Effectiveness of Mouth Rinsing versus Ingesting Pickle Juice for Alleviating Electrically Induced Cramp in Physically Active Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.3. Experimental Procedures
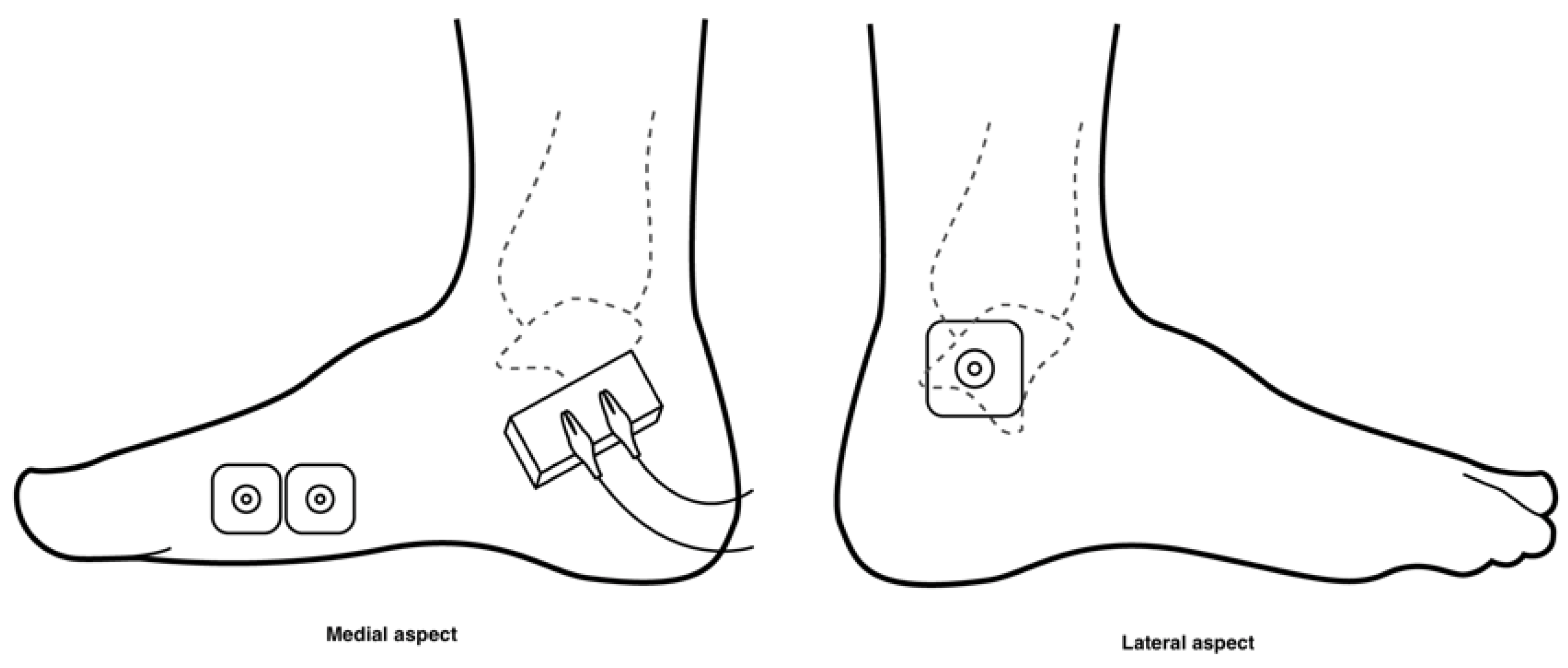
2.3.1. Familiarisation Session
2.3.2. Experimental Sessions
2.4. Statistical Analysis
3. Results
3.1. Urine Specific Gravity
3.2. Cramp Duration
3.3. Severity and Duration of Perceived Cramp Discomfort (VAS)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schwellnus, M.P.; Derman, E.W.; Noakes, T.D. Aetiology of skeletal muscle ‘cramps’ during exercise: A novel hypothesis. J. Sports Sci. 1997, 15, 277–285. [Google Scholar] [CrossRef]
- Swash, M.; Czesnik, D.; De Carvalho, M. Muscular cramp: Causes and management. Eur. J. Neurol. 2018, 26, 214–221. [Google Scholar] [CrossRef]
- Miller, K.C.; Stone, M.S.; Huxel, K.C.; Edwards, J.E. Exercise-Associated Muscle Cramps. Sports Health A Multidiscip. Approach 2010, 2, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Minetto, M.A.; Holobar, A.; Botter, A.; Farina, D. Origin and Development of Muscle Cramps. Exerc. Sport Sci. Rev. 2013, 41, 3–10. [Google Scholar] [CrossRef]
- Hoffman, M.D.; Stuempfle, K.J. Muscle Cramping during a 161-km Ultramarathon: Comparison of Characteristics of Those with and without Cramping. Sports Med.-Open 2015, 1, 24. [Google Scholar] [CrossRef]
- Martínez-Navarro, I.; Montoya-Vieco, A.; Collado-Boira, E.; Hernando, B.; Panizo, N.; Hernando, C. Muscle Cramping in the Marathon. J. Strength Cond. Res. 2020, 37, 364–367. [Google Scholar] [CrossRef] [PubMed]
- Schwellnus, M.P. Cause of Exercise Associated Muscle Cramps (EAMC)-altered neuromuscular control, dehydration or electrolyte depletion? Br. J. Sports Med. 2008, 43, 401–408. [Google Scholar] [CrossRef]
- Edouard, P. Exercise associated muscle cramps: Discussion on causes, prevention and treatment. Sci. Sports 2014, 29, 299–305. [Google Scholar] [CrossRef]
- Jahic, D.; Begic, E. Exercise-Associated Muscle Cramp-Doubts About the Cause. Mater. Soc.-Med. 2018, 30, 67–69. [Google Scholar] [CrossRef] [PubMed]
- Maughan, R.J.; Shirreffs, S.M. Muscle Cramping During Exercise: Causes, Solutions, and Questions Remaining. Sports Med. 2019, 49, 115–124. [Google Scholar] [CrossRef]
- Swash, M.; De Carvalho, M. Testing electrolyte supplementation for muscle cramp. Muscle Nerve 2019, 60, 499–500. [Google Scholar] [CrossRef]
- Schwellnus, P.M.P. Muscle Cramping in the Marathon: Aetiology and risk factors. Sports Med. 2007, 37, 364–367. [Google Scholar] [CrossRef]
- Schwellnus, M.P.; Drew, N.; Collins, M. Increased running speed and previous cramps rather than dehydration or serum sodium changes predict exercise-associated muscle cramping: A prospective cohort study in 210 Ironman triathletes. Br. J. Sports Med. 2011, 45, 650–656. [Google Scholar] [CrossRef] [PubMed]
- Nelson, N.L.; Churilla, J.R. A narrative review of exercise-associated muscle cramps: Factors that contribute to neuromuscular fatigue and management implications. Muscle Nerve 2016, 54, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Giuriato, G.; Pedrinolla, A.; Schena, F.; Venturelli, M. Muscle cramps: A comparison of the two-leading hypothesis. J. Electromyogr. Kinesiol. 2018, 41, 89–95. [Google Scholar] [CrossRef]
- Panza, G.; Stadler, J.; Murray, D.; Lerma, N.; Barrett, T.; Pettit-Mee, R.; Edwards, J.E. Acute Passive Static Stretching and Cramp Threshold Frequency. J. Athl. Train. 2017, 52, 918–924. [Google Scholar] [CrossRef]
- Miller, K.C.; Mack, G.W.; Knight, K.L.; Hopkins, J.T.; Draper, D.O.; Fields, P.J.; Hunter, I. Reflex Inhibition of Electrically Induced Muscle Cramps in Hypohydrated Humans. Med. Sci. Sports Exerc. 2010, 42, 953–961. [Google Scholar] [CrossRef]
- Simon, S.A.; De Araujo, I.E.; Gutierrez, R.; Nicolelis, M.A.L. The neural mechanisms of gustation: A distributed processing code. Nat. Rev. Neurosci. 2006, 7, 890–901. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Berdugo, D.; Rofes, L.; Farre, R.; Casamitjana, J.F.; Enrique, A.; Chamizo, J.M.G.; Padrón, A.; Navarro, X.; Clave, P. Localization and expression of TRPV1 and TRPA1 in the human oropharynx and larynx. Neurogastroenterol. Motil. 2015, 28, 91–100. [Google Scholar] [CrossRef]
- Chang, R.B.; Waters, H.; Liman, E.R. A proton current drives action potentials in genetically identified sour taste cells. Proc. Natl. Acad. Sci. USA 2010, 107, 22320–22325. [Google Scholar] [CrossRef]
- Huang, Y.A.; Maruyama, Y.; Stimac, R.; Roper, S.D. Presynaptic (Type III) cells in mouse taste buds sense sour (acid) taste. J. Physiol. 2008, 586, 2903–2912. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.Y.; Chang, R.B.; Allgood, S.; Silver, W.L.; Liman, E.R. A TRPA1-dependent mechanism for the pungent sensation of weak acids. J. Gen. Physiol. 2011, 137, 493–505. [Google Scholar] [CrossRef] [PubMed]
- Craighead, D.H.; Shank, S.W.; Gottschall, J.S.; Passe, D.H.; Murray, B.; Alexander, L.M.; Kenney, W.L. Ingestion of transient receptor potential channel agonists attenuates exercise-induced muscle cramps. Muscle Nerve 2017, 56, 379–385. [Google Scholar] [CrossRef]
- Mandadi, S.; Nakanishi, S.; Takashima, Y.; Dhaka, A.; Patapoutian, A.; McKemy, D.; Whelan, P. Locomotor networks are targets of modulation by sensory transient receptor potential vanilloid 1 and transient receptor potential melastatin 8 channels. Neuroscience 2009, 162, 1377–1397. [Google Scholar] [CrossRef]
- Ataide-Silva, T.; Souza, M.E.D.C.A.D.; De Amorim, J.F.; Stathis, C.G.; Leandro, C.G.; Lima-Silva, A.E. Can Carbohydrate Mouth Rinse Improve Performance during Exercise? A Systematic Review. Nutrients 2013, 6, 1–10. [Google Scholar] [CrossRef]
- Wickham, K.A.; Spriet, L.L. Administration of Caffeine in Alternate Forms. Sports Med. 2018, 48, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Fares, E.-J.M.; Kayser, B. Carbohydrate Mouth Rinse Effects on Exercise Capacity in Pre- and Postprandial States. J. Nutr. Metab. 2011, 2011, 385962. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, J.; Bottoms, L.; Flynn, C.; Bradley, E.; Alexander, G.; Mccullagh, S.; Finn, T.; Hurst, H.T. The effect of different durations of carbohydrate mouth rinse on cycling performance. Eur. J. Sport Sci. 2014, 14, 259–264. [Google Scholar] [CrossRef]
- Rennie, S. Electrophysical agents contraindications and precautions: An evidence-based approach to clinical decision making in physical therapy foreword. Physiother. Can. 2010, 62, 1–80. [Google Scholar] [CrossRef]
- Miller, K.C.; Knight, K.L.; Wilding, S.R.; Stone, M.B. Duration of electrically induced muscle cramp increased by increasing stimulation frequency. J. Sport Rehabil. 2012, 21, 182–185. [Google Scholar] [CrossRef]
- Ms, M.B.S.; Edwards, J.E.; Babington, J.P.; Ingersoll, C.D.; Ms, R.M.P. Reliability of an electrical method to induce muscle cramp. Muscle Nerve 2003, 27, 122–123. [Google Scholar] [CrossRef]
- Behringer, M.; Nowak, S.; Leyendecker, J.; Mester, J. Effects of TRPV1 and TRPA1 activators on the cramp threshold frequency: A randomized, double-blind placebo-controlled trial. Graefe’s Arch. Clin. Exp. Ophthalmol. 2017, 117, 1641–1647. [Google Scholar] [CrossRef]
- Tanner, R.K.; Gore, C.J. Physiological Tests for Elite Athletes, 2nd ed.; Australian Institute of Sport: Champaign, IL, USA, 2013. [Google Scholar]
- Ohno, M.; Lavender, A.P.; Sawai, A. Heat-induced Body Fluid Loss Causes Muscle Cramp during Maximal Voluntary Contraction for the Knee Flexors. Int. J. Sport Health Sci. 2018, 16, 191–199. [Google Scholar] [CrossRef][Green Version]
- Marosek, S.E.H.; Antharam, V.; Dowlatshahi, K. Quantitative Analysis of the Acetic Acid Content in Substances Used by Athletes for the Possible Prevention and Alleviation of Exercise-Associated Muscle Cramps. J. Strength Cond. Res. 2020, 34, 1539–1546. [Google Scholar] [CrossRef] [PubMed]
- Craighead, D.H.; Shank, S.; Alexander, L.M.; Kenney, W.L. Orally ingested transient receptor potential (TRP) channel activators attenuate the intensity-duration of voluntarily induced muscle cramps in humans. Faseb. J. 2016, 30, lb706. [Google Scholar] [CrossRef]
- Earp, J.E.; Stearns, R.L.; Stranieri, A.; Agostinucci, J.; Lepley, A.S.; Matson, T.; Ward-Ritacco, C.L. Electrolyte beverage consumption alters electrically induced cramping threshold. Muscle Nerve 2019, 60, 598–603. [Google Scholar] [CrossRef]
- Miller, K.C. Myths and Misconceptions About Exercise-Associated Muscle Cramping. ACSM’s Health Fit. J. 2016, 20, 37–39. [Google Scholar] [CrossRef]
- Manning, C.D.; McDonald, P.L.L.; Murnaghan, C.D.; Bawa, P. Reciprocal inhibition versus unloading response during stretch reflex in humans. Exp. Brain Res. 2013, 226, 33–43. [Google Scholar] [CrossRef]

| Condition | Median Point Estimate | Median 95% Confidence Interval | |
|---|---|---|---|
| Lower Bound | Upper Bound | ||
| PJI | 82.81 | 54.13 | 111.50 |
| PJMR | 68.56 | 0.00 | 162.21 |
| Water | 100.000 | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Georgieva, J.; Brade, C.J.; Ducker, K.J.; Davey, P.; Jacques, A.; Ohno, M.; Lavender, A.P. Effectiveness of Mouth Rinsing versus Ingesting Pickle Juice for Alleviating Electrically Induced Cramp in Physically Active Adults. Appl. Sci. 2021, 11, 12096. https://doi.org/10.3390/app112412096
Georgieva J, Brade CJ, Ducker KJ, Davey P, Jacques A, Ohno M, Lavender AP. Effectiveness of Mouth Rinsing versus Ingesting Pickle Juice for Alleviating Electrically Induced Cramp in Physically Active Adults. Applied Sciences. 2021; 11(24):12096. https://doi.org/10.3390/app112412096
Chicago/Turabian StyleGeorgieva, Julia, Carly J. Brade, Kagan J. Ducker, Paul Davey, Angela Jacques, Masato Ohno, and Andrew P. Lavender. 2021. "Effectiveness of Mouth Rinsing versus Ingesting Pickle Juice for Alleviating Electrically Induced Cramp in Physically Active Adults" Applied Sciences 11, no. 24: 12096. https://doi.org/10.3390/app112412096
APA StyleGeorgieva, J., Brade, C. J., Ducker, K. J., Davey, P., Jacques, A., Ohno, M., & Lavender, A. P. (2021). Effectiveness of Mouth Rinsing versus Ingesting Pickle Juice for Alleviating Electrically Induced Cramp in Physically Active Adults. Applied Sciences, 11(24), 12096. https://doi.org/10.3390/app112412096







