Featured Application
This study provides valuable information on the Syzygium formosum as a new cosmeceutical ingredient.
Abstract
Syzygium formosum (SF) leaves have long been used in traditional medicine for the treatment of skin-related diseases such as atopic dermatitis and psoriasis. To understand the underpinning pharmacological mechanism of skin relief functionality, the quantitative profile of phytochemicals in the leaf extract has been performed. Twenty phytochemical components including eight flavonoids, three phenolic acids, and nine triterpenoids were identified and quantitatively analyzed from the SF leaves. The dominant flavonoids of the SF leaves were found to be catechin and myricetin-3-O-rhamnoside. Gallic acid was the most abundant phenolic acid. Among triterpenoids, asiatic acid, corosolic acid, and betulinic acid exhibited a high concentration ranging 2.78–8.59, 3.19–3.91, and 3.58–6.14 mg/g dry leaf, respectively. The total triterpenoid contents were 14.18–19.00 mg/g dry leaf which were 3-fold higher than those in the Indonesian Centella asiatica leaves. In human keratinocytes assay, industrial scale extract of SF significantly reduced expressions of proinflammatory cytokines and cyclooxygenase-2. Furthermore, that SF extract exhibited very strong antibacterial activity on Staphylococcus aureus and Cutibacterium acnes that possibly induce serious skin problems. Our results proved that S. formosum leaf extract with high content of biologically active compounds could be a superior bioactive ingredient in cosmetic and pharmaceutical products.
1. Introduction
Syzygium formosum (Wall.) Masam, a medicinal plant belonging to the Myrtaceae family, is widely found in South Asian countries including Vietnam, Thailand, Cambodia, India, and Bangladesh [1]. In Vietnam, Syzygium formosum is mainly distributed in the Northern region and has been used as a traditional medicinal material for the treatment of skin-related diseases such as furuncle, scabies, itching, rashes, atopic dermatitis, psoriasis, and some other respiratory diseases like sore throat and bronchitis [2]. S. formosum leaves possess the biological functionalities including antibacterial [3,4], antioxidative [3,5], anti-allergic, and anti-inflammatory activities [6]. A variety of bioactive compounds, particularly flavonoids, phenolic acids, and triterpenoids have been reported to exhibit remarkable pharmacological and biological properties of plants [7,8,9]. Recent studies have revealed that S. formosum leaves contain those bioactive secondary metabolites, including phenolics and triterpenoids [6,10].
Triterpenoids are a class of terpenes formed by the polymerization of isoprene unit as a monomer. Triterpenoids are biosynthesized by the cyclization of their precursor squalene and can be grouped into amyrin, lupeol, and lanosterol based on their chemical structures. Triterpenoids have been found in the traditional oriental herbal medicine. Among the biofunctional phytochemicals, triterpenoids are currently identified as promising pharmaceutical and cosmeceutical ingredients. It has been reported that they exhibit strong biological activities such as anti-oxidation, anti-inflammation, anti-allergy, or anticancer [11]. For instance, ginsenosides, which are famous and well-studied phytochemicals from Ginseng, are also a glycoconjugated triterpenoid modified from β-amyrin or dammarenediol. As another example, Alisma orientale has been used for a long time in China to treat edema, leukorrhea, diarrhea, and dizziness. The phytochemicals identified as characteristic constituents in A. orientale are terpenoids including protostane triterpenoids and guaiane sesquiterpenoids. These active constituents exhibit a wide range of pharmacological functionalities such as diuretic, anti-urolithiatic, antinephritic, anti-atherosclerotic, immunomodulatory, and hepatoprotective activities [12].
Centella asiatica (L.) Urban is also a famous medicinal plant, the use of which is not limited to a functional food but has also been extended to the pharmaceuticals. C. asiatica has been used in traditional medicine for hundreds of years in a wide range of Asian countries to treat various diseases such as neurological, endocrine, cardiovascular, gastrointestinal, immune, gynecological, and particularly dermatological symptoms such as skin smoothing or wound healing, and to act as a brain tonic [13,14,15]. Among phytochemicals, triterpenoids are biologically active substances in C. asiatica. Asiaticoside and madecassoside are triterpenoids which are responsible for its pharmacological activities [16,17]. Indeed, recent studies have reported that the methanolic extracts of triterpenoids from Centella asiatica exhibited anti-inflammation activity and therapeutic potential on some skin diseases such as wounds, atopic dermatitis, and acne [14,18,19]. Due to the beneficial effects on various conditions, particularly skin diseases like atopic dermatitis, C. asiatica has been used as a good source for skin therapy and cosmetic formulation. Currently, it has been consumed as a commercial cosmeceutical ingredient [14,20,21].
Profiling the phytochemicals in medicinal plants has attracted widespread interest and promoted a much better understanding of traditional herbal medicines in recent years. Medicinal plants contain diverse phytochemicals including tannins, glycosides, saponins, alkaloids, flavonoids, and triterpenoids which have shown the biofunctionalities. Tannins help in wound healing and inflamed mucosal membranes [22]. Glycosides have purgative qualities and are used for the treatment of skin diseases and as anticancer agents [23]. Saponins, alkaloids, and flavonoids have beneficial effects on neurological disorders and oxidative stress [24].
Despite the historical applications of S. formosum leaves in traditional Vietnamese medicine, little has been reported in in-depth studies of the SF leaves and their phytochemical components effective for medicinal use in Vietnam. For a better understanding of the underpinning pharmacological mechanism of skin relief functionality of S. formosum leaves, in this study, the comprehensive profiling of phytochemical components in the SF leaves has been performed. We categorized the phytochemicals in three abundant groups that include the flavonoids, phenolics, and triterpenoids, then quantified each component by the multiple reaction monitoring (MRM) using triple quadrupole mass spectrometry. Additionally, comparative analyses of phytochemicals in S. formosum and C. asiatica leaves as well as their biological functionalities on the human keratinocytes and antimicrobial activity were performed.
2. Materials and Methods
2.1. Chemicals
The standards including asiatic acid, corosolic acid, betulin, betulinic acid, and oleanolic acid were purchased from Sigma-Aldrich (St. Louis, MO, USA). Madecassic acid was purchased from Toronto Research Chemicals (Ontario, ON, Canada). Hederagenin and ursolic acid were obtained from Tokyo Chemical Industry (Tokyo, Japan). Maslinic acid, gallic acid, 3,4-dihydrobenzoic acid (3,4-DHBA), and p-coumaric acid were obtained from Chengdu Biopurify Phytochemicals Ltd. (Chengdu, China). Catechin, epigallocatechin gallate (EGCG), myricetin-3-O-glucoside, myricetin-3-O-rhamnoside, quercetin-3-O-glucoside, and quercetin-3-O-rhamnoside were obtained from Extrasynthese SAS (Genay Cedex, France). Quercetin and quercetin-3-O-arabinoside were purchased from ChemFaces Biochemical Co., Ltd. (Wuhan, China). All standard compounds have a purity of ≥95%, except ursolic acid (purity >90%). HPLC-grade acetonitrile, methanol, and formic acid were purchased from Sigma-Aldrich.
Preparation of Standard Solutions
Each standard was prepared at a concentration of 1.0 mg/mL in methanol (stock solution). Then, standard mix series including triterpenoid, flavonoid, and phenolic acid series were prepared from the stock solutions and diluted to the appropriate range for calibration curves. All solutions were stored in a brown vial at −60 °C until analysis.
2.2. Plant Materials
The Syzygium formosum leaves were collected from Hanoi (December 2019) and Phu Tho provinces, a mid-land and mountainous province of the northwestern Vietnam (February 2020). The SF leaves were identified by Dr. Van Dao Nguyen (Faculty of Biotechnology, Hanoi Open University, Hanoi, Vietnam). The fresh leaves were dried by a hot air oven at 45 °C for 48 h, before delivering to Korea. The Centella asiatica dried leaves originated from Indonesia and were purchased from Jung Woo-dang (Seoul, Korea). The Centella asiatica dried leaves originated from Korea and were purchased from Nanastore (Seoul, Korea).
The dried leaves were crushed to the powder before extraction. Syzygium formosum and Centella asiatica powder were extracted four times by ethanol. Briefly, 50 g of dry leaves powder was first extracted with 600 mL of 70% ethanol for 12 h. After each incubation period, the mixtures were centrifuged at 700 rpm for 30 min. The supernatant was collected, and the residue was used for further extraction steps. Similarly, the second extraction was conducted with 600 mL of 100% ethanol for 12 h. The third and fourth extractions were done with 200 mL of 100% ethanol for 4 h. All four extraction steps were performed at 50 °C. Finally, all extracts were combined and lyophilized to yield fine powder.
2.3. Sample Purification
Samples were purified by solid-phase extraction (SPE) before LC-MS/MS analysis. A 1 mg of the extracted powder was suspended in 1 mL of methanol and diluted ten times in 10% methanol. The flavonoids were purified by using a C18 cartridge (200 mg, 3 mL, Waters). The triterpenoid and phenolic acid were extracted by Oasis HLB Cartridge (30 mg, 6 mL, Waters, Millford, MA, USA). After SPE, the samples were evaporated and reconstituted in 50% methanol before analysis by LC-MS/MS.
2.4. LC-MS/MS Analysis
Analysis of all compounds was performed by using ultraperformance liquid chromatography tandem mass spectrometry (6470 Triple Quadrupole System, Agilent, Palo Alto, CA, USA). The MRM parameters and MS/MS transitions are shown in Supplementary Table S1.
For flavonoids and phenolic acids, the separation was performed using an Agilent Zorbax Eclipse Plus C18 column (2.1 × 50 mm, 1.8 µm) and the column temperature was maintained at 40 °C. The mobile phases contained 0.1% (v/v) formic acid in distilled water (solvent A) and 0.1% (v/v) formic acid in ACN (solvent B). The flow rate was 0.3 mL/min.
The gradients between the time points were set up for flavonoids separation as follows: 0–5 min, 0–15% B; 5–12 min, 15–40% B; 12–13 min, 40–100% B; 13.5–20 min, 100% B; and re-equilibration 0% B for from 21–27 min. For phenolic acids, the gradients between the time points were as follows: 0–2 min, 0–5% B; 2–10 min, 5–25% B; 10–13 min, 25–75% B; 13–14 min, 75–95% B; 95% B was maintained in 7 min followed by an 8-min re-equilibration.
To quantify triterpenoids, the Agilent Zorbax Eclipse Rapid Resolution High Definition C18 column (2.1 × 100 mm, 1.8 µm) was used. The mobile phases consisted of solvent 5 mM of ammonium formate in distilled water (solvent A) and a mixture of methanol: IPA (95:5), containing 5 mM of ammonium formate (solvent B). The gradient elution program for triterpenoid separation was as follows: 0–1 min, 70% B; 1–3 min, 80% B; 3–8 min, 82% B; 8–20 min, 84% B; 20.5–25.5 min, 100% B; and re-equilibration to 70% B for 10 min. The injection volume was 2 μL, the mobile phase flow rate was 0.2 mL/min, and the column temperature was stabilized at 40 °C.
2.5. Method Validation
The evaluation of linearity, the limit of detection (LOD), the limit of quantification (LOQ), and recovery of the target compounds were performed to validate the reliability of the quantitative analysis. The linearity was demonstrated by the calibration curve of analytes, which was expressed by plotting the peak area versus the concentration of standard using a linear regression model. The LOD and LOQ were calculated using peaks with signal to noise (S/N) ratios of 3.3 and 10.0 as the following formula: standard deviation/slope × 3.3 or 10.0.
The SPE recovery was performed by employing the standard addition method, i.e., using a known concentration of standards added into the sample to evaluate matrix effect on the recovery and the accuracy of the analytical method. The standard solutions at low, medium, and high concentrations were used, and recovery was calculated by the following formula: (Concentration of spiked sample − concentration of non-spiked sample)/Concentration of spiked blank × 100%.
2.6. Anti-Inflammation Assay Using HaCaT Cells
The HaCaT cell line was kindly provided from the Department of Dermatology, College of Medicine, Chungnam National University, Korea. S. formosum leaf extract prepared in an industrial scale (50 kg dry leaves) was compared with a commercial extract of C. asiatica produced in Korea. The HaCaT cell (2 mL) culture with 0.5 × 106 cells/mL was seeded into a 6-well plate and incubated overnight. The extracts of S. formosum and C. asiatica were added into the cell culture as a final concentration of 15 μg/mL, then the cells were incubated 6 h more. One milliliter of Dulbecco’s phosphate buffer saline (DPBS, WELGENE, Daegu, Korea) was added after removing the culture medium. Ultraviolet-B was radiated on the HaCaT cells with the intensity of 20 mJ/cm2 for 30 s. DPBS was replaced with serum-free Dulbecco’s Modified Eagle’s Medium (Gibco, Waltham, MA, USA), then the cells were further incubated for 18 h. The cell culture was conducted with 5% CO2 at 37 °C.
After harvesting the cultured cells, total RNA was extracted with AccuPrep® Universal RNA Extraction Kit (Bioneer, Daejeon, Korea), and cDNA library was prepared with reverse transcription mix (5× Master Premix, Elpis-Biotech, Daejeon, Korea). For the PCR, actin was used as a house keeping gene to compare relative mRNA levels of proinflammatory genes. Sequences of PCR primers of IL-1β, IL-6, IL-8, COX-2, and a housekeeping gene, actin, are given in Table 1.

Table 1.
Sequences of PCR primers of IL-1β, IL-6, IL-8, COX-2, and a housekeeping gene, actin.
2.7. Antibacterial Effect of Syzygium formosum Leaves
The industrial scale extract of S. formosum leaves was used for the antibacterial activity assays.
2.7.1. Antibacterial Assay on Staphylococcus Species
S. aureus (ATCC 25923) and S. epidermidis (KCTC 1917) were used to determine the antibacterial activity. Stock cultures of strains were stored in frozen medium at −70 °C. Strains were firstly seeded into a fresh Cation adjusted Mueller–Hinton broth (CAMHB) and incubated at 37 °C with shaking 200 rpm for 12 h.
The antibacterial agent (mupirocin) and extracts were dissolved in the dimethyl sulfoxide (DMSO, Junsei). CAMHB (1600 μL) and 200 μL of antibacterial agent or extract were dispensed into the 24-well plate. Bacterial solution (200 μL), which was adjusted the concentration to OD595 = 0.4–0.6, was added to the mixed solution. The final concentration of DMSO was controlled to 5%. The 24-well plate was incubated at 37 °C with shaking 180 rpm. Bacterial growth inhibitory effect was monitored by measuring the OD595 every 2 h. Negative control was wells containing media adjusted 5% of DMSO concentration and bacterial suspension. Positive control was mupirocin. Minimum inhibitory concentration, MIC50 values were determined the concentrations of antibacterial agent or the extract that inhibit 50% of the strains.
2.7.2. Antibacterial Assay on Cutibacterium acnes
Cutibacterium acnes used for culturing was stored at −80 °C. For activation, C. acnes was cultured on BHI (Brain Heart Infusion, Bifco) agar in anaerobic chamber at 37 °C for 48 h. A single colony of C. acnes was inoculated on BHI broth for the preculture allowing to grow cell OD (optical density) up to 1.5. The C. acnes in the BHI broth were diluted from 101 to 108 for the inoculation on the colony counting plate.
To identify the antibacterial effect of S. formosum leaf extract on C. acnes, 3 types of modifications of the BHI media were prepared. A negative control regular BHI media has been used. Modified BHI with 1% (v/v) of 70% ethanol and 0.05% (w/v) S. formosum leaf extract by 70% ethanol was used for the growth inhibition study. Plates were prepared in triplicates for both the controls and the S. formosum test. One-hundred microliters of preculture containing logarithmically growing C. acnes was spread onto each plate. All media were incubated in the 37 °C anaerobic chamber for 24 h, then the colonies were counted.
2.8. Data Analysis
The MS data were analyzed using Agilent Mass Hunter Quantitative Analysis, version B.07.01. SP1 (Agilent). The data were expressed as a mean ± standard deviation. All statistical analyses were performed using IBM SPSS statistics 24 (IBM, Armonk, NY, USA).
3. Results
3.1. Phytochemical Profiling of Syzygium formosum Leaves Using LC-MS/MS
The phytochemicals in S. formosum leaves were tentatively identified based on the retention time and mass to charge (m/z) ratio using both positive and negative ionization full scan modes. In the scan mode, we can find that glycoconjugates of myricetin and quercetin were the main flavonoids detected in the SF leaves extract. Meanwhile, phenolic acid was primarily exhibited to be gallic acid. Similarly, some triterpenoid aglycons were predicted to be present in this material. For further confirmation, reference standards corresponding to expected compounds were purchased and simultaneously analyzed.
In a mass spectroscopic analysis, positive-ion mode has been used for the analysis of flavonoid and triterpenoid demonstration as more intense signals were achieved as the protonated molecular ions, [M+H]+; in contrast, the negative-ion mode, [M−H]−, was used for phenolic acids due to its fewer signals. The fragmentation of the expected compounds was conducted by product ion scan and the major fragment ions were selected based on their high intensity and were used under the multiple reaction monitoring (MRM) condition for quantitative analysis. Table 2 summarized the parameters of the compounds identified in S. formosum leaves. Twenty components found in the SF leaf extract including eight flavonoids, three phenolic acids, and nine triterpenoids were confirmed based on a comparison of reference standards and MS/MS analysis (Figure 1, Supplementary Figures S1 and S2). MS/MS fragmentation of phenolic components and triterpenoids is presented in Supplementary Figure S3.

Table 2.
List of phenolic compounds and triterpenoids detected in Syzygium formosum leaves by UPLC-MS/MS.
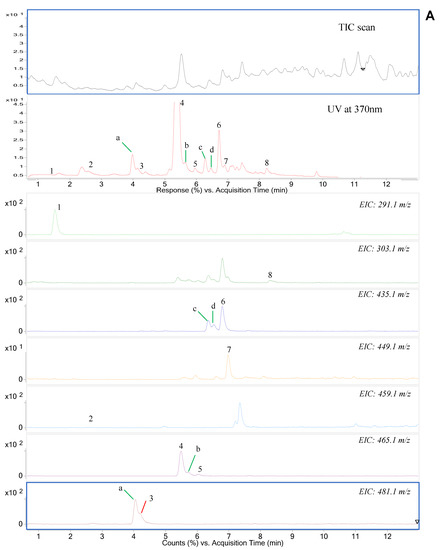
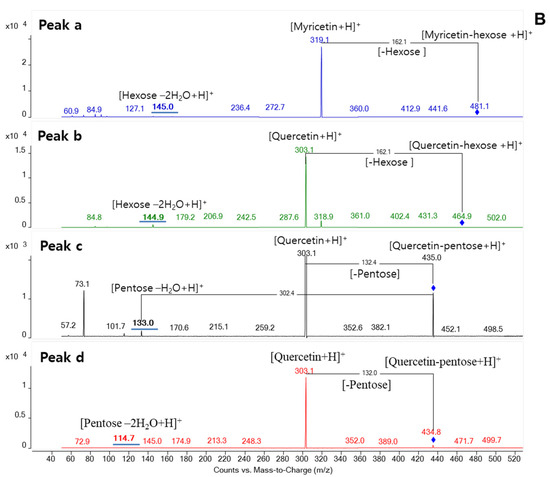
Figure 1.
(A) The total ion (TIC), UV, and extracted-ion (EIC) chromatogram in the positive scan mode, (B) The product ion scan (PIS) chromatogram of flavonoids in Syzygium formosum leaves (1-catechin, 2-EGCG, 3-myricetin glucoside, 4-myricetin rhamnoside, 5-muercetin glucoside, 6-muercetin arabinoside, 7-muercetin rhamnoside, 8-quercetin; peaks from a–d: unknown flavonoids).
We used both UV spectrometric and mass spectrometric methods to determine the phytochemicals in the SF leaves. To determine the major components of potential flavonoids and phenolics, a UV detector which coupled with mass spectrometry has been used at the wavelength of 370 nm. Then, the components were primarily identified by the molecular ion mass and fragmentation pattern at positive mode as well as the comparison of retention time with the reference standard. For example, a positive molecular ion at m/z 291, and product ions at m/z 139 and 123, were initially determined as catechin or its isomer epicatechin, then we confirmed it to be catechin based on the reference standard.
Structural isomers could be determined by the MS/MS analysis without reference standards. By extraction of molecular ion mass of m/z 435.1, three independent peaks were found at different retention time (Figure 1A). Among three peaks, one with the retention time of 6.79 min was identified as quercetin-arabinose by reference standard. Showing the unique fragmentation pattern in MS/MS spectra, two isobaric components were determined to be a quercetin-pentose glycoconjugate (Figure 1B). Those possibly could be a linkage isomer of quercetin-arabinose or different glycoconjugate such as quercetin-xylose. Similarly, when extracting molecular ion mass of m/z 465.1, three peaks appeared at different time. Two among them with the retention time of 5.49 min and 6.02 min were confirmed to be myricetin-rhamnose and quercetin-glucose, respectively, based on a comparison of reference standards. By observing in MS/MS spectra, one remaining compound was glycoconjugate of quercetin with a hexose moiety. This compound could be a linkage isomer of quercetin-glucose. Regarding the precursor ion mass of m/z 481.1, a peak with the retention time of 4.06 min was identified as myricetin-glucoside by a reference standard. Additionally, one isobaric component was also found at different retention time, and it was determined as a myricetin-hexose glycoconjugate that was predicted to be a linkage isomer of myricetin-glucose.
Under extracted ion chromatogram of m/z 303.1, in addition to a peak that appeared at the retention time of 8.30 min, which was confirmed to be quercetin aglycon by a reference standard, there were some other peaks with the same mass found at different retention time. The m/z 303.1 was the product ion mass formed by the fragmentation of precursor ions including glycoconjugates of quercetin based on the MS/MS spectra. By extraction of molecular ion mass of m/z 459.0, we can see that some independent peaks appeared at distinct retention time. A peak with retention time at 2.46 min was confirmed to be epigallocatechin gallate (EGCG) when comparing to retention time of a reference standard.
For phenolic acids, from peak 1 to peak 3 shown in Supplementary Figure S1, the deprotonated molecular ions were m/z 169, m/z 153, and m/z 163, and their corresponding product ions were m/z 125, m/z 109, and m/z 119. These fragment ions were formed by losing carbonyl group (-COO) and based on the reference standards we can conclude that three of these peaks were gallic acid, 3,4-dihydrobenzoic acid, and p-coumaric acid, respectively.
For triterpenoids, nine triterpene acids were found in S. formosum leaves. During positive-ion mass spectrometry, the protonated molecular ions with losing two water molecules of peak 1 and peak 2 demonstrated molecular ion mass of m/z 469.3 and 453.3, respectively (Supplementary Figure S2). Peaks from 3 to 5 with the same molecular ion mass of m/z 455.3 that was formed by the protonation with losing one water molecule. Similarly, peaks from 7 to 9 had the same molecular ion mass of m/z 439.3; meanwhile, peak 6 had the m/z of 425.3. Most of these compounds performed the fragmentation in similar ways thereby generated the product ions with the same mass. By comparison of the retention time with the authentic standards, these compounds were confirmed to be madecassic acid, asiatic acid, hederagenin, betulin, betulinic acid, maslinic acid, corosolic acid, oleanolic acid, and ursolic acid corresponding to peaks from 1 to 9. The fragmentation pathways of detected triterpenoids in the SF leaves were illustrated in Supplementary Figure S3.
3.2. Method Validation
The quantification of the identified compounds was implemented under the multiple reaction monitoring (MRM) condition and by a comparison of peak areas with calibration curves from corresponding standard compounds. The results showed that the calibration curves of most standards exhibited the regression coefficients with a high R2-value (R2 ≥ 0.997). The limit of detection (LOD) of most of analytes was in the range of 1.0–5.0 μg/L and limit of quantification (LOQ) ranged 3.0–15.0 μg/L, except for gallic acid and betulinic acid which exhibited the limit of detection at 10 μg/L. The values expressed a high sensitivity of analytical method. The recovery values of most compounds were ~100%, except for quercetin and gallic acid with ~80% (Supplementary Table S2).
3.3. Phytochemical Composition in Syzygium formosum Leaves
Syzygium formosum is mainly distributed through the Northern region in Vietnam. In this study, the SF leaves collected from Hanoi in October of 2019 and Phu Tho in February of 2020 were analyzed. Phu Tho is a mid-land and mountainous province in the Northwestern part of Vietnam and it is about 90 km Northwest of Hanoi.
3.3.1. Flavonoids
The SF leaves collected at the Hanoi area had a higher content of flavonoids with 2.72 ± 0.24 mg/g dry leaf. This is 1.5-fold more than the sample collected in the Phu Tho area with 1.85 ± 0.11 mg/g dry leaf (p < 0.05).
Except for catechin and EGCG, myricetin glycoside and quercetin glycoside were identified from SF leaves (Table 3). Myricetin-3-O-rhamnoside was 54% of the total flavonoids in the Hanoi sample, followed by catechin and quercetin-3-O-arabinoside each of which were ~20% of the total. Meanwhile, the Phu Tho sample contained ~60% catechin and 30% myricetin-3-O-rhamnoside, five times higher than that of quercetin-3-O-arabinoside. Generally, catechin, myricetin-3-O-rhamnoside, and quercetin-3-O-arabinoside were the three dominant flavonoids of the SF leaves in both samples. The results also showed that the contents of the remaining individual flavonoids in the Hanoi sample were 2–3 folds higher than those in the Phu Tho sample.

Table 3.
Phenolic compound contents (mg/g dry leaf) in Syzygium formosum leaves.
3.3.2. Phenolic Acids
Gallic acid, 3,4-dihydrobenzoic acid, and p-coumaric acid were the phenolic acids found in Syzygium formosum leaves. Total phenolic contents between Hanoi and Phu Tho sample was not significantly different each with approximately 0.85 mg/g dry leaf (p < 0.05). It was less than half of the flavonoids and only 4–6% of triterpenoids. Gallic acid was the most abundant component consisting 95% of total phenolic acid content. The content of gallic acid in the Phu Tho sample was 0.85 ± 0.09 mg/g dry leaf, which was similar to the Hanoi batch with 0.78 ± 0.15 mg/g dry leaf (Table 3).
3.3.3. Triterpenoids
Triterpenoids were the most abundant phytochemicals identified in SF leaves showing 84% of total phytochemicals. Triterpenoids in Hanoi and Phu Tho samples were similar: 19.00 ± 1.51 and 14.18 ± 1.15 mg/g dry leaf, respectively, which occupied 84% of total phytochemicals (Table 4).

Table 4.
Triterpenoid contents (mg/g dry leaf) in Syzygium formosum and Centella asiatica leaves.
Nine triterpenoids were found in SF leaves. Asiatic acid, corosolic acid, and betulinic acid were the three most abundant ones. Betulinic acid composed over 40–45% of total triterpenoids, followed by asiatic acid and corosolic acid each of which were occupying about 20% of the total. Ursolic and maslinic acid was present at ~1.0 mg/g dry leaf. In addition, madecassic, oleanolic acid, hederagenin, and betulin were found less than 0.5 mg/g dry leaf.
Overall quantitative profiles of nine triterpenoids between the samples from two different regions were similar. However, the SF leaves from Phu Tho area contained more betulinic acid and less asiatic acid than the samples obtained from Hanoi. The asiatic acid concentration from the Hanoi sample was 8.59 ± 0.48 mg/g dry leaf which was 3.1-fold higher than the sample from Phu Tho. Meanwhile, the SF leaves from Phu Tho contained 1.7-fold more betulinic acid than the Hanoi sample with 6.14 ± 0.49 mg/g dry leaf.
3.4. Comparison of Triterpenoids between Syzygium formosum and Centella asiatica Leaves
Centella asiatica is a herbaceous medicinal plant recognized as an important drug by the Indian Herbal Pharmacopoeia, European Pharmacopoeia, Pharmacopoeia of the People’s Republic of China, and German Homoeopathic Pharmacopoeia [25]. It has been used to relieve skin rashes, heal wounds, and sooth itching. The functional molecule of Centella asiatica has been reported the triterpenoids, particularly the asiaticosides. To compare the triterpenoid profiles and contents, C. asiatica leaves from Indonesia and Korea were analyzed and compared to S. formosum leaves.
While nine triterpenoid compounds were found in the ethanolic extracts of S. formosum leaves, only asiatic acid and madecassic acid were found in C. asiatica samples from both Indonesia and Korea.
The results showed that the triterpenoid content in Korea C. asiatica was significantly lower than in other countries. The concentration of masdecassic acid was 0.53 ± 0.11 mg/g dry leaf that was similar or slightly higher than that observed in Vietnam samples. However, the asiatic acid concentration in the Korean sample was less than 1.0 mg/mL, showing only 9–30% of other samples from Vietnam and Indonesia. The madecassic acid content in Indonesia samples were 5- to 10-fold higher than that of other countries. Asiatic acid concentration was also higher than the samples from Korea and Phu Tho region, albeit no other triterpenoids were found including their hexose or pentose glycoconjugates (Table 4).
3.5. Anti-Inflammatory Effects of Syzygium formosum
S. formosum leaf extract and C. asiatica extract did not exert significant cytotoxicity up to 20 μg/mL on the human keratinocyte cell line, HaCaT (data not shown). To evaluate its anti-inflammatory effects, the S. formosum leaf extract was treated with 15 μg/mL of final concentration on HaCaT, which was radiated with UVB to induce proinflammatory responses. A commercial product of C. asiatica extract was also used for the experiment.
mRNA levels of proinflammatory cytokines IL-1β, IL-6, and IL-8 were greatly reduced by 15 μg/mL of S. formosum leaf extract, while C. asiatica extract did not induced any significant changes of cytokine expressions. COX-2 was also downregulated by S. formosum, but the reduction was not great compared with the three cytokines (Figure 2).
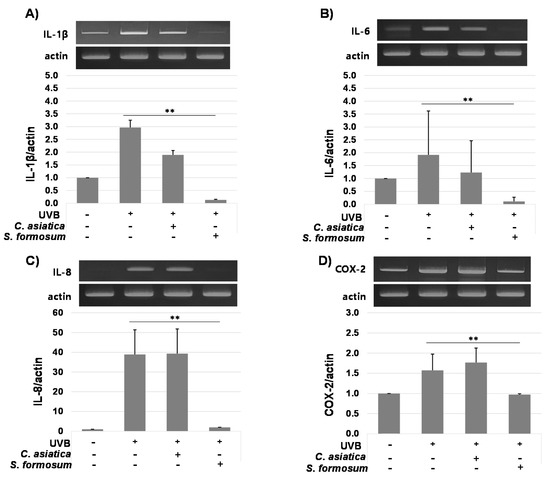
Figure 2.
Relative mRNA expression of proinflammatory cytokines (A–C) and COX-2 (D) after treatment of Syzygium formosum leaf extract or Centella asiatica extract on the HaCaT cells radiated with ultraviolet-B. Gene expression levels were compared with a house keeping gene, actin. Statistical significance (p < 0.01) is indicated with double asterisks (**).
3.6. Antibacterial Effect of Syzygium formosum Extract
Antibacterial effect of S. formosum leaf extract was evaluated on the bacteria which might induce serious skin problems such as atopic dermatitis and acne. First, MIC50 of the SF extract was determined on the S. aureus and S. epidermidis. The SF extract had strong antibacterial activities on Staphylococcus genus, and MIC50 was 95.52 μg/mL and 78.83 μg/mL against S. aureus and S. epidermidis, respectively. Mupirocin, pesudomonic acid A, was used as a positive control. Unlike mupirocin, the SF extract showed similar antibacterial effects on the two strains (Figure 3).
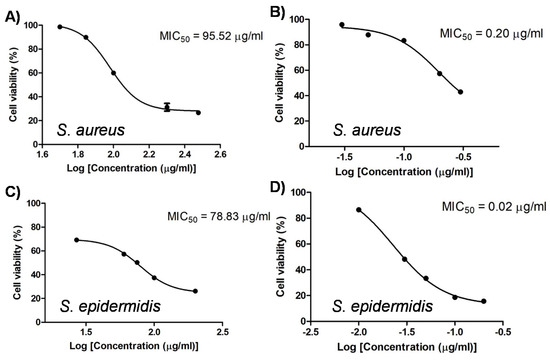
Figure 3.
Antibacterial effects of the Syzygium formosum leaf extract (A,C). Important skin bacteria Staphylococcus aureus and S. epidermidis were used. The commercial antibacterial drug, mupirocin was used as a positive control (B,D). Minimal inhibition concentration 50 (MIC50) was estimated and given in each graph.
The antibacterial activity of S. formosum leaf extract was also investigated for C. acnes. The number of bacteria obtained by spreading on each plate were presented in Table 5. When the number of colonies was over 250 on the plate, it was marked >250. In BHI media, the average number of 181 ± 12 colonies (182, 196, 166) were found at 106 dilutions. In BHI with the 1% (v/v) of 70% ethanol, the similar but slightly fewer colonies with 113 ± 19 (95, 105, 140) on average were counted at same dilutions. However, with S. formosum leaf extract, only a couple of colonies (3, 4, 0) were found at 103 dilutions showing 99.999% (10−5-fold) sterilization efficacy.

Table 5.
The number of Cutibacterium acnes colonies per dilution of each media treated with the Syzygium formosum extract.
4. Discussion
For many decades, medicinal herbs have attracted the interest of scientists as they possess various bioactive phytochemical constituents including phenolics, alkaloids, flavonoids, tannins, saponins, and triterpenoids which exhibit positive pharmacological functionalities on the human body.
In this study, we have aimed to provide more comprehensive information of bioactive compounds in Syzygium formosum regarding the cosmetic or dermatological uses for soothing or relieving the rashes, itching, atopic dermatitis or psoriasis, anti-inflammatory, and anti-bacterial activities. By the UVB-induced HaCaT inflammation model, it has been clearly demonstrated that the extract of S. formosum has great anti-inflammatory effects. When compared with C. asiatica which is currently proven to be effective, S. formosum leaf extract exhibited higher efficacy in the down-regulations of IL-1β, IL-6, and IL-8 expressions. In the stratum corneum and fibroblasts, IL-1β is a major cytokine affecting skin condition. IL-6 and IL-8 are also important in proinflammation responses of skin.
In addition, S. formosum leaf extract also showed a great antibacterial effect on S. aureus and C. acnes that is commensal skin bacteria species. Especially, by adding the S. formosum leaf extract, the growth of C. acnes growth was significantly suppressed showing the efficacy of 105 magnitudes that suggest the 99.999% sterilization. S. aureus generates toxins which could aggravate symptoms of atopic dermatitis. Proliferation of C. acnes is regarded as the main factor correlated with a severe form of acne. Therefore, controlling S. aureus and C. acnes is very important for skin health.
Despite the effective functionalities, there have been few in-depth studies on the comprehensive and systemic analysis of phytochemical components in S. formosum leaves. In our study, twenty phytochemical components including eight flavonoids (catechin, epigallocatechin gallate, quercetin aglycone, glycoconjugates of quercetin, and myricetin), three phenolic acids (gallic acid, 3,4-dihydrobenzoic acid, p-coumaric acid), and nine triterpenoids (madecassic acid, asiatic acid, hederagenin, maslinic acid, corosolic acid, betulin, betulinic acid, oleanolic acid, and ursolic acid) were identified in S. formosum leaves. Besides, four different glycoconjugates of quercetin and myricetin were determined as well. Each component of flavonoids, phenolic acids, and triterpenoids was quantified by the MRM using QQQ-MS/MS. Among the twenty-four compounds identified in the SF leaves, epigallocatechin gallate, p-coumaric acid, madecassic acid, hederagenin, and betulin were the five phytochemical components firstly recognized in our study.
S. formosum leaf contained a broad spectrum of triterpenoids derived from lupeol and amyrin. Betulin and betulinic acid, which is one of the major triterpenoids in the SF leaves, are derived from lupeol and are known to be major functional constituents in birch extract that has been used in cosmetic and herbal essence products [26,27]. Derived from β-amyrin, oleanolic acid is a precursor of maslinic acid and hederagenin, and importantly, ginsenoside Pg, Pq, and Pj [28]. Maslinic acid is abundant in the wax-like coating of the leaves and fruits of olive and is reported to exhibit anti-cancer and anti-inflammatory activities [29,30]. Instead of β-conformation, ursolic, corosolic, asiatic, and madecassic acids are synthesized by the sequential hydroxylation from the α-amyrin. Ursolic acid was a precursor of madecassic and asiatic acid and has been widely found in the peels of fruits and herbs [31]. Although the systematic human clinical trial has not been evaluated, numerous beneficial effects on health and disease were reported such as anti-inflammatory, antioxidant, anti-carcinogenic, antiobesity, antidiabetic, cardioprotective, neuroprotective, hepatoprotective, anti-skeletal muscle atrophy, and thermogenic effects [31,32]. The hydroxylated ursolic acid is corosolic acid. Corosolic acid is known to the main active constituents of Banaba, Lagerstroemia speciosa, that exhibit antidiabetic activity. With the oleanolic and maslinic acid, corosolic acid could inhibit the glycogen phosphorylase and consequently lower the blood glucose level in adrenaline-induced diabetic mice [33,34]. Asiatic acid is also one of the most abundant triterpenoids in SF leaves. With the madecassic acid, asiatic acid is a major functional phytochemical in Centella asiatica. It has been used cosmetically and pharmaceutically in skin and dermatological diseases.
Centella asiatica has currently been used as a commercial cosmeceutical ingredient. Compare to the C. asiatica grown in south and east Asia, S. formosum leaves were found to have more diverse and higher amounts of triterpenoids. While only asiatic and madecassic acid were identified, nine different triterpenoids were found from SF leaves extract. The asiatic acid and madecassic acid concentrations in SF leaves were similar or higher concentrations than those in C. asiatica. Only madecassic acid in the C. asiatica grown in Indonesia contained 5- to 10-fold more than SF leaves showing a similar amount to the asiatic acid. Seven additional triterpenoids in SF leaves were extraordinary, however, their amounts were not negligible as well. The concentrations of betulinic and corosolic acid were 3.2–6.1 mg/g dry leaf that was a similar amount of asiatic acid. Betulinic acid and corosolic acid have strong beneficial functionalities. Recent studies have provided evidence that betulinic acid ameliorates psoriatic symptoms and skin inflammation in psoriatic mice, thereby suggesting the compound could be a natural anti-psoriatic drug for treating human psoriasis [35,36]. Corosolic acid is also a triterpenoid widely found in plants and is known to regulate various cellular processes such as cellular oxidative stress and signaling kinase in some diseases, including skin cancer [37].
For analysis of phenolic compounds, myricetin-3-O-rhamnoside, a member of the flavonol class of the flavonoids, was the most abundance compound with ~50% of total flavonoids in S. formosum leaves, followed by quercetin-3-O-arabinoside with 20% of the total. Meanwhile, catechin, a flavanol, was found to be a dominant component with 20–60% of the total flavonoids in this plant. Myricetin-3-O-rhamnoside is thought to offer a variety of potential health benefits, displaying anti-mutagenic, anti-inflammatory, antioxidant, and antinociceptive activities [38]. Catechin is widely known to be a biologically active compound of green tea. Catechins have many benefits including prevention or reduction of skin damage [39,40]. Additionally, the three components of phenolic acids including gallic acid, 3,4-dihydrobenzoic acid, and p-coumaric acid were found in S. formosum leaves. Among them, gallic acid consisted of 95% of total phenolic acid content in S. formosum leaves. Recently, some studies have reported that gallic acid brought new perspective efficacy in cosmetics and nanocosmeceuticals [41]. Its anti-inflammatory properties make it valuable components to prevent inflammatory-status diseases such as skin inflammation, wound healing, skin infections [42,43,44].
In summary, the quantitative analysis of bioactive phytochemicals in S. formosum leaf extract was first performed in our study. S. formosum exhibited high levels of phytochemicals as well as its anti-inflammatory effect and antibacterial capacity, which make the SF leaves attractive for pharmacological and cosmetic applications in the future.
5. Conclusions
In this study, the detailed phytochemical profiling of S. formosum leaves was conducted. Our results showed that this plant contains valuable secondary metabolites including eight flavonoids, three phenolic acids, and nine triterpenoids. Our study is the first report involving the quantitative analysis of functional phytochemicals identified in the ethanolic extracts of S. formosum leaves as well as comparative analysis with the ethanolic extracts of C. asiatica leaves derived from Indonesia and Korea. The total content of functional phytochemical groups in the SF leaves was significantly higher than that in C. asiatica leaves. The high content of bioactive compounds in the SF leaves as well as the exhibition of biological functionalities of the SF leaves such as anti-inflammatory and antibacterial effects can explain its traditional use for the treatment of some skin-related diseases in Vietnam. S. formosum could be a promising potential candidate for cosmetic and pharmacological applications in the future.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/app112210552/s1, Table S1: The MRM parameters and MS/MS transitions for analysis of phenolic compounds and triterpenoids; Table S2: Regression equations and coefficients, linear ranges, limit of detection, limit of quantification, and recovery of the analytes; Figure S1: The MRM chromatogram of detected phenolic acids in Syzygium formosum leaves (A: Mixed standard, B: Sample; 1-Gallic acid, 2-3,4-dihydrobenzoic acid, 3-p-coumaric acid); Figure S2: The MRM chromatogram of detected triterpenoids in Syzygium formosum leaves (A: Mixed standard, B: Sample; 1-Madecassic acid, 2-Asiatic acid, 3-Hederagenin, 4-Maslinic acid, 5-Corosolic acid, 6-Betulin, 7-Betulinic acid, 8-Oleanolic acid, 9-Ursolic acid); Figure S3: Proposed fragmentation of phenolic compounds (A) and triterpenoids (B and C) in Syzygium formosum leaves.
Author Contributions
Data curation, M.T.T.N.; Formal analysis, J.L., M.T.T.N. and H.-L.K.; Funding acquisition, C.-K.L.; Investigation, K.H.T.H. and N.-Y.L.; Project administration, J.K.; Resources, C.-K.L. and V.D.N.; Supervision, J.-T.P.; Writing—original draft, K.H.T.H.; Writing—review and editing, J.-T.P. and J.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HP20C0211).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Soh, W.-K.; Parnell, J. A revision of Syzygium Gaertn (Myrtaceae) in Indochina (Cambodia, Laos and Vietnam). Adansonia 2015, 37, 179–275. [Google Scholar]
- Do, T. Medicinal Plants and Remedies of Vietnam; Medicine Publishing House: Hanoi, Vietnam, 2001; 826p. [Google Scholar]
- Parul, R.; Das, A.K.; Rana, M.S. Comparative pharmacological evaluation in respect to non-polar and polar solvent extracts of the leaves of Syzygium balsameum & Syzygium formosum. J. Pharmacogn. Phytochem. 2020, 9, 45–52. [Google Scholar]
- Nong, T. Research on Phytochemical Constituents of Syzygium Formosum in Thai Nguyen-Vietnam; Chemistry Department, Thai Nguyen University: Taiyuan, China, 2009. [Google Scholar]
- Thuong, P.T.; Na, M.-K.; Dang, N.H.; Hung, T.M.; Ky, P.T.; Thanh, T.V.; Nam, N.H.; Thuan, N.D.; Sok, D.-E.; Bae, K.-H. Antioxidant activities of Vietnamese medicinal plants. Nat. Prod. Sci. 2006, 12, 29–37. [Google Scholar]
- Nguyen, T.M.N.; Lomunova, M.; Vu, T.P.D.; Le, B.V.; Kim, Y.H.; Kang, J.S.; Hwang, I. Anti-allergic effects of the ethanol extract of Syzygium formosum (Wall.) Masam leaves and its immunoregulatory mechanisms. J. Ethnopharmacol. 2018, 211, 171–179. [Google Scholar] [CrossRef]
- Zhang, J.; Yamada, S.; Ogihara, E.; Kurita, M.; Banno, N.; Qu, W.; Feng, F.; Akihisa, T. Biological activities of triterpenoids and phenolic compounds from Myrica cerifera bark. Chem. Biodivers. 2016, 13, 1601–1609. [Google Scholar] [CrossRef] [PubMed]
- Nzogong, R.T.; Ndjateu, F.S.T.; Ekom, S.E.; Fosso, J.-A.M.; Awouafack, M.D.; Tene, M.; Tane, P.; Morita, H.; Choudhary, M.I.; Tamokou, J.-d.-D. Antimicrobial and antioxidant activities of triterpenoid and phenolic derivatives from two Cameroonian Melastomataceae plants: Dissotis senegambiensis and Amphiblemma monticola. BMC Complementary Altern. Med. 2018, 18, 159. [Google Scholar] [CrossRef]
- Manosroi, A.; Jantrawut, P.; Ogihara, E.; Yamamoto, A.; Fukatsu, M.; Yasukawa, K.; Tokuda, H.; Suzuki, N.; Manosroi, J.; Akihisa, T. Biological activities of phenolic compounds and triterpenoids from the galls of Terminalia chebula. Chem. Biodivers. 2013, 10, 1448–1463. [Google Scholar] [CrossRef]
- Vu, T.P.D.; Khong, T.Q.; Nguyen, T.M.N.; Kim, Y.H.; Kang, J.S. Phytochemical profile of Syzygium formosum (Wall.) Masam leaves using HPLC–PDA–MS/MS and a simple HPLC–ELSD method for quality control. J. Pharm. Biomed. Anal. 2019, 168, 1–12. [Google Scholar]
- Dzubak, P.; Hajduch, M.; Vydra, D.; Hustova, A.; Kvasnica, M.; Biedermann, D.; Markova, L.; Urban, M.; Sarek, J. Pharmacological activities of natural triterpenoids and their therapeutic implications. Nat. Prod. Rep. 2006, 23, 394–411. [Google Scholar] [CrossRef]
- Shu, Z.; Pu, J.; Chen, L.; Zhang, Y.; Rahman, K.; Qin, L.; Zheng, C. Alisma orientale: Ethnopharmacology, phytochemistry and pharmacology of an important traditional Chinese medicine. Am. J. Chin. Med. 2016, 44, 227–251. [Google Scholar] [CrossRef]
- Thakurdesai, P.A. Centella asiatica (Gotu kola) leaves: Potential in neuropsychiatric conditions. In Nutraceuticals in Brain Health and Beyond; Elsevier: Cham, Switzerland, 2021; pp. 307–328. [Google Scholar]
- Ho, P.J.; Sung, J.J.; Cheon, K.K.; Tae, H.J. Anti-inflammatory effect of Centella asiatica phytosome in a mouse model of phthalic anhydride-induced atopic dermatitis. Phytomedicine 2018, 43, 110–119. [Google Scholar]
- Brinkhaus, B.; Lindner, M.; Schuppan, D.; Hahn, E. Chemical, pharmacological and clinical profile of the East Asian medical plant Centella aslatica. Phytomedicine 2000, 7, 427–448. [Google Scholar] [CrossRef]
- Sun, B.; Wu, L.; Wu, Y.; Zhang, C.; Qin, L.; Hayashi, M.; Kudo, M.; Gao, M.; Liu, T. Therapeutic potential of Centella asiatica and its triterpenes: A review. Front. Pharmacol. 2020, 11, 1373. [Google Scholar] [CrossRef]
- Roy, A.; Krishnan, L.; Bharadvaja, N. Qualitative and quantitative phytochemical analysis of Centella asiatica. Nat. Prod. Chem. Res. 2018, 6, 4. [Google Scholar] [CrossRef]
- Shen, X.; Guo, M.; Yu, H.; Liu, D.; Lu, Z.; Lu, Y. Propionibacterium acnes related anti-inflammation and skin hydration activities of madecassoside, a pentacyclic triterpene saponin from Centella asiatica. Biosci. Biotechnol. Biochem. 2019, 83, 561–568. [Google Scholar] [CrossRef]
- Sawatdee, S.; Choochuay, K.; Chanthorn, W.; Srichana, T. Evaluation of the topical spray containing Centella asiatica extract and efficacy on excision wounds in rats. Acta Pharm. 2016, 66, 233–244. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seo, S.; Kim, Y. Improving cosmetic activity by optimizing Centella asiatica extraction process. Nat. Prod. Commun. 2019, 14, 1934578X19867188. [Google Scholar] [CrossRef] [Green Version]
- Ratz-Łyko, A.; Arct, J.; Pytkowska, K. Moisturizing and antiinflammatory properties of cosmetic formulations containing Centella asiatica extract. Indian J. Pharm. Sci. 2016, 78, 27. [Google Scholar] [CrossRef] [Green Version]
- Yadav, M.; Chatterji, S.; Gupta, S.K.; Watal, G. Preliminary phytochemical screening of six medicinal plants used in traditional medicine. Int. J. Pharm. Pharm. Sci. 2014, 6, 539–542. [Google Scholar]
- Nahar, L.; Sarker, S.D. Chemistry for Pharmacy Students: General, Organic and Natural Product Chemistry; John Wiley & Sons: Hoboken, NJ, USA, 2019. [Google Scholar]
- Ahmad, S.; Ullah, F.; Ayaz, M.; Sadiq, A.; Imran, M. Antioxidant and anticholinesterase investigations of Rumex hastatus D. Don: Potential effectiveness in oxidative stress and neurological disorders. Biol. Res. 2015, 48, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loc, N.H.; An, N.T.T. Asiaticoside production from centella (Centella asiatica L. Urban) cell culture. Biotechnol. Bioprocess Eng. 2010, 15, 1065–1070. [Google Scholar] [CrossRef]
- Šiman, P.; Filipová, A.; Tichá, A.; Niang, M.; Bezrouk, A.; Havelek, R. Effective method of purification of betulin from birch bark: The importance of its purity for scientific and medicinal use. PLoS ONE 2016, 11, e0154933. [Google Scholar] [CrossRef] [Green Version]
- Rastogi, S.; Pandey, M.M.; Rawat, A.K.S. Medicinal plants of the genus Betula—Traditional uses and a phytochemical–pharmacological review. J. Ethnopharmacol. 2015, 159, 62–83. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-J.; Zhang, D.; Yang, D.-C. Biosynthesis and biotechnological production of ginsenosides. Biotechnol. Adv. 2015, 33, 717–735. [Google Scholar] [CrossRef]
- Lee, W.; Kim, J.; Park, E.K.; Bae, J.-S. Maslinic acid ameliorates inflammation via the downregulation of NF-κB and STAT-1. Antioxidants 2020, 9, 106. [Google Scholar] [CrossRef] [Green Version]
- Juan, M.E.; Planas, J.M. Cancer chemopreventive activity of maslinic acid, a pentacyclic triterpene from olives and olive oil. In Olives and Olive Oil in Health and Disease Prevention; Elsevier: Cham, Switzerland, 2021; pp. 525–535. [Google Scholar]
- Lee, S.Y.; Kim, Y.J.; Chung, S.O.; Park, S.U. Recent studies on ursolic acid and its biological and pharmacological activity. EXCLI J. 2016, 15, 221. [Google Scholar] [PubMed]
- Nguyen, H.N.; Ullevig, S.L.; Short, J.D.; Wang, L.; Ahn, Y.J.; Asmis, R. Ursolic Acid and Related Analogues: Triterpenoids with Broad Health Benefits. Antioxidants 2021, 10, 1161. [Google Scholar] [CrossRef]
- Yamada, K.; Hosokawa, M.; Fujimoto, S.; Fujiwara, H.; Fujita, Y.; Harada, N.; Yamada, C.; Fukushima, M.; Ueda, N.; Kaneko, T. Effect of corosolic acid on gluconeogenesis in rat liver. Diabetes Res. Clin. Pract. 2008, 80, 48–55. [Google Scholar] [CrossRef]
- Shi, L.; Zhang, W.; Zhou, Y.-Y.; Zhang, Y.-N.; Li, J.-Y.; Hu, L.-H.; Li, J. Corosolic acid stimulates glucose uptake via enhancing insulin receptor phosphorylation. Eur. J. Pharmacol. 2008, 584, 21–29. [Google Scholar] [CrossRef]
- Weber, L.A.; Meißner, J.; Delarocque, J.; Kalbitz, J.; Feige, K.; Kietzmann, M.; Michaelis, A.; Paschke, R.; Michael, J.; Pratscher, B. Betulinic acid shows anticancer activity against equine melanoma cells and permeates isolated equine skin in vitro. BMC Vet. Res. 2020, 16, 44. [Google Scholar] [CrossRef]
- Liu, C.; Chen, Y.; Lu, C.; Chen, H.; Deng, J.; Yan, Y.; Xu, Y.-Y.; Liu, H.; Huang, H.; Wei, J. Betulinic acid suppresses Th17 response and ameliorates psoriasis-like murine skin inflammation. Int. Immunopharmacol. 2019, 73, 343–352. [Google Scholar] [CrossRef]
- Hudlikar, R.R.; Sargsyan, D.; Wu, R.; Su, S.; Zheng, M.; Kong, A.-N. Triterpenoid corosolic acid modulates global CpG methylation and transcriptome of tumor promotor TPA induced mouse epidermal JB6 P+ cells. Chem. Biol. Interact. 2020, 321, 109025. [Google Scholar] [CrossRef]
- Domitrović, R.; Rashed, K.; Cvijanović, O.; Vladimir-Knežević, S.; Škoda, M.; Višnić, A. Myricitrin exhibits antioxidant, anti-inflammatory and antifibrotic activity in carbon tetrachloride-intoxicated mice. Chem. Biol. Interact. 2015, 230, 21–29. [Google Scholar] [CrossRef]
- Katiyar, S.K.; Ahmad, N.; Mukhtar, H. Green tea and skin. Arch. Dermatol. 2000, 136, 989–994. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.; Kim, N.; Shin, Y.; Kim, S.-Y.; Kim, Y.-J. Activity of catechins and their applications. Biomed. Dermatol. 2020, 4, 8. [Google Scholar] [CrossRef] [Green Version]
- Khan, B.A.; Mahmood, T.; Menaa, F.; Shahzad, Y.; Yousaf, A.M.; Hussain, T.; Ray, S.D. New perspectives on the efficacy of gallic acid in cosmetics & nanocosmeceuticals. Curr. Pharm. Des. 2018, 24, 5181–5187. [Google Scholar]
- Yang, D.J.; Moh, S.H.; Son, D.H.; You, S.; Kinyua, A.W.; Ko, C.M.; Song, M.; Yeo, J.; Choi, Y.-H.; Kim, K.W. Gallic acid promotes wound healing in normal and hyperglucidic conditions. Molecules 2016, 21, 899. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vitonyte, J.; Manca, M.L.; Caddeo, C.; Valenti, D.; Peris, J.E.; Usach, I.; Nacher, A.; Matos, M.; Gutiérrez, G.; Orrù, G. Bifunctional viscous nanovesicles co-loaded with resveratrol and gallic acid for skin protection against microbial and oxidative injuries. Eur. J. Pharm. Biopharm. 2017, 114, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Tsang, M.S.; Jiao, D.; Chan, B.C.; Hon, K.-L.; Leung, P.C.; Lau, C.; Wong, E.C.; Cheng, L.; Chan, C.K.; Lam, C.W. Anti-inflammatory activities of pentaherbs formula, berberine, gallic acid and chlorogenic acid in atopic dermatitis-like skin inflammation. Molecules 2016, 21, 519. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).