Synthetic Transformations and Medicinal Significance of 1,2,3-Thiadiazoles Derivatives: An Update
Abstract
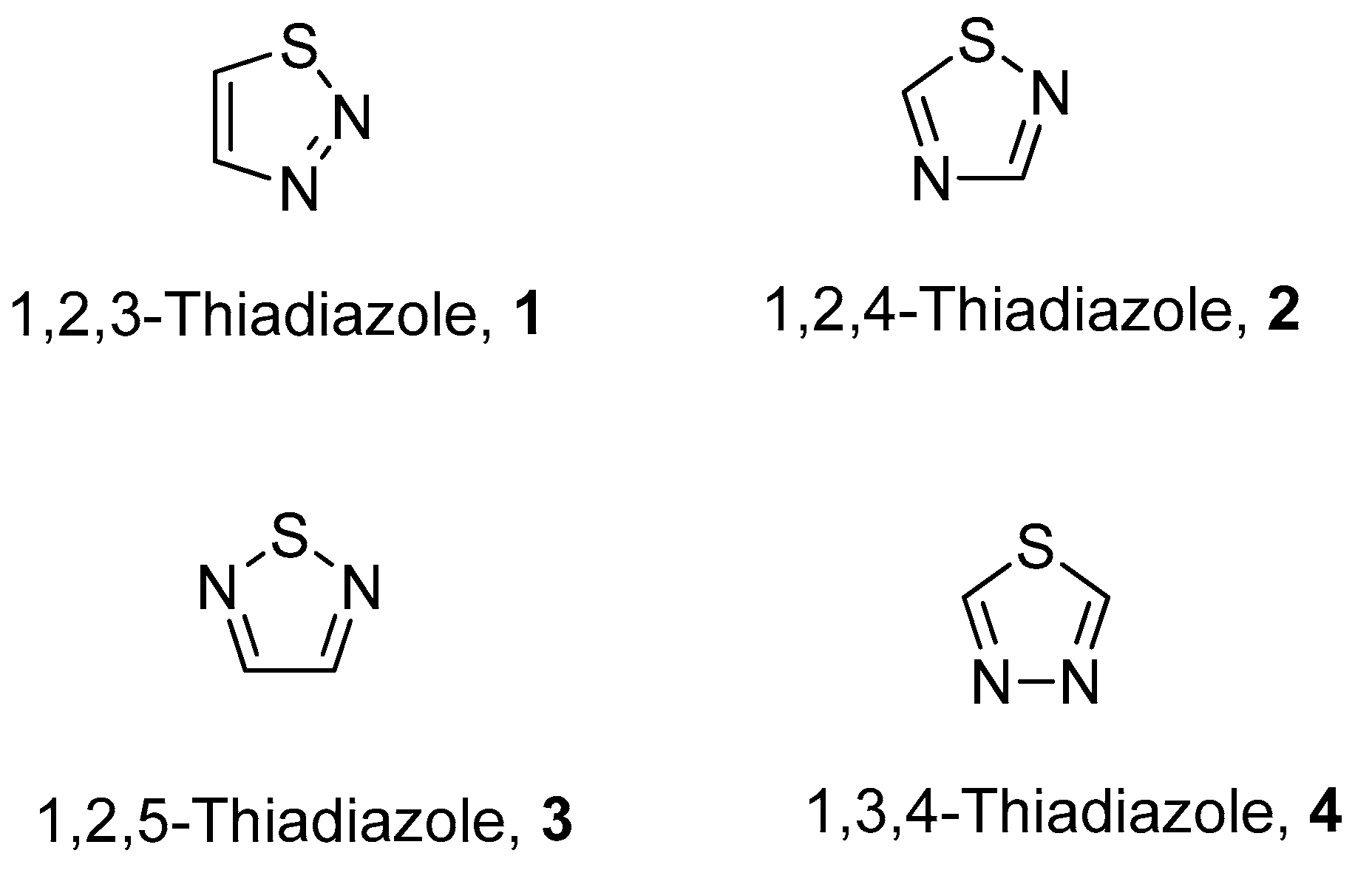
1. Introduction
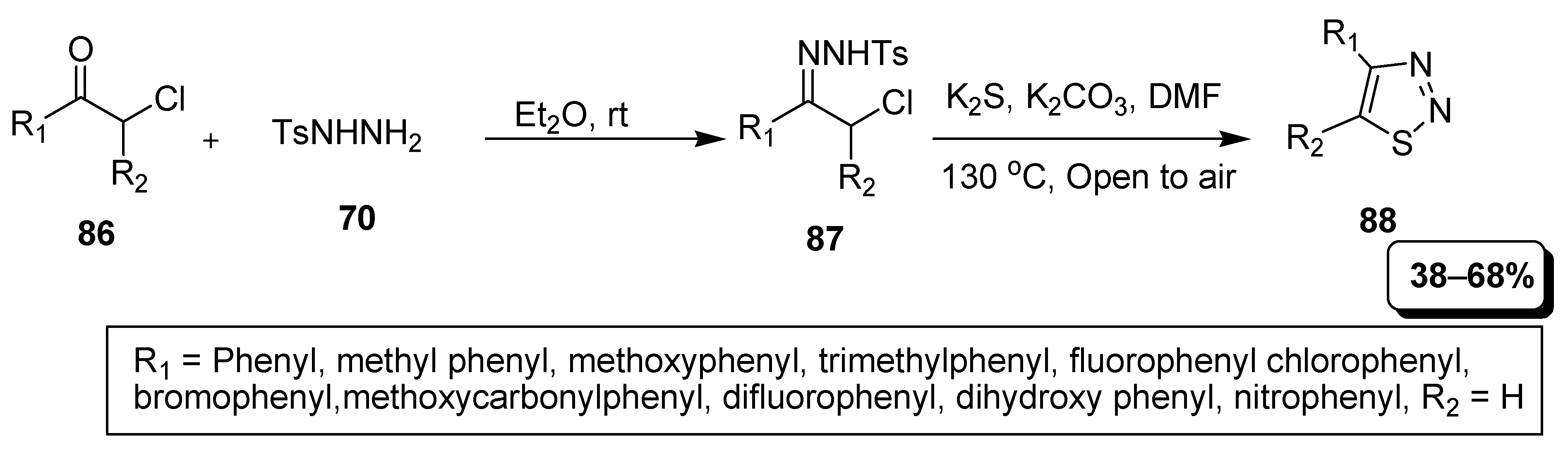
2. Synthetic Approaches
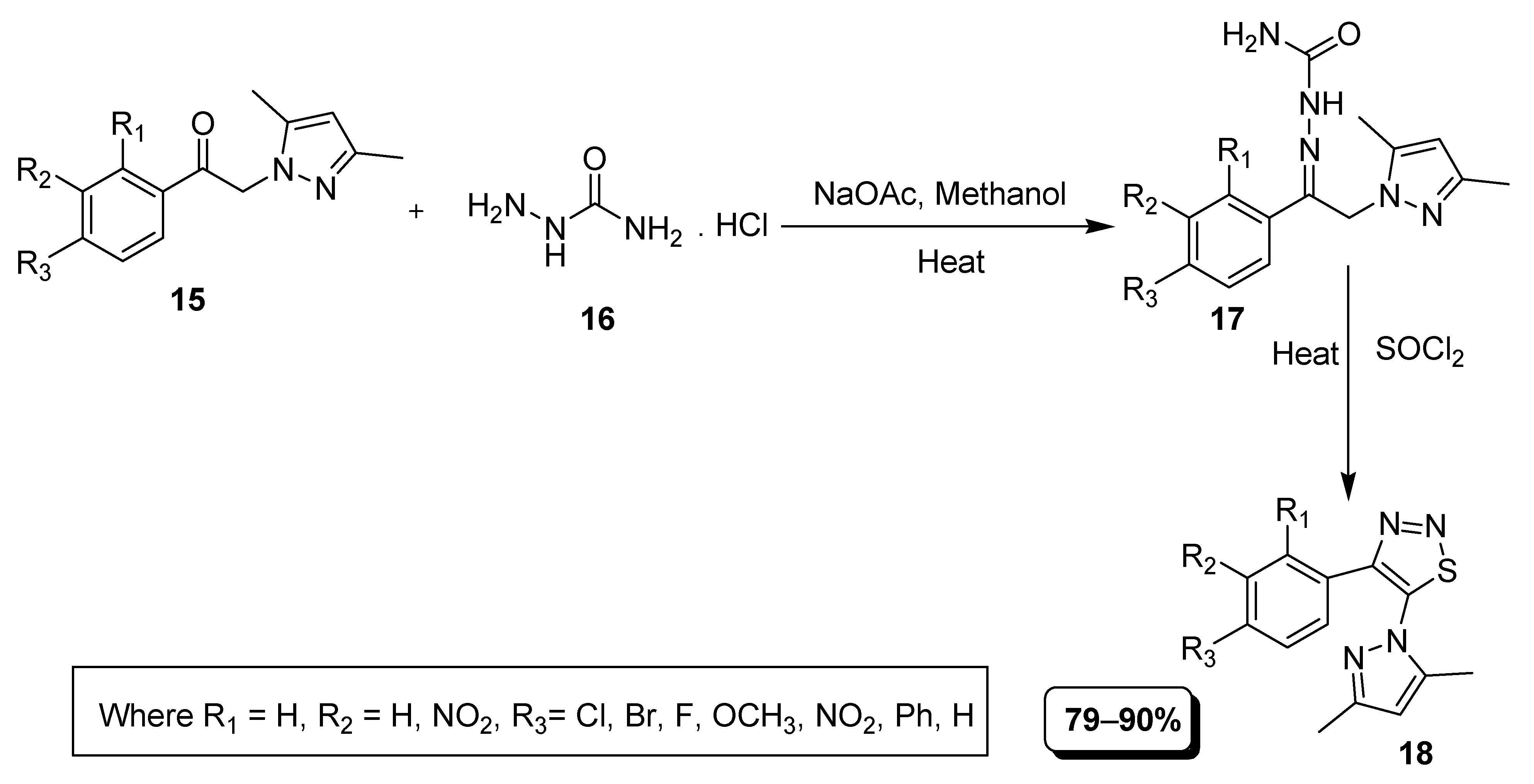
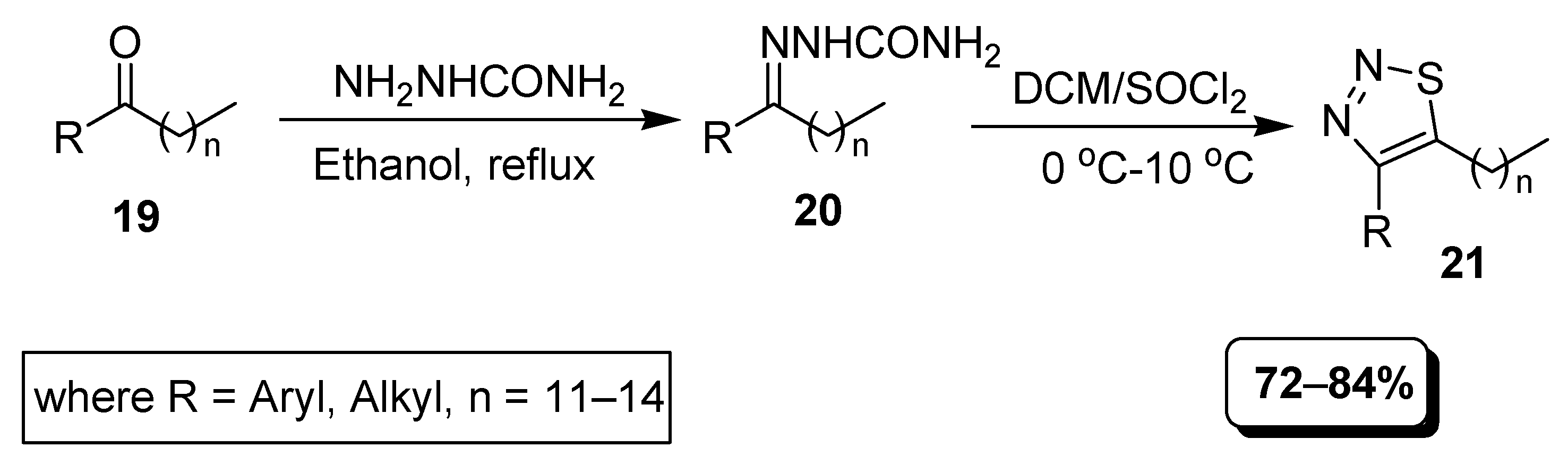
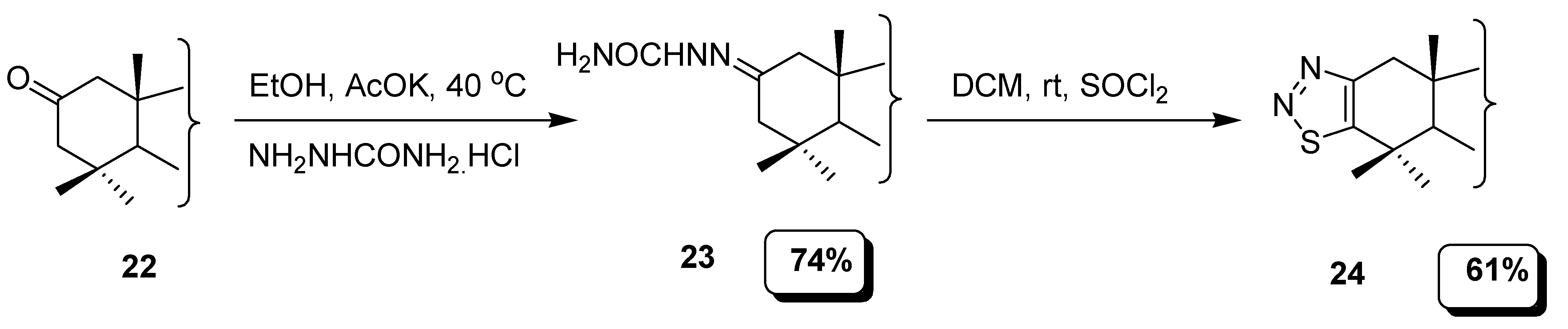
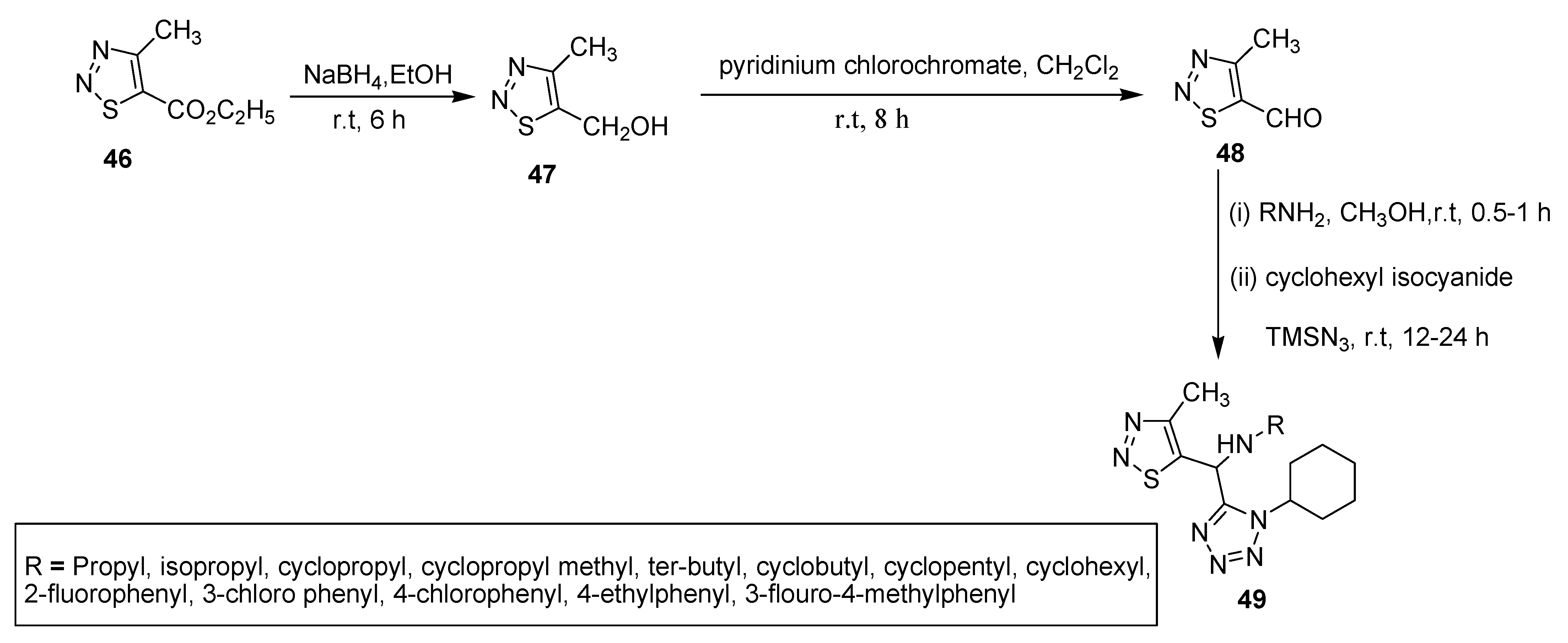
2.1. Hurd–Mori and Lalezari Cyclization
2.2. Microwave-Assisted Synthesis of 1,2,3-Thiadiazoles
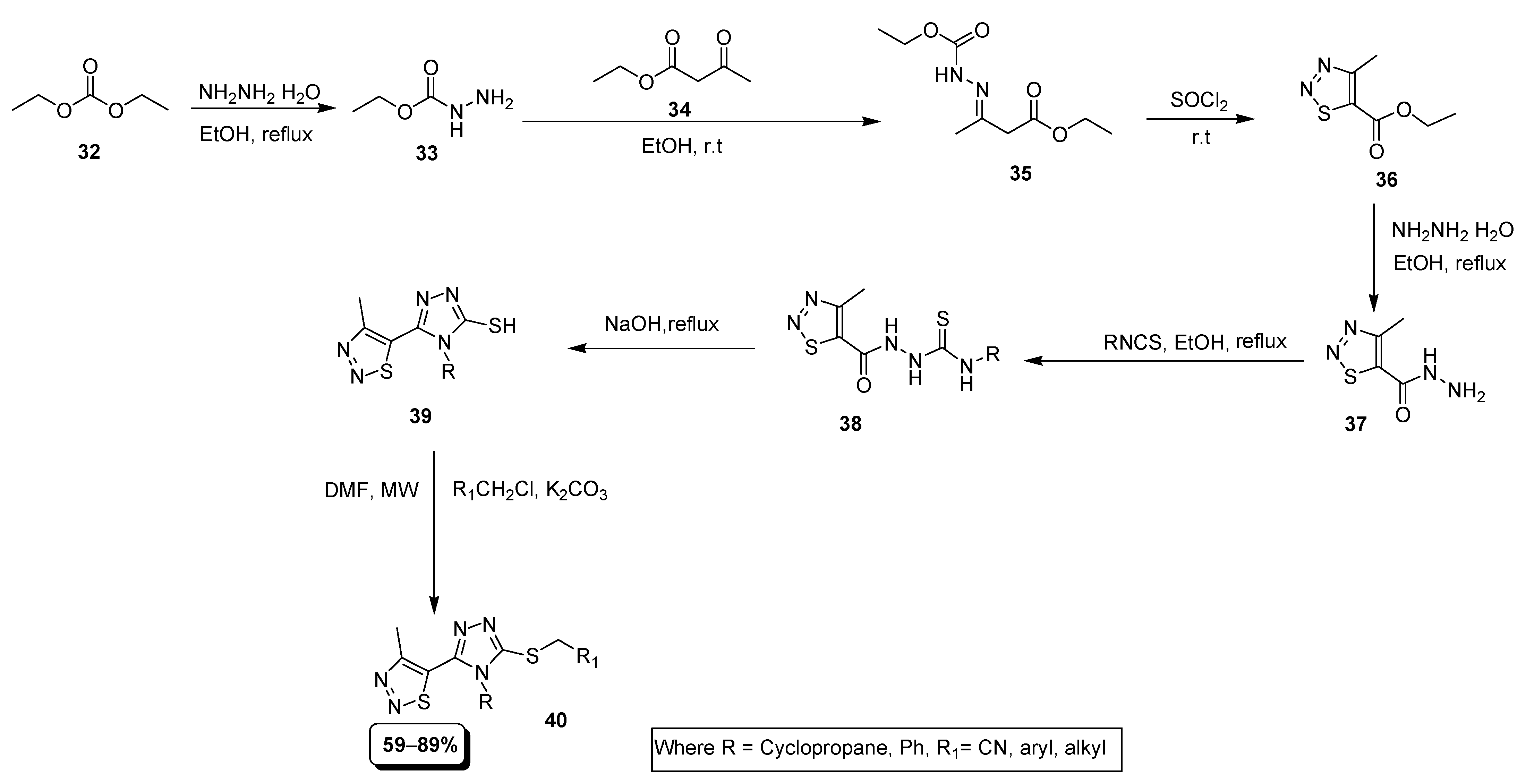
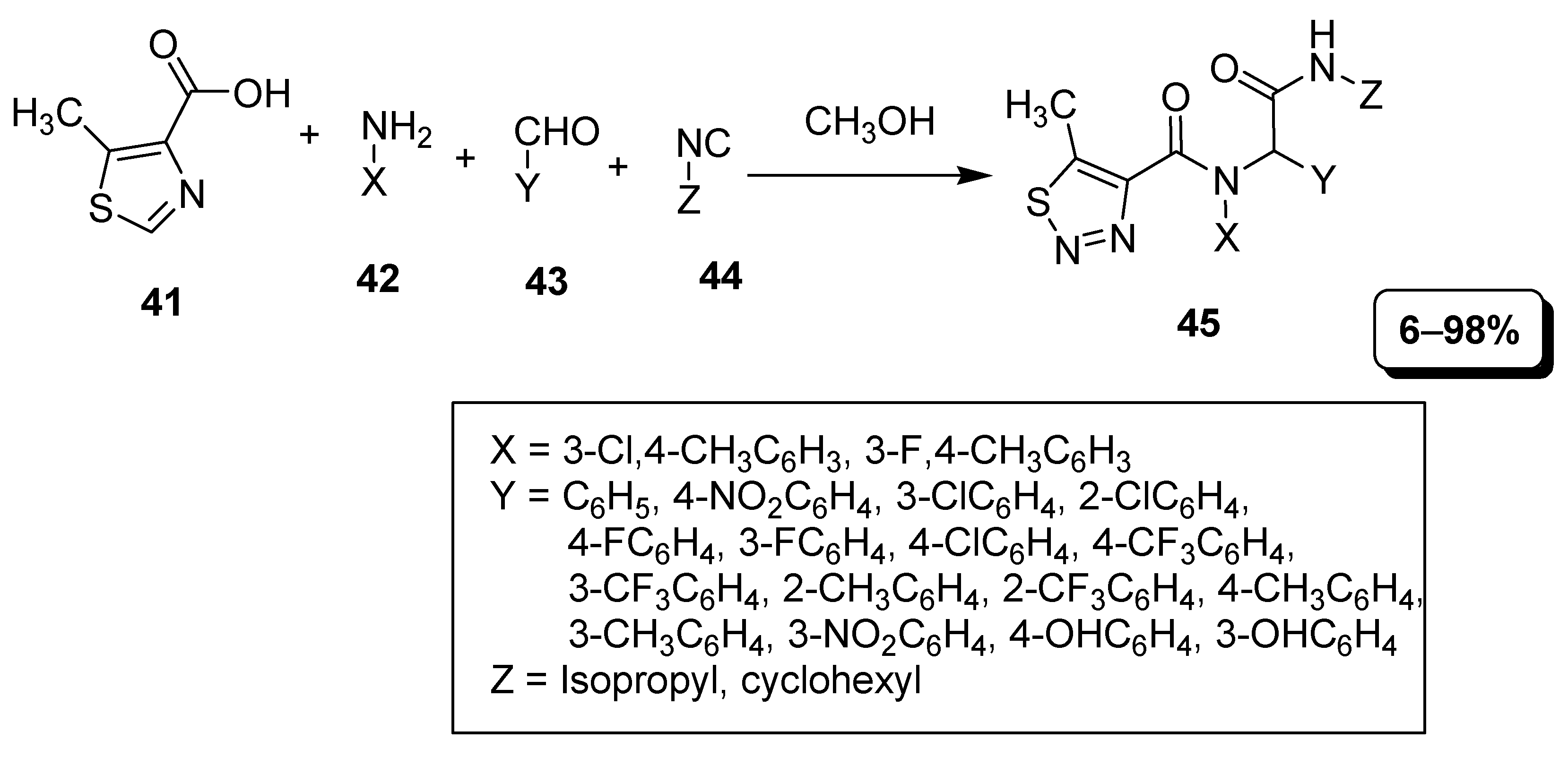
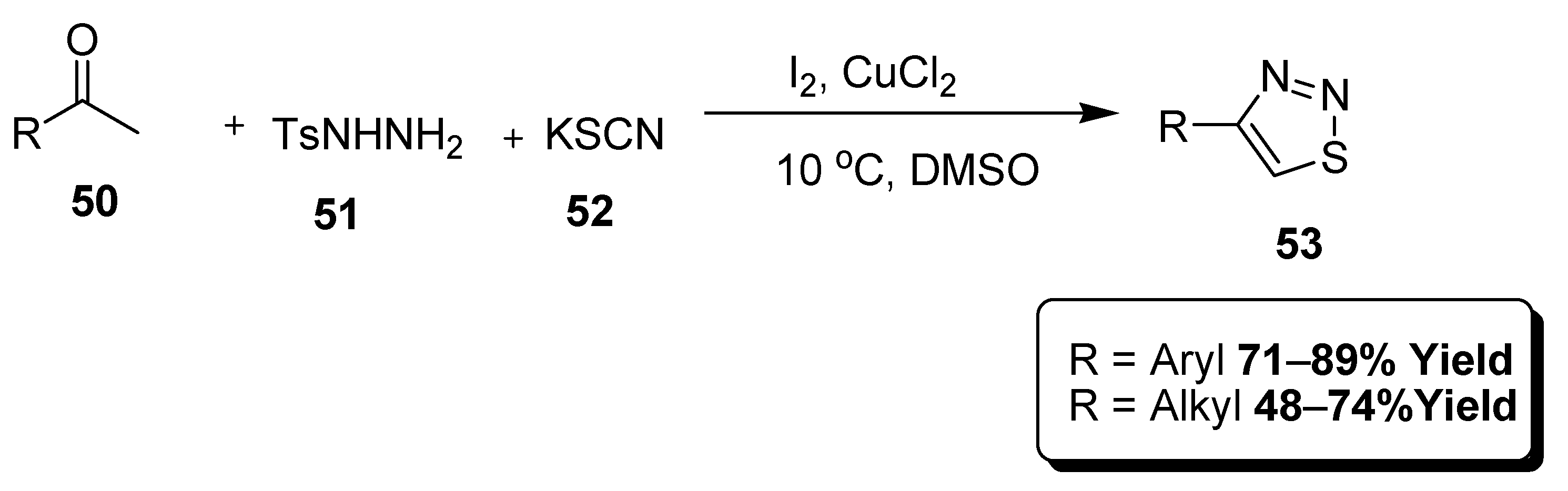
2.3. Multi Component Synthetic Strategies
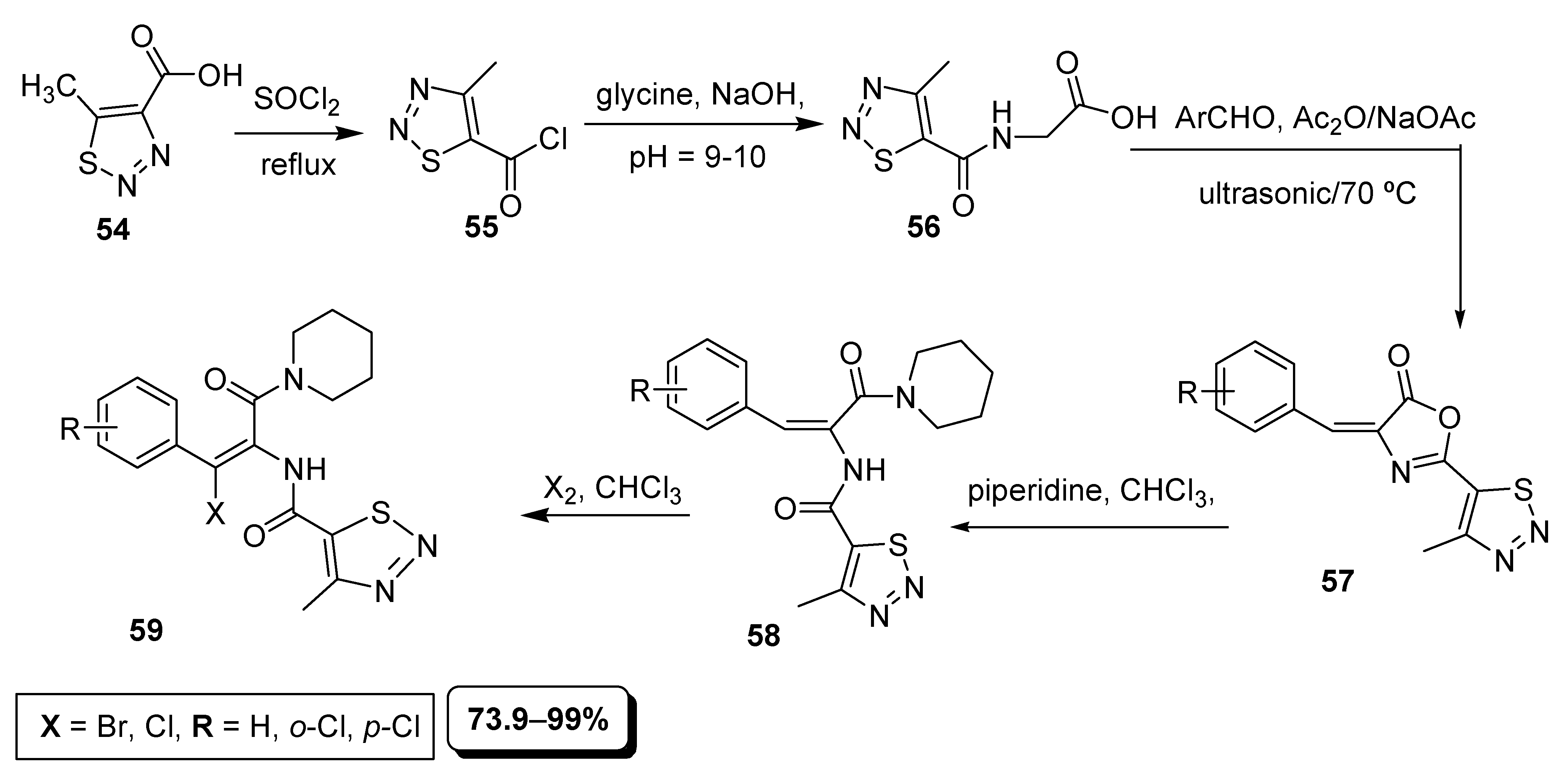
2.4. Ultrasonic Assisted Synthesis of 1,2,3-Thiadiazoles
2.5. Multi-Step Synthesis of 1,2,3-Thiadiazole from Quinolin-8-ol
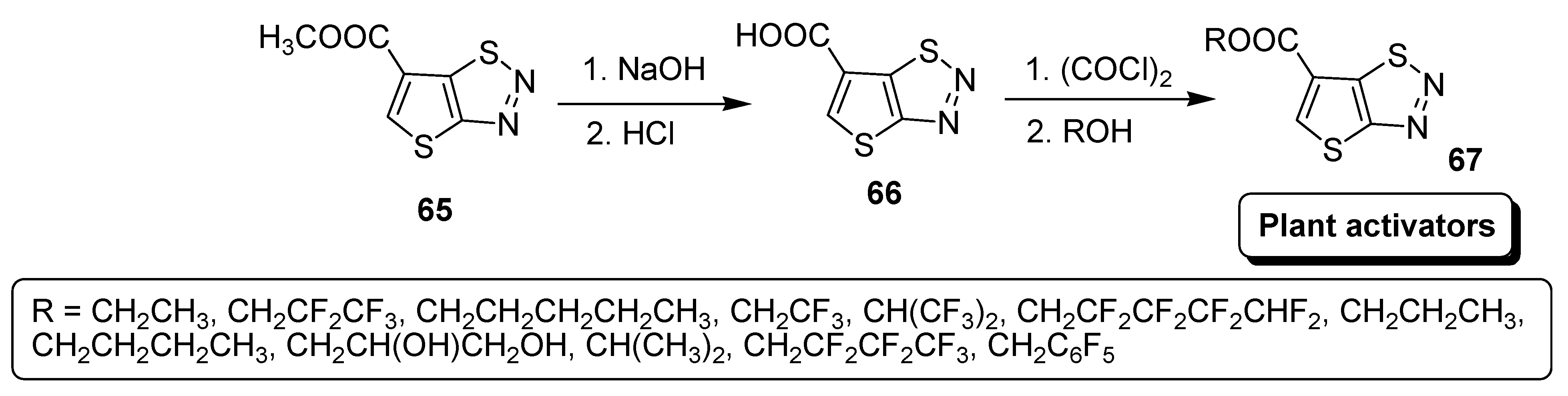
2.6. Hydrolization and Esterification Approach
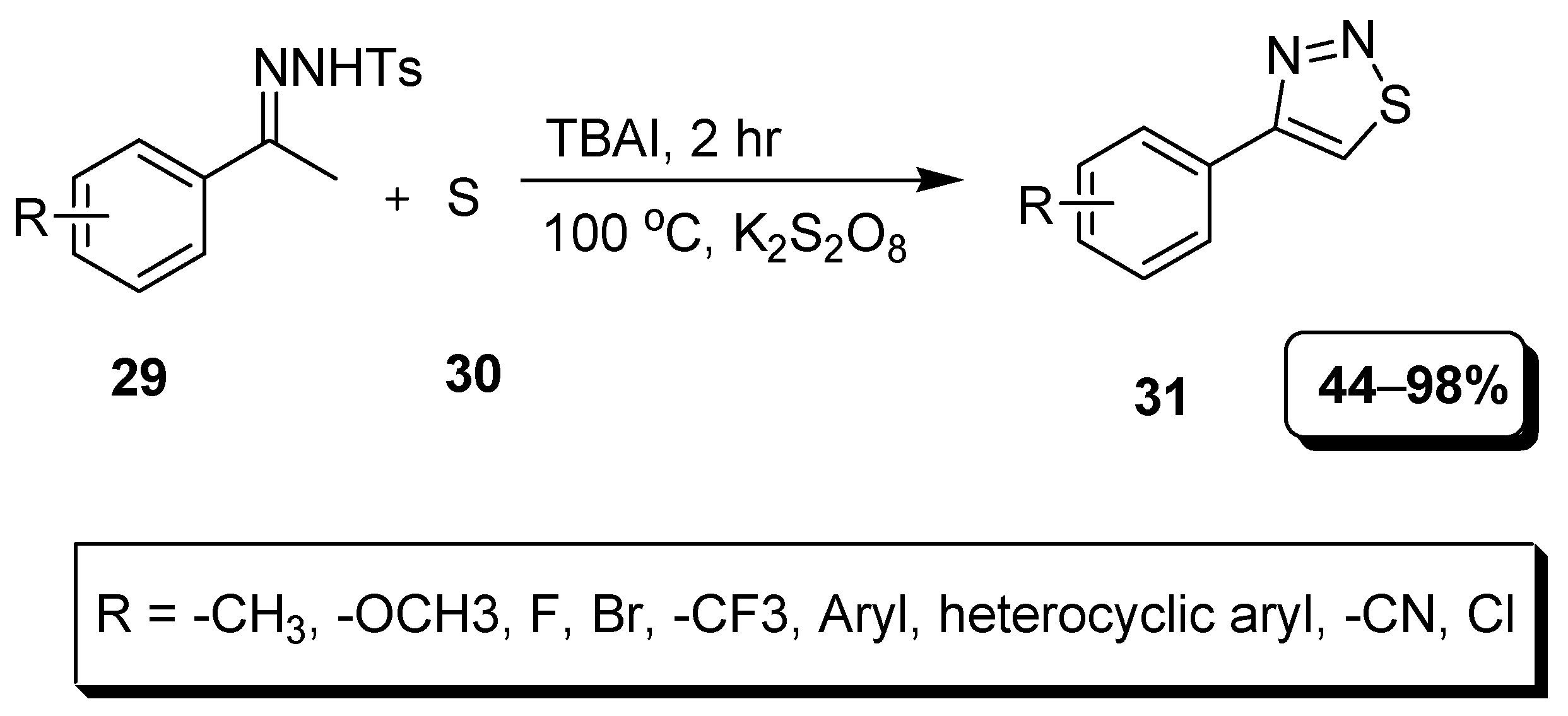
2.7. Transition Metal-Free Synthetic Approach
2.8. Nucleophilic Addition
2.9. [4 + 1]. Annulation of Azoalkenes
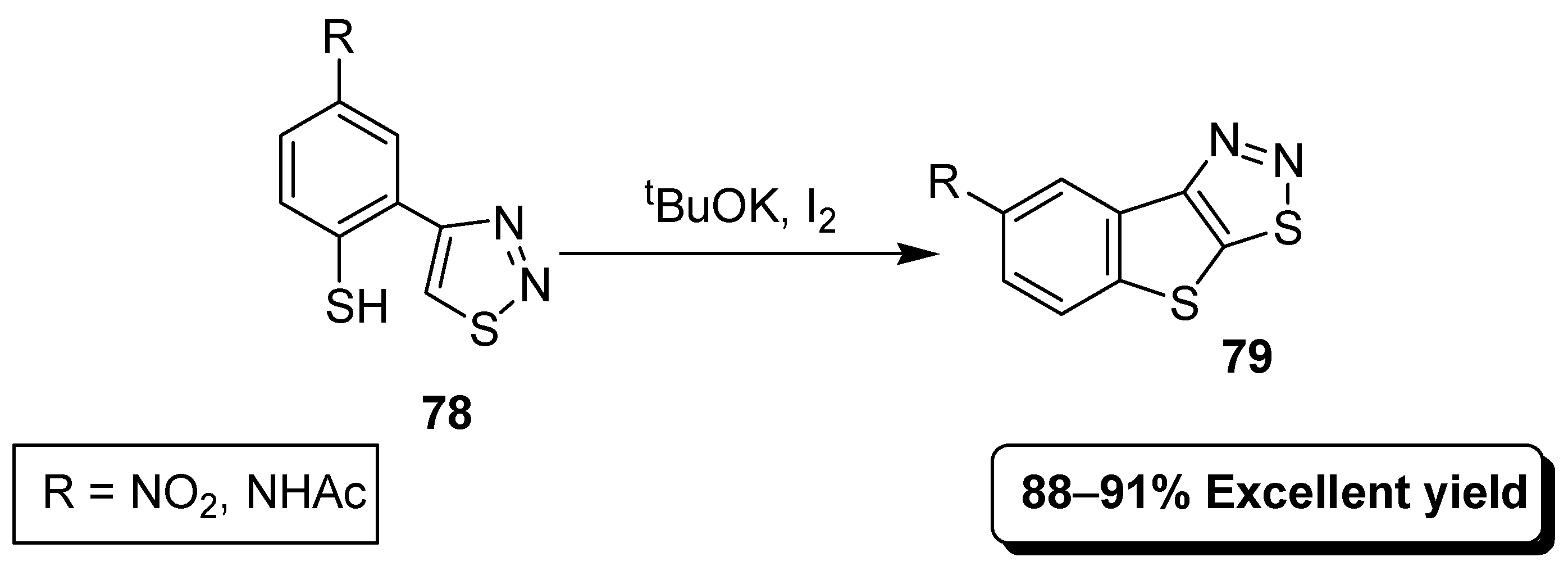
2.10. Intramolecular Oxidative Nucleophilic Substitution of Hydrogen (ONSH)
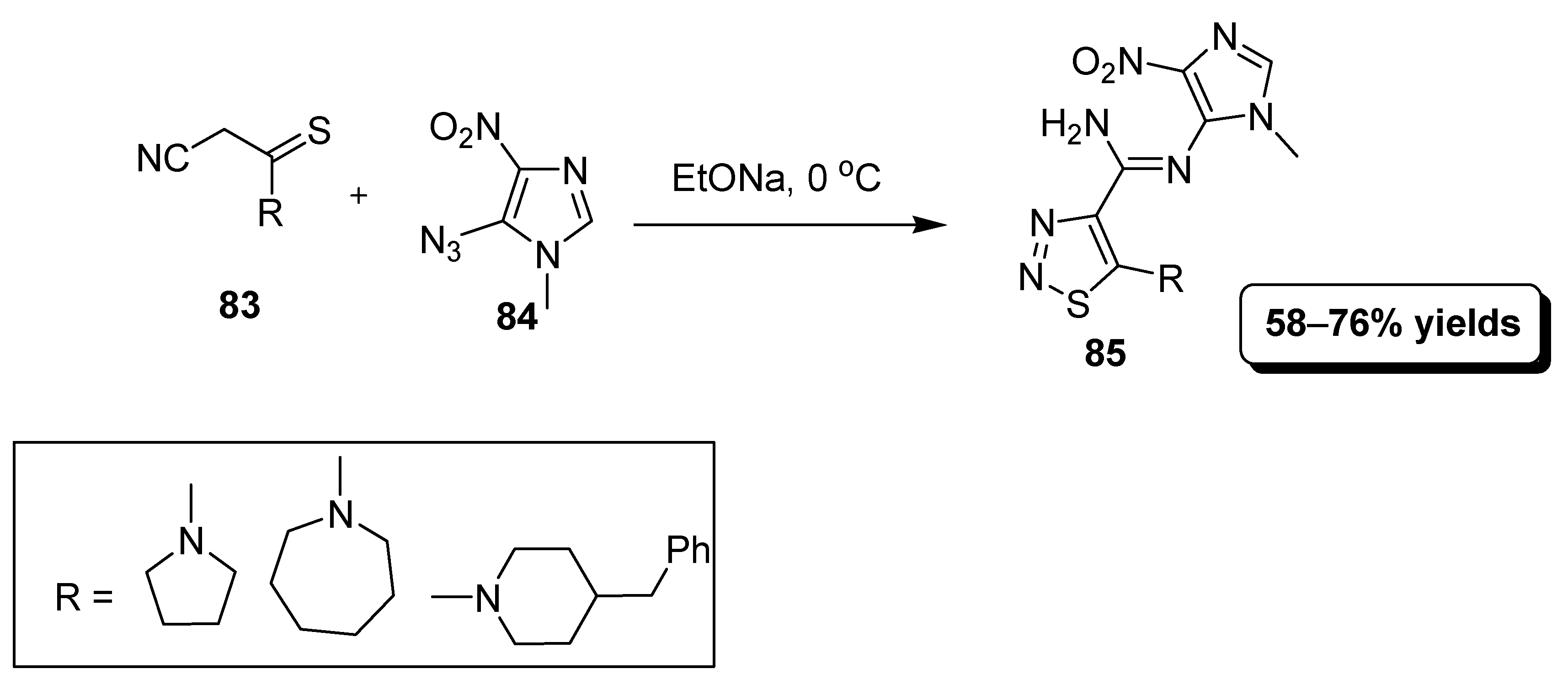
2.11. Intermolecular [3 + 2] Heterocyclization
2.12. Cornforth Rearrangement Approach
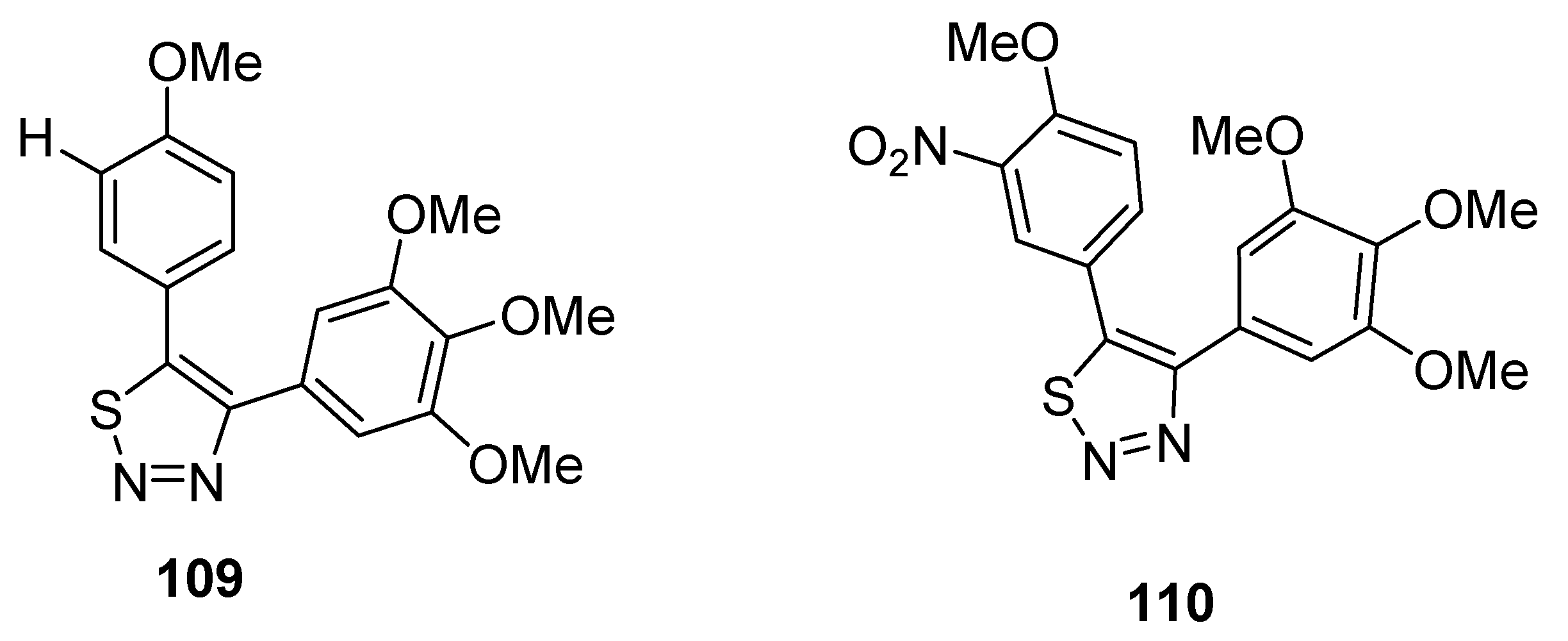
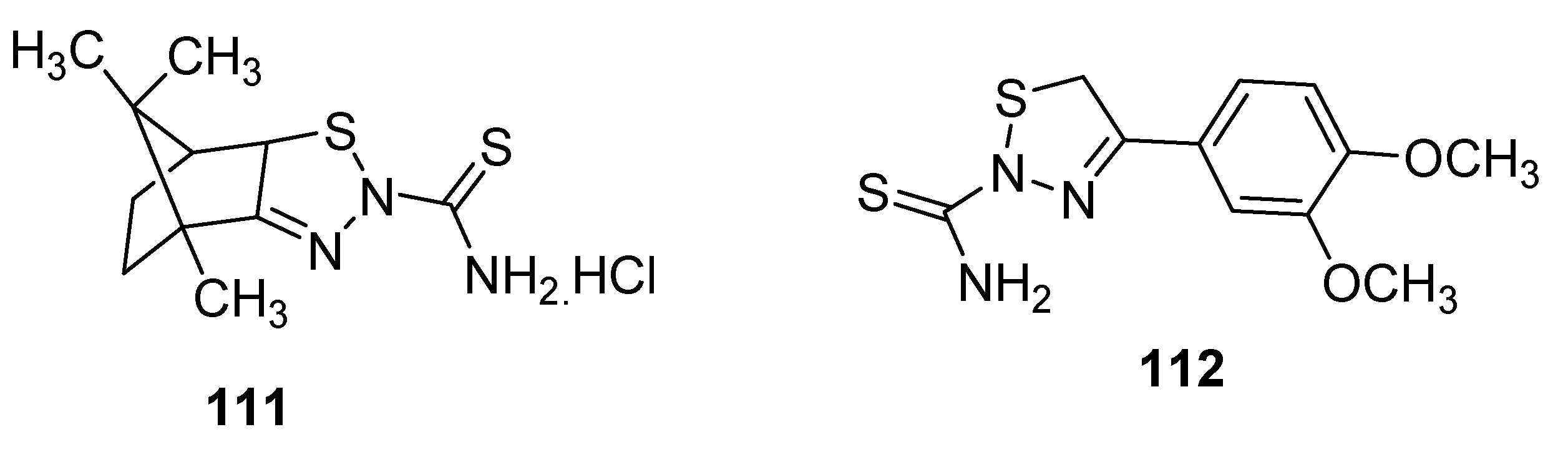
2.13. Cascade Process
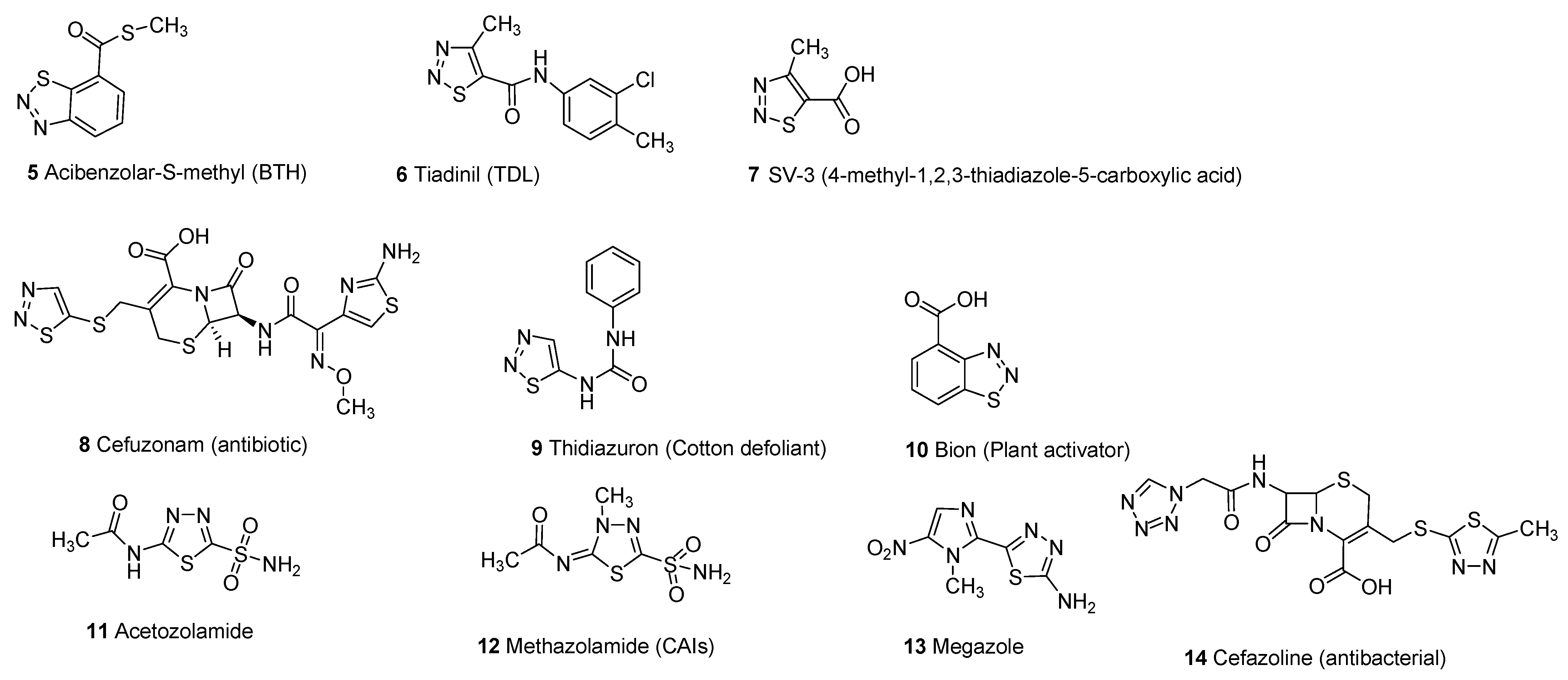
3. Biological Activities of 1,2,3-Thiadiazole Derivatives
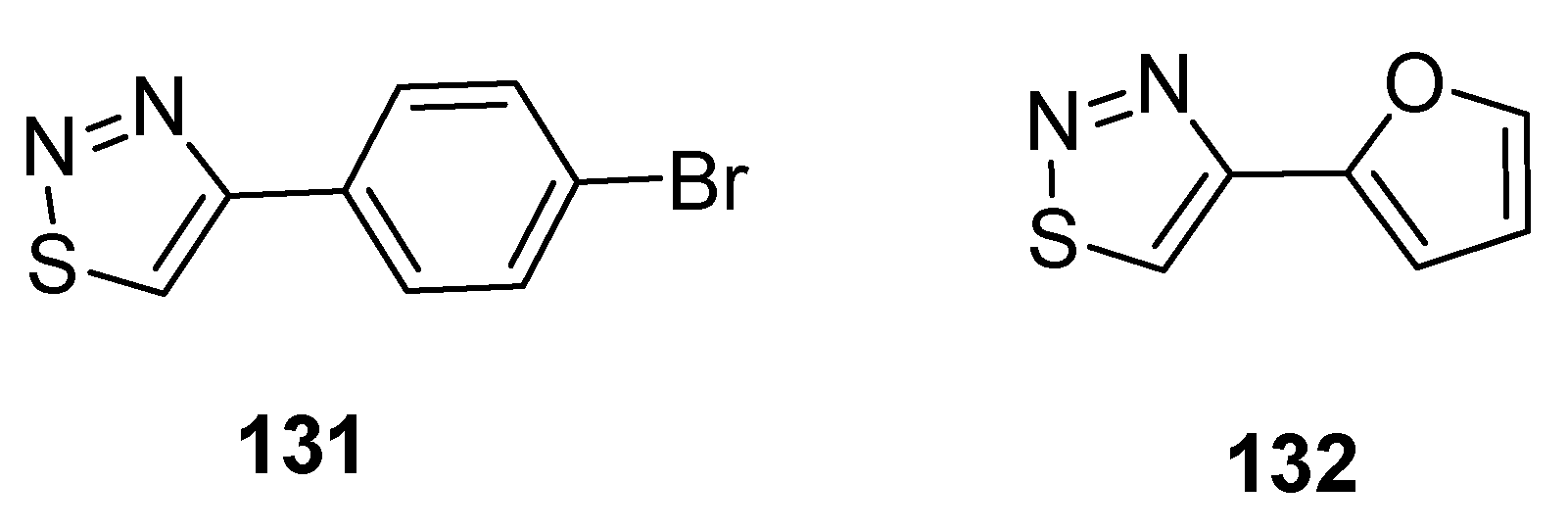
3.1. Antiviral Agents
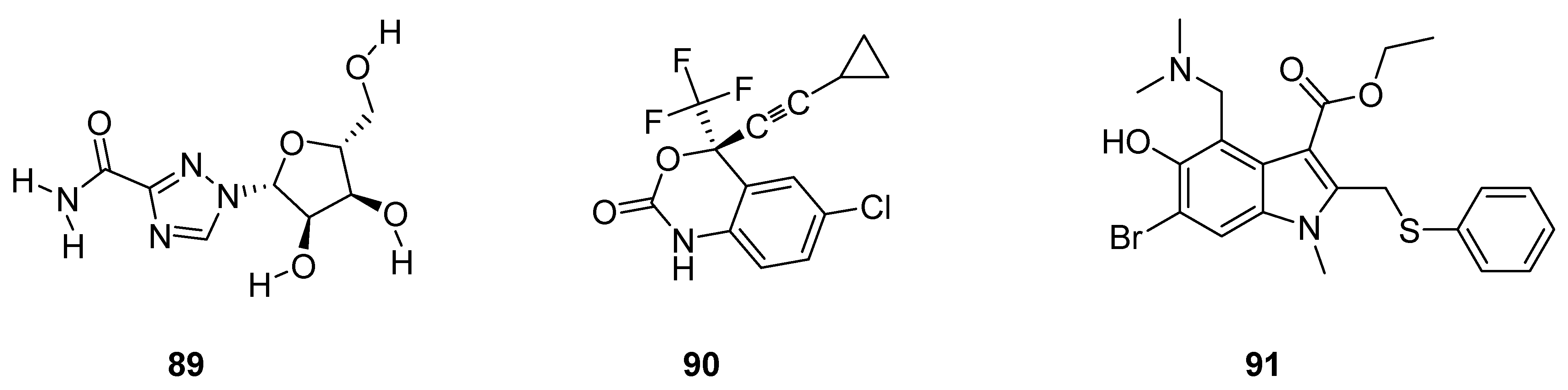
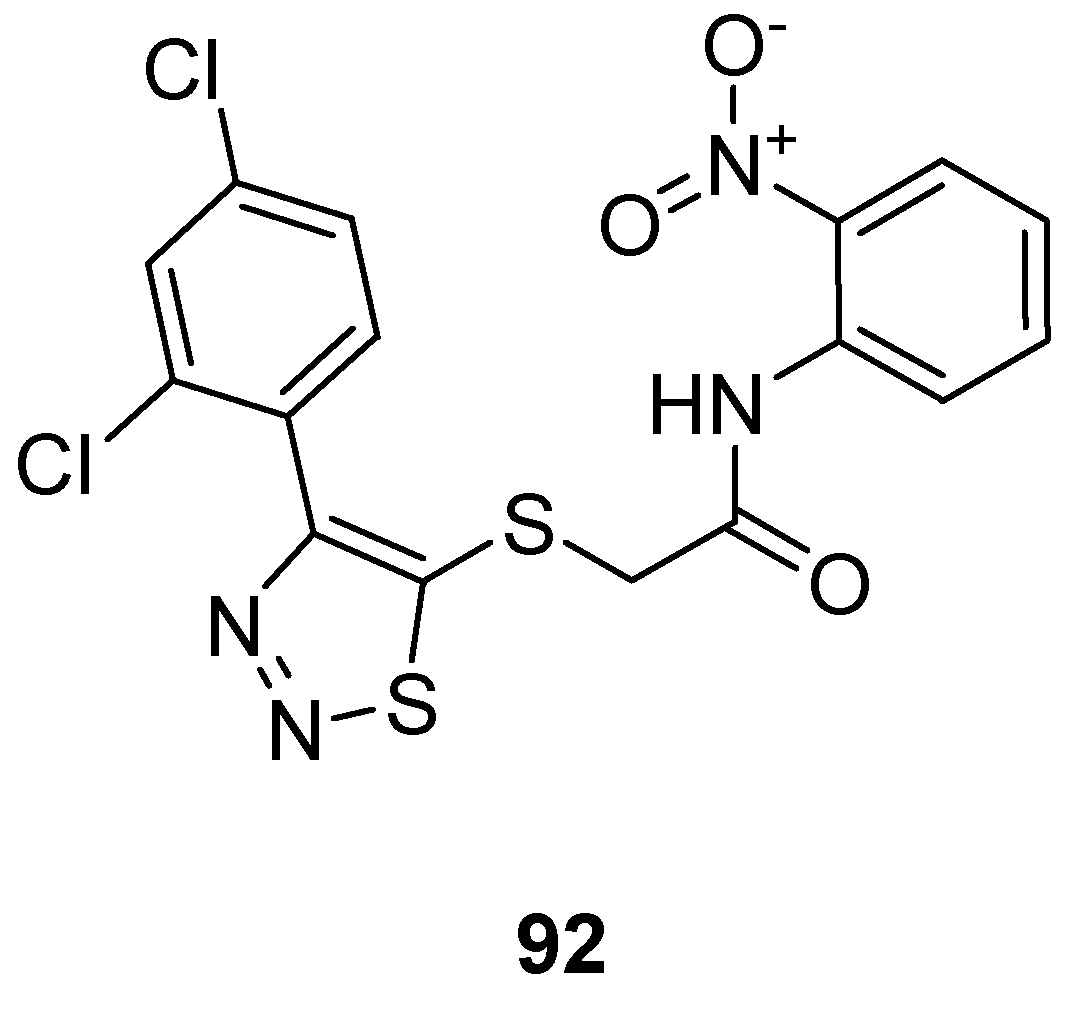
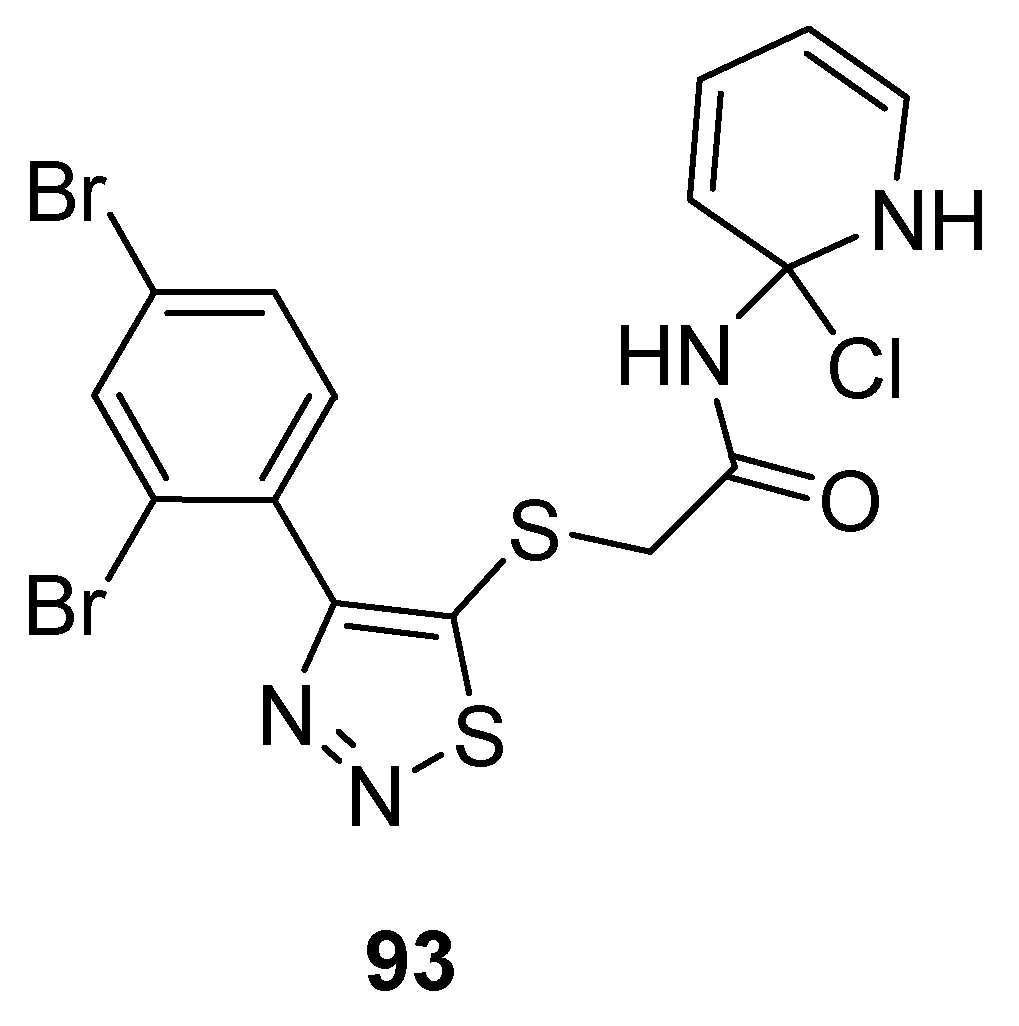
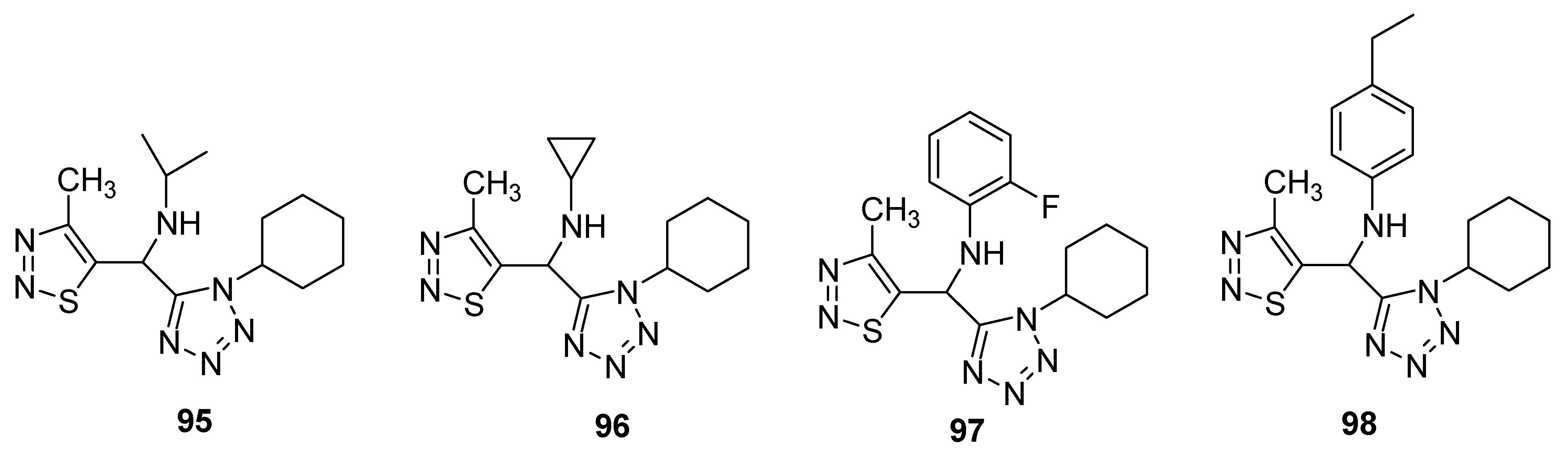
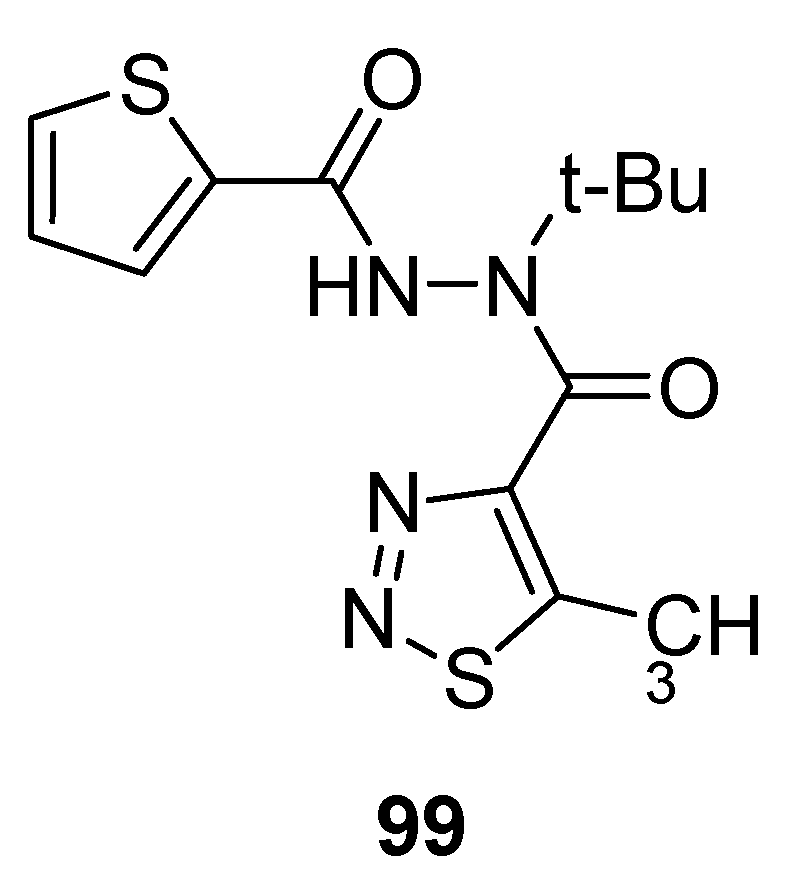
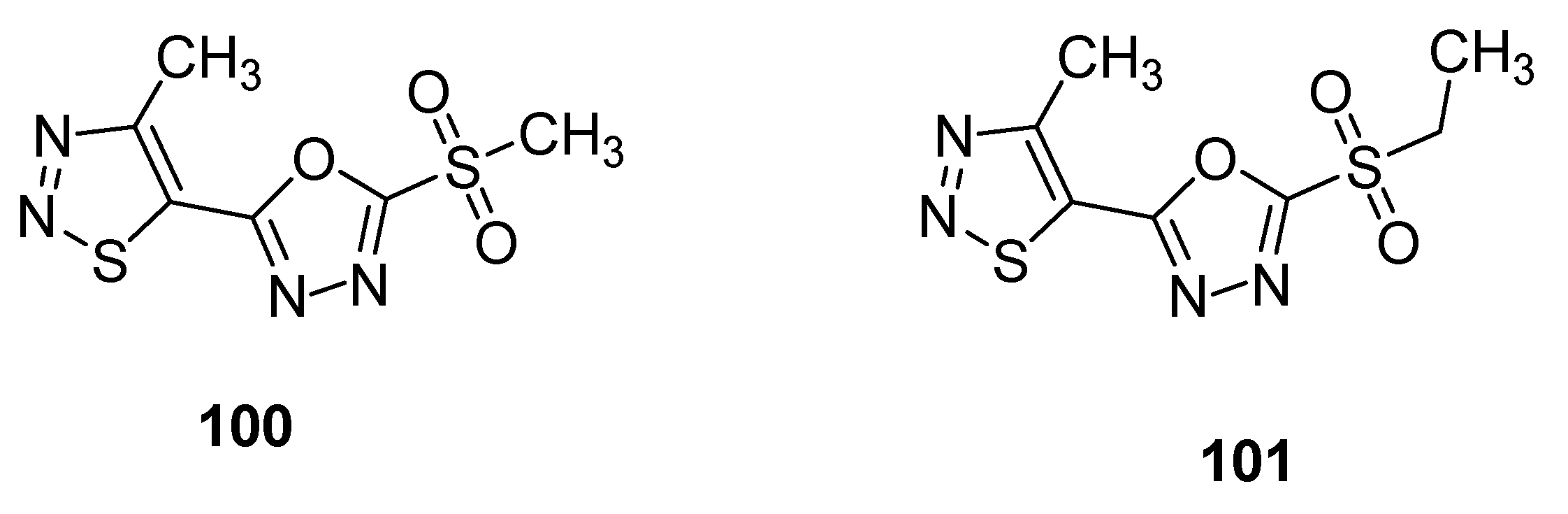
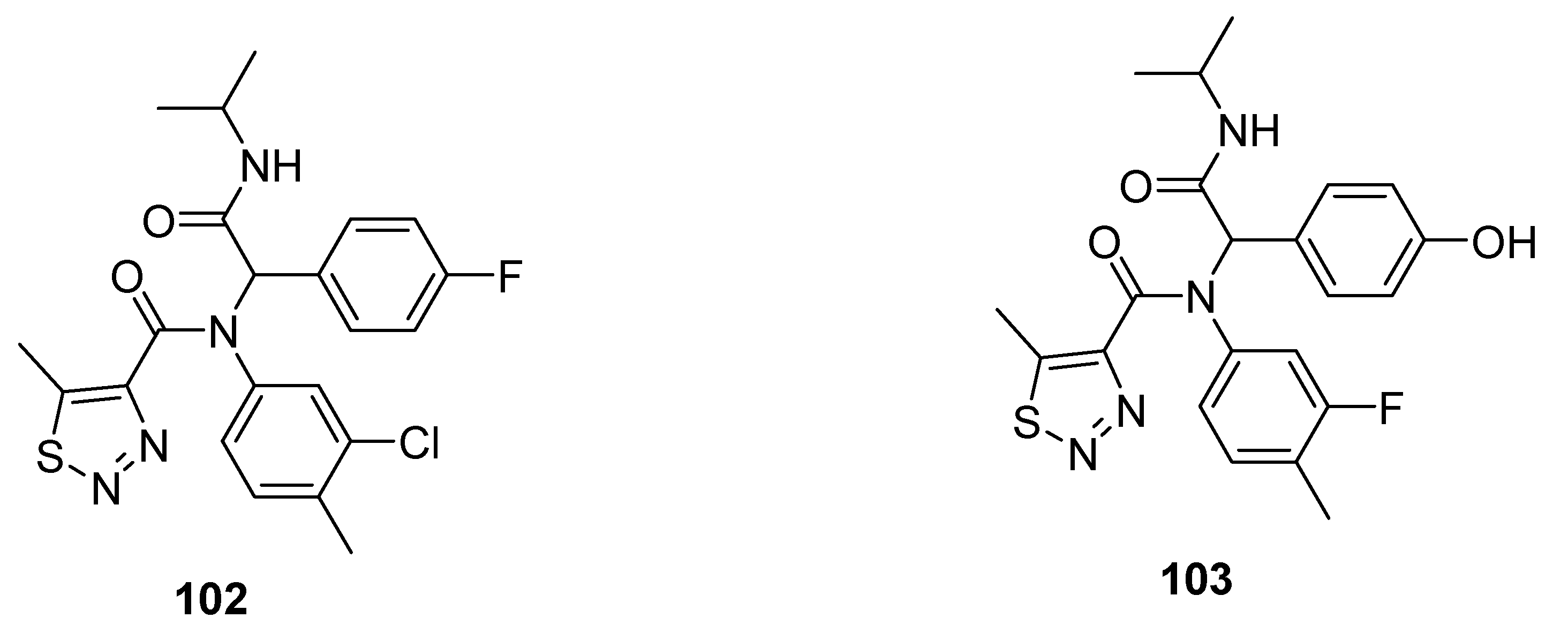
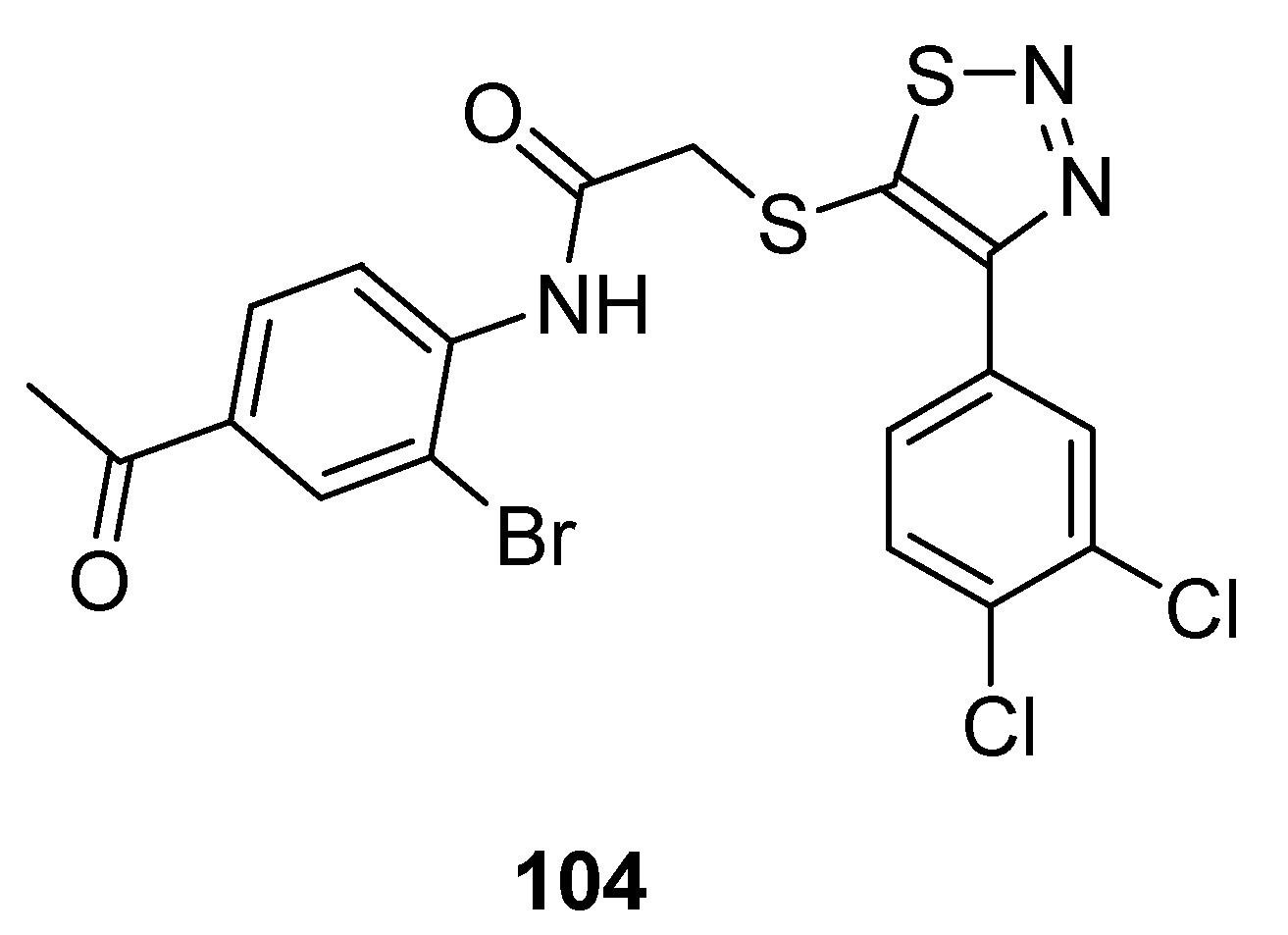
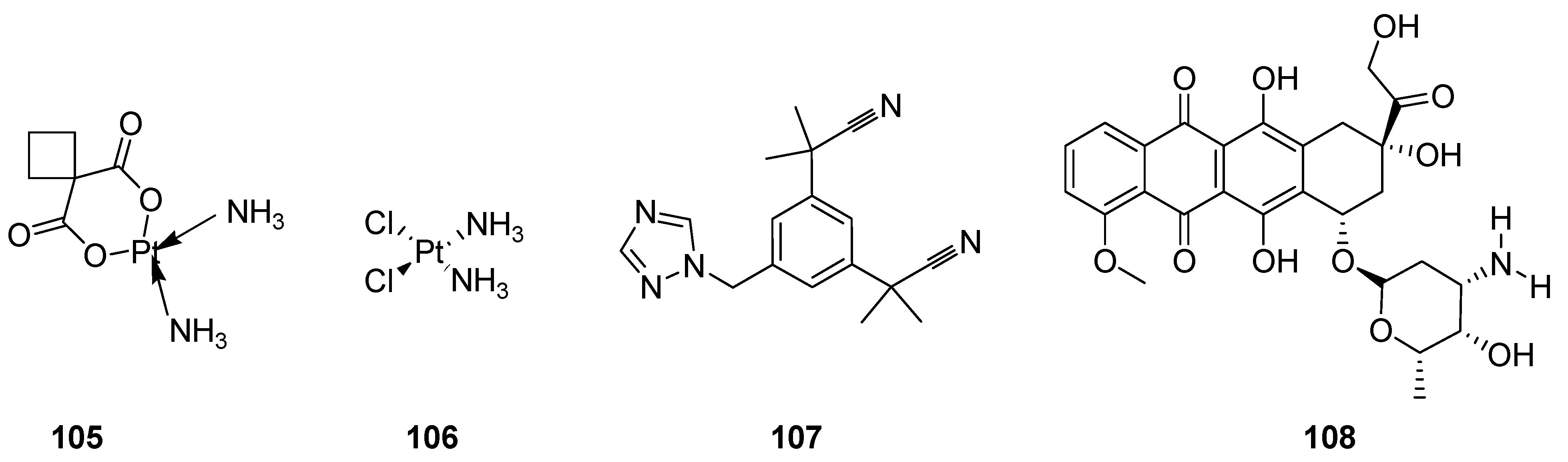
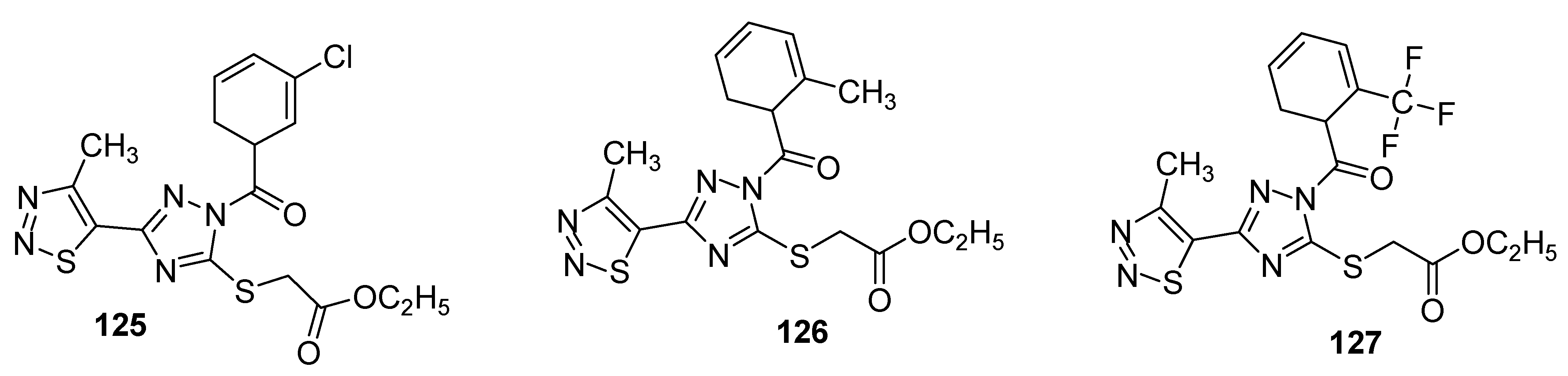
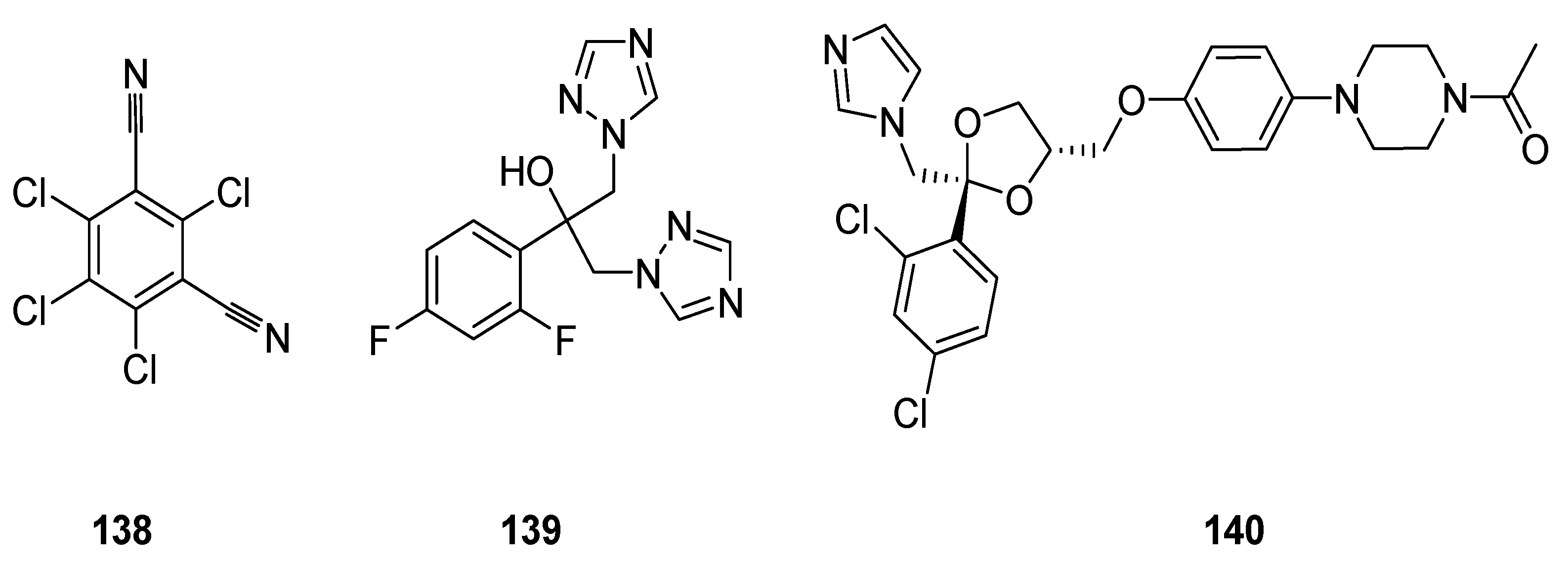
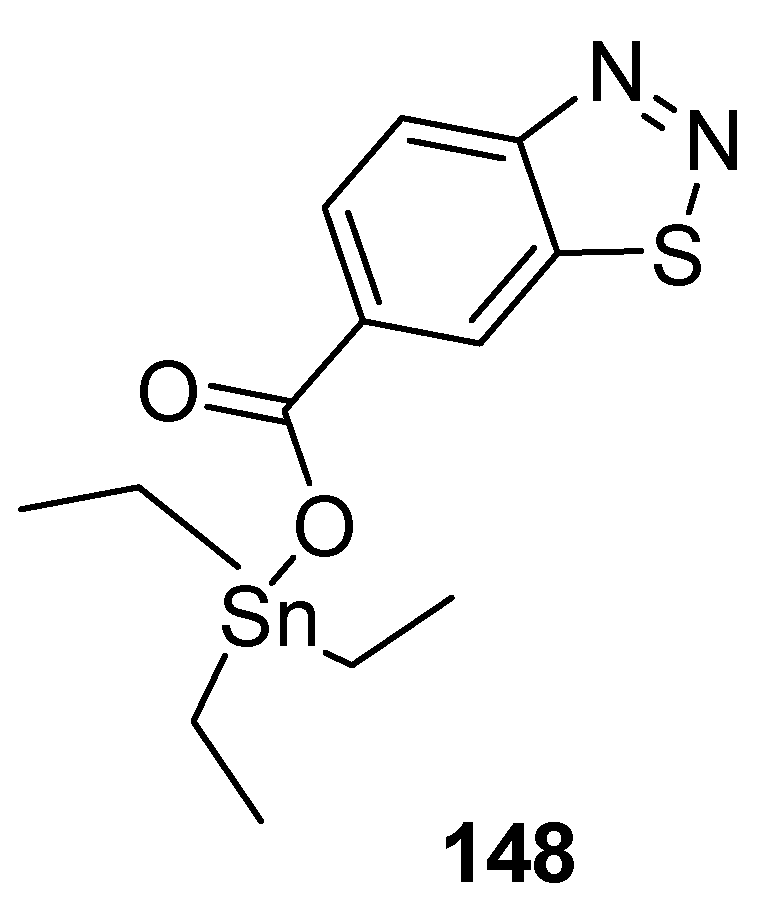
3.2. Anticancer Agents
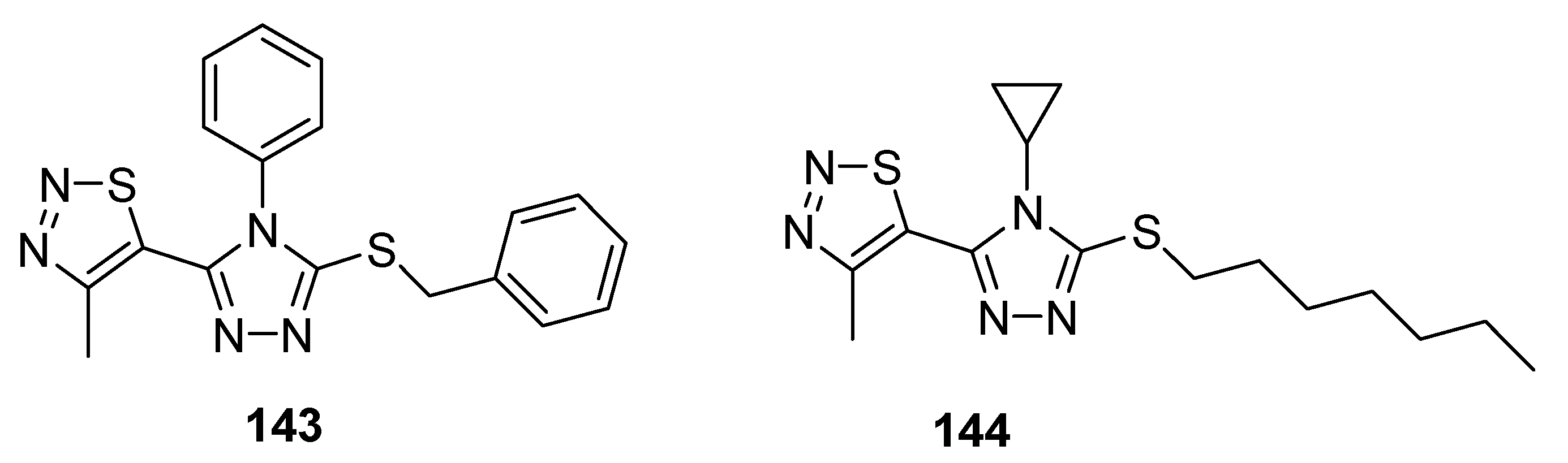
3.3. Insecticidal Agents
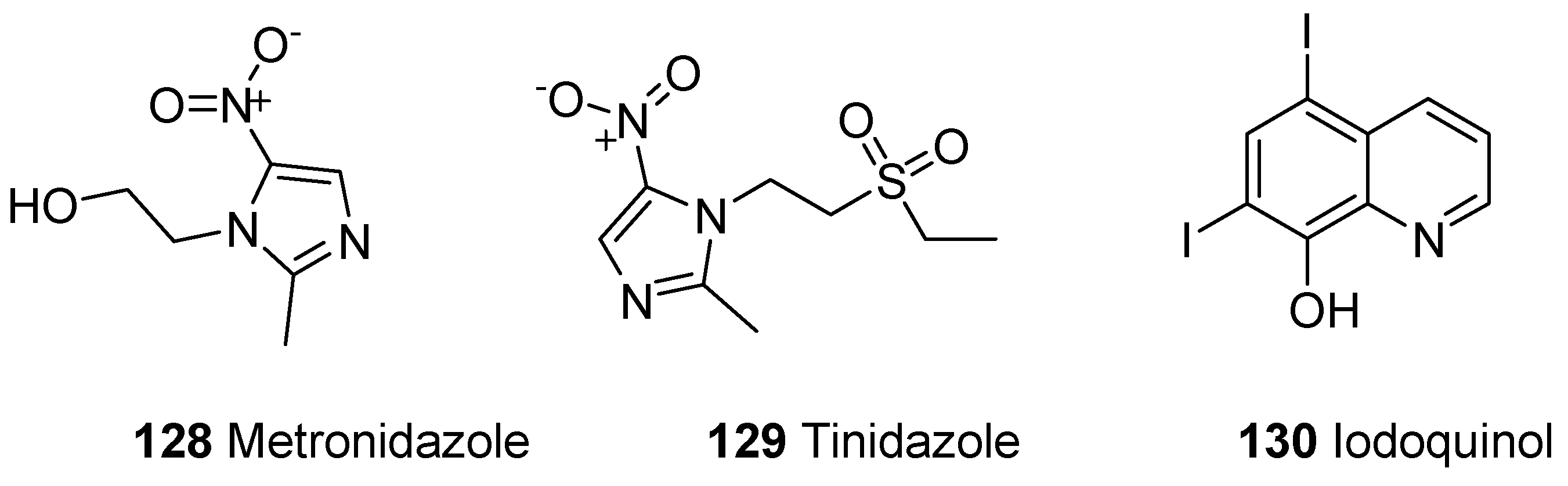
3.4. Amoebicidal Agents
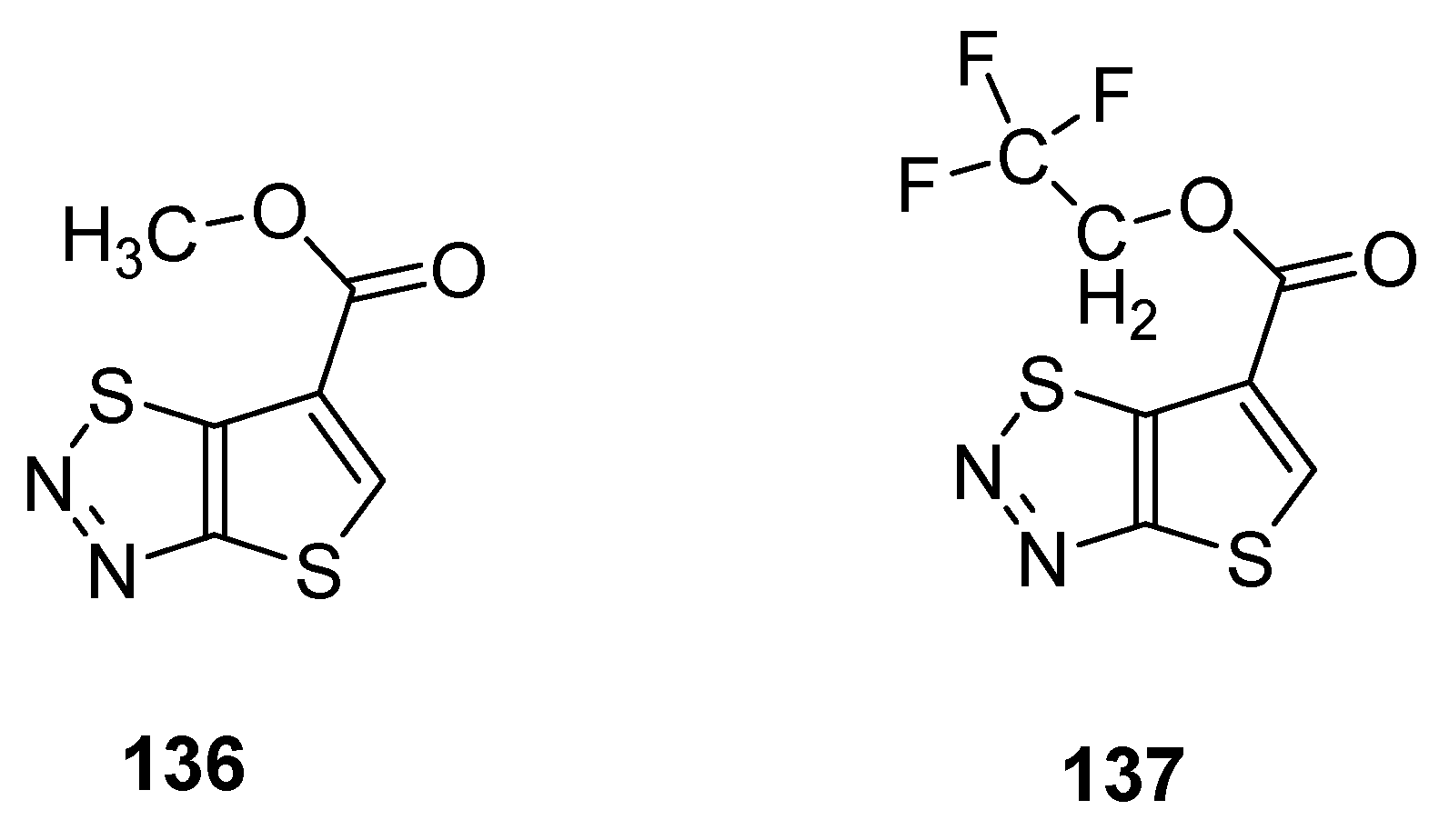
3.5. Plant Activator Agents
3.6. Fungicidal Agents
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Irfan, A.; Batool, F.; Ahmad, S.; Ullah, R.; Sultan, A.; Sattar, R.; Nisar, B.; Rubab, L. Recent trends in the synthesis of 1,2,3-thiadiazoles. Phosphorus Sulfur Silicon Relat. Elem. 2019, 194, 1098–1115. [Google Scholar] [CrossRef]
- Singh, A.K.; Parthsarthy, R.; Kshitiz, J.; Mishra, G. Synthesis, characterization and antibacterial activity of 1,3,4-thiadiazole derivatives. Int. J. Sci. Innov. Discov. 2011, 3, 353–361. [Google Scholar]
- Siddiqui, N.; Ahuja, P.; Ahsan, W.; Pandeya, S.N.; Alam, S.M. Thiadiazoles: Progress report on biological activities. J. Chem. Pharm. Res. 2009, 1, 19–30. [Google Scholar]
- Jalhan, S.; Jindal, A.; Gupta, A.; Hemraj, H. Synthesis, biological activities and chemistry of thiadiazole derivatives and schiff bases. Asian J. Pharm. Clin. Res. 2012, 3, 199–208. [Google Scholar]
- Srivastava, S.; Prasad, K.R.; Saini, R. Thiadiazole: A brief review. WJPPS 2014, 3, 1198–1212. [Google Scholar]
- Paulrasu, K.; Duraikannu, A.; Palrasu, M.; Shanmugasundaram, A.; Kuppusamy, M.; Thirunavukkarasu, B. Synthesis of 4-methyl-N’-(3-alkyl-2r,6cdiarylpiperidin-4-ylidene)-1,2,3-thiadiazole-5-carbohydrazides with antioxidant, antitumor and antimicrobial activities. Org. Biomol. Chem. 2014, 12, 911–921. [Google Scholar] [CrossRef]
- Kanakaraju, S.; Suresh, L. Design, synthesis, in vitro antimicrobial and cytotoxic evaluation of novel 1,2,3-selena/thiadiazolyltetrazole derivatives. RSC Adv. 2015, 5, 29325–29334. [Google Scholar] [CrossRef]
- Dogan, H.N.; Duran, A.; Rollas, S.; Sener, G.; Uysal, M.K.; Gulen, D. Synthesis of new 2,5-disubstituted-1,3,4-thiadiazoles and preliminary evaluation of anticonvulsant and antimicrobial activities. Bioorg. Med. Chem. 2002, 10, 2893–2898. [Google Scholar] [CrossRef]
- Hussain, S.; Sharma, J.; Amir, M. Synthesis and Antimicrobial activities of 1,2,4-triazole and 1,3,4-thiadiazole derivatives of 5-amino-2-hydroxybenzoic acid. E J. Chem. 2008, 5, 963–968. [Google Scholar] [CrossRef]
- Demirbas, A.; Sahin, D.; Demirbas, N.; Karaoglu, A.S. Synthesis of some new 1,3,4-thiadiazol-2-ylmethyl-1,2,4-triazole derivatives and investigation of their antimicrobial activities. Eur. J. Med. Chem. 2009, 44, 2896–2903. [Google Scholar] [CrossRef]
- Farshori, N.N.; Banday, M.R.; Ahmad, A.; Khan, A.U.; Rauf, A. Synthesis, characterization and in vitro antimicrobial activities of 5-alkenyl/hydroxyalkenyl-2-phenylamine-1,3,4-oxadiazoles and thiadiazoles. Bioorg. Med. Chem. Lett. 2010, 20, 1933–1938. [Google Scholar] [CrossRef] [PubMed]
- Almajan, G.L.; Innocenti, A.; Puccetti, L.; Manole, G.; Barbuceanu, S.; Saramet, I.; Scozzafava, A.; Supuran, C.T. Carbonic anhydrase inhibitors. Inhibition of the cytosolic and tumor-associated carbonic anhydrase isozymes I, II, and IX with a series of 1,3,4-thiadiazole- and 1,2,4-triazole-thiols. Bioorg. Med. Chem. Lett. 2005, 15, 2347–2352. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Hamid, M.K.; Abdel-Hafez, A.A.; El-Koussi, N.A.; Mahfouz, N.M.; Innocenti, A.; Supuran, C.T. Design, synthesis, and docking studies of new 1,3,4-thiadiazole-2-thione derivatives with carbonic anhydrase inhibitory activity. Bioorg. Med. Chem. 2007, 15, 6975–6984. [Google Scholar] [CrossRef]
- Dudutien, V.; Baranauskien, L.; Matulis, D. Benzimidazo[1,2-c][1,2,3]thiadiazole-7-sulfonamides as inhibitors of carbonic anhydrase. Bioorg. Med. Chem. Lett. 2007, 17, 3335–3338. [Google Scholar] [CrossRef]
- Jalilian, A.R.; Sattari, S.; Bineshmarvasti, M.; Daneshtalab, M.; Shafiee, A. Synthesis and in vitro antifungal and cytotoxicity evaluation of substituted 4,5-dihydronaphtho[1,2-d][1,2,3]thia(orselena)diazoles. Il Farmaco 2003, 58, 63–68. [Google Scholar] [CrossRef]
- Chu, C.H.; Hui, X.P.; Xu, P.F.; Zhang, Z.Y.; Li, Z.C.; Liao, R.A. Synthesis and antifungal activities of ω-(5-arylamino-1,3,4-thiadiazol-2-thio-)-ω-(1H-1,2,4-triazol-1-yl)acetophenones. Indian J. Chem. 2002, 41B, 2436–2438. [Google Scholar] [CrossRef]
- Moawad, E.B.; Yousif, M.Y.; Metwally, M.A. Synthesis of certain heteroaryl-fused pyrimidines and pyridines and selena- and thia-diazoles with naphthyl substituent as potential antifungal agents. Pharmazie 1989, 44, 820–822. [Google Scholar] [CrossRef]
- Yadav, L.S.; Zaidi, M.G.H.; Singh, B.N. Synthesis and fungicidal evaluation of some 1,3,4-thiadiazoles against P. oryzae and R. solani. Asian J. Chem. 2003, 15, 1805–1807. [Google Scholar]
- Zou, X.-J.; Lai, L.H.; Jin, G.Y.; Zhang, Z.X. Synthesis, fungicidal activity, and 3D-QSAR of pyridazinone-substituted 1,3,4-oxadiazoles and 1,3,4-thiadiazoles. J. Agric. Food. Chem. 2002, 50, 3757–3760. [Google Scholar] [CrossRef]
- Balasankar, T.; Gopalakrishnan, M.; Nagarajan, S. Synthesis and antibacterial activity of some 5-(4-biphenylyl)-7-aryl[3,4-d] [1,2,3]-benzothiadiazoles. Eur. J. Med. Chem. 2005, 40, 728–731. [Google Scholar] [CrossRef]
- Padmavathi, V.; Mahesh, K.; Mohan, N.V.A.; Mohan, N.; Padmaja, A. Synthesis and bioassay of oxazolyl/thiazolyl selenadiazoles, thiadiazoles and diazaphospholes. Chem. Pharm. Bull. 2009, 57, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Taha, S.S.; Ahmad, A.A.; Mawlood, S.I.; Ali, N.O. Synthesis of some series of 2-amino-1,3,4-thiadiazole derivatives with their pathogenic bacterial activity. J. Raparin Univ. 2017, 4, 63–78. [Google Scholar]
- Othman, A.A.; Kihel, M.; Amara, S. 1,3,4-Oxadiazole, 1,3,4-thiadiazole and 1,2,4-triazole derivatives as potential antibacterial agents. Arab. J. Chem. 2014, 12, 1660–1675. [Google Scholar] [CrossRef]
- .Paruch, K.; Popiołek, Ł.; Biernasiuk, A.; Berecka-Rycerz, A.; Malm, A.; Gumieniczek, A.; Wujec, M. Novel derivatives of 4-methyl-1,2,3-thiadiazole-5-carboxylic acid hydrazide: Synthesis, lipophilicity, and in vitro antimicrobial activity screening. Appl. Sci. 2021, 11, 1180. [Google Scholar] [CrossRef]
- Camoutsis, C.; Geronikaki, A.; Ciric, A.; Sokovic, M.; Zoumpoulakis, P.; Zervou, M. Sulfonamide-1,2,4-thiadiazole derivatives as antifungal and antibacterial agents: Synthesis, biological evaluation, lipophilicity and conformational studies. Chem. Pharm. Bull. 2010, 58, 160–167. [Google Scholar] [CrossRef]
- Yasuda, M.; Kusajima, M.; Nakajima, M.; Akutsu, K.; Kudo, T.; Yoshida, S.; Nakashita, H. Thiadiazole carboxylic acid moiety of tiadinil, SV-03, induces systemic acquired resistance in tobacco without salicylic acid accumulation. J. Pest. Sci. 2006, 31, 329–334. [Google Scholar] [CrossRef]
- Xu, Y.F.; Zhao, Z.J.; Qian, X.H.; Qian, Z.; Tian, W.; Zhong, J. Novel, unnatural benzo-1,2,3-thiadiazole-7-carboxylate elicitors of taxoid biosynthesis. J. Agric. Food Chem. 2006, 54, 8793–8798. [Google Scholar] [CrossRef] [PubMed]
- Nombela, G.; Pascual, S.; Aviles, M.; Guillard, E.; Muñizet, M. Benzothiadiazole induces local resistance to Bemisia tabaci (Hemiptera:Aleyrodidae) in tomato plants. J. Econ. Entomol. 2005, 98, 2266–2271. [Google Scholar] [CrossRef]
- Wang, H.; Yang, Z.; Fan, Z.; Wu, Q.; Zhang, Y.; Mi, N.; Wang, S.; Zhang, Z.; Song, H.; Liu, F. Synthesis and insecticidal activity of N-tert- 314 butyl-N,N0-diacylhydrazines containing 1,2,3-thiadiazoles. J. Agric. Food Chem. 2011, 59, 628–634. [Google Scholar] [CrossRef]
- Bloom, J.D.; DiGrandi, M.J.; Dushin, R.G.; Curran, K.J.; Ross, A.A.; Norton, E.B.; Terefenko, E.; Jones, T.R.; Feld, B.; Lang, S.A. Thiourea inhibitors of herpes viruses. Part 1: Bis-(aryl)thiourea inhibitors of CMV. Bioorg. Med. Chem. Lett. 2003, 13, 2929–2932. [Google Scholar] [CrossRef]
- Bloom, J.D.; Dushin, R.G.; Curran, K.J.; Donahue, F.; Norton, E.B.; Terefenko, E.; Jones, T.R.; Ross, A.A.; Feld, B.; Lang, S.A.; et al. Thiourea inhibitors of herpes viruses. Part 2: N-Benzyl-N0-arylthiourea inhibitors of CMV. Bioorg. Med. Chem. Lett. 2004, 14, 3401–3406. [Google Scholar] [CrossRef] [PubMed]
- Pannecouque, C.; Szafarowicz, B.; Volkova, N.; Bakulev, V.; Dehaen, W.; Mely, Y.; Daelemans, D. N,N′-Bis(1,2,3-thiadiazol-5-yl)benzene-1,2-diamine targets the HIV-1 retroviral nucleocapsid zinc fingers. Antimicrob. Agents Chemother. 2010, 54, 1461–1468. [Google Scholar] [CrossRef]
- Tatar, E.; Kucukguzel, S.G.; Karakus, S.; Clercq, E.D.; Andrei, G.; Snoeck, R.; Pannecouque, C.; Okullu, S.O.; Ünübol, N.; Kocagöz, T.; et al. Synthesis and biological evaluation of some new 1,3,4-thiadiazole and 1,2,4-triazole derivatives from L-methionine as antituberculosis and antiviral agents. Marmara Pharm. J. 2015, 19, 88–102. [Google Scholar] [CrossRef]
- Azaam, M.M.; Kenawy, E.; El-Din, A.S.B.; Khamis, A.A.; El-Magdd, M.A. Antioxidant and Anticancer Activities of α-Aminophosphonates Containing Thiadiazole. J. Saudi Chem. Soc. 2018, 22, 34–41. [Google Scholar] [CrossRef]
- Cressier, D.; Prouillac, C.; Hernandez, P.; Amourette, C.; Diserbo, M.; Lion, C.; Rima, G. Synthesis, antioxidant properties and radioprotective effects of new benzothiazoles and thiadiazoles. Bioorg. Med. Chem. 2009, 17, 5275–5284. [Google Scholar] [CrossRef] [PubMed]
- Kaur, H.; Kumar, S.; Vishwakarma, P.; Sharma, M.; Saxena, K.K.; Kumar, A. Synthesis and antipsychotic and anticonvulsant activity of some new substituted oxa/thiadiazolyl azetidinonyl/thiazolidinonyl carbazoles. Eur. J. Med. Chem. 2010, 45, 2777–2783. [Google Scholar] [CrossRef]
- Sddiqui, N.; Ahuja, P.; Ahsan, W.; Pandeya, S.N.; Alam, S.M. Synthesis of 3-arylamino-4-aryl-5-(N-arylthiocarbonylimino)-4,5-dihydro-1,2,4-thiadiazoles as anticonvulsant agents. Indian J. Heterocycl. Chem. 2004, 14, 159–160. [Google Scholar]
- Siddiqui, N.; Arshad, M.F.; Khan, S.A.; Ahsan, W. Synthesis, anticonvulsant and neurotoxicity screening of 1-(substituted phenyl)-3-[(5-substituted phenyl)-1,3,4-thiadiazole-2-yl]-2-thioxodihydro pyrimidine-4,6 (1H, 5H)-diones. J. Pharm. Res. 2008, 7, 122–125. [Google Scholar] [CrossRef]
- Siddiqui, N.; Ahsan, W. Synthesis, anticonvulsant and toxicity screening of thiazolyl-thiadiazole derivatives. Med. Chem. Res. 2011, 20, 261–268. [Google Scholar] [CrossRef]
- Mullick, P.; Khan, S.A.; Verma, S.; Alam, O. Thiadiazole derivatives as potential anticonvulsant agents. B Korean Chem. Soc. 2011, 32, 1011–1016. [Google Scholar] [CrossRef]
- Chapleo, C.B.; Myers, M.; Myers, P.L.; Saville, J.F.; Smith, A.C.B.; Stillings, M.R.; Tulloch, J.F.; Walter, D.S.; Welbourn, A.P. Substituted 1,3,4-thiadiazoles with anticonvulsant activity. 1.Hydrazines. J. Med. Chem. 1986, 29, 2273–2280. [Google Scholar] [CrossRef]
- Rajak, H.; Behera, C.K.; Pawar, R.S.; Singour, P.K.; Kharya, M.D. Synthesis and anticonvulsant evaluation of some novel 2,5-disubstituted 1,3,4-thiadiazoles: Pharmacophore model studies. Acta Pol. Pharm. 2010, 67, 503–510. [Google Scholar]
- Samel, A.B.; Pai, N.R. Synthesis of novel aryloxypropanoylthiadiazoles as potential antihypertensive agents. Chin. Chem. Soc. TAIP 2010, 57, 1327–1330. [Google Scholar] [CrossRef]
- Turner, S.; Myers, M.; Gadie, B.; Nelson, A.J.; Pape, R.; Saville, J.F.; Doxey, J.C.; Berridge, T.L. Antihypertensive thiadiazoles 1. Synthesis of some 2-aryl-5-hydrazino-1,3,4- thiadiazole with vasodilator activity. J. Med. Chem. 1988, 31, 902–906. [Google Scholar] [CrossRef]
- Wei, M.-X.; Feng, L.; Li, X.-Q.; Zhou, X.-Z.; Shao, Z.-H. Synthesis of new chiral 2,5-disubstituted 1,3,4-thiadiazoles possessing γ-butenolide moiety and preliminary evaluation of in vitro anticancer activity. Eur. J. Med. Chem. 2009, 44, 3340–3344. [Google Scholar] [CrossRef]
- Tripathy, R.; Ghose, A.; Singh, J.; Bacon, E.R.; Angeles, T.S.; Yang, S.X.; Albom, M.S.; Aimone, L.D.; Herman, J.L.; Mallamo, J.P. 1,2,3-Thiadiazole substituted pyrazolones as potent KDR/VEGFR-2 kinase inhibitors. Bioorg. Med. Chem. Lett. 2007, 17, 1793–1798. [Google Scholar] [CrossRef]
- Ibrahim, D.A. Synthesis and biological evaluation of 3,6-disubstituted [1,2,4]triazolo[3,4-b][1,3,4]thiadiazole derivatives as a novel class of potential anti-tumor agents. Eur. J. Med. Chem. 2009, 44, 776–2781. [Google Scholar] [CrossRef]
- Padmavathi, V.; Reddy, G.S.; Padmaja, A.; Kondaiah, P.P.; Shazia, A. Synthesis, antimicrobial and cytotoxic activities of 1,3,4-oxadiazoles, 1,3,4-thiadiazoles and 1,2,4-triazoles. Eur. J. Med. Chem. 2009, 44, 2106–2112. [Google Scholar] [CrossRef] [PubMed]
- Karakuü, S.; Çoruh, U.; Barlas-Durgun, B.; Vázquez-López, E.M.; Özbaü-Turan, S.; Akbuùa, J.; Rollas, S. Synthesis and cytotoxic activity of some 1,2,4-triazoline-3-thione and 2,5-disubstituted-1,3,4-thiadiazole derivatives. Marmara Pharm. J. 2010, 14, 84–90. [Google Scholar] [CrossRef]
- Holla, B.S.; Poorjary, K.N.; Rao, B.S.; Shivananda, M.K. New bis-aminomercaptotriazoles and bis-triazolothiadiazoles as possible anti-cancer agents. Eur. J. Med. Chem. 2002, 37, 511–517. [Google Scholar] [CrossRef]
- Upadhyay, P.K.; Mishra, P. Synthesis, antimicrobial and anticancer activities of 5-(4-substituted phenyl)-1,3,4-thiadiazole-2-amines. Rasayan J. Chem. 2017, 10, 254–262. [Google Scholar] [CrossRef]
- Schenone, S.; Brullo, C.; Bruno, O.; Bondavalli, F.; Ranise, A.; Filippelli, W.; Rinaldi, B.; Capuano, A.; Falcone, G. New 1,3,4-thiadiazole derivatives endowed with analgesic and anti-inflammatory activities. Bioorg. Med. Chem. 2006, 14, 1698–1705. [Google Scholar] [CrossRef]
- Rostom, S.A.F.; El-Ashmawy, I.M.; el Razik, H.A.A.; Badr, M.H.; Ashour, H.M.A. Design and synthesis of some thiazolyl and thiadiazolyl derivatives of antipyrine as potential non-acidic anti-inflammatory, analgesic and antimicrobial agents. Bioorg. Med. Chem. 2009, 17, 882–895. [Google Scholar] [CrossRef]
- Labanauskas, L.; Kalcas, V.; Udrenaite, E.; Gaidelis, P.; Brukstus, A.; Dauksas, A. Synthesis of 3-(3,4-dimethoxyphenyl)-1H-1,2,4-triazole-5-thiol and 2-amino-5-(3,4-dimethoxyphenyl)-1,3,4-thiadiazole derivatives exhibiting anti-inflammatory activity. Pharmazie 2001, 56, 617–619. [Google Scholar] [CrossRef]
- Kadi, A.A.; El-Brolossy, N.R.; Al-Deeb, O.A.; Habib, E.E.; Ibrahim, T.M.; El-Emam, A.A. Synthesis, antimicrobial, and anti-inflammatory activities of novel 2-(1-adamantyl)-5-substituted-1,3,4-oxadiazoles and 2-(1-adamantylamino)-5-substituted-1,3,4-thiadiazoles. Eur. J. Med. Chem. 2007, 42, 235–242. [Google Scholar] [CrossRef]
- Kadi, A.A.; Al-Abdullah, E.S.; Shehata, I.A.; Habib, E.E.; Ibrahim, T.M.; El-Emam, A.A. Synthesis, antimicrobial and anti-inflammatory activi¬ties of novel 5-(1-adamantyl)-1,3,4-thiadiazole derivatives. Eur. J. Med. Chem. 2010, 45, 5006–5011. [Google Scholar] [CrossRef]
- Bekhit, A.A.; Ashour, H.M.A.; Ghany, Y.S.A.; Bekhit, A.E.A.; Baraka, A. Synthesis and biological evaluation of some thiazolyl and thiadiazolyl derivatives of 1H-pyrazole as anti-inflammatory antimicrobial agents. Eur. J. Med. Chem. 2008, 43, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Kamal, M.D.; Gawad, H.A.; Rageb, E.A.; Ellithey, M.; Mohamed, H.A. Synthesis, anticonvulsant, and anti-inflammatory evaluation of some new benzotriazole and benzofuran-based heterocycles. Bioorg. Med. Chem. 2006, 14, 3672–3680. [Google Scholar] [CrossRef]
- Du, Q.S.; Zhu, W.P.; Zhao, Z.J.; Qian, X.H.; Xu, Y.F. Novel benzo-1,2,3-thiadiazole-7-carboxylate derivatives as plant activators and the development of their agricultural applications. J. Agric. Food Chem. 2012, 60, 346–353. [Google Scholar] [CrossRef]
- Stanetty, P.; Kremslehner, M.; Veollenkle, H. A new type of plant activator: Synthesis of thieno[2,3-d][1,2,3]-thiadiazole-6-carboxylic acid derivatives via Hurd-Mori cyclization. J. Chem. Soc. Perkin Trans. 1998, 1, 853–856. [Google Scholar] [CrossRef]
- Pattan, S.R.; Kekare, P.; Dighe, N.S.; Nirmal, S.A.; Musmade, D.S.; Parjane, S.K.; Daithankar, A.V. Synthesis and biological evaluation of some 1,3,4-thiadiazoles. J. Chem. Pharm. Res. 2009, 1, 191–198. [Google Scholar]
- Ram, V.J.; Goel, A.; Kandpal, M. Tetraazaacenaphthene, tetraazaphenalene and 1,3,4-thiadiazole derivatives as potential leishmanicides. Bioorg. Med. Chem. Lett. 1997, 7, 651–656. [Google Scholar] [CrossRef]
- Silva, E.F.; Canto-Cavalheiro, M.M.; Braz, V.R.; Cysne-Finkelstein, L.; Leon, L.L.; Echevarria, A. Synthesis, and biological evaluation of new 1,3,4-thiadiazolium-2-phenylaminenderivatives against Leishmania amazonensis promastigotes and amastigotes. Eur. J. Med. Chem. 2002, 37, 979–984. [Google Scholar] [CrossRef]
- Behrouzi-Fardmoghadam, M.; Poorrajab, F.; Ardestani, S.K.; Emami, S.; Shafiee, A.; Foroumadi, A. Synthesis and in vitro anti-leishmanial activity of 1-[5-(5-nitrofuran-2-yl)- 1,3,4-thiadiazol-2-yl]- and 1-[5-(5-nitrothiophen-2-yl)-1,3,4-thiadiazol-2-yl]-4-aroyl piperazines. Bioorg. Med. Chem. 2008, 16, 4509–4515. [Google Scholar] [CrossRef]
- Foroumadi, A.; Emami, S.; Pournourmohammadi, S.; Kharazmi, A.; Shafiee, A. Synthesis and in vitro leishmanicidal activity of 2-(1-methyl-5-nitro-1H- imidazol-2-yl)-5-substituted-1,3,4-thiadiazole derivatives. Eur. J. Med. Chem. 2005, 40, 1346–1350. [Google Scholar] [CrossRef]
- Oruc, E.E.; Rollas, S.; Kandermirli, S.; Shvets, N.; Dimoglo, A.S. 1,3,4-thiadiazole derivatives. Synthesis, structure elucidation, and structure-antituberculosis activity relationship investigation. J. Med. Chem. 2004, 47, 6760–6767. [Google Scholar] [CrossRef]
- Karakus, S.; Rollas, S. Synthesis and antituberculosis activity of new N-phenyl-N′-[4-(5-alkyl/arylamino-1,3,4-thiadiazole-2-yl)phenyl]thioureas. Farmaco 2002, 57, 577–581. [Google Scholar] [CrossRef]
- Solak, N.; Rollas, S. Synthesis and antituberculosis activity of 2-(aryl/alkylamino)-5-(4-aminophenyl)-1,3,4-thiadiazoles and their Schiff bases. ARKIVOC 2006, 12, 173–181. [Google Scholar] [CrossRef]
- Patole, J.; Shingnapurkar, D.; Padhye, S.; Ratledge, C. Schiff base conjugates of p-aminosalicylic acid as antimycobacterial agents. Bioorg. Med. Chem. Lett. 2006, 16, 1514–1517. [Google Scholar] [CrossRef]
- Foroumadi, A.; Kargar, Z.; Sakhteman, A.; Sharifzadeh, Z.; Mohammadi, R.F.; Kazemi, M.; Shafiee, A. Synthesis and antimycobacterial activity of some alkyl [5-(nitroaryl)-1,3,4-thiadiazol-2-ylthio]propionates. Bioorg. Med. Chem. Lett. 2006, 16, 1164–1167. [Google Scholar] [CrossRef]
- Tahghighi, A.; Babalouei, F. Thiadiazoles: The appropriate pharmacological scaffolds with leishmanicidal and antimalarial activities: A review. Iran. J. Basic Med. Sci. 2017, 20, 613–622. [Google Scholar] [CrossRef]
- Gudala, S.; Ambati, S.R.; Patel, J.L.; Vedula, R.R.; Penta, S. An Efficient Synthesis of Pyrazolyl-1,2,3-thiadiazoles viaHurd-Mori Reaction. J. Heterocycl. Chem. 2019, 56, 2163–2169. [Google Scholar] [CrossRef]
- Jain, K.A.; Sharma, S.; Vaidya, A.; Ravichandran, V.; Agrawal, R.K. 1,3,4-Thiadiazole and its Derivatives: A Review on Recent Progress in Biological Activities. Chem. Biol. Drug Des. 2013, 81, 557–576. [Google Scholar] [CrossRef]
- Supuran, C.T.; Scozzafava, A. Carbonic anhydrase inhibitors. Curr. Med. Chem. Immunol. Endocrinol. Metab. Agents 2001, 1, 61–97. [Google Scholar] [CrossRef]
- Georgeta, S.; Oana, S.; Eugenia, S.; Santa, B. 2-Amino-1,3,4-thiadiazole as a potential scaffold for promising antimicrobial agents. Drug Des. Devel. Ther. 2018, 12, 1545–1566. [Google Scholar] [CrossRef]
- Iizawa, Y.; Okonogi, K.; Hayashi, R.; Iwahi, T.; Yamazaki, T.; Imada, A. Therapeutic effect of cefozopran (SCE-2787), a new parenteral cephalosporin, against experimental infections in mice. Antimicrob. Agents Chemother. 1993, 37, 100–105. [Google Scholar] [CrossRef]
- Sharma, B.; Verma, A.; Prajapati, S.; Sharma, K.U. Synthetic methods, chemistry, and the anticonvulsant activity of thiadiazoles. Int. J. Med. Chem. 2013, 2013. [Google Scholar] [CrossRef]
- Kucuk, H.B.; Salt, Z.B.; Kara, E.M.; Mehan, A.S.; Yusufoglu, A.S. Synthesis of novel 1,2,3-thiadizoles and 1,2,3-selenadiazoles as new antimicrobial agents. Phosphorus Sulfur Silicon Relat. Elem. 2019, 194. [Google Scholar] [CrossRef]
- Ngoc, T.D. Synthesis and characterization of some new 1,2,3-thiadiazole and 1,2,3-selenadiazole triterpene derivatives from allobetulone and 2-oxoallobetulin. Synth. Commun. 2020, 50, 1665–1671. [Google Scholar] [CrossRef]
- Kumar, A.; Muthyala, K.M.; Choudhary, S.; Tiwari, R.K.; Parang, K. Ionic Liquid as Soluble Support for Synthesis of 1,2,3-Thiadiazoles and 1,2,3-Selenadiazoles. J. Org. Chem. 2012, 77, 9391–9396. [Google Scholar] [CrossRef]
- Chen, J.; Jiang, Y.; Yu, J.T.; Cheng, J. TBAI-Catalyzed Reaction between N-Tosylhydrazones and Sulfur: A Procedure toward 1,2,3-Thiadiazole. J. Org. Chem. 2016, 81, 271–275. [Google Scholar] [CrossRef]
- Sun, N.-B.; Fu, J.Q.; Weng, J.-Q.; Jin, J.-Z.; Tan, C.-X.; Liu, X.-H. Microwave Assisted Synthesis, Antifungal Activity and DFTTheoretical Study of Some Novel 1,2,4-Triazole Derivatives Containing the 1,2,3-Thiadiazole Moiety. Molecules 2013, 18, 12725–12739. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Mi, N.; Fan, Z.; Zuo, X.; Zhang, H.; Wang, H.; Yang, Z. 5-Methyl-1,2,3-thiadiazoles Synthesized via Ugi Reaction and Their Fungicidal and Antiviral Activities. J. Agric. Food Chem. 2010, 58, 7846–7855. [Google Scholar] [CrossRef]
- Wang, S.-X.; Fang, Z.; Fan, Z.J.; Wang, D.; Li, Y.-D.; Li, X.-T.; Hua, X.-W.; Huang, Y.; Kalinina, A.T.; Bakulev, A.V.; et al. Synthesis of tetrazole containing 1,2,3-thiadiazole derivatives via U-4CR and their anti-TMV activity. Chin. Chem. Lett. 2013, 24, 889–892. [Google Scholar] [CrossRef]
- Wang, C.; Geng, X.; Zhao, P.; Zhou, Y.; Wu, Y.D.; Cui, Y.F.; Wu, A.X. I2/CuCl2-promoted one-pot three-component synthesis of aliphatic or aromatic substituted 1,2,3-thiadiazoles. Chem. Commun. 2019, 55, 8134–8137. [Google Scholar] [CrossRef]
- Dong, W.-L.; Liu, Z.-X.; Liu, X.-H.; Li, Z.-M.; Zhao, W.-G. Synthesis and antiviral activity of new acrylamide derivatives containing1,2,3-thiadiazole as inhibitors of hepatitis B virus replication. Eur. J. Med. Chem. 2010, 45, 1919–1926. [Google Scholar] [CrossRef]
- Hayat, F.; Salahuddin, A.; Zargan, J.; Azam, A. Synthesis, characterization, antiamoebic activity and cytotoxicity of novel 2-(quinolin-8-yloxy) acetohydrazones and their cyclized products (1,2,3-thiadiazole and 1,2,3-selenadiazole derivatives). Eur. J. Med. Chem. 2010, 45, 6127–6134. [Google Scholar] [CrossRef]
- Du, Q.-S.; Shi, Y.-X.; Li, P.-F.; Zhao, Z.-J.; Zhu, W.-P.; Qian, X.-H.; Li, B.-J.; Xu, Y.-F. Novel plant activators with thieno[2,3-d]-1,2,3-thiadiazole-6-carboxylate scaffold: Synthesis and bioactivity. Chin. Chem. Lett. 2013, 24, 967–969. [Google Scholar] [CrossRef]
- Yang, Z.; Liang, Y.; Li, A.; Liu, K.; Li, L.; Yang, T.; Zhou, C. One-Pot Synthesis of 5-Acyl-1,2,3-Thiadiazoles from Enaminones, Tosylhydrazine and Elemental Sulfur under Transition Metal-Free Conditions. J. Org. Chem. 2019, 84, 16262–16267. [Google Scholar] [CrossRef]
- Zhang, L.; Sun, B.; Liu, Q.; Mo, F. Addition of Diazo Compounds ipso-C-H Bond to Carbon Disulfide: Synthesis of 1,2,3-Thiadiazoles under Mild Conditions. J. Org. Chem. 2018, 83, 4275–4278. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Cao, Y.; Lu, L.; Zhang, S.; Bao, W.; Huang, S.; Rao, Y. Perylenequinonoid-Catalyzed [4 + 1] and [4 + 2] Annulations of Azoalkenes: Photocatalytic Access to 1,2,3-Thiadiazole/1,4,5,6-Tetrahydropyridazine Derivatives. J. Org. Chem. 2019, 84, 7711–7721. [Google Scholar] [CrossRef]
- Teplyakov, S.F.; Vasileva, G.T.; Petrov, L.M.; Androsov, A.D. A New Synthesis of Benzo[b]thiophene-2-thiolates and Their Derivatives via Base-Promoted Transformation of 4-(2-Mercaptophenyl)-1,2,3-thiadiazoles. Org. Lett. 2013, 15, 4038–4041. [Google Scholar] [CrossRef]
- Fan, W.; Li, Q.; Li, Y.; Sun, H.; Jiang, B.; Li, G. I2/O2-Enabled N-S Bond Formation to Access Functionalized 1,2,3-Thiadiazoles. Org. Lett. 2016, 18, 1258–1261. [Google Scholar] [CrossRef]
- Belyaev, A.N.; Beryozkina, V.T.; Bakulev, A.V. Synthesis of N-heteroarylamidines of 1,2,3-thiadiazole-4-carboxylic acid from 2-cyanothioacetamides and 5-azido-1-methyl-4-nitroimidazole. Chem. Heterocycl. Compd. 2016, 52, 206–208. [Google Scholar] [CrossRef]
- Liu, B.-B.; Bai, H.-W.; Liu, H.; Wang, S.-Y.; Ji, S.-J. A cascade trisulfur radical anion (S3•-) addition/electron detosylation process for the synthesis of 1,2,3-thiadiazoles and isothiazoles. J. Org. Chem. 2018, 83, 10281–10288. [Google Scholar] [CrossRef] [PubMed]
- Pawar, P.; Chawla, A.; Sharma, P.; Arora, S. Novel drug delivery approaches on antiviral and antiretroviral agents. J. Adv. Pharm. Technol. Res. 2012, 3, 147–159. [Google Scholar] [CrossRef]
- Rossignol, J.F. Nitazoxanide: A first-in-class broad-spectrum antiviral agent. Antivir. Res. 2014, 110, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Raymund, R.; Razonable, M.D. Antiviral Drugs for Viruses Other Than Human Immunodeficiency Virus. Mayo Clin. Proc. 2011, 86, 1009–1026. [Google Scholar] [CrossRef]
- Nystrom, K.; Waldenstrom, J.; Tang, K.-W.; Lagging, M. Ribavirin: Pharmacology, multiple modes of action and possible future perspectives. Future Virol. 2019, 14, 153–160. [Google Scholar] [CrossRef]
- Sidwell, R.W.; Huffman, J.H.; Khare, G.P.; Allen, L.B.; Witkowski, J.T.; Robins, R.K. Broad-spectrum antiviral activity of virazole: 1-beta-D-ribofuranosyl-1,2,4-triazole-3-carboxamide. J. Sci. 1972, 177, 705–706. [Google Scholar] [CrossRef]
- Rakhmanina, N.Y.; Vanda, V.J.N. Efavirenz in the therapy of HIV infection. Expert Opin. Drug Metab. Toxicol. 2009, 6, 95–103. [Google Scholar] [CrossRef]
- Larru, B.; Eby, J.; Lowenthal, E.D. Antiretroviral treatment in HIV-1 infected pediatric patients: Focus on efavirenz. Pediatric Health Med. Ther. 2014, 2014, 29–42. [Google Scholar] [CrossRef][Green Version]
- Kryst, J.; Kawalec, P.; Pilc, A. Efavirenz-Based Regimens in Antiretroviral-Naive HIV-Infected Patients: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. PLoS ONE 2015, 10, e0124279. [Google Scholar] [CrossRef]
- Leneva, I.A.; Russell, R.J.; Boriskin, Y.S.; Hay, A.J. Characteristics of arbidol-resistant mutants of influenza virus: Implications for the mechanism of anti-influenza action of arbidol. Antivir. Res. 2009, 81, 132–140. [Google Scholar] [CrossRef]
- Liu, Q.; Xiong, H.; Lu, L.; Liu, Y.; Luo, F.; Hou, W.; Yang, Z. Antiviral and anti-inflammatory activity of arbidol hydrochloride in influenza A (H1N1) virus infection. Acta Pharmacol. Sin. 2013, 34, 1075–1083. [Google Scholar] [CrossRef]
- Zhan, P.; Liu, X.; Cao, Y.; Wang, Y.; Pannecouque, C.; Clercq, E.D. 1,2,3-thiadiazole thioacetanilides as a novel class of potent HIV-1 non-nucleoside reverse transcriptase inhibitors. Bioorg. Med. Chem. Lett. 2008, 18, 5368–5371. [Google Scholar] [CrossRef]
- Zhan, P.; Liu, X.; Li, Z.; Fang, Z.; Li, Z.; Wang, D.; Pannecouque, C.; Clercq, E.D. Novel 1,2,3-thiadiazole derivatives As HIV-1 NNRTIs with improved potency: Synthesis and preliminary SAR studies. Bioorg. Med. Chem. Lett. 2009, 17, 5920–5927. [Google Scholar] [CrossRef]
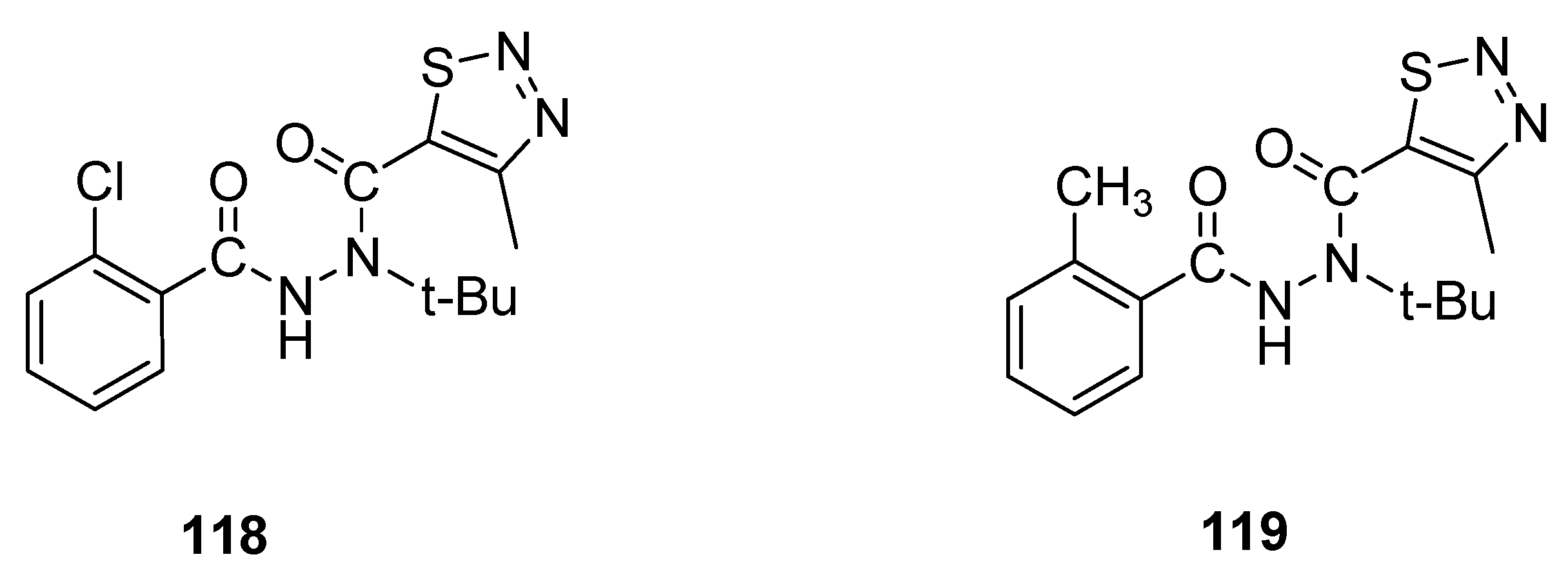
- Mao, W.T.; Zhao, H.; Fan, Z.J.; Ji, X.T.; Hua, X.W.; Kalinina, T.; Yury, Y.M.; Vasiliy, A.B. Synthesis and bioactivity of N-tert-butyl-N′-acyl-5-methy l-1,2,3-thiadiazole-4-carbohydrazides. Chin. Chem. Lett. 2012, 23, 1233–1236. [Google Scholar] [CrossRef]
- Xu, W.-M.; Li, S.-Z.; He, M.; Yang, S.; Li, X.-Y.; Li, P. Synthesis and bioactivities of novel thioether/sulfone derivatives containing 1,2,3-thiadiazole and 1,3,4-oxadiazole/thiadiazole moiety. Bioorg. Med. Chem. Lett. 2013, 23, 5821–5824. [Google Scholar] [CrossRef]
- Zhan, P.; Liu, X.Y.; Li, Z.Y.; Fang, Z.-J.; Pannecouque, C.; Clercqet, E.D. Synthesis and biological evaluation of a new series of 2-{[4-(3,4-dichlorophenyl)-1,2,3-thiadiazol-5-yl] sulfanyl}acetanilides as HIV-1 inhibitors. Chem. Biodivers. 2010, 7, 1717–1727. [Google Scholar] [CrossRef]
- Grasso, C.S.; Wu, Y.M.; Robinson, D.R.; Cao, X.; Dhanasekaran, S.M.; Khan, A.P.; Quist, M.J.; Jing, X.J.; Lonigro, R.J.; Brenner, J.C.; et al. The mutational landscape of lethal castration-resistant prostate cancer. Nature 2012, 487, 239–243. [Google Scholar] [CrossRef]
- Irfan, A.; Batool, F.; Naqvi, S.A.Z.; Islam, A.; Osman, S.M.; Nocentini, A.; Alissa, S.A.; Supuran, C.T. Benzothiazole derivatives as anticancer agents. J. Enzym. Inhib. Med. Chem. 2020, 35, 265–279. [Google Scholar] [CrossRef]
- Pattabiraman, V.R.; Bode, J.W. Rethinking amide bond synthesis. Nature 2011, 480, 471–479. [Google Scholar] [CrossRef]
- He, Z.C.; Ling, G.L.; Yi, Z.Y.; Fei, Z.F.; Zhou, W.G.; Lei, J.; Xia, G.R. Review on supermolecules as chemical drugs. Sci. China Ser. B 2009, 52, 415–458. [Google Scholar] [CrossRef]
- Dasari, S.; Tchounwou, P.B. Cisplatin in cancer therapy: Molecular mechanisms of action. Eur. J. Pharmacol. 2014, 740, 364–378. [Google Scholar] [CrossRef]
- Gangadhara, S.; Bertelli, G. Long-term efficacy and safety of anastrozole for adjuvant treatment of early breast cancer in postmenopausal women. Ther. Clin. Risk Manag. 2009, 5, 291–300. [Google Scholar] [CrossRef]
- Barros-Oliveira, M.C.; Costa-Silva, D.R.; Andrade, D.B.; Borges, U.S.; Tavares, C.B.; Borges, R.S.; Silva, J.M.; Silva, B.B. Use of anastrozole in the chemoprevention and treatment of breast cancer: A literature review. Rev. Assoc. Med. Bras. 2017, 63, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Tacar, O.; Sriamornsak, P.; Dass, C.R. Doxorubicin: An update on anticancer molecular action, toxicity and novel drug delivery systems. J. Pharm. Pharmacol. 2013, 65, 157–170. [Google Scholar] [CrossRef]
- Pereyra, C.E.; Dantas, R.F.; Ferreira, S.B.; Gomes, L.P.; Silva, F.P. The diverse mechanisms and anticancer potential of naphthoquinones. Cancer Cell Int. 2019, 19, 207. [Google Scholar] [CrossRef]
- Thorn, C.F.; Oshiro, C.; Marsh, S.; Hernandez, B.T.; McLeod, H.; Klein, T.E.; Altman, R.B. Doxorubicin pathways: Pharmacodynamics and adverse effects. Pharm. Genom. 2011, 21, 440–446. [Google Scholar] [CrossRef]
- Al-Malky, S.H.; Damanhouri, Z.A.; Aama, J.Y.A.; Qahtani, A.A.A.; Ramadan, W.S.; AlKreathy, H.M.; Harthi, S.E.A.; Osman, A.M.M. Diltiazem potentiation of doxorubicin cytotoxicity and cellular uptake in human breast cancer cells. Breast Cancer Manag. 2019, 8, BMT31. [Google Scholar] [CrossRef]
- Wu, M.; Sun, Q.; Yang, C.; Chen, D.; Ding, J.; Chen, Y.; Lin, L.; Xie, Y. Synthesis and activity of Combretastatin A-4 analogues: 1,2,3-thiadiazoles as potent antitumor agents. Bioorg. Med. Chem. Lett. 2007, 17, 869–873. [Google Scholar] [CrossRef]
- Hosny, M.A.; El-Sayed, T.H.; El-Sawi, E.A. A New Type of Synthesis of 1,2,3-thiadiazole and 1,2,3-diazaphosphole derivatives Via-Hurd-Mori cyclization. J. Chem. 2012, 9, 1276–1287. [Google Scholar] [CrossRef]
- Cikotiene, I.; Kazlauskas, E.; Matuliene, J.; Michailoviene, V.; Torresan, J.; Jachno, J.; Matuliset, D. 5-Aryl-4-(5-substituted-2,4-dihydroxyphenyl)-1,2,3-thiadiazoles as inhibitors of Hsp90 chaperone. Bioorg. Med. Chem. Lett. 2009, 19, 1089–1092. [Google Scholar] [CrossRef]
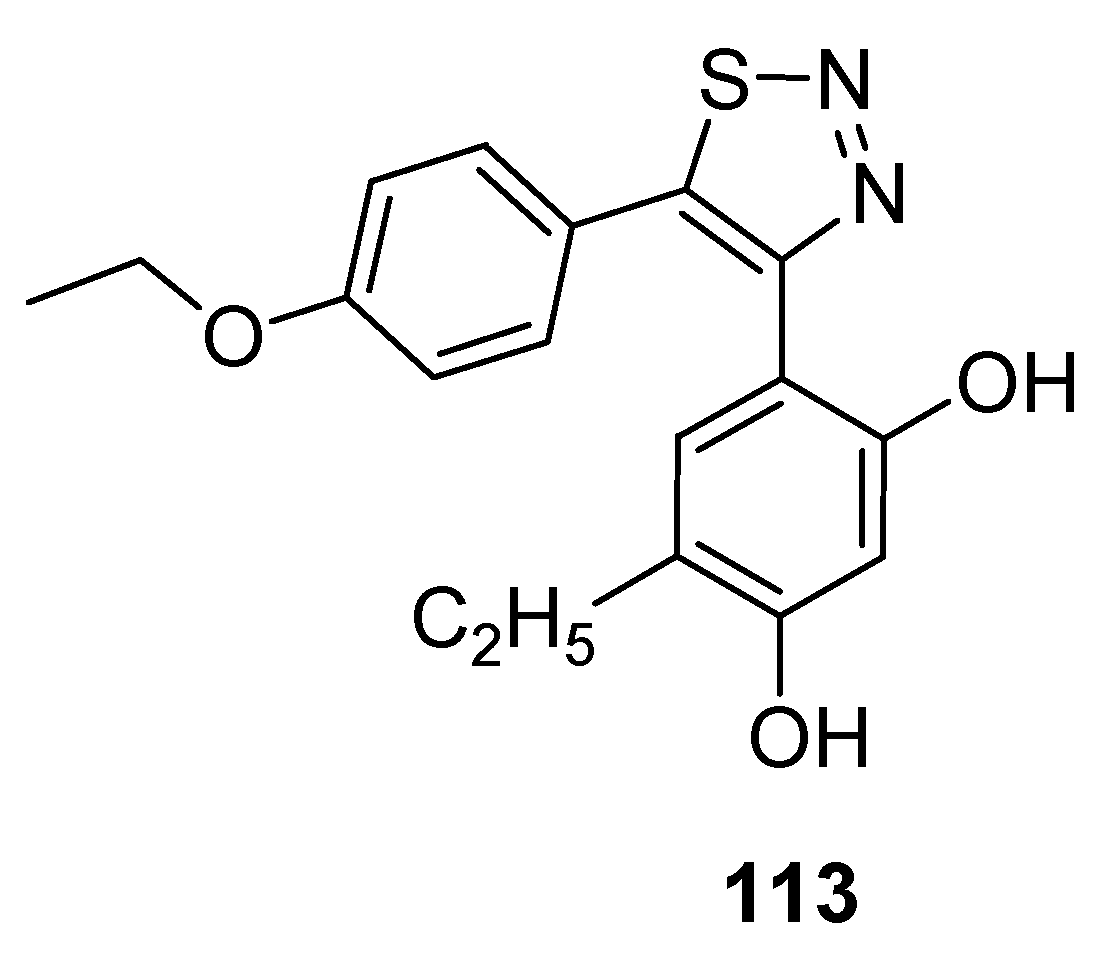
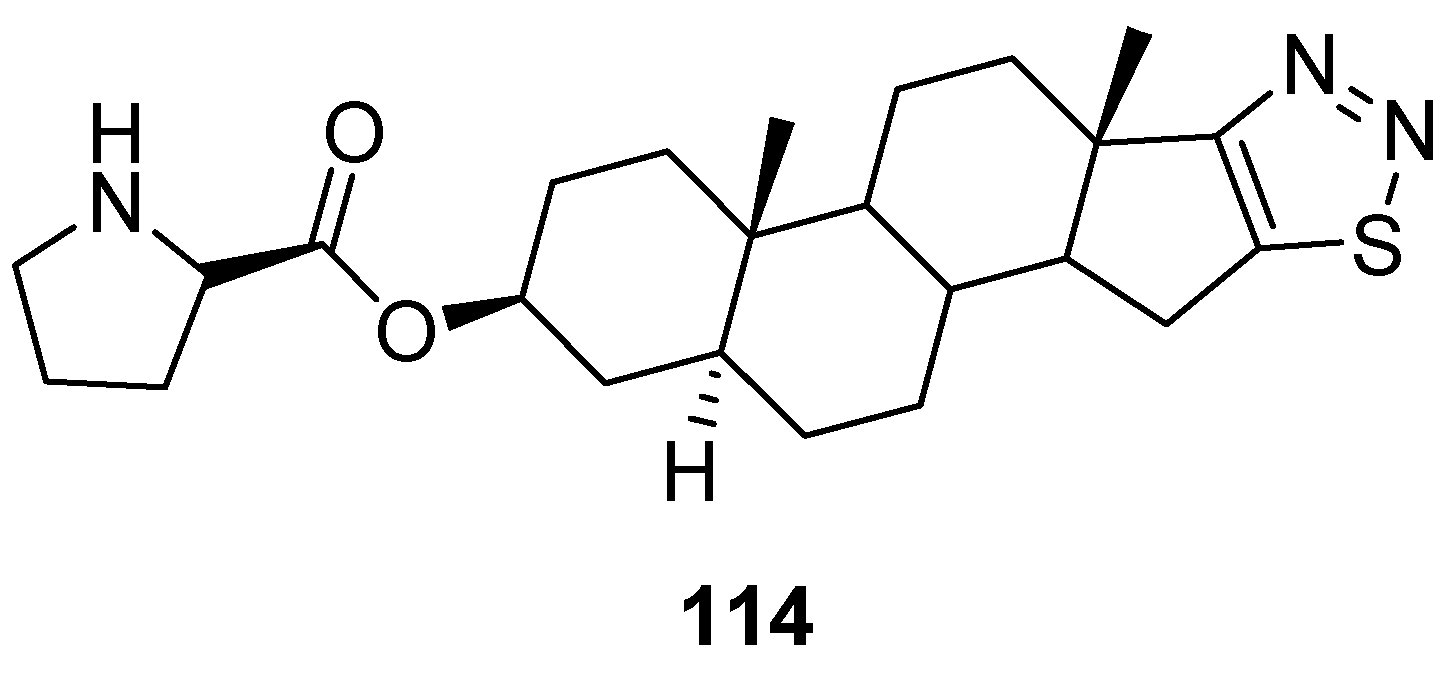
- Cui, H.W.; Peng, S.; Gu, X.Z.; Chen, H.; He, Y.; Gao, W.; Lv, F.; Wang, J.-H.; Wang, Y.; Xie, J.; et al. Synthesis and biological evaluation of D-ring fused 1,2,3-thiadiazole dehydro epiandro sterone derivatives as antitumor agents. Eur. J. Med. Chem. 2016, 111, 126–137. [Google Scholar] [CrossRef] [PubMed]
- Oberemok, V.V.; Laikova, K.V.; Gninenko, Y.I.; Zaitsev, A.S.; Nyadar, P.M.; Adeyemi, T.A. A short history of insecticides. J. Plant Prot. Res. 2015, 55, 221–226. [Google Scholar] [CrossRef]
- Xu, Z.; Shi, L.; Jiang, D.; Cheng, J.; Shao, X.; Li, Z. Azobenzene Modified Imidacloprid Derivatives as Photoswitchable Insecticides: Steering Molecular Activity in a Controllable Manner. Sci. Rep. 2015, 5, 13962. [Google Scholar] [CrossRef]
- Carlson, G.R. Tebufenozide: A Novel Caterpillar Control Agent with Unusually High Target Selectivity. Green Chemical Syntheses and Processes. ACS Symp. Ser. 2000, 767, 8–17. [Google Scholar] [CrossRef]
- Zhang, A.; Kayser, H.; Maeinfisch, P. Insect nicotinic acetylcholine receptor: Conserved neonicotinoid specificity of imidacloprid binding site. J. Neurochem. 2002, 75, 1294–1303. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Verma, A.; Kumar, A. Accidental human poisoning with a neonicotinoid insecticide, imidacloprid: A rare case report from rural India with a brief review of literature. Egypt. J. Forensic Sci. 2013, 3, 123–126. [Google Scholar] [CrossRef]
- Mundhe, S.A.; Birajdar, S.V.; Chavan, S.S.; Pawar, N.R. Imidacloprid Poisoning: An Emerging Cause of Potentially Fatal Poisoning. Indian J. Crit. Care Med. 2017, 21, 786–788. [Google Scholar] [CrossRef]
- Ausborn, J.; Wolf, H.; Mader, W.; Kayser, H. The insecticide pymetrozine selectively affects chordotonal mechanoreceptors. J. Exp. Biol. 2005, 28, 4451–4466. [Google Scholar] [CrossRef]
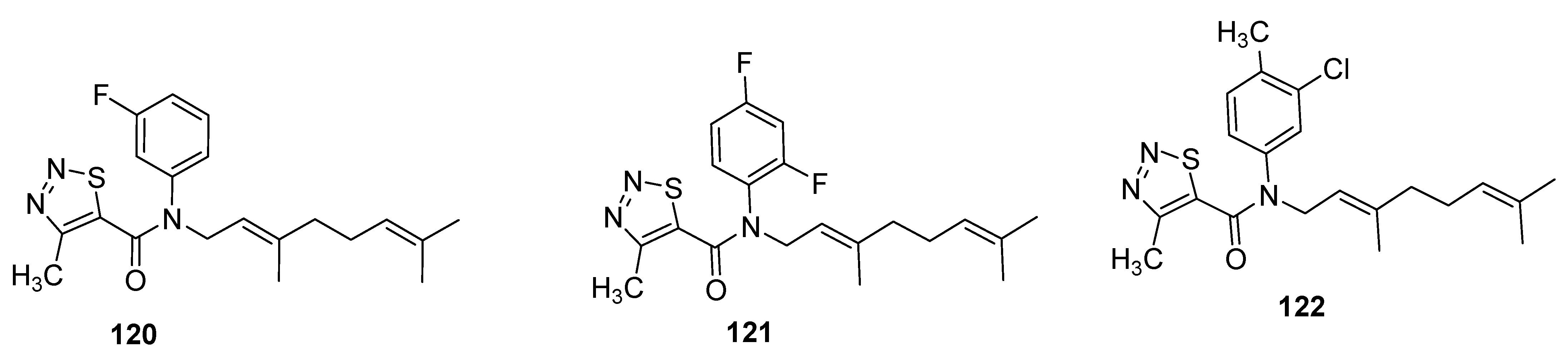
- Zhang, J.P.; Qin, Y.G.; Dong, Y.W.; Song, D.-L.; Duan, H.-X.; Yang, X.-L. Synthesis and biological activities of (E)-B-farnesene analogues 4 containing 1,2,3-thiadiazole. Chin. Chem. Lett. 2016, 28, 372–376. [Google Scholar] [CrossRef]
- Dai, H.; Ge, S.; Li, G.; Chen, J.; Shi, Y.; Ye, L.; Ling, Y. Synthesis and bioactivities of novel pyrazole oxime derivatives containing a 1,2,3-thiadiazole moiety. Bioorg. Med. Chem. Lett. 2016, 28, 4504–4507. [Google Scholar] [CrossRef]
- Li, Y.-D.; Mao, W.-T.; Fan, Z.-J.; Li, J.-J.; Fang, Z.; Ji, X.-T.; Hua, X.-W.; Zong, G.-N.; Li, F.-Y.; Liu, C.-L.; et al. Synthesis and biological evaluation of novel 1,2,4-triazole containing 1,2,3-thiadiazole derivatives. Chin. Chem. Lett. 2013, 24, 1134–1136. [Google Scholar] [CrossRef]
- Bercu, T.E.; Petri, W.A.; Behm, J.W. Amebic colitis: New insights into pathogenesis and treatment. Curr. Gastroenterol. Rep. 2007, 9, 429–433. [Google Scholar] [CrossRef]
- Choudhuri, G.; Rangan, M. Amebic infection in humans. Indian J. Gastroenterol. 2012, 31, 153–162. [Google Scholar] [CrossRef]
- Shirley, D.T.; Farr, L.; Watanabe, K.; Moonah, S. A review of the global burden, new diagnostics, and current therapeutics for amebiasis. Open Forum Infect. Dis. 2018, 5, ofy161. [Google Scholar] [CrossRef] [PubMed]
- Farrar, J.; Hotez, P.; Junghanss, T.; Kang, G.; Lalloo, D.; White, N. Manson’s Tropical Diseases; Elsevier Health Sciences: Amsterdam, The Netherlands, 2013; pp. 664–671. ISBN 9780702053061. [Google Scholar]
- Lofmark, S.; Edlund, C.; Nord, C.E. Metronidazole Is Still the Drug of Choice for Treatment of Anaerobic Infections. Clin. Infect. Dis. 2010, 50, S16–S23. [Google Scholar] [CrossRef]
- Steinitz, H. The Treatment of Chronic Amoebic Infection with Metronidazole (Flagyl). Digestion 1972, 6, 75–82. [Google Scholar] [CrossRef]
- Gonzales, M.L.M.; Dans, L.F.; Martinez, E.G. Antiamoebic drugs for treating amoebic colitis. Cochrane Database Syst. Rev. 2009, 2, CD006085. [Google Scholar] [CrossRef]
- Ghaskadbi, S.; Vaidya, V.G. In vivo antimutagenic effect of ascorbic acid against mutagenicity of the common antiamebic drug diiodohydroxyquinoline. Mutat. Res. 1989, 222, 219–222. [Google Scholar] [CrossRef]
- Aranega-Bou, P.O.; Leyva, M.; Finiti, I.; Garcia-Agustin, P.; Gonzalez-Bosch, C. Priming of plant resistance by natural compounds. Hexanoic acid as a model. Front. Plant Sci. 2014, 5, 488. [Google Scholar] [CrossRef]
- Li, J.; Long, T.; Sun, T.-J.; Lu, Y.; Yin, J.; Yang, Y.-B.; Dai, G.Y.; Zhu, X.Y.; Yao, N. Pyrimidin-Like Plant Activator Stimulates Plant Disease Resistance and Promotes the Synthesis of Primary Metabolites. Int. J. Mol. Sci. 2020, 21, 2705. [Google Scholar] [CrossRef]
- Conrath, U.; Beckers, G.J.M.; Langenbach, C.J.G.; Jaskiewicz, M.R. Priming for enhanced defense. Annu. Rev. Phytopathol. 2015, 53, 97–119. [Google Scholar] [CrossRef] [PubMed]
- Cavalcanti, F.R.; Resendeb, M.L.V.; Limac, J.P.M.S.; Silveirac, J.A.G.; Oliveira, J.T.A. Activities of antioxidant enzymes and photosynthetic responses in tomato pretreated by plant activators and inoculated by Xanthomonas vesicatoria. Int. J. Plant Pathol. 2006, 68, 198–208. [Google Scholar]
- Martinez-Medina, A.; Flors, V.; Heil, M.; Mauch-Mani, B.; Pieterse, C.M.J.; Pozo, M.J.; Ton, J.; Dam, N.M.V. Recognizing Plant Defense Priming. Trends Plant Sci. 2016, 21, 818–822. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.K.; Agarwal, M. Toxicity of fungicides. In Veterinary Toxicology: Basic and Clinical Principles, 2nd ed.; Gupta, R.C., Ed.; Elsevier: Amsterdam, The Netherland, 2012; pp. 653–670. [Google Scholar]
- Ramudu, A.C.; Mohiddin, G.J.; Srinivasulu, M.; Madakka, M.; Rangaswamy, V. Impact of Fungicides Chlorothalonil and Propiconazole on Microbial Activities in Groundnut (Arachis hypogaea L.) Soils. ISRN Microbiol. 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- Bacmaga, T.M.; Wyszkowska, J.; Kucharski, J. The influence of chlorothalonil on the activity of soil microorganisms and enzymes. Ecotoxicology 2018, 27, 1188–1202. [Google Scholar] [CrossRef]
- Martin, M.V. The use of fluconazole and itraconazole in the treatment of Candida albicans infections: A review. J. Antimicrob. 1999, 44, 429–437. [Google Scholar] [CrossRef]
- Greer, D.; Jolly, H.W. Topical ketoconazole treatment of cutaneous candidiasis. J. Am. Acad. Dermatol. 1988, 18, 748–750. [Google Scholar] [CrossRef]
- Fan, Z.; Shi, Z.; Zhang, H.; Liu, X.; Bao, L.; Ma, L.; Zuo, X.; Zheng, Q.; Mi, N. Synthesis and biological activity evaluation of 1,2,3-thiadiazole derivatives as potential elicitors with highly systemic acquired resistance. J. Agric. Food Chem. 2009, 57, 4279–4286. [Google Scholar] [CrossRef]
- Tan, C.-X.; Weng, J.-Q.; Liu, Z.-X.; Liu, X.-H.; Zhao, W.-G. Synthesis, Crystal Structure, and Fungicidal Activity of a Novel 1,2,3-Thiadiazole Compound. Phosphorus Sulfur Silicon Relat. Elem. 2012, 187, 990–996. [Google Scholar] [CrossRef]
- Fan, Z.; Yang, Z.; Zhang, H.; Mi, N.; Wang, H.; Cai, F.; Zuo, X.; Zheng, Q.; Song, H. Synthesis, crystal structure, and biological activity of 4-methyl-1,2,3-thiadiazole-containing 1,2,4-triazolo[3,4-b][1,3,4]thiadiazoles. J. Agric. Food Chem. 2010, 58, 2630–2636. [Google Scholar] [CrossRef]
- Wang, Z.-H.; Guo, Y.-Z.; Zhang, J.; Ma, L.; Song, H.-B.; Fan, Z.-J. Synthesis and biological activity of organotin 4-Methyl-1,2,3-thiadiazole-5-carboxylates and benzo[1,2,3]thiadiazole-7-carboxylates. J. Agric. Food Chem. 2010, 58, 2715–2719. [Google Scholar] [CrossRef] [PubMed]
- Zuo, X.; Mi, N.; Fan, Z.; Zheng, Q.; Zhang, H.; Wang, H.; Yang, Z. Synthesis of 4-methyl-1,2,3-thiadiazole derivatives via Ugi reaction and their biological Activities. J. Agric. Food Chem. 2010, 58, 2755–2762. [Google Scholar] [CrossRef]


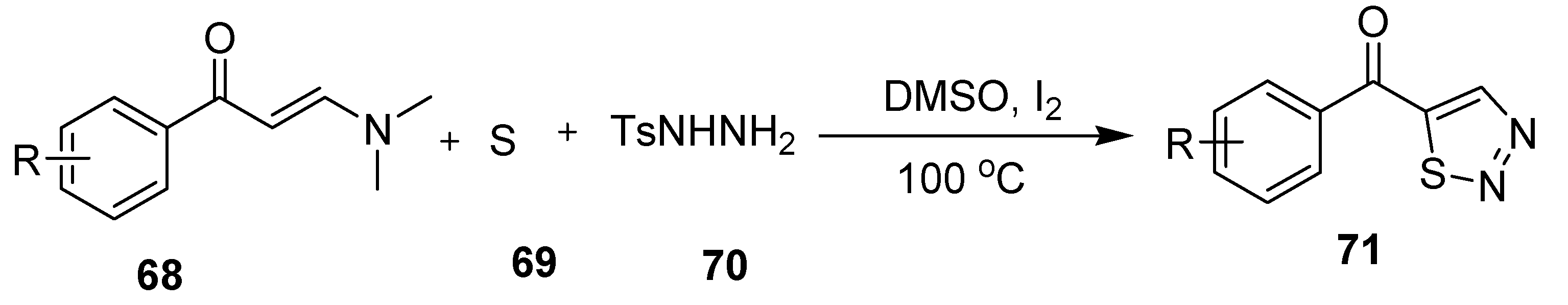


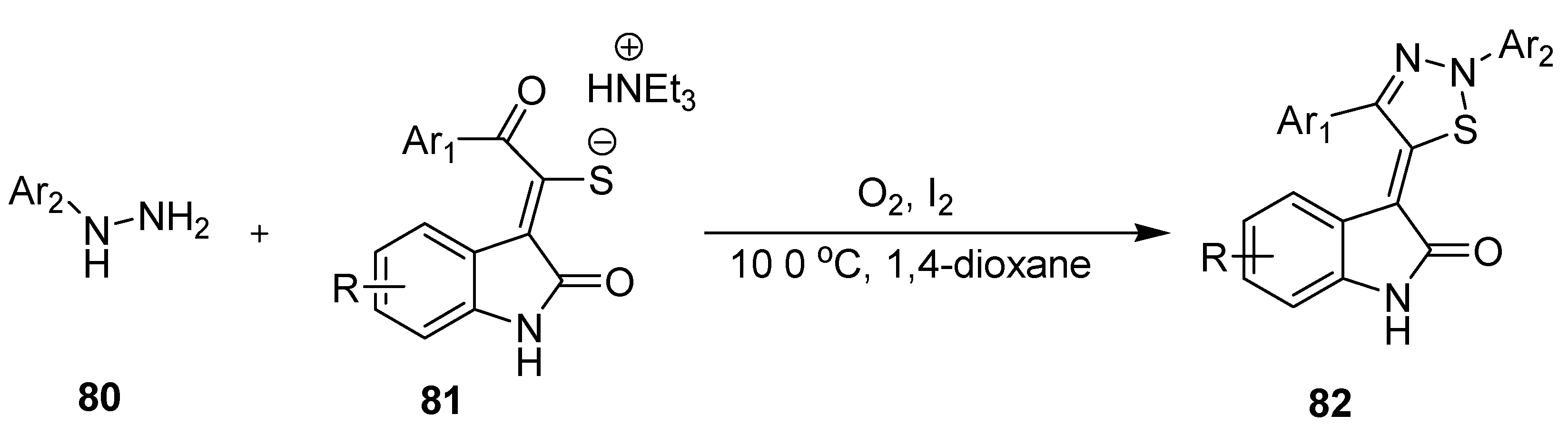








Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Irfan, A.; Ullah, S.; Anum, A.; Jabeen, N.; Zahoor, A.F.; Kanwal, H.; Kotwica-Mojzych, K.; Mojzych, M. Synthetic Transformations and Medicinal Significance of 1,2,3-Thiadiazoles Derivatives: An Update. Appl. Sci. 2021, 11, 5742. https://doi.org/10.3390/app11125742
Irfan A, Ullah S, Anum A, Jabeen N, Zahoor AF, Kanwal H, Kotwica-Mojzych K, Mojzych M. Synthetic Transformations and Medicinal Significance of 1,2,3-Thiadiazoles Derivatives: An Update. Applied Sciences. 2021; 11(12):5742. https://doi.org/10.3390/app11125742
Chicago/Turabian StyleIrfan, Ali, Sami Ullah, Ayesha Anum, Nazish Jabeen, Ameer Fawad Zahoor, Hafza Kanwal, Katarzyna Kotwica-Mojzych, and Mariusz Mojzych. 2021. "Synthetic Transformations and Medicinal Significance of 1,2,3-Thiadiazoles Derivatives: An Update" Applied Sciences 11, no. 12: 5742. https://doi.org/10.3390/app11125742
APA StyleIrfan, A., Ullah, S., Anum, A., Jabeen, N., Zahoor, A. F., Kanwal, H., Kotwica-Mojzych, K., & Mojzych, M. (2021). Synthetic Transformations and Medicinal Significance of 1,2,3-Thiadiazoles Derivatives: An Update. Applied Sciences, 11(12), 5742. https://doi.org/10.3390/app11125742








