Impact of Environmental Stressors on Gene Expression in the Embryo of the Italian Wall Lizard
Abstract
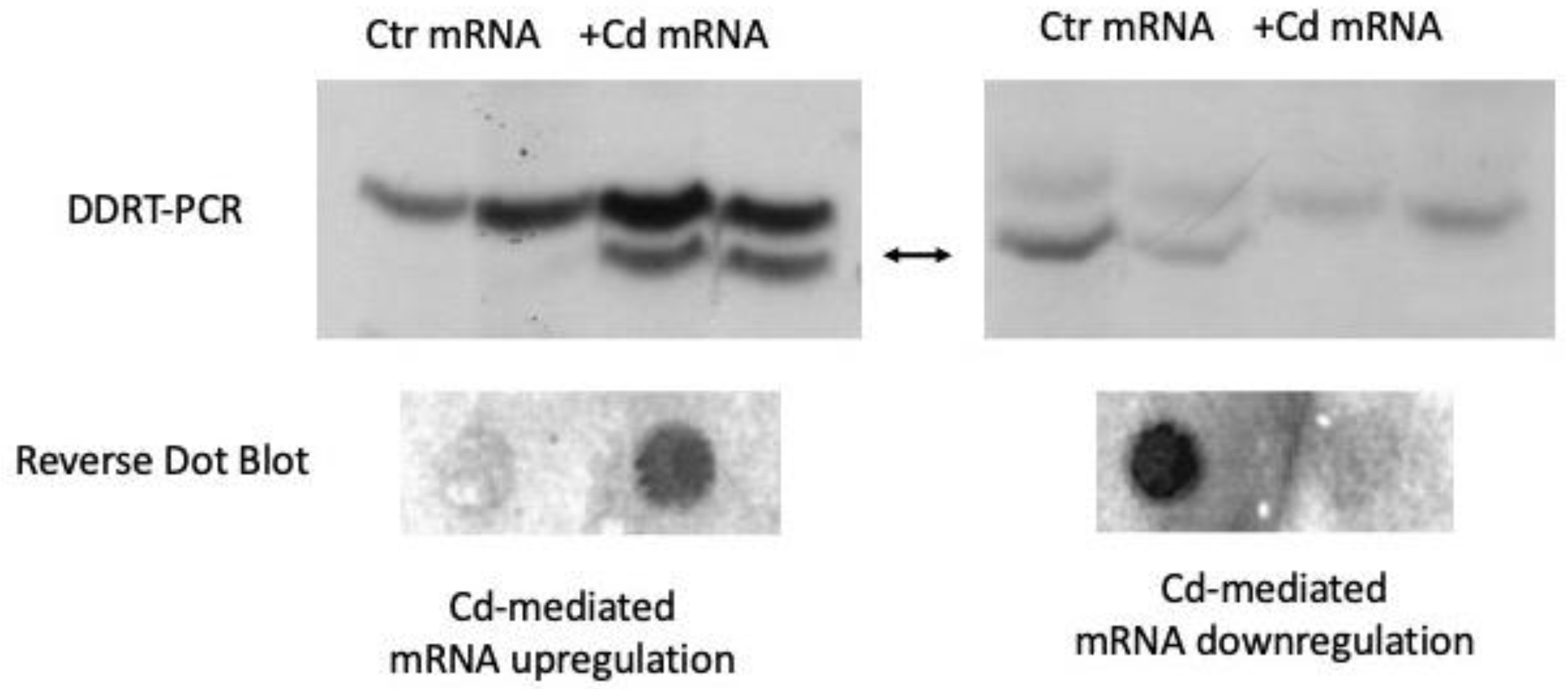
1. Introduction
Cadmium-Responsive Genes in Podarcis Siculus Embryos
2. Cd-Mediated Genes Downregulation
2.1. Transcription Regulation
Basic Transcription Factor 3
2.2. Cellular Metabolism
Three-hydroxy-3-methylglutaryl coenzyme A reductase
2.3. Cell Cycle Regulation
Fizzy and Cell Division Cycle 20 Related 1
3. Cd-Mediated Genes Upregulation
3.1. Transcription Regulation
3.1.1. RLF Zinc Finger Protein
3.1.2. Topoisomerase 1-Binding Protein with a BTB/POZ Domain
3.2. Master Regulatory Genes in Eye Development
Otx 2 and Pax6
3.3. Membrane Trafficking
Development and Differentiation-Enhancing Factor 1
3.4. Sensory Transduction and Synaptic Plasticity
3.4.1. Voltage-Dependent Sodium Channel
3.4.2. γ-Aminobutyric Acid Type B Receptor
3.5. Immune Response
Lymphocyte Function Associated Antigen 3
3.6. Cellular Detoxification
3.6.1. Metallothionein
3.6.2. Cold Stress-Responsive Genes in Podarcis Embryos
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Weigel, D.; Izaurralde, E. A tiny helper lightens the maternal load. Cell 2006, 124, 1117–1118. [Google Scholar] [CrossRef]
- Davis, T.L.; Rebay, I. Master regulators in development: Views from the Drosophila retinal determination and mammalian pluripotency gene networks. Dev. Biol. 2017, 421, 93–107. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Cao, F.; Li, X. Epigenetic Programming and Fetal Metabolic Programming. Front. Endocrinol. 2019, 10, 764. [Google Scholar] [CrossRef] [PubMed]
- Packard, G.C.; Packard, M.J. Evolution of the Cleidoic Egg Among Reptilian Antecedents of Birds. Am. Zool. 1980, 20, 351–362. [Google Scholar] [CrossRef]
- Marco, A.; López-Vicente, M.; Pérez-Mellado, V. Arsenic uptake by reptile flexible-shelled eggs from contaminated nest substrates and toxic effect on embryos. Bull. Environ. Contam. Toxicol. 2004, 72, 983–990. [Google Scholar] [CrossRef]
- Gómara, B.; Gómez, G.; Diaz-Paniagua, C.; Marco, A.; Gonzalez, M.J. PCB, DDT, arsenic, and heavy metal (Cd, Cu, Pb, and Zn) concentrations in chameleon (Chamaeleo chamaeleon) eggs from Southwest Spain. Chemosphere 2007, 68, 25–31. [Google Scholar] [CrossRef]
- Simoniello, P.; Motta, C.M.; Scudiero, R.; Trinchella, F.; Filosa, S. Cadmium-induced teratogenicity in lizard embryos: Correlation with metallothionein gene expression. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2011, 153, 119–127. [Google Scholar] [CrossRef]
- Trinchella, F.; Cannetiello, M.; Simoniello, P.; Filosa, S.; Scudiero, R. Differential gene expression profiles in embryos of the lizard Podarcis sicula under in ovo exposure to cadmium. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2010, 151, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Simoniello, P.; Trinchella, F.; Filosa, S.; Scudiero, R.; Magnani, D.; Theil, T.; Motta, C.M. Cadmium contaminated soil affects retinogenesis in lizard embryos. J. Exp. Zool. 2014, 321A, 207–219. [Google Scholar] [CrossRef] [PubMed]
- Scudiero, R.; Verderame, M.; Motta, C.M.; Simoniello, P. Unravelling the role of metallothionein on development, reproduction and detoxification in the wall lizard Podarcis sicula. Int. J. Mol. Sci. 2017, 18, 1569. [Google Scholar] [CrossRef]
- Dufaure, J.P.; Hubert, J. Table de developpement du lezard vivipare: Lacerta (Zootoca) vivipara Jacquin. Arch. Anat. Microscop. Morphol. Exp. 1961, 50, 309–328. [Google Scholar]
- Sanger, T.J.; Losos, J.B.; Gibson-Brown, J.J. A developmental staging series for the lizard genus Anolis: A new system for the integration of evolution, development, and ecology. J. Morphol. 2008, 269, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Boback, S.M.; Dichter, E.K.; Mistry, H.L. A developmental staging series for the African house snake, Boaedon (Lamprophis) fuliginosus. Zoology 2012, 115, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Lazić, M.M.; Carretero, M.A.; Crnobrnja-Isailović, J.; Kaliontzopoulou, A. Effects of environmental disturbance on phenotypic variation: An integrated assessment of canalization, developmental stability, modularity, and allometry in lizard head shape. Am. Nat. 2015, 185, 44–58. [Google Scholar] [CrossRef]
- Singh, S.K.; Das, D.; Rhen, T. Embryonic Temperature Programs Phenotype in Reptiles. Front. Physiol. 2020, 11, 35. [Google Scholar] [CrossRef] [PubMed]
- Suarez, M.E.; Wilson, H.R.; McPherson, B.N.; Mather, F.B.; Wilcox, C.J. Low temperature effects on embryonic development and hatch time. Poult. Sci. 1996, 75, 924–932. [Google Scholar] [CrossRef] [PubMed]
- Rhen, T.; Schroeder, A. Molecular mechanisms of sex determination in reptiles. Sex Dev. 2010, 4, 16–28. [Google Scholar] [CrossRef]
- Simoniello, P.; Filosa, S.; Scudiero, R.; Trinchella, F.; Motta, C.M. Cadmium impairment of reproduction in the female wall lizard Podarcis sicula. Environ. Toxicol. 2013, 28, 553–562. [Google Scholar] [CrossRef]
- Yang, L.; Shen, Q.; Zeng, T.; Li, J.; Li, W.; Wang, Y. Enrichment of imidacloprid and its metabolites in lizards and its toxic effects on gonads. Environ. Pollut. 2020, 258, 113748. [Google Scholar] [CrossRef]
- Elhamamsy, A.R. DNA methylation dynamics in plants and mammals: Overview of regulation and dysregulation. Cell Biochem. Funct. 2016, 34, 289–298. [Google Scholar] [CrossRef]
- Li, P.; Zhang, X.; Murphy, A.J.; Costa, M.; Zhao, X.; Sun, H. Downregulation of hedgehog-interacting protein (HHIP) contributes to hexavalent chromium-induced malignant transformation of human bronchial epithelial cells. Carcinogenesis 2021, 42, 136–147. [Google Scholar] [CrossRef]
- Motta, C.M.; Simoniello, P.; Filosa, S. Control of oocyte recruitment and selection in Podarcis sicula, the Italian wall lizard. In Advances in Medicine and Biology; Nova Science Publishers: Happauge, NY, USA, 2011; Volume 24, pp. 247–263. [Google Scholar]
- Verderame, M.; Scudiero, R. Health status of the lizard Podarcis siculus (Rafinesque-Schmaltz, 1810) subject to different anthropogenic pressures. CR Biol. 2019, 342, 81–89. [Google Scholar] [CrossRef]
- Scudiero, R.; Filosa, S.; Motta, C.M.; Simoniello, P.; Trinchella, F. Cadmium in the wall lizard Podarcis Sicula: Morphological and molecular effects on embryonic and adult tissues. In Reptiles: Biology, Behavior and Conservation; Baker, K.J., Ed.; Nova Science Publishers: Happauge, NY, USA, 2011; pp. 147–162. [Google Scholar]
- Liang, P.; Bauer, D.; Averboukh, L.; Warthoe, P.; Rohrwild, M.; Muller, H.; Strauss, M.; Pardee, A.B. Analysis of altered gene expression by differential display. Methods Enzymol. 1995, 254, 304–321. [Google Scholar]
- Carginale, V.; Capasso, C.; Scudiero, R.; Parisi, E. Identification of cadmium-sensitive genes in the Antarctic fish Chionodraco hamatus by mRNA differential display. Gene 2002, 299, 117–124. [Google Scholar] [CrossRef]
- Kirstein-Miles, J.; Scior, A.; Deuerling, E.; Morimoto, R.I. The nascent polypeptide-associated complex is a key regulator of proteostasis. EMBO J. 2013, 32, 1451–1468. [Google Scholar] [CrossRef]
- Zheng, X.M.; Moncollin, V.; Egly, J.M.; Chambon, P. A general transcription factor forms a stable complex with RNA polymerase B (II). Cell 1987, 50, 361–368. [Google Scholar] [CrossRef]
- Thiede, B.; Dimmler, C.; Siejak, F.; Rudel, T. Predominant identification of RNA-binding proteins in Fas-induced apoptosis by proteome analysis. J. Biol. Chem. 2001, 276, 26044–26050. [Google Scholar] [CrossRef]
- Deng, J.M.; Behringer, R.R. An insertional mutation in the BTF3 transcription factor gene leads to an early postimplantation lethality in mice. Transgenic Res. 1995, 4, 264–269. [Google Scholar] [CrossRef]
- Li, X.; Sui, J.; Xing, J.; Cao, F.; Wang, H.; Fu, C.; Wang, H. Basic transcription factor 3 expression silencing attenuates colon cancer cell proliferation and migration in vitro. Oncol. Lett. 2019, 17, 113–118. [Google Scholar] [CrossRef]
- Luo, J.; Yang, H.; Song, B.L. Mechanisms and regulation of cholesterol homeostasis. Nat. Rev. Mol. Cell Biol. 2020, 21, 225–245. [Google Scholar] [CrossRef]
- Cooper, M.K.; Wassif, C.A.; Krakowiak, P.A.; Taipale, J.; Gong, R.; Kelley, R.I.; Porter, F.D.; Beachy, P.A. A defective response to Hedgehog signaling in disorders of cholesterol biosynthesis. Nat. Genet. 2003, 33, 508–513. [Google Scholar] [CrossRef] [PubMed]
- Ohashi, K.; Osuga, J.; Tozawa, R.; Kitamine, T.; Yagyu, H.; Sekiya, M.; Tomita, S.; Okazaki, H.; Tamura, Y.; Yahagi, N.; et al. Early embryonic lethality caused by targeted disruption of the 3- hydroxy-3-methylglutaryl-CoA reductase gene. J. Biol. Chem. 2003, 278, 42936–42941. [Google Scholar] [CrossRef]
- Qiao, X.; Zhang, L.; Gamper, A.M.; Fujita, T.; Wan, Y. APC/C-Cdh1. From cell cycle to cellular differentiation and genomic integrity. Cell Cycle 2010, 9, 3904–3912. [Google Scholar] [CrossRef] [PubMed]
- Simoniello, P.; Trinchella, F.; Borrelli, L.; De Stasio, R.; Motta, C.M.; Filosa, S.; Scudiero, R. Effects of cadmium on retinal development in lizard embryo: A molecular and morphological study. Comp. Biochem. Physiol. 2009, 154A, S21–S22. [Google Scholar] [CrossRef]
- .Razin, S.V.; Borunova, V.V.; Maksimenko, O.G.; Kantidze, O.L. Cys2His2 zinc finger protein family: Classification, functions, and major members. Biochemistry 2012, 77, 217–226. [Google Scholar] [CrossRef]
- Laity, J.H.; Lee, B.M.; Wright, P.E. Zinc finger proteins: New insights into structural and functional diversity. Curr. Opin. Struct. Biol. 2001, 11, 39–46. [Google Scholar] [CrossRef]
- Cassandri, M.; Smirnov, A.; Novelli, F.; Pitolli, C.; Agostini, M.; Malewicz, M.; Melino, G.; Raschellà, G. Zinc-finger proteins in health and disease. Cell Death Discov. 2017, 3, 17071. [Google Scholar] [CrossRef]
- Xu, L.; Yang, L.; Hashimoto, K.; Anderson, M.; Kohlhagen, G.; Pommier, Y.; D’Arpa, P. Characterization of BTBD1 and BTBD2, two similar BTB-domain-containing Kelch-like proteins that interact with Topoisomerase I. BMC Genom. 2002, 3, 1. [Google Scholar] [CrossRef]
- Bardwell, V.J.; Treisman, R. The POZ domain: A conserved protein–protein interaction motif. Genes Dev. 1994, 8, 1664–1677. [Google Scholar] [CrossRef]
- Qi, J.; Zhang, X.; Zhang, H.K.; Yang, H.M.; Zhou, Y.B.; Han, Z.G. ZBTB34, a novel human BTB/POZ zinc finger protein, is a potential transcriptional repressor. Mol. Cell. Biochem. 2006, 290, 159–167. [Google Scholar] [CrossRef]
- Wen, Y.; Nguyen, D.; Li, Y.; Lai, Z.C. The N-terminal BTB/POZ domain and C- terminal sequences are essential for Tramtrack69 to specify cell fate in the developing Drosophila eye. Genetics 2000, 156, 195–203. [Google Scholar] [CrossRef]
- Yoshitomi, Y.; Osada, H.; Satake, H.; Kojima, M.; Saito-Takatsuji, H.; Ikeda, T.; Yoshitake, Y.; Ishigaki, Y.; Kubo, E.; Sasaki, H.; et al. Ultraviolet B-induced Otx2 expression in lens epithelial cells promotes epithelial-mesenchymal transition. Biol. Open 2019, 8, bio035691. [Google Scholar] [CrossRef]
- Acampora, D.; Mazan, S.; Lallemand, Y.; Avantaggiato, V.; Maury, M.; Simeone, A.; Brûlet, P. Forebrain and midbrain regions are deleted in Otx2−/− mutants due to a defective anterior neuroectoderm specification during gastrulation. Development 1995, 121, 3279–3290. [Google Scholar] [CrossRef]
- Wyatt, A.; Bakrania, P.; Bunyan, D.J.; Osborne, R.J.; Crolla, J.A.; Salt, A.; Ayuso, C.; Newbury-Ecob, R.; Abou-Rayyah, Y.; Collin, J.R.O.; et al. Novel heterozygous Otx2 mutations and whole gene deletions in anophthalmia, microphthalmia and coloboma. Hum. Mutat. 2008, 29, E278–E283. [Google Scholar] [CrossRef] [PubMed]
- Manuel, M.; Pratt, T.; Liu, M.; Jeffery, G.; Price, D.J. Overexpression of Pax6 results in microphthalmia, retinal dysplasia and defective retinal ganglion cell axon guidance. BMC Dev. Biol. 2008, 8, 59. [Google Scholar] [CrossRef]
- Grindley, J.C.; Hargett, L.K.; Hill, R.E.; Ross, A.; Hogan, B.L. Disruption of PAX6 function in mice homozygous for the Pax6Sey-1Neu mutation produces abnormalities in the early development and regionalization of the diencephalon. Mech. Dev. 1997, 64, 111–126. [Google Scholar] [CrossRef]
- Collinson, J.M.; Quinn, J.C.; Buchanan, M.A.; Kaufman, M.H.; Wedden, S.E.; West, J.D.; Hill, R.E. Primary defects in the lens underlie complex anterior segment abnormalities of the Pax6 heterozygous eye. Proc. Natl. Acad. Sci. USA 2001, 98, 9688–9693. [Google Scholar] [CrossRef]
- Mort, R.L.; Bentley, A.J.; Martin, F.L.; Collinson, J.M.; Douvaras, P.; Hill, R.E.; Morley, S.D.; Fullwood, N.J.; West, J.D. Effects of aberrant Pax6 gene dosage on mouse corneal pathophysiology and corneal epithelial homeostasis. PLoS ONE 2011, 6, e28895. [Google Scholar] [CrossRef]
- Yang, J.S.; Lee, S.Y.; Gao, M.; Bourgoin, S.; Randazzo, P.A.; Premont, R.T.; Hsu, V.W. ARFGAP1 promotes the formation of COPI vesicles, suggesting function as a component of the coat. J. Cell Biol. 2002, 159, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Soundararajan, M.; Yang, X.; Elkins, J.M.; Sobott, F.; Doyle, D.A. The centaurin gamma-1 GTPase-like domain functions as an NTPase. Biochem. J. 2007, 401, 679–688. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zuk, M.; Prescha, A.; Kepczynski, J.; Szopa, J. ADP ribosylation factor regulates metabolism and antioxidant capacity of transgenic potato tubers. J. Agric. Food Chem. 2003, 51, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Minglin, L.; Yuxiu, Z.; Tuanyao, C. Identification of genes up-regulated in response to Cd exposure in Brassica juncea L. Gene 2005, 363, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Voronin, D.A.; Kiseleva, E.V. Functional role of proteins containing ankyrin repeats. Tsitologiia 2007, 49, 989–999. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.Y.; Lu, Z.W.; Sun, Y.; Fang, Z.W.; Chen, J.; Zhou, Y.B.; Chen, M.; Ma, Y.Z.; Xu, Z.S.; Min, D.H. The Ankyrin-Repeat Gene GmANK114 Confers Drought and Salt Tolerance in Arabidopsis and Soybean. Front. Plant Sci. 2020, 11, 584167. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Chen, C.; Wei, X.; Li, X.; Luo, G.; Zhang, J.; Bin, J.; Huang, X.; Cao, S.; Li, G.; et al. Overexpression of ankyrin repeat domain 1 enhances cardiomyocyte apoptosis by promoting p53 activation and mitochondrial dysfunction in rodents. Clin. Sci. 2015, 128, 665–678. [Google Scholar] [CrossRef] [PubMed]
- Catterall, W.A.; Goldin, A.L.; Waxman, S.G. International Union of Pharmacology. XLVII. Nomenclature and structure–function relationships of voltage-gated sodium channels. Pharmacol. Rev. 2005, 57, 397–409. [Google Scholar] [CrossRef] [PubMed]
- Wada, A. Roles of voltage-dependent sodium channels in neuronal development, pain, and neurodegeneration. J. Pharmacol. Sci. 2006, 102, 253–268. [Google Scholar] [CrossRef]
- Lévesque, M.; Martineau, C.; Jumarie, C.; Moreau, R. Characterization of cadmium uptake and cytotoxicity in human osteoblast-like MG-63 cells. Toxicol. Appl. Pharmacol. 2008, 15, 308–317. [Google Scholar] [CrossRef]
- Ogawa, T.; Kuwagata, M.; Hori, Y.; Shioda, S. Valproate-induced developmental neurotoxicity is affected by maternal conditions including shipping stress and environmental change during early pregnancy. Toxicol. Lett. 2007, 174, 18–24. [Google Scholar] [CrossRef]
- Evenseth, L.; Gabrielsen, M.; Sylte, I. The GABABReceptor—Structure, Ligand Binding and Drug Development. Molecules 2020, 25, 3093. [Google Scholar] [CrossRef]
- Kornau, H.C. GABA(B) receptors and synaptic modulation. Cell Tissue Res. 2006, 326, 517–533. [Google Scholar] [CrossRef]
- Pranzatelli, M.R.; Nadi, N.S. Mechanism of action of antiepileptic and antimyoclonic drugs. Adv. Neurol. 1995, 67, 329–360. [Google Scholar]
- Dengler, T.J.; Hoffmann, J.C.; Knolle, P.; Albert-Wolf, M.; Roux, M.; Wallich, R.; Meuer, S.C. Structural and functional epitopes of the human adhesion receptor CD58 (LFA-3). Eur. J. Immunol. 1992, 22, 2809–2817. [Google Scholar] [CrossRef]
- Binder, C.; Cvetkovski, F.; Sellberg, F.; Berg, S.; Paternina Visbal, H.; Sachs, D.H.; Berglund, E.; Berglund, D. CD2 Immunobiology. Front. Immunol. 2020, 9, 1090. [Google Scholar] [CrossRef] [PubMed]
- Shao, T.; Shi, W.; Zheng, J.Y.; Xu, X.X.; Lin, A.F.; Xiang, L.X.; Shao, J.Z. Costimulatory Function of Cd58/Cd2 Interaction in Adaptive Humoral Immunity in a Zebrafish Model. Front. Immunol. 2018, 9, 1204. [Google Scholar] [CrossRef] [PubMed]
- Kimura, T.; Kambe, T. The Functions of Metallothionein and ZIP and ZnT Transporters: An Overview and Perspective. Int. J. Mol. Sci. 2016, 17, 336. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, D.E.; Stillman, M.J. The “magic numbers” of metallothionein. Metallomics 2011, 3, 444–463. [Google Scholar] [CrossRef]
- Waeytens, A.; De Vos, M.; Laukens, D. Evidence for a potential role of metallothioneins in inflammatory bowel diseases. Mediat. Inflamm. 2009, 2009, 729172. [Google Scholar] [CrossRef]
- Kumari, M.V.; Hiramatsu, M.; Ebadi, M. Free radical scavenging actions of metallothionein isoforms I and II. Free Radic Res. 1998, 29, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Atif, F.; Kaur, M.; Ansari, R.A.; Raisuddin, S. Channa punctata brain metallothionein is a potent scavenger of superoxide radicals and prevents hydroxyl radical-induced in vitro DNA damage. J. Biochem. Mol. Toxicol. 2008, 22, 202–208. [Google Scholar] [CrossRef]
- Carginale, V.; Scudiero, R.; Capasso, C.; Capasso, A.; Kille, P.; di Prisco, G.; Parisi, E. Cadmium-induced differential accumulation of metallothionein isoforms in the Antarctic icefish which exhibits no basal protein but high endogenous mRNA levels. Biochem. J. 1998, 332, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Samson, S.L.; Gedamu, L. Molecular analyses of metallothionein gene regulation. Prog. Nucleic Acid Res. Mol. Biol. 1998, 59, 257–288. [Google Scholar] [CrossRef] [PubMed]
- Trinchella, F.; Riggio, M.; Filosa, S.; Volpe, M.G.; Parisi, E.; Scudiero, R. Cadmium distribution and metallothionein expression in lizard tissues following acute and chronic cadmium intoxication. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2006, 144, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, S. Positive and negative regulators of the metallothionein gene (review). Mol. Med. Rep. 2015, 12, 795–799. [Google Scholar] [CrossRef]
- Klaassen, C.D.; Liu, J.; Diwan, B.A. Metallothionein protection of cadmium toxicity. Toxicol. Appl. Pharmacol. 2009, 238, 215–220. [Google Scholar] [CrossRef]
- Günes, C.; Heuchel, R.; Georgiev, O.; Müller, K.H.; Lichtlen, P.; Blüthmann, H.; Marino, S.; Aguzzi, A.; Schaffner, W. Embryonic lethality and liver degeneration in mice lacking the metal-responsive transcriptional activator MTF-1. EMBO J. 1998, 17, 2846–2854. [Google Scholar] [CrossRef]
- Durliat, M.; Muller, J.P.; André, M.; Wegnez, M. Expression of the Xenopus laevis metallothionein gene during ontogeny. Int. J. Dev. Biol. 1999, 43, 575–578. [Google Scholar]
- Romero, M.B.; Polizzi, P.; Chiodi, L.; Das, K.; Gerpe, M. The role of metallothioneins, selenium and transfer to offspring in mercury detoxification in Franciscana dolphins (Pontoporia blainvillei). Mar. Pollut. Bull. 2016, 109, 650–654. [Google Scholar] [CrossRef]
- Riggio, M.; Trinchella, F.; Parisi, E.; Filosa, S.; Scudiero, R. Accumulation of zinc, copper and metallothionein mRNA in lizard ovary proceeds without a concomitant increase in metallothionein content. Mol. Repr. Dev. 2003, 66, 347–382. [Google Scholar] [CrossRef]
- Simoniello, P.; Motta, C.M.; Scudiero, R.; Trinchella, F.; Filosa, S. Spatiotemporal changes in metallothionein gene expression during embryogenesis in the wall lizard Podarcis sicula. J. Exp. Zool. 2010, 313A, 410–420. [Google Scholar] [CrossRef]
- Borrelli, L.; De Stasio, R.; Motta, C.M.; Parisi, E.; Filosa, S. Seasonal-dependent effect of temperature on the response of adenylate cyclase to FSH stimulation in the oviparous lizard, Podarcis sicula. J. Endocrinol. 2000, 167, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Scudiero, R.; Esposito, M.G.; Simoniello, P. Tolerance to Thermal Stress in Lizard Embryos. In Lizards: Thermal Ecology, Genetic Diversity and Functional Role in Ecosystems; Kiernan, M.P., Ed.; Nova Science Publishers: Happauge, NY, USA, 2014; pp. 29–44. [Google Scholar]
- Simoniello, P.; Esposito, M.G.; Trinchella, F.; Motta, C.M.; Scudiero, R. Alterations in brain morphology and HSP70 expression in lizard embryos exposed to thermal stress. CR Biol. 2016, 339, 380–390. [Google Scholar] [CrossRef]
- Dayananda, B.; Webb, J.K. Incubation under climate warming affects learning ability and survival in hatchling lizards. Biol. Lett. 2017, 13, 20170002. [Google Scholar] [CrossRef]
- Feiner, N.; Rago, A.; While, G.M.; Uller, T. Developmental plasticity in reptiles: Insights from temperature-dependent gene expression in wall lizard embryos. J. Exp. Zool. A Ecol. Integr. Physiol. 2018, 329, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Scudiero, R.; Verderame, M.; Motta, C.M.; Migliaccio, V.; Simoniello, P. HSP70 localization in Podarcis siculus embryos under natural thermal regime and following a non-lethal cold shock. CR Biol. 2019, 342, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Tam, Y.; Heikkila, J.J. Identification of members of the hsp30 small heat shock protein family and characterization of their developmental regulation in heat-shocked Xenopus laevis embryos. Dev. Genet. 1995, 17, 331–339. [Google Scholar] [CrossRef]
- Heikkila, J.J. The expression and function of hsp30-like small heat shock protein genes in amphibians, birds, fish, and reptiles. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2017, 203, 179–192. [Google Scholar] [CrossRef]
- Yang, H.; Pesavento, J.J.; Starnes, T.W.; Cryderman, D.E.; Wallrath, L.L.; Kelleher, N.L.; Mizzen, C.A. Preferential dimethylation of histone H4 lysine 20 by Suv4-20. J. Biol. Chem. 2008, 283, 12085–12092. [Google Scholar] [CrossRef]
- Yizhar-Barnea, O.; Valensisi, C.; Jayavelu, N.D.; Kishore, K.; Andrus, C.; Koffler-Brill, T.; Ushakov, K.; Perl, K.; Noy, Y.; Bhonker, Y.; et al. DNA methylation dynamics during embryonic development and postnatal maturation of the mouse auditory sensory epithelium. Sci. Rep. 2018, 8, 17348. [Google Scholar] [CrossRef]
- Fan, L.H.; Wang, Z.B.; Li, Q.N.; Meng, T.G.; Dong, M.Z.; Hou, Y.; Ouyang, Y.C.; Schatten, H.; Sun, Q.Y. Absence of mitochondrial DNA methylation in mouse oocyte maturation, aging and early embryo development. Biochem. Biophys. Res. Commun. 2019, 513, 912–918. [Google Scholar] [CrossRef]
- Klutstein, M.; Nejman, D.; Greenfield, R.; Cedar, H. DNA Methylation in Cancer and Aging. Cancer Res. 2016, 76, 3446–3450. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, S.; Schotta, G.; Sørensen, C.S. Histone H4 lysine 20 methylation: Key player in epigenetic regulation of genomic integrity. Nucleic Acids Res. 2013, 41, 2797–2806. [Google Scholar] [CrossRef] [PubMed]
- Wickramasekara, R.N.; Stessman, H.A.F. Histone 4 Lysine 20 Methylation: A Case for Neurodevelopmental Disease. Biology 2019, 8, 11. [Google Scholar] [CrossRef] [PubMed]
- Paredes, U.; Radersma, R.; Cannell, N.; While, G.M.; Uller, T. Low Incubation Temperature Induces DNA Hypomethylation in Lizard Brains. J. Exp. Zool. A Ecol. Genet. Physiol. 2016, 325, 390–395. [Google Scholar] [CrossRef] [PubMed]
| Gene | Gene Symbol | Biological Pathway | Cd-Effect |
|---|---|---|---|
| Basic transcription factor 3 | BTF3 | Transcription regulation | Inhibition |
| Three-hydroxy-3-methylglutaryl coenzyme A reductase | HMGR | Cholesterol biosynthesis | inhibition |
| Fizzy and cell division cycle 20 related 1 | Fzr1 | Control of cell proliferation | inhibition |
| Rearranged L-myc fusion | RLF | Zinc finger transcription factor | Induction |
| Topoisomerase 1-binding protein with a broad-complex, Tramtrack and bric a brac/Pox virus and Zinc finger | Topo1-bp, BTB/POZ domain | Transcription regulation—protein–protein interactions | Induction |
| Ortho-denticle protein homolog 2 | Otx2 | Transcription factor—Brain and eye development | Induction |
| Paired Box 6 | Pax6 | Transcription factor—Eye development | Induction |
| Development and differentiation-enhancing factor 1 | DDEF1 | ADP-ribosylation factor—Vesicular trafficking | Induction |
| Voltage-gated sodium channel | VGSC | Sensory transduction and synaptic plasticity | Induction |
| γ-aminobutyric acid type B receptor | GABABR | G protein-coupled inhibitory receptors | Induction |
| Lymphocyte function associated antigen 3 | CD58 | Production of inflammatory cytokine | Induction |
| Metallothionein | MT | Heavy metals homeostasis and detoxification | Induction |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scudiero, R.; Motta, C.M.; Simoniello, P. Impact of Environmental Stressors on Gene Expression in the Embryo of the Italian Wall Lizard. Appl. Sci. 2021, 11, 4723. https://doi.org/10.3390/app11114723
Scudiero R, Motta CM, Simoniello P. Impact of Environmental Stressors on Gene Expression in the Embryo of the Italian Wall Lizard. Applied Sciences. 2021; 11(11):4723. https://doi.org/10.3390/app11114723
Chicago/Turabian StyleScudiero, Rosaria, Chiara Maria Motta, and Palma Simoniello. 2021. "Impact of Environmental Stressors on Gene Expression in the Embryo of the Italian Wall Lizard" Applied Sciences 11, no. 11: 4723. https://doi.org/10.3390/app11114723
APA StyleScudiero, R., Motta, C. M., & Simoniello, P. (2021). Impact of Environmental Stressors on Gene Expression in the Embryo of the Italian Wall Lizard. Applied Sciences, 11(11), 4723. https://doi.org/10.3390/app11114723








