1. Introduction
Neurons are viewed as the basic cells that process and transmit information. Trees and neurons share similar structure and neurotransmitter-like substances [
1,
2]. No evidence for structures such as neurons, synapses, or a brain has been found inside plants [
3]. Signal processing by trees has been known since Salix sitchensis trees were found to be sensitive to and respond to signals generated by trees attacked by tent caterpillars and webworms [
4]. Interplant communication can occur from leaves of one species of plant to leaves of another species to activate the expression of defensive genes [
5]. N-(17-hydroxylinolenoyl)-L-glutamine was found to be a key compound in communication between leaves of corn seedlings eaten by beet armyworms and in calls for help to the enemy of their enemy, parasitic wasps [
6]. Furthermore, species in ecological communities show the ability to build complex webs of communication based on plant fungus [
7,
8].
Communication of plants through airborne signals has been analyzed [
9,
10]. In addition, plants have memory defined by reversible changes in behavior (plasticity) that has been altered by experience [
11]. Previous work has shown changes in neural substrates of memory [
12] based on predictions of rewarding events using adaptive optimization control [
13]. However, there is a lack of knowledge about the mechanisms that lead to plant communication that are beneficial for both plant emitter and receiver [
10].
A roadmap towards a functional paradigm for learning and memory in plants has been proposed [
14]. From the seven requirements proposed to substantiate plant learning and memory, one aspect included information processing which was in initial stages and needed additional clarification. Another requirement aspect proposed was independent internal representation, which is currently theoretical and needs further investigation. Here, instead of viewing each plant as an independent mechanism, we view the forest as a system that is processing information and has representation of the knowledge being processed.
We show how viewing a forest as a network rather than as separate tree entities can help explain the benefit of communication as resource optimization. It is suggested that each tree’s goal of survival is directed by the forest trying to optimize its resources, which in our case is access to sunlight. Comparison of a forest network to a neural network shows interesting similarities in both the physical structure and some of the functional properties of both networks.
We define Knowledge Processing as the analysis of information intended to try to optimize the use of available resources. Animals are viewed as having the ability to process knowledge at different levels in order to use their available resources. To bypass the philosophical argument of whether animals or trees “think”, we analyze the use of resources needed to survive. In the case of animals, neuron brain activity allows the processing of the knowledge of available resources. Since trees lack neurons but can build a communication network [
15], their use of the resources required for survival, including light, nutrients, and water, can be analyzed. We chose to analyze the communication between trees in a forest that tries to optimize the use of light.
The analysis is inspired by epilepsy resection, the action performed on neurons when they exhibit high neuron activity. In epilepsy resection, a part of the brain, a set of neurons, is removed to allow the neurons residing in the surrounding area to become less active and resume normal neuron activity [
16]. Here we try to imitate the idea behind this procedure and look at forests where a section of trees has been removed. The idea is that resection causes nearby neurons (trees) to become less active, or rather, less efficient at processing knowledge. This means “resume normal activity” in hyper-acting neurons, but “falls down to abnormal activity” in trees. We analyze trees on both sides of an area in the forest which was “surgically” removed by humans—a trail. We look at the behavior of trees and analyze the change of the use of light as the distance from the trails increases.
When the forest canopy increases near the resection, it increases logarithmically as the distance from the resection increases. Some exceptions to this phenomenon showed forest structure similar to the neuron semi-sphere shape proliferate structure occurring in brain tumors. The results show that neuron-like relations can occur in a forest knowledge processing system.
The similarity between human neurons transmitting knowledge and forest trees transmitting knowledge can be seen from the network structure and the network behavior. The ability to process the knowledge regarding the available resources while optimizing their use appears to be similar. Furthermore, each processing unit, neuron or tree, seems to prioritize the resource needs of the network rather than its own needs.
The importance of the results is in the ability to analyze neuron-like behavior in an environment which processes knowledge much slower than human neurons. The forest environment, representing nature-inspired knowledge processing, has similar characteristics to brain neuron processing but is easier to manipulate and control.
2. Methods
ArcGIS [
17], a spatial analysis tool, was used for canopy coverage and distance from trail calculations. The set of sampled points representing changes in landscape was used to observe the continuity and variability of the data across a surface using interpolation tools. Deterministic and geo-statistical techniques were used for interpolation. Inverse Distance Weight (IDW) and Spline are deterministic techniques based on the extent of similarity of cells. Kriging is a geo-statistical interpolation technique.
IDW is based on a linear weighted combination of a sample of points. The weight is assigned as a descending function when the distance of the input point to the cell location is growing.
is the predicted value for given location
.
is the number of measured points near the predicted location.
are weights assigned to each measured point used. The weights value decreases as the distance increases.
is the measured value at location
.
As distance increases, the weight is reduced by a factor of p. is the distance between prediction location and each measured location . The optimal value is determined by minimizing the root-mean square prediction error (RMSPE). The RMSPE is the statistic that is calculated from cross-validation where each measured point is removed and compared to the predicted value of the location. The IDW aims at identifying the value that produces the minimum RMSPE.
Kriging performs statistical interpolation by fitting a function to a specified number of points with a specified radius to determine the output value for each location. Kriging provides information on the spatial autocorrelation of the dataset, the forest canopy coverage. We use the spatial resolution of a 30 m
2 cell covered by tree canopy. Fitting and loss of tree canopy coverage values were produced using a Random Forests regression. It assumes the distance between sample points reflects a spatial correlation that explains the variation in the surface.
where
is the value for location
,
is the deterministic trend, and
are the random correlated errors.
representing the second-order polynomial trend surface linear regression on x and y coordinates.
Spline estimates values by minimizing the overall surface curvature. This is based on a smooth surface that passes through the input points.
3. Results
Forest canopy tree coverage degradation and regeneration in areas surrounding trails were analyzed in 19 locations of national forests across the US. We sampled 2802 trails in these forests. The total number of trails sampled in each forest ranged from 6 to 825 per forest.
The tree canopy coverage of each trail is based on data from the USGS/MRLC National Land Cover Database [
18]. Tree canopy coverage is available for 2011 (13.2 GB) for the entire US. Each spatial resolution represents the proportion of a 30m
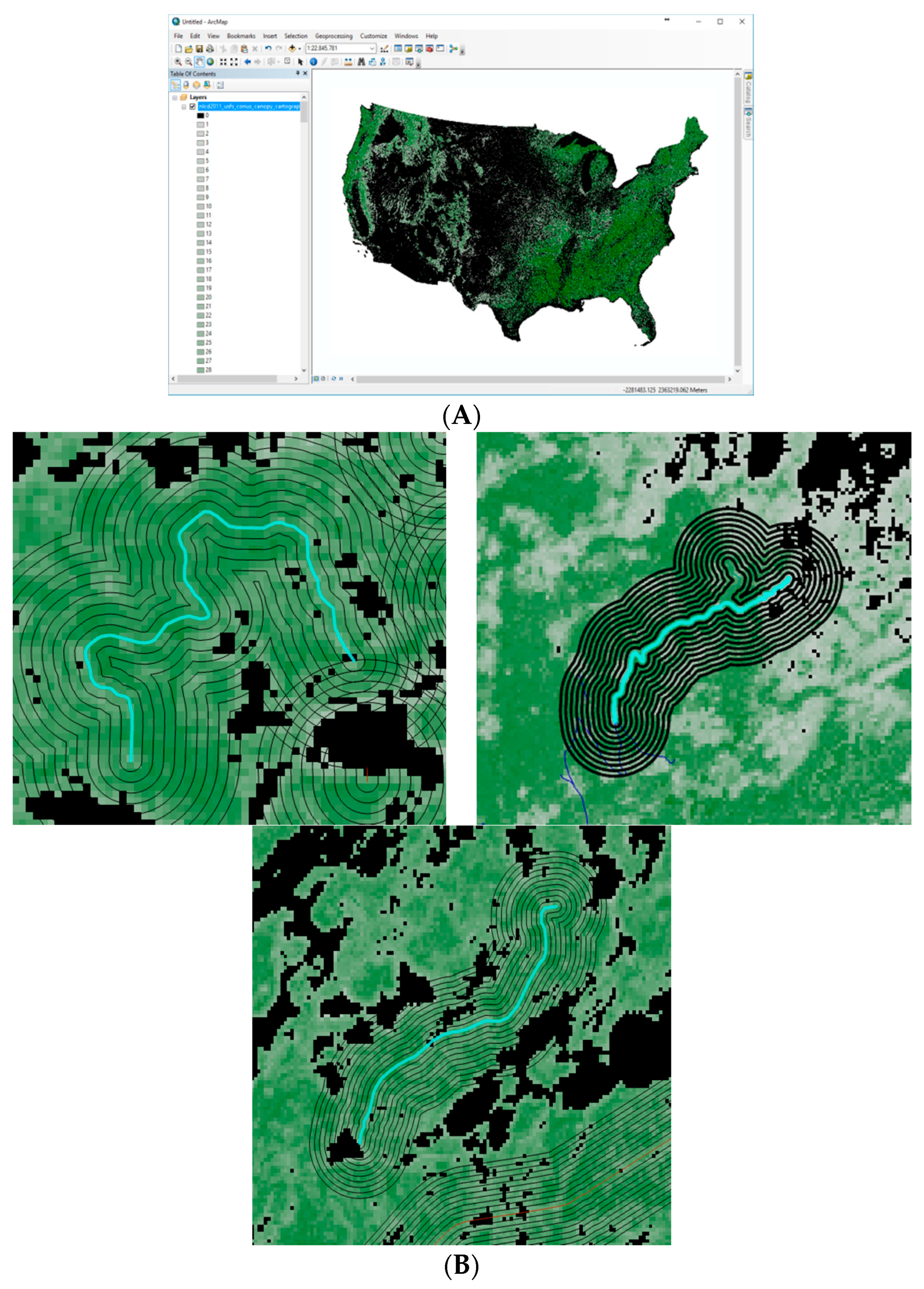
2 cell covered by tree canopy. Tree canopy coverage values were produced using a Random Forests regression algorithm. ArcMap was used as a Geographical Information System (GIS) to analyze the different information layers, placing the trails on top of the tree canopy coverage (
Figure 1A). Next, we analyzed the canopy coverage change range from each trail to the distance of 450 m from the trail (
Figure 1B). Around each trail a series of rings was created. Each ring represents an additional growing distance of 45 m from the center of the trail. The average canopy coverage at a fixed time was analyzed for each ring.
The total number of trails analyzed that exhibited at least 10% change in canopy tree coverage within the 450 m proximal was 323. The number of trails with the 10% change varied in each national forest from 1 to 96. Trails with high canopy change were selected to analyze the network structure after resection.
Table 1 presents the sampled trail distribution in forests across the US. The overall results show that 86.51% of the cases analyzed present increased canopy coverage as the distance from the resection increases. The results can be viewed as knowledge processing in forests based on the consistency between tree network resection and canopy coverage as distances of up to 450 m. While influence on the nearby trees can be attributed to people walking on the trails, trees at a further distance from the trail should not be influenced by the trail themselves, and this absence of a direct influence was confirmed in multiple locations. The attempt of the forest to optimize the use of light resources through canopy coverage can be viewed as knowledge processing using the trees as a network.
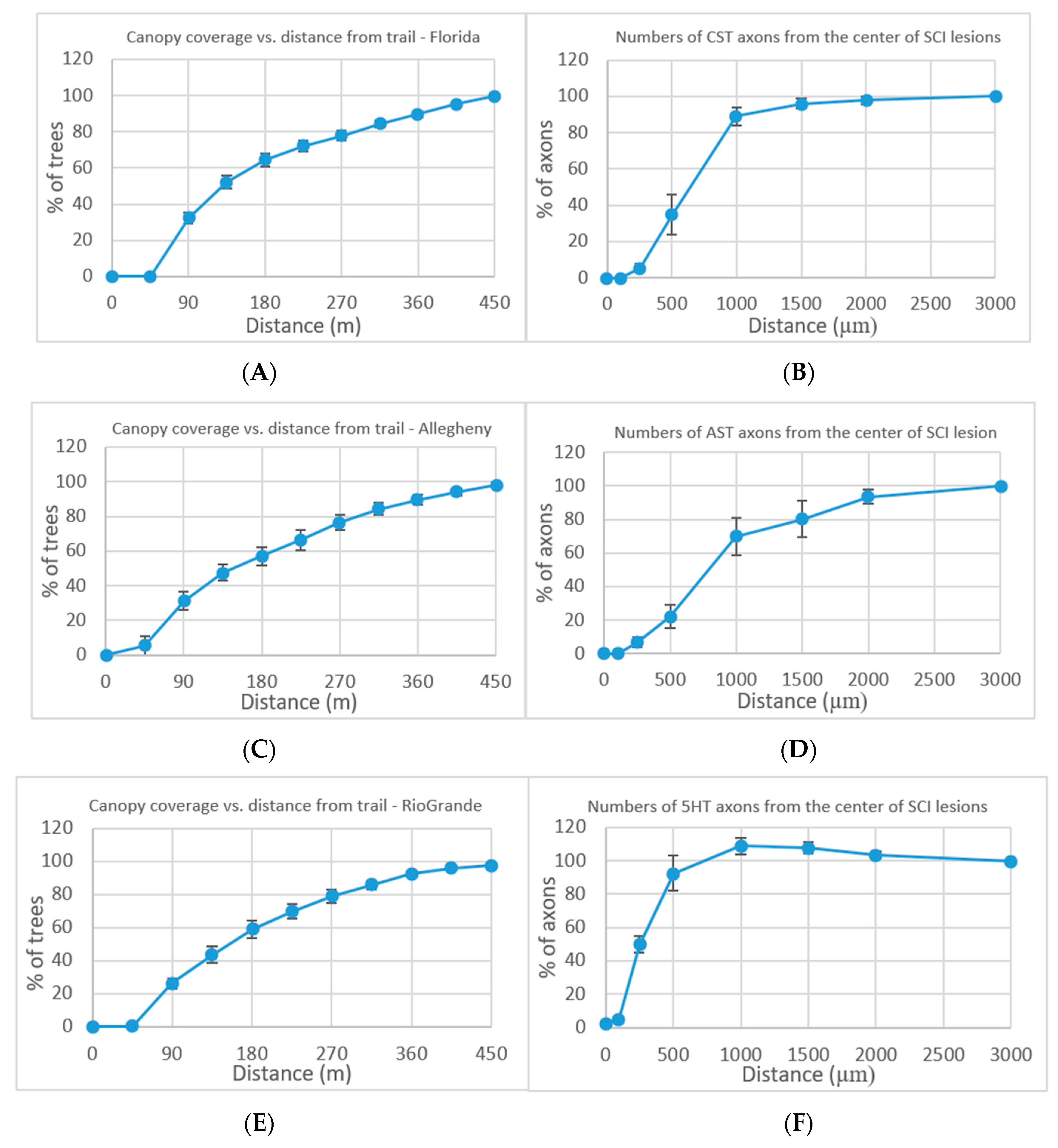
Forest resection by trails is analyzed in multiple locations across the US. From all the occurrences of at least 10% increase in canopy from the trail, examples of three locations are presented (
Figure 2A,C,E): Florida, Allegheny, and the Rio Grande National Forests. Additional locations appear in the
Supplementary Information (
Figures S1 and S2). The canopy coverage was evaluated at an additional growing distance of 45 m surrounding the trail. The mean value ± standard error of the mean (s.e.m.) is displayed. The results show dieback away from the trail center which continues to a distance of 450 m.
Forest resection by trails was compared to neuron resection. The neuronal comparison values are taken from Anderson et al. [
19], who performed in their research on severe crush spinal cord injuries (SCI) in wild-type mice. Following the severe crush SCI, dense astrocytic scars were formed in the mice over a period of two weeks and remained until eight weeks. The consequence of the astrocyte scar formation on axon regeneration was analyzed by three evaluation measurements: descending corticospinal tract (CST), ascending sensory tract (AST), and descending serotonergic (5HT) tract. As a result of the severe SCI in the mice, transected CST, AST, and 5HT axons exhibit dieback away from the lesion center (
Figure 2B,D,F).
The results of the canopy tree coverage as the distance from the trail extends compared to the results of the neuron axon coverage as the distance from the lesion center extends show a high level of similarity. The expected results of a set of trees acting independently, attempting to maximize their canopy coverage, would yield different results from a growing logarithmic graph consistently appearing in all locations. We can infer from the results that the trees, after resection, attempt to form two independent entities, each trying to perform network knowledge processing to optimize their light resource usage. The alternative explanation of damage by trail travelers which expands past the trail area on both sides would not explain the effect reaching a distance of 450 m on both sides of each trail in all the cases described. A possible explanation is the communication of the tree network trying to recover from the trail resection while reformatting the tree canopy coverage in an attempt to maximize the light coverage as a network of trees.
The cases in which the forests did not exhibit an increase in the canopy coverage as the distance from the trails increased (
Table 1, 13.49% of the cases) were analyzed manually for each case. Some of the cases had multiple reasons. The most common reason for the inconsistent increase included large bodies of water such as lakes or rivers within the vicinity of the trails (25 cases). Other reasons included manmade forest-empty areas used for agriculture, residences, or roads (nine cases). Another reason that explained the inconsistency was a sharp geographical drop in height, which can be defined as a mountain cliff, which is another unfavorable environment for tree canopies (four cases).
Past studies on tree regeneration in felling and natural tree fall disturbances showed similar results appearing in logging areas [
20]. The research found that forest gap area and canopy openness were positively related in both logging gap areas and natural gap areas. The results stated that the lack of response to canopy openness in natural gaps was likely a result of relatively low levels of understory and substrate disturbance coupled with the absence of large natural gaps with high canopy openness. These results show that direct forest gap due to logging does effect the canopy. However, our work shows a possible explanation for both the increased damage and the regrowth pattern in both the natural and logging gaps.
Other work on areas of disturbed and residual stand damage following logging in a Bolivian tropical forest [
21], and damage and carbon export by reduced-impact logging in southern Amazonia [
22], also showed logging as an important factor in impacting canopy openness—which is similar to our work and supports our results. An assessment of canopy damage and ground disturbance in skid trails and tree-fall gaps was performed. In both of these cases, the emphasis in the analysis was on directly impacted trees. Our results show the damage can be far deeper than seen in these previous works and the process of recovery of the forest is similar to the recovery of a neural network from an error value. In the neural network the process is known as readjusting the weights of the neural network.
4. Discussion
An additional interesting increase in canopy coverage that is inconsistent with the distance from the trail resection is due to mountain rock or land formation (3 cases). The tree formation in these cases seems to be concentrated in specific spherical areas, with few trees in the surrounding circumference. For a forest in growth distress, the land formation limiting forest growth can be compared to brain cancer (
Figure 3). The trees form a spherical canopy growth pattern (
Figure 3A–E), which displays some similarities to neurons in a brain tumor [
23,
24].
We show that when resection of forests occurs, each part of the forest tries to optimize its light resources, measured by canopy coverage independently. Therefore, the forests can be viewed as a knowledge processing network as opposed to just a communication network. The trees in the forest attempt to maximize their use of resources; in our case, only the resource of light was analyzed. The knowledge processing network of trees in the forest resembles the activity of neurons working in the brain system. Other resource usage of trees can be analyzed to examine whether the use of nutrients and water also involves a neural network like pattern. Furthermore, knowledge processing without neurons might appear in other systems in nature.
5. Conclusions
Many recent artificial intelligence models rely more on neural networks. These models have been proven to be more successful than some of the other directions of artificial intelligence. The success of the neural network models in the field of computing leads to the question of whether knowledge processing is based on neural networks. Another issue that arises is how common neural network knowledge processing is in nature-inspired systems.
The results show that trees use mechanisms which could be interpreted as similar to neural network knowledge processing. The communication between neurons in the brain is similar to the communication of knowledge in forests when trying to optimize a decision. In our study, the results show the optimization of maximizing the canopy over the entire forest as a process of sharing knowledge between all trees. Each interference in one part of the network is transmitted, and the entire network attempts to overcome the loss and reoptimize the use of resources, in this case sunlight. Similarly, neurons in the brain can be viewed as trying to optimize the use of available resources.
The results can lead to the philosophical question of how unique knowledge processing in the human brain is if similar knowledge processing happens to commonly appear in nature. The use of models which can be defined as equivalent to neural network processing by trees might be just one example of nature-inspired knowledge processing behavior. Other neural network knowledge processing models might be common, and therefore the knowledge processing which we call “thinking” might be widely used by processing mechanisms other than human ones.