Bioengineered Skin Substitutes: Advances and Future Trends
Abstract
1. Introduction
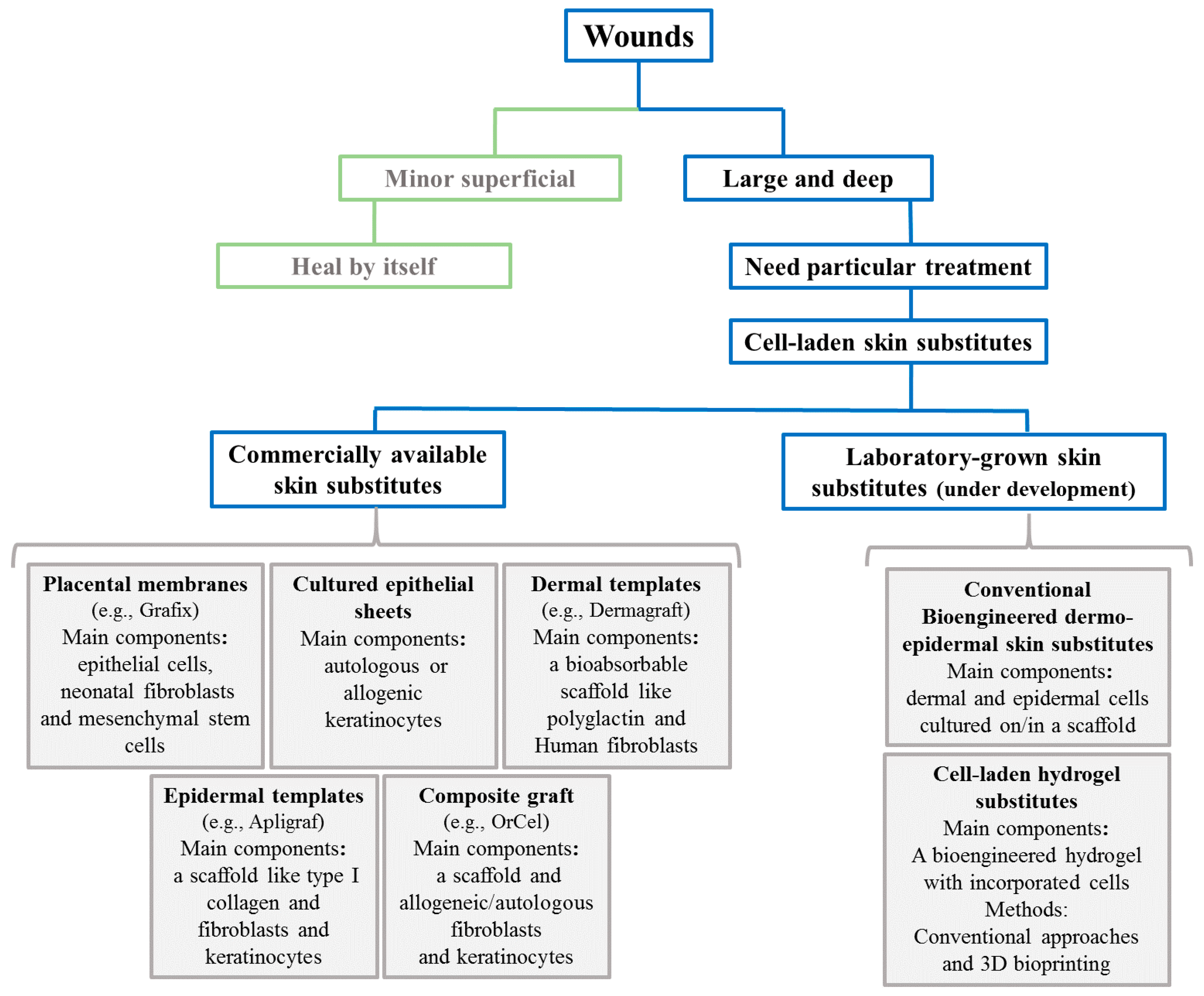
2. Skin Injuries and Common Treatment Techniques
3. Cell-Based Skin Substitutes
3.1. Cell-Laden Commercial Skin Templates
3.1.1. Placental Membranes
3.1.2. Cultured Epithelial Sheets
3.1.3. Dermal Templates
3.1.4. Epidermal Templates
3.1.5. Dermo-Epidermal Skin Equivalents (Composite Graft)
3.2. Bioengineered Dermo-Epidermal Skin Substitutes (Under Development)
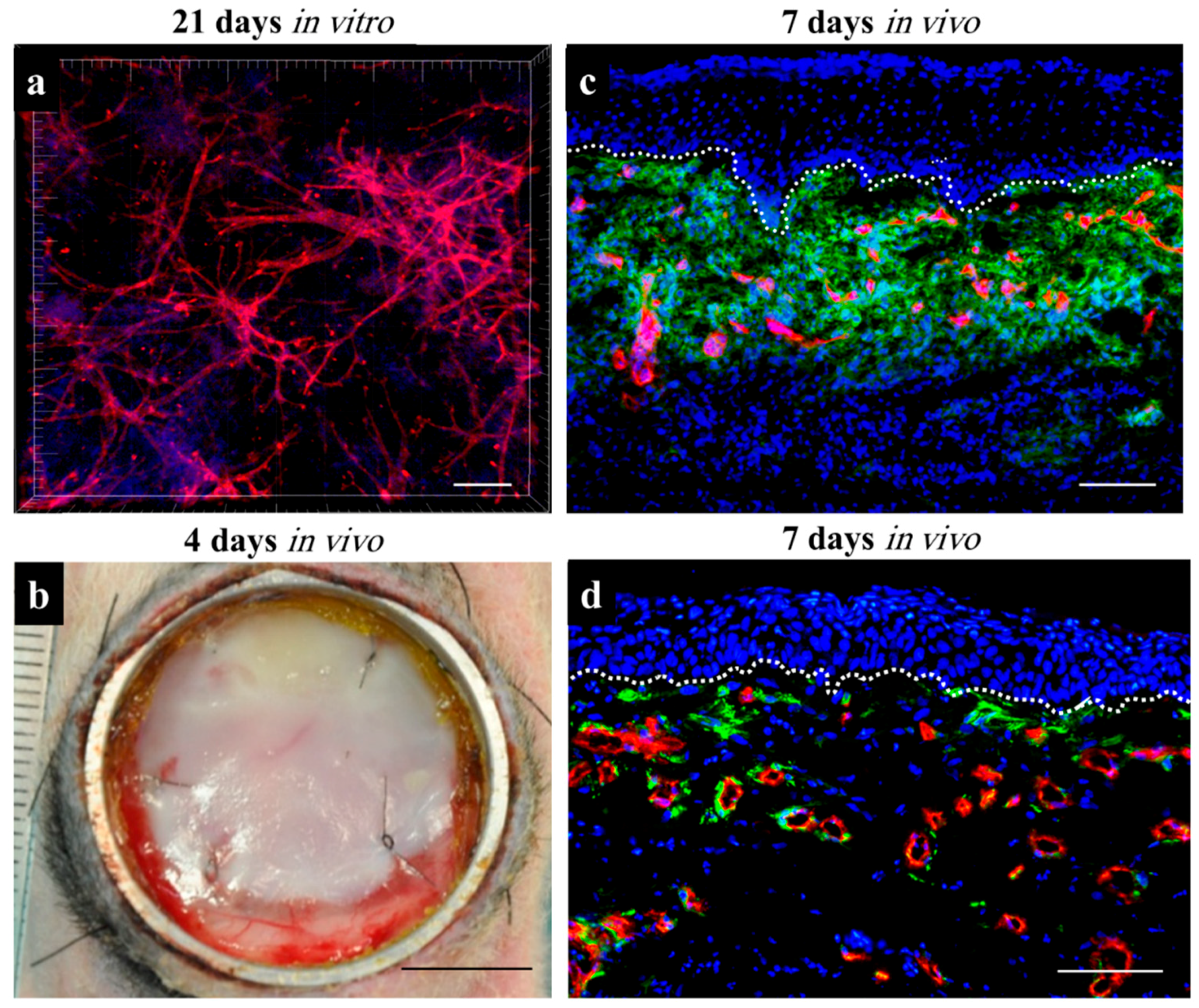
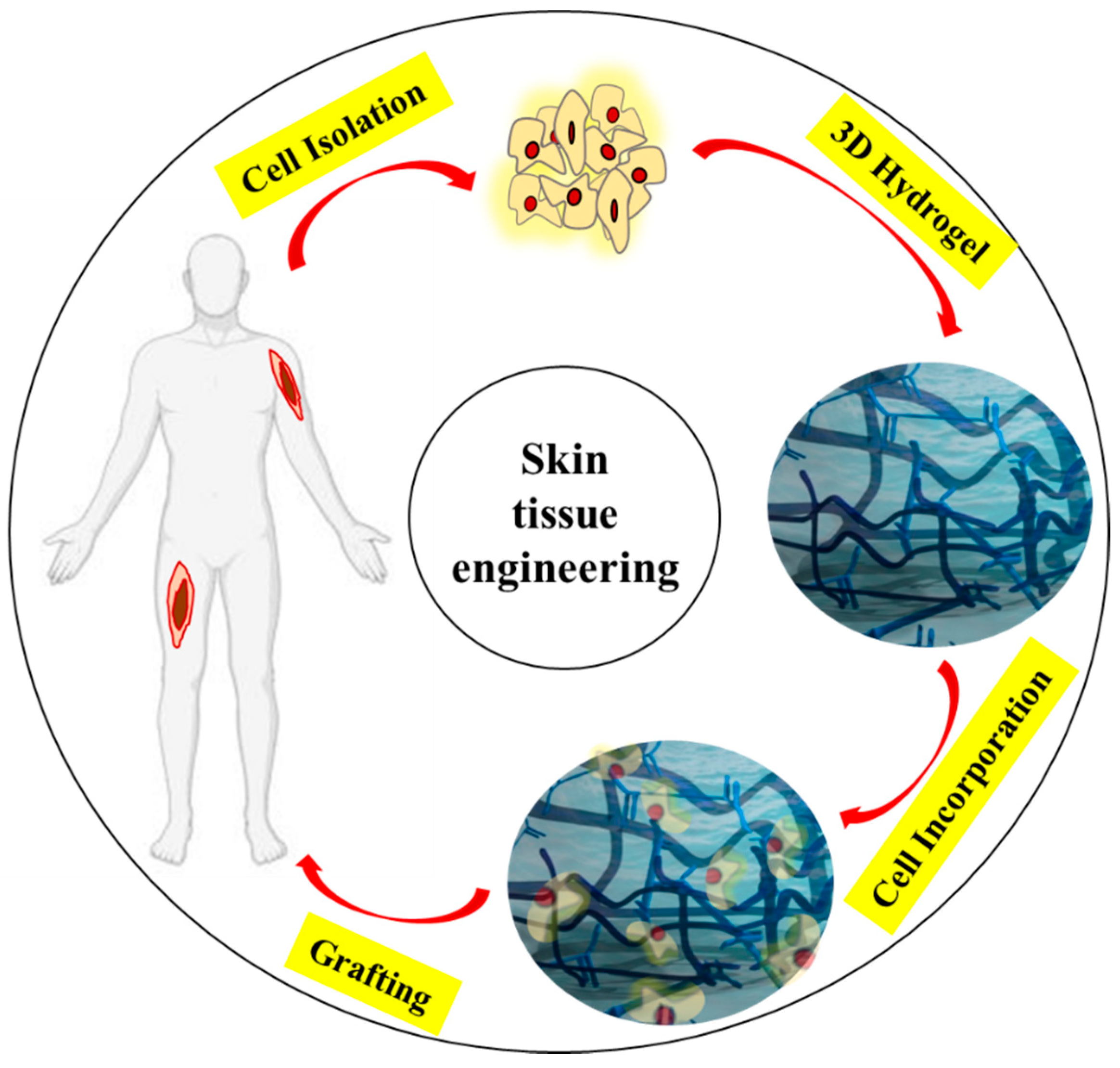
3.2.1. Cell-Laden Hydrogels as Wound Dressings
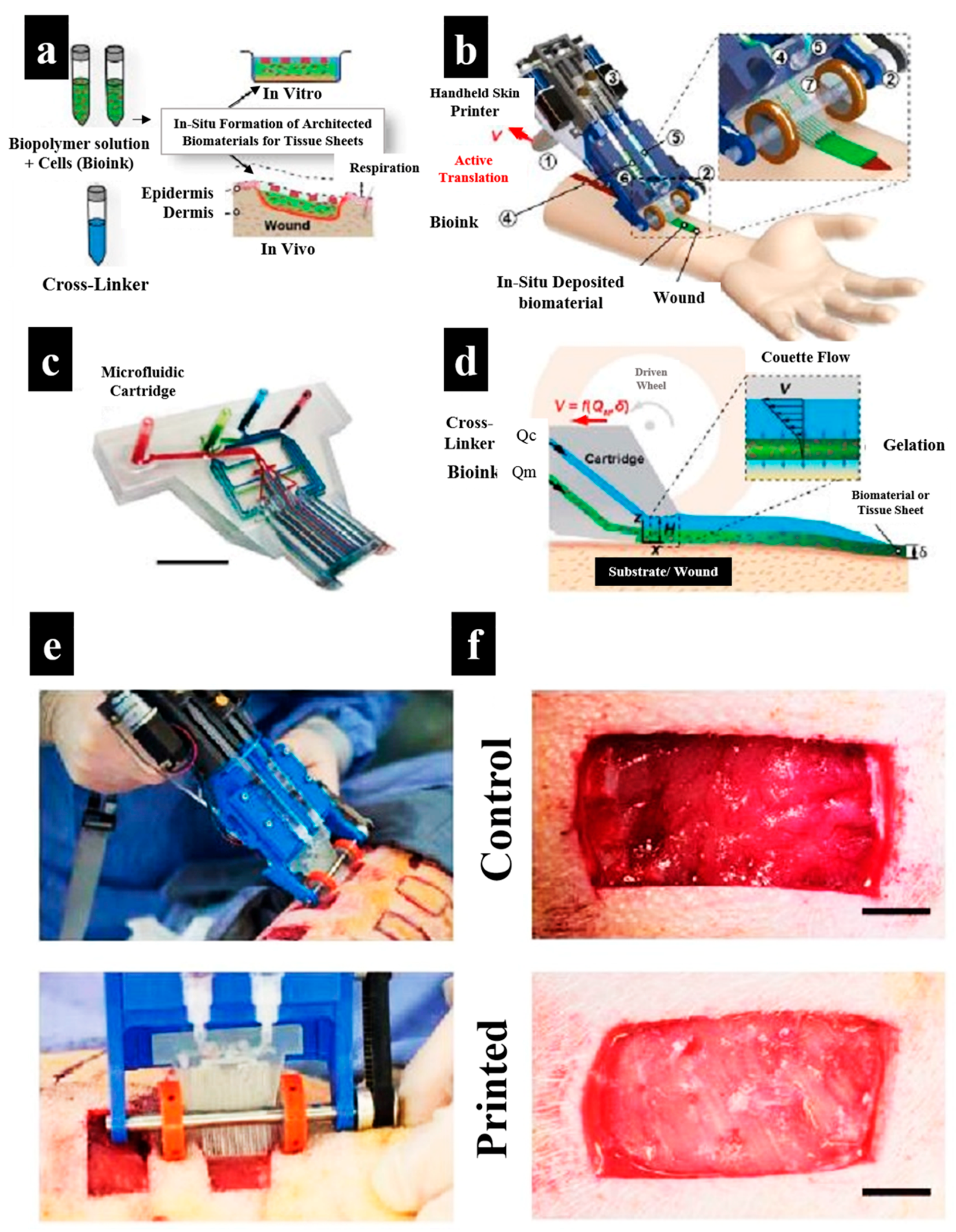
3.2.2. 3D Bioprinting of Cell-Laden Hydrogels for Wound Dressings
4. Conclusions and Future Direction
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Shevchenko, R.V.; James, S.E. A review of tissue-engineered skin bioconstructs available for skin reconstruction. J. R. Soc. Interface 2009, 7, 229–258. [Google Scholar] [CrossRef] [PubMed]
- Lazarus, G.S.; Cooper, D.M.; Knighton, D.R.; Margolis, D.J.; Percoraro, R.E.; Rodeheaver, G.; Robson, M.C. Definitions and guidelines for assessment of wounds and evaluation of healing. Wound Repair Regen. 1994, 2, 165–170. [Google Scholar] [CrossRef]
- Tavakoli, S.; Mokhtari, H.; Kharaziha, M.; Kermanpur, A.; Talebi, A.; Moshtaghian, J. A multifunctional nanocomposite spray dressing of Kappa-carrageenan-polydopamine modified ZnO/L-glutamic acid for diabetic wounds. Mater. Sci. Eng. C 2020, 111, 110837. [Google Scholar] [CrossRef] [PubMed]
- Kanitakis, J. Anatomy, histology and immunohistochemistry of normal human skin. Eur. J. Dermatol. 2002, 12, 390–401. [Google Scholar]
- Schmook, F.P.; Meingassner, J.G.; Billich, A. Comparison of human skin or epidermis models with human and animal skin in in-vitro percutaneous absorption. Int. J. Pharm. 2001, 215, 51–56. [Google Scholar] [CrossRef]
- Tavakoli, S.; Klar, A.S. Advanced Hydrogels as Wound Dressings. Biomolecules 2020, 10, 1169. [Google Scholar] [CrossRef]
- Subramanian, A.; Krishnan, U.M.; Sethuraman, S. Skin tissue regeneration. In Electrospinning for Tissue Regeneration; Woodhead Publishing: Cambridge, UK, 2011; Volume 1, pp. 298–316. [Google Scholar] [CrossRef]
- Rowan, M.P.; Cancio, L.C.; Elster, E.A.; Burmeister, D.M.; Rose, L.F.; Natesan, S.; Chan, R.K.; Christy, R.J.; Chung, K.K. Burn wound healing and treatment: Review and advancements. Crit. Care 2015, 19, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Boateng, J.S.; Matthews, K.H.; Stevens, H.N.; Eccleston, G.M. Wound Healing Dressings and Drug Delivery Systems: A Review. J. Pharm. Sci. 2008, 97, 2892–2923. [Google Scholar] [CrossRef]
- Gaur, M.; Dobke, M.K.; Lunyak, V.V. Mesenchymal Stem Cells from Adipose Tissue in Clinical Applications for Dermatological Indications and Skin Aging. Int. J. Mol. Sci. 2017, 18, 208. [Google Scholar] [CrossRef]
- Roupé, K.M.; Nybo, M.; Sjöbring, U.; Alberius, P.; Schmidtchen, A.; Sørensen, O.E.; Roup, M.N.K.M. Injury Is a Major Inducer of Epidermal Innate Immune Responses during Wound Healing. J. Investig. Dermatol. 2010, 130, 1167–1177. [Google Scholar] [CrossRef]
- Klar, A.S.; Güven, S.; Zimoch, J.; Zapiórkowska, N.A.; Biedermann, T.; Böttcher-Haberzeth, S.; Meuli-Simmen, C.; Martin, I.; Scherberich, A.; Reichmann, E.; et al. Characterization of vasculogenic potential of human adipose-derived endothelial cells in a three-dimensional vascularized skin substitute. Pediatr. Surg. Int. 2015, 32, 17–27. [Google Scholar] [CrossRef]
- Klar, A.S.; Michalak-Mićka, K.; Biedermann, T.; Simmen-Meuli, C.; Reichmann, E.; Meuli, M. Characterization of M1 and M2 polarization of macrophages in vascularized human dermo-epidermal skin substitutes in vivo. Pediatr. Surg. Int. 2017, 34, 129–135. [Google Scholar] [CrossRef]
- Delavary, B.M.; Van Der Veer, W.M.; Van Egmond, M.; Niessen, F.B.; Beelen, R.H. Macrophages in skin injury and repair. Immunobiology 2011, 216, 753–762. [Google Scholar] [CrossRef]
- Sukari, H.A.; Khoo, T.L.; Yussof, S.J. Biologic and synthetic skin substitutes: An overview. Indian J Plast Surg 2010, 43, 23–28. [Google Scholar] [CrossRef]
- Harding, K.G.; Morris, H.L.; Patel, G.K. Science, medicine, and the future: Healing chronic wounds. BMJ 2002, 324, 160–163. [Google Scholar] [CrossRef]
- Guo, S.; DiPietro, L.A. Critical Review in Oral Biology & Medicine: Factors Affecting Wound Healing. J. Dent. Res. 2010, 89, 219–229. [Google Scholar] [CrossRef]
- Johnson, K.E.; Wilgus, T.A. Vascular Endothelial Growth Factor and Angiogenesis in the Regulation of Cutaneous Wound Repair. Adv. Wound Care (New Rochelle) 2014, 3, 647–661. [Google Scholar] [CrossRef]
- Demidova-Rice, T.N.; Durham, J.T.; Herman, I.M. Wound Healing Angiogenesis: Innovations and Challenges in Acute and Chronic Wound Healing. Adv. Wound Care 2012, 1, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Klar, A.S.; Güven, S.; Biedermann, T.; Luginbühl, J.; Böttcher-Haberzeth, S.; Meuli-Simmen, C.; Meuli, M.; Martin, I.; Scherberich, A.; Reichmann, E. Tissue-engineered dermo-epidermal skin grafts prevascularized with adipose-derived cells. Biomaterials 2014, 35, 5065–5078. [Google Scholar] [CrossRef] [PubMed]
- Frueh, F.S.; Macedo, N.; Calcagni, M.; Giovanoli, P.; Lindenblatt, N. The Crucial Role of Vascularization and Lymphangiogenesis in Skin Reconstruction. Eur. Surg. Res. 2018, 59, 242–254. [Google Scholar] [CrossRef] [PubMed]
- Shahin, H.; Elmasry, M.; Steinvall, L.; Söberg, F.; Serafi, A. Vascularization is the next challenge for skin tissue engineering as a solution for burn management. Burns Trauma 2020, 8, 022. [Google Scholar] [CrossRef] [PubMed]
- Klar, A.S.; Böttcher-Haberzeth, S.; Biedermann, T.; Schiestl, C.; Reichmann, E.; Meuli, M. Analysis of blood and lymph vascularization patterns in tissue-engineered human dermo-epidermal skin analogs of different pigmentation. Pediatr. Surg. Int. 2014, 30, 223–231. [Google Scholar] [CrossRef]
- Pourmoussa, A.; Gardner, D.J.; Johnson, M.B.; Wong, A.K. An update and review of cell-based wound dressings and their integration into clinical practice. Ann. Transl. Med. 2016, 4, 457. [Google Scholar] [CrossRef]
- Klar, A.S.; Biedermann, T.; Simmen-Meuli, C.; Reichmann, E.; Meuli, M. Comparison of in vivo immune responses following transplantation of vascularized and non-vascularized human dermo-epidermal skin substitutes. Pediatr. Surg. Int. 2016, 33, 377–382. [Google Scholar] [CrossRef]
- Pogorielov, M.; Hapchenko, A.; Pogorielov, O.O.M. Tissue Engineering: Challenges and Selected Application. Adv. Tissue Eng. Regen. Med. Open Access 2017, 3, 1–6. [Google Scholar] [CrossRef]
- Li, C.; Guan, G.; Reif, R.; Huang, Z.; Wang, R.K. Determining elastic properties of skin by measuring surface waves from an impulse mechanical stimulus using phase-sensitive optical coherence tomography. J. R. Soc. Interface 2011, 9, 831–841. [Google Scholar] [CrossRef]
- Olena, P.; Prokopyuk, V.; Figueiredo, C.; Pogozhykh, D. Placenta and Placental Derivatives in Regenerative Therapies: Experimental Studies, History, and Prospects. Stem Cells Int. 2018, 2018, 1–14. [Google Scholar] [CrossRef]
- Maxson, S.; Lopez, E.A.; Yoo, D.; Danilkovitch-Miagkova, A.; Leroux, M.A. Concise Review: Role of Mesenchymal Stem Cells in Wound Repair. STEM CELLS Transl. Med. 2012, 1, 142–149. [Google Scholar] [CrossRef]
- Lavery, L.A.; Fulmer, J.; Shebetka, K.A.; Regulski, M.; Vayser, D.; Fried, D.; Kashefsky, H.; Owings, T.M.; Nadarajah, J. The Grafix Diabetic Foot Ulcer Study Group The efficacy and safety of Grafix® for the treatment of chronic diabetic foot ulcers: Results of a multi-centre, controlled, randomised, blinded, clinical trial. Int. Wound J. 2014, 11, 554–560. [Google Scholar] [CrossRef] [PubMed]
- Wong, T.; McGrath, J.A.; Navsaria, H. The role of fibroblasts in tissue engineering and regeneration. Br. J. Dermatol. 2007, 156, 1149–1155. [Google Scholar] [CrossRef]
- Gibbons, G.W. Grafix®, a Cryopreserved Placental Membrane, for the Treatment of Chronic/Stalled Wounds. Adv. Wound Care 2015, 4, 534–544. [Google Scholar] [CrossRef]
- Landsman, A.S.; Cook, J.J.; Cook, A.E.; Landsman, A.R.; Garrett, P.; Yoon, J.; Kirkwood, A.; Desman, E. A Retrospective Clinical Study of 188 Consecutive Patients to Examine the Effectiveness of a Biologically Active Cryopreserved Human Skin Allograft (TheraSkin®) on the Treatment of Diabetic Foot Ulcers and Venous Leg Ulcers. Foot Ankle Spéc. 2010, 4, 29–41. [Google Scholar] [CrossRef]
- Wood, F.M.; Kolybaba, M.; Allen, P. The use of cultured epithelial autograft in the treatment of major burn wounds: Eleven years of clinical experience. Burns 2006, 32, 538–544. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.-R.; Hao, Z.-Q.; Nie, L.-J.; Liu, G.-F. Coverage of full skin thickness burns with allograft inoculated with autogenous epithelial cells. Burns 1986, 12, 220–224. [Google Scholar] [CrossRef]
- Barret, P.J.; Wolf, S.E.; Desai, M.H.; Herndon, D.N. Cost-Efficacy of Cultured Epidermal Autografts in Massive Pediatric Burns. Ann. Surg. 2000, 231, 869–876. [Google Scholar] [CrossRef]
- Rheinwatd, J.G.; Green, H. Seria cultivation of strains of human epidemal keratinocytes: The formation keratinizin colonies from single cell is. Cell 1975, 6, 331–343. [Google Scholar] [CrossRef]
- Rheinwald, J.G.; Green, H. Epidermal growth factor and the multiplication of cultured human epidermal keratinocytes. Nat. Cell Biol. 1977, 265, 421–424. [Google Scholar] [CrossRef]
- Gobet, R.; Raghunath, M.; Altermatt, S.; Meuli-Simmen, C.; Benathan, M.; Dietl, A.; Meuli, M. Efficacy of cultured epithelial autografts in pediatric burns and reconstructive surgery. Surgery 1997, 121, 654–661. [Google Scholar] [CrossRef]
- Atiyeh, B.S.; Costagliola, M. Cultured epithelial autograft (CEA) in burn treatment: Three decades later. Burns 2007, 33, 405–413. [Google Scholar] [CrossRef]
- Ronfard, V.; Rives, J.M.; Neveux, Y.; Carsin, H.; Barrandon, Y. Long-term regeneration of human epidermis on third degree burns transplanted with autologous cultured epithelium grown on a fibrin matrix1, 2. Transplantation 2000, 70, 1588–1598. [Google Scholar] [CrossRef] [PubMed]
- Cuono, C.; Langdon, R.; McGuire, J. Use of cultured epidermal autografts and dermal allografts as skin replacement after burn injury. Lancet 1986, 327, 1123–1124. [Google Scholar] [CrossRef]
- Meuli, M.; Raghunath, M. Tops and flops using cultured epithelial autografts in children. Pediatr. Surg. Int. 1997, 12, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Krupp, S.; Benathan, M.; Meuli, M.; Déglise, B.; Holzer, E.; Wiesner, L.; Delacrétaz, F.; Chioléro, R. Current Concepts in Pediatric Burn Care: Management of Burn Wounds with Cultured Epidermal Autografts*. Eur. J. Pediatr. Surg. 1992, 2, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, N.; Sow, W.T.; Devi, D.; Ting, S.W.; Mandal, B.B.; Cho, N.-J. Silk fibroin–keratin based 3D scaffolds as a dermal substitute for skin tissue engineering. Integr. Biol. 2014, 7, 53–63. [Google Scholar] [CrossRef]
- Marston, W.A.; Hanft, J.; Norwood, P.; Pollak, R. The Efficacy and Safety of Dermagraft in Improving the Healing of Chronic Diabetic Foot Ulcers: Results of a prospective randomized trial. Diabetes Care 2003, 26, 1701–1705. [Google Scholar] [CrossRef]
- Hart, C.E.; Loewen-Rodriguez, A.; Lessem, J. Dermagraft: Use in the Treatment of Chronic Wounds. Adv. Wound Care 2012, 1, 138–141. [Google Scholar] [CrossRef]
- MacEwan, M.R.; MacEwan, S.R.; Kovacs, T.R.; Batts, J. What Makes the Optimal Wound Healing Material? A Review of Current Science and Introduction of a Synthetic Nanofabricated Wound Care Scaffold. Cureus 2017, 9, e1736. [Google Scholar] [CrossRef]
- Naughton, G.; Mansbridge, J.; Gentzkow, G. A Metabolically Active Human Dermal Replacement for the Treatment of Diabetic Foot Ulcers. Artif. Organs 1997, 21, 1203–1210. [Google Scholar] [CrossRef] [PubMed]
- Dermagraft: Use in the Treatment of Chronic Wounds—Google Scholar. Available online: https://scholar.google.com/scholar?hl=en&as_sdt=0%2C5&q=Dermagraft%3A+Use+in+the+Treatment+of+Chronic+Wounds&btnG= (accessed on 28 December 2020).
- Marston, W.A. Dermagraft®, a bioengineered human dermal equivalent for the treatment of chronic nonhealing diabetic foot ulcer. Expert Rev. Med. Devices 2004, 1, 21–31. [Google Scholar] [CrossRef]
- Omar, A.A.; Mavor, A.I.D.; Jones, A.M.; Homer-Vanniasinkam, S. Treatment of venous leg ulcers with Dermagraft®. Eur. J. Vasc. Endovas. Surg. 2004, 27, 666–672. [Google Scholar] [CrossRef] [PubMed]
- Gentzkow, G.D.; Iwasaki, S.D.; Hershon, K.S.; Mengel, M.; Prendergast, J.J.; Ricotta, J.J.; Lipkin, S. Use of dermagraft, a cultured human dermis, to treat diabetic foot ulcers. Diabetes Care 1996, 19, 350–354. [Google Scholar] [CrossRef]
- Kumar, S.; Kang, H.J.; Berthiaume, F. Scaffolds for epidermal tissue engineering. In Handbook of Tissue Engineering Scaffolds; Woodhead Publishing: Cambridge, UK, 2019; Volume 2, pp. 173–191. [Google Scholar]
- Curran, M.P.; Plosker, G.L. Bilayered Bioengineered Skin Substitute (Apligraf®): A Review of Its Use in the Treatment of Venous Leg Ulcers and Diabetic Foot Ulcers. BioDrugs 2002, 16, 439–455. [Google Scholar] [CrossRef] [PubMed]
- Eaglstein, W.H.; Alvarez, O.M.; Auletta, M.; Leffel, D.; Rogers, G.S.; Zitelli, J.A.; Norris, J.E.; Thomas, I.; Irondo, M.; Fewkes, J.; et al. Acute Excisional Wounds Treated with a Tissue-Engineered Skin (Apligraf). Dermatol. Surg. 1999, 25, 195–201. [Google Scholar] [CrossRef]
- Foley, E.; Robinson, A.; Maloney, M. Skin Substitutes and Dermatology: A Review. Curr. Dermatol. Rep. 2013, 2, 101–112. [Google Scholar] [CrossRef]
- Sibbald, R.G.; Zuker, R.; Coutts, P.; Coelho, S.; Williamson, D.; Queen, D. Using a dermal skin substitute in the treatment of chronic wounds secondary to recessive dystrophic epidermolysis bullosa: A case series. Ostomy Wound Manag. 2005, 51, 22–46. [Google Scholar]
- Hasegawa, T.; Suga, Y.; Mizoguchi, M.; Ikeda, S.; Ogawa, H.; Kubo, K.; Matsui, H.; Kagawa, S.; Kuroyanagi, Y. Clinical trial of allogeneic cultured dermal substitute for the treatment of intractable skin ulcers in 3 patients with recessive dystrophic epidermolysis bullosa*. J. Am. Acad. Dermatol. 2004, 50, 803–804. [Google Scholar] [CrossRef]
- Braziulis, E.; Biedermann, T.; Hartmann-Fritsch, F.; Schiestl, C.; Pontiggia, L.; Böttcher-Haberzeth, S.; Reichmann, E.; Meuli, M. Skingineering I: Engineering porcine dermo-epidermal skin analogues for autologous transplantation in a large animal model. Pediatr. Surg. Int. 2011, 27, 241–247. [Google Scholar] [CrossRef]
- Still, J.; Glat, P.; Silverstein, P.; Griswold, J.; Mozingo, D. The use of a collagen sponge/living cell composite material to treat donor sites in burn patients. Burns 2003, 29, 837–841. [Google Scholar] [CrossRef]
- Böttcher-Haberzeth, S.; Kapoor, S.; Meuli, M.; Neuhaus, K.; Biedermann, T.; Reichmann, E.; Schiestl, C. Osmotic expanders in children: No filling—No control—No problem? Eur. J. Pediatr. Surg. 2011, 21, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Schiestl, C.; Neuhaus, K.; Biedermann, T.; Böttcher-Haberzeth, S.; Reichmann, E.; Meuli, M. Novel Treatment for Massive Lower Extremity Avulsion Injuries in Children: Slow, but Effective with Good Cosmesis. Eur. J. Pediatr. Surg. 2010, 21, 106–110. [Google Scholar] [CrossRef]
- Schiestl, C.; Stiefel, D.; Meuli, M. Giant naevus, giant excision, eleg (i) ant closure? Reconstructive surgery with Integra Artificial Skin® to treat giant congenital melanocytic naevi in children. J. Plast. Reconstr. Aesthet. Surg. 2010, 63, 610–615. [Google Scholar] [CrossRef]
- Klar, A.S.; Biedermann, T.; Michalak, K.; Michalczyk, T.; Meuli-Simmen, C.; Scherberich, A.; Meuli, M.; Reichmann, E. Human Adipose Mesenchymal Cells Inhibit Melanocyte Differentiation and the Pigmentation of Human Skin via Increased Expression of TGF-β1. J. Invest. Dermatol. 2017, 137, 2560–2569. [Google Scholar] [CrossRef] [PubMed]
- Klar, A.S.; Zimoch, J.; Biedermann, T. Skin Tissue Engineering: Application of Adipose-Derived Stem Cells. BioMed Res. Int. 2017, 2017, 1–12. [Google Scholar] [CrossRef]
- Boyce, S.T.; Hansbrough, J.F. Biologic attachment, growth, and differentiation of cultured human epidermal keratinocytes on a graftable collagen and chondroitin-6-sulfate substrate. Surgery 1988, 103, 421–431. [Google Scholar]
- Boyce, S.T.; Greenhalgh, D.G.; Kagan, R.J.; Housinger, T.; Sorrell, J.M.; Childress, C.P.; Rieman, M.; Warden, G.D. Skin Anatomy and Antigen Expression after Burn Wound Closure with Composite Grafts of Cultured Skin Cells and Biopolymers. Plast. Reconstr. Surg. 1993, 91, 632–641. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Klar, A.S.; Michalak, K.; Böttcher-Haberzeth, S.; Reichmann, E.; Meuli, M.; Biedermann, T. The expression pattern of keratin 24 in tissue-engineered dermo-epidermal human skin substitutes in an in vivo model. Pediatr. Surg. Int. 2018, 34, 237–244. [Google Scholar] [CrossRef]
- Hartmann-Fritsch, F.; Biedermann, T.; Braziulis, E.; Luginbuehl, J.; Pontiggia, L.; Böttcher-Haberzeth, S.; Van Kuppevelt, T.H.; Faraj, K.A.; Schiestl, C.; Meuli, M.; et al. Collagen hydrogels strengthened by biodegradable meshes are a basis for dermo-epidermal skin grafts intended to reconstitute human skin in a one-step surgical intervention. J. Tissue Eng. Regen. Med. 2012, 10, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Braziulis, E.; Diezi, M.; Biedermann, T.; Pontiggia, L.; Schmucki, M.; Hartmann-Fritsch, F.; Luginbühl, J.; Schiestl, C.; Meuli, M.; Reichmann, E. Modified Plastic Compression of Collagen Hydrogels Provides an Ideal Matrix for Clinically Applicable Skin Substitutes. Tissue Eng. Part C Methods 2012, 18, 464–474. [Google Scholar] [CrossRef]
- Kisiel, M.A.; Klar, A.S. Isolation and Culture of Human Dermal Fibroblasts. Methods Mol. Biol. 2019, 1993, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Klar, A.S.; Böttcher-Haberzeth, S.; Biedermann, T.; Kisiel, M.A.; Reichmann, E.; Meuli, M. Differential expression of granulocyte, macrophage, and hypoxia markers during early and late wound healing stages following transplantation of tissue-engineered skin substitutes of human origin. Pediatr. Surg. Int. 2014, 30, 1257–1264. [Google Scholar] [CrossRef]
- Micka, K.; Biedermann, T.; Reichmann, E.; Meuli, M.; Klar, A.S. Induction of angiogenic and inflammation-associated dermal biomarkers following acute UVB exposure on bio-engineered pigmented dermo-epidermal skin substitutes in vivo. Pediatr. Surg. Int. 2019, 35, 129–136. [Google Scholar] [CrossRef]
- Meuli, M.; Hartmann-Fritsch, F.; Hüging, M.; Marino, D.; Saglini, M.; Hynes, S.; Neuhaus, K.; Manuel, E.; Middelkoop, E.; Reichmann, E.; et al. A Cultured Autologous Dermo-epidermal Skin Substitute for Full-Thickness Skin Defects: A Phase I, Open, Prospective Clinical Trial in Children. Plast. Reconstr. Surg. 2019, 144, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Amirsadeghi, A.; Jafari, A.; Eggermont, L.J.; Hashemi, S.-S.; Bencherif, S.A.; Khorram, M. Vascularization strategies for skin tissue engineering. Biomater. Sci. 2020, 8, 4073–4094. [Google Scholar] [CrossRef] [PubMed]
- Dehkordi, A.N.; Babaheydari, F.M.; Chehelgerdi, M.; Dehkordi, S.R. Skin tissue engineering: Wound healing based on stem-cell-based therapeutic strategies. Stem Cell Res. Ther. 2019, 10, 1–20. [Google Scholar] [CrossRef]
- Nilforoushzadeh, M.A.; Sisakht, M.M.; Amirkhani, M.A.; Seifalian, A.M.; Banafshe, H.R.; Verdi, J.; Nouradini, M. Engineered skin graft with stromal vascular fraction cells encapsulated in fibrin-collagen hydrogel: A clinical study for diabetic wound healing. J. Tissue Eng. Regen. Med. 2020, 14, 424–440. [Google Scholar] [CrossRef]
- Güven, S.; Mehrkens, A.; Saxer, F.; Schaefer, D.J.; Martinetti, R.; Martin, I.; Scherberich, A. Engineering of large osteogenic grafts with rapid engraftment capacity using mesenchymal and endothelial progenitors from human adipose tissue. Biomaterials 2011, 32, 5801–5809. [Google Scholar] [CrossRef]
- Annabi, N.; Tamayol, A.; Uquillas, J.A.; Akbari, M.; Bertassoni, L.E.; Cha, C.; Camci-Unal, G.; Dokmeci, M.R.; Peppas, N.A.; Khademhosseini, A. 25th Anniversary Article: Rational Design and Applications of Hydrogels in Regenerative Medicine. Adv. Mater. 2013, 26, 85–124. [Google Scholar] [CrossRef]
- Rana, D.; Kumar, T.S.; Ramalingam, M. Cell-laden hydrogels for tissue engineering. J. Biomater. Tissue Eng. 2014, 4, 507–535. [Google Scholar] [CrossRef]
- Zimoch, J.; Padial, J.S.; Klar, A.S.; Vallmajo-Martin, Q.; Meuli, M.; Biedermann, T.; Wilson, C.J.; Rowan, A.; Reichmann, E. Polyisocyanopeptide hydrogels: A novel thermo-responsive hydrogel supporting pre-vascularization and the development of organotypic structures. Acta Biomater. 2018, 70, 129–139. [Google Scholar] [CrossRef]
- Tavakoli, S.; Kharaziha, M.; Nemati, S.; Kalateh, A. Nanocomposite hydrogel based on carrageenan-coated starch/cellulose nanofibers as a hemorrhage control material. Carbohydr. Polym. 2021, 251, 117013. [Google Scholar] [CrossRef]
- Tavakoli, S.; Kharaziha, M.; Kermanpur, A.; Mokhtari, H. Sprayable and injectable visible-light Kappa-carrageenan hydrogel for in-situ soft tissue engineering. Int. J. Biol. Macromol. 2019, 138, 590–601. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Marchant, R.; Hu, J. Biomimetic hydrogels as scaffolds for tissue-engineering applications. In Biomimetic Biomaterials: Structure and Applications; Woodhead Publishing: Cambridge, UK, 2013; pp. 238–275. [Google Scholar] [CrossRef]
- Kumacheva, E.; Prince, E. Design and applications of man-made biomimetic fibrillar hydrogels. Nat. Rev. Mater. 2019, 4, 99–115. [Google Scholar]
- Pham, C.; Greenwood, J.; Cleland, H.; Woodruff, P.; Maddern, G. Bioengineered skin substitutes for the management of burns: A systematic review. Burns 2007, 33, 946–957. [Google Scholar] [CrossRef]
- Kumar, R.J.; Kimble, R.M.; Boots, R.; Pegg, S.P. Treatment of partial-thickness burns: A prospective, randomized trial using TranscyteTM. ANZ J. Surg. 2004, 74, 622–626. [Google Scholar] [CrossRef]
- Sharma, Y.; Tiwari, A.; Hattori, S.; Terada, D.; Sharma, A.K.; Ramalingam, M.; Kobayashi, H. Fabrication of conducting electrospun nanofibers scaffold for three-dimensional cells culture. Int. J. Biol. Macromol. 2012, 51, 627–631. [Google Scholar] [CrossRef]
- Mokhtari, H.; Kharaziha, M.; Karimzadeh, F.; Tavakoli, S. An injectable mechanically robust hydrogel of Kappa-carrageenan-dopamine functionalized graphene oxide for promoting cell growth. Carbohydr. Polym. 2019, 214, 234–249. [Google Scholar] [CrossRef]
- El-Sherbiny, I.M.; Yacoub, M.H. Hydrogel scaffolds for tissue engineering: Progress and challenges. Glob. Cardiol. Sci. Pract. 2013, 2013, 316–342. [Google Scholar] [CrossRef]
- Park, J.H.; Chung, B.G.; Lee, W.G.; Kim, J.; Brigham, M.D.; Shim, J.; Lee, S.; Hwang, C.M.; Durmus, N.G.; Demirci, U.; et al. Microporous cell-laden hydrogels for engineered tissue constructs. Biotechnol. Bioeng. 2010, 106, 138–148. [Google Scholar] [CrossRef] [PubMed]
- Cordell, J.M.; Vogl, M.L.; Johnson, A.J.W. The influence of micropore size on the mechanical properties of bulk hydroxyapatite and hydroxyapatite scaffolds. J. Mech. Behav. Biomed. Mater. 2009, 2, 560–570. [Google Scholar] [CrossRef]
- Li, Q.; Mai, Y.-W. Biomaterials for Implants and Scaffolds. In Biomaterials Science and Engineering; Springer: Berlin, Germany, 2017; Volume 8, ISBN 978-3-662-53572-1. [Google Scholar]
- Klawitter, J.J.; Hulbert, S.F. Application of porous ceramics for the attachment of load bearing internal orthopedic applications. J. Biomed. Mater. Res. 1971, 5, 161–229. [Google Scholar] [CrossRef]
- Yeh, J.; Blumling, J.; Karp, J.M.; Gantz, J.; Chandawarkar, A.; Eng, G.; Iii, J.B.; Langer, R.; Khademhosseini, A. Micromolding of shape-controlled, harvestable cell-laden hydrogels. Biomaterials 2006, 27, 5391–5398. [Google Scholar] [CrossRef] [PubMed]
- Galperin, A.; Long, T.J.; Ratner, B.D. Degradable, Thermo-Sensitive Poly(N-isopropyl acrylamide)-Based Scaffolds with Controlled Porosity for Tissue Engineering Applications. Biomacromolecules 2010, 11, 2583–2592. [Google Scholar] [CrossRef]
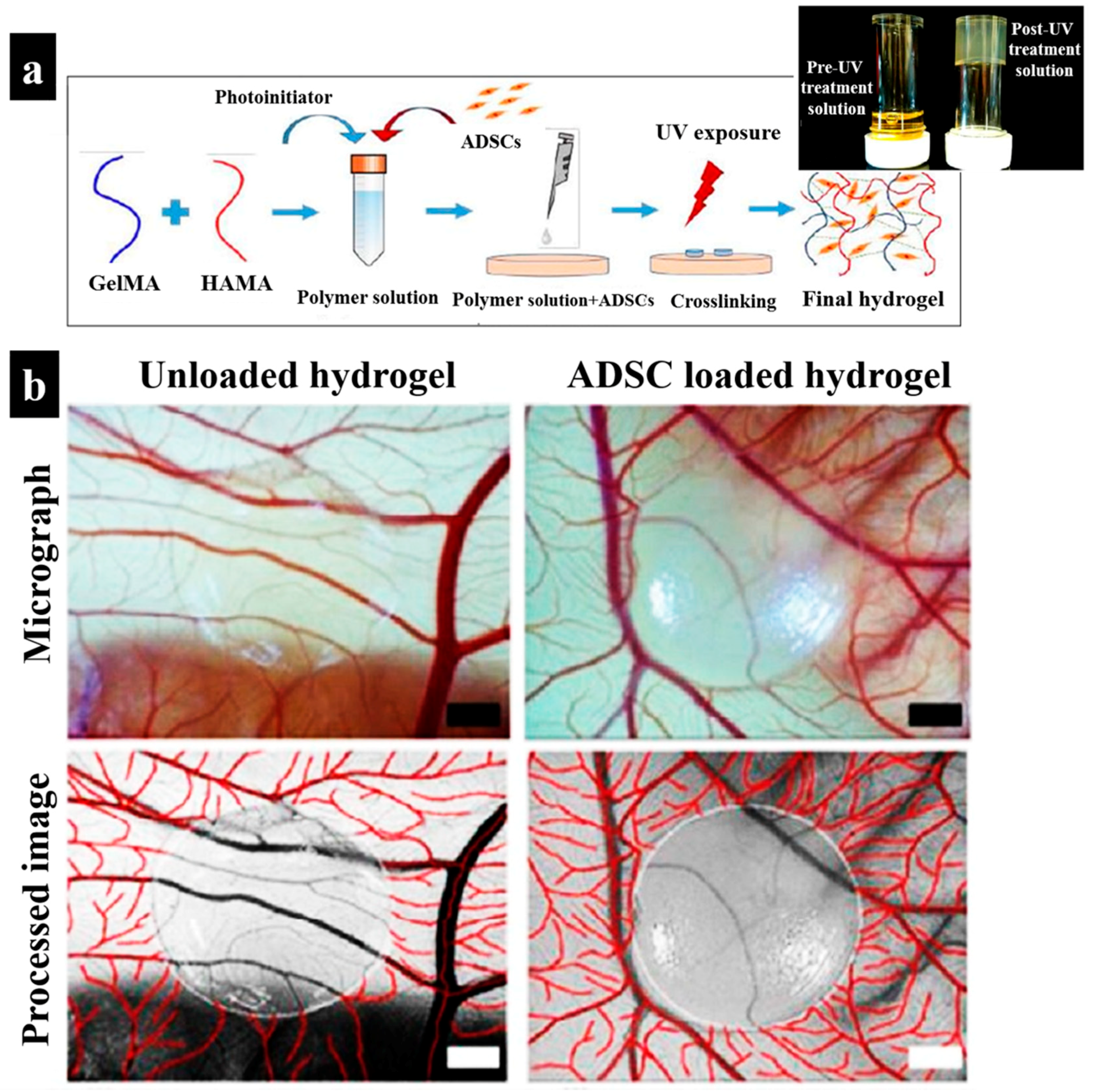
- Eke, G.; Mangir, N.; Hasirci, N.; MacNeil, S.; Hasirci, V. Development of a UV crosslinked biodegradable hydrogel containing adipose derived stem cells to promote vascularization for skin wounds and tissue engineering. Biomaterials 2017, 129, 188–198. [Google Scholar] [CrossRef]
- Pereira, R.F.; Sousa, A.; Barrias, C.C.; Bayat, A.; Granja, P.L.; Bártolo, P. Advances in bioprinted cell-laden hydrogels for skin tissue engineering. Biomanuf. Rev. 2017, 2, 1. [Google Scholar] [CrossRef]
- Murphy, S.V.; Atalaa, A. 3D bioprinting of tissues and organs. Nat. Biotechnol. 2014, 32, 773–785. [Google Scholar] [CrossRef]
- El-Serafi, A.T.; El-Serafi, I.T.; Elmasry, M.; Sjöberg, F. Skin regeneration in three dimensions, current status, challenges and opportunities. Differentiation 2017, 96, 26–29. [Google Scholar] [CrossRef]
- VijayaVenkataRaman, S.; Lu, W.F.; Fuh, J.Y.H. 3D bioprinting of skin: A state-of-the-art review on modelling, materials, and processes. Biofabrication 2016, 8, 032001. [Google Scholar] [CrossRef]
- Yu, J.R.; Navarro, J.; Coburn, J.C.; Mahadik, B.; Molnar, J.; Iv, J.H.H.; Nam, A.J.; Fisher, P.J.P. Current and Future Perspectives on Skin Tissue Engineering: Key Features of Biomedical Research, Translational Assessment, and Clinical Application. Adv. Healtc. Mater. 2019, 8, e1801471. [Google Scholar] [CrossRef]
- Li, H.; Tan, C.; Li, L. Review of 3D printable hydrogels and constructs. Mater. Des. 2018, 159, 20–38. [Google Scholar] [CrossRef]
- Huang, G.; Li, F.; Zhao, X.; Ma, Y.; Li, Y.; Min, L.; Jin, G.; Lu, T.J.; Genin, G.M.; Xu, F. Functional and Biomimetic Materials for Engineering of the Three-Dimensional Cell Microenvironment. Chem. Rev. 2017, 117, 12764–12850. [Google Scholar] [CrossRef] [PubMed]
- Cubo, N.; Garcia, M.; Del Cañizo, J.F.; Velasco, D.; Jorcano, J.L. 3D bioprinting of functional human skin: Production and in vivo analysis. Biofabrication 2016, 9, 015006. [Google Scholar] [CrossRef] [PubMed]
- Yanez, M.; Rincon, J.; Dones, A.; De Maria, C.; Gonzales, R.; Boland, T. In Vivo Assessment of Printed Microvasculature in a Bilayer Skin Graft to Treat Full-Thickness Wounds. Tissue Eng. Part A 2015, 21, 224–233. [Google Scholar] [CrossRef]
- Hakimi, N.; Cheng, R.; Leng, L.; Sotoudehfar, M.; Ba, P.Q.; Bakhtyar, N.; Amini-Nik, S.; Jeschke, M.G.; Günther, A. Handheld skin printer: In situ formation of planar biomaterials and tissues. Lab Chip 2018, 18, 1440–1451. [Google Scholar] [CrossRef]
- Liu, P.; Shen, H.; Zhi, Y.; Si, J.; Shi, J.; Guo, L.; Shen, S.G. 3D bioprinting and in vitro study of bilayered membranous construct with human cells-laden alginate/gelatin composite hydrogels. Coll. Surf. B Biointerfaces 2019, 181, 1026–1034. [Google Scholar] [CrossRef]
- Albanna, M.; Binder, K.W.; Murphy, S.V.; Kim, J.; Qasem, S.A.; Zhao, W.; Tan, J.; El-Amin, I.B.; Dice, D.D.; Marco, J.; et al. In Situ Bioprinting of Autologous Skin Cells Accelerates Wound Healing of Extensive Excisional Full-Thickness Wounds. Sci. Rep. 2019, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Hafezi, F.; Scoutaris, N.; Douroumis, D.; Boateng, J.S. 3D printed chitosan dressing crosslinked with genipin for potential healing of chronic wounds. Int. J. Pharm. 2019, 560, 406–415. [Google Scholar] [CrossRef]
- Admane, P.; Gupta, A.C.; Jois, P.; Roy, S.; Lakshmanan, C.C.; Kalsi, G.; Bandyopadhyay, B.; Ghosh, S. Direct 3D bioprinted full-thickness skin constructs recapitulate regulatory signaling pathways and physiology of human skin. Bioprinting 2019, 15, e00051. [Google Scholar] [CrossRef]
- Chinga-Carrasco, G.; Ehman, N.V.; Filgueira, D.; Johansson, J.; Vallejos, M.E.; Felissia, F.E.; Area, M.C. Bagasse—A major agro-industrial residue as potential resource for nanocellulose inks for 3D printing of wound dressing devices. Addit. Manuf. 2019, 28, 267–274. [Google Scholar] [CrossRef]
- Intini, C.; Elviri, L.; Cabral, J.; Mros, S.; Bergonzi, C.; Bianchera, A.; Flammini, L.; Govoni, P.; Barocelli, E.; McConnell, M. 3D-printed chitosan-based scaffolds: An in vitro study of human skin cell growth and an in-vivo wound healing evaluation in experimental diabetes in rats. Carbohydr. Polym. 2018, 199, 593–602. [Google Scholar] [CrossRef] [PubMed]
- Seol, Y.J.; Lee, H.; Copus, J.S.; Kang, H.W.; Cho, D.W.; Atala, A.; Lee, S.J.; Yoo, J.J. 3D bioprinted biomask for facial skin reconstruction. Bioprinting 2018, 10, e00028. [Google Scholar] [CrossRef] [PubMed]
- Si, H.; Xing, T.; Ding, Y.; Zhang, H.; Yin, R.; Zhang, W. 3D bioprinting of the sustained drug release wound dressing with double-crosslinked hyaluronic-acid-based hydrogels. Polymers 2019, 11, 1584. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tavakoli, S.; Klar, A.S. Bioengineered Skin Substitutes: Advances and Future Trends. Appl. Sci. 2021, 11, 1493. https://doi.org/10.3390/app11041493
Tavakoli S, Klar AS. Bioengineered Skin Substitutes: Advances and Future Trends. Applied Sciences. 2021; 11(4):1493. https://doi.org/10.3390/app11041493
Chicago/Turabian StyleTavakoli, Shima, and Agnes S. Klar. 2021. "Bioengineered Skin Substitutes: Advances and Future Trends" Applied Sciences 11, no. 4: 1493. https://doi.org/10.3390/app11041493
APA StyleTavakoli, S., & Klar, A. S. (2021). Bioengineered Skin Substitutes: Advances and Future Trends. Applied Sciences, 11(4), 1493. https://doi.org/10.3390/app11041493






