Abstract
The study of Technosols development, spatial distribution and physicochemical characteristics is becoming more and more important in the Anthropocene Era. The aim of the present study was to assess soil features and potential heavy metal release risk of soils developed on different mine tailing types after the waste disposal derived from mining activity in Central Italy. Soils were analyzed for their morphological, physical and chemical properties, and a chemical sequential extraction of heavy metals was performed. The investigated soils were classified as Technosols toxic having in some layer within 50 cm of the soil surface inorganic materials with high concentrations of toxic elements. Our findings showed that the bioavailability of potentially toxic element concentrations in the soil changed according to the origin of the mine tailing. However, because of the acidic pH, there is a serious risk of metals leaching which was reduced where the soil organic matter content was higher.
1. Introduction
The awareness that human activities have an extremely relevant impact in landscape transformation and environmental characterization has been recently embraced in the first theory on “Anthropocene” as a new Geological Era [1,2,3]. The process of soil formation and development, in fact, can be strongly influenced by human activity in many parts of the world [4,5].
In soil sciences, this awareness has been assumed by the World Reference Base [6], that has introduced the concept of soils developed by strong human influence due to long and intensive agricultural use (Anthrosols) or soils containing significant amounts of artefacts and trace elements acc+umulation (Technosols) [4,6,7]. Similarly, the Soil Taxonomy classification has highlighted the relevance of the “anthropic epipedon”, as soil epipedons characterized by mineral soil material that shows evidence of the purposeful alteration of soil properties or of earth-surface features [8,9].
According to these definitions, anthropic soils are drastically disturbed pedons developed under intense human pressure [10,11] and include soil materials formed by stripping of the natural soil, deposition of refuse or spoil, or by severe soil mixing [12,13,14].
The interaction between human activities and natural processes may promote negative effects on soil, and the understanding of these effects is becoming more and more relevant for the correct management of soils for future generations [15,16,17].
For instance, a wrong soil manipulation, e.g., soil deforestation, liming, intense irrigation practices, use of biocides, etc., may lead to different kind of soil degradation and to the reduction soil fertility and stability [18]. Waste material disposal may lead to deep soil acidification/alkalinization, making soil unhospitable for several organisms and inducing the release toxic elements on the underground waters [19,20].
Metals mine areas have been strongly exploited over time, and in these areas the genetic of soils has drastically changed [4,5,21,22]. Soils developed in these areas or on mine tailings rich in heavy metals generally result as strongly acidic areas as consequence of Acid Mine Drainage [23,24,25]. In addition, these soils may present a very high content of weathered oxidic materials that induce a net positive charge of soil and a low sorption capacity [26,27].
These conditions may also change the metal chemical speciation, thus increasing their mobility, leaching capacity and toxicity [28,29] with important consequences in the ecological equilibrium of the environment.
In this “Anthropocene Era”, one of the roles of soil scientists for the sustainable development goals is to assess the quality of our soils and attempt to foresee the best soil management for both environmental and human protection [30]. Soil research has largely increased the possibility to improve soil quality through controlled soil development of Technosols. The knowledge of soil pedogenesis in these areas can be very useful to land reclamation operations in mining areas [31,32], or in abandoned industrial one [33].
In the Italian context, the Apuan Alps (Central Italy) represent an example of a deep human excavation and mining activity. In the Baccatoio River Valley (Municipality of Pietrasanta, Tuscany) sulfosalts mineralization originated by low-grade metamorphism of previous sedimentary rocks [34]. These rocks, contain several orebodies, made of pyrite, barite, iron oxide rich in heavy metal such as Pb, Zn, Ag, [35]. This area has been exploited for decades for extraction of barites used as a heavy additive in oil-well-drilling mud for oil extraction [36,37]. During this time, waste material of different nature (e.g., mine gangue, mineral flotation) have been deposited in the surrounding area, leading to weathering processes that favored the mobilization of trace elements included in the sulfosalts, and to soil development.
In the last years concerns about the security of the area have grown among the population because of the risk of water and soil contamination, that can deeply affect agricultural products, drinkable waters, and so on.
The serious risk linked to the potential availability of heavy metals released by pyrite oxidation processes is therefore a priority to assure the best soil management of the area [25].
It is largely known that heavy metals availability in soils depends on many abiotic and biotic factors, such as soil pH, soil organic matter dynamics and many other soil physicochemical characteristics [38,39].
In a so complex soil forming context, the pedological approach may be very useful to assess the potential risk of the area, starting from the study of the anthropic origin of these soils.
The aim of this study was to study and classify the soils generated from different kind of mine tailings in the Apuan Alps. In addition, as these soils were very rich in heavy metals, the aim of the paper was also to assess the different environmental risk linked to the bioavailability of the potentially toxic elements (PTEs) present in these soils, according to their origin.
2. Materials and Methods
2.1. Study Area
Pietrasanta is a small village in Tuscany (Lucca District, central Italy) located in the Apuan Alps. This area is characterized by several tectonic-stratigraphic units involved in the Northern Apennines nappe stack [40,41]. The basement is mostly composed by carbonatic rocks and cataclastic breccia, covered by different orebodies made of pyrite, barite and iron oxides rich in potential toxic elements such as Pb, Zn, Ag and Tl. The area has an historical vocation to mining activity. Since Etruscan and Romans era, the area has been exploited for iron and silver extraction, while starting from the XVIII century different industrial mining activities has been placed for the extraction of other metals. After the first world war the main interest of the extracting companies was linked to the high presence of barites, uses as a heavy additive in oil-well-drilling mud for oil extraction and in the cement buildings of nuclear power stations. During these years, extraction, purification and transformation of barite was a common practice in the area, and mining tailings were usually discharged in the surrounding area. In the 80s, the use of barites in these productive processes was dismissed and the mines were abandoned, with the consequent non-management of the related environmental consequences.
The area is characterized by udic, mesic pedoclimatic regime [42]. Soil sampling campaign was conducted in autumn 2015–2016 in different parts of the Pietrasanta municipality (Figure 1), where the waste materials were disposed. In particular, soil profiles were distributed in the northern part of the village, next to the ancient mine ores, in the surrounding forest and in the low part of the valley.
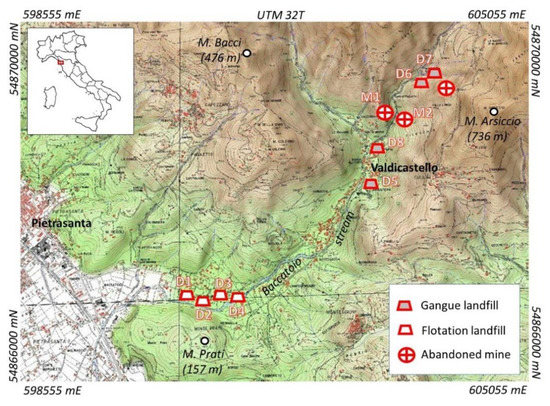
Figure 1.
Map of the study area and georeferenciated sampling sites (WGS-84 UTM 32T coordinates).
2.2. Soil Sampling and Analysis
Soil profiles were excavated in different part of the valley, and the sampling points were chosen according to the typology of the mine tailings deposited. In particular, M1 and M2 sites represent soils developed on mining rocks deposits, close to the abandoned mine ores (GR1); D5, D6, D7 and D9 sites represent soils formed on coarse debris of mining landfill or on altering mine debris (GR2); D1, D2, D3 and D4 sites represent soils formed on mining flotation deposits (GR3). All details are reported in Table S1, while examples of some typical soil profiles are displayed in Figure S1.
Soil profiles were observed in order to identify their genetic horizons and A-AC-C sequence was identified. Each soil horizon was described in field according to Schoeneberger et al. [43] and soil horizon samples were collected in duplicate for laboratory analysis.
Soils samples were dried at room temperature and sieved a 2 mm before analysis. All laboratory analyses were performed in triplicate and the accuracy was checked with standard deviation at 95% of replicability. Texture analysis were performed with the pipette method according to Gee and Bauder [44], soil pH was measured on 1:2.5 (w:v) soil:water solution. Total lime (CaCO3) content was quantified by volumetric method [45] but soils presented almost no carbonate content (data not shown). Soil organic matter was measured by loss of ignition according to Schulte and Hopkins [46] and the total organic carbon (TOC) was calculated using a 1.72 factor [47].
A finely grounded aliquot of each sample (0.25 g) was weighed in Teflon containers and digested with aqua regia (suprapure HNO3 and HCl 1:3, v:v, Fukla) in a microwave oven (Start D 1200, Milestone, Sorisole (BG)-Italy) for 3 min at 250 W, 4 min at 450 W and 3 min at 700 W. After this treatment, the solution was collected in 20 mL volumetric flasks and filtered through Watmann 42 filter paper. The concentration of total macro nutrients and heavy metals were then recorded by Inductively Coupled Plasma Emission Spectrometer (ICP-OES, Ametek, Spectro Arcos, Zeloforamagno (MI), Italy). Chemical analyses have been calibrated and crosschecked against international references materials and laboratory internal standards, as described by Vittori Antisari et al. [48].
The available fraction of metals was extracted by sequential dissolution technique.
For metal partitioning, metals were extracted sequentially from 1 g of 2 mm soil samples by following the method of Ferronato et al. [38] with some modification: being acidic soils, in fact, phase I, extracting pH dependent metals, was performed with 0.1 M CH3COONa (pH 5). Phase II, extracting metals associated to oxides and oxyhydroxides (reducible), was performed in 0.1 M NH2OH·HCl in 25% CH3COOH w/v (pH 2) while phase III, extracting metals associated to organic matter and suphides (oxidable), was performed in 30% H2O2 + 3 mL of 0.02 M HNO3 and 1 M CH3COONH4. Finally phase IV, which records the residual metals associated to the mineral soil particles, was calculated as the difference between pseudo total metal concentration in aqua regia and the sum of the metal content previously extracted. All solutions were then analyzed by Inductively Coupled Plasma Emission Spectrometer (ICP-OES, Ametek, Spectro ArcosZeloforamagno (MI), Italy).
2.3. Statistical Analysis
Data analysis were analyzed as mean values of the replicates of the original parent material of the area that composes the C soil horizons, and of the above soil horizons (A and AC horizons) where it was possible to observe the pedogenetic soil evolution according to the mine tailing deposited. Data of A and AC horizons were analyzed as mean values in order to highlight significant changes among soil groups.
All statistical analysis were assisted by Statistica 10 software (Stat Soft). The analysis of variance was performed with Krukal-Wallis test, and the significance was checked with p-value < 0.05. A principal components and classification analysis (PCCA) was performed to find the relationships among variables and cases (samples).
3. Results
3.1. Soil Morphological and Physicochemical Features
Soils were little developed and generally presented a A-AC-C sequence (Table 1). GR1 presented meanly 5/10 cm of A horizon, while GR2 and GR3 generally showed a thinner A horizon (ranging from 2 to 7 cm) (Table 1).

Table 1.
General features and morphology of the Technosols within the Baccatoio basin, Pietrasanta, Italy. For symbols see legend [43].
The horizon limits, generally, were abrupt/smooth or abrupt/wavy, highlighting the very little human manipulation of these soils after the first mining tailing disposal, and the low soil development. Rock fragments were consistent in GR 1 profiles (up to 70%) and decreased in GR 2 (20–68%), while in GR 3 soils, very few rock fragments were observed (up to 20%) together with silty horizons.
Weak fine granular soil structure was observed in most of the A horizons of GR2 and GR3 soils, followed by a weak fine angular or subangular structure in the deeper horizons (e.g., GR2) or moderate medium platy (e.g., GR3). Diversely, GR1 soils had almost weak fine angular or subangular structure.
Table 2 presents the main physicochemical characteristics of the C horizons (parent material) of the soil groups divided according to the different kind of mine tailings deposited. All parameters presented a high intra-group variability and no significant differences were highlighted. Soil texture was sandy loam in all groups, and high content of S (up to 17 g kg−1) and Ba (up to 2500 mg kg−1) was also recorded.

Table 2.
Physicochemical characteristics’ mean and standard deviation (SD) of soil samples collected from the C horizons of soil profiles in mining rocks deposits (GR1), soils formed on coarse debris of mining landfill or on altering mine debris (GR2), soils formed on mining flotation deposits (GR3).
As expected, the contribution of microelements such as As, Pb and Tl was consistent and in different samples the content of these element overhead the threshold limit of Italian law for soil residential and even industrial use (D.lsg 152/2006: 20, 100, 1 mg kg−1 respectively (residential use) and 50, 1000, 10 mg kg−1 respectively (industrial use) [49]. Notably, even Zn content was particularly high, and all samples were higher that the legislative threshold for soil residential use (D.lsg 152/2006: 150 mg kg−1) [49].
Table 3 shows the physicochemical characteristics of soil obtained by mean of A and AC horizons of the different soil groups.

Table 3.
Physicochemical characteristics’ mean and standard deviation (SD) of soil samples collected from the different soil profile (mean value of A and AC horizons) of soil profiles in mining rocks deposits (GR1), soils formed on coarse debris of mining landfill or on altering mine debris (GR2), soils formed on mining flotation deposits (GR3). Different letters within the row (a, ab and b) indicate significant differences among the values (p < 00.5).
All horizons of soil groups showed a sub-acid or acid pH, and the acidity generally increased from GR1 to GR3, even if not significant. Total organic carbon was less than 100 g kg−1 in all groups, with exception of some samples of GR2 group, and soils were generally poor in Ca content. Carbonates were absent, coherently with the low pH values and most macronutrients did not show significant differences among groups.
GR1 horizons showed a sandy loam texture, similarly to GR2 soil, and presented significantly higher Cu amount than GR3 ones without changes with depth (meanly 59 mg kg−1) and with values lower than the threshold of 120 and 600 mg kg−1 reported in Italian legislative Decree for both residential and industrial use [49]. GR1 A and AC soil horizons also showed three to four times significantly higher amount of Pb and Zn (1096 and 976 mg kg−1, respectively) than both GR2 and GR3 soils and high concentration of geogenic elements (e.g., Ba and S) was also determined.
GR2 soil horizons showed significant higher concentration of P, Fe and As than GR1 as well as higher Ni amount than that found in GR3.
Interesting to note that GR3, which showed a silty loam texture, presented the lowest content of both macroelements (e.g., S) and of PTEs (e.g., As, Cu, Ni and Zn).
Figure 2a displays the distribution of variables used in the PCCA model in the orthogonal graph according to their correlation. The first group of active variables represent the mineral and macro elements concentration of the soils (blu lines), while the second supplementary group of variables represents the contribution of the PTEs in discriminating the three soil groups (red lines).
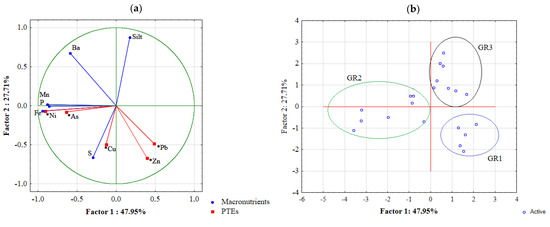
Figure 2.
Principal component and classification analysis output: (a) Scatterplot of variables correlations; (b) Scatterplot of case correlations.
The first factor equation extracted by the analysis of variables explained 48% of the variance and highlighted the good correlation between Zn and Pb, in the positive sector while on the negative sector, P was strongly associated to Fe, Ni and As; similarly, Cu and S were highly correlated.
The second factor function extracted 28% of the total variance of the population. This function highlighted the correlation of Silt and Ba against all the other variables.
Given the variable distribution in the orthogonal graph, Figure 2b display the case distribution by using the factor scores extracting by the two functions and could thus be correlated with the variable distribution. This method allowed to confirm that soils of GR1 differed from the other soils for their high content of Zn and Pb and for the lower content of Ba, while GR2 samples could be discriminated for the higher content of P associated to Fe minerals, rich in Ni and As. Finally, the PCCA highlighted the diversity of GR3 soil horizons mostly due to the high silt component and to the lower content of PTEs with respect to the other soil groups.
3.2. Potentially Toxic Elements Sequential Extraction
Figure 3 displays the percentage of selected metals availability in soil (A and AC horizon) according to the affinity of the metals for different processes occurring in soil (e.g., Phase 1—weak absorption and pH change; Phase 2—oxides and oxyhydroxides bound or reducible processes; Phase 3—soil organic matter/sulphides bound or oxidable processes; Phase 4—stability to the mineral phase or residual ph.

Figure 3.
Histograms of the Cu, Ni, Pb and Zn sequential extraction (a–d) respectively in the topsoil horizons (A and AC) of soil profiles in mining rocks deposits (GR1), soils formed on coarse debris of mining landfill or on altering mine debris (GR2), soils formed on mining flotation depos-its (GR3).
The sequential analysis showed that out of the residual and stable phase, the potential available Cu was up to 40% in GR1 soils and decrease up to 20% in GR2 and GR3 one. Most of the Cu present in soil was found in the oxidable phase. The highest percentage was present in GR1 as (30%, p < 0.05) and it decreased to 13% in GR 2 and GR3, highlighting its affinity with soil organic matter and sulphides. Notably, up to 7% of Cu was also present in exchangeable or pH dependent phase in GR3 soils.
Potential available Ni ranged between 20 and 30% in the different soils. Notably, Ni was generally weakly bounded in GR2 soil horizons (12%). Diversely, in GR3, Ni was mostly bounded to oxides and oxyhydroxides (14%).
Out of the residual and stable phase, 25% of Pb was found in available or potentially available form in GR1 epipedons (p > 0.05), decreasing to 15% and 13% in GR2 and GR3 respectively. Most of the potential available Pb was found in Phase II (GR1 > GR2 > GR3), which recorded Pb linked to oxides and hydroxides, and in Phase III (GR1 < GR3 < GR2) which recorded Pb linked to weathered minerals (e.g., PbS).
Zn showed a greatest potential availability among the analyzed elements, ranging between 45% in GR2 soils (p < 0.05) to 35% in GR1 and GR3 soil horizons. Notably, up to 20% of exchangeable Zn (Phase I) was found in GR2 soils, while in GR3 soils Zn was mostly associated to Phase III which recorded Zn associated to soil organic matter or sulphides.
4. Discussion
According to the type of the mine tailings deposited, the World Reference Base for Soil Resources [6] allows to classify these soils as Technosols as they contain more than 20% mine artefacts transported and deposited to the surface directly or after transformation by industrial activities. All the investigated soils were classified Technosols toxic having in some layer within 50 cm of the soil surface inorganic materials with high concentrations of toxic elements. Leptic Hyperskeletic Technosols (Toxic) soils were found in those stony areas where soil developed on abandoned mining area (GR1) having <20% (by volume) fine earth over continuous rock starting <75 cm from soil surface. Spolic Technosols (Toxic, Transportic) were classified in those areas where soils were characterized by transported mine gangue materials (GR2), containing >35% (by volume) mine debris that have been transported from the mining area. Isolatic Technosols (Toxic, Relocatic) were described in those areas where soils were characterized by deposition of mineral flotation materials derived by rock manipulation (GR3), limited to defined areas, and covered and remodeled in situ with fill soil.
As underlined by the soil classification, soils were very little developed and were all characterized by the presence of high content of PTEs. The high PTEs concentration was expected as these soils resulted in an abandoned mining area, characterized by pyrite, barite, iron oxide rich in heavy metal [50,51,52]. As soils showed acidic pH, there is a serious risk of metals leaching into underground water, with the consequent spread of the metal contamination in the surrounding area. Concern was arisen for the high content of As Pb, Zn and Tl (up to 297, 1096, 1081 and 143 mg kg−1, respectively), that exceed the threshold limit defined by Italian law [49], but also for the high concentration of other potentially toxic elements such as Ag, Ni and Cu (up to 29, 49 and 135 mg kg−1 respectively). According to the sequential partitioning of metals in A and AC horizons more information about potential risk linked to the high concentration of PTEs were recorded.
Soils of GR1 were characterized by the highest content of Cu, Pb and Zn, linked to the presence of low-weathered pyrites minerals. Pyrite, in fact, is the most common sulfide associated with other sulfide minerals such as sphalerite (ZnS), chalcopyrite (CuFeS2), and galena (PbS) [53] and in this area such minerals are very common as confirmed by D’Orazio et al. [50]. These soils are also characterized by a lower Ba content, probably because they are the only natural mine soils that do not contain anthropic Ba accumulation occurred in the other soils as results of tailings manipulation.
Soils of GR2, developed on gangue deposits, recorded the highest content of TOC and P. This result is probably related to the position of these soils, almost located in natural forested area, that promote wild vegetation growth and accumulation of fresh organic matter [54,55]. In addition, GR2 soils showed the highest content Fe and Mn, which are probably present as oxide and hydroxides, as the results of pyrite oxidation. The steps of pyrite oxidation due e.g., to atmospheric contact, oxygenated waters, soil humidity, etc., may lead to soil acidification [56,57] and to different reactions according to the activation energy of the site and redox potential. According to Dos Santos et al. [53], the pyrite weathering at low Eh potential and low activation energy, may lead to oxidation of Fe(II) to Fe(III)–OH– species, while in presence of higher activation energy the weathering product may lead to the formation of S═O bond and Fe(II)–OH2 species. As a result of mineral oxidation, the total concentration of S-linked metal such as Pb and Zn were significantly lower in GR2 compared to GR 1 soils, but their potential availability is higher. In fact, as confirmed by the analysis of sequential-partitioning metals, both metals are mostly linked to exchangeable or Eh-dependent soil phases usually linked to the presence of oxides and oxyhydroxides and to soil organic matter (SOM).
GR 3 soils group resulted to be the less contaminated soils from the point of view of PTEs total concentration, coherently with the fact that these soils resulted from flotation deposits after the separation of pyrite from the associated metals [58]. The analysis of metal sequential partitioning (Figure 3) showed that these critical metals were much less available in GR3 soils than in GR1 (e.g., Cu and Pb) confirming that GR 3 soils have been already “washed”. Beside the acidic pH, which is one of the most important factors driving metals mobility [59], the low content of clay and organic matter in these soils can be considered other important elements that promote metals leaching, thus increasing the vulnerability of these soils to release heavy metals into the surrounding environment. On the other hand, the sequential extraction showed that in GR3 soils the available Zn and Ni is mostly bound to soil oxides and oxyhydroxides (e.g., Ni) and to organic matter and sulphides (e.g., Zn).
These metals in fact, are the product of human rock manipulation (ex situ mineral alteration processes); consequently, the high soil hazard in GR3 could be partially controlled by SOM and by metal alteration products, which are essential soil driving factors, and play an important role in metals chelation. In order to assess the environmental risk of the area and to avoid future water contamination, these soils and the underneath water should be strictly monitored during time. Given the important role of SOM in chelating heavy metals, promoting soil organic matter enrichment could be a successful method to restore these sites and control soil development.
The higher Zn and Ni availability in GR2 and GR1 soils than in GR3 one could be due to the affinity of such metals to the pH dependent phase. These metals are products of in situ mineral alteration processes that characterize the pedogenesis in these areas, and are easily still available. For this reason, urgent reclamation operation should be necessary in these areas.
5. Conclusions
In abandoned mining areas soils developed because of both human-induced and natural processes. In our case, the kind of deposited waste material strongly influenced soil physicochemical characteristics of soils, and this is reflected on the analysis of pseudo-total elements in the different soil groups. According to the material deposited, however, the pedogenesis can differ according to the degree of mineral alteration (e.g., pyrite oxidation, oxides and oxydroxides formation, organo-mineral complex formation) leading to different level of soil vulnerability to heavy metals release. Our study demonstrated as the analysis of pseudo-total elements in soil is not enough for assessing the environmental risk in the area, and that partial dissolution techniques may explain much better the mobility of the elements linked to the mineral or organo-mineral complexes that they form in situ.
As a result, in this specific case, GR1 soils do not results the highest contaminated soils for pseudo-total content of heavy metals, but they show the highest environmental risk from the point of view of metal mobility (e.g., for Cu, Ni, Pb and Zn), while GR2 soils, which are characterized by the highest Ni, Pb and Zn content results to be less environmentally critical from the point of view of metal mobility. In fact, although the potentially available metals resulted from the alteration of pyrite minerals, the presence of a good SOM content and the pH around 6 can act as powerful soil buffer that prevent metal leaching.
Finally, GR3 characterized by the lowest concentration of PTEs and the lowest metal mobility behavior but with the lowest pH and SOM content, can be considered environmentally critical because of the high potential metal leacheability. In these areas, in fact, the low SOM content and the low soil pH, enhance the potential risk of metal mobility in the surrounding area (e.g., in the underground water).
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/app11156982/s1, Figure S1 Representation of some typical landscape and soil profiles treated in this study; Table S1 Georeferenciated location and description of the investigated soils.
Author Contributions
Writing original draft, C.F.; writing review and editing, G.V., M.D.F., and L.V.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research was partially funded by Pietrasanta Municipality (Lucca, Tuscani), grant n. 3656/2015.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Certini, G.; Scalenghe, R. Anthropogenic soils are the golden spikes for the Anthropocene. Holocene 2011, 21, 1269–1274. [Google Scholar] [CrossRef]
- Crutzen, P.J. Geology of mankind. Nature 2002, 415, 211–215. [Google Scholar] [CrossRef]
- Crutzen, P.J.; Stoermer, E. The ‘Anthropocene’. Glob. Chang. Newsl. 2000, 41, 17–18. [Google Scholar]
- Dazzi, C.; Monteleone, S. Anthropogenic processes in the evolution of a soil chronosequence on marly-limestone substrata in an Italian Mediterranean environment. Geoderma 2007, 141, 201–209. [Google Scholar] [CrossRef]
- Burghardt, W. Soils in urban and industrial environments. Z. Für Pflanz. Und Bodenkd 1994, 157, 205–214. [Google Scholar] [CrossRef]
- IUSS Working Group WRB. World Reference Base for Soil Resources 2014, Update 2015 International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; World Soil Resources Reports No. 106; FAO: Rome, Italy, 2015; ISBN 978-92-5-108369-7. [Google Scholar]
- Food and Agriculture Organization of the United Nations. World Reference Base for Soil Resources; Food and Agriculture Organization of the United Nations: Rome, Italy, 1998; ISBN 92-5-104141-5. [Google Scholar]
- Soil Survey Staff. Keys to Soil Taxonomy, 12th ed.; United States Department of Agriculture, Natural Resources Conservation Service: Lincoln, NE, USA, 2014. [Google Scholar]
- ICOMANTH. Circular Letter. 2003; Volume 4. Available online: https://www.nrcs.usda.gov/wps/portal/nrcs/detail/soils/survey/class/taxonomy/?cid=stelprdb1262283 (accessed on 30 January 2006).
- Blum, W.E.H. Forms of Energy Editorials Forms of Energy Involved in Soil and Sediment Processes. J. Soils Sediments 2008, 8, 1–2. [Google Scholar] [CrossRef]
- Papa, G.L.; Antisari, L.V.; Vianello, G.; Dazzi, C. Soil interpretation in the context of anthropedoge-nic transformations and pedotechniques application. Catena 2018, 166, 240–248. [Google Scholar] [CrossRef]
- Soils Portal. 2020. Anthropic Soils [A] » New Zealand Soils Portal. Available online: https://soils.landcareresearch.co.nz/describing-soils/nzsc/soil-order/anthropic-soils/ (accessed on 23 December 2020).
- Schwenke, G.; Mulligan, D.R.; Bell, L.C. Stripping and replacement for the rehabilitation of bauxite-mined land at Weipa. I. Initial changes to soil organic matter and related parameters. Aust. J. Soil Res. 2000, 38, 345–369. [Google Scholar] [CrossRef]
- Tedesco, M.J.; Teixeira, E.C.; Medina, C.; Bugin, A. Reclamation of Spoil and Refuse Material Produced by Coal Mining using Bottom Ash and Lime. Environ. Technol. 1999, 20, 523–529. [Google Scholar] [CrossRef]
- Keesstra, S.; Mol, G.; de Leeuw, J.; Okx, J.; Molenaar, C.; de Cleen, M.; Visser, S. Soil-Related Sustainable Development Goals: Four Concepts to Make Land Degradation Neutrality and Restoration Work. Land 2018, 7, 133. [Google Scholar] [CrossRef] [Green Version]
- Lal, R.; Horn, R.; Kosaki, T. Soil and Sustainable Development Goals. 2018. US-ISBN: 1-59326-269-8. Available online: https://www.schweizerbart.de/publications/detail/isbn/9783510654253/Soil_and_Sustainable_Development_Goals_ (accessed on 25 July 2021).
- Huot, H.; Simonnot, M.-O.; Morel, J.-L. Pedogenetic Trends in Soils Formed in Technogenic Parent Materials. Soil Sci. 2015, 180, 182–192. [Google Scholar] [CrossRef] [Green Version]
- Dazzi, C.; Papa, G.L. Anthropogenic soils: General aspects and features. Ecocycles 2015, 1, 3–8. [Google Scholar] [CrossRef] [Green Version]
- Shaharoona, B.; Al-Ismaily, S.; Al-Mayahi, A.; Al-Harrasi, N.; Al-Kindi, R.; Al-Sulaimi, A.; Al-Busaidi, H.; Al-Abri, M. The role of urbanization in soil and groundwater contamination by heavy metals and pathogenic bacteria: A case study from Oman. Heliyon 2019, 5, e01771. [Google Scholar] [CrossRef] [Green Version]
- Naveen, B.P.; Sumalatha, J.; Malik, R.K. A study on contamination of ground and surface water bodies by leachate leakage from a landfill in Bangalore, India. Int. J. Geo-Eng. 2018, 9, 27. [Google Scholar] [CrossRef]
- Haering, K.C.; Daniels, W.L.; Galbraith, J.M. Mapping and Classification of Southwest Virginia Mine Soils. Soil Sci. Soc. Am. J. 2005, 69, 463–472. [Google Scholar] [CrossRef]
- Ciolkosz, E.J.; Cronce, R.C.; Cunnigham, R.L.; Petersen, G.W. Characteristics, genesis and classification of Pennsylvania Minesoils. Soil Sci. 1985, 139, 232–238. [Google Scholar] [CrossRef]
- El Amari, K.; Valera, P.; Hibti, M.; Pretti, S.; Marcello, A.; Essarraj, S. Impact of mine tailings on surrounding soils and ground water: Case of Kettara old mine, Morocco. J. Afr. Earth Sci. 2014, 100, 437–449. [Google Scholar] [CrossRef]
- Antunes, S.C.; de Figueiredo, D.R.; Marques, S.M.; Castro, B.B.; Pereira, R.; Gonçalves, F. Evaluation of water column and sediment toxicity from an abandoned uranium mine using a battery of bioassays. Sci. Total Environ. 2007, 374, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Clemente, R.; Walker, D.J.; Roig, A.; Bernal, M.P. Heavy metal bioavailability in a soil affected by mineral sulphides contamination following the mine spillage at Aznalcóllar (Spain). Biodegradation 2003, 14, 199–205. [Google Scholar] [CrossRef]
- Buch, A.C.; Niemeyer, J.C.; Marques, E.D.; Silva-Filho, E.V. Ecological risk assessment of trace metals in soils affected by mine tailings. J. Hazard. Mater. 2021, 403, 123852. [Google Scholar] [CrossRef]
- Audry, S.; Schäfer, J.; Blanc, G.; Jouanneau, J.-M. Fifty-year sedimentary record of heavy metal pollution (Cd, Zn, Cu, Pb) in the Lot River reservoirs (France). Environ. Pollut. 2004, 132, 413–426. [Google Scholar] [CrossRef]
- Liu, J.; Yin, M.; Xiao, T.; Zhang, C.; Tsang, D.C.W.; Bao, Z.; Zhou, Y.; Chen, Y.; Luo, X.; Yuan, W.; et al. Thallium isotopic fractionation in industrial process of pyrite smelting and environmental implications. J. Hazard. Mater. 2020, 384, 121378. [Google Scholar] [CrossRef]
- Kabata-Pendias, A. Trace Elements in Soils and Plants; CRC Press. Boca Raton Florida, Taylor and Francis: Boca Raton, FL, USA, 2010. [Google Scholar] [CrossRef]
- Fitzpatrick, R.W. Demands on Soil Classification and Soil Survey Strategies: Special-Purpose Soil Classification Systems for Local Practical Use. In Developments in Soil Classification, Land Use Planning and Policy Implications; Springer: Dordrecht, The Netherlands, 2013; pp. 51–83. [Google Scholar] [CrossRef]
- Benidire, L.; Madline, A.; Pereira, S.I.A.; Castro, P.M.L.; Boularbah, A. Synergistic effect of organo-mineral amendments and plant growth-promoting rhizobacteria (PGPR) on the establishment of vegetation cover and amelioration of mine tailings. Chemosphere 2021, 262, 127803. [Google Scholar] [CrossRef]
- Santini, T.C.; Fey, M.V. From tailings to soil: Long-term effects of amendments on progress and trajectory of soil formation and in situ remediation in bauxite residue. J. Soils Sediments 2018, 18, 1935–1949. [Google Scholar] [CrossRef]
- Antisari, L.V.; Papa, G.L.; Ferronato, C.; Falsone, G.; Vianello, G.; Dazzi, C. In situ remediation of polluted Spolic Technosols using Ca(OH)2 and smectitic marlstone. Geoderma 2014, 232–234. [Google Scholar] [CrossRef] [Green Version]
- Antisari, L.V.; Bini, C.; Ferronato, C.; Gherardi, M.; Vianello, G. Translocation of potential toxic elements from soil to black cabbage (Brassica oleracea L.) growing in an abandoned mining district area of the Apuan Alps (Tuscany, Italy). Environ. Geochem. Health 2020, 42, 2413–2423. [Google Scholar] [CrossRef]
- Biagioni, C.; D’Orazio, M.; Vezzoni, S.; Dini, A.; Orlandi, P.; Biagioni, C.; D’Orazio, M.; Vezzoni, S.; Dini, A.; Orlandi, P. Mobilization of Tl-Hg-As-Sb-(Ag,Cu)-Pb sulfosalt melts during low-grade metamorphism in the Alpi Apuane (Tuscany, Italy). Geology 2013, 41, 747–750. [Google Scholar] [CrossRef]
- Neff, J.M. Estimation of bioavailability of metals from drilling mud barite. Integr. Environ. Assess. Manag. 2008, 4. [Google Scholar] [CrossRef] [PubMed]
- Bruton, J.R.; Bacho, J.P.; Newcaster, J. The Future of Drilling-Grade Barite Weight Material-A Case for a Substitute Specification, in: SPE Annual Technical Conference and Exhibition. Soc. Pet. Eng. 2006. [Google Scholar] [CrossRef]
- Ferronato, C.; Vianello, G.; Antisari, L.V. Heavy metal risk assessment after oxidation of dredged sediments through speciation and availability studies in the Reno river basin, Northern Italy. J. Soils Sediments 2015, 15, 1235–1245. [Google Scholar] [CrossRef]
- Landner, L.; Reuther, R. Speciation, mobility and bioavailability of metals in the environment. In Metals in Society and in the Environment; Kluwer Academic Publisher: Dordrecht, The Netherlands, 2005; pp. 139–274. [Google Scholar] [CrossRef]
- Meccheri, M. Structural inheritance and style of reactivation at mid-crustal levels: A case study from the Alpi Apuane (Tuscany, Italy). Tectonophysics 2012, 579, 74–87. [Google Scholar] [CrossRef]
- Carmignani, L.; Kligfield, R. Crustal extension in the northern Apennines: The transition from compression to extension in the Alpi Apuane Core Complex. Tectonics 1990, 9, 1275–1303. [Google Scholar] [CrossRef]
- Vinci, A.; Gardin, L.; Ripiani, A. Legenda della Carta dei Suoli della Toscana in scala 1:250.000, Carta dei suoli d’Italia in scala 1:250,000. 2002. Available online: http://www.regione.toscana.it/documents/10180/14299445/7-La+carta+dei+suoli+della+Regione+Toscana.pdf/a6a5e77e-df09-4b8c-ac5e-084682355f46 (accessed on 25 July 2021).
- Schoeneberger, P.; Wysocki, D.A.; Benham, E.C.J. Field Book for Describing and Sampling Soils, Version 3.0; Natural Resources Conservation Service, National Soil Survey Center: Lincoln, NE, USA, 2012. [Google Scholar]
- Gee, G.W.; Bauder, J.W. Methods of Soil Analysis: Part 1—Physical and Mineralogical Methods; SSSA Book Series; Soil Science Society of America, American Society of Agronomy; Accademic Press: New York, NY, USA, 1986. [Google Scholar] [CrossRef]
- Loeppert, R.H.; Suarez, D.L. Carbonate and Gypsum; Publication from USDA-ARS/UNL Faculty: Lincoln, NE, USA, 1996; Chapter 15; 39p. [Google Scholar] [CrossRef]
- Schulte, E.E.; Hopkins, B.G. Estimation of Soil Organic Matter by Weight Loss-On-Ignition; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2015; pp. 21–31. [Google Scholar] [CrossRef]
- Abella, S.R.; Zimmer, B.W. Estimating Organic Carbon from Loss-On-Ignition in Northern Arizona Forest Soils. Soil Sci. Soc. Am. J. 2007, 71, 545. [Google Scholar] [CrossRef]
- Antisari, L.V.; Carbone, S.; Ferronato, C.; Simoni, A.; Vianello, G. Characterization of heavy metals atmospheric deposition for urban environmental quality in the Bologna city (Italy). EQA-Int. J. Environ. Qual. 2011, 7, 49–63. [Google Scholar] [CrossRef]
- Decreto legislativo 2006/152/ITA. Codice dell’Ambiente, Gazzetta Ufficiale n 88, 14 Aprile 2006, Supplemento Ordinario n 96. Available online: https://www.gazzettaufficiale.it/home (accessed on 25 July 2021).
- D’Orazio, M.; Biagioni, C.; Dini, A.; Vezzoni, S. Thallium-rich pyrite ores from the Apuan Alps, Tuscany, Italy:constraints for their origin and environmental concerns. Miner. Depos. 2017, 52, 687–707. [Google Scholar] [CrossRef]
- Biagioni, C.; Bonaccorsi, E.; Moëlo, Y.; Orlandi, P. Mercury-arsenic sulfosalts from Apuan Alps (Tuscany, Italy). I. Routhierite, (Cu0.8Ag0.2)Hg2Tl(As1.4Sb0.6)S=2S6, from Monte Arsiccio mine: Occurrence and crystal structure. Eur. J. Mineral. 2014, 26, 163–170. [Google Scholar] [CrossRef]
- Petrini, R.; Pennisi, M.; Antisari, L.V.; Cidu, R.; Vianello, G.; Aviani, U. Geochemistry and stable isotope composition of surface waters from the Ravenna plain (Italy): Implications for the management of water resources in agricultural lands. Environ. Earth Sci. 2014, 71, 5099–5111. [Google Scholar] [CrossRef]
- Dos Santos, E.C.; de Mendonça Silva, J.C.; Duarte, H.A. Pyrite Oxidation Mechanism by Oxygen in Aqueous Medium. J. Phys. Chem. C 2016, 120, 2760–2768. [Google Scholar] [CrossRef] [Green Version]
- Lair, G.J.; Gerzabek, M.H.; Haberhauer, G. Sorption of heavy metals on organic and inorganic soil constituents. Environ. Chem. Lett. 2007, 5, 23–27. [Google Scholar] [CrossRef]
- Stietiya, M.H.; Wang, J.J. Effect of organic matter oxidation on the fractionation of copper, zinc, lead, and arsenic in sewage sludge and amended soils. J. Environ. Qual. 2006, 40, 1162–1171. [Google Scholar] [CrossRef]
- Ferronato, C.; Falsone, G.; Natale, M.; Zannoni, D.; Buscaroli, A.; Vianello, G.; Vittori, L. Chemical and pedological features of subaqueous and hydromorphic soils along a hydrosequence within a coastal system (San Vitale Park, Northern Italy). Geoderma 2016, 265, 141–151. [Google Scholar] [CrossRef]
- Dent, D.L.; Pons, L.J. A world perspective on acid sulphate soils. Geoderma 1995, 67, 263–276. [Google Scholar] [CrossRef]
- Stirling, A.; Bernasconi, M.; Parrinello, M. Ab initio simulation of water interaction with the (100) surface of pyrite. J. Chem. Phys. 2003, 118, 8917–8926. [Google Scholar] [CrossRef]
- Houben, D.; Evrard, L.; Sonnet, P. Mobility, bioavailability and pH-dependent leaching of cadmium, zinc and lead in a contaminated soil amended with biochar. Chemosphere 2013, 92, 1450–1457. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).