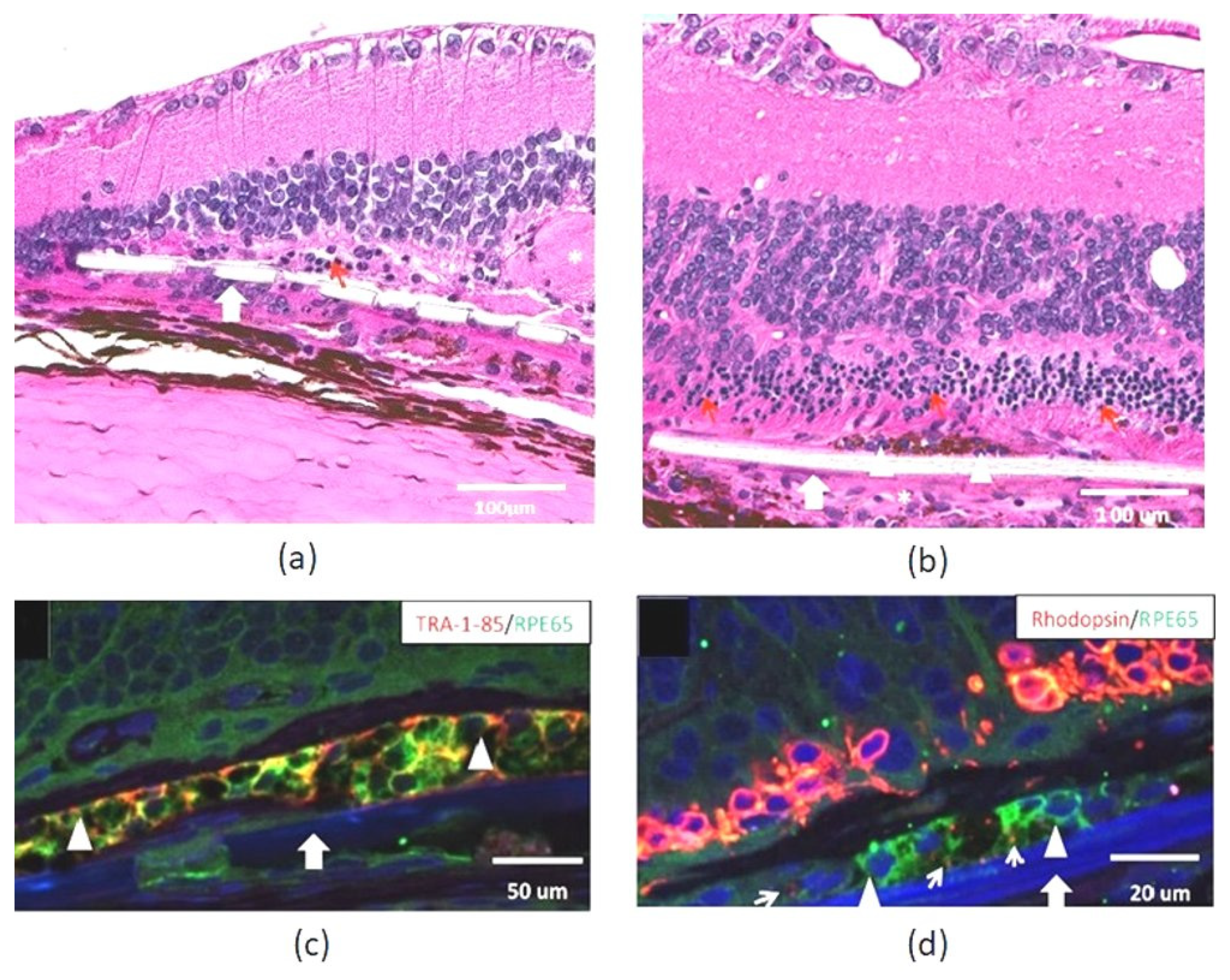

| Biomaterial | Thickness (μm) | Advantages | Studies | References |
|---|---|---|---|---|
| Collagen type I membrane | 7 | Non-toxic, no inflammatory response, controllable, stability (10 weeks), degrade (within 24 weeks) | Long term biocompatibility and membrane degradation evaluated (rabbits) | (Bhatt et al., 1994; Booij et al., 2010; Lu et al., 2007; Thumann et al., 2009) |
| Gelatin | 30–35 | Lower immunogenicity, crosslinking ability, and better solubility in aqueous systems | Biocompatibility, improved survival, and formation of laminar structures (rabbits) | (Hsiue et al., 2002; Lai and Li, 2010) |
| Alginate | Thin film | Purified alginate- high cell proliferative rate | Ability to support the growth of RPE cells and their high proliferative rates (in vitro) | (Heidari et al., 2015; Hunt et al., 2017; Jeong et al., 2011) |
| Silk Fibroin | 3 | Great mechanical strength, good biodegradability, and biocompatibility | Evaluate BMSF as a substrate for RPE cell transplantation (in vitro) | (Shadforth et al., 2012; Tran et al., 2018) |
| PLGA | Remarkable mechanical properties, adjustable degradation rates (80-90 days), and good processability | To demonstrate safety and cell integration in the eye (rodent and porcine preclinical models) | (Pan and Ding, 2012; Sharma et al., 2019) | |
| PCL | 20–40 | Thinnest scaffold, permeable, slow degradation, adverse tissue responses not observed | Assess the tolerance and durability of micro and nanostructured PCL thin films (rabbits) | (Bernards et al., 2013; Redenti et al., 2008) |
| PTMC | 100 | Elastomeric properties similar to BM, thickness tunable | Demonstrate adherence and maturation of hESC-RPE cells on PTMC compared to PDLLA films | (Sorkio et al., 2017) |
| PMMA | 6 | Reduced risk of trauma | Evaluate adhesion of RPCs and its differentiation and migration to host retina (mice) | (Tao et al., 2007) |
| PGS | 45 | A suitable candidate for RPC delivery with great novel properties | Evaluate mechanical properties | (Neeley et al., 2008) |
| Parylene-C | 0.15–0.30, 0.3 μm thickness supported on a 6.0 μm thick mesh frame | Macromolecules and nutrients can diffuse, nonimmunogenic, Promotes cell adhesion after vitronectin/matrigel coating | Evaluate safety, survival, and functionality of hESC-RPE cells on parylene in animal models | (Kashani et al., 2018; Koss et al., 2016; Lu et al., 2012; Thomas et al., 2016) |
| 0.3 μm thickness supported on a 6.0 μm thick mesh frame | Assess safety and efficacy of hESC-RPE on parylene in patients with AMD. (clinical study) | |||
| Check cell adherence and proliferation (in vitro) | ||||
| PLLA & PLGA | Week 1: 133.1 Week 2: 131.5 Week 3: 103.5 | 25:75 (PLLA: PLGA) thinnest, most porous, and minimal cell death | Evaluate the variety of suitable scaffolds for RPE transplantation (in vitro) | (Thomson et al., 2011) |
| SF & PLCL | 60–100 | Quick RPC proliferation, preferential differentiation towards retinal neurons like photoreceptors | Understand effects of blended nanofibrous membranes of silk fibroin and PLCL (in vitro) | (Zhang et al., 2015) |
| Honeycomb like films and collagen IV | Increased hydrophilicity, high permeability | Investigate honeycomb-like film as a promising scaffold for hESC-RPE tissue engineering | (Calejo et al., 2016) | |
| PNIPAAm – Thermoresponsive polymer | scalable | Allows cell sheet harvest by temperature reduction from 37–20 °C | Demonstrate fabrication of transplantable retinal pigment epithelium cell sheets | (Kubota et al., 2006; Kushida et al., 1999) |
| Decellularized matrix | 10–20 | micro- and macro-scale structural components and functional ECM proteins present Photoreceptor differentiation | Develop novel biomaterial by decellularizing retina using ionic detergents | (Kundu et al., 2016) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rajendran Nair, D.S.; Seiler, M.J.; Patel, K.H.; Thomas, V.; Martinez Camarillo, J.C.; Humayun, M.S.; Thomas, B.B. Tissue Engineering Strategies for Retina Regeneration. Appl. Sci. 2021, 11, 2154. https://doi.org/10.3390/app11052154
Rajendran Nair DS, Seiler MJ, Patel KH, Thomas V, Martinez Camarillo JC, Humayun MS, Thomas BB. Tissue Engineering Strategies for Retina Regeneration. Applied Sciences. 2021; 11(5):2154. https://doi.org/10.3390/app11052154
Chicago/Turabian StyleRajendran Nair, Deepthi S., Magdalene J. Seiler, Kahini H. Patel, Vinoy Thomas, Juan Carlos Martinez Camarillo, Mark S. Humayun, and Biju B. Thomas. 2021. "Tissue Engineering Strategies for Retina Regeneration" Applied Sciences 11, no. 5: 2154. https://doi.org/10.3390/app11052154
APA StyleRajendran Nair, D. S., Seiler, M. J., Patel, K. H., Thomas, V., Martinez Camarillo, J. C., Humayun, M. S., & Thomas, B. B. (2021). Tissue Engineering Strategies for Retina Regeneration. Applied Sciences, 11(5), 2154. https://doi.org/10.3390/app11052154







