Synthesis of Porous Biomimetic Composites: A Sea Urchin Skeleton Used as a Template
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Methods of Synthesis
2.2.1. Synthesis of PFPS
2.2.2. Synthesis of the PFPS-Based Composite
2.3. Characterization of Research Methods
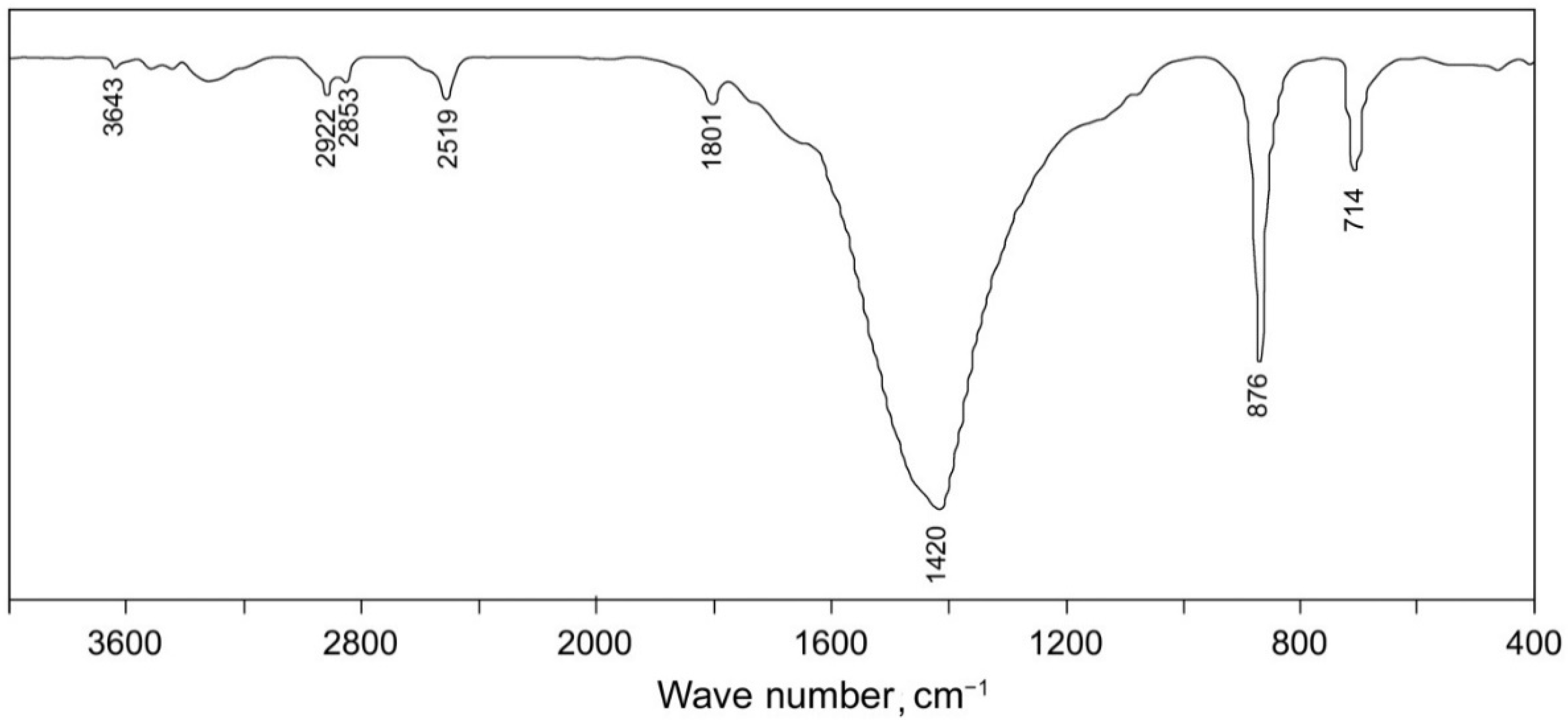
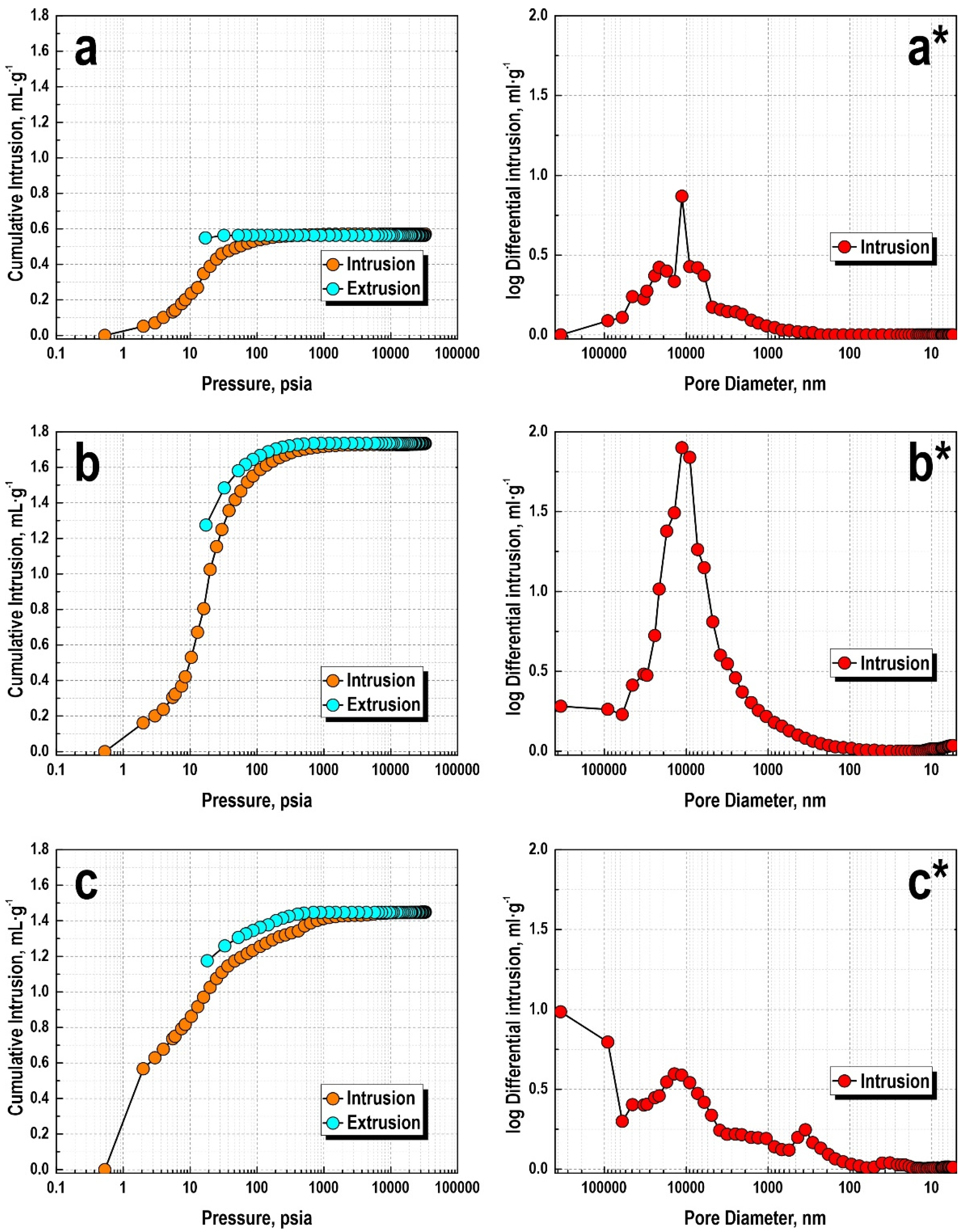
3. Results
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Martín-Palma, R.J.; Kolle, M. Biomimetic photonic structures for optical sensing. Opt. Laser Technol. 2019, 109, 270–277. [Google Scholar] [CrossRef]
- Romanholo, P.V.V.; Razzino, C.A.; Raymundo-Pereira, P.A.; Prado, T.M.; Machado, S.A.S.; Sgobbi, L.F. Biomimetic electrochemical sensors: New horizons and challenges in biosensing applications. Biosens. Bioelectron. 2021, 185, 113242. [Google Scholar] [CrossRef]
- Zhao, L.; Yang, H.; He, F.; Yao, Y.; Xu, R.; Wang, L.; He, L.; Zhang, H.; Li, S.; Huang, F. Biomimetic N-doped sea-urchin-structured porous carbon for the anode material of high-energy-density potassium-ion batteries. Electrochim. Acta 2021, 388, 138565. [Google Scholar] [CrossRef]
- Han, X.; Wu, J.; Zhang, X.; Shi, J.; Wei, J.; Yang, Y.; Wu, B.; Feng, Y. Special issue on advanced corrosion-resistance materials and emerging applications. The progress on antifouling organic coating: From biocide to biomimetic surface. J. Mater. Sci. Technol. 2021, 61, 46–62. [Google Scholar] [CrossRef]
- Li, W.; Pei, Y.; Zhang, C.; Kottapalli, A.G.P. Bioinspired designs and biomimetic applications of triboelectric nanogenerators. Nano Energy 2021, 84, 105865. [Google Scholar] [CrossRef]
- Al-Ketan, O.; Rowshan, R.; Alami, A.H. Biomimetic Materials for Engineering Applications. In Reference Module in Materials Science and Materials Engineering; Elsevier: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Cojocaru, F.D.; Balan, V.; Tanase, C.E.; Popa, I.M.; Butnaru, M.; Bredetean, O.; Mares, M.; Nastasa, V.; Pasca, S.; Verestiuc, L. Development and characterisation of microporous biomimetic scaffolds loaded with magnetic nanoparticles as bone repairing material. Ceram. Int. 2021, 47, 11209–11219. [Google Scholar] [CrossRef]
- Cruz, E.; Hubert, T.; Chancoco, G.; Naim, O.; Chayaamor-Heil, N.; Cornette, R.; Badarnah, L.; Raskin, K.; Aujard, F. Design processes and multi-regulation of biomimetic building skins: A comparative analysis. Energy Build. 2021, 246, 111034. [Google Scholar] [CrossRef]
- Wang, R.; Yan, H.; Yu, A.; Ye, L.; Zhai, G. Cancer targeted biomimetic drug delivery system. J. Drug Deliv. Sci. Technol. 2021, 63, 102530. [Google Scholar] [CrossRef]
- Baldino, L.; Aragón, J.; Mendoza, G.; Irusta, S.; Cardea, S.; Reverchon, E. Production, characterization and testing of antibacterial PVA membranes loaded with HA-Ag3PO4 nanoparticles, produced by SC-CO2 phase inversion. J. Chem. Technol. Biotechnol. 2019, 94, 98–108. [Google Scholar] [CrossRef] [Green Version]
- Ajmal, G.; Bonde, G.V.; Mittal, P.; Khan, G.; Pandey, V.K.; Bakade, B.V.; Mishra, B. Biomimetic PCL-gelatin based nanofibers loaded with ciprofloxacin hydrochloride and quercetin: A potential antibacterial and anti-oxidant dressing material for accelerated healing of a full thickness wound. Int. J. Pharm. 2019, 567, 118480. [Google Scholar] [CrossRef]
- Fuwad, A.; Ryu, H.; Malmstadt, N.; Kim, S.M.; Jeon, T.J. Biomimetic membranes as potential tools for water purification: Preceding and future avenues. Desalination 2019, 458, 97–115. [Google Scholar] [CrossRef]
- Chen, W.; Mo, J.; Du, X.; Zhang, Z.; Zhang, W. Biomimetic dynamic membrane for aquatic dye removal. Water Res. 2019, 151, 243–251. [Google Scholar] [CrossRef]
- Liu, H.; Liu, L.; Yu, J.; Yin, X.; Ding, B. High-efficiency and super-breathable air filters based on biomimetic ultrathin nanofiber networks. Compos. Commun. 2020, 22, 100493. [Google Scholar] [CrossRef]
- Jakšić, Z.; Jakšić, O. Biomimetic nanomembranes: An overview. Biomimetics 2020, 5, 24. [Google Scholar] [CrossRef] [PubMed]
- Tu, Y.M.; Samineni, L.; Ren, T.; Schantz, A.B.; Song, W.; Sharma, S.; Kumar, M. Prospective applications of nanometer-scale pore size biomimetic and bioinspired membranes. J. Memb. Sci. 2021, 620, 118968. [Google Scholar] [CrossRef]
- Park, C.W.; Lee, J.H.; Seo, J.K.; Ran, W.T.A.; Whang, D.; Hwang, S.M.; Kim, Y.J. Graphene/pvdf composites for ni-rich oxide cathodes toward high-energy density li-ion batteries. Materials 2021, 14, 2271. [Google Scholar] [CrossRef]
- Kamedulski, P.; Lukaszewicz, J.P.; Witczak, L.; Szroeder, P.; Ziolkowski, P. The importance of structural factors for the electrochemical performance of graphene/carbon nanotube/melamine powders towards the catalytic activity of oxygen reduction reaction. Materials 2021, 14, 2448. [Google Scholar] [CrossRef] [PubMed]
- Chae, G.S.; Youn, D.H.; Lee, J.S. Nanostructured iron sulfide/n, s dual-doped carbon nanotube-graphene composites as efficient electrocatalysts for oxygen reduction reaction. Materials 2021, 14, 2146. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.L.; Masoomi, M.Y.; Morsali, A. Template strategies with MOFs. Coord. Chem. Rev. 2019, 387, 415–435. [Google Scholar] [CrossRef]
- Wang, C.Y.; Jiao, K.; Yan, J.F.; Wan, M.C.; Wan, Q.Q.; Breschi, L.; Chen, J.H.; Tay, F.R.; Niu, L.N. Biological and synthetic template-directed syntheses of mineralized hybrid and inorganic materials. Prog. Mater. Sci. 2021, 116, 100712. [Google Scholar] [CrossRef]
- Yang, T.; Wu, Z.; Chen, H.; Zhu, Y.; Li, L. Quantitative 3D structural analysis of the cellular microstructure of sea urchin spines (I): Methodology. Acta Biomater. 2020, 107, 204–217. [Google Scholar] [CrossRef] [PubMed]
- Shapkin, N.P.; Khalchenko, I.G.; Panasenko, A.E.; Drozdov, A.L. Sea urchin skeleton: Structure, composition, and application as a template for biomimetic materials. In Proceedings of the AIP Conference Proceedings, St. Petersburg, Russia, 19–21 May 2017; Volume 1858, p. 020006. [Google Scholar] [CrossRef]
- Donnay, G.; Pawson, D.L. X-ray Diffraction Studies of Echinoderm Plates. Science 1969, 166, 1147–1150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weber, J.N. The incorporation of magnesium into the skeletal calcites of echinoderms. Am. J. Sci. 1969, 267, 537–566. [Google Scholar] [CrossRef]
- Voronkov, M.G.; Shapkin, N.P. Phosphorus-containing polymetalloorganosiloxanes. J. Organomet. Chem. 1990, 389, 169–186. [Google Scholar] [CrossRef]




| No. | Sample | Impregnation with Polymer | Calcination after Impregnation with Polymer | Acid Treatment after Calcination |
|---|---|---|---|---|
| 1 | c-PFPS | PFPS | - | - |
| 2 | c-PFPS/1000 | PFPS | 1000 °C | - |
| 3 | c-PFPS/1000/HCl | PFPS | 1000 °C | HCl |
| Object | Content of Elements, wt% * | |||||||
|---|---|---|---|---|---|---|---|---|
| СаО | MgO | Na2O | K2O | SiO2 | P2O5 | SrO | Fe2O3 | |
| Test of the sea urchin (original) | 45.98 | 11.03 | 40.70 | 1.52 | 0.30 | - | 0.20 | 0.11 |
| Test of the urchin (calcined at 700 °C) | 87.40 | 11.67 | - | - | - | - | 0.46 | 0.11 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shapkin, N.P.; Papynov, E.K.; Panasenko, A.E.; Khalchenko, I.G.; Mayorov, V.Y.; Drozdov, A.L.; Maslova, N.V.; Buravlev, I.Y. Synthesis of Porous Biomimetic Composites: A Sea Urchin Skeleton Used as a Template. Appl. Sci. 2021, 11, 8897. https://doi.org/10.3390/app11198897
Shapkin NP, Papynov EK, Panasenko AE, Khalchenko IG, Mayorov VY, Drozdov AL, Maslova NV, Buravlev IY. Synthesis of Porous Biomimetic Composites: A Sea Urchin Skeleton Used as a Template. Applied Sciences. 2021; 11(19):8897. https://doi.org/10.3390/app11198897
Chicago/Turabian StyleShapkin, Nikolay P., Evgeniy K. Papynov, Alexandr E. Panasenko, Irina G. Khalchenko, Vitaly Yu. Mayorov, Anatoliy L. Drozdov, Natalya V. Maslova, and Igor Yu. Buravlev. 2021. "Synthesis of Porous Biomimetic Composites: A Sea Urchin Skeleton Used as a Template" Applied Sciences 11, no. 19: 8897. https://doi.org/10.3390/app11198897
APA StyleShapkin, N. P., Papynov, E. K., Panasenko, A. E., Khalchenko, I. G., Mayorov, V. Y., Drozdov, A. L., Maslova, N. V., & Buravlev, I. Y. (2021). Synthesis of Porous Biomimetic Composites: A Sea Urchin Skeleton Used as a Template. Applied Sciences, 11(19), 8897. https://doi.org/10.3390/app11198897








