Performances Assessment of Tricalcium Aluminate as an Innovative Material for Thermal Energy Storage Applications
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
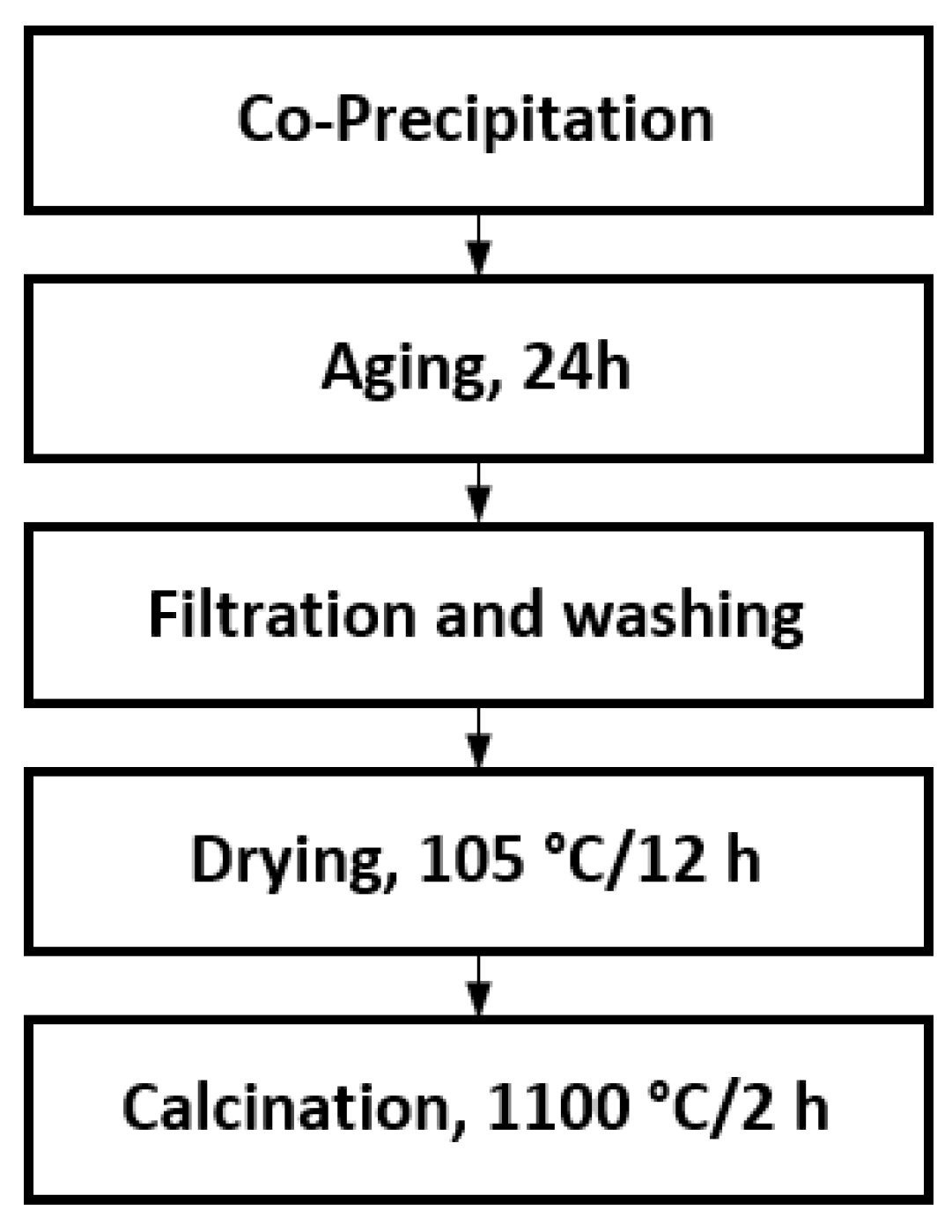
2.1. Samples Preparation
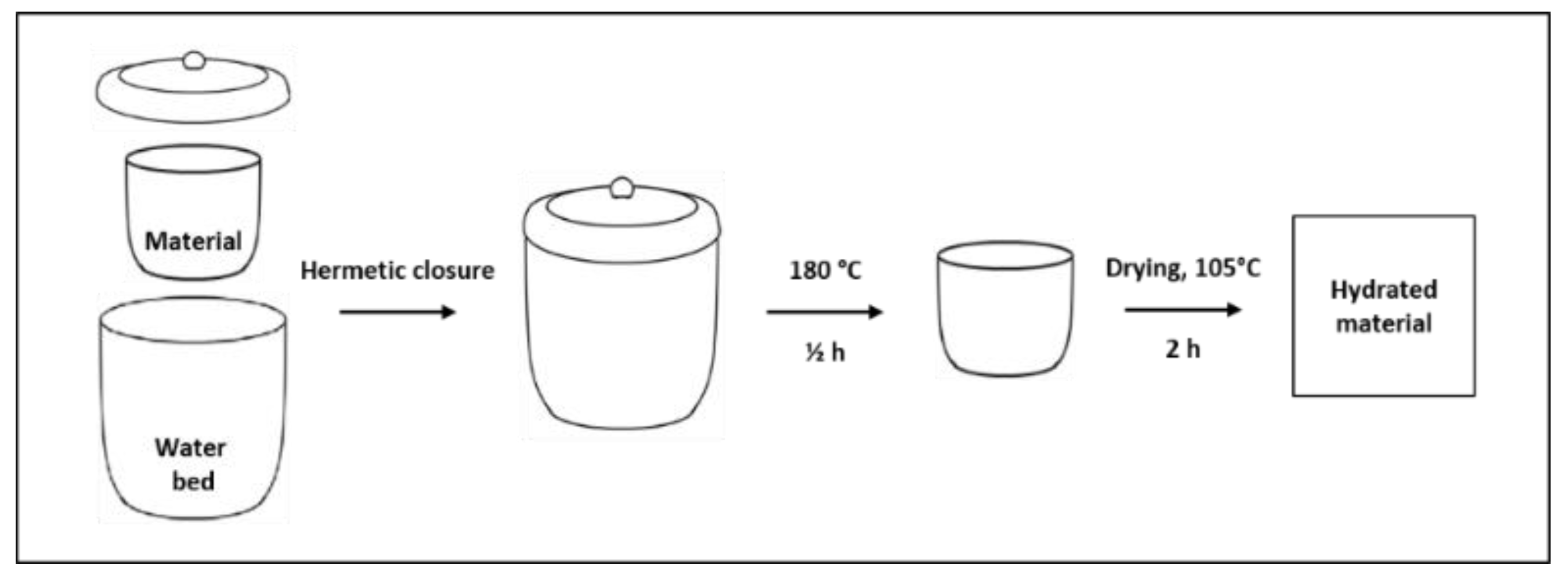
2.2. Samples Hydration
2.3. Characterization
2.3.1. Structural and Morphological Characterization
2.3.2. Differential Scanning Calorimetry (DSC)
2.3.3. Thermogravimetric Analysis (TGA)
3. Results and Discussion
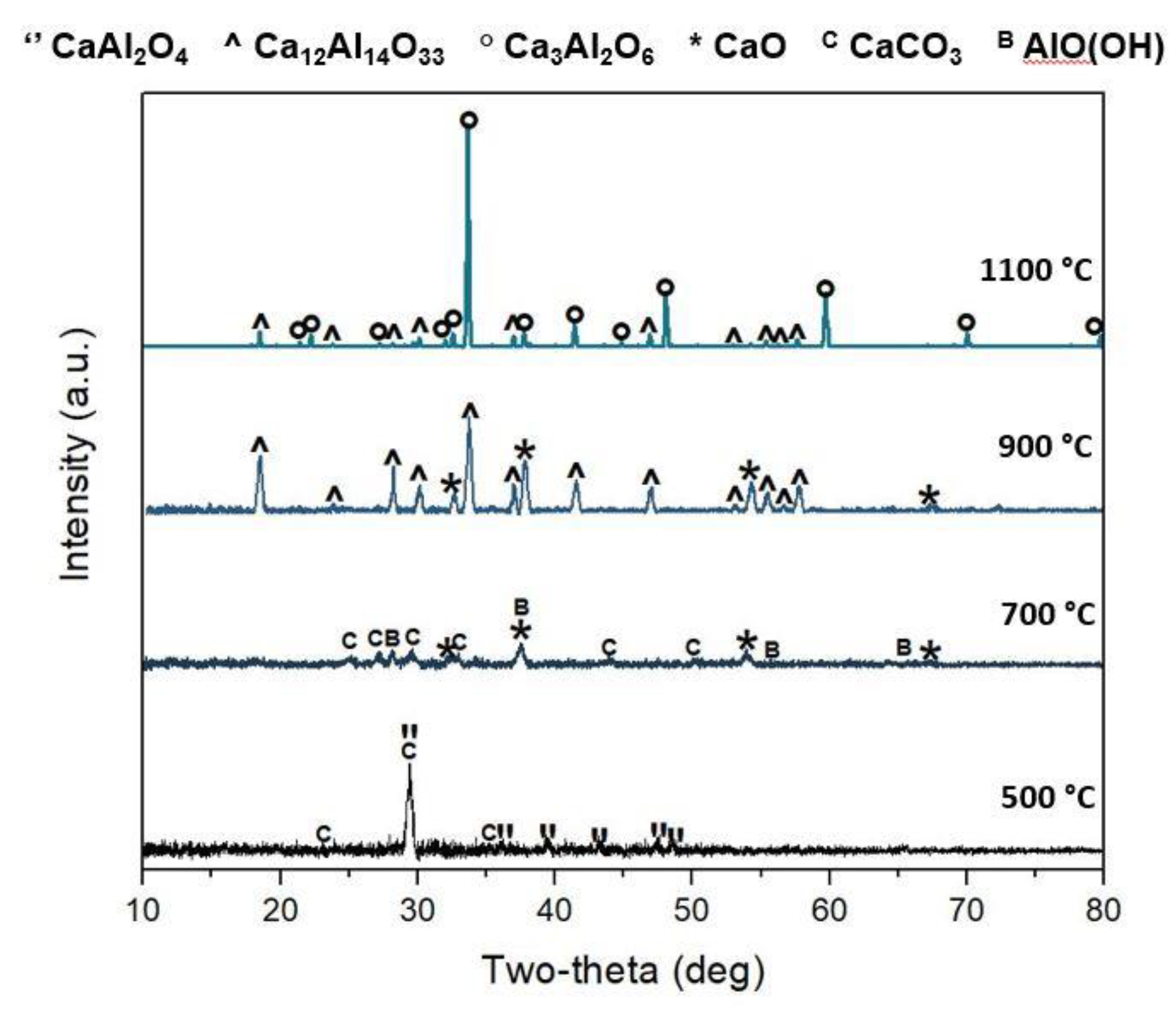
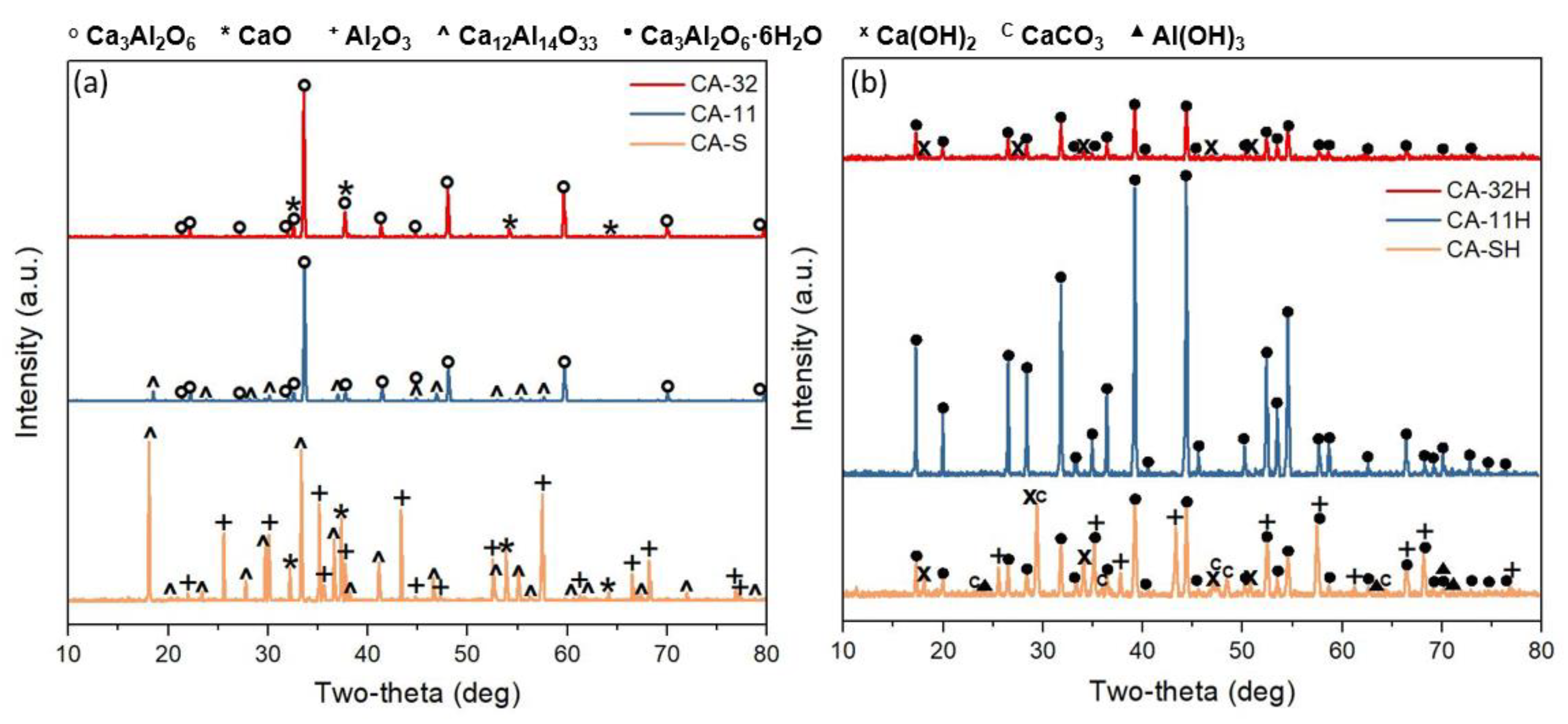
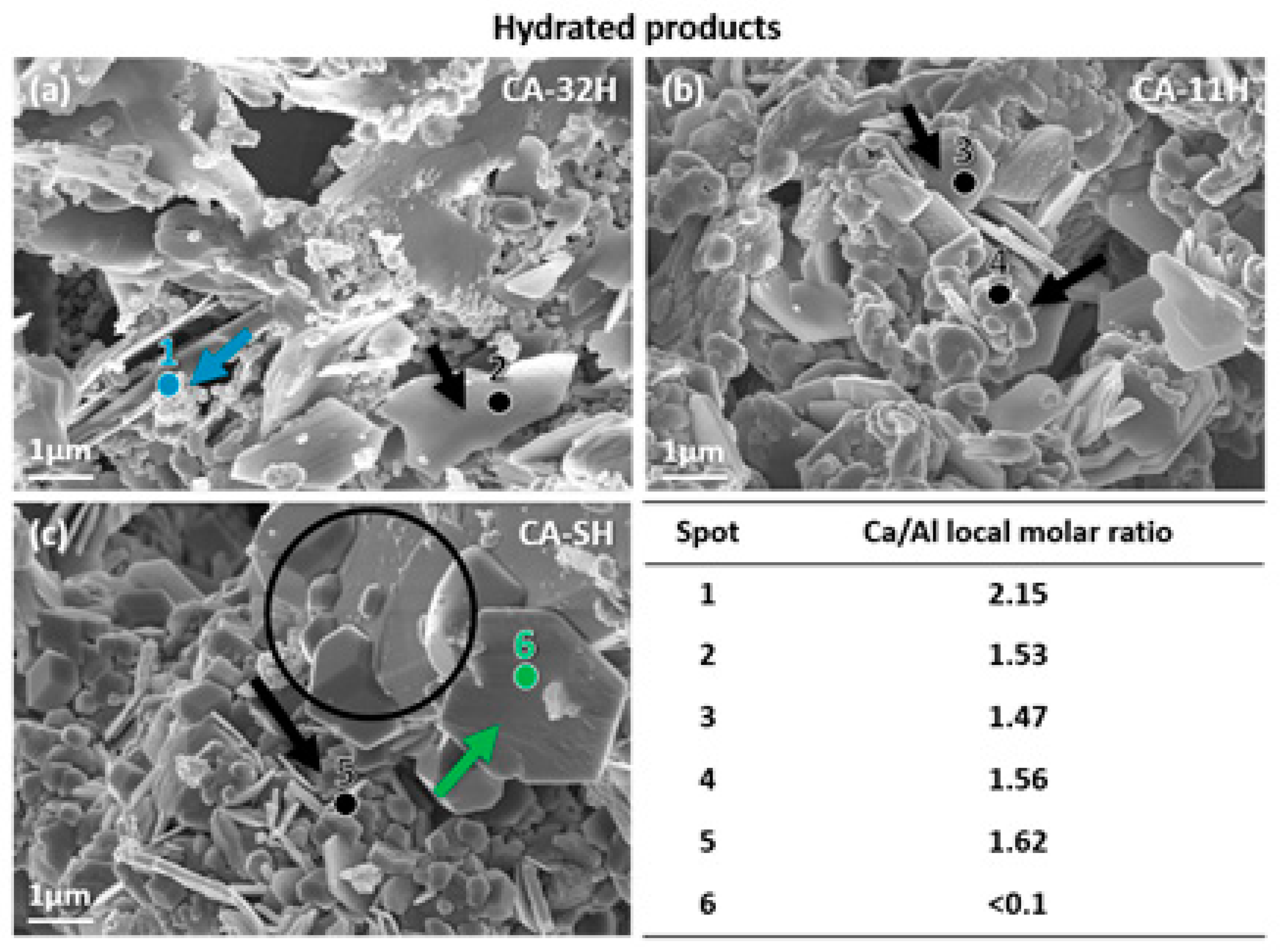
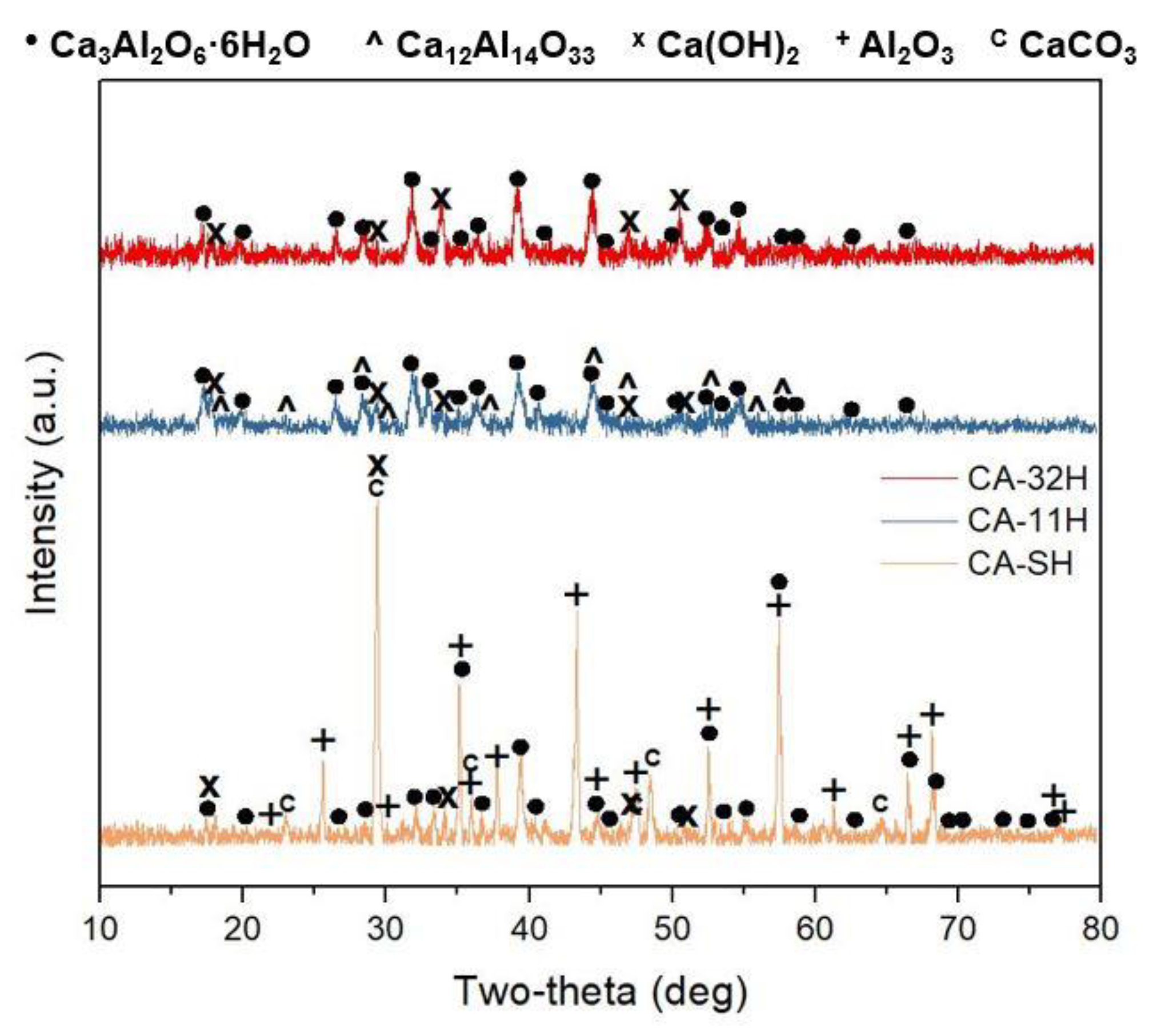
3.1. Ca3Al2O6 Synthesis in Anhydrous and Hydrated Forms
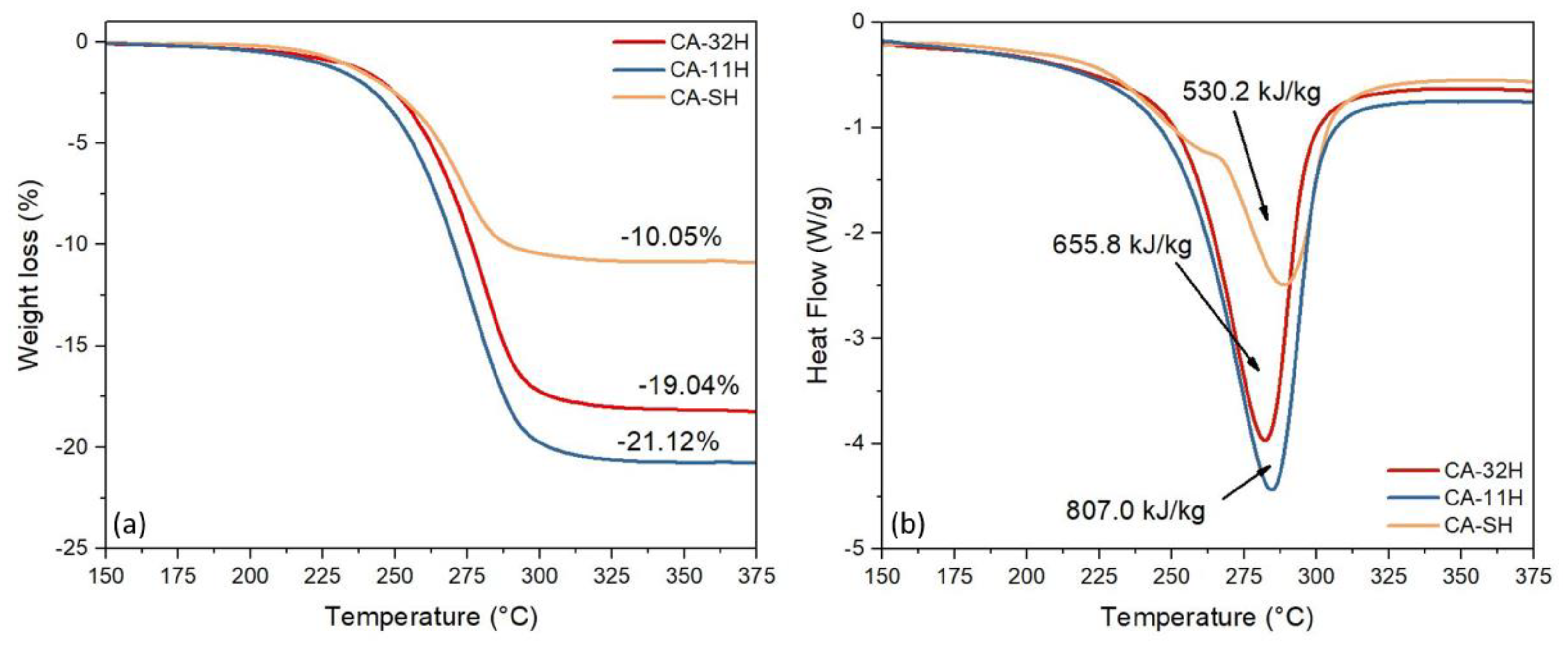
3.2. Dehydration and Heat of Reaction
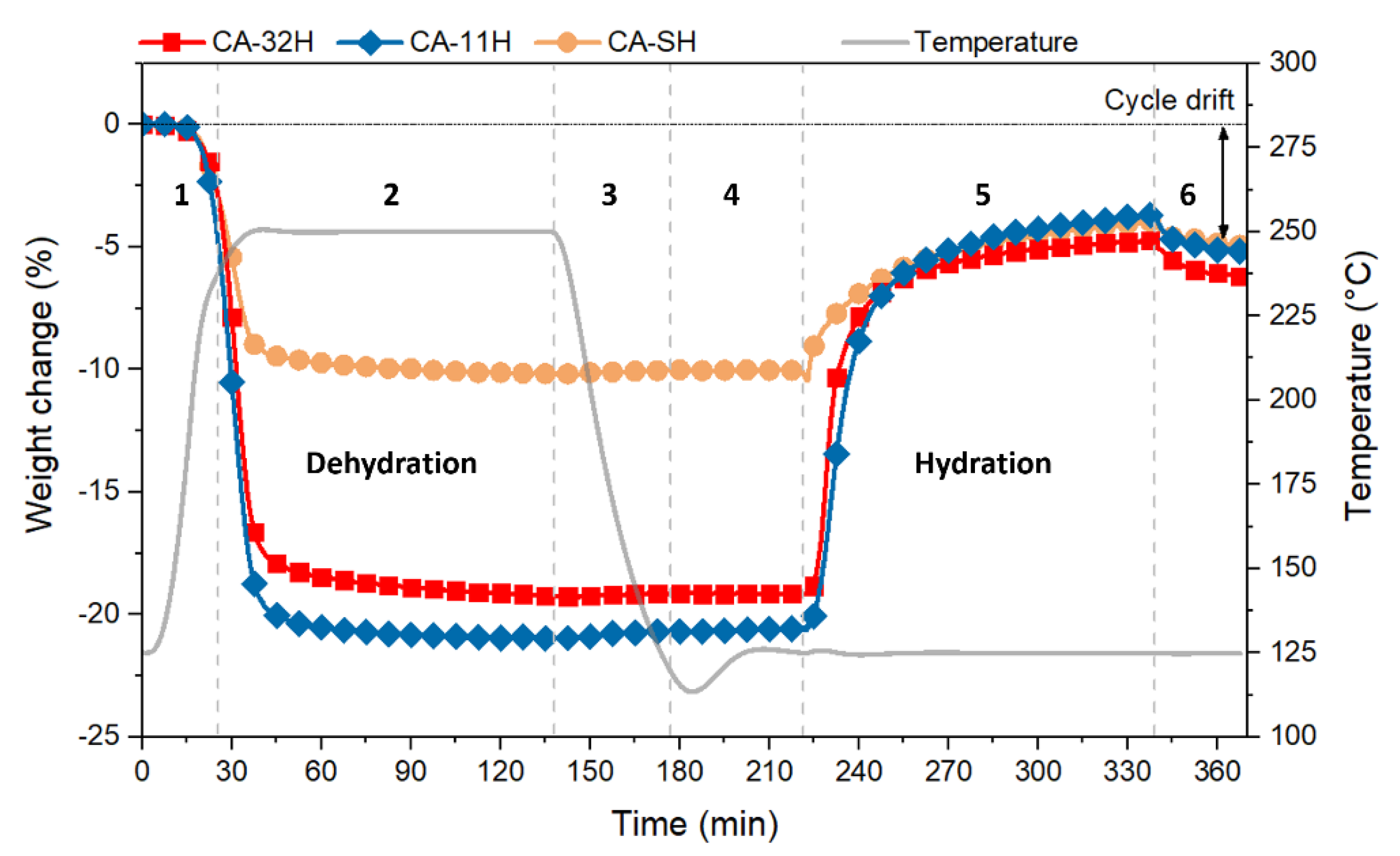
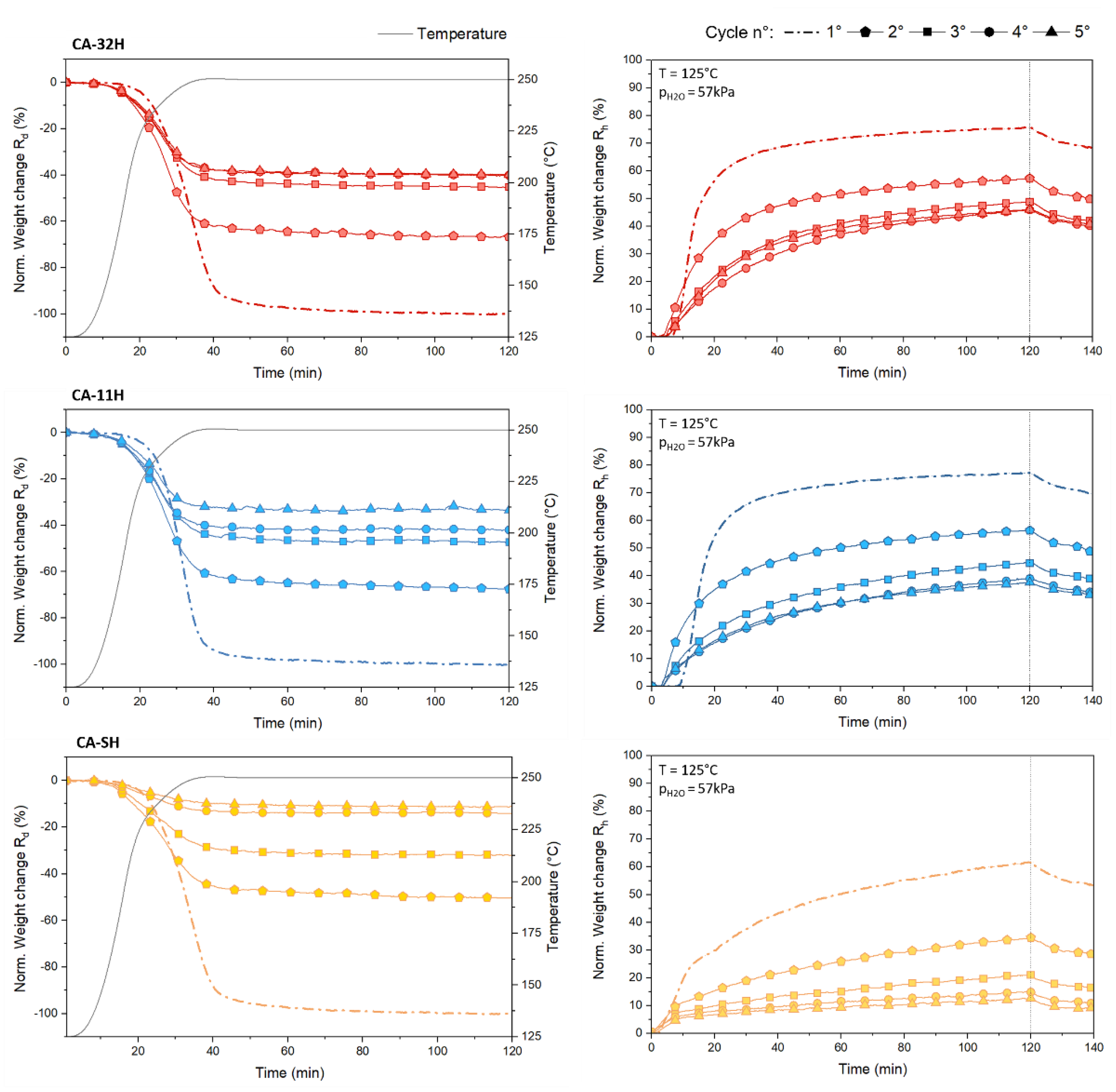
3.3. Dehydration/Hydration Stability
4. Conclusions
- Co-precipitation and solid–solid methods have been studied and compared as possible strategies to obtain low-cost tricalcium aluminate hexahydrate materials. The solid–solid reaction pointed out that it is not possible to obtain a material with appropriate thermochemical features without passing through a preliminary synthetic route that leads to a better chemical interaction between Ca2+ and Al3+.
- The structural and thermochemical characterization showed higher katoite content (98.6%) if a 1:1 Ca/Al ratio was initially used in co-precipitation, which also involves a greater heat of dehydration (807.0 kJ/kg), compared to the other products.
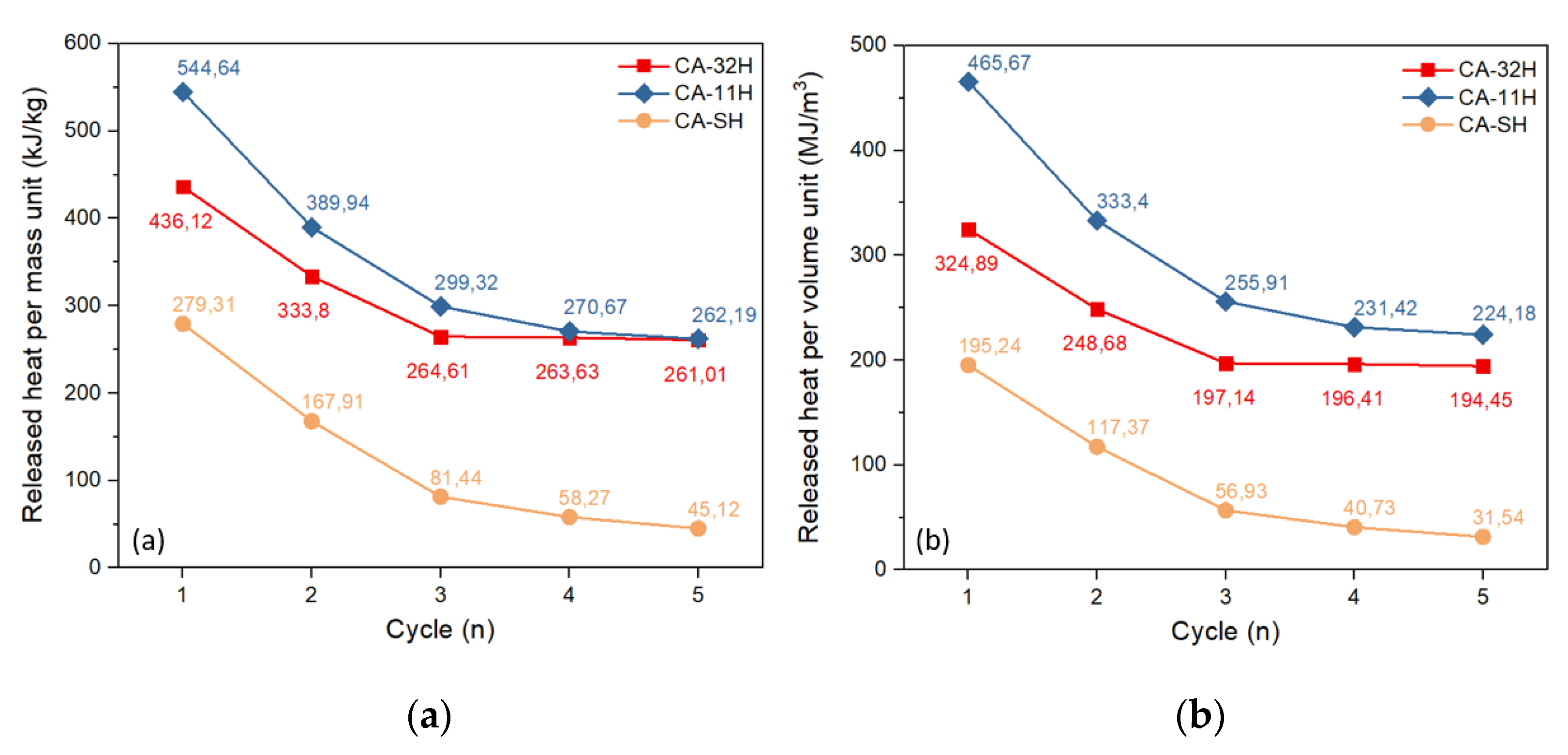
- The study on dehydration/hydration cycled materials evinced that the initial samples underwent a structural change during the cycles. As a consequence, a decrease in conversion yields and, therefore, in stored–released heat, is observed, with respect to the starting materials.
- Calcium hydroxide appears to be important on the stabilization of the material, partially preventing decomposition phenomena.
- Once a cycle stability is reached, the amount of stored/released heat per mass unit is similar for both co-precipitated products (about 260 kJ/kg of hydrated material), while in terms of storage density, CA-11H exhibits the highest capacity (224.2 MJ/m3).
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Haddad, C.; Périlhon, C.; Danlos, A.; François, M.X.; Descombes, G. Some efficient solutions to recover low and medium waste heat: Competitiveness of the thermoacoustic technology. Energy Procedia 2014, 50, 1056–1069. [Google Scholar] [CrossRef]
- Liu, J.; Chang, Z.; Wang, L.; Xu, J.; Kuang, R.; Wu, Z. Exploration of Basalt Glasses as High-Temperature Sensible Heat Storage Materials. ACS Omega 2020, 5, 19236–19246. [Google Scholar] [CrossRef]
- McGillicuddy, R.D.; Thapa, S.; Wenny, M.B.; Gonzalez, M.I.; Mason, J.A. Metal-organic phase-change materials for thermal energy storage. J. Am. Chem. Soc. 2020, 142, 19170–19180. [Google Scholar] [CrossRef]
- Cabeza, L.F.; Martorell, I.; Miró, L.; Fernández, A.I.; Barreneche, C. Introduction to Thermal Energy Storage (TES) Systems; Woodhead Publishing Limited: Cambridge, UK, 2015; ISBN 9781782420965. [Google Scholar]
- Henninger, S.K.; Jeremias, F.; Kummer, H.; Schossig, P.; Henning, H.M. Novel sorption materials for solar heating and cooling. Energy Procedia 2012, 30, 279–288. [Google Scholar] [CrossRef]
- Cabeza, L.F.; Solé, A.; Barreneche, C. Review on sorption materials and technologies for heat pumps and thermal energy storage. Renew. Energy 2017, 110, 3–39. [Google Scholar] [CrossRef]
- Alva, G.; Lin, Y.; Fang, G. An overview of thermal energy storage systems. Energy 2018, 144, 341–378. [Google Scholar] [CrossRef]
- Linder, M. Using Thermochemical Reactions in Thermal Energy Storage Systems; Woodhead Publishing Limited: Cambridge, UK, 2014; ISBN 9781782420965. [Google Scholar]
- Sunku Prasad, J.; Muthukumar, P.; Desai, F.; Basu, D.N.; Rahman, M.M. A critical review of high-temperature reversible thermochemical energy storage systems. Appl. Energy 2019, 254, 113733. [Google Scholar] [CrossRef]
- Chan, C.W.; Ling-Chin, J.; Roskilly, A.P. A review of chemical heat pumps, thermodynamic cycles and thermal energy storage technologies for low grade heat utilisation. Appl. Therm. Eng. 2013, 53, 160–176. [Google Scholar] [CrossRef]
- Neveu, P.; Castaing, J. Solid-gas chemical heat pumps: Field of application and performance of the internal heat of reaction recovery process. Heat Recover. Syst. CHP 1993, 13, 233–251. [Google Scholar] [CrossRef]
- Cot-Gores, J.; Castell, A.; Cabeza, L.F. Thermochemical energy storage and conversion: A-state-of-the-art review of the experimental research under practical conditions. Renew. Sustain. Energy Rev. 2012, 16, 5207–5224. [Google Scholar] [CrossRef]
- Albrecht, K.J.; Jackson, G.S.; Braun, R.J. Thermodynamically consistent modeling of redox-stable perovskite oxides for thermochemical energy conversion and storage. Appl. Energy 2016, 165, 285–296. [Google Scholar] [CrossRef]
- Huo, J.; Ge, R.; Liu, Y.; Guo, J.; Lu, L.; Chen, W.; Liu, C.; Gao, H.; Liu, H. Recent advances of two-dimensional molybdenum disulfide based materials: Synthesis, modification and applications in energy conversion and storage. Sustain. Mater. Technol. 2020, 24. [Google Scholar] [CrossRef]
- Zhao, R.; Cui, D.; Dai, J.; Xiang, J.; Wu, F. Morphology controllable NiCo2O4 nanostructure for excellent energy storage device and overall water splitting. Sustain. Mater. Technol. 2020, 24, e00151. [Google Scholar] [CrossRef]
- André, L.; Abanades, S.; Cassayre, L. Experimental Investigation of Co-Cu, Mn-Co, and Mn-Cu Redox Materials Applied to Solar Thermochemical Energy Storage. ACS Appl. Energy Mater. 2018, 1, 3385–3395. [Google Scholar] [CrossRef]
- Gravogl, G.; Knoll, C.; Artner, W.; Welch, J.M.; Eitenberger, E.; Friedbacher, G.; Harasek, M.; Hradil, K.; Werner, A.; Weinberger, P.; et al. Pressure effects on the carbonation of MeO (Me = Co, Mn, Pb, Zn) for thermochemical energy storage. Appl. Energy 2019, 252, 113451. [Google Scholar] [CrossRef]
- André, L.; Abanades, S.; Flamant, G. Screening of thermochemical systems based on solid-gas reversible reactions for high temperature solar thermal energy storage. Renew. Sustain. Energy Rev. 2016, 64, 703–715. [Google Scholar] [CrossRef]
- Mastronardo, E.; Bonaccorsi, L.; Kato, Y.; Piperopoulos, E.; Milone, C. Efficiency improvement of heat storage materials for MgO/H2O/Mg(OH)2 chemical heat pumps. Appl. Energy 2016, 162, 31–39. [Google Scholar] [CrossRef]
- Kato, Y.; Takahashi, R.; Sekiguchi, T.; Ryu, J. Study on medium-temperature chemical heat storage using mixed hydroxides. Int. J. Refrig. 2009, 32, 661–666. [Google Scholar] [CrossRef]
- Sakellariou, K.G.; Karagiannakis, G.; Criado, Y.A.; Konstandopoulos, A.G. Calcium oxide based materials for thermochemical heat storage in concentrated solar power plants. Sol. Energy 2015, 122, 215–230. [Google Scholar] [CrossRef]
- Sánchez Jiménez, P.E.; Perejón, A.; Benítez Guerrero, M.; Valverde, J.M.; Ortiz, C.; Pérez Maqueda, L.A. High-performance and low-cost macroporous calcium oxide based materials for thermochemical energy storage in concentrated solar power plants. Appl. Energy 2019, 235, 543–552. [Google Scholar] [CrossRef]
- Álvarez Criado, Y.; Alonso, M.; Abanades, J.C. Composite Material for Thermochemical Energy Storage Using CaO/Ca(OH)2. Ind. Eng. Chem. Res. 2015, 54, 9314–9327. [Google Scholar] [CrossRef]
- Shkatulov, A.; Krieger, T.; Zaikovskii, V.; Chesalov, Y.; Aristov, Y. Doping magnesium hydroxide with sodium nitrate: A new approach to tune the dehydration reactivity of heat-storage materials. ACS Appl. Mater. Interfaces 2014, 6, 19966–19977. [Google Scholar] [CrossRef]
- Wang, X.; Chen, H.; Xu, Y.; Wang, L.; Hu, S. Advances and prospects in thermal energy storage: A critical review. Kexue Tongbao Chin. Sci. Bull. 2017, 62, 1602–1610. [Google Scholar] [CrossRef]
- Scrivener, K.L.; Capmas, A. Calcium Aluminate Cements. Lea’s Chem. Cem. Concr. 2003, 713–782. [Google Scholar] [CrossRef]
- Kapeluszna, E.; Kotwica, Ł.; Pichór, W.; Nocuń-Wczelik, W. Cement-based composites with waste expanded perlite—Structure, mechanical properties and durability in chloride and sulphate environments. Sustain. Mater. Technol. 2020, 24. [Google Scholar] [CrossRef]
- Liiv, J.; Teppand, T.; Rikmann, E.; Tenno, T. Novel ecosustainable peat and oil shale ash-based 3D-printable composite material. Sustain. Mater. Technol. 2018, 17, e00067. [Google Scholar] [CrossRef]
- Geiger, C.A.; Dachs, E.; Benisek, A. Thermodynamic behavior and properties of katoite ( hydrogrossular ): A calorimetric study. Am. Mineral. 2012, 97, 1252–1255. [Google Scholar] [CrossRef]
- Stein, H.N. Thermodynamic considerations on the hydration mechanisms of Ca3SiO5 and Ca3Al2O6. Cem. Concr. Res. 1972, 2, 167–177. [Google Scholar] [CrossRef]
- Lacivita, V.; Mahmoud, A.; Arco, P.D.; Mustapha, S. Hydrogrossular, Ca3Al2(SiO4)3–x(H4O4)x: An ab initio investigation of its structural and energetic properties. Am. Mineral. 2015, 100, 2637–2649. [Google Scholar] [CrossRef]
- Cavenett The Dehydration of Tricalcium aluminate exahydrate. J. Chem. Inf. Model. 1941, 19, 1689–1699. [CrossRef]
- Ball, M.C.; Ladner, N.G.; Taylor, J.A. The dehydroxylation of tricalcium aluminate 6-hydrate. Thermochim. Acta 1976, 17, 207–215. [Google Scholar] [CrossRef]
- Rivas-Mercury, J.M.; Pena, P.; de Aza, A.H.; Turrillas, X. Dehydration of Ca3Al2(SiO4)y(OH)4(3−y) (0 < y < 0.176) studied by neutron thermodiffractometry J.M. J. Eur. Ceram. Soc. 2008, 28, 1737–1748. [Google Scholar] [CrossRef]
- Antonovič, V.; Keriene, J.; Boris, R.; Aleknevičius, M. The effect of temperature on the formation of the hydrated calcium aluminate cement structure. Procedia Eng. 2013, 57, 99–106. [Google Scholar] [CrossRef]
- Ukrainczyk, N.; Matusinović, T. Thermal properties of hydrating calcium aluminate cement pastes. Cem. Concr. Res. 2010, 40, 128–136. [Google Scholar] [CrossRef]
- Mohamed, B.M.; Sharp, J.H. Kinetics and mechanism of formation of tricalcium aluminate, Ca3Al2O6. Thermochim. Acta 2002, 388, 105–114. [Google Scholar] [CrossRef]
- Hem, J.D.; Roberson, C.E. Form and Stability of Aluminum Hydroxide Complexes in Dilute Solution. USGS Water Supply Pap. 1967, 1827-A, 55. [Google Scholar] [CrossRef]
- Whittington, B.I.; Fallows, T.M.; Willing, M.J. Tricalcium aluminate hexahydrate (TCA) filter aid in the Bayer industry: Factors affecting TCA preparation and morphology. Int. J. Miner. Process. 1997, 49, 1–29. [Google Scholar] [CrossRef]
- Wefers, K.; Misra, C. Oxides and Hydroxides of Aluminum. Alcoa Tech. Pap. 1987, 19, 100. [Google Scholar]


| Sample Code | nCa (mmol) | nAl (mmol) | Method | Ca/Al Molar Ratio |
|---|---|---|---|---|
| CA-S | 15 | 10 | Solid–solid | 3:2 |
| CA-11 | 10 | 10 | Co-precipitation | 1:1 |
| CA-32 | 15 | 10 | Co-precipitation | 3:2 |
| Hydration Temperature (°C) | Main Formed Phase(s) | Ref. |
|---|---|---|
| <15 | CaAl2O4·10H2O | [36] |
| 15–27 | CaAl2O4·10H2O/Ca2Al2O5·8H2O | [36] |
| 27–70 | Ca2Al2O5·8H2O/Ca3Al2O6·6H2O | [36] |
| >70 | Ca3Al2O6·6H2O | [15] |
| Step n° | Temperature (°C) | Atmosphere | Method | Time (Min) |
|---|---|---|---|---|
| 1 | 125→250 | N2 | Heating | - |
| 2 | 250 | N2 | Isothermal | 120 |
| 3 | 250→125 | N2 | Cooling | - |
| 4 | 125 | N2 | Stabilization | - |
| 5 | 125 | N2/H2O | Hydration | 120 |
| 6 | 125 | N2 | Release of adsorbed water | 30 |
| Sample ID | Tonset (°C) | Weight Loss (%) | Heat (kJ/kg) | Katoite (%) |
|---|---|---|---|---|
| CA-32H | 247.5 | −19.04 | 655.8 | 88.9 |
| CA-11H | 245.8 | −21.12 | 807.0 | 98.6 |
| CA-SH | 248.7 | −10.15 | 530.2 | <46.9 |
| Sample ID | Cycle | Dehydration Tonset (°C) | Rd (%) | Rh (%) |
|---|---|---|---|---|
| CA-32H | 1 | 241.5 | 100 | 66.5 |
| 2 | 231.1 | 67.6 | 50.9 | |
| 3 | 229.8 | 45.7 | 40.4 | |
| 4 | 230.2 | 40.4 | 40.2 | |
| 5 | 230.0 | 40.1 | 39.8 | |
| CA-11H | 1 | 242.7 | 100 | 67.5 |
| 2 | 231.4 | 68.1 | 48.3 | |
| 3 | 229.4 | 47.6 | 37.1 | |
| 4 | 229.3 | 42.5 | 33.6 | |
| 5 | 229.6 | 33.3 | 32.5 | |
| CA-SH | 1 | 241.0 | 100 | 52.7 |
| 2 | 233.2 | 51.0 | 31.7 | |
| 3 | 233.3 | 32.1 | 15.4 | |
| 4 | 231.7 | 12.2 | 11.0 | |
| 5 | 230.5 | 11.4 | 8.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alvaro, F.; Piperopoulos, E.; Calabrese, L.; La Mazza, E.; Lanza, M.; Milone, C. Performances Assessment of Tricalcium Aluminate as an Innovative Material for Thermal Energy Storage Applications. Appl. Sci. 2021, 11, 1958. https://doi.org/10.3390/app11041958
Alvaro F, Piperopoulos E, Calabrese L, La Mazza E, Lanza M, Milone C. Performances Assessment of Tricalcium Aluminate as an Innovative Material for Thermal Energy Storage Applications. Applied Sciences. 2021; 11(4):1958. https://doi.org/10.3390/app11041958
Chicago/Turabian StyleAlvaro, Fabrizio, Elpida Piperopoulos, Luigi Calabrese, Emanuele La Mazza, Maurizio Lanza, and Candida Milone. 2021. "Performances Assessment of Tricalcium Aluminate as an Innovative Material for Thermal Energy Storage Applications" Applied Sciences 11, no. 4: 1958. https://doi.org/10.3390/app11041958
APA StyleAlvaro, F., Piperopoulos, E., Calabrese, L., La Mazza, E., Lanza, M., & Milone, C. (2021). Performances Assessment of Tricalcium Aluminate as an Innovative Material for Thermal Energy Storage Applications. Applied Sciences, 11(4), 1958. https://doi.org/10.3390/app11041958









