More Than 50-Fold Enhanced Nonlinear Optical Response of Porphyrin Molecules in Aqueous Solution Induced by Mixing Base and Organic Solvent
Abstract
1. Introduction
2. Materials and Methods
3. Results
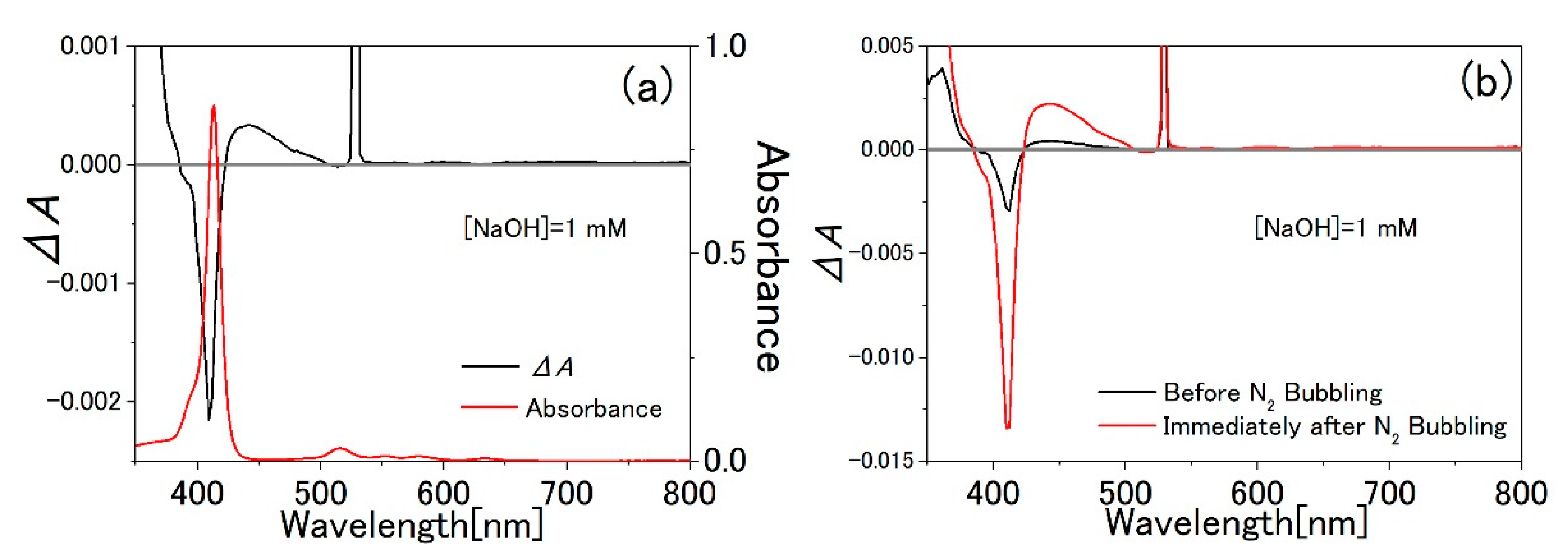
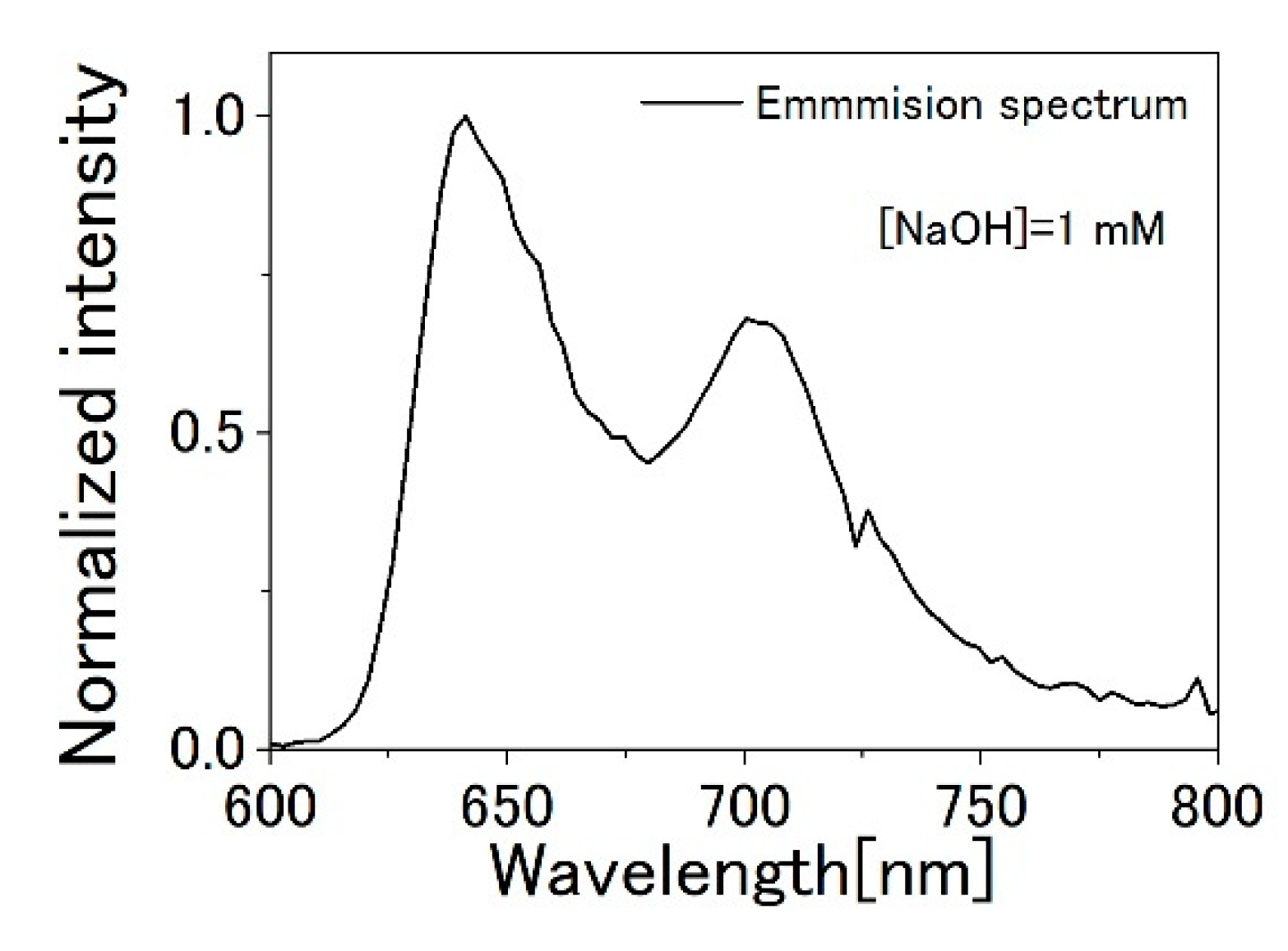
3.1. TPPS Aqueous Solution
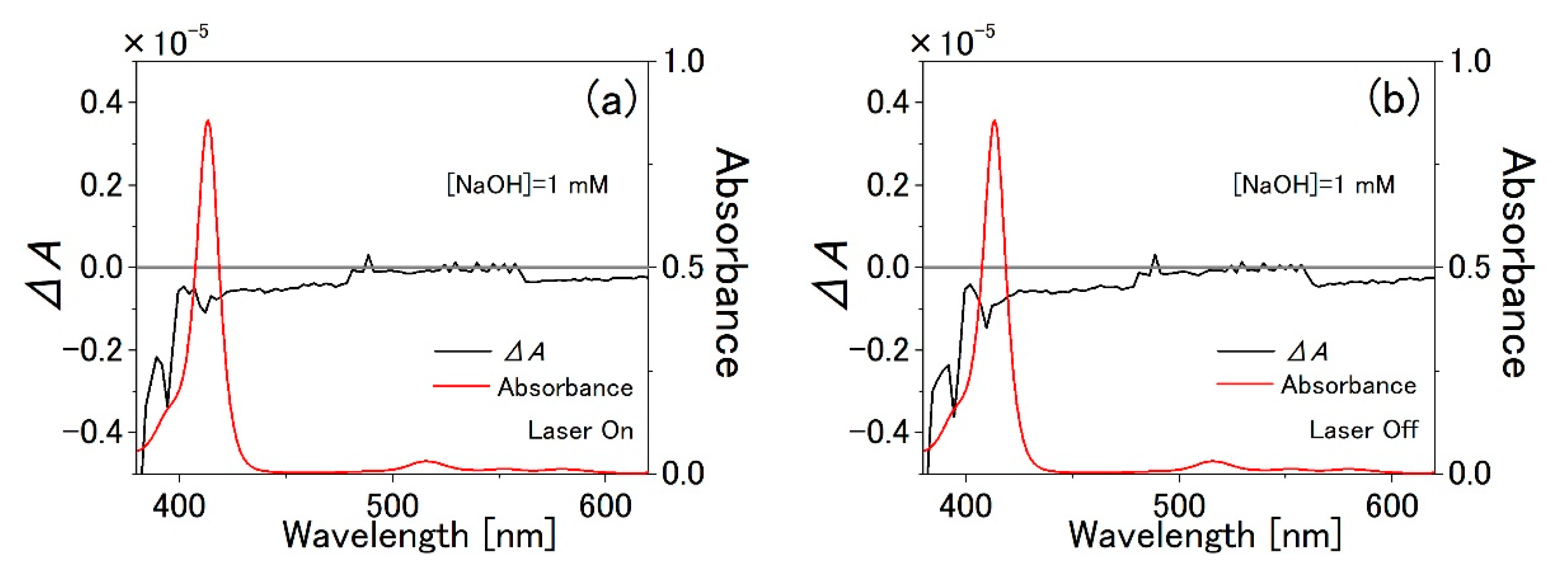
3.2. PVA Sample
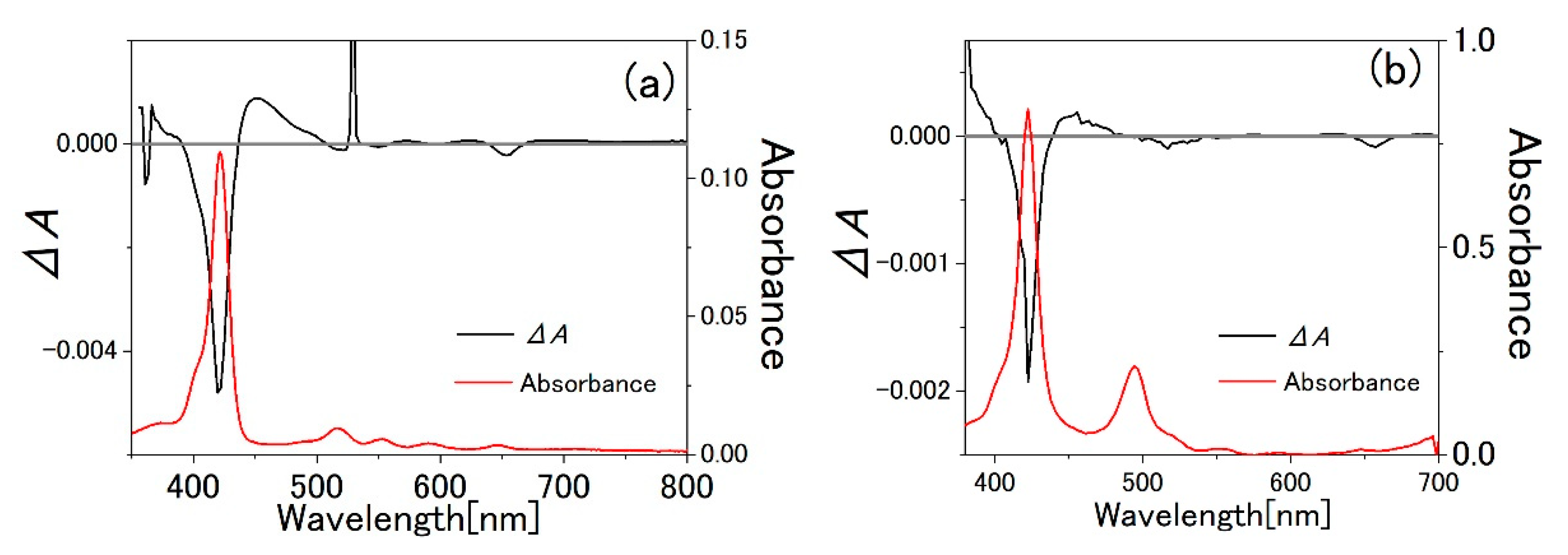
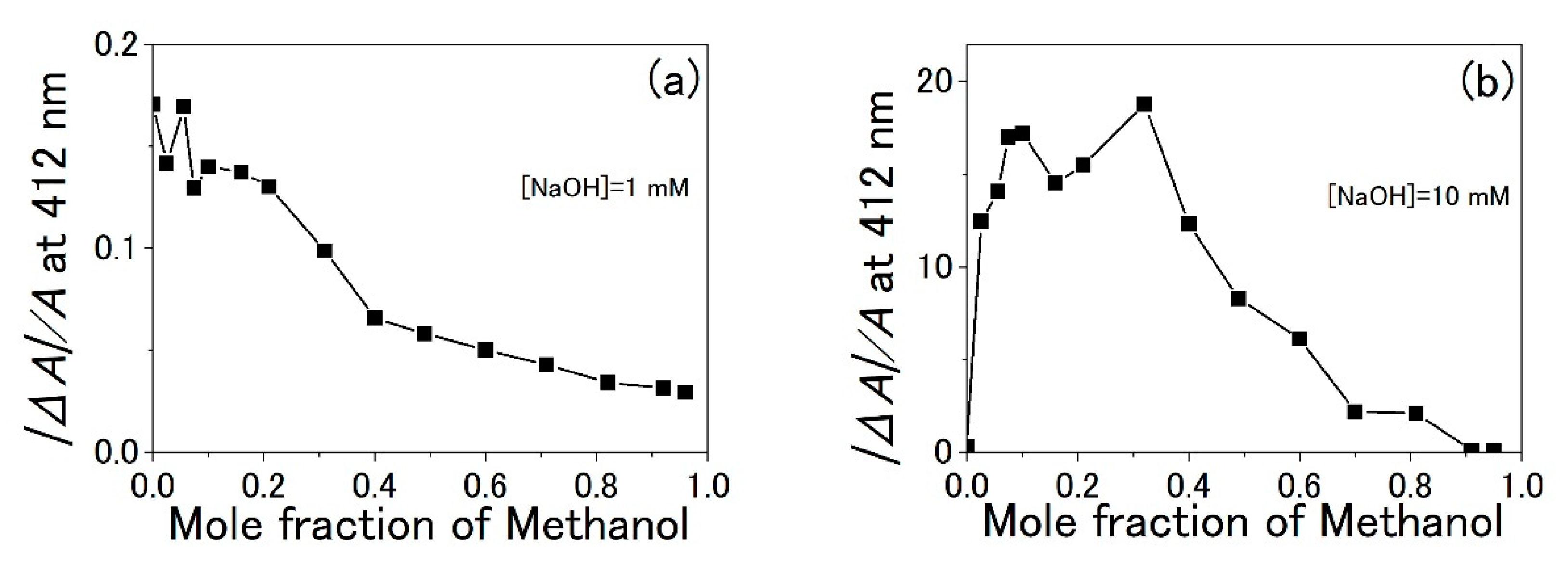
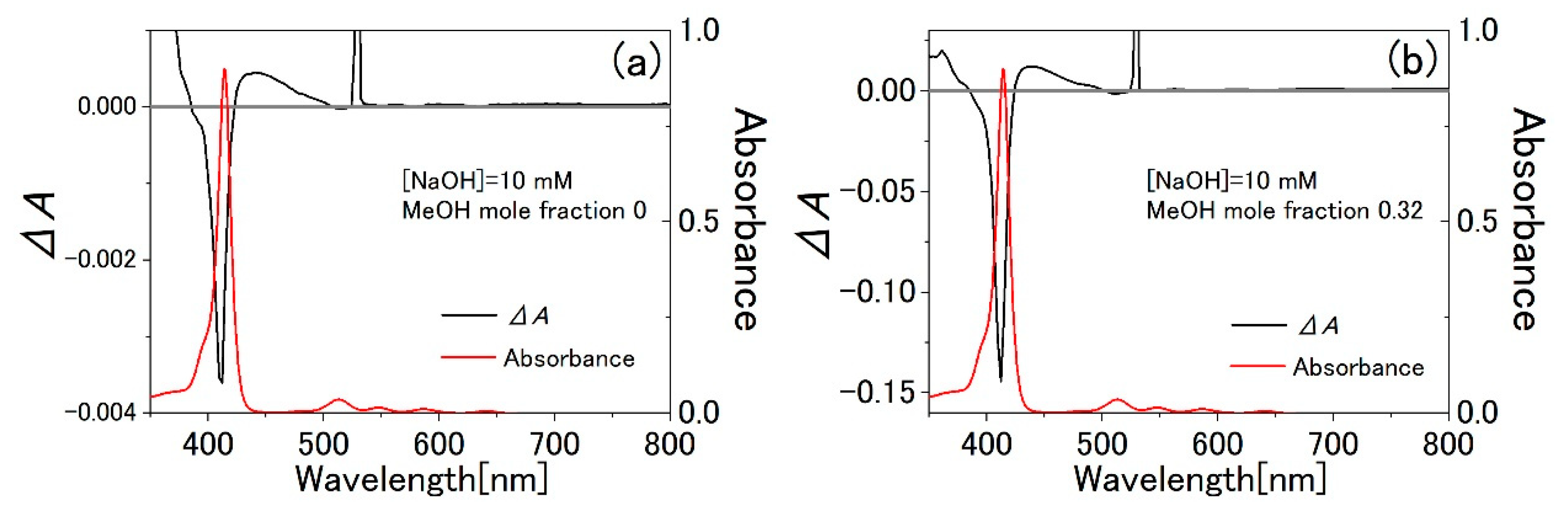
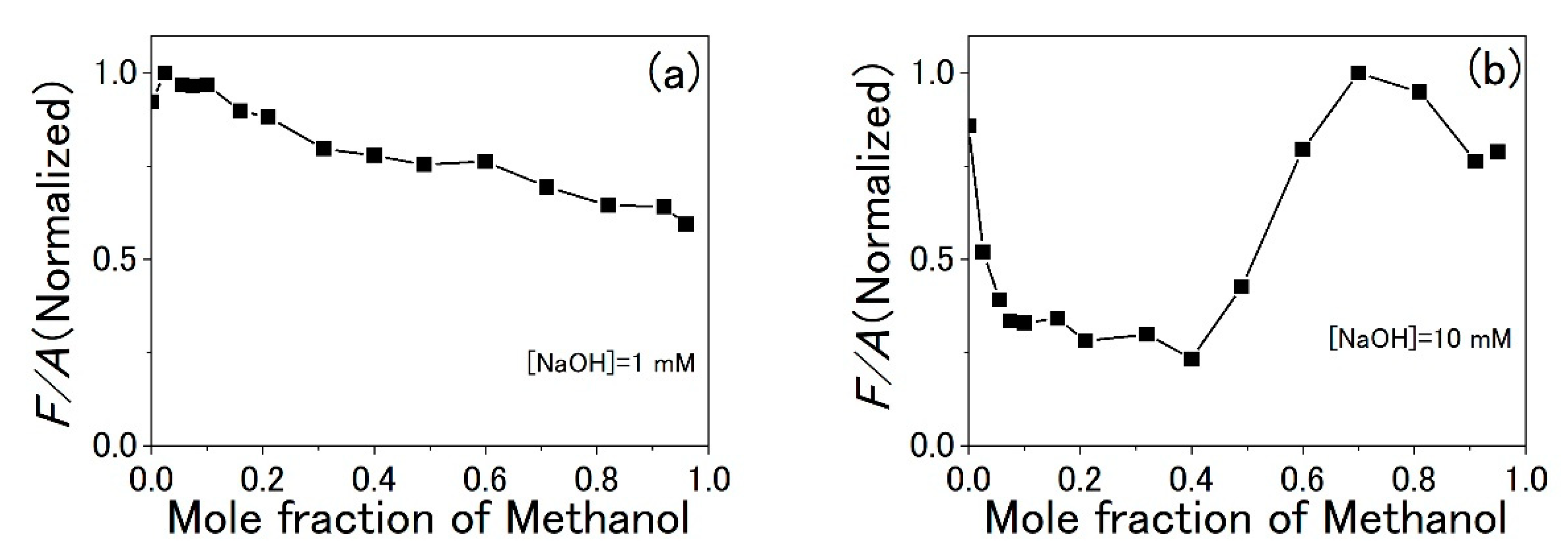
3.3. Water-Methanol Mixed Solution of TPPS
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shirakawa, M.; Nakata, K.; Suzuki, M.; Kobayashi, T.; Tokunaga, E. Nonlinear absorption spectroscopy of porphyrin j-aggregates in aqueous solution: Evidence for control of degree of association by light-induced force. J. Phys. Soc. Jpn. 2017, 86, 044703. [Google Scholar] [CrossRef]
- Shirakawa, M.; Kobayashi, T.; Tokunaga, E. Solvent effects in highly efficient light-induced molecular aggregation. Appl. Sci. 2019, 9, 5381. [Google Scholar] [CrossRef]
- Tsukahara, S.; Watarai, H. Kinetics for acid-dissociation of tetraphenylporphinetetrasulfonate in the ground state measured by laser photolysis relaxation method. Phys. Chem. Chem. Phys. 2002, 4, 1592–1597. [Google Scholar] [CrossRef]
- Bonnet, R.; Ridge, R.J. Pulsed irradiation of water-soluble porphyrins. J. Chem. Soc. Faraday Trans. 1982, 78, 127–136. [Google Scholar] [CrossRef]
- Kalyanasundaram, K.; Neumann-Spallart, M. Photophysical and redox properties of water-soluble porphyrins in aqueous media. J. Chem. Phys. 1982, 86, 5163–5169. [Google Scholar] [CrossRef]
- Sreedhar, S.; Illyaskutty, N.; Sreedhanya, S.; Philip, R.; Muneera, C.I. An organic dye-polymer (phenol red-poly (vinyl alcohol)) composite architecture towards tunable-optical and -saturable absorption characteristics. J. Appl. Phys. 2016, 119, 193106. [Google Scholar] [CrossRef]
- Zhu, G.; Long, Y.; Ren, H.; Zhou, Y.; Zhang, L.; Shi, Z.; Shehzad, F.K.; Asif, H.M. Notable third-order optical nonlinearities realized in layer-by-layer assembled composite films by intercalation of porphyrin/polyoxometalate into layered double hydroxide. J. Phys. Chem. C 2016, 120, 22549–22557. [Google Scholar] [CrossRef]
- Gonçalves, P.J.; Sciuti, L.F.; Neto, N.M.B.; Silva, R.C.E.; Silveira-Alves, E., Jr.; Mendonça, C.R.; Zílio, S.C.; Borissevitch, I.E.; Boni, L.D. Effects of pH on the ultrafast transient absorption of iron (III) meso-tetrakis (4-N-methyl-pyridiniumyl) porphyrin (Fe3 + TMPyP) molecular complexes. J. Photochem. Photobiol. A 2021, 408, 113082. [Google Scholar] [CrossRef]
- Vijisha, M.V.; Ramesh, J.; Arunkumar, C.; Chandrasekharan, K. Impressive nonlinear optical responses of a cationic porphyrin derivative in a flexible all-polymer Bragg stack on optical Tamm mode coupling. J. Mater. Chem. C 2020, 8, 12689–12697. [Google Scholar] [CrossRef]
- Badran, H.A.; Abul-Hail, R.C.; Shaker, H.S.; Musa, A.I.; Hassan, Q.M.A. An all-optical switch and third-order optical nonlinearity of 3,4-pyridinediamine. Appl. Phys. B 2017, 123, 31. [Google Scholar] [CrossRef]
- Shi, Z.; Zhou, Y.; Zhang, L.; Mu, C.; Ren, H.; Hassan, D.; Yanga, D.; Asifa, H.M. New supramolecular compounds based on porphyrin and polyoxometalate: Synthesis, characterization and nonlinear optical and optical limiting properties. RSC Adv. 2014, 4, 50277–50284. [Google Scholar] [CrossRef]
- Sreekumar, G.; Frobel, P.G.L.; Muneera, C.I.; Sathiyamoorthy, K.; Vijayan, C.; Mukherjee, C. Saturable and reverse saturable absorption and nonlinear refraction in nanoclustered Amido Black dye–polymer films under low power continuous wave He–Ne laser light excitation. J. Opt. A Pure Appl. Opt. 2009, 11, 125204. [Google Scholar] [CrossRef]
- Srinivas, N.K.M.N.; Rao, S.V.; Rao, D.N. Saturable and reverse saturable absorption of Rhodamine B in methanol and water. J. Opt. Soc. Am. B 2003, 20, 2470. [Google Scholar] [CrossRef]
- Hirata, S.; Vacha, M. Large reverse saturable absorption at the sunlight power level using the ultralong lifetime of triplet excitons. J. Phys. Chem. Lett. 2017, 8, 3683–3689. [Google Scholar] [CrossRef]
- Wang, C.; Lystrom, L.; Yin, H.; Hetu, M.; Kilina, S.; McFarland, S.A.; Sun, W. Increasing the triplet lifetime and extending the ground-state absorption of biscyclometalated Ir(III) complexes for reverse saturable absorption and photodynamic therapy applications. Dalton Trans. 2016, 45, 16366–16378. [Google Scholar] [CrossRef] [PubMed]
- Blau, W.; Byrne, H.; Dennis, W.M. Reverse saturable absorption in tetraphenylporphyrins. Opt. Commun. 1985, 56, 25–29. [Google Scholar] [CrossRef]
- Wrthner, F.; Kaiser, T.E.; Saha-Mller, C.R. J-Aggregates: From serendipitous discovery to supramolecular engineering of functional dye materials. Angew. Chem. Int. Ed. 2011, 50, 3376–3410. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Misawa, K. Hierarchical structure of one-dimensional J-aggregates. J. Lumin. 1997, 72, 38–40. [Google Scholar] [CrossRef]
- Biroli, A.O.; Tessore, F.; Righetto, S.; Forni, A.; Macchioni, A.; Rocchigiani, L.; Pizzotti, M.; Carlo, G.D. Intriguing influence of −cooh-driven intermolecular aggregation and acid−base interactions with n,n-dimethylformamide on the second-order nonlinear-optical response of 5,15 push−pull diarylzinc(ii) porphyrinates. Inorg. Chem. 2017, 56, 6438–6450. [Google Scholar] [CrossRef] [PubMed]
- Ray, P.C.; Leszczynski, J. Nonlinear optical properties of highly conjugated push–pull porphyrin aggregates: Role of intermolecular interaction. Chem. Phys. Lett. 2006, 419, 578–583. [Google Scholar] [CrossRef]
- Bergendahl, L.T.; Paterson, M.J. Excited states of porphyrin and porphycene aggregates: Computation insights. Comput. Theor. Chem. 2014, 1040, 274–286. [Google Scholar] [CrossRef]
- Ogawa, T.; Tokunaga, E.; Kobayashi, T. Giant electrooptic response of excitons in porphyrin J-aggregates. Chem. Phys. Lett. 2005, 408, 186–191. [Google Scholar] [CrossRef]
- Lapes, M.; Petera, J.; Jirsa, M. Photodynamic therapy of cutaneous metastases of breast cancer after local application of meso-tetra-(para-sulphophenyl)-porphin (TPPS4). J. Photochem. Photobiol. B 1996, 36, 205–207. [Google Scholar] [CrossRef]
- Singh, S.; Aggarwal, A.; Bhupathiraju, N.V.S.; Dinesh, K.; Arianna, G.; Tiwari, K.; Drain, C.M. Glycosylated porphyrins, phthalocyanines, and other porphyrinoids for diagnostics and therapeutics. Chem. Rev. 2015, 115, 10261–10306. [Google Scholar] [CrossRef] [PubMed]
- Ethirajan, M.; Chen, Y.; Joshi, P.; Pandey, R.K. The role of porphyrin chemistry in tumor imaging and photodynamic therapy. Chem. Soc. Rev. 2011, 40, 340–362. [Google Scholar] [CrossRef] [PubMed]
- Nyman, E.S.; Hynninen, P.H. Research advances in the use of tetrapyrrolic photosensitizers for photodynamic therapy. J. Photochem. Photobiol. B 2004, 73, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Bhaumik, J.; Mittal, A.K.; Banerjee, A.; Chisti, Y.; Banerjee, U.C. Applications of phototheranostic nanoagents in photodynamic therapy. Nano Res. 2015, 8, 1373–1394. [Google Scholar] [CrossRef]
- Hou, L.; Zhang, X.; Pijper, T.C.; Browne, W.R.; Feringa, B. Reversible photochemical control of singlet oxygen generation using diarylethene photochromic switches. J. Am. Chem. Soc. 2014, 136, 910–913. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Ding, L.; Xu, H.; Shen, Z.; Ju, H.; Jia, L.; Bao, L.; Yu, J. Cell-specific and pH-activatable rubyrin-loaded nanoparticles for highly selective near-infrared photodynamic therapy against cancer. J. Am. Chem. Soc. 2013, 135, 18850–18858. [Google Scholar] [CrossRef]
- Ge, J.; Lan, M.; Zhou, B.; Liu, W.; Guo, L.; Wang, H.; Jia, Q.; Niu, G.; Huang, X.; Zhou, H.; et al. A graphene quantum dot photodynamic therapy agent with high singlet oxygen generation. Nat. Commun. 2014, 5, 4596. [Google Scholar] [CrossRef]
- Ochsner, M. Photophysical and photobiological processes in the photodynamic therapy of tumours. J. Photochem. Photobiol. B 1997, 39, 1–18. [Google Scholar] [CrossRef]
- Kalyanasundaram, K.; Gratzel, M. Light induced redox reactions of water soluble porphyrins, sensitization of hydrogen generation from water by zincporphyrin derivatives. Helv. Chim. Acta 1980, 63, 478–485. [Google Scholar] [CrossRef]
- McLendon, G.; Miller, D.S. Metalloporphyrins catalyse the photo-reduction of water to H2. J. Chem. Soc. Chem. Commun. 1980, 533–534. [Google Scholar] [CrossRef]
- Harriman, A.; Richoux, M. Photoproduction of hydrogen from reductive quenching of a water-soluble zinc porphyrin. J. Photochem. 1981, 15, 335–339. [Google Scholar] [CrossRef]
- Harriman, A.; Porter, G.; Richoux, M. Photosensitised reduction of water to hydrogen using water-soluble zinc porphyrins. J. Chem. Soc. Faraday Trans. 1981, 77, 833–844. [Google Scholar] [CrossRef]
- Okura, I.; Kim-Thuan, N. Hydrogen generation by visible light with tris-(2,2′-bipyridine) ruthenium dication. J. Mol. Catal. 1979, 5, 311–314. [Google Scholar] [CrossRef]
- Choi, M.Y.; Pollard, J.A.; Webb, M.A.; McHale, J.L. Counterion-dependent excitonic spectra of tetra(p-carboxyphenyl)porphyrin aggregates in acidic aqueous solution. J. Am. Chem. Soc. 2003, 125, 810–820. [Google Scholar] [CrossRef]
- Ishii, N.; Tokunaga, E.; Adachi, S.; Kimura, T.; Matsuda, H.; Kobayashi, T. Optical frequency- and vibrational time-resolved two-dimensional spectroscopy by real-time impulsive resonant coherent Raman scattering in polydiacetylene. Phys. Rev. A 2004, 70, 023811. [Google Scholar] [CrossRef]
- Akins, D.L.; Özçelik, S.; Zhu, H.; Guo, C. Fluorescence decay kinetics and structure of aggregated tetrakis(p-sulfonatophenyl)porphyrin. J. Phys. Chem. 1996, 100, 14390–14396. [Google Scholar] [CrossRef]
- Eaton, D.F. Reference materials for fluorescence measurement. Pure Appl. Chem. 1988, 60, 1107–1114. [Google Scholar] [CrossRef]
- Kobayashi, T. J-Aggregates, 1st ed.; World Scientific: Singapore, 1996; pp. 95–110. [Google Scholar]
- Nakata, K.; Kobayashi, T.; Tokunaga, E. Electric field-controlled dissociation and association of porphyrin J-aggregates in aqueous solution. Phys. Chem. Chem. Phys. 2011, 13, 17756–17767. [Google Scholar] [CrossRef] [PubMed]
- Kudo, T.; Ishihara, H. Proposed nonlinear resonance laser technique for manipulating nanoparticles. Phys. Rev. Lett. 2012, 109, 087402. [Google Scholar] [CrossRef]
- Tanaka, Y.; Yoshikawa, H.; Masuhara, H. Laser-induced self-assembly of pseudoisocyanine j-aggregates. J. Phys. Chem. C 2007, 111, 18457–18460. [Google Scholar] [CrossRef]
- Osborne, M.A.; Balasubramanian, S.; Furey, W.S.; Klenerman, D. Optically biased diffusion of single molecules studied by confocal fluorescence microscopy. J. Phys. Chem. B 1998, 102, 3160–3167. [Google Scholar] [CrossRef]
- Mototsuji, A.; Shoji, T.; Wakisaka, Y.; Murakoshi, K.; Yao, H.; Tsuboi, Y. Plasmonic optical trapping of nanometer-sized J-/H-dye aggregates as explored by fluorescence microspectroscopy. Opt. Express 2017, 25, 13617–13625. [Google Scholar] [CrossRef]
- Rollie, M.E.; Patonay, G.; Warner, I.M. Deoxygenation of solutions and its analytical applications. Ind. Eng. Chem. Res. 1987, 26, 1–6. [Google Scholar] [CrossRef]
- Bakalyar, S.R.; Bradley, M.P.T.; Honganen, R. The role of dissolved gases in high -performance liquid chromatography. J. Chromatogr. 1978, 158, 277–293. [Google Scholar] [CrossRef]
- Benjamin, L.; Benson, G.C. A deuterium isotope effect on the excess enthalpy of methanol-water solutions. J. Phys. Chem. 1963, 67, 858–861. [Google Scholar] [CrossRef]
- Wakisaka, A.; Abdoul-Carime, H.; Yamamoto, Y.; Kiyozumi, Y. Non-ideality of binary mixtures Water–methanol and water–acetonitrile from the viewpoint of clustering structure. J. Chem. Soc. Faraday Trans. 1998, 94, 369–374. [Google Scholar] [CrossRef]
- Nose, A.; Hojo, M.; Ueda, T. Effects of salts, acids, and phenols on the hydrogen-bonding structure of water−ethanol mixtures. J. Phys. Chem. B 2004, 108, 798–804. [Google Scholar] [CrossRef]

| [TPPS] | [NaOH] | Mole Fraction of MeOH |
|---|---|---|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Imura, S.; Kobayashi, T.; Tokunaga, E. More Than 50-Fold Enhanced Nonlinear Optical Response of Porphyrin Molecules in Aqueous Solution Induced by Mixing Base and Organic Solvent. Appl. Sci. 2021, 11, 4892. https://doi.org/10.3390/app11114892
Imura S, Kobayashi T, Tokunaga E. More Than 50-Fold Enhanced Nonlinear Optical Response of Porphyrin Molecules in Aqueous Solution Induced by Mixing Base and Organic Solvent. Applied Sciences. 2021; 11(11):4892. https://doi.org/10.3390/app11114892
Chicago/Turabian StyleImura, Satoshi, Takayoshi Kobayashi, and Eiji Tokunaga. 2021. "More Than 50-Fold Enhanced Nonlinear Optical Response of Porphyrin Molecules in Aqueous Solution Induced by Mixing Base and Organic Solvent" Applied Sciences 11, no. 11: 4892. https://doi.org/10.3390/app11114892
APA StyleImura, S., Kobayashi, T., & Tokunaga, E. (2021). More Than 50-Fold Enhanced Nonlinear Optical Response of Porphyrin Molecules in Aqueous Solution Induced by Mixing Base and Organic Solvent. Applied Sciences, 11(11), 4892. https://doi.org/10.3390/app11114892








