A Multivariate Randomized Controlled Experiment about the Effects of Mindfulness Priming on EEG Neurofeedback Self-Regulation Serious Games
Featured Application
Abstract
1. Introduction
2. Materials and Methods
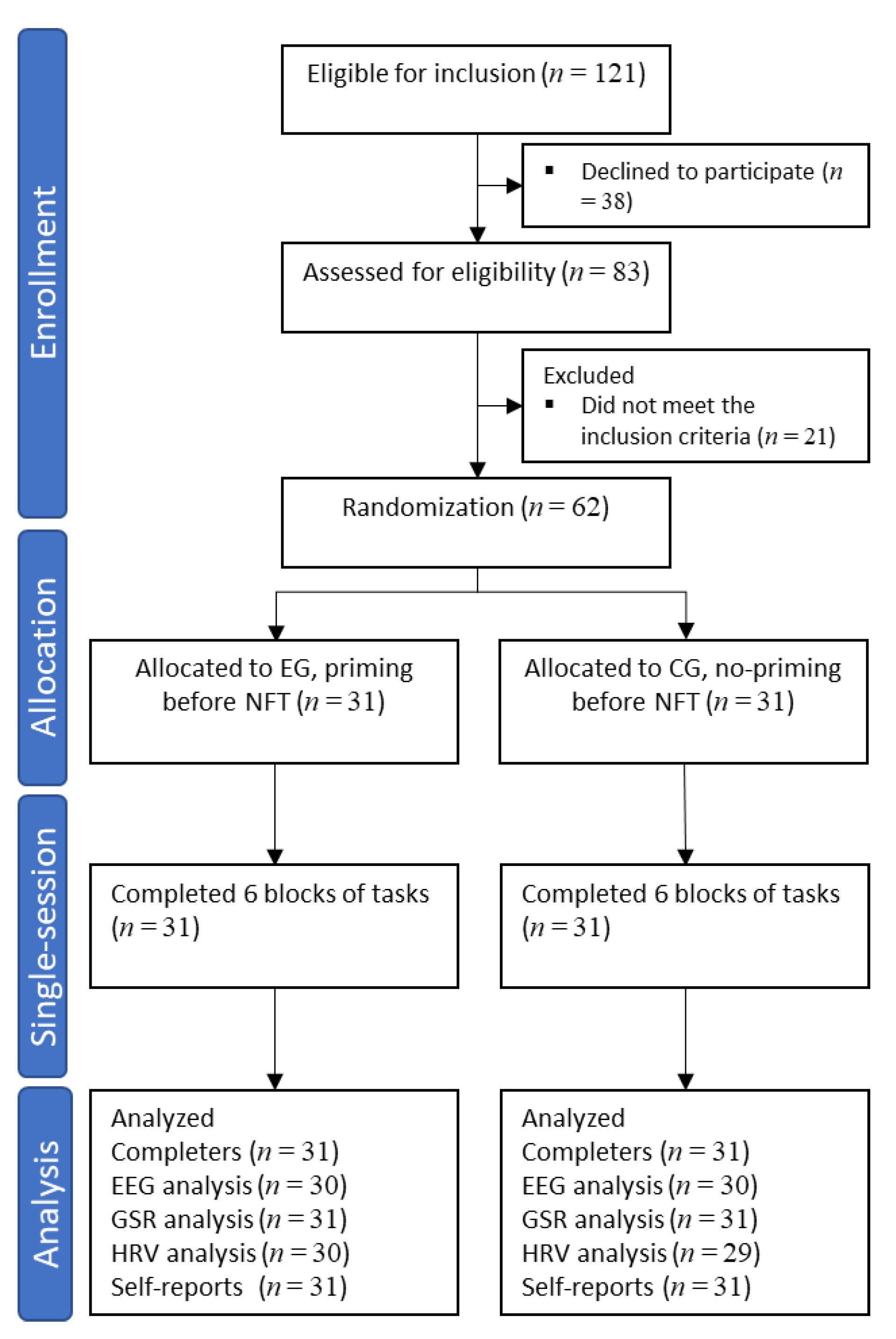
2.1. Participants
2.2. Randomizations and Study Blinding
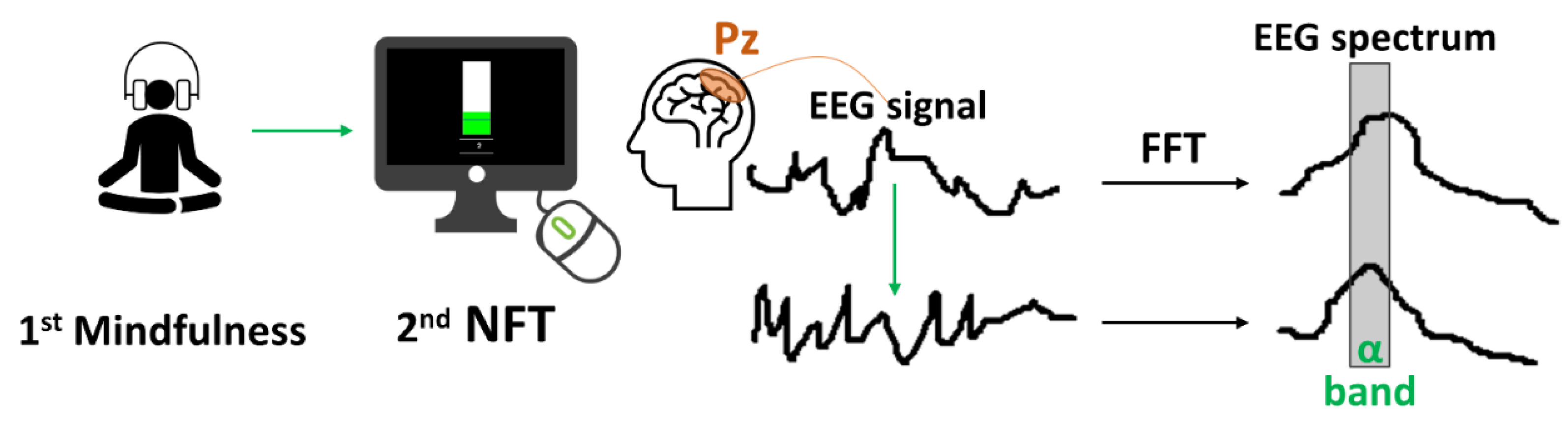
2.3. Interventions and Control Condition
2.4. Experimental Design
2.5. Questionnaires
2.6. Physiological Measures
2.7. Recordings
2.8. Multidimensional Signals Processing
2.9. Data Analysis
3. Results
3.1. Group Characteristics
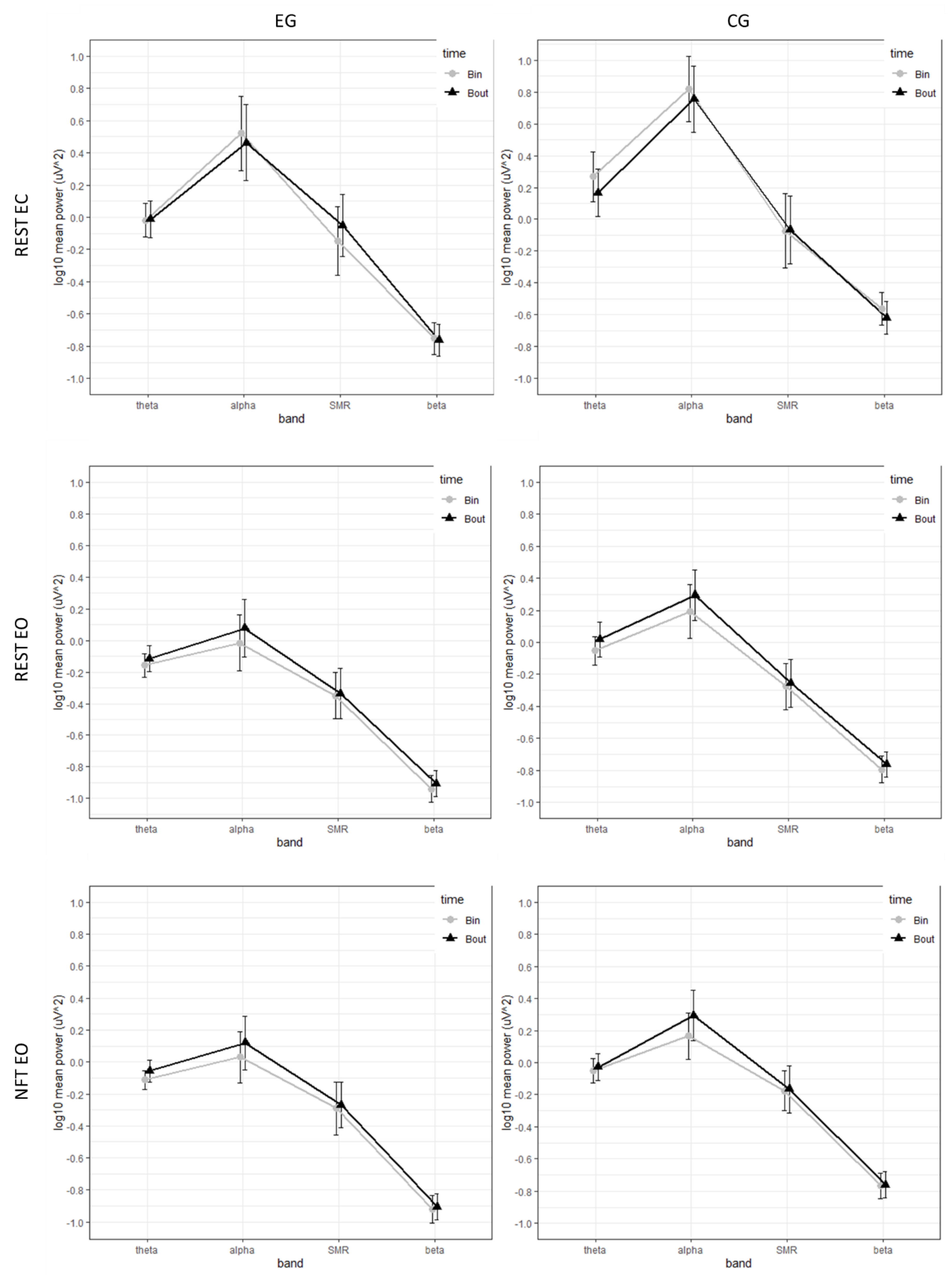
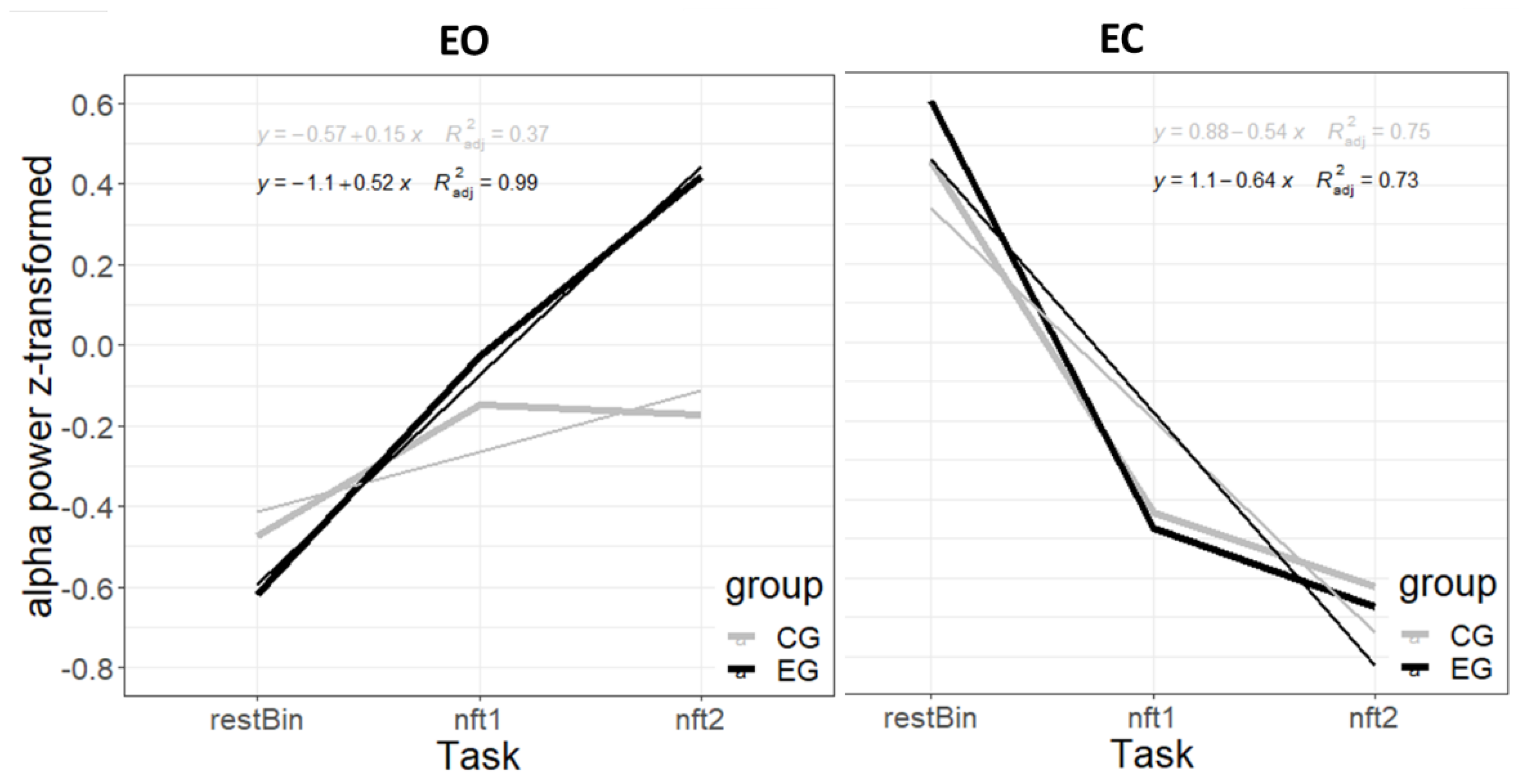
3.2. EEG Power Spectrum at Pre and Post Priming Intervention
3.3. NFT Performance in Different Group Domains at Intervention Blocks
4. Discussion
General Discussion and Future Proposals
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Appendix A
Appendix A.1. EEG Features
- [theta, alpha, SMR, beta]: list of bands extracted.
- epoch_a: epochs array of each task = [[band mean, standard deviation]…, [n-epoch]]. Bands power spectrum density (PSD) is calculated from 1000 samples per second.
- Mean: changes in absolute values of frequency band mean amplitude (power spectra measures were log10-transformed to obtain normally distributed data), reflecting brief and temporally unstable increases over time from the learner.
Appendix A.2. GSR Features
- epoch_a: epochs array of each task = [[TIMESTAMP, SAMPLE_COUNTER, GSR_VALUE,], …, [n-epoch]]. Each epoch is 1 sample of GSR value, calculated from the 100 samples per second.
- scl_mean: GSR mean per task.
- scl_std: GSR standard deviation per task.
- scr_sumResp: sum of response amplitude per task.
Appendix A.3. HRV Features
- epoch_a: epochs array of each task = [[TIMESTAMP, SAMPLE_COUNTER, BPM_VALUE, RR_VALUE], …, [n-epoch]]. Each epoch is 1 sample of RR value, calculated from the 100 samples per second.
- sdnn: The standard deviation of the time interval between successive normal heart beats (i.e., the RR-intervals).
- rmssd: The square root of the mean of the sum of the squares of differences between adjacent NN-intervals. Reflects high frequency (fast or parasympathetic) influences on HRV (i.e., those influencing larger changes from one beat to the next).
Appendix A.4. Self-Reports Features
- First questionnaire at T0 (pre-intervention): https://forms.gle/2uT7f7oH3pd4c9FD9 (accessed on 21 August 2021).
- Second questionnaire at Bin: https://forms.gle/nQNRQkBWEVtbKySo8 (accessed on 21 August 2021).
- Third questionnaire at Bout: https://forms.gle/k1zVwzwVacu7hBYRA (accessed on 21 August 2021).
Appendix A.4.1. Traits (TG)
FFMQ
- Observe. “I notice the smells and aromas of things.”
- Describe. “I am good at finding words to describe my feelings.”
- Actaware (acting with awareness). “I find myself doing things without paying awareness attention” (R).
- Nonjudge (nonjudging of inner emotions). “I think some of my emotions are bad or experience inappropriate and I should not feel them”(R).
- Nonreact (nonreactivity to inner emotions). “I perceive my feelings and emotions experience without having to react to them.”
ERQ
- Cognitive reappraisal. Where a person attempts to change how he or she thinks about a situation in order to change its emotional impact.
- Expressive suppression. “I keep my emotions to myself”—where a person attempts to inhibit the behavioural expression of his or her emotions.
DASS
Appendix A.4.2. Sates (SG)
TMS
POMS
- Tension: state of preoccupation and muscle tension.
- Fatigue: state of tiredness, inertia, boredom.
- Confusion: state of confusion.
- Vigour: state of energy and physical and psychological vigour.
References
- Damasio, A.R. Self Comes to Mind: Constructing the Conscious Brain; Pantheon Books: New York, NY, USA, 2010; ISBN 9780307378750. [Google Scholar]
- Gruzelier, J. Differential effects on mood of 12–15 (SMR) and 15–18 (beta1) Hz neurofeedback. Int. J. Psychophysiol. 2014, 93, 112–115. [Google Scholar] [CrossRef]
- Heatherton, T.F. Neuroscience of Self and Self-Regulation. Annu. Rev. Psychol 2011, 62, 363–390. [Google Scholar] [CrossRef]
- Tang, Y.-Y.; Hölzel, B.K.; Posner, M.I. The neuroscience of mindfulness meditation. Nat. Rev. Neurosci. 2015, 16, 213–225. [Google Scholar] [CrossRef]
- Gruzelier, J. EEG-neurofeedback for optimising performance. II: Creativity, the performing arts and ecological validity. Neurosci. Biobehav. Rev. 2013, 44, 142–158. [Google Scholar] [CrossRef]
- Jin, J.; Xiao, R.; Daly, I.; Miao, Y.; Wang, X.; Cichocki, A. Internal Feature Selection Method of CSP Based on L1-Norm and Dempster-Shafer Theory. IEEE Trans. Neural Netw. Learn. Syst. 2020, 1–12. [Google Scholar] [CrossRef]
- Pinheiro, J.; de Almeida, R.S.; Marques, A. Emotional self-regulation, virtual reality and neurofeedback. Comput. Hum. Behav. Rep. 2021, 4, 100101. [Google Scholar] [CrossRef]
- Ninaus, M.; Witte, M.; Kober, S.E.; Friedrich, E.V.C.; Kurzmann, J.; Hartsuiker, E.; Neuper, C.; Wood, G. Neurofeedback and serious games. Psychol. Pedagog. Assess. Serious Games 2013, 82–109. [Google Scholar] [CrossRef]
- Coenen, F.; Scheepers, F.E.; Palmen, S.J.M.; de Jonge, M.V.; Oranje, B. Serious Games as Potential Therapies: A Validation Study of a Neurofeedback Game. Clin. EEG Neurosci. 2020, 51, 87–93. [Google Scholar] [CrossRef]
- Israsena, P.; Jirayucharoensak, S.; Hemrungrojn, S.; Pan-Ngum, S. Brain Exercising Games with Consumer-Grade Single-Channel Electroencephalogram Neurofeedback: Pre-Post Intervention Study. JMIR Serious Games 2021, 9, e26872. [Google Scholar] [CrossRef]
- Ros, T.; Baars, B.; Lanius, R.R.A.; Vuilleumier, P.; Baars, B.J.; Lanius, R.R.A.; Vuilleumier, P. Tuning pathological brain oscillations with neurofeedback: A systems neuroscience framework. Front. Hum. Neurosci. 2014, 8, 1008. [Google Scholar] [CrossRef]
- Sitaram, R.; Ros, T.; Stoeckel, L.; Haller, S.; Scharnowski, F.; Lewis-Peacock, J.; Weiskopf, N.; Blefari, M.L.; Rana, M.; Oblak, E.; et al. Closed-loop brain training: The science of neurofeedback. Nat. Rev. Neurosci. 2016, 18, 86–100. [Google Scholar] [CrossRef]
- Enriquez-Geppert, S.; Huster, R. EEG-neurofeedback as a tool to modulate cognition and behavior: A review tutorial. Front. Hum. 2017, 11, 51. [Google Scholar] [CrossRef]
- Davelaar, E. Mechanisms of neurofeedback: A computation-theoretic approach. Neuroscience 2017, 378, 175–188. [Google Scholar] [CrossRef] [PubMed]
- Gu, S.; Cieslak, M.; Baird, B.; Muldoon, S.F.; Grafton, S.T.; Pasqualetti, F.; Bassett, D.S. The Energy Landscape of Neurophysiological Activity Implicit in Brain Network Structure. Sci. Rep. 2018, 8, 2507. [Google Scholar] [CrossRef] [PubMed]
- Ros, T.; Enriquez-Geppert, S.; Zotev, V.; Young, K.D.; Wood, G.; Whitfield-Gabrieli, S.; Wan, F.; Vuilleumier, P.; Vialatte, F.; Van De Ville, D.; et al. Consensus on the reporting and experimental design of clinical and cognitive-behavioural neurofeedback studies (CRED-nf checklist). Brain 2020, 143, 1674–1685. [Google Scholar] [CrossRef] [PubMed]
- Zoefel, B.; Huster, R.J.; Herrmann, C.S. Neurofeedback training of the upper alpha frequency band in EEG improves cognitive performance. NeuroImage 2010, 54, 1427–1431. [Google Scholar] [CrossRef]
- Gruzelier, J. EEG-neurofeedback for optimising performance. I: A review of cognitive and affective outcome in healthy participants. Neurosci. Biobehav. Rev. 2013, 44, 124–141. [Google Scholar] [CrossRef]
- Thibault, R.; Veissière, S.; Olson, J.; Raz, A. Treating ADHD with suggestion: Neurofeedback and placebo therapeutics. J. Atten. Disord. 2018, 22, 707–711. [Google Scholar] [CrossRef]
- Zuberer, A.; Brandeis, D. Are treatment effects of neurofeedback training in children with ADHD related to the successful regulation of brain activity? A review on the learning of regulation of brain activity and a contribution to the discussion on specificity. Front. Hum. 2015, 9, 135. [Google Scholar] [CrossRef]
- Alkoby, O.; Abu-Rmileh, A.; Shriki, O.; Todder, D. Can We Predict Who Will Respond to Neurofeedback? A Review of the Inefficacy Problem and Existing Predictors for Successful EEG Neurofeedback Learning. Neuroscience 2018, 378, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Nan, W.; Wan, F.; Tang, Q.; Wong, C.M.; Wang, B.; Rosa, A. Eyes-Closed Resting EEG Predicts the Learning of Alpha Down-Regulation in Neurofeedback Training. Front. Psychol. 2018, 9, 1607. [Google Scholar] [CrossRef] [PubMed]
- Witte, M.; Kober, S.E.S.; Ninaus, M.; Neuper, C.; Wood, G. Control beliefs can predict the ability to up-regulate sensorimotor rhythm during neurofeedback training. Front. Hum. Neurosci. 2013, 7, 478. [Google Scholar] [CrossRef]
- Ninaus, M.; Kober, S.E.; Witte, M.; Koschutnig, K.; Stangl, M.; Neuper, C.; Wood, G. Neural substrates of cognitive control under the belief of getting neurofeedback training. Front. Hum. Neurosci. 2013, 7, 914. [Google Scholar] [CrossRef]
- Wood, G.; Kober, S.; Witte, M. On the need to better specify the concept of “control” in brain-computer-interfaces/neurofeedback research. Front. Syst. 2014, 8, 171. [Google Scholar] [CrossRef]
- Davelaar, E.J.; Barnby, J.M.; Almasi, S.; Eatough, V. Differential Subjective Experiences in Learners and Non-learners in Frontal Alpha Neurofeedback: Piloting a Mixed-Method Approach. Front. Hum. Neurosci. 2018, 12, 402. [Google Scholar] [CrossRef]
- Chow, T.; Javan, T.; Ros, T.; Frewen, P. EEG Dynamics of Mindfulness Meditation Versus Alpha Neurofeedback: A Sham-Controlled Study. Mindfulness 2017, 8, 572–584. [Google Scholar] [CrossRef]
- Tang, Y. The Neuroscience of Mindfulness Meditation: How the Body and Mind Work Together to Change Our Behaviour; Palgrave Macmillan: London, UK, 2017. [Google Scholar] [CrossRef]
- Lutz, A.; Slagter, H.A.; Dunne, J.D.; Davidson, R.J. Attention regulation and monitoring in meditation. Trends Cogn. Sci. 2008, 12, 163–169. [Google Scholar] [CrossRef]
- Kober, S.; Witte, M.; Ninaus, M. Ability to Gain Control Over One’s Own Brain Activity and its Relation to Spiritual Practice: A Multimodal Imaging Study. Front. Hum. 2017, 11, 271. [Google Scholar] [CrossRef]
- Zhao, Z.; Yao, S.; Zweerings, J.; Zhou, X.; Zhou, F.; Kendrick, K.M.; Chen, H.; Mathiak, K.; Becker, B. Putamen volume predicts real-time fMRI neurofeedback learning success across paradigms and neurofeedback target regions. Hum. Brain Mapp. 2021, 42, 1879–1887. [Google Scholar] [CrossRef]
- Haugg, A.; Sladky, R.; Skouras, S.; McDonald, A.; Craddock, C.; Kirschner, M.; Herdener, M.; Koush, Y.; Papoutsi, M.; Keynan, J.N.; et al. Can we predict real-time fMRI neurofeedback learning success from pre-training brain activity? Hum. Brain Mapp. 2020, 41, 3839–3854. [Google Scholar] [CrossRef]
- Haugg, A.; Renz, F.M.; Nicholson, A.A.; Lor, C.; Götzendorfer, S.J.; Sladky, R.; Skouras, S.; McDonald, A.; Craddock, C.; Hellrung, L.; et al. Predictors of real-time fMRI neurofeedback performance and improvement—A machine learning mega-analysis. Neuroimage 2021, 237, 118207. [Google Scholar] [CrossRef] [PubMed]
- Diaz Hernandez, L.; Rieger, K.; Koenig, T. Low Motivational Incongruence Predicts Successful EEG Resting-state Neurofeedback Performance in Healthy Adults. Neuroscience 2018, 378, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Reichert, J.L.; Kober, S.E.; Neuper, C.; Wood, G. Resting-state sensorimotor rhythm (SMR) power predicts the ability to up-regulate SMR in an EEG-instrumental conditioning paradigm. Clin. Neurophysiol. 2015, 126, 2068–2077. [Google Scholar] [CrossRef] [PubMed]
- Wan, F.; Nan, W.; Vai, M.; Rosa, A. Resting alpha activity predicts learning ability in alpha neurofeedback. Front. Hum. 2014, 8, 500. [Google Scholar] [CrossRef] [PubMed]
- Hardman, E.; Gruzelier, J.; Cheesman, K.; Jones, C. Frontal interhemispheric asymmetry: Self regulation and individual differences in humans. Neuroscience 1997, 221, 117–120. [Google Scholar] [CrossRef]
- Egner, T.; Gruzelier, J. Learned self-regulation of EEG frequency components affects attention and event-related brain potentials in humans. NeuroReport 2001, 12, 4155–4159. [Google Scholar] [CrossRef]
- Nijboer, F.; Sellers, E.; Mellinger, J.; Jordan, M. A P300-based brain–computer interface for people with amyotrophic lateral sclerosis. Clinical 2008, 119, 1909–1916. [Google Scholar] [CrossRef]
- Dickhaus, T.; Sannelli, C.; Müller, K.R.; Curio, G.; Blankertz, B. Predicting BCI performance to study BCI illiteracy. BMC Neurosci 2009, 10, P84. [Google Scholar] [CrossRef]
- Kleih, S.; Nijboer, F.; Halder, S.; Kübler, A. Motivation modulates the P300 amplitude during brain–computer interface use. Clin. Neurophysiol. 2010, 121, 1023–1031. [Google Scholar] [CrossRef]
- Hammer, E.M.; Halder, S.; Blankertz, B.; Sannelli, C.; Dickhaus, T.; Kleih, S.; Müller, K.R.; Kübler, A. Psychological predictors of SMR-BCI performance. Biol. Psychol. 2012, 89, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Kober, S.E.S.; Witte, M.; Ninaus, M.; Neuper, C.; Wood, G. Learning to modulate one’s own brain activity: The effect of spontaneous mental strategies. Front. Hum. Neurosci. 2013, 7, 695. [Google Scholar] [CrossRef] [PubMed]
- Enriquez-Geppert, S.; Huster, R.J.; Herrmann, C.S. Boosting brain functions: Improving executive functions with behavioral training, neurostimulation, and neurofeedback. Int. J. Psychophysiol. 2013, 88, 1–16. [Google Scholar] [CrossRef]
- Ruiz, S.; Buyukturkoglu, K.; Rana, M.; Birbaumer, N.; Sitaram, R. Real-time fMRI brain computer interfaces: Self-regulation of single brain regions to networks. Biol. Psychol. 2014, 95, 4–20. [Google Scholar] [CrossRef] [PubMed]
- Hammer, E.; Kaufmann, T.; Kleih, S. Visuo-motor coordination ability predicts performance with brain-computer interfaces controlled by modulation of sensorimotor rhythms (SMR). Front. Hum. 2014, 8, 574. [Google Scholar] [CrossRef]
- Kotchoubey, B.; Strehl, U.; Holzapfel, S. Negative potential shifts and the prediction of the outcome of neurofeedback therapy in epilepsy. Clinical 1999, 110, 683–686. [Google Scholar] [CrossRef]
- Neumann, N.; Birbaumer, N. Predictors of successful self control during brain-computer communication. J. Neurol. Neurosurg. 2003, 74, 1117–1121. [Google Scholar] [CrossRef] [PubMed]
- Blankertz, B.; Sannelli, C.; Halder, S.; Hammer, E.M.; Kübler, A.; Müller, K.R.; Curio, G.; Dickhaus, T. Neurophysiological predictor of SMR-based BCI performance. NeuroImage 2010, 51, 1303–1309. [Google Scholar] [CrossRef]
- Weber, E.; Köberl, A.; Frank, S.; Doppelmayr, M. Predicting successful learning of SMR neurofeedback in healthy participants: Methodological considerations. Appl. Psychophysiol. Biofeedback 2011, 36, 37–45. [Google Scholar] [CrossRef]
- Nan, W.; Rodrigues, J.P.; Ma, J.; Qu, X.; Wan, F.; Mak, P.-I.; Mak, P.U.; Vai, M.I.; Rosa, A. Individual alpha neurofeedback training effect on short term memory. Int. J. Psychophysiol. 2012, 86, 83–87. [Google Scholar] [CrossRef]
- Halder, S.; Varkuti, B.; Bogdan, M.; Kübler, A.; Rosenstiel, W.; Sitaram, R.; Birbaumer, N. Prediction of brain-computer interface aptitude from individual brain structure. Front. Hum. Neurosci. 2013, 7, 105. [Google Scholar] [CrossRef]
- Enriquez-Geppert, S.; Huster, R.; Scharfenort, R. The morphology of midcingulate cortex predicts frontal-midline theta neurofeedback success. Front. Hum. Neurosci. 2013, 7, 453. [Google Scholar] [CrossRef] [PubMed]
- Nan, W.; Wan, F.; Vai, M.I.; Da Rosa, A.C. Resting and Initial Beta Amplitudes Predict Learning Ability in Beta/Theta Ratio Neurofeedback Training in Healthy Young Adults. Front. Hum. Neurosci. 2015, 9, 677. [Google Scholar] [CrossRef] [PubMed]
- Weber, L.A.; Ethofer, T.; Ehlis, A.C. Predictors of neurofeedback training outcome: A systematic review. NeuroImage Clin. 2020, 27, 102301. [Google Scholar] [CrossRef] [PubMed]
- Klimesch, W.; Sauseng, P.; Hanslmayr, S. EEG alpha oscillations: The inhibition-timing hypothesis. Brain Res. Rev. 2007, 53, 63–88. [Google Scholar] [CrossRef]
- Gruzelier, J.; Thompson, T.; Redding, E.; Brandt, R. Application of alpha/theta neurofeedback and heart rate variability training to young contemporary dancers: State anxiety and creativity. Int. J. Psychophysiol. 2014, 93, 105–111. [Google Scholar] [CrossRef]
- Kober, S.E.; Schweiger, D.; Witte, M.; Reichert, J.L.; Grieshofer, P.; Neuper, C.; Wood, G. Specific effects of EEG based neurofeedback training on memory functions in post-stroke victims. J. Neuroeng. Rehabil. 2015, 12, 107. [Google Scholar] [CrossRef] [PubMed]
- Hanslmayr, S.; Sauseng, P.; Doppelmayr, M.; Schabus, M.; Klimesch, W. Increasing individual upper alpha power by neurofeedback improves cognitive performance in human subjects. Appl. Psychophysiol. Biofeedback 2005, 30, 1–10. [Google Scholar] [CrossRef]
- Gruzelier, J.; Inoue, A.; Smart, R.; Steed, A.; Steffert, T. Acting performance and flow state enhanced with sensory-motor rhythm neurofeedback comparing ecologically valid immersive VR and training screen scenarios. Neurosci. Lett. 2010, 480, 112–116. [Google Scholar] [CrossRef]
- Van Lutterveld, R.; Houlihan, S.; Pal, P.; Sacchet, M.; McFarlane-Blake, C.; Patel, P.R.; Sullivan, J.S.; Ossadtchi, A.; Druker, S.; Bauer, C.; et al. Source-space EEG neurofeedback links subjective experience with brain activity during effortless awareness meditation. NeuroImage 2016, 151, 117–127. [Google Scholar] [CrossRef]
- Aftanas, L.; Golosheikin, S. Changes in cortical activity in altered states of consciousness: The study of meditation by high-resolution EEG. Hum. Physiol. 2003, 29, 143–151. [Google Scholar] [CrossRef]
- Cahn, B.R.; Polich, J. Meditation states and traits: EEG, ERP, and neuroimaging studies. Psychol. Bull. 2006, 132, 180–211. [Google Scholar] [CrossRef]
- Lagopoulos, J.; Xu, J.; Rasmussen, I.; Vik, A.; Malhi, G.S.; Eliassen, C.F.; Arntsen, I.E.; Sæther, J.G.; Hollup, S.; Holen, A.; et al. Increased theta and alpha EEG activity during nondirective meditation. J. Altern. Complement. Med. 2009, 15, 1187–1192. [Google Scholar] [CrossRef]
- Chiesa, A.; Serretti, A. A systematic review of neurobiological and clinical features of mindfulness meditations. Psychol. Med. 2010, 40, 1239–1252. [Google Scholar] [CrossRef] [PubMed]
- Lomas, T.; Ivtzan, I.; Fu, C.H.Y. A systematic review of the neurophysiology of mindfulness on EEG oscillations. Neurosci. Biobehav. Rev. 2015, 57, 401–410. [Google Scholar] [CrossRef] [PubMed]
- Navarro Gil, M.; Escolano Marco, C.; Montero-Marín, J.; Minguez Zafra, J.; Shonin, E.; García Campayo, J. Efficacy of Neurofeedback on the Increase of Mindfulness-Related Capacities in Healthy Individuals: A Controlled Trial. Mindfulness 2018, 9, 303–311. [Google Scholar] [CrossRef]
- Da Costa, N.M.C. Nmc-Costa/Neuroprime: A Framework for Real-Time HCI/BCI. Specifically Developed for Advanced Human-Computer Assisted Self-Regulation of Neurofeedback. Available online: https://github.com/nmc-costa/neuroprime (accessed on 19 April 2020).
- Da Costa, N.M.C.; Bicho, E.G.; Dias, N.S. Priming with mindfulness affects our capacity to self-regulate brain activity? In Proceedings of the 2020 IEEE 8th International Conference on Serious Games and Applications for Health (SeGAH), Online, 12–14 August 2020; pp. 1–8. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Lang, A.G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. In Proceedings of the Behavior Research Methods; Psychonomic Society Inc.: Chicago, IL, USA, 2007; Volume 39, pp. 175–191. Available online: https://link.springer.com/content/pdf/10.3758/BF03193146.pdf (accessed on 21 August 2021).
- Frewen, P.A.; Unholzer, F.; Logie-Hagan, K.R.J.; MacKinley, J.D. Meditation Breath Attention Scores (MBAS): Test-Retest Reliability and Sensitivity to Repeated Practice. Mindfulness 2014, 5, 161–169. [Google Scholar] [CrossRef]
- Ros, T.; Théberge, J.; Frewen, P.A.; Kluetsch, R.; Densmore, M.; Calhoun, V.D.; Lanius, R.A. Mind over chatter: Plastic up-regulation of the fMRI salience network directly after EEG neurofeedback. NeuroImage 2013, 65, 324–335. [Google Scholar] [CrossRef]
- Peeters, F.; Ronner, J.; Bodar, L.; van Os, J.; Lousberg, R. Validation of a neurofeedback paradigm: Manipulating frontal EEG alpha-activity and its impact on mood. Int. J. Psychophysiol. 2013, 93, 116–120. [Google Scholar] [CrossRef] [PubMed]
- Wells, R.; Outhred, T.; Heathers, J.A.J.; Quintana, D.S.; Kemp, A.H. Matter over mind: A randomised-controlled trial of single-session biofeedback training on performance anxiety and heart rate variability in musicians. PLoS ONE 2012, 7, e46597. [Google Scholar] [CrossRef] [PubMed]
- Angelakis, E.; Stathopoulou, S.; Frymiare, J.L.; Green, D.L.; Lubar, J.F.; Kounios, J. EEG neurofeedback: A brief overview and an example of peak alpha frequency training for cognitive enhancement in the elderly. Clin. Neuropsychol. 2007, 21, 110–129. [Google Scholar] [CrossRef]
- Van Boxtel, G.J.M.; Denissen, A.J.M.; Jäger, M.; Vernon, D.; Dekker, M.K.J.; Mihajlović, V.; Sitskoorn, M.M. A novel self-guided approach to alpha activity training. Int. J. Psychophysiol. 2012, 83, 282–294. [Google Scholar] [CrossRef]
- Vernon, D.; Dempster, T.; Bazanova, O.; Rutterford, N.; Pasqualini, M.; Andersen, S. Alpha Neurofeedback Training for Performance Enhancement: Reviewing the Methodology. J. Neurother. 2009, 13, 214–227. [Google Scholar] [CrossRef]
- Barry, R.J.; Clarke, A.R.; Johnstone, S.J.; Magee, C.A.; Rushby, J.A. EEG differences between eyes-closed and eyes-open resting conditions. Clin. Neurophysiol. 2007, 118, 2765–2773. [Google Scholar] [CrossRef]
- Baer, R.A.; Smith, G.T.; Lykins, E.; Button, D.; Krietemeyer, J.; Sauer, S.; Walsh, E.; Duggan, D.; Williams, J.M.G. Construct validity of the five facet mindfulness questionnaire in meditating and nonmeditating samples. Assessment 2008, 15, 329–342. [Google Scholar] [CrossRef]
- Gregório, S.; Gouveia, J.P. Facetas de mindfulness: Características psicométricas de um instrumento de avaliação. Psychologica 2011, 259–279. [Google Scholar] [CrossRef]
- Crawford, J.R.; Henry, J.D. The Depression Anxiety Stress Scales (DASS): Normative data and latent structure in a large non-clinical sample. Br. J. Clin. Psychol. 2003, 42, 111–131. [Google Scholar] [CrossRef] [PubMed]
- Pais-Ribeiro, J.L.; Honrado, A.; Leal, I. Contribuição para o Estudo da Adaptação Portuguesa das Escalas de Ansiedade, Depressão e Stress (EADS) de 21 itens de Lovibond e Lovibond. Psicol. Saúde Doenças 2004, 5, 229–239. [Google Scholar]
- Lovibond, P.; Lovibond, S. The structure of negative emotional states: Comparison of the Depression Anxiety Stress Scales (DASS) with the Beck Depression and Anxiety Inventories. Behav. Res. Ther. 1995, 33, 335–343. [Google Scholar] [CrossRef]
- Vaz, F.; Martins, C.; Martins, E.C. Diferenciação emocional e regulação emocional em adultos portugueses. Psicologia 2008, 22, 123. [Google Scholar] [CrossRef]
- Viana, M.; Almeida, P.; Santos, R. Adaptação Portuguesa da Versão Reduzida do Perfil de Estados de Humor: POMS. Aná. Psicol. 2001, 19, 77–92. [Google Scholar] [CrossRef]
- Lau, M.A.; Bishop, S.R.; Segal, Z.V.; Buis, T.; Anderson, N.D.; Carlson, L.; Shapiro, S.; Carmody, J.; Abbey, S.; Devins, G. The toronto mindfulness scale: Development and validation. J. Clin. Psychol. 2006, 62, 1445–1467. [Google Scholar] [CrossRef]
- Moreira, P.S.; Chaves, P.; Dias, R.; Dias, N.; Almeida, P.R. Validation of wireless sensors for psychophysiological studies. Sensors 2019, 19, 4824. [Google Scholar] [CrossRef] [PubMed]
- Correia, B.; Dias, N.; Costa, P.; Pêgo, J.M. Validation of a Wireless Bluetooth Photoplethysmography Sensor Used on the Earlobe for Monitoring Heart Rate Variability Features during a Stress-Inducing Mental Task in Healthy Individuals. Sensors 2020, 20, 3905. [Google Scholar] [CrossRef] [PubMed]
- Dempster, T.; Vernon, D. Identifying indices of learning for alpha neurofeedback training. Appl. Psychophysiol. Biofeedback 2009, 34, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Louis, E.K.S.; Frey, L.C.; Britton, J.W.; Frey, L.C.; Hopp, J.L.; Korb, P.; Koubeissi, M.Z.; Lievens, W.E.; Pestana-Knight, E.M.; Louis, E.K.S. The Normal EEG. 2016. Available online: https://www.ncbi.nlm.nih.gov/books/NBK390343/ (accessed on 21 August 2021).
- Boucsein, W.; Fowles, D.C.; Grimnes, S.; Ben-Shakhar, G.; Roth, W.T.; Dawson, M.E.; Filion, D.L. Publication recommendations for electrodermal measurements. Psychophysiology 2012, 49, 1017–1034. [Google Scholar] [CrossRef]
- Posada-Quintero, H.F.; Chon, K.H. Innovations in Electrodermal Activity Data Collection and Signal Processing: A Systematic Review. Sensors 2020, 20, 479. [Google Scholar] [CrossRef]
- Malik, M.; John Camm, A.; Thomas Bigger, J.; Breithardt, G.; Cerutti, S.; Cohen, R.J.; Coumel, P.; Fallen, E.L.; Kennedy, H.L.; Kleiger, R.E.; et al. Heart rate variability: Standards of measurement, physiological interpretation, and clinical use. Circulation 1996, 93, 1043–1065. [Google Scholar] [CrossRef]
- Zahn, D.; Adams, J.; Krohn, J.; Wenzel, M.; Mann, C.G.; Gomille, L.K.; Jacobi-Scherbening, V.; Kubiak, T. Heart rate variability and self-control—A meta-analysis. Biol. Psychol. 2016, 115, 9–26. [Google Scholar] [CrossRef]
- Beauchaine, T. Vagal tone, development, and Gray’s motivational theory: Toward an integrated model of autonomic nervous system functioning in psychopathology. Dev. Psychopathol. 2001, 13, 183–214. [Google Scholar] [CrossRef]
- Graziano, P.; Derefinko, K. Cardiac vagal control and children’s adaptive functioning: A meta-analysis. Biol. Psychol. 2013, 94, 22–37. [Google Scholar] [CrossRef]
- Pimenta, M.G.; Brown, T.; Arns, M.; Enriquez-Geppert, S. Treatment Efficacy and Clinical Effectiveness of EEG Neurofeedback as a Personalized and Multimodal Treatment in ADHD: A Critical Review. Neuropsychiatr. Dis. Treat. 2021, 17, 637. [Google Scholar] [CrossRef] [PubMed]
- Champseix, R. Aura-Healthcare/Hrv-Analysis: Package for Heart Rate Variability Analysis in Python. Available online: https://github.com/Aura-healthcare/hrv-analysis (accessed on 12 May 2021).

| EG (n = 30) | CG (n = 30) | p-Value | ||||
|---|---|---|---|---|---|---|
| M | SD | M | SD | F | p | |
| Demographic | ||||||
| Age (years) | 28.87 | 7.40 | 27.50 | 6.38 | 0.587 | ns |
| Gender (F/M) | 18/12 | 19/11 | 0.00 a | ns | ||
| Education (9/12/15/17/21) | 0/3/10/12/5 | 0/8/8/11/3 | 5.01 b | ns | ||
| Conditions | ||||||
| ES (ES1/ES2) | 16/14 | 15/15 | 0.0 a | ns | ||
| SS (RS/PS1/PS2) | 0/15/15 | 30/0/0 | - c | - c | ||
| Baseline Bands | ||||||
| theta (EC/EO) | −0.02/−0.16 | 0.28/0.2 | 0.27/−0.05 | 0.42/0.24 | 9.75/3.92 | **/ns |
| alpha (EC/EO) | 0.52/−0.015 | 0.61/0.47 | 0.82/0.19 | 0.56/0.45 | 3.90/3.04 | ns/ns |
| SMR (EC/EO) | −0.15/−0.35 | 0.55/0.40 | −0.07/−0.28 | 0.55/0.38 | 0.23/0.53 | ns/ns |
| beta (EC/EO) | −0.75/−0.94 | 0.27/0.23 | −0.56/−0.79 | 0.28/0.22 | 7.31/6.30 | **/* |
| Task | T | T × G | ||||
|---|---|---|---|---|---|---|
| F | ηp2 | Bout-Bin | F | ηp2 | ||
| REST EC | theta | 3.98 | 0.06 | −0.050 | 4.86 * | 0.08 |
| alpha | 5.04 * | 0.08 | −0.061 | 0.01 | <0.001 | |
| SMR | 4.67 * | 0.09 | 0.054 | <0.001 | <0.001 | |
| beta | 4.45 * | 0.07 | −0.033 | 2.13 | 0.04 | |
| REST EO | theta | 8.89 ** | 0.13 | 0.056 | 0.64 | 0.01 |
| alpha | 18.17 *** | 0.24 | 0.096 | 0.07 | 0.001 | |
| SMR | 33.62 *** | 0.38 | 0.015 | 0.61 | 0.01 | |
| beta | 5.21 * | 0.08 | 0.033 | 0.01 | <0.001 | |
| NFT EO | theta | 4.41 * | 0.07 | 0.039 | 0.77 | 0.01 |
| alpha | 20.67 *** | 0.26 | 0.109 | 0.65 | 0.01 | |
| SMR | 0.02 | <0.001 | 0.015 | 0.02 | <0.001 | |
| beta | 0.79 | 0.01 | 0.012 | 0.17 | 0.003 | |
| Domain | Feature | HV Frequencies (EG/CG) | EO EG [HVp1/LVp2]p3 | EO CG [HVp1/LVp2]p3 |
|---|---|---|---|---|
| TG | FFMQ actaware | 13/16 | [0.16/0.79 ***] ++ | [0.35/−0.08] |
| SG | TMS decentering (Bin) | 18/19 | [0.69 ***/0.27] | [0.07/0.29] |
| POMS Vigour (Bout) | 13/15 | [0.2/0.76 ***] + | [0.22/0.09] | |
| POMS confusion (Bout-Bin) | 12/13 | [0.64 **/0.44 *] | [−0.23/0.44 *] ++ | |
| POMS fatigue (Bout-Bin) | 18/21 | [0.48 **/0.58 *] | [−0.06/0.63 *] + | |
| POMS tension (Bout_Bin) | 17/20 | [0.69 ***/0.3] | [0.04/0.38] | |
| GSR | GSR scl_mean | 19/15 | [0.39 */0.74 **] | [0.46 **/−0.15] + |
| GSR scl_std | 15/17 | [0.36/0.68 ***] | [0.1/0.22] | |
| GSR scr_sumResp | 11/10 | [0.4/0.59 ***] | [0.24/0.11] | |
| HRV | HRV sdnn | 15/15 | [0.48 */0.56 *] | [0.44 */−0.12] + |
| HRV rmssd | 14/9 | [0.35/0.69 ***] | [0.23/0.17] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
da Costa, N.M.C.; Bicho, E.; Ferreira, F.; Vilhena, E.; Dias, N.S. A Multivariate Randomized Controlled Experiment about the Effects of Mindfulness Priming on EEG Neurofeedback Self-Regulation Serious Games. Appl. Sci. 2021, 11, 7725. https://doi.org/10.3390/app11167725
da Costa NMC, Bicho E, Ferreira F, Vilhena E, Dias NS. A Multivariate Randomized Controlled Experiment about the Effects of Mindfulness Priming on EEG Neurofeedback Self-Regulation Serious Games. Applied Sciences. 2021; 11(16):7725. https://doi.org/10.3390/app11167725
Chicago/Turabian Styleda Costa, Nuno M. C., Estela Bicho, Flora Ferreira, Estela Vilhena, and Nuno S. Dias. 2021. "A Multivariate Randomized Controlled Experiment about the Effects of Mindfulness Priming on EEG Neurofeedback Self-Regulation Serious Games" Applied Sciences 11, no. 16: 7725. https://doi.org/10.3390/app11167725
APA Styleda Costa, N. M. C., Bicho, E., Ferreira, F., Vilhena, E., & Dias, N. S. (2021). A Multivariate Randomized Controlled Experiment about the Effects of Mindfulness Priming on EEG Neurofeedback Self-Regulation Serious Games. Applied Sciences, 11(16), 7725. https://doi.org/10.3390/app11167725








