Why Do Tethered-Bilayer Lipid Membranes Suit for Functional Membrane Protein Reincorporation?
Abstract
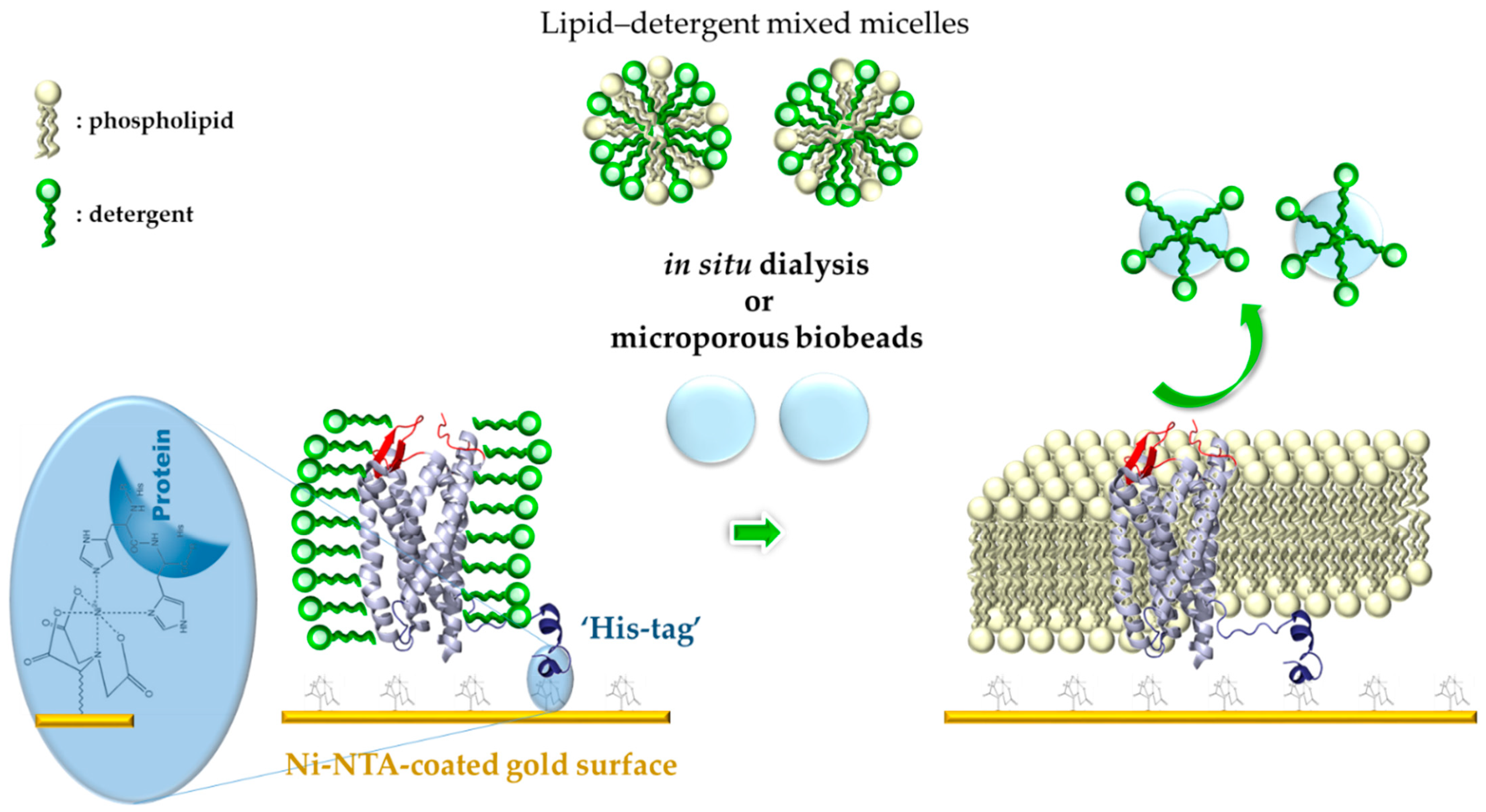
:1. Introduction
2. Design of Tethered-Bilayer Lipid Membranes
3. Characterization of Tethered-Bilayer Lipid Membranes
4. Different Types of Tethered-Bilayer Lipid Membranes
4.1. Polymer-tBLMs
4.2. Anchorlipid-tBLMs
4.3. Sparsely-tBLMs or stBLMs
4.4. Peptide-tBLMs or Pep-BLMs
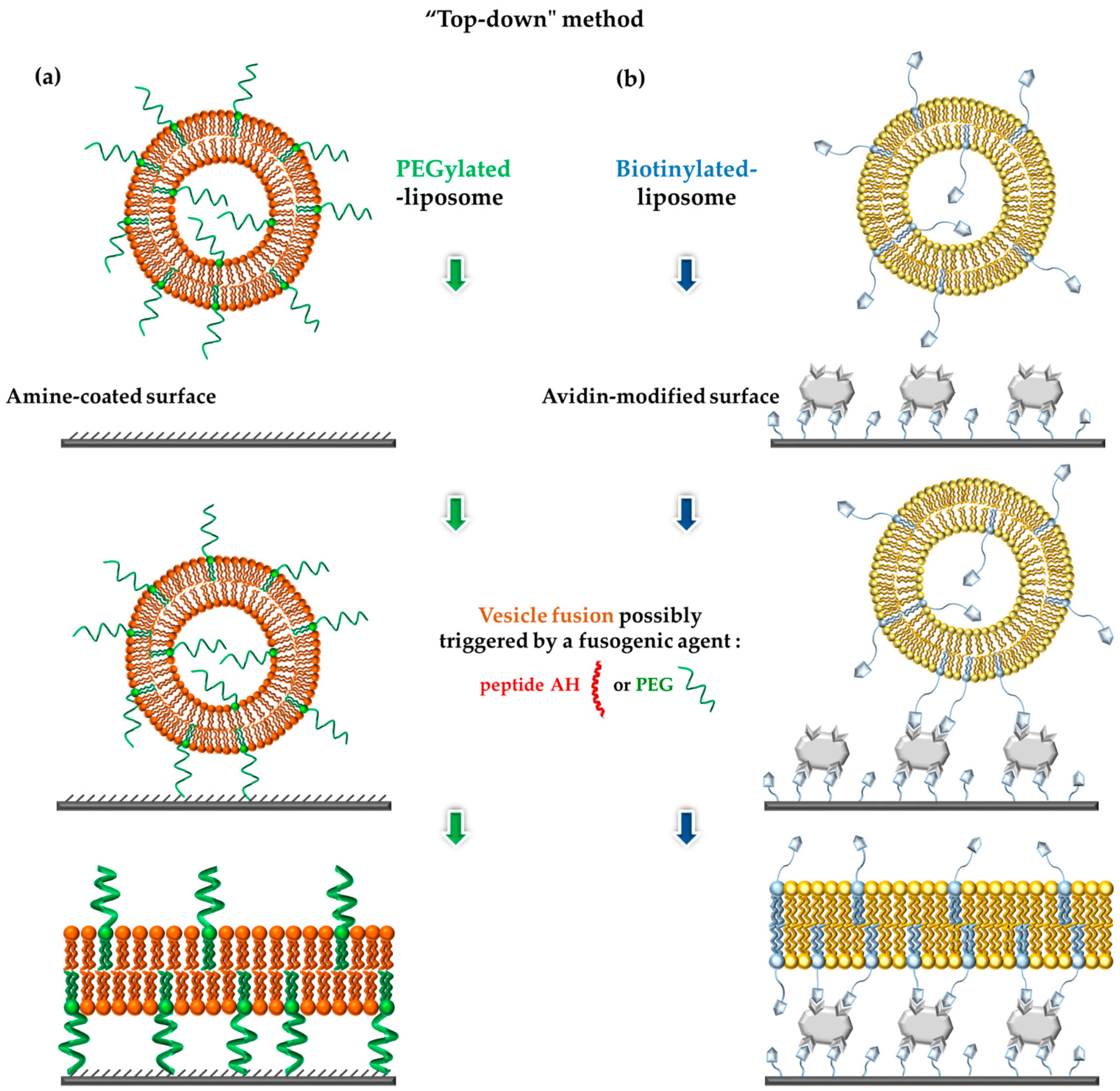
4.5. Vesicle Fusion Method to Form tBLMs: A “Top-Down” Approach
4.5.1. Using PEG as Anchoring Spacer
4.5.2. Using Biotin/Avidin as Spacers
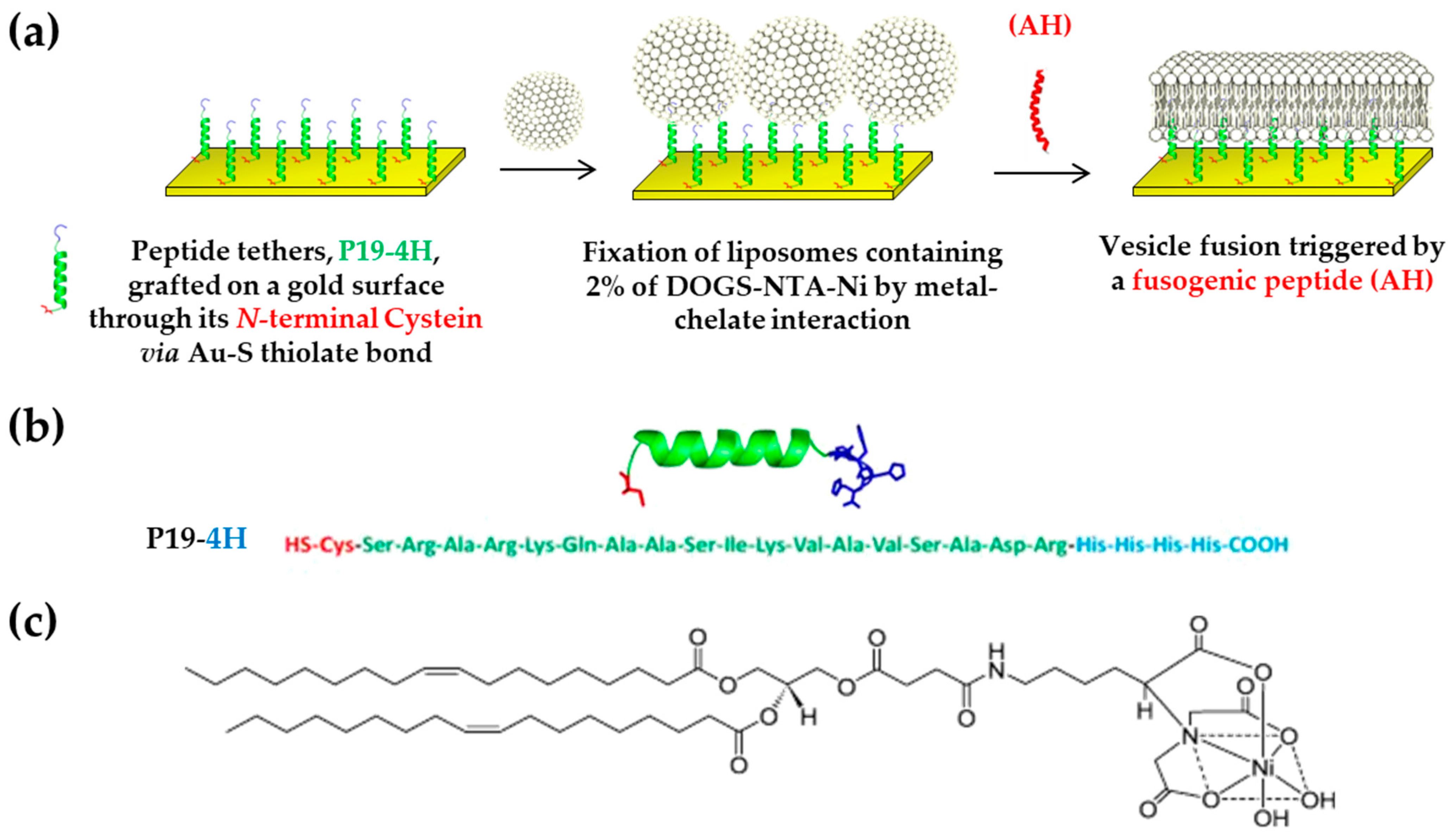
4.5.3. With Peptide as Tethers
4.6. Protein-tBLMs or ptBLMs
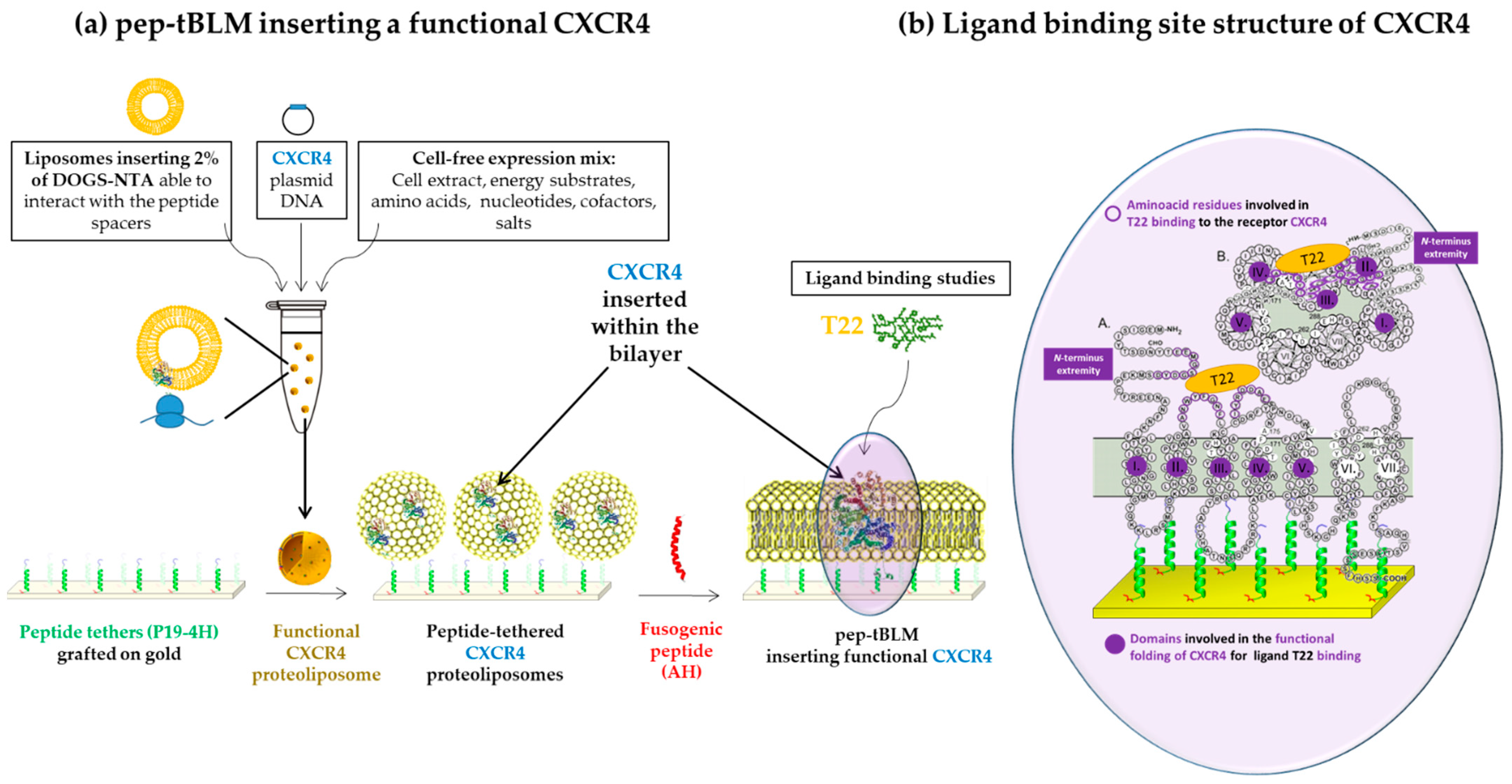
5. Cell-Free Expression for Reinsertion of Membrane Proteins before or after Formation of tBLMs
6. Perspectives: Are tBLMs Placed in the 3R System?
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Nicolson, G.L. The Fluid—Mosaic Model of Membrane Structure: Still relevant to understanding the structure, function and dynamics of biological membranes after more than 40 years. Biochim. Biophys. Acta Biomembr. 2014, 1838, 1451–1466. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, J.; Rost, B. Comparing function and structure between entire proteomes. Protein Sci. 2001, 10, 1970–1979. [Google Scholar] [CrossRef] [Green Version]
- Van Meer, G.; Voelker, D.R.; Feigenson, G.W. Membrane lipids: Where they are and how they behave. Nat. Rev. Mol. Cell Biol. 2008, 9, 112–124. [Google Scholar] [CrossRef]
- Overington, J.P.; Al-Lazikani, B.; Hopkins, A.L. How many drug targets are there? Nat. Rev. Drug Discov. 2006, 5, 993–996. [Google Scholar] [CrossRef]
- Yin, H.; Flynn, A.D. Drugging Membrane Protein Interactions. Annu. Rev. Biomed. Eng. 2016, 18, 51–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Simons, K.; Ikonen, E. Functional rafts in cell membranes. Nature 1997, 387, 569–572. [Google Scholar] [CrossRef] [PubMed]
- Lingwood, D.; Kaiser, H.-J.; Levental, I.; Simons, K. Lipid rafts as functional heterogeneity in cell membranes. Biochem. Soc. Trans. 2009, 37, 955–960. [Google Scholar] [CrossRef] [Green Version]
- Lingwood, D.; Simons, K. Lipid rafts as a membrane-organizing principle. Science 2010, 327, 46–50. [Google Scholar] [CrossRef] [Green Version]
- Dart, C. SYMPOSIUM REVIEW: Lipid microdomains and the regulation of ion channel function. J. Physiol. 2010, 588, 3169–3178. [Google Scholar] [CrossRef]
- Simons, K.; Sampaio, J.L. Membrane organization and lipid rafts. Cold Spring Harbor Perspect. Biol. 2011, 3, a004697. [Google Scholar] [CrossRef]
- Escribá, P.V. Membrane-lipid therapy: A new approach in molecular medicine. Trends Mol. Med. 2006, 12, 34–43. [Google Scholar] [CrossRef]
- Escribá, P.V.; González-Ros, J.M.; Goñi, F.M.; Kinnunen, P.K.J.; Vigh, L.; Sánchez-Magraner, L.; Fernández, A.M.; Busquets, X.; Horváth, I.; Barceló-Coblijn, G. Membranes: A meeting point for lipids, proteins and therapies. J. Cell. Mol. Med. 2008, 12, 829–875. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Escribá, P.V.; Busquets, X.; Inokuchi, J.-i.; Balogh, G.; Török, Z.; Horváth, I.; Harwood, J.L.; Vígh, L. Membrane lipid therapy: Modulation of the cell membrane composition and structure as a molecular base for drug discovery and new disease treatment. Prog. Lipid Res. 2015, 59, 38–53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Penkauskas, T.; Preta, G. Biological applications of tethered bilayer lipid membranes. Biochimie 2019, 157, 131–141. [Google Scholar] [CrossRef]
- Van den Brink-van der Laan, E.; Antoinette Killian, J.; de Kruijff, B. Nonbilayer lipids affect peripheral and integral membrane proteins via changes in the lateral pressure profile. Biochim. Biophys. Acta Biomembr. 2004, 1666, 275–288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dlouhý, O.; Kurasová, I.; Karlický, V.; Javornik, U.; Šket, P.; Petrova, N.Z.; Krumova, S.B.; Plavec, J.; Ughy, B.; Špunda, V.; et al. Modulation of non-bilayer lipid phases and the structure and functions of thylakoid membranes: Effects on the water-soluble enzyme violaxanthin de-epoxidase. Sci. Rep. 2020, 10, 11959. [Google Scholar] [CrossRef] [PubMed]
- Koldsø, H.; Sansom, M.S.P. Local Lipid Reorganization by a Transmembrane Protein Domain. J. Phys. Chem. Lett. 2012, 3, 3498–3502. [Google Scholar] [CrossRef] [Green Version]
- Tamm, L.K.; McConnell, H.M. Supported phospholipid bilayers. Biophys. J. 1985, 47, 105–113. [Google Scholar] [CrossRef] [Green Version]
- Brian, A.A.; McConnell, H.M. Allogeneic stimulation of cytotoxic T cells by supported planar membranes. Proc. Natl. Acad. Sci. USA 1984, 81, 6159–6163. [Google Scholar] [CrossRef] [Green Version]
- Wagner, M.L.; Tamm, L.K. Tethered Polymer-Supported Planar Lipid Bilayers for Reconstitution of Integral Membrane Proteins: Silane-Polyethyleneglycol-Lipid as a Cushion and Covalent Linker. Biophys. J. 2000, 79, 1400–1414. [Google Scholar] [CrossRef] [Green Version]
- Sackmann, E. Supported Membranes: Scientific and Practical Applications. Science 1996, 271, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Sackmann, E.; Tanaka, M. Supported membranes on soft polymer cushions: Fabrication, characterization and applications. Trends Biotechnol. 2000, 18, 58–64. [Google Scholar] [CrossRef]
- Silin, V.I.; Wieder, H.; Woodward, J.T.; Valincius, G.; Offenhausser, A.; Plant, A.L. The Role of Surface Free Energy on the Formation of Hybrid Bilayer Membranes. J. Am. Chem. Soc. 2002, 124, 14676–14683. [Google Scholar] [CrossRef] [Green Version]
- Terrettaz, S.; Mayer, M.; Vogel, H. Highly Electrically Insulating Tethered Lipid Bilayers for Probing the Function of Ion Channel Proteins. Langmuir 2003, 19, 5567–5569. [Google Scholar] [CrossRef]
- Cullison, J.K.; Hawkridge, F.M.; Nakashima, N.; Yoshikawa, S. A Study of Cytochrome c Oxidase in Lipid Bilayer Membranes on Electrode Surfaces. Langmuir 1994, 10, 877–882. [Google Scholar] [CrossRef]
- Ogier, S.D.; Bushby, R.J.; Cheng, Y.; Evans, S.D.; Evans, S.W.; Jenkins, A.T.A.; Knowles, P.F.; Miles, R.E. Suspended Planar Phospholipid Bilayers on Micromachined Supports. Langmuir 2000, 16, 5696–5701. [Google Scholar] [CrossRef]
- Römer, W.; Steinem, C. Impedance Analysis and Single-Channel Recordings on Nano-Black Lipid Membranes Based on Porous Alumina. Biophys. J. 2004, 86, 955–965. [Google Scholar] [CrossRef] [Green Version]
- Römer, W.; Lam, Y.H.; Fischer, D.; Watts, A.; Fischer, W.B.; Göring, P.; Wehrspohn, R.B.; Gösele, U.; Steinem, C. Channel Activity of a Viral Transmembrane Peptide in Micro-BLMs: Vpu1-32 from HIV-1. J. Am. Chem. Soc. 2004, 126, 16267–16274. [Google Scholar] [CrossRef]
- Guidelli, R.; Aloisi, G.; Becucci, L.; Dolfi, A.; Rosa Moncelli, M.; Tadini Buoninsegni, F. Bioelectrochemistry at metal/water interfaces. J. Electroanal. Chem. 2001, 504, 1–28. [Google Scholar] [CrossRef]
- Naumann, R.; Schiller, S.M.; Giess, F.; Grohe, B.; Hartman, K.B.; Kärcher, I.; Köper, I.; Lübben, J.; Vasilev, K.; Knoll, W. Tethered Lipid Bilayers on Ultraflat Gold Surfaces. Langmuir 2003, 19, 5435–5443. [Google Scholar] [CrossRef]
- Lang, H.; Duschl, C.; Vogel, H. A new class of thiolipids for the attachment of lipid bilayers on gold surfaces. Langmuir 1994, 10, 197–210. [Google Scholar] [CrossRef]
- Schiller, S.M.; Naumann, R.; Lovejoy, K.; Kunz, H.; Knoll, W. Archaea Analogue Thiolipids for Tethered Bilayer Lipid Membranes on Ultrasmooth Gold Surfaces. Angew. Chem. Int. Ed. 2003, 42, 208–211. [Google Scholar] [CrossRef]
- Förtig, A.; Jordan, R.; Graf, K.; Schiavon, G.; Purrucker, O.; Tanaka, M. Solid-supported biomimetic membranes with tailored lipopolymer tethers. Macromol. Symp. 2004, 210, 329–338. [Google Scholar] [CrossRef]
- Knoll, W.; Frank, C.W.; Heibel, C.; Naumann, R.; Offenhäusser, A.; Rühe, J.; Schmidt, E.K.; Shen, W.W.; Sinner, A. Functional tethered lipid bilayers. Rev. Mol. Biotechnol. 2000, 74, 137–158. [Google Scholar] [CrossRef]
- Rossi, C.; Homand, J.; Bauche, C.; Hamdi, H.; Ladant, D.; Chopineau, J. Differential Mechanisms for Calcium-Dependent Protein/Membrane Association as Evidenced from SPR-Binding Studies on Supported Biomimetic Membranes†. Biochemistry 2003, 42, 15273–15283. [Google Scholar] [CrossRef]
- Deniaud, A.; Rossi, C.; Berquand, A.; Homand, J.; Campagna, S.; Knoll, W.; Brenner, C.; Chopineau, J. Voltage-Dependent Anion Channel Transports Calcium Ions through Biomimetic Membranes. Langmuir 2007, 23, 3898–3905. [Google Scholar] [CrossRef] [PubMed]
- Yildiz, A.A.; Yildiz, U.H.; Liedberg, B.; Sinner, E.K. Biomimetic membrane platform: Fabrication, characterization and applications. Colloids Surf. B 2013, 103, 510–516. [Google Scholar] [CrossRef]
- Zieleniecki, J.L.; Nagarajan, Y.; Waters, S.; Rongala, J.; Thompson, V.; Hrmova, M.; Köper, I. Cell-Free Synthesis of a Functional Membrane Transporter into a Tethered Bilayer Lipid Membrane. Langmuir 2016, 32, 2445–2449. [Google Scholar] [CrossRef] [PubMed]
- Coutable, A.; Thibault, C.; Chalmeau, J.; François, J.M.; Vieu, C.; Noireaux, V.; Trévisiol, E. Preparation of Tethered-Lipid Bilayers on Gold Surfaces for the Incorporation of Integral Membrane Proteins Synthesized by Cell-Free Expression. Langmuir 2014, 30, 3132–3141. [Google Scholar] [CrossRef]
- Clifton, L.A.; Campbell, R.A.; Sebastiani, F.; Campos-Terán, J.; Gonzalez-Martinez, J.F.; Björklund, S.; Sotres, J.; Cárdenas, M. Design and use of model membranes to study biomolecular interactions using complementary surface-sensitive techniques. Adv. Colloid Interface Sci. 2020, 277, 102118. [Google Scholar] [CrossRef] [PubMed]
- A Biosensor That Uses Ion-Channel Switches. Available online: http://www.nature.com/nature/journal/v387/n6633/suppinfo/387580a0_S1.html (accessed on 26 May 2021).
- Jackman, J.; Knoll, W.; Cho, N.-J. Biotechnology Applications of Tethered Lipid Bilayer Membranes. Materials 2012, 5, 2637. [Google Scholar] [CrossRef] [Green Version]
- Chadli, M.; Maniti, O.; Marquette, C.; Tillier, B.; Cortes, S.; Girard-Egrot, A. A new functional membrane protein microarray based on tethered phospholipid bilayers. Analyst 2018, 143, 2165–2173. [Google Scholar] [CrossRef] [PubMed]
- Rebaud, S.; Maniti, O.; Girard-Egrot, A.P. Tethered bilayer lipid membranes (tBLMs): Interest and applications for biological membrane investigations. Biochimie 2014, 107 Pt A, 135–142. [Google Scholar] [CrossRef]
- Koenig, B.W.; Krueger, S.; Orts, W.J.; Majkrzak, C.F.; Berk, N.F.; Silverton, J.V.; Gawrisch, K. Neutron Reflectivity and Atomic Force Microscopy Studies of a Lipid Bilayer in Water Adsorbed to the Surface of a Silicon Single Crystal. Langmuir 1996, 12, 1343–1350. [Google Scholar] [CrossRef]
- Richter, R.P.; Bérat, R.; Brisson, A.R. Formation of Solid-Supported Lipid Bilayers: An Integrated View. Langmuir 2006, 22, 3497–3505. [Google Scholar] [CrossRef] [PubMed]
- Keller, C.A.; Glasmästar, K.; Zhdanov, V.P.; Kasemo, B. Formation of Supported Membranes from Vesicles. Phys. Rev. Lett. 2000, 84, 5443–5446. [Google Scholar] [CrossRef]
- Lind, T.K.; Cárdenas, M. Understanding the formation of supported lipid bilayers via vesicle fusion—A case that exemplifies the need for the complementary method approach (Review). Biointerphases 2016, 11, 020801. [Google Scholar] [CrossRef] [Green Version]
- Johnson, S.J.; Bayerl, T.M.; McDermott, D.C.; Adam, G.W.; Rennie, A.R.; Thomas, R.K.; Sackmann, E. Structure of an adsorbed dimyristoylphosphatidylcholine bilayer measured with specular reflection of neutrons. Biophys. J. 1991, 59, 289–294. [Google Scholar] [CrossRef] [Green Version]
- Tero, R. Substrate Effects on the Formation Process, Structure and Physicochemical Properties of Supported Lipid Bilayers. Materials 2012, 5, 2658–2680. [Google Scholar] [CrossRef]
- Andersson, J.; Köper, I. Tethered and Polymer Supported Bilayer Lipid Membranes: Structure and Function. Membranes 2016, 6, 30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Groves, J.T.; Dustin, M.L. Supported planar bilayers in studies on immune cell adhesion and communication. J. Immunol. Methods 2003, 278, 19–32. [Google Scholar] [CrossRef]
- Reviakine, I.; Brisson, A. Streptavidin 2D Crystals on Supported Phospholipid Bilayers: Toward Constructing Anchored Phospholipid Bilayers. Langmuir 2001, 17, 8293–8299. [Google Scholar] [CrossRef]
- Milhiet, P.-E.; Giocondi, M.-C.; Baghdadi, O.; Ronzon, F.; Roux, B.; Le Grimellec, C. Spontaneous insertion and partitioning of alkaline phosphatase into model lipid rafts. EMBO Rep. 2002, 3, 485–490. [Google Scholar] [CrossRef] [Green Version]
- Bouter, A.; Gounou, C.; Bérat, R.; Tan, S.; Gallois, B.; Granier, T.; d’Estaintot, B.L.; Pöschl, E.; Brachvogel, B.; Brisson, A.R. Annexin-A5 assembled into two-dimensional arrays promotes cell membrane repair. Nat. Commun. 2011, 2, 270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heath, G.R.; Scheuring, S. High-speed AFM height spectroscopy reveals µs-dynamics of unlabeled biomolecules. Nat. Commun. 2018, 9, 4983. [Google Scholar] [CrossRef]
- Melby, E.S.; Mensch, A.C.; Lohse, S.E.; Hu, D.; Orr, G.; Murphy, C.J.; Hamers, R.J.; Pedersen, J.A. Formation of supported lipid bilayers containing phase-segregated domains and their interaction with gold nanoparticles. Environ. Sci. Nano 2016, 3, 45–55. [Google Scholar] [CrossRef] [Green Version]
- Waldie, S.; Lind, T.K.; Browning, K.; Moulin, M.; Haertlein, M.; Forsyth, V.T.; Luchini, A.; Strohmeier, G.A.; Pichler, H.; Maric, S.; et al. Localization of Cholesterol within Supported Lipid Bilayers Made of a Natural Extract of Tailor-Deuterated Phosphatidylcholine. Langmuir 2018, 34, 472–479. [Google Scholar] [CrossRef]
- Waldie, S.; Moulin, M.; Porcar, L.; Pichler, H.; Strohmeier, G.A.; Skoda, M.; Forsyth, V.T.; Haertlein, M.; Maric, S.; Cárdenas, M. The Production of Matchout-Deuterated Cholesterol and the Study of Bilayer-Cholesterol Interactions. Sci. Rep. 2019, 9, 5118. [Google Scholar] [CrossRef]
- Shilts, K.; Naumann, C.A. Tunable cell-surface mimetics as engineered cell substrates. Biochim. Biophys. Acta Biomembr. 2018, 1860, 2076–2093. [Google Scholar] [CrossRef]
- Yu, C.-h.; Groves, J. Engineering supported membranes for cell biology. Med. Biol. Eng. Comput. 2010, 48, 955–963. [Google Scholar] [CrossRef] [Green Version]
- Hartman, N.C.; Nye, J.A.; Groves, J.T. Cluster size regulates protein sorting in the immunological synapse. Proc. Natl. Acad. Sci. USA 2009, 106, 12729–12734. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torres, A.J.; Contento, R.L.; Gordo, S.; Wucherpfennig, K.W.; Love, J.C. Functional single-cell analysis of T-cell activation by supported lipid bilayer-tethered ligands on arrays of nanowells. Lab. Chip 2013, 13, 90–99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Groves, J.T.; Parthasarathy, R.; Forstner, M.B. Fluorescence Imaging of Membrane Dynamics. Annu. Rev. Biomed. Eng. 2008, 10, 311–338. [Google Scholar] [CrossRef]
- Loose, M.; Schwille, P. Biomimetic membrane systems to study cellular organization. J. Struct. Biol. 2009, 168, 143–151. [Google Scholar] [CrossRef]
- Cho, N.-J.; Frank, C.W.; Kasemo, B.; Hook, F. Quartz crystal microbalance with dissipation monitoring of supported lipid bilayers on various substrates. Nat. Protoc. 2010, 5, 1096–1106. [Google Scholar] [CrossRef]
- Przybylo, M.; Sýkora, J.; Humpolíčková, J.; Benda, A.; Zan, A.; Hof, M. Lipid Diffusion in Giant Unilamellar Vesicles Is More than 2 Times Faster than in Supported Phospholipid Bilayers under Identical Conditions. Langmuir 2006, 22, 9096–9099. [Google Scholar] [CrossRef]
- Macháň, R.; Hof, M. Lipid diffusion in planar membranes investigated by fluorescence correlation spectroscopy. Biochim. Biophys. Acta Biomembr. 2010, 1798, 1377–1391. [Google Scholar] [CrossRef] [Green Version]
- Wu, H.-L.; Tong, Y.; Peng, Q.; Li, N.; Ye, S. Phase transition behaviors of the supported DPPC bilayer investigated by sum frequency generation (SFG) vibrational spectroscopy and atomic force microscopy (AFM). Phys. Chem. Chem. Phys. 2016, 18, 1411–1421. [Google Scholar] [CrossRef] [Green Version]
- Sondhi, P.; Lingden, D.; Stine, K.J. Structure, Formation, and Biological Interactions of Supported Lipid Bilayers (SLB) Incorporating Lipopolysaccharide. Coatings 2020, 10, 981. [Google Scholar] [CrossRef]
- Sinner, E.-K.; Ritz, S.; Naumann, R.; Schiller, S.; Knoll, W. Self-Assembled Tethered Bimolecular Lipid Membranes. In Advances in Clinical Chemistry; Gregory, S.M., Ed.; Elsevier: Amsterdam, The Netherlands, 2009; Volume 49, pp. 159–179. [Google Scholar]
- Köper, I.; Schiller, S.M.; Giess, F.; Naumann, R.; Knoll, W. Functional Tethered Bimolecular Lipid Membranes (tBLMs). In Advances in Planar Lipid Bilayers and Liposomes; Liu, A.L., Ed.; Academic Press: Cambridge, MA, USA, 2006; Volume 3, pp. 37–53. [Google Scholar]
- Tanaka, M.; Rosseti, F.F.; Kaufmann, S. Native supported membranes: Creation of two-dimensional cell membranes on polymer supports (Review). Biointerphases 2008, 3, FA12–FA16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lind, T.K.; Wacklin, H.; Schiller, J.; Moulin, M.; Haertlein, M.; Pomorski, T.G.; Cárdenas, M. Formation and Characterization of Supported Lipid Bilayers Composed of Hydrogenated and Deuterated Escherichia coli Lipids. PLoS ONE 2015, 10, e0144671. [Google Scholar] [CrossRef]
- Reimhult, E.; Kumar, K. Membrane biosensor platforms using nano- and microporous supports. Trends Biotechnol. 2008, 26, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Knoll, W.; Köper, I.; Naumann, R.; Sinner, E.-K. Tethered bimolecular lipid membranes—A novel model membrane platform. Electrochim. Acta 2008, 53, 6680–6689. [Google Scholar] [CrossRef]
- Junghans, A.; Köper, I. Structural Analysis of Tethered Bilayer Lipid Membranes. Langmuir 2010, 26, 11035–11040. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Faller, R. Conformational, Dynamical. and Tensional Study of Tethered Bilayer Lipid Membranes in Coarse-Grained Molecular Simulations. Langmuir 2012, 28, 15907–15915. [Google Scholar] [CrossRef] [PubMed]
- Montal, M.; Mueller, P. Formation of Bimolecular Membranes from Lipid Monolayers and a Study of Their Electrical Properties. Proc. Natl. Acad. Sci. USA 1972, 69, 3561–3566. [Google Scholar] [CrossRef] [Green Version]
- Tien, H.T.; Ottova, A.L. The lipid bilayer concept and its experimental realization: From soap bubbles, kitchen sink, to bilayer lipid membranes. J. Membr. Sci. 2001, 189, 83–117. [Google Scholar] [CrossRef]
- Berquand, A.; Mazeran, P.-E.; Pantigny, J.; Proux-Delrouyre, V.; Laval, J.-M.; Bourdillon, C. Two-Step Formation of Streptavidin-Supported Lipid Bilayers by PEG-Triggered Vesicle Fusion. Fluorescence and Atomic Force Microscopy Characterization†. Langmuir 2003, 19, 1700–1707. [Google Scholar] [CrossRef]
- Jeuken, L.J.C.; Daskalakis, N.N.; Han, X.; Sheikh, K.; Erbe, A.; Bushby, R.J.; Evans, S.D. Phase separation in mixed self-assembled monolayers and its effect on biomimetic membranes. Sens. Actuators B 2007, 124, 501–509. [Google Scholar] [CrossRef]
- Lee, B.K.; Lee, H.Y.; Kim, P.; Suh, K.Y.; Kawai, T. Nanoarrays of tethered lipid bilayer rafts on poly(vinyl alcohol) hydrogels. Lab. Chip 2009, 9, 132–139. [Google Scholar] [CrossRef] [Green Version]
- Vockenroth, I.K.; Rossi, C.; Shah, M.R.; Köper, I. Formation of tethered bilayer lipid membranes probed by various surface sensitive techniques. Biointerphases 2009, 4, 19–26. [Google Scholar] [CrossRef] [Green Version]
- Basit, H.; Van der Heyden, A.; Gondran, C.; Nysten, B.; Dumy, P.; Labbé, P. Tethered Bilayer Lipid Membranes on Mixed Self-Assembled Monolayers of a Novel Anchoring Thiol: Impact of the Anchoring Thiol Density on Bilayer Formation. Langmuir 2011, 27, 14317–14328. [Google Scholar] [CrossRef] [PubMed]
- Bronder, A.M.; Bieker, A.; Elter, S.; Etzkorn, M.; Häussinger, D.; Oesterhelt, F. Oriented Membrane Protein Reconstitution into Tethered Lipid Membranes for AFM Force Spectroscopy. Biophys. J. 2016, 111, 1925–1934. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, W.; Burke, P.J. Versatile Bottom-Up Synthesis of Tethered Bilayer Lipid Membranes on Nanoelectronic Biosensor Devices. ACS Appl. Mater. Interfaces 2017, 9, 14618–14632. [Google Scholar] [CrossRef]
- Shenoy, S.; Moldovan, R.; Fitzpatrick, J.; Vanderah, D.J.; Deserno, M.; Lösche, M. In-plane homogeneity and lipid dynamics in tethered bilayer lipid membranes (tBLMs). Soft Matter 2010, 6, 1263–1274. [Google Scholar] [CrossRef]
- Eicher-Lorka, O.; Charkova, T.; Matijoška, A.; Kuodis, Z.; Urbelis, G.; Penkauskas, T.; Mickevičius, M.; Bulovas, A.; Valinčius, G. Cholesterol-based tethers and markers for model membranes investigation. Chem. Phys. Lipids 2016, 195, 71–86. [Google Scholar] [CrossRef]
- Naumann, C.A.; Prucker, O.; Lehmann, T.; Rühe, J.; Knoll, W.; Frank, C.W. The Polymer-Supported Phospholipid Bilayer: Tethering as a New Approach to Substrate−Membrane Stabilization. Biomacromolecules 2002, 3, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Munro, J.C.; Frank, C.W. In Situ Formation and Characterization of Poly(ethylene glycol)-Supported Lipid Bilayers on Gold Surfaces. Langmuir 2004, 20, 10567–10575. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.-Y.; Chen, W.-L.; Ober, C.K.; Daniel, S. Biologically Complex Planar Cell Plasma Membranes Supported on Polyelectrolyte Cushions Enhance Transmembrane Protein Mobility and Retain Native Orientation. Langmuir 2018, 34, 1061–1072. [Google Scholar] [CrossRef]
- Roder, F.; Waichman, S.; Paterok, D.; Schubert, R.; Richter, C.; Liedberg, B.; Piehler, J. Reconstitution of Membrane Proteins into Polymer-Supported Membranes for Probing Diffusion and Interactions by Single Molecule Techniques. Anal. Chem. 2011, 83, 6792–6799. [Google Scholar] [CrossRef]
- McGillivray, D.J.; Valincius, G.; Vanderah, D.J.; Febo-Ayala, W.; Woodward, J.T.; Heinrich, F.; Kasianowicz, J.J.; Lösche, M. Molecular-scale structural and functional characterization of sparsely tethered bilayer lipid membranes. Biointerphases 2007, 2, 21–33. [Google Scholar] [CrossRef] [Green Version]
- Vockenroth, I.K.; Ohm, C.; Robertson, J.W.F.; McGillivraya, D.J.; Lösche, M.; Köper, I. Stable insulating tethered bilayer lipid membranes. Biointerphases 2008, 3, FA68–FA73. [Google Scholar] [CrossRef] [Green Version]
- Budvytyte, R.; Valincius, G.; Niaura, G.; Voiciuk, V.; Mickevicius, M.; Chapman, H.; Goh, H.-Z.; Shekhar, P.; Heinrich, F.; Shenoy, S.; et al. Structure and Properties of Tethered Bilayer Lipid Membranes with Unsaturated Anchor Molecules. Langmuir 2013, 29, 8645–8656. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hertrich, S.; Stetter, F.; Rühm, A.; Hugel, T.; Nickel, B. Highly Hydrated Deformable Polyethylene Glycol-Tethered Lipid Bilayers. Langmuir 2014, 30, 9442–9447. [Google Scholar] [CrossRef] [PubMed]
- Yap, T.L.; Jiang, Z.; Heinrich, F.; Gruschus, J.M.; Pfefferkorn, C.M.; Barros, M.; Curtis, J.E.; Sidransky, E.; Lee, J.C. Structural features of membrane-bound glucocerebrosidase and α-synuclein probed by neutron reflectometry and fluorescence spectroscopy. J. Biol. Chem. 2015, 290, 744–754. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maccarini, M.; Watkins, E.B.; Stidder, B.; Alcaraz, J.-P.; Cornell, B.A.; Martin, D.K. Nanostructural determination of a lipid bilayer tethered to a gold substrate. Eur. Phys. J. E 2016, 39, 123. [Google Scholar] [CrossRef] [PubMed]
- Cranfield, C.G.; Berry, T.; Holt, S.A.; Hossain, K.R.; Le Brun, A.P.; Carne, S.; Al Khamici, H.; Coster, H.; Valenzuela, S.M.; Cornell, B. Evidence of the Key Role of H3O+ in Phospholipid Membrane Morphology. Langmuir 2016, 32, 10725–10734. [Google Scholar] [CrossRef]
- Andersson, J.; Knobloch, J.J.; Perkins, M.V.; Holt, S.A.; Köper, I. Synthesis and Characterization of Novel Anchorlipids for Tethered Bilayer Lipid Membranes. Langmuir 2017, 33, 4444–4451. [Google Scholar] [CrossRef]
- Alharbi, A.R.M.; Andersson, J.M.; Köper, I.; Andersson, G.G. Investigating the Structure of Self-Assembled Monolayers Related to Biological Cell Membranes. Langmuir 2019, 35, 14213–14221. [Google Scholar] [CrossRef]
- Squillace, O.; Perrault, T.; Gorczynska, M.; Caruana, A.; Bajorek, A.; Brotons, G. Design of tethered bilayer lipid membranes, using wet chemistry via aryldiazonium sulfonic acid spontaneous grafting on silicon and chrome. Colloids Surf. B 2021, 197, 111427. [Google Scholar] [CrossRef]
- Atanasov, V.; Knorr, N.; Duran, R.S.; Ingebrandt, S.; Offenhäusser, A.; Knoll, W.; Köper, I. Membrane on a Chip: A Functional Tethered Lipid Bilayer Membrane on Silicon Oxide Surfaces. Biophys. J. 2005, 89, 1780–1788. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leitch, J.; Kunze, J.; Goddard, J.D.; Schwan, A.L.; Faragher, R.J.; Naumann, R.; Knoll, W.; Dutcher, J.R.; Lipkowski, J. In Situ PM-IRRAS Studies of an Archaea Analogue Thiolipid Assembled on a Au(111) Electrode Surface. Langmuir 2009, 25, 10354–10363. [Google Scholar] [CrossRef]
- Su, Z.; Ran, X.; Leitch, J.J.; Schwan, A.L.; Faragher, R.; Lipkowski, J. How Valinomycin Ionophores Enter and Transport K+ across Model Lipid Bilayer Membranes. Langmuir 2019, 35, 16935–16943. [Google Scholar] [CrossRef] [PubMed]
- Ataka, K.; Giess, F.; Knoll, W.; Naumann, R.; Haber-Pohlmeier, S.; Richter, B.; Heberle, J. Oriented Attachment and Membrane Reconstitution of His-Tagged Cytochrome c Oxidase to a Gold Electrode: In Situ Monitoring by Surface-Enhanced Infrared Absorption Spectroscopy. J. Am. Chem. Soc. 2004, 126, 16199–16206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kozuch, J.; Weichbrodt, C.; Millo, D.; Giller, K.; Becker, S.; Hildebrandt, P.; Steinem, C. Voltage-dependent structural changes of the membrane-bound anion channel hVDAC1 probed by SEIRA and electrochemical impedance spectroscopy. Phys. Chem. Chem. Phys. 2014, 16, 9546–9555. [Google Scholar] [CrossRef] [Green Version]
- Wiebalck, S.; Kozuch, J.; Forbrig, E.; Tzschucke, C.C.; Jeuken, L.J.C.; Hildebrandt, P. Monitoring the Transmembrane Proton Gradient Generated by Cytochrome bo3 in Tethered Bilayer Lipid Membranes Using SEIRA Spectroscopy. J. Phys. Chem. B 2016, 120, 2249–2256. [Google Scholar] [CrossRef]
- Forbrig, E.; Staffa, J.K.; Salewski, J.; Mroginski, M.A.; Hildebrandt, P.; Kozuch, J. Monitoring the Orientational Changes of Alamethicin during Incorporation into Bilayer Lipid Membranes. Langmuir 2018, 34, 2373–2385. [Google Scholar] [CrossRef]
- Schmidt, E.K.; Liebermann, T.; Kreiter, M.; Jonczyk, A.; Naumann, R.; Offenhäusser, A.; Neumann, E.; Kukol, A.; Maelicke, A.; Knoll, W. Incorporation of the acetylcholine receptor dimer from Torpedo californica in a peptide supported lipid membrane investigated by surface plasmon and fluorescence spectroscopy. Biosens. Bioelectron. 1998, 13, 585–591. [Google Scholar] [CrossRef]
- Giess, F.; Friedrich, M.G.; Heberle, J.; Naumann, R.L.; Knoll, W. The Protein-Tethered Lipid Bilayer: A Novel Mimic of the Biological Membrane. Biophys. J. 2004, 87, 3213–3220. [Google Scholar] [CrossRef] [Green Version]
- Wiltschi, B.; Knoll, W.; Sinner, E.-K. Binding assays with artificial tethered membranes using surface plasmon resonance. Methods 2006, 39, 134–146. [Google Scholar] [CrossRef]
- Becucci, L.; Moncelli, M.R.; Naumann, R.; Guidelli, R. Potassium Ion Transport by Valinomycin across a Hg-Supported Lipid Bilayer. J. Am. Chem. Soc. 2005, 127, 13316–13323. [Google Scholar] [CrossRef] [PubMed]
- Naumann, R.; Baumgart, T.; Gräber, P.; Jonczyk, A.; Offenhäusser, A.; Knoll, W. Proton transport through a peptide-tethered bilayer lipid membrane by the H+−ATP synthase from chloroplasts measured by impedance spectroscopy. Biosens. Bioelectron. 2002, 17, 25–34. [Google Scholar] [CrossRef]
- Krishna, G.; Schulte, J.; Cornell, B.A.; Pace, R.J.; Osman, P.D. Tethered Bilayer Membranes Containing Ionic Reservoirs: Selectivity and Conductance. Langmuir 2003, 19, 2294–2305. [Google Scholar] [CrossRef]
- Cranfield, C.G.; Bettler, T.; Cornell, B. Nanoscale Ion Sequestration To Determine the Polarity Selectivity of Ion Conductance in Carriers and Channels. Langmuir 2015, 31, 292–298. [Google Scholar] [CrossRef]
- Proux-Delrouyre, V.; Elie, C.; Laval, J.-M.; Moiroux, J.; Bourdillon, C. Formation of Tethered and Streptavidin-Supported Lipid Bilayers on a Microporous Electrode for the Reconstitution of Membranes of Large Surface Area. Langmuir 2002, 18, 3263–3272. [Google Scholar] [CrossRef]
- Jeuken, L.J.C.; Connell, S.D.; Henderson, P.J.F.; Gennis, R.B.; Evans, S.D.; Bushby, R.J. Redox Enzymes in Tethered Membranes. J. Am. Chem. Soc. 2006, 128, 1711–1716. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Friedrich, M.G.; Robertson, J.W.F.; Walz, D.; Knoll, W.; Naumann, R.L.C. Electronic Wiring of a Multi-Redox Site Membrane Protein in a Biomimetic Surface Architecture. Biophys. J. 2008, 94, 3698–3705. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeuken, L.J.C. Electrodes for integral membrane enzymes. Nat. Prod. Rep. 2009, 26, 1234–1240. [Google Scholar] [CrossRef] [PubMed]
- Nowak, C.; Schach, D.; Gebert, J.; Grosserueschkamp, M.; Gennis, R.B.; Ferguson-Miller, S.; Knoll, W.; Walz, D.; Naumann, R.L.C. Oriented immobilization and electron transfer to the cytochrome c oxidase. J. Solid State Electrochem. 2011, 15, 105–114. [Google Scholar] [CrossRef] [Green Version]
- Becucci, L.; Guidelli, R. Can gramicidin ion channel affect the dipole potential of neighboring phospholipid headgroups? Bioelectrochemistry 2015, 106, 343–352. [Google Scholar] [CrossRef] [PubMed]
- McGillivray, D.J.; Valincius, G.; Heinrich, F.; Robertson, J.W.F.; Vanderah, D.J.; Febo-Ayala, W.; Ignatjev, I.; Lösche, M.; Kasianowicz, J.J. Structure of Functional Staphylococcus aureus α-Hemolysin Channels in Tethered Bilayer Lipid Membranes. Biophys. J. 2009, 96, 1547–1553. [Google Scholar] [CrossRef] [Green Version]
- Andersson, J.; Köper, I.; Knoll, W. Tethered Membrane Architectures—Design and Applications. Front. Mater. 2018, 5. [Google Scholar] [CrossRef] [Green Version]
- Rossi, C.; Chopineau, J. Biomimetic tethered lipid membranes designed for membrane-protein interaction studies. Eur. Biophys. J. 2007, 36, 955–965. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Sackmann, E. Polymer-supported membranes as models of the cell surface. Nature 2005, 437, 656–663. [Google Scholar] [CrossRef]
- Knoll, W.; Bender, K.; Förch, R.; Frank, C.; Götz, H.; Heibel, C.; Jenkins, T.; Jonas, U.; Kibrom, A.; Kügler, R.; et al. Polymer-Tethered Bimolecular Lipid Membranes. In Polymer Membranes/Biomembranes; Meier, W.P., Knoll, W., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 87–111. [Google Scholar]
- Veneziano, R.; Rossi, C.; Chenal, A.; Devoisselle, J.-M.; Ladant, D.; Chopineau, J. Bordetella pertussis adenylate cyclase toxin translocation across a tethered lipid bilayer. Proc. Natl. Acad. Sci. USA 2013, 110, 20473–20478. [Google Scholar] [CrossRef] [Green Version]
- Rossi, C.; Doumiati, S.; Lazzarelli, C.; Davi, M.; Meddar, F.; Ladant, D.; Chopineau, J. A Tethered Bilayer Assembled on Top of Immobilized Calmodulin to Mimic Cellular Compartmentalization. PLoS ONE 2011, 6, e19101. [Google Scholar] [CrossRef]
- Naumann, R.; Schmidt, E.K.; Jonczyk, A.; Fendler, K.; Kadenbach, B.; Liebermann, T.; Offenhäusser, A.; Knoll, W. The peptide-tethered lipid membrane as a biomimetic system to incorporate cytochrome c oxidase in a functionally active form. Biosens. Bioelectron. 1999, 14, 651–662. [Google Scholar] [CrossRef]
- Naumann, R.L.C.; Knoll, W. Protein tethered lipid bilayer: An alternative mimic of the biological membrane (Mini Review). Biointerphases 2008, 3, FA101–FA107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Naumann, R.L.C.; Geiss, A.F.; Steininger, C.; Knoll, W. Biomimetic Membranes for Multi-Redox Center Proteins. Int. J. Mol. Sci. 2016, 17, 330. [Google Scholar] [CrossRef] [Green Version]
- Elie-Caille, C.; Fliniaux, O.; Pantigny, J.; Mazière, J.-C.; Bourdillon, C. Self-Assembly of Solid-Supported Membranes Using a Triggered Fusion of Phospholipid-Enriched Proteoliposomes Prepared from the Inner Mitochondrial Membrane1. Langmuir 2005, 21, 4661–4668. [Google Scholar] [CrossRef] [Green Version]
- Su, Z.; Jiang, Y.; Velázquez-Manzanares, M.; Jay Leitch, J.; Kycia, A.; Lipkowski, J. Electrochemical and PM-IRRAS studies of floating lipid bilayers assembled at the Au(111) electrode pre-modified with a hydrophilic monolayer. J. Electroanal. Chem. 2013, 688, 76–85. [Google Scholar] [CrossRef]
- Deverall, M.A.; Gindl, E.; Sinner, E.K.; Besir, H.; Ruehe, J.; Saxton, M.J.; Naumann, C.A. Membrane Lateral Mobility Obstructed by Polymer-Tethered Lipids Studied at the Single Molecule Level. Biophys. J. 2005, 88, 1875–1886. [Google Scholar] [CrossRef] [Green Version]
- Maynard, J.A.; Lindquist, N.C.; Sutherland, J.N.; Lesuffleur, A.; Warrington, A.E.; Rodriguez, M.; Oh, S.-H. Surface plasmon resonance for high-throughput ligand screening of membrane-bound proteins. Biotechnol. J. 2009, 4, 1542–1558. [Google Scholar] [CrossRef] [Green Version]
- Wagner, M.L.; Tamm, L.K. Reconstituted Syntaxin1A/SNAP25 Interacts with Negatively Charged Lipids as Measured by Lateral Diffusion in Planar Supported Bilayers. Biophys. J. 2001, 81, 266–275. [Google Scholar] [CrossRef] [Green Version]
- Kiessling, V.; Tamm, L.K. Measuring Distances in Supported Bilayers by Fluorescence Interference-Contrast Microscopy: Polymer Supports and SNARE Proteins. Biophys. J. 2003, 84, 408–418. [Google Scholar] [CrossRef] [Green Version]
- Purrucker, O.; Gönnenwein, S.; Förtig, A.; Jordan, R.; Rusp, M.; Bärmann, M.; Moroder, L.; Sackmann, E.; Tanaka, M. Polymer-tethered membranes as quantitative models for the study of integrin-mediated cell adhesion. Soft Matter 2007, 3, 333–336. [Google Scholar] [CrossRef]
- Inci, F.; Celik, U.; Turken, B.; Özer, H.Ö.; Kok, F.N. Construction of P-glycoprotein incorporated tethered lipid bilayer membranes. Biochem. Biophys. Rep. 2015, 2, 115–122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schiller, S.M.; Reisinger-Friebis, A.; Götz, H.; Hawker, C.J.; Frank, C.W.; Naumann, R.; Knoll, W. Biomimetic Lipoglycopolymer Membranes: Photochemical Surface Attachment of Supramolecular Architectures with Defined Orientation. Angew. Chem. Int. Ed. 2009, 48, 6896–6899. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Lin, Y.H.; Lautscham, L.A.; Goldmann, W.H.; Fabry, B.; Naumann, C.A. N-cadherin-functionalized polymer-tethered multi-bilayer: A cell surface-mimicking substrate to probe cellular mechanosensitivity. Soft Matter 2016, 12, 8274–8284. [Google Scholar] [CrossRef] [Green Version]
- Atanasov, V.; Atanasova, P.P.; Vockenroth, I.K.; Knorr, N.; Köper, I. A Molecular Toolkit for Highly Insulating Tethered Bilayer Lipid Membranes on Various Substrates. Bioconjug. Chem. 2006, 17, 631–637. [Google Scholar] [CrossRef]
- Kunze, J.; Leitch, J.; Schwan, A.L.; Faragher, R.J.; Naumann, R.; Schiller, S.; Knoll, W.; Dutcher, J.R.; Lipkowski, J. New Method to Measure Packing Densities of Self-Assembled Thiolipid Monolayers. Langmuir 2006, 22, 5509–5519. [Google Scholar] [CrossRef] [PubMed]
- Becucci, L.; Faragher, R.J.; Schwan, A. The effect of the hydrophilic spacer length on the functionality of a mercury-supported tethered bilayer lipid membrane. Bioelectrochemistry 2015, 101, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Becucci, L.; Aloisi, G.; Scaloni, A.; Caira, S.; Guidelli, R. On the interaction of the highly charged peptides casocidins with biomimetic membranes. Bioelectrochemistry 2018, 123, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Becucci, L.; Guidelli, R. What Ion Flow along Ion Channels Can Tell us about Their Functional Activity. Membranes 2016, 6, 53. [Google Scholar] [CrossRef] [Green Version]
- Becucci, L.; Guidelli, R. Mercury-supported biomimetic membranes for the investigation of antimicrobial peptides. Pharmaceuticals 2014, 7, 136–168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niu, L.; Wohland, T.; Knoll, W.; Köper, I. Interaction of a synthetic antimicrobial peptide with a model bilayer platform mimicking bacterial membranes. Biointerphases 2017, 12, 04E404. [Google Scholar] [CrossRef] [PubMed]
- Andersson, J.; Fuller, M.A.; Wood, K.; Holt, S.A.; Köper, I. A tethered bilayer lipid membrane that mimics microbial membranes. Phys. Chem. Chem. Phys. 2018, 20, 12958–12969. [Google Scholar] [CrossRef]
- Silin, V.; Kasianowicz, J.J.; Michelman-Ribeiro, A.; Panchal, R.G.; Bavari, S.; Robertson, J.W. Biochip for the Detection of Bacillus anthracis Lethal Factor and Therapeutic Agents against Anthrax Toxins. Membranes 2016, 6, 36. [Google Scholar] [CrossRef] [Green Version]
- Budvytyte, R.; Pleckaityte, M.; Zvirbliene, A.; Vanderah, D.J.; Valincius, G. Reconstitution of cholesterol-dependent vaginolysin into tethered phospholipid bilayers: Implications for bioanalysis. PLoS ONE 2013, 8, e82536. [Google Scholar] [CrossRef]
- Heinrich, F.; Ng, T.; Vanderah, D.J.; Shekhar, P.; Mihailescu, M.; Nanda, H.; Lösche, M. A New Lipid Anchor for Sparsely Tethered Bilayer Lipid Membranes. Langmuir 2009, 25, 4219–4229. [Google Scholar] [CrossRef] [PubMed]
- Boden, N.; Bushby, R.J.; Clarkson, S.; Evans, S.D.; Knowles, P.F.; Marsh, A. The design and synthesis of simple molecular tethers for binding biomembranes to a gold surface. Tetrahedron 1997, 53, 10939–10952. [Google Scholar] [CrossRef]
- Jenkins, A.T.A.; Boden, N.; Bushby, R.J.; Evans, S.D.; Knowles, P.F.; Miles, R.E.; Ogier, S.D.; Schönherr, H.; Vancso, G.J. Microcontact Printing of Lipophilic Self-Assembled Monolayers for the Attachment of Biomimetic Lipid Bilayers to Surfaces. J. Am. Chem. Soc. 1999, 121, 5274–5280. [Google Scholar] [CrossRef]
- Kendall, J.K.R.; Johnson, B.R.G.; Symonds, P.H.; Imperato, G.; Bushby, R.J.; Gwyer, J.D.; van Berkel, C.; Evans, S.D.; Jeuken, L.J.C. Effect of the Structure of Cholesterol-Based Tethered Bilayer Lipid Membranes on Ionophore Activity. ChemPhysChem 2010, 11, 2191–2198. [Google Scholar] [CrossRef]
- Achalkumar, A.S.; Bushby, R.J.; Evans, S.D. Cholesterol-based anchors and tethers for phospholipid bilayers and for model biological membranes. Soft Matter 2010, 6, 6036–6051. [Google Scholar] [CrossRef]
- He, L.; Robertson, J.W.F.; Li, J.; Kärcher, I.; Schiller, S.M.; Knoll, W.; Naumann, R. Tethered Bilayer Lipid Membranes Based on Monolayers of Thiolipids Mixed with a Complementary Dilution Molecule. 1. Incorporation of Channel Peptides. Langmuir 2005, 21, 11666–11672. [Google Scholar] [CrossRef]
- Becucci, L.; Maran, F.; Guidelli, R. Probing membrane permeabilization by the antibiotic lipopeptaibol trichogin GA IV in a tethered bilayer lipid membrane. Biochim. Biophys. Acta Biomembr. 2012, 1818, 1656–1662. [Google Scholar] [CrossRef] [PubMed]
- Becucci, L.; Tramonti, V.; Fiore, A.; Fogliano, V.; Scaloni, A.; Guidelli, R. Channel-forming activity of syringomycin E in two mercury-supported biomimetic membranes. Biochim. Biophys. Acta Biomembr. 2015, 1848, 932–941. [Google Scholar] [CrossRef] [Green Version]
- Becucci, L.; Rossi, M.; Fiore, A.; Scaloni, A.; Guidelli, R. Channel-forming activity of syringopeptin 25A in mercury-supported lipid bilayers with a phosphatidylcholine distal leaflet. Bioelectrochemistry 2016, 108, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Becucci, L.; Toppi, A.; Fiore, A.; Scaloni, A.; Guidelli, R. Channel-forming activity of syringopeptin 25A in mercury-supported phospholipid monolayers and negatively charged bilayers. Bioelectrochemistry 2016, 111, 131–142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Becucci, L.; Innocenti, M.; Salvietti, E.; Rindi, A.; Pasquini, I.; Vassalli, M.; Foresti, M.L.; Guidelli, R. Potassium ion transport by gramicidin and valinomycin across a Ag(111)-supported tethered bilayer lipid membrane. Electrochim. Acta 2008, 53, 6372–6379. [Google Scholar] [CrossRef]
- Vockenroth, I.K.; Atanasova, P.P.; Jenkins, A.T.A.; Köper, I. Incorporation of α-Hemolysin in Different Tethered Bilayer Lipid Membrane Architectures. Langmuir 2008, 24, 496–502. [Google Scholar] [CrossRef]
- Braunagel, J.; Junghans, A.; Köper, I. Membrane-Based Sensing Approaches. Aust. J. Chem. 2011, 64, 54–61. [Google Scholar] [CrossRef]
- Guidelli, R.; Becucci, L. Ion transport across biomembranes and model membranes. J. Solid State Electrochem. 2011, 15, 1459–1470. [Google Scholar] [CrossRef]
- Naumann, R.; Walz, D.; Schiller, S.M.; Knoll, W. Kinetics of valinomycin-mediated K+ ion transport through tethered bilayer lipid membranes. J. Electroanal. Chem. 2003, 550–551, 241–252. [Google Scholar] [CrossRef]
- Roskamp, R.F.; Vockenroth, I.K.; Eisenmenger, N.; Braunagel, J.; Köper, I. Functional Tethered Bilayer Lipid Membranes on Aluminum Oxide. ChemPhysChem 2008, 9, 1920–1924. [Google Scholar] [CrossRef]
- Vockenroth, I.K.; Atanasova, P.P.; Long, J.R.; Jenkins, A.T.A.; Knoll, W.; Köper, I. Functional incorporation of the pore forming segment of AChR M2 into tethered bilayer lipid membranes. Biochim. Biophys. Acta Biomembr. 2007, 1768, 1114–1120. [Google Scholar] [CrossRef]
- Datta, S.A.K.; Heinrich, F.; Raghunandan, S.; Krueger, S.; Curtis, J.E.; Rein, A.; Nanda, H. HIV-1 Gag extension: Conformational changes require simultaneous interaction with membrane and nucleic acid. J. Mol. Biol. 2011, 406, 205–214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clifton, L.A.; Holt, S.A.; Hughes, A.V.; Daulton, E.L.; Arunmanee, W.; Heinrich, F.; Khalid, S.; Jefferies, D.; Charlton, T.R.; Webster, J.R.P.; et al. An Accurate In Vitro Model of the E. coli Envelope. Angew. Chem. Int. Ed. 2015, 54, 11952–11955. [Google Scholar] [CrossRef] [Green Version]
- Becucci, L.; Moncelli, M.R.; Guidelli, R. Impedance Spectroscopy of OmpF Porin Reconstituted into a Mercury-Supported Lipid Bilayer. Langmuir 2006, 22, 1341–1346. [Google Scholar] [CrossRef] [PubMed]
- Becucci, L.; Carbone, M.V.; Biagiotti, T.; D’Amico, M.; Olivotto, M.; Guidelli, R. Incorporation of the HERG Potassium Channel in a Mercury Supported Lipid Bilayer. J. Phys. Chem. B 2008, 112, 1315–1319. [Google Scholar] [CrossRef] [PubMed]
- Naumann, R.L.C.; Nowak, C.; Knoll, W. Proteins in biomimetic membranes: Promises and facts. Soft Matter 2011, 7, 9535–9548. [Google Scholar] [CrossRef]
- Hoiles, W.; Gupta, R.; Cornell, B.; Cranfield, C.; Krishnamurthy, V. The Effect of Tethers on Artificial Cell Membranes: A Coarse-Grained Molecular Dynamics Study. PLoS ONE 2016, 11, e0162790. [Google Scholar] [CrossRef] [PubMed]
- Ragaliauskas, T.; Mickevicius, M.; Rakovska, B.; Penkauskas, T.; Vanderah, D.J.; Heinrich, F.; Valincius, G. Fast formation of low-defect-density tethered bilayers by fusion of multilamellar vesicles. Biochim. Biophys. Acta Biomembr. 2017, 1859, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Preta, G.; Jankunec, M.; Heinrich, F.; Griffin, S.; Sheldon, I.M.; Valincius, G. Tethered bilayer membranes as a complementary tool for functional and structural studies: The pyolysin case. Biochim. Biophys. Acta Biomembr. 2016, 1858, 2070–2080. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Penkauskas, T.; Zentelyte, A.; Ganpule, S.; Valincius, G.; Preta, G. Pleiotropic effects of statins via interaction with the lipid bilayer: A combined approach. Biochim. Biophys. Acta Biomembr. 2020, 1862, 183306. [Google Scholar] [CrossRef] [PubMed]
- Ragaliauskas, T.; Plečkaitytė, M.; Jankunec, M.; Labanauskas, L.; Baranauskiene, L.; Valincius, G. Inerolysin and vaginolysin, the cytolysins implicated in vaginal dysbiosis, differently impair molecular integrity of phospholipid membranes. Sci. Rep. 2019, 9, 10606. [Google Scholar] [CrossRef] [PubMed]
- Valincius, G.; Heinrich, F.; Budvytyte, R.; Vanderah, D.J.; McGillivray, D.J.; Sokolov, Y.; Hall, J.E.; Lösche, M. Soluble Amyloid Beta-Oligomers Affect Dielectric Membrane Properties by Bilayer Insertion and Domain Formation: Implications for Cell Toxicity. Biophys. J. 2008, 95, 4845–4861. [Google Scholar] [CrossRef] [Green Version]
- Kozuch, J.; Steinem, C.; Hildebrandt, P.; Millo, D. Combined Electrochemistry and Surface-Enhanced Infrared Absorption Spectroscopy of Gramicidin A Incorporated into Tethered Bilayer Lipid Membranes. Angew. Chem. Int. Ed. 2012, 51, 8114–8117. [Google Scholar] [CrossRef] [PubMed]
- Weiss, S.A.; Bushby, R.J.; Evans, S.D.; Henderson, P.J.F.; Jeuken, L.J.C. Characterization of cytochrome bo3 activity in a native-like surface-tethered membrane. Biochem. J. 2009, 417, 555–560. [Google Scholar] [CrossRef] [Green Version]
- Weiss, S.A.; Bushby, R.J.; Evans, S.D.; Jeuken, L.J.C. A study of cytochrome bo3 in a tethered bilayer lipid membrane. Biochim. Biophys. Acta Bioenerg. 2010, 1797, 1917–1923. [Google Scholar] [CrossRef] [Green Version]
- McMillan, D.G.G.; Marritt, S.J.; Firer-Sherwood, M.A.; Shi, L.; Richardson, D.J.; Evans, S.D.; Elliott, S.J.; Butt, J.N.; Jeuken, L.J.C. Protein–Protein Interaction Regulates the Direction of Catalysis and Electron Transfer in a Redox Enzyme Complex. J. Am. Chem. Soc. 2013, 135, 10550–10556. [Google Scholar] [CrossRef]
- Johnson, B.R.G.; Bushby, R.J.; Colyer, J.; Evans, S.D. Self-Assembly of Actin Scaffolds at Ponticulin-Containing Supported Phospholipid Bilayers. Biophys. J. 2006, 90, L21–L23. [Google Scholar] [CrossRef] [Green Version]
- Spencelayh, M.J.; Cheng, Y.; Bushby, R.J.; Bugg, T.D.H.; Li, J.-j.; Henderson, P.J.F.; O’Reilly, J.; Evans, S.D. Antibiotic Action and Peptidoglycan Formation on Tethered Lipid Bilayer Membranes. Angew. Chem. Int. Ed. 2006, 45, 2111–2116. [Google Scholar] [CrossRef]
- Raguse, B.; Braach-Maksvytis, V.; Cornell, B.A.; King, L.G.; Osman, P.D.J.; Pace, R.J.; Wieczorek, L. Tethered Lipid Bilayer Membranes: Formation and Ionic Reservoir Characterization. Langmuir 1998, 14, 648–659. [Google Scholar] [CrossRef]
- Cranfield, C.G.; Cornell, B.A.; Grage, S.L.; Duckworth, P.; Carne, S.; Ulrich, A.S.; Martinac, B. Transient Potential Gradients and Impedance Measures of Tethered Bilayer Lipid Membranes: Pore-Forming Peptide Insertion and the Effect of Electroporation. Biophys. J. 2014, 106, 182–189. [Google Scholar] [CrossRef] [Green Version]
- Yin, P.; Burns, C.J.; Osman, P.D.J.; Cornell, B.A. A tethered bilayer sensor containing alamethicin channels and its detection of amiloride based inhibitors. Biosens. Bioelectron. 2003, 18, 389–397. [Google Scholar] [CrossRef]
- Al Khamici, H.; Hossain, K.R.; Cornell, B.A.; Valenzuela, S.M. Investigating Sterol and Redox Regulation of the Ion Channel Activity of CLIC1 Using Tethered Bilayer Membranes. Membranes 2016, 6, 51. [Google Scholar] [CrossRef] [Green Version]
- Alghalayini, A.; Garcia, A.; Berry, T.; Cranfield, C.G. The Use of Tethered Bilayer Lipid Membranes to Identify the Mechanisms of Antimicrobial Peptide Interactions with Lipid Bilayers. Antibiotics 2019, 8, 12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cranfield, C.G.; Henriques, S.T.; Martinac, B.; Duckworth, P.; Craik, D.J.; Cornell, B. Kalata B1 and Kalata B2 Have a Surfactant-Like Activity in Phosphatidylethanolomine-Containing Lipid Membranes. Langmuir 2017, 33, 6630–6637. [Google Scholar] [CrossRef] [PubMed]
- Berry, T.; Dutta, D.; Chen, R.; Leong, A.; Wang, H.; Donald, W.A.; Parviz, M.; Cornell, B.; Willcox, M.; Kumar, N.; et al. Lipid Membrane Interactions of the Cationic Antimicrobial Peptide Chimeras Melimine and Cys-Melimine. Langmuir 2018, 34, 11586–11592. [Google Scholar] [CrossRef] [PubMed]
- Maccarini, M.; Gayet, L.; Alcaraz, J.-P.; Liguori, L.; Stidder, B.; Watkins, E.B.; Lenormand, J.-L.; Martin, D.K. Functional Characterization of Cell-Free Expressed OprF Porin from Pseudomonas aeruginosa Stably Incorporated in Tethered Lipid Bilayers. Langmuir 2017. [Google Scholar] [CrossRef]
- Alghalayini, A.; Jiang, L.; Gu, X.; Yeoh, G.H.; Cranfield, C.G.; Timchenko, V.; Cornell, B.A.; Valenzuela, S.M. Real-time monitoring of heat transfer between gold nanoparticles and tethered bilayer lipid membranes. Biochim. Biophys. Acta Biomembr. 2020, 1862, 183334. [Google Scholar] [CrossRef]
- Garcia, A.; Deplazes, E.; Aili, S.; Padula, M.P.; Touchard, A.; Murphy, C.; Mirissa Lankage, U.; Nicholson, G.M.; Cornell, B.; Cranfield, C.G. Label-Free, Real-Time Phospholipase-A Isoform Assay. ACS Biomater. Sci. Eng. 2020, 6, 4714–4721. [Google Scholar] [CrossRef] [PubMed]
- Facey, J.A.; Steele, J.R.; Violi, J.P.; Mitrovic, S.M.; Cranfield, C. An examination of microcystin-LR accumulation and toxicity using tethered bilayer lipid membranes (tBLMs). Toxicon 2019, 158, 51–56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Squillace, O.; Esnault, C.; Pilard, J.-F.; Brotons, G. Electrodes for Membrane Surface Science. Bilayer Lipid Membranes Tethered by Commercial Surfactants on Electrochemical Sensors. ACS Sens. 2019, 4, 1337–1345. [Google Scholar] [CrossRef] [PubMed]
- Chadli, M.; Rebaud, S.; Maniti, O.; Tillier, B.; Cortès, S.; Girard-Egrot, A. New Tethered Phospholipid Bilayers Integrating Functional G-Protein-Coupled Receptor Membrane Proteins. Langmuir 2017, 33, 10385–10401. [Google Scholar] [CrossRef]
- Naumann, R.; Jonczyk, A.; Kopp, R.; van Esch, J.; Ringsdorf, H.; Knoll, W.; Gräber, P. Incorporation of Membrane Proteins in Solid-Supported Lipid Layers. Angew. Chem. Int. Ed. 1995, 34, 2056–2058. [Google Scholar] [CrossRef]
- Bunjes, N.; Schmidt, E.K.; Jonczyk, A.; Rippmann, F.; Beyer, D.; Ringsdorf, H.; Gräber, P.; Knoll, W.; Naumann, R. Thiopeptide-Supported Lipid Layers on Solid Substrates. Langmuir 1997, 13, 6188–6194. [Google Scholar] [CrossRef]
- Yildiz, A.A.; Knoll, W.; Gennis, R.B.; Sinner, E.K. Cell-free synthesis of cytochrome bo(3) ubiquinol oxidase in artificial membranes. Anal. Biochem. 2012, 423, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Naumann, R.; Jonczyk, A.; Hampel, C.; Ringsdorf, H.; Knoll, W.; Bunjes, N.; Gräber, P. Coupling of proton translocation through ATPase incorporated into supported lipid bilayers to an electrochemical process. Bioelectrochem. Bioenerg. 1997, 42, 241–247. [Google Scholar] [CrossRef]
- Yildiz, A.A.; Kang, C.B.; Sinner, E.K. Biomimetic membrane platform containing hERG potassium channel and its application to drug screening. Analyst 2013, 138, 2007–2012. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sinner, E.-K.; Reuning, U.; Kök, F.N.; Saccà, B.; Moroder, L.; Knoll, W.; Oesterhelt, D. Incorporation of integrins into artificial planar lipid membranes: Characterization by plasmon-enhanced fluorescence spectroscopy. Anal. Biochem. 2004, 333, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Becucci, L.; Guidelli, R.; Peggion, C.; Toniolo, C.; Moncelli, M.R. Incorporation of channel-forming peptides in a Hg-supported lipid bilayer. J. Electroanal. Chem. 2005, 576, 121–128. [Google Scholar] [CrossRef]
- Song, H.; Sinner, E.-K.; Knoll, W. Peptid-tethered bilayer lipid membranes and their interaction with Amyloid ß-peptide. Biointerphases 2007, 2, 151–158. [Google Scholar] [CrossRef] [Green Version]
- Peggion, C.; Formaggio, F.; Toniolo, C.; Becucci, L.; Moncelli, M.R.; Guidelli, R. A Peptide-Tethered Lipid Bilayer on Mercury as a Biomimetic System. Langmuir 2001, 17, 6585–6592. [Google Scholar] [CrossRef]
- Baumgart, T.; Kreiter, M.; Lauer, H.; Naumann, R.; Jung, G.; Jonczyk, A.; Offenhäusser, A.; Knoll, W. Fusion of small unilamellar vesicles onto laterally mixed self-assembled monolayers of thiolipopeptides. J. Colloid Interface Sci. 2003, 258, 298–309. [Google Scholar] [CrossRef]
- Albertorio, F.; Diaz, A.J.; Yang, T.; Chapa, V.A.; Kataoka, S.; Castellana, E.T.; Cremer, P.S. Fluid and Air-Stable Lipopolymer Membranes for Biosensor Applications. Langmuir 2005, 21, 7476–7482. [Google Scholar] [CrossRef]
- Diaz, A.J.; Albertorio, F.; Daniel, S.; Cremer, P.S. Double Cushions Preserve Transmembrane Protein Mobility in Supported Bilayer Systems. Langmuir 2008, 24, 6820–6826. [Google Scholar] [CrossRef] [Green Version]
- Wallin, M.; Choi, J.-H.; Oh Kim, S.; Cho, N.-J.; Andersson, M. Peptide-induced formation of a tethered lipid bilayer membrane on mesoporous silica. Eur. Biophys. J. 2015, 44, 27–36. [Google Scholar] [CrossRef]
- Proux-Delrouyre, V.; Laval, J.-M.; Bourdillon, C. Formation of Streptavidin-Supported Lipid Bilayers on Porous Anodic Alumina: Electrochemical Monitoring of Triggered Vesicle Fusion. J. Am. Chem. Soc. 2001, 123, 9176–9177. [Google Scholar] [CrossRef]
- Veneziano, R.; Rossi, C.; Chenal, A.; Brenner, C.; Ladant, D.; Chopineau, J. Synthesis and characterization of tethered lipid assemblies for membrane protein reconstitution (Review). Biointerphases 2017, 12, 04E301. [Google Scholar] [CrossRef] [PubMed]
- Voegele, A.; O’Brien, D.P.; Subrini, O.; Sapay, N.; Cannella, S.E.; Enguéné, V.Y.N.; Hessel, A.; Karst, J.; Hourdel, V.; Perez, A.C.S.; et al. Translocation and calmodulin-activation of the adenylate cyclase toxin (CyaA) of Bordetella pertussis. Pathog. Dis. 2018, 76. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.D.; Linman, M.J.; Wilkop, T.; Cheng, Q. Regenerable Tethered Bilayer Lipid Membrane Arrays for Multiplexed Label-Free Analysis of Lipid−Protein Interactions on Poly(dimethylsiloxane) Microchips Using SPR Imaging. Anal. Chem. 2009, 81, 1146–1153. [Google Scholar] [CrossRef]
- Anderson, R.G.W.; Jacobson, K. A Role for Lipid Shells in Targeting Proteins to Caveolae, Rafts, and Other Lipid Domains. Science 2002, 296, 1821–1825. [Google Scholar] [CrossRef]
- Gamper, N.; Shapiro, M.S. Regulation of ion transport proteins by membrane phosphoinositides. Nat. Rev. Neurosci. 2007, 8, 921–934. [Google Scholar] [CrossRef] [PubMed]
- De Kruijff, B.; Cullis, P.R.; Verkleij, A.J. Non-bilayer lipid structures in model and biological membranes. Trends Biochem. Sci. 1980, 5, 79–81. [Google Scholar] [CrossRef]
- Cullis, P.R.; De Kruijff, B. Lipid polymorphism and the functional roles of lipids in biological membranes. Biochim. Biophys. Acta 1979, 559, 399–420. [Google Scholar] [CrossRef]
- Garab, G.; Ughy, B.; Goss, R. Role of MGDG and Non-bilayer Lipid Phases in the Structure and Dynamics of Chloroplast Thylakoid Membranes. Subcell. Biochem. 2016, 86, 127–157. [Google Scholar] [CrossRef] [PubMed]
- Epand, R.M. Lipid polymorphism and protein–lipid interactions. Biochim. Biophys. Acta 1998, 1376, 353–368. [Google Scholar] [CrossRef]
- Pace, H.; Simonsson Nyström, L.; Gunnarsson, A.; Eck, E.; Monson, C.; Geschwindner, S.; Snijder, A.; Höök, F. Preserved Transmembrane Protein Mobility in Polymer-Supported Lipid Bilayers Derived from Cell Membranes. Anal. Chem. 2015, 87, 9194–9203. [Google Scholar] [CrossRef]
- Roder, F.; Birkholz, O.; Beutel, O.; Paterok, D.; Piehler, J. Spatial Organization of Lipid Phases in Micropatterned Polymer-Supported Membranes. J. Am. Chem. Soc. 2013, 135, 1189–1192. [Google Scholar] [CrossRef]
- Richards, M.J.; Hsia, C.-Y.; Singh, R.R.; Haider, H.; Kumpf, J.; Kawate, T.; Daniel, S. Membrane Protein Mobility and Orientation Preserved in Supported Bilayers Created Directly from Cell Plasma Membrane Blebs. Langmuir 2016, 32, 2963–2974. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.-Y.; Pappa, A.-M.; Pavia, A.; Pitsalidis, C.; Thiburce, Q.; Salleo, A.; Owens, R.M.; Daniel, S. Self-Assembly of Mammalian-Cell Membranes on Bioelectronic Devices with Functional Transmembrane Proteins. Langmuir 2020, 36, 7325–7331. [Google Scholar] [CrossRef] [PubMed]
- Sumino, A.; Dewa, T.; Takeuchi, T.; Sugiura, R.; Sasaki, N.; Misawa, N.; Tero, R.; Urisu, T.; Gardiner, A.T.; Cogdell, R.J.; et al. Construction and Structural Analysis of Tethered Lipid Bilayer Containing Photosynthetic Antenna Proteins for Functional Analysis. Biomacromolecules 2011, 12, 2850–2858. [Google Scholar] [CrossRef] [PubMed]
- Girard-Egrot, A.; Maniti, O.; Rebaud, S. Subtrate/Peptide/Lipid Bilayer Assembly, Preparation Methods and Associated Dectection Methods. International Patent WO2016066947, 6 May 2016. [Google Scholar]
- Robelek, R.; Lemker, E.S.; Wiltschi, B.; Kirste, V.; Naumann, R.; Oesterhelt, D.; Sinner, E.-K. Incorporation of In Vitro Synthesized GPCR into a Tethered Artificial Lipid Membrane System. Angew. Chem. Int. Ed. 2007, 46, 605–608. [Google Scholar] [CrossRef]
- Bornhorst, J.A.; Falke, J.J. Purification of proteins using polyhistidine affinity tags. Methods Enzymol. 2000, 326, 245–254. [Google Scholar] [CrossRef] [Green Version]
- Santafé, A.l.A.M.; Blum, L.c.J.; Marquette, C.A.; Girard-Egrot, A.s.P. Chelating Langmuir−Blodgett Film: A New Versatile Chemiluminescent Sensing Layer for Biosensor Applications. Langmuir 2010, 26, 2160–2166. [Google Scholar] [CrossRef] [PubMed]
- Nye, J.A.; Groves, J.T. Kinetic Control of Histidine-Tagged Protein Surface Density on Supported Lipid Bilayers. Langmuir 2008, 24, 4145–4149. [Google Scholar] [CrossRef] [Green Version]
- Elazar, M.; Cheong, K.H.; Liu, P.; Greenberg, H.B.; Rice, C.M.; Glenn, J.S. Amphipathic helix-dependent localization of NS5A mediates hepatitis C virus RNA replication. J. Virol. 2003, 77, 6055–6061. [Google Scholar] [CrossRef] [Green Version]
- Cho, N.-J.; Wang, G.; Edvardsson, M.; Glenn, J.S.; Hook, F.; Frank, C.W. Alpha-Helical Peptide-Induced Vesicle Rupture Revealing New Insight into the Vesicle Fusion Process As Monitored in Situ by Quartz Crystal Microbalance-Dissipation and Reflectometry. Anal. Chem. 2009, 81, 4752–4761. [Google Scholar] [CrossRef] [PubMed]
- Cho, N.-J.; Dvory-Sobol, H.; Xiong, A.; Cho, S.-J.; Frank, C.W.; Glenn, J.S. Mechanism of an Amphipathic α-Helical Peptide’s Antiviral Activity Involves Size-Dependent Virus Particle Lysis. ACS Chem. Biol. 2009, 4, 1061–1067. [Google Scholar] [CrossRef] [PubMed]
- Hardy, G.J.; Nayak, R.; Munir Alam, S.; Shapter, J.G.; Heinrich, F.; Zauscher, S. Biomimetic supported lipid bilayers with high cholesterol content formed by [small alpha]-helical peptide-induced vesicle fusion. J. Mater. Chem. 2012, 22, 19506–19513. [Google Scholar] [CrossRef] [Green Version]
- Cullis, P.R.; Hope, M.J.; Tilcock, C.P.S. Lipid polymorphism and the roles of lipids in membranes. Chem. Phys. Lipids 1986, 40, 127–144. [Google Scholar] [CrossRef]
- Siegel, D.P.; Epand, R.M. The mechanism of lamellar-to-inverted hexagonal phase transitions in phosphatidylethanolamine: Implications for membrane fusion mechanisms. Biophys. J. 1997, 73, 3089–3111. [Google Scholar] [CrossRef] [Green Version]
- Friedrich, M.G.; Gieβ, F.; Naumann, R.; Knoll, W.; Ataka, K.; Heberle, J.; Hrabakova, J.; Murgida, D.H.; Hildebrandt, P. Active site structure and redox processes of cytochrome c oxidase immobilised in a novel biomimetic lipid membrane on an electrode. Chem. Commun. 2004, 2376–2377. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, M.G.; Plum, M.A.; Santonicola, M.G.; Kirste, V.U.; Knoll, W.; Ludwig, B.; Naumann, R.L.C. In Situ Monitoring of the Catalytic Activity of Cytochrome c Oxidase in a Biomimetic Architecture. Biophys. J. 2008, 95, 1500–1510. [Google Scholar] [CrossRef] [Green Version]
- Ataka, K.; Richter, B.; Heberle, J. Orientational Control of the Physiological Reaction of Cytochrome c Oxidase Tethered to a Gold Electrode. J. Phys. Chem. B 2006, 110, 9339–9347. [Google Scholar] [CrossRef] [Green Version]
- Friedrich, M.G.; Kirste, V.U.; Zhu, J.; Gennis, R.B.; Knoll, W.; Naumann, R.L.C. Activity of Membrane Proteins Immobilized on Surfaces as a Function of Packing Density. J. Phys. Chem. B 2008, 112, 3193–3201. [Google Scholar] [CrossRef]
- Jiang, X.; Zaitseva, E.; Schmidt, M.; Siebert, F.; Engelhard, M.; Schlesinger, R.; Ataka, K.; Vogel, R.; Heberle, J. Resolving voltage-dependent structural changes of a membrane photoreceptor by surface-enhanced IR difference spectroscopy. Proc. Natl. Acad. Sci. USA 2008, 105, 12113–12117. [Google Scholar] [CrossRef] [Green Version]
- Nedelkovski, V.; Schwaighofer, A.; Wraight, C.A.; Nowak, C.; Naumann, R.L.C. Surface-Enhanced Infrared Absorption Spectroscopy (SEIRAS) of Light-Activated Photosynthetic Reaction Centers from Rhodobacter sphaeroides Reconstituted in a Biomimetic Membrane System. J. Phys. Chem. C 2013, 117, 16357–16363. [Google Scholar] [CrossRef]
- Kriegel, S.; Uchida, T.; Osawa, M.; Friedrich, T.; Hellwig, P. Biomimetic Environment to Study, E. coli Complex I through Surface-Enhanced IR Absorption Spectroscopy. Biochemistry 2014, 53, 6340–6347. [Google Scholar] [CrossRef]
- Kibrom, A.; Roskamp, R.F.; Jonas, U.; Menges, B.; Knoll, W.; Paulsen, H.; Naumann, R.L.C. Hydrogel-supported protein-tethered bilayer lipid membranes: A new approach toward polymer-supported lipid membranes. Soft Matter 2011, 7, 237–246. [Google Scholar] [CrossRef]
- Geiss, A.F.; Khandelwal, R.; Baurecht, D.; Bliem, C.; Reiner-Rozman, C.; Boersch, M.; Ullmann, G.M.; Loew, L.M.; Naumann, R.L.C. pH and Potential Transients of the bc1 Complex Co-Reconstituted in Proteo-Lipobeads with the Reaction Center from Rb. sphaeroides. J. Phys. Chem. B 2017, 121, 143–152. [Google Scholar] [CrossRef] [Green Version]
- Geiss, A.F.; Bliem, C.; Frank, P.; Reiner-Rozman, C.; Kewney, J.; Boersch, M.; Naumann, R.L.C. Proteo-lipobeads to encapsulate cytochrome c oxidase from Paracoccus denitrificans. J. Colloid Interface Sci. 2017, 500, 119–125. [Google Scholar] [CrossRef]
- Schadauer, F.; Geiss, A.F.; Srajer, J.; Siebenhofer, B.; Frank, P.; Reiner-Rozman, C.; Ludwig, B.; Richter, O.M.; Nowak, C.; Naumann, R.L. Silica nanoparticles for the oriented encapsulation of membrane proteins into artificial bilayer lipid membranes. Langmuir 2015, 31, 2511–2516. [Google Scholar] [CrossRef]
- Frank, P.; Siebenhofer, B.; Hanzer, T.; Geiss, A.F.; Schadauer, F.; Reiner-Rozman, C.; Durham, B.; Loew, L.M.; Ludwig, B.; Richter, O.H.; et al. Proteo-lipobeads for the oriented encapsulation of membrane proteins. Soft Matter 2015, 11, 2906–2908. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leutenegger, M.; Lasser, T.; Sinner, E.K.; Robelek, R. Imaging of G protein-coupled receptors in solid-supported planar lipid membranes. Biointerphases 2008, 3, FA136. [Google Scholar] [CrossRef]
- Damiati, S.; Zayni, S.; Schrems, A.; Kiene, E.; Sleytr, U.B.; Chopineau, J.; Schuster, B.; Sinner, E.-K. Inspired and stabilized by nature: Ribosomal synthesis of the human voltage gated ion channel (VDAC) into 2D-protein-tethered lipid interfaces. Biomater. Sci. 2015, 3, 1406–1413. [Google Scholar] [CrossRef]
- Kim, D.-M.; Swartz, J.R. Prolonging Cell-Free Protein Synthesis by Selective Reagent Additions. Biotechnol. Progr. 2000, 16, 385–390. [Google Scholar] [CrossRef]
- Kim, T.-W.; Kim, D.-M.; Choi, C.-Y. Rapid production of milligram quantities of proteins in a batch cell-free protein synthesis system. J. Biotechnol. 2006, 124, 373–380. [Google Scholar] [CrossRef]
- Liguori, L.; Marques, B.; Lenormand, J.-L. A Bacterial Cell-Free Expression System to Produce Membrane Proteins and Proteoliposomes: From cDNA to Functional Assay. In Current Protocols in Protein Science; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2001. [Google Scholar]
- Liguori, L.; Marques, B.; Villegas-Méndez, A.; Rothe, R.; Lenormand, J.L. Production of membrane proteins using cell-free expression systems. Expert Rev. Proteom. 2007, 4, 79–90. [Google Scholar] [CrossRef]
- Ryabova, L.A.; Morozov, I.; Spirin, A.S. Continuous-flow cell-free translation, transcription-translation, and replication-translation systems. Methods Mol. Biol. 1998, 77, 179–193. [Google Scholar] [CrossRef] [PubMed]
- Spirin, A.S. High-throughput cell-free systems for synthesis of functionally active proteins. Trends Biotechnol. 2004, 22, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Swartz, J. Cell-Free Protein Expression; Springer: Berlin/Heidelberg, Germany, 2003; Volume 5. [Google Scholar]
- Swartz, J. Developing cell-free biology for industrial applications. J. Ind. Microbiol. Biotechnol. 2006, 33, 476–485. [Google Scholar] [CrossRef] [PubMed]
- Lamla, T.; Mammeri, K.; Erdmann, V.A. The cell-free protein biosynthesis applications and analysis of the system. Acta Biochim. Pol. 2001, 48, 453–465. [Google Scholar] [CrossRef]
- Ryu, H.; Fuwad, A.; Yoon, S.; Jang, H.; Lee, J.C.; Kim, S.M.; Jeon, T.-J. Biomimetic Membranes with Transmembrane Proteins: State-of-the-Art in Transmembrane Protein Applications. Int. J. Mol. Sci. 2019, 20, 1437. [Google Scholar] [CrossRef] [Green Version]
- Russell, W.M.S.; Burch, R.L. The Principles of Humane Experimental Technique. Med. J. Aust. 1960, 1, 500. [Google Scholar] [CrossRef]
 : represents either anchorlipid (i.e., Telechelics) mainly 2,3-di-O-phytanyl-sn-glycerol-1-tetraethylene glycol-d,l-α-lipoic acid ester lipid (DPhyTL), lipopolymer tethers or thio(lipo)peptides;
: represents either anchorlipid (i.e., Telechelics) mainly 2,3-di-O-phytanyl-sn-glycerol-1-tetraethylene glycol-d,l-α-lipoic acid ester lipid (DPhyTL), lipopolymer tethers or thio(lipo)peptides;  : Backfillers, mainly β-mercaptoethaonl (βME) or 6-mercaptohexanol (6-MH);
: Backfillers, mainly β-mercaptoethaonl (βME) or 6-mercaptohexanol (6-MH);  : benzyl-disulfide (tetra-ethyleneglycol)n=2 C20-phytanyl tether (or DLP);
: benzyl-disulfide (tetra-ethyleneglycol)n=2 C20-phytanyl tether (or DLP);  : ethyleneoxy-linked cholesterol (or EO3C).
: ethyleneoxy-linked cholesterol (or EO3C).
 : represents either anchorlipid (i.e., Telechelics) mainly 2,3-di-O-phytanyl-sn-glycerol-1-tetraethylene glycol-d,l-α-lipoic acid ester lipid (DPhyTL), lipopolymer tethers or thio(lipo)peptides;
: represents either anchorlipid (i.e., Telechelics) mainly 2,3-di-O-phytanyl-sn-glycerol-1-tetraethylene glycol-d,l-α-lipoic acid ester lipid (DPhyTL), lipopolymer tethers or thio(lipo)peptides;  : Backfillers, mainly β-mercaptoethaonl (βME) or 6-mercaptohexanol (6-MH);
: Backfillers, mainly β-mercaptoethaonl (βME) or 6-mercaptohexanol (6-MH);  : benzyl-disulfide (tetra-ethyleneglycol)n=2 C20-phytanyl tether (or DLP);
: benzyl-disulfide (tetra-ethyleneglycol)n=2 C20-phytanyl tether (or DLP);  : ethyleneoxy-linked cholesterol (or EO3C).
: ethyleneoxy-linked cholesterol (or EO3C).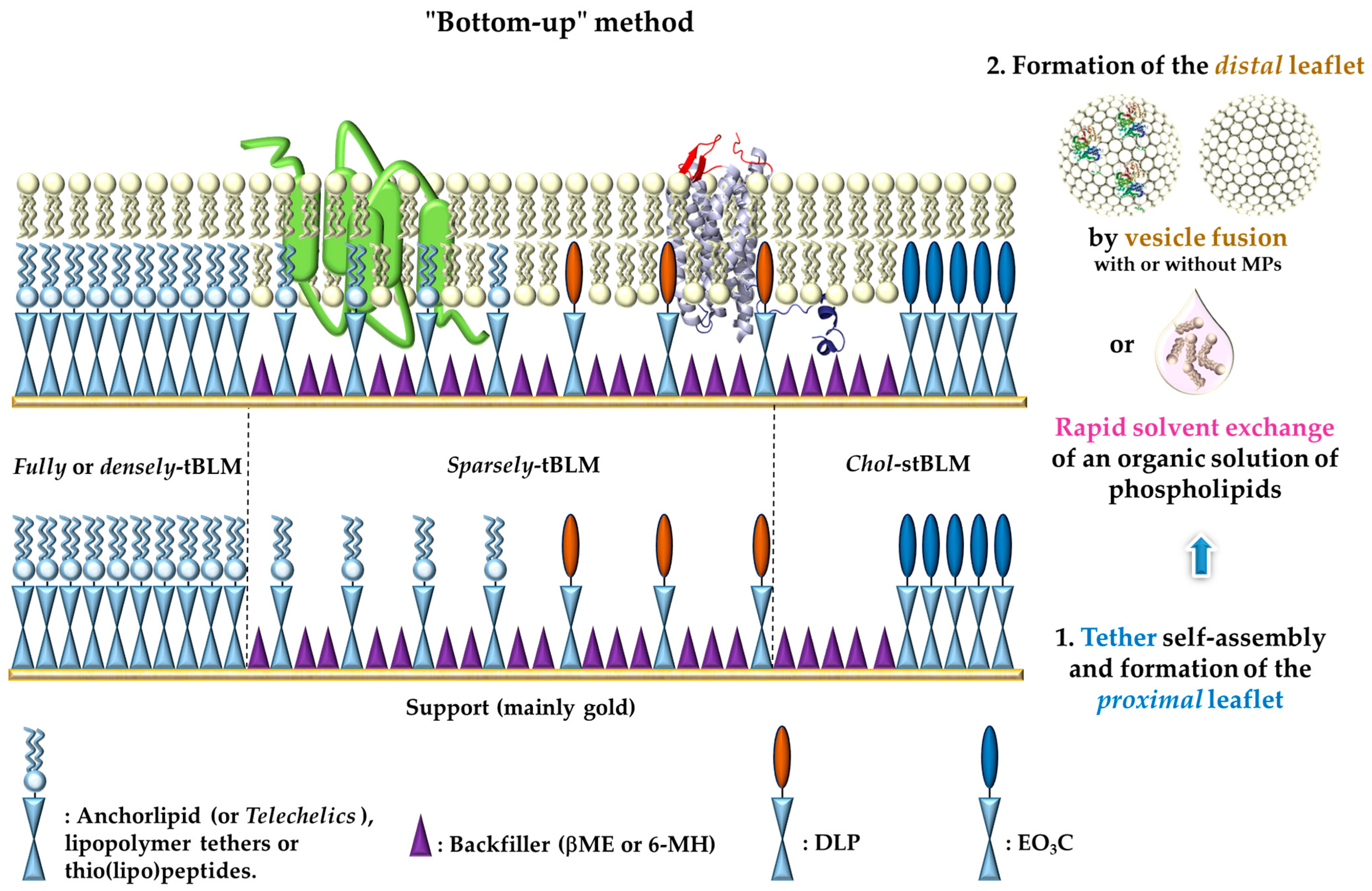
| Techniques | Bilayer Characterization | Surfaces |
|---|---|---|
| Surface plasmon resonance (SPR) imaging | Optical thickness of the bilayer, highly sensitive real-time monitoring of interactions without labeling of the analytes or the ligand, real-time monitoring of bilayer formation | Gold, silver, aluminum |
| Quartz crystal microbalance with dissipation (QCM-D) | Interfacial wet mass determination and viscoelasticity (dissipation sensitive to viscoelastic properties of the adsorbed material), (acoustic) film thickness, real-time monitoring of bilayer formation | Gold, SiO2, mica, metal oxides |
| Imaging ellipsometry (IE) | Indirect quantitative characterization of structural and functional properties of bilayers such as thickness and dry adsorbed mass (i.e., lipids in the adsorbed layer), anisotropy (lateral uniformity and phase separation), molecular area, and receptor-protein interaction affinities. Real-time large area imaging with high sensitivity | Oxide (silicon) substrates |
| Fluorescence recovery after photobleaching (FRAP) | Dynamics, fluidity, and mobility characterisation of lipids and proteins (peripheral or integral), intergrity of artificial membranes | Optically transparent substrates: glass, silica, silcon, gold |
| Electrochemical impedance spectroscopy (EIS) | Electrical properties (resistance and capacitance) of lipid bilayer membranes, formation process in real-time, stability of the membrane, characterization of incorporated ion channels | Gold, silicon |
| Atomic force microscopy (AFM) | In-plane structure and morphology: surface roughness determination, investigation of bilayer surface at the nanoscale range in real-time and in aqueous environment, direct measure of physical properties at high spatial resolution, phase separation (domain formation) and quantification of bilayer thickness | Atomically flat surfaces: mica, silicon, quartz, flat gold |
| (AFM) single-molecule Force Spectroscopy (FS) | Membrane stiffness and mechanical stability on the nanometer length scale, in-depth insight of the orientation of reconstituted transmembrane proteins | Mica, silicon, quartz, flat gold |
| Neutron Reflectometry (NR) | Non-damaging technique giving high structural information on lipid bilayer and internal distribution of components (lipid or protein) within the bilayer (thickness of stratified layers normal to the interface), roughness and interaction with inserted proteins (easy differentiation of lipid and polypeptide components across the membrane structure after interaction) | Gold, silicon |
| X-ray photoelectron spectroscopy (XPS) | Provides quantitaive analysis of elemental composition of a surface and its chemical state | Quartz |
| Grazing incidence small angle neutron or X-ray scattering (GISANS and GISAXS) | Non-destructive method for the structural investigation of biomembranes and mixed lipids systems with different topologies | Performed in quartz glass |
| Name | Amino Acid Sequence |
|---|---|
| P5 | Lip-Ala-Ala-Ala-Ala-Ala-COOH 1 |
| P7 | HS-(CH2)2-Ala-Ser-Ser-Ala-Ala-Ser-Ala-COOH |
| LP12 2 | HS-Cys-Ala-Ser-Ala-Ala-Ser-Ser-Ala-Pro-Ser-Ser-Lys(Myr)-Myr 1 |
| P19 3 | HS-Cys-Ser-Arg-Ala-Arg-Lys-Gln-Ala-Ala-Ser-Ile-Lys-Val-Ala-Val-Ser-Ala-Asp-Arg-COOH |
| P19-4H | HS-Cys-Ser-Arg-Ala-Arg-Lys-Gln-Ala-Ala-Ser-Ile-Lys-Val-Ala-Val-Ser-Ala-Asp-Arg-His-His-His-His-COOH |
| Lipid Composition | Molar Percentage (Mol%) 1 |
|---|---|
| POPC | 100 |
| DOPC | 100 |
| DOPC/DOPS 2 | 75:25 |
| DOPC/DOPS doped with fluorophores | 75:25 |
| Egg PC/brain PS | 68:32 |
| Egg PC/brain PS/brain PIP2 3 | 68:30:2 |
| DOPC/DOPE/DMPA 4/Chol | 31:17:20:32 |
| POPC/SM 5/POPE 6/Chol 7 | 44:35:10:11 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Girard-Egrot, A.P.; Maniti, O. Why Do Tethered-Bilayer Lipid Membranes Suit for Functional Membrane Protein Reincorporation? Appl. Sci. 2021, 11, 4876. https://doi.org/10.3390/app11114876
Girard-Egrot AP, Maniti O. Why Do Tethered-Bilayer Lipid Membranes Suit for Functional Membrane Protein Reincorporation? Applied Sciences. 2021; 11(11):4876. https://doi.org/10.3390/app11114876
Chicago/Turabian StyleGirard-Egrot, Agnès P., and Ofelia Maniti. 2021. "Why Do Tethered-Bilayer Lipid Membranes Suit for Functional Membrane Protein Reincorporation?" Applied Sciences 11, no. 11: 4876. https://doi.org/10.3390/app11114876
APA StyleGirard-Egrot, A. P., & Maniti, O. (2021). Why Do Tethered-Bilayer Lipid Membranes Suit for Functional Membrane Protein Reincorporation? Applied Sciences, 11(11), 4876. https://doi.org/10.3390/app11114876







