Method for Quantitative Broadband Diffuse Optical Spectroscopy of Tumor-Like Inclusions
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
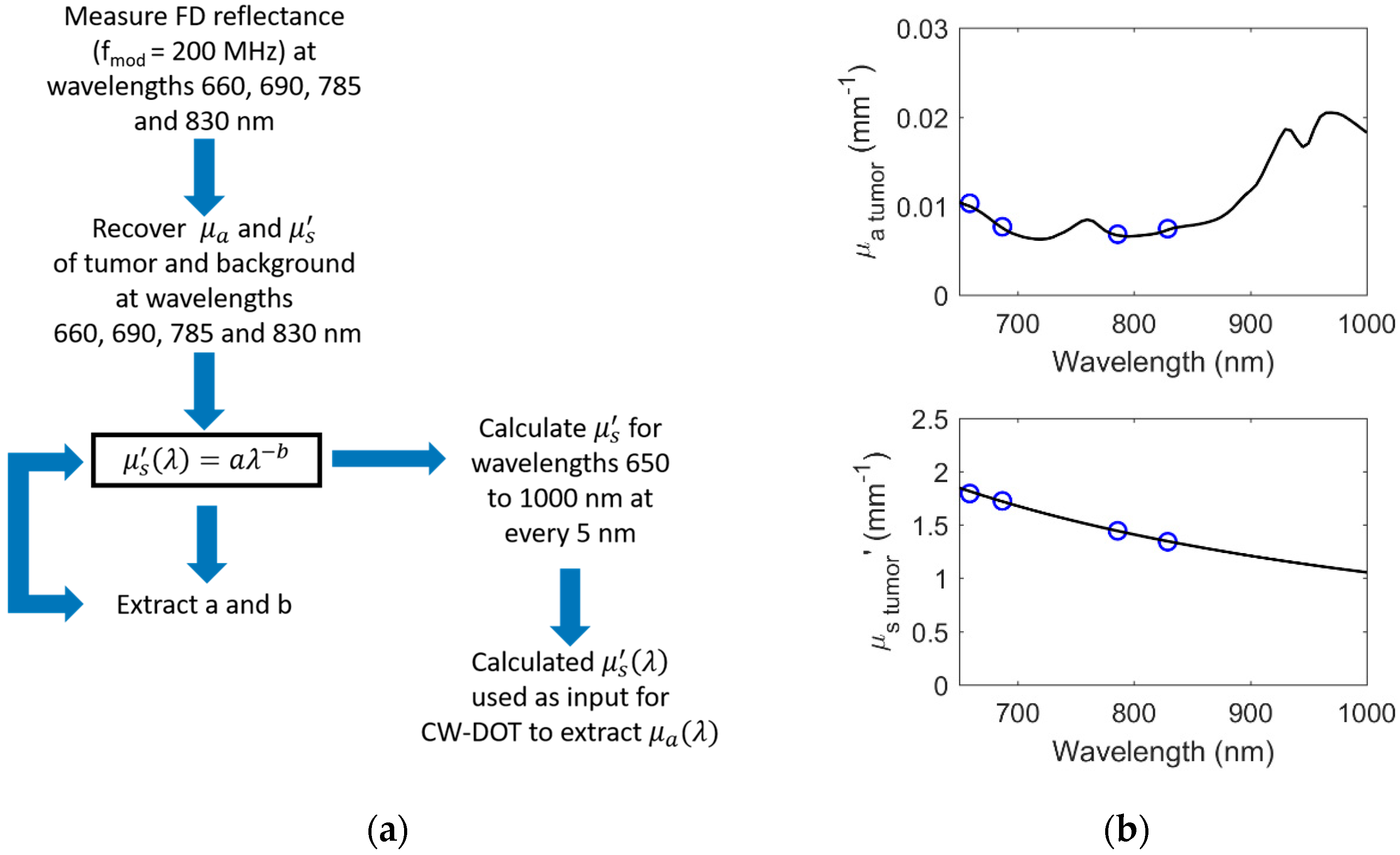
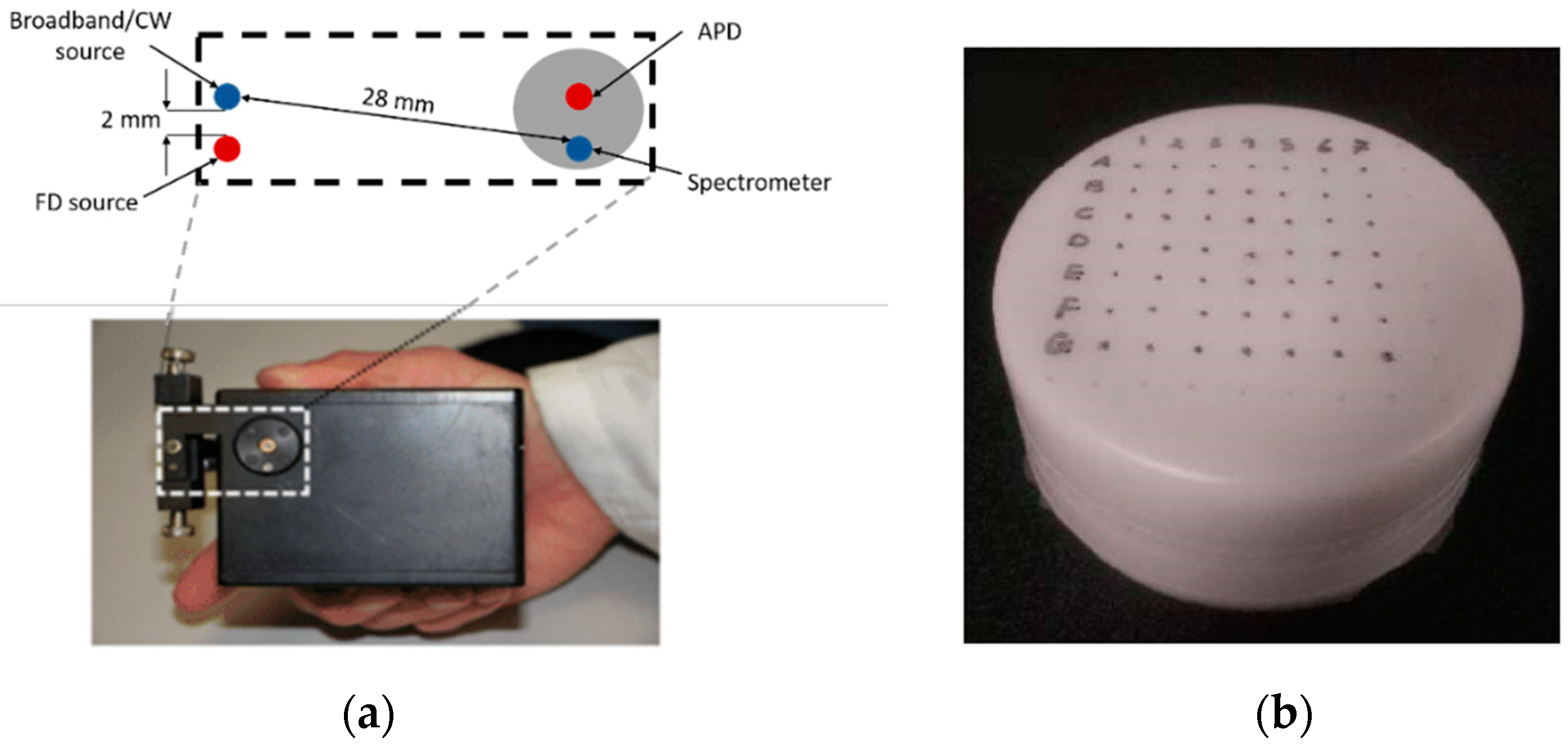
2.1. Hyperspectral Hybrid Frequency-Domain (FD) and Continuous-Wave (CW) Diffuse Optical Imaging DOI
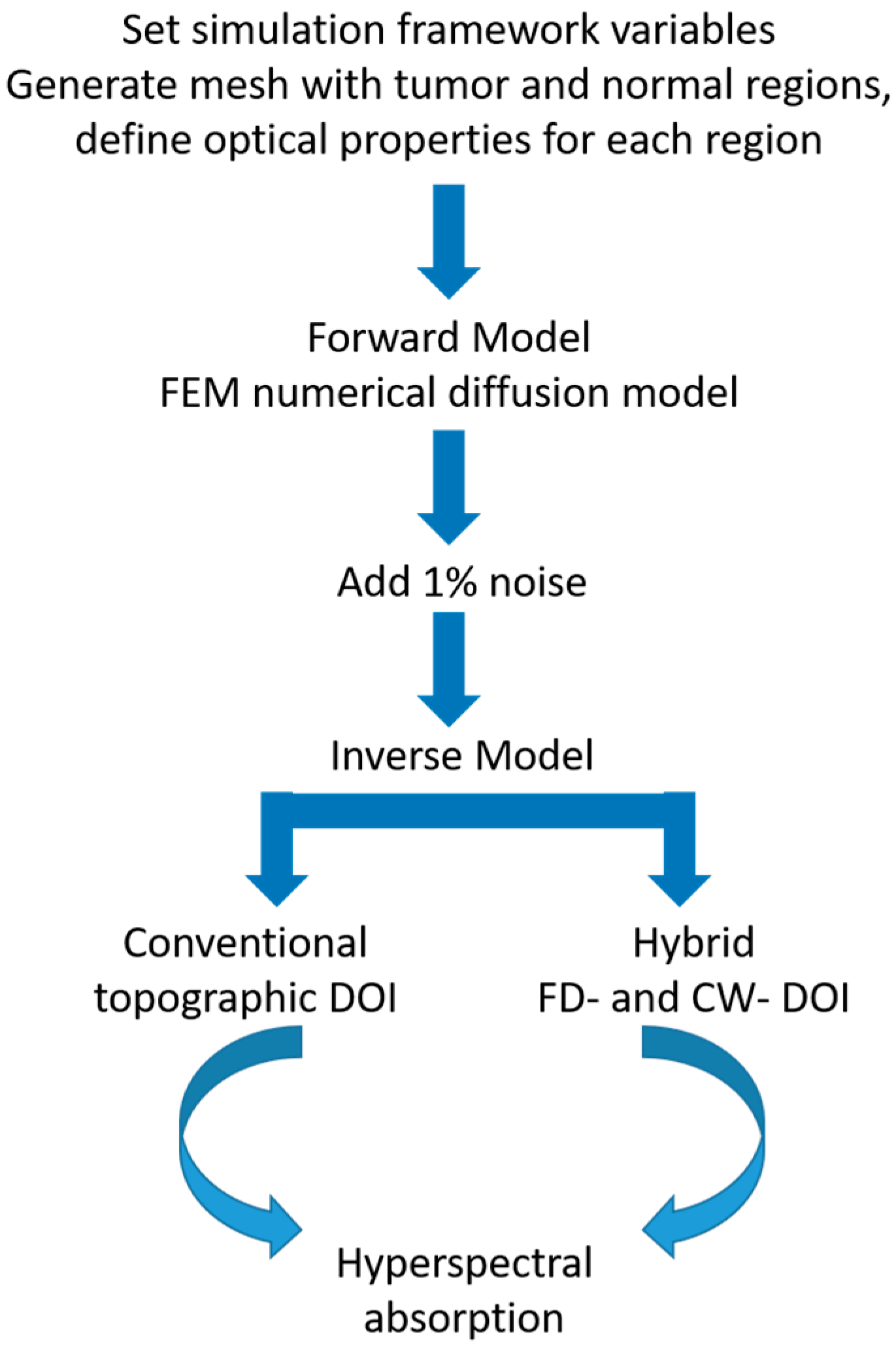
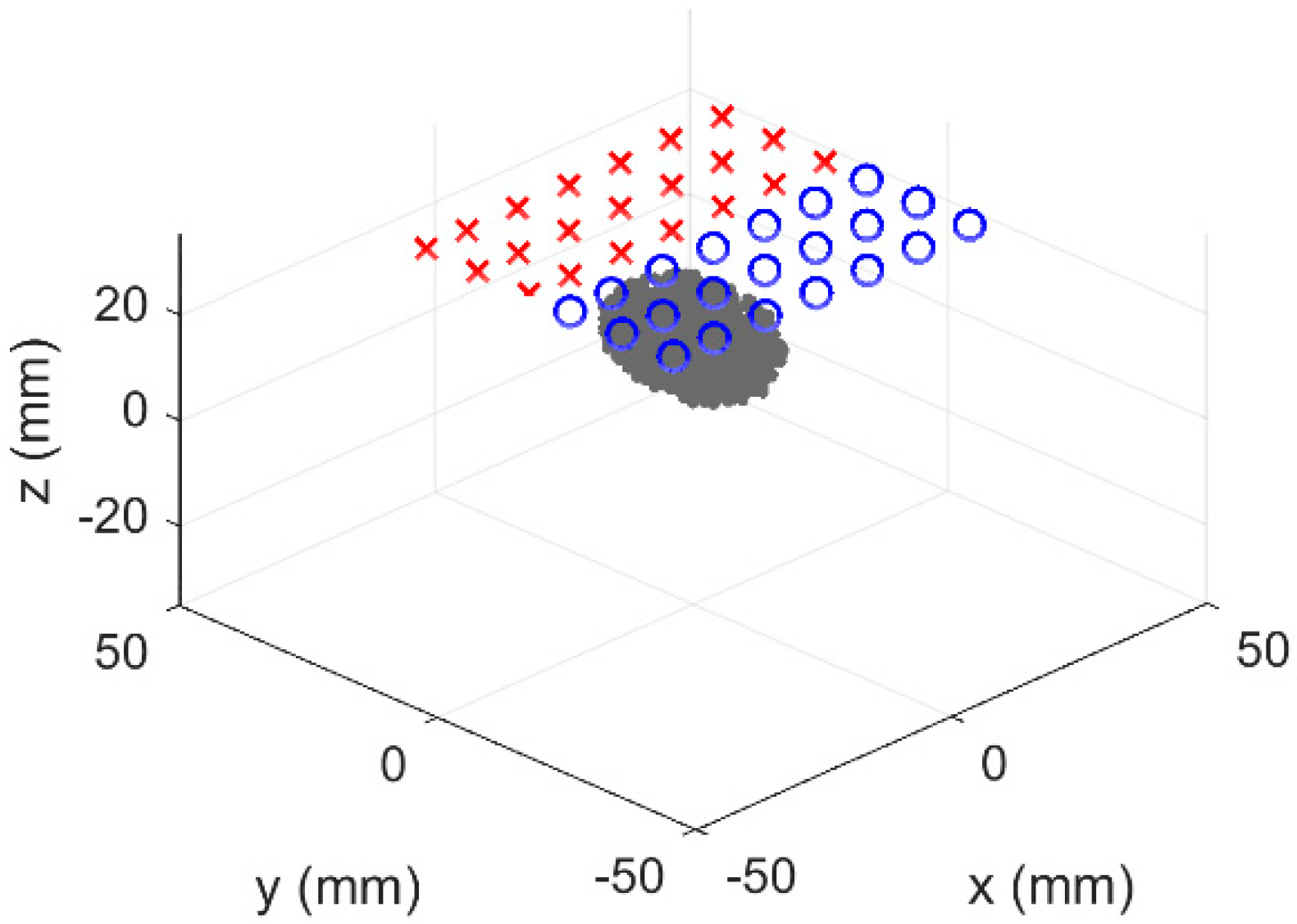
2.2. Simulation Overview
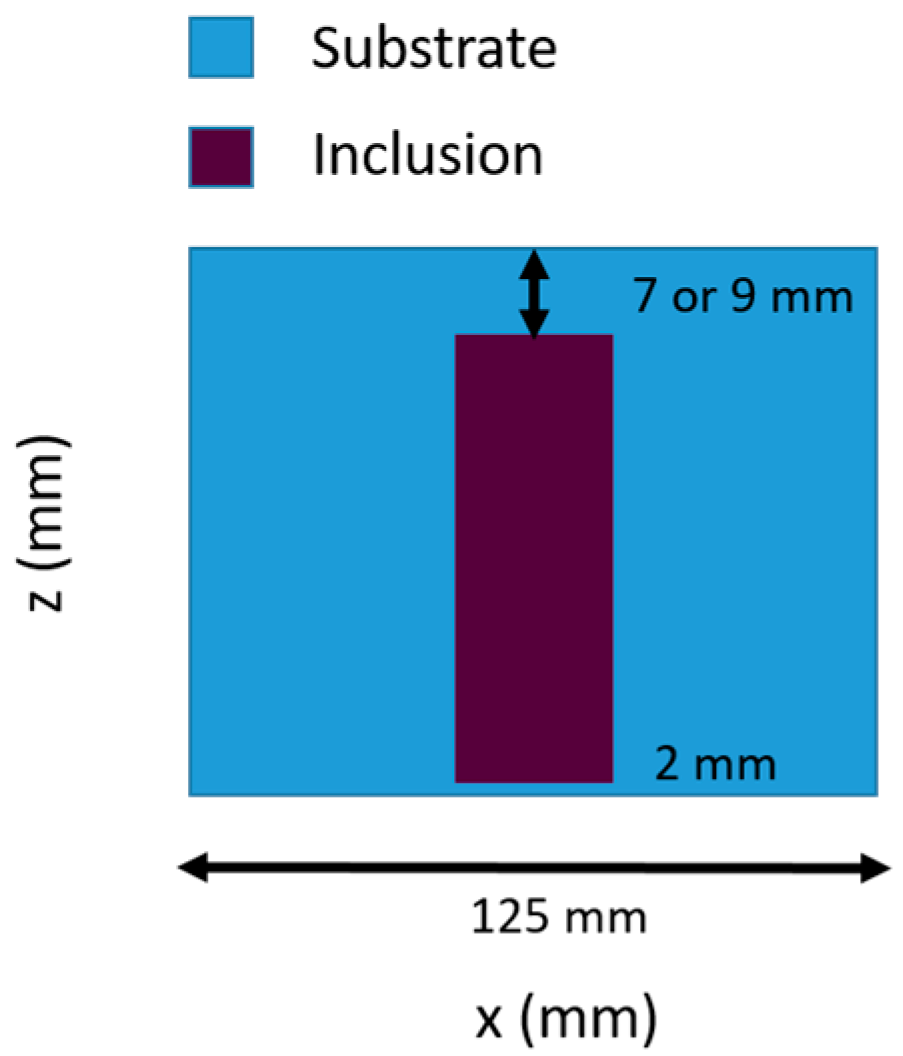
2.3. Tissue-Simulating Phantom Validation
2.3.1. Phantom Composition
2.3.2. Phantom Measurements
3. Results
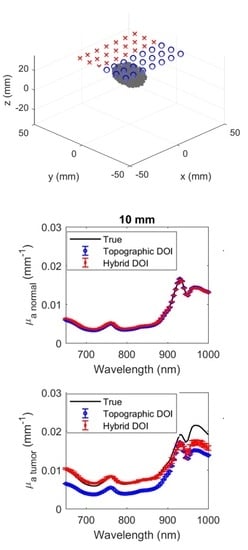
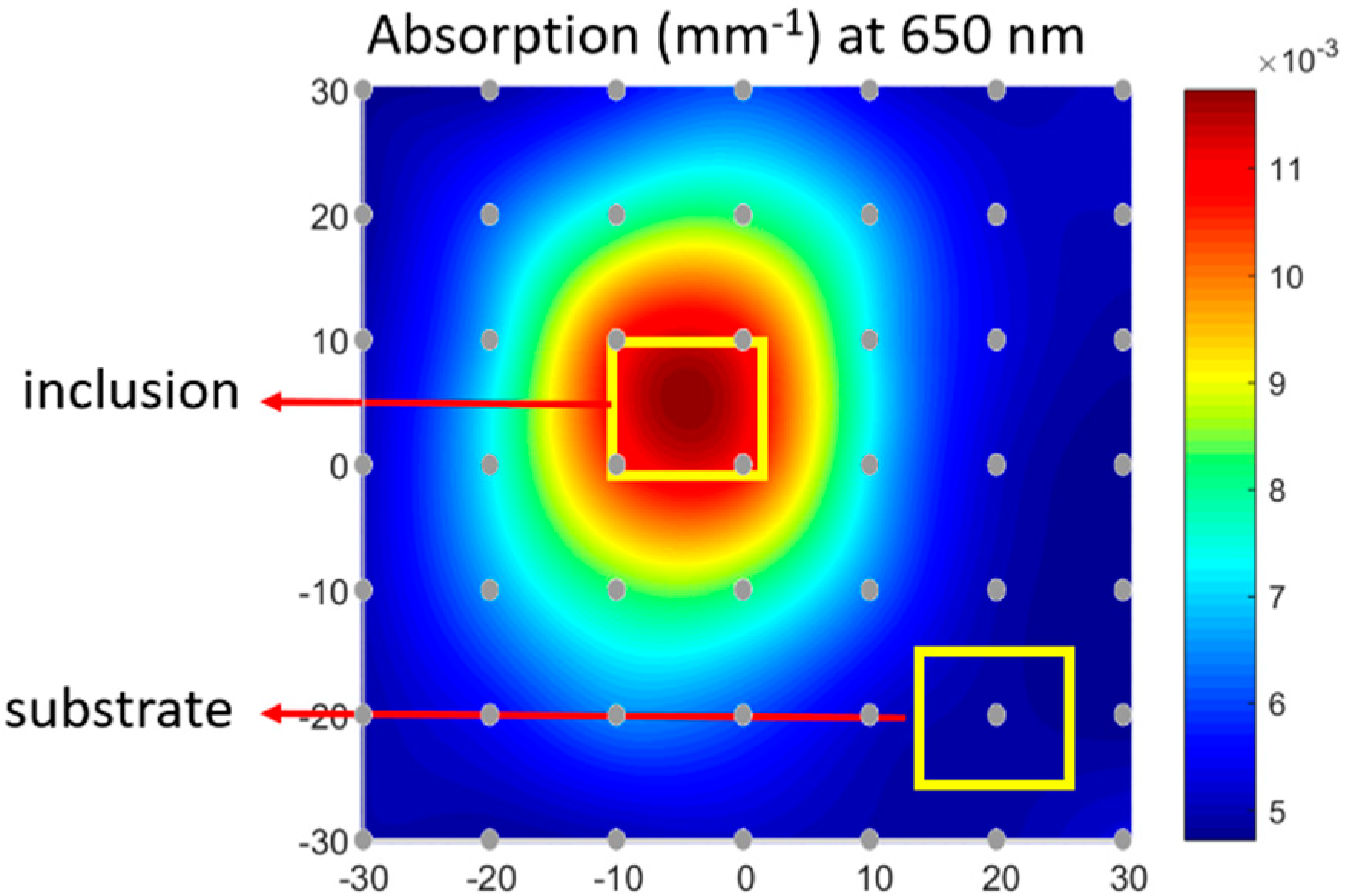
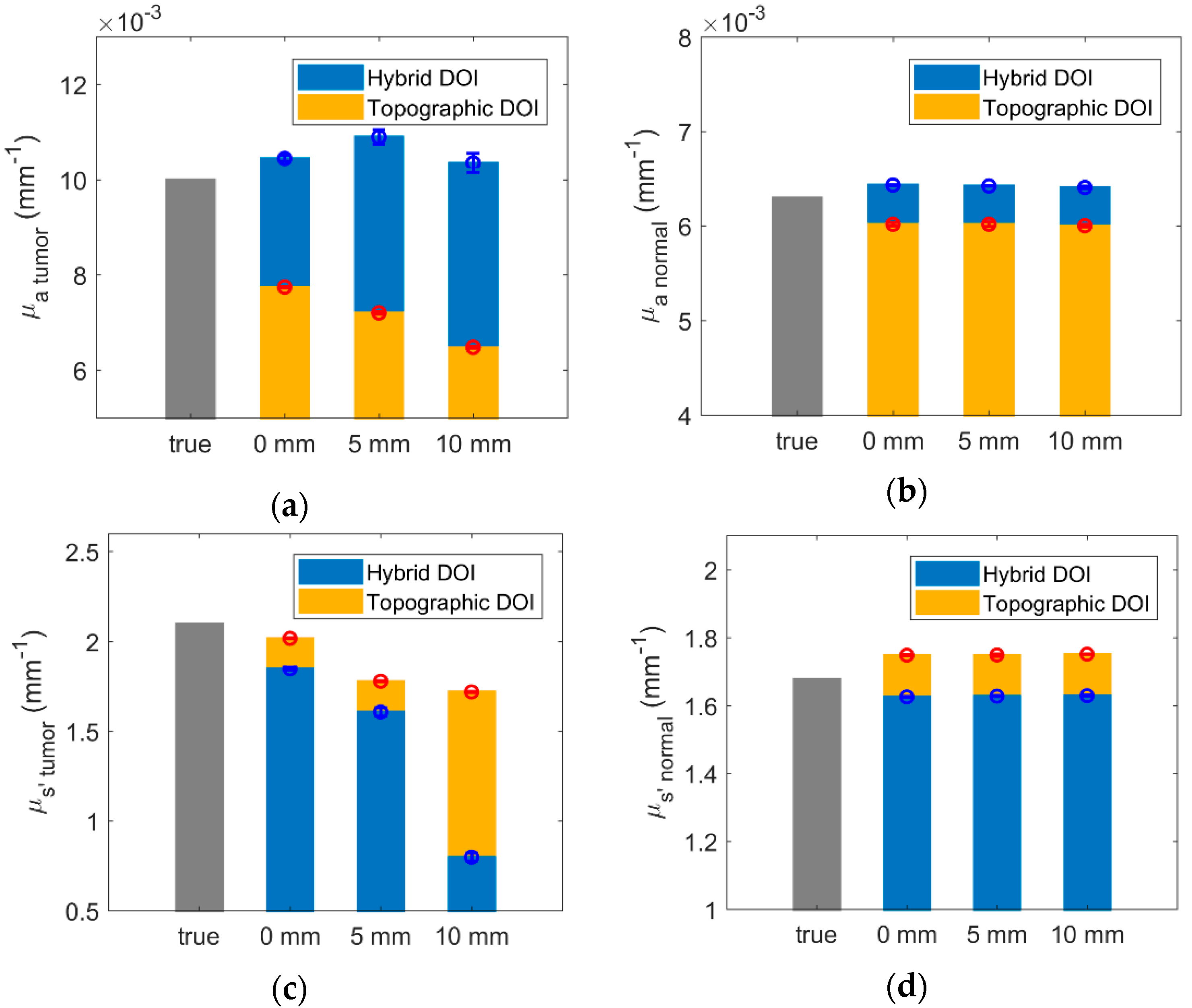
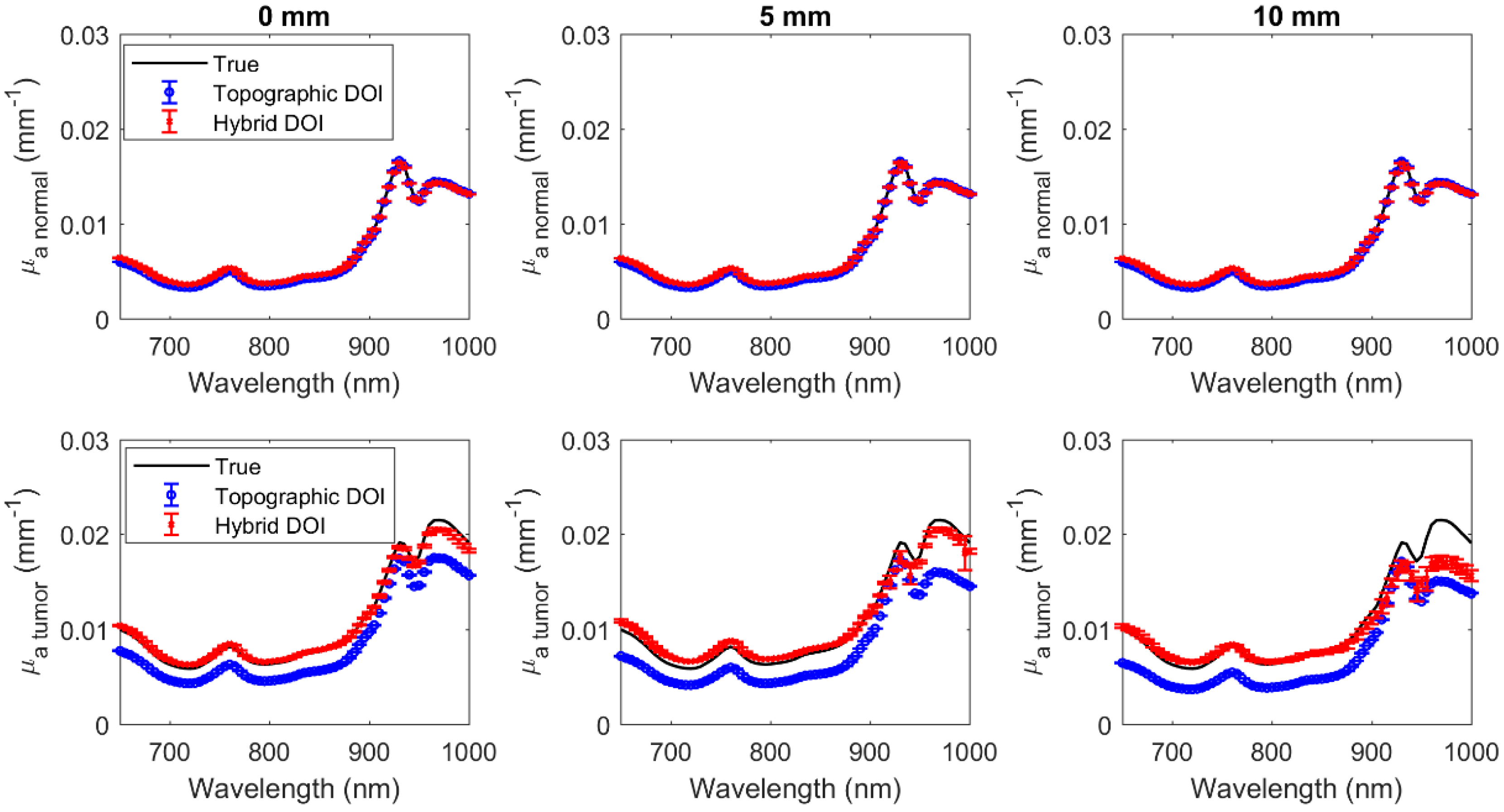
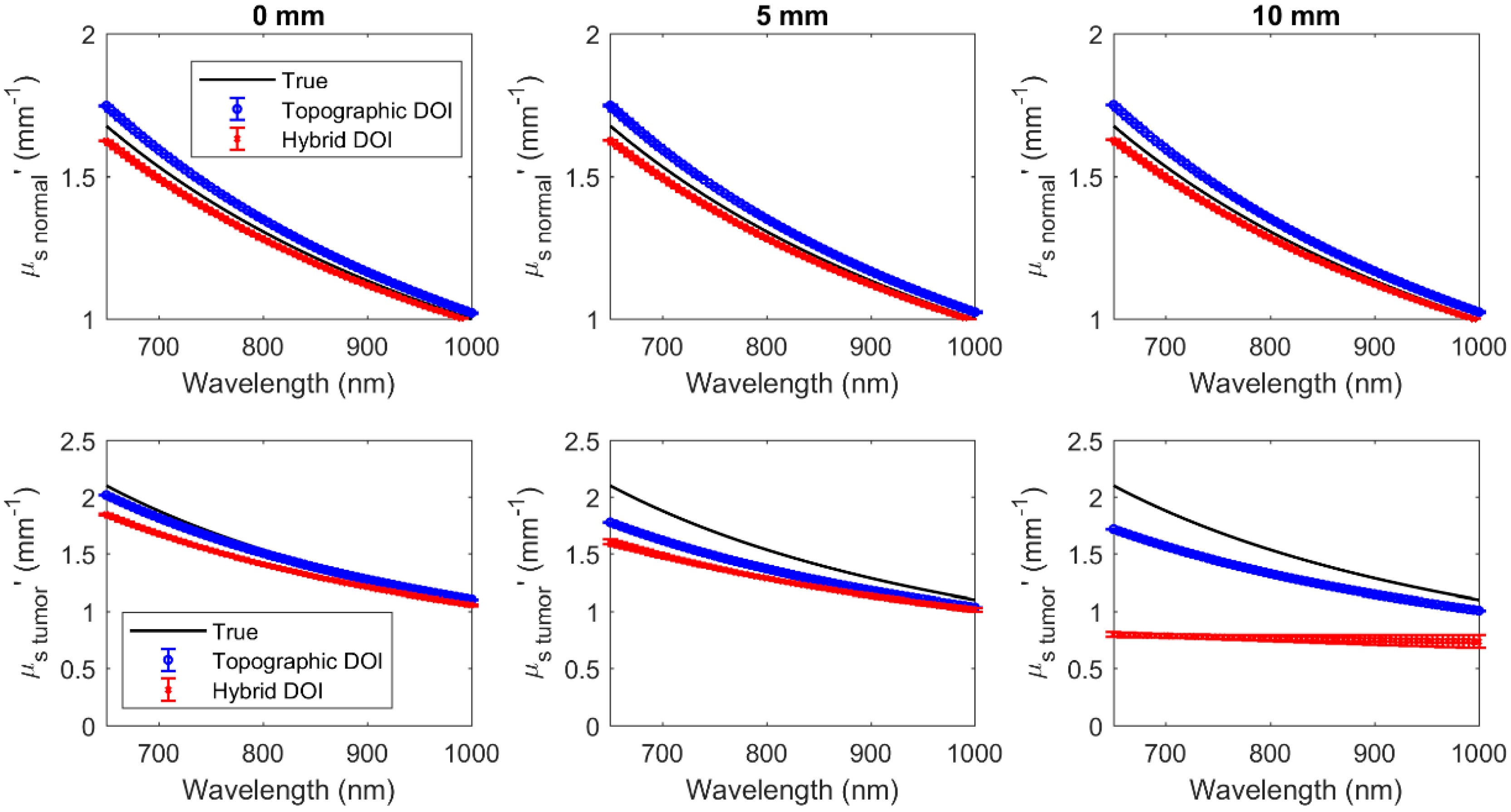
3.1. Simulated Results of Hybrid DOI Method
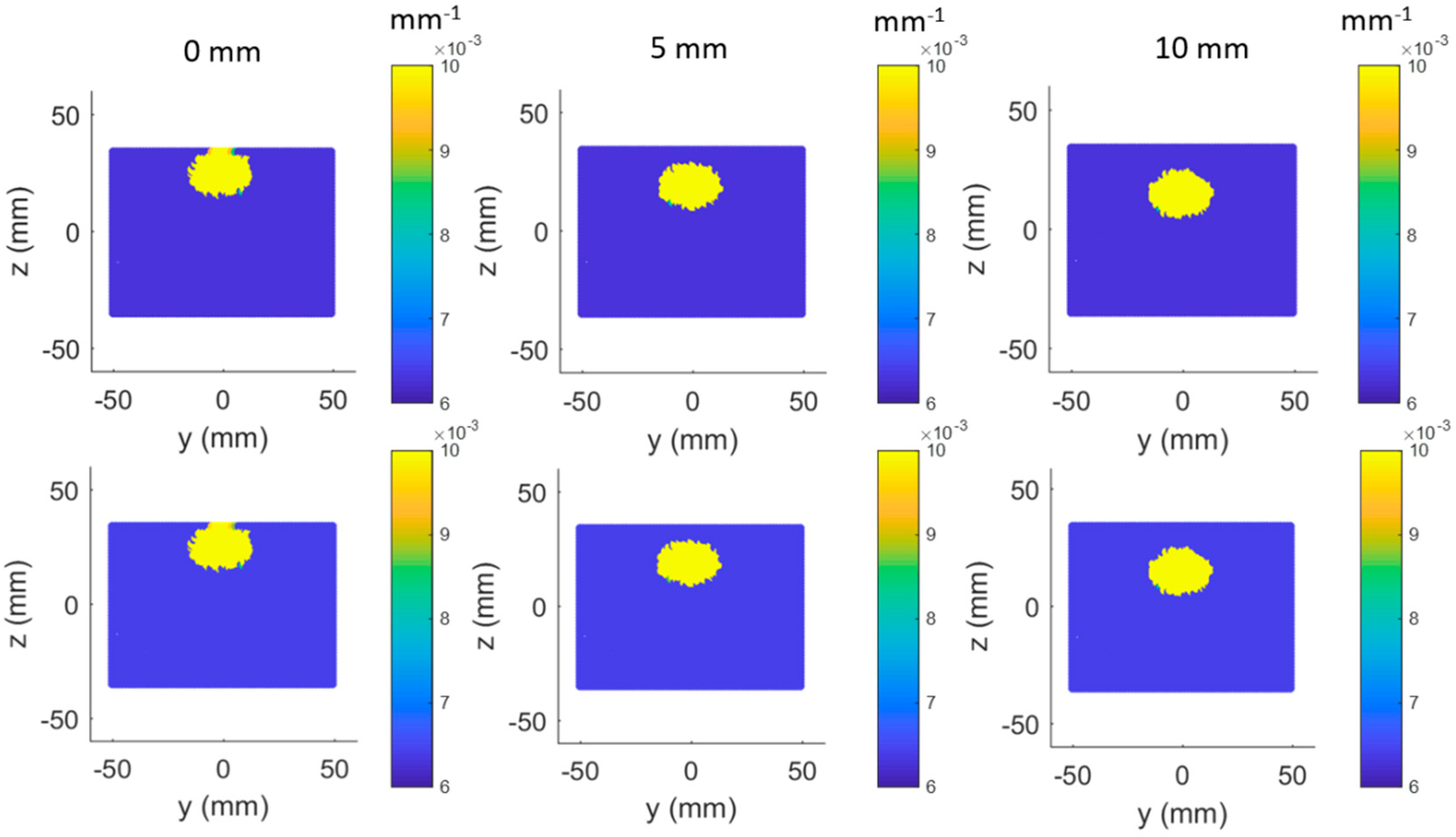
3.2. Simulated Results of Topographic DOI Method
3.3. Phantom Experimental Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- O’Sullivan, T.; Cerussi, A.E.; Cuccia, D.J.; Tromberg, B.J. Diffuse optical imaging using spatially and temporally modulated light. J. Biomed. Opt. 2012, 17, 0713111. [Google Scholar] [CrossRef]
- Cerussi, A.E.; Shah, N.S.; Hsiang, D.; Durkin, A.; Butler, J.A.; Tromberg, B.J. In vivo absorption, scattering, and physiologic properties of 58 malignant breast tumors determined by broadband diffuse optical spectroscopy. J. Biomed. Opt. 2006, 11, 044005. [Google Scholar] [CrossRef] [PubMed]
- Cerussi, A.E.; Hsiang, D.; Shah, N.; Mehta, R.; Durkin, A.; Butler, J.; Tromberg, B.J. Predicting response to breast cancer neoadjuvant chemotherapy using diffuse optical spectroscopy. Proc. Natl. Acad. Sci. USA 2007, 104, 4014–4019. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Kurtzman, S.H.; Hegde, P.; Tannenbaum, S.; Kane, M.; Huang, M.; Chen, N.; Jagjivan, B.; Zarfos, K. Utilizing Optical Tomography with Ultrasound Localization to Image Heterogeneous Hemoglobin Distribution in Large Breast Cancers1. Neoplasia 2005, 7, 263–270. [Google Scholar] [CrossRef] [PubMed][Green Version]
- O’Sullivan, T.; Leproux, A.; Chen, J.H.; Bahri, S.; Matlock, A.; Roblyer, D.M.; McLaren, C.E.; Chen, W.-P.; Cerussi, A.E.; Su, M.; et al. Optical imaging correlates with magnetic resonance imaging breast density and reveals composition changes during neoadjuvant chemotherapy. Breast Cancer Res. 2013, 15, R14. [Google Scholar] [CrossRef]
- Shah, N.; Cerussi, A.E.; Jakubowski, D.B.; Hsiang, D.; Butler, J.A.; Tromberg, B.J. Spatial variations in optical and physiological properties of healthy breast tissue. J. Biomed. Opt. 2004, 9, 534–541. [Google Scholar] [CrossRef]
- Choe, R.; Corlu, A.; Lee, K.; Durduran, T.; Konecky, S.D.; Grosicka-Koptyra, M.; Arridge, S.R.; Czerniecki, B.J.; Fraker, D.L.; DeMichele, A.; et al. Diffuse Optical Tomography of Breast Cancer during Neoadjuvant Chemotherapy: A Case Study with Comparison to MRI. Med. Phys. 2005, 32, 1128–1139. [Google Scholar] [CrossRef]
- Shah, N.; Gibbs, J.; Wolverton, D.; Cerussi, A.; Hylton, N.; Tromberg, B.J. Combined diffuse optical spectroscopy and contrast-enhanced magnetic resonance imaging for monitoring breast cancer neoadjuvant chemotherapy: A case study. J. Biomed. Opt. 2005, 10, 051503. [Google Scholar] [CrossRef]
- Kukreti, S.; Cerussi, A.; Tromberg, B.; Gratton, E. Intrinsic tumor biomarkers revealed by novel double-differential spectroscopic analysis of near-infrared spectra. J. Biomed. Opt. 2007, 12, 20509. [Google Scholar] [CrossRef]
- Kukreti, S.; Cerussi, A.; Tromberg, B.; Gratton, E. Intrinsic near-infrared spectroscopic markers of breast tumors. Dis. Markers 2008, 25, 281–290. [Google Scholar] [CrossRef]
- Kukreti, S.; Cerussi, A.E.; Tanamai, W.; Hsiang, D.; Tromberg, B.J.; Gratton, E. Characterization of metabolic differences between benign and malignant tumors: High-spectral-resolution diffuse optical spectroscopy. Radiology 2010, 254, 277–284. [Google Scholar] [CrossRef]
- Leproux, A.; Durkin, A.F.; Compton, M.; Cerussi, A.E.; Gratton, E.; Tromberg, B.J. Assessing tumor contrast in radiographically dense breast tissue using Diffuse Optical Spectroscopic Imaging (DOSI). Breast Cancer Res. 2013, 15, R89. [Google Scholar] [CrossRef]
- Leproux, A.; Kim, Y.M.; Min, J.W.; McLaren, C.E.; Chen, W.-P.; O’Sullivan, T.; Lee, S.-H.; Chung, P.-S.; Tromberg, B.J. Differential diagnosis of breast masses in South Korean premenopausal women using diffuse optical spectroscopic imaging. J. Biomed. Opt. 2016, 21, 74001. [Google Scholar] [CrossRef] [PubMed]
- Tromberg, B.J.; Pogue, B.W.; Paulsen, K.D.; Yodh, A.G.; Boas, D.A.; Cerussi, A.E. Assessing the future of diffuse optical imaging technologies for breast cancer management. Med. Phys. 2008, 35, 2443–2451. [Google Scholar] [CrossRef]
- Hull, E.L.; Nichols, M.; Foster, T.H. Quantitative broadband near-infrared spectroscopy of tissue-simulating phantoms containing erythrocytes. Phys. Med. Boil. 1998, 43, 3381–3404. [Google Scholar] [CrossRef] [PubMed]
- Bevilacqua, F.; Berger, A.J.; Cerussi, A.E.; Jakubowski, D.; Tromberg, B.J. Broadband absorption spectroscopy in turbid media by combined frequency-domain and steady-state methods. Appl. Opt. 2000, 39, 6498–6507. [Google Scholar] [CrossRef] [PubMed]
- Sekar, S.K.V.; Mora, A.D.; Bargigia, I.; Martinenghi, E.; Lindner, C.; Farzam, P.; Pagliazzi, M.; Durduran, T.; Taroni, P.; Pifferi, A.; et al. Broadband (600–1350 nm) Time-Resolved Diffuse Optical Spectrometer for Clinical Use. IEEE J. Sel. Top. Quantum Electron. 2015, 22, 406–414. [Google Scholar] [CrossRef]
- Tachtsidis, I.; Leung, T.S.; Tahir, B.; Elwell, C.; Kohl-Bareis, M.; Gramer, M.; Cooper, C. A Hybrid Multi-Distance Phase and Broadband Spatially Resolved Algorithm for Resolving Absolute Concentrations of Chromophores in the Near-Infrared Light Spectrum: Results from Studies in Dynamic Phantoms. Adv. Exp. Med. Biol. 2010, 662, 169–175, Published online 2010. [Google Scholar]
- Tachtsidis, I.; Gao, L.; Leung, T.S.; Kohl-Bareis, M.; Cooper, C.; Elwell, C. A Hybrid Multi-Distance Phase and Broadband Spatially Resolved Spectrometer and Algorithm for Resolving Absolute Concentrations of Chromophores in the Near-Infrared Light Spectrum. In Single Molecule and Single Cell Sequencing; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2010; Volume 662, pp. 169–175. [Google Scholar]
- Kitsmiller, V.; Dummer, M.; Johnson, K.; O’Sullivan, T. Tunable vertical cavity surface emitting lasers for use in the near infrared biological window. Proc. SPIE 10488, Optical Fibers and Sensors for Medical Diagnostics and Treatment Applications XVIII. Available online: https://www.spiedigitallibrary.org/conference-proceedings-of-spie/10488/104880L/Tunable-vertical-cavity-surface-emitting-lasers-for-use-in-the/10.1117/12.2290899.short (accessed on 13 February 2018).
- Boas, D.A.; Dale, A.M.; Franceschini, M.A. Diffuse optical imaging of brain activation: Approaches to optimizing image sensitivity, resolution, and accuracy. NeuroImage 2004, 23, S275–S288. [Google Scholar] [CrossRef]
- Bigio, I.J.; Fantini, S. Quantitative Biomedical Optics: Theory, Methods, and Applications; Cambridge University Press: Cambridge, UK, 2016. [Google Scholar]
- Wang, J.; Davis, S.C.; Srinivasan, S.; Jiang, S.; Pogue, B.W.; Paulsen, K.D. Spectral tomography with diffuse near-infrared light: Inclusion of broadband frequency domain spectral data. J. Biomed. Opt. 2008, 13, 041305. [Google Scholar] [CrossRef]
- Culver, J.P.; Choe, R.; Holboke, M.J.; Zubkov, L.; Durduran, T.; Slemp, A.; Ntziachristos, V.; Chance, B.; Yodh, A.G. Three-dimensional diffuse optical tomography in the parallel plane transmission geometry: Evaluation of a hybrid frequency domain/continuous wave clinical system for breast imaging. Med. Phys. 2003, 30, 235–247. [Google Scholar] [CrossRef] [PubMed]
- Corlu, A.; Choe, R.; Durduran, T.; Lee, K.; Schweiger, M.; Arridge, S.; Hillman, E.M.C.; Yodh, A.G. Diffuse optical tomography with spectral constraints and wavelength optimization. Appl. Opt. 2005, 44, 2082–2093. [Google Scholar] [CrossRef] [PubMed]
- Kitsmiller, V.J.; O’Sullivan, T.; Stillwell, R.A. Toward handheld real time frequency domain diffuse optical spectroscopy. Proc. SPIE 10874, Optical Tomography and Spectroscopy of Tissue XIII, 1087405. Available online: https://www.spiedigitallibrary.org/conference-proceedings-of-spie/10874/1087405/Toward-handheld-real-time-frequency-domain-diffuse-optical-spectroscopy/10.1117/12.2508911.full (accessed on 1 March 2019).
- Chen, N.G.; Guo, P.; Yan, S.; Piao, D.; Zhu, Q. Simultaneous near-infrared diffusive light and ultrasound imaging. Appl. Opt. 2001, 40, 6367–6380. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, X.; Pogue, B.W.; Jiang, S.; Dehghani, H.; Song, X.; Srinivasan, S.; Brooksby, B.A.; Paulsen, K.D.; Kogel, C.; Poplack, S.P.; et al. Image reconstruction of effective Mie scattering parameters of breast tissue in vivo with near-infrared tomography. J. Biomed. Opt. 2006, 11, 041106. [Google Scholar] [CrossRef]
- Jacques, S.L. Optical properties of biological tissues: A review. Phys. Med. Boil. 2013, 58, R37–R61. [Google Scholar] [CrossRef]
- Dehghani, H.; Eames, M.E.; Yalavarthy, P.; Davis, S.C.; Srinivasan, S.; Carpenter, C.M.; Pogue, B.W.; Paulsen, K.D. Near Infrared Optical Tomography Using NIRFAST: Algorithm for Numerical Model and Image Reconstruction. Commun. Numer. Methods Eng. 2008, 25, 711–732. [Google Scholar] [CrossRef]
- Dehghani, H.; Srinivasan, S.; Pogue, B.W.; Gibson, A. Numerical Modelling and Image Reconstruction in Diffuse Optical Tomography. Philos. Trans. A Math. Phys. Eng. Sci. 2009, 367, 3073–3093. [Google Scholar] [CrossRef]
- Haskell, R.C.; Svaasand, L.O.; Tsay, T.-T.; Feng, T.-C.; McAdams, M.S.; Tromberg, B.J. Boundary conditions for the diffusion equation in radiative transfer. J. Opt. Soc. Am. A 1994, 11, 2727. [Google Scholar] [CrossRef]
- Cerussi, A.; Tromberg, B. Photon Migration Spectroscopy Frequency- Domain Techniques. In Biomedical Photonics Handbook; CRC Press: Boca Raton, FL, USA, 2003. [Google Scholar]
- Lionheart, W. EIT reconstruction algorithms: Pitfalls, challenges and recent developments. Physiol. Meas. 2004, 25, 125–142. [Google Scholar] [CrossRef]
- Ueda, S.; Yoshizawa, N.; Shigekawa, T.; Takeuchi, H.; Ogura, H.; Osaki, A.; Saeki, T.; Ueda, Y.; Yamane, T.; Kuji ISakahara, H. Near-Infrared Diffuse Optical Imaging for Early Prediction of Breast Cancer Response to Neoadjuvant Chemotherapy: A Comparative Study Using 18F-FDG PET/CT. J. Nucl. Med. 2016, 57, 1189–1195. [Google Scholar] [CrossRef]
- Tromberg, B.J.; Zhang, Z.; Leproux, A.; O’Sullivan, T.; Cerussi, A.E.; Carpenter, P.M.; Mehta, R.; Roblyer, D.M.; Yang, W.; Paulsen, K.D.; et al. Predicting Responses to Neoadjuvant Chemotherapy in Breast Cancer: ACRIN 6691 Trial of Diffuse Optical Spectroscopic Imaging. Cancer Res. 2016, 76, 5933–5944. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Pogue, B.W.; Jiang, S.; Paulsen, K.D. Near-infrared tomography of breast cancer hemoglobin, water, lipid, and scattering using combined frequency domain and cw measurement. Opt. Lett. 2010, 35, 82–84. [Google Scholar] [CrossRef]
- Cerussi, A.E.; Warren, R.; Hill, B.; Roblyer, D.; Leproux, A.; Durkin, A.F.; O’Sullivan, T.; Keene, S.; Haghany, H.; Quang, T.; et al. Tissue phantoms in multicenter clinical trials for diffuse optical technologies. Biomed. Opt. Express 2012, 3, 966–971. [Google Scholar] [CrossRef] [PubMed]
- Bosschaart, N.; Leproux, A.; Abdalsalam, O.; Chen, W.-P.; McLaren, C.E.; Tromberg, B.J.; O’Sullivan, T. Diffuse optical spectroscopic imaging for the investigation of human lactation physiology: A case study on mammary involution. J. Biomed. Opt. 2019, 24, 1–8. [Google Scholar] [CrossRef]
- Biswal, N.C.; Xu, Y.; Zhu, Q. Imaging tumor oxyhemoglobin and deoxyhemoglobin concentrations with ultrasound-guided diffuse optical tomography. Technol. Cancer Res. Treat. 2011, 10, 417–429. [Google Scholar] [CrossRef]
- Zhu, Q.; Huang, M.; Chen, N.; Zarfos, K.; Jagjivan, B.; Kane, M.; Hedge, P.; Kurtzman, S.H. Ultrasound-Guided Optical Tomographic Imaging of Malignant and Benign Breast Lesions: Initial Clinical Results of 19 Cases. Neoplasia 2003, 5, 379–388. [Google Scholar] [CrossRef]
- Larusson, F.; Fantini, S.; Miller, E. Hyperspectral image reconstruction for diffuse optical tomography. Biomed. Opt. Express 2011, 2, 946–965. [Google Scholar] [CrossRef]
- Eames, M.; Wang, J.; Pogue, B.W.; Dehghani, H. Wavelength band optimization in spectral near-infrared optical tomography improves accuracy while reducing data acquisition and computational burden. J. Biomed. Opt. 2008, 13, 054037. [Google Scholar] [CrossRef]
- Brooksby, B.; Dehghani, H.; Pogue, B.; Paulsen, K. Near-infrared (nir) tomography breast image reconstruction with a priori structural information from mri: Algorithm development for reconstructing heterogeneities. IEEE J. Sel. Top. Quantum Electron. 2003, 9, 199–209. [Google Scholar] [CrossRef]
- Brooksby, B.; Jiang, S.; Dehghani, H.; Pogue, B.W.; Paulsen, K.D.; Weaver, J.; Kogel, C.; Poplack, S.P. Combining near-infrared tomography and magnetic resonance imaging to study in vivo breast tissue: Implementation of a Laplacian-type regularization to incorporate magnetic resonance structure. J. Biomed. Opt. 2005, 10, 051504. [Google Scholar] [CrossRef] [PubMed]


| Parameters | Normal | Tumor |
|---|---|---|
| Absorbing Chromophores | ||
| HbO (µM) | 8.6 | 17.5 |
| HHb (µM) | 6.3 | 9.8 |
| H2O (%) | 21.8 | 31.3 |
| Lipids (%) | 80.6 | 66.3 |
| NGB (g/L) | -- | 0.05 |
| Scattering Parameters | ||
| Scattering amplitude (a) (mm−1) | 2.29 | 3.11 |
| Scattering power (b) | 1.2 | 1.5 |
| Depths (mm) | Tumor | |||||||
| Hybrid DOI | Topographic DOI | |||||||
| % Difference of mean from true (%) | Difference from True | % Difference of Mean from True (%) | ||||||
| 0 | 4.4 × 10−4 ± 6.3 × 10−5 | −0.25 ± 7.9 × 10−3 | 4.4 | −12.0 | −2.3 × 10−3 ± 1.6 × 10−5 | −0.08 ± 2.6 × 10−3 | −22.6 | −3.9 |
| 5 | 8.9 × 10−4 ± 1.5 × 10−4 | −0.49 ± 0.02 | 8.9 | −23.5 | −2.8 × 10−3 ± 2.3 × 10−5 | −0.32 ± 2.4 × 10−3 | −28.0 | −15.3 |
| 10 | 3.5 × 10−4 ± 1.9 × 10−4 | −1.3 ± 0.02 | 3.5 | −62.0 | −3.5 × 10−3 ± 2.1 × 10−5 | −0.38 ± 1.9 × 10−3 | −35.2 | −18.2 |
| Normal | ||||||||
| Hybrid DOI | Topographic DOI | |||||||
| % Difference of mean from true (%) | Difference from true | % Difference of mean from true (%) | ||||||
| 0 | 1.3 × 10−4 ± 5.7 × 10−6 | −0.05 ± 1.0 × 10−3 | 2.1 | −3.1 | −2.8 × 10−4 ± 1.6 × 10−5 | 0.07 ± 2.3 × 10−3 | −4.5 | 4.2 |
| 5 | 1.2 × 10−4 ± 3.5 × 10−6 | −0.05 ± 1.2 × 10−3 | 1.9 | −3.0 | −2.9 × 10−4 ± 2.2 × 10−5 | 0.07 ± 3.5 × 10−3 | −4.5 | 4.3 |
| 10 | 1.1 × 10−4 ± 1.2 × 10−5 | −0.05 ± 1.5 × 10−3 | 1.7 | −2.9 | −2.9 × 10−4 ± 1.1 × 10−5 | 0.07 ± 1.1 × 10−3 | −4.7 | 4.4 |
| Depths (mm) | Tumor | |||||||
| Hybrid DOI | Topographic DOI | |||||||
| Mean% Change from True Over all Wavelengths (%) | Mean% Change from True Over all Wavelengths (%) | |||||||
| 0 | 3.9 × 10−4 | 0.13 | 3.87 | 7.86 | 2.2 × 10−3 | 0.03 | 22.35 | 1.78 |
| 5 | 7.2 × 10−4 | 0.25 | 7.49 | 15.34 | 2.6 × 10−3 | 0.16 | 26.52 | 10.38 |
| 10 | 1.2 × 10−3 | 0.75 | 8.39 | 47.86 | 3.2 × 10−3 | 0.21 | 32.26 | 13.04 |
| Normal | ||||||||
| Hybrid DOI | Topographic DOI | |||||||
| Mean% change from true over all wavelengths (%) | Mean% change from true over all wavelengths (%) | |||||||
| 0 | 1.1 × 10−4 | 0.03 | 1.64 | 1.9 | 1.9 × 10−4 | 0.04 | 3.65 | 3.14 |
| 5 | 1.0 × 10−4 | 0.02 | 1.50 | 1.76 | 2.1 × 10−4 | 0.04 | 3.89 | 3.30 |
| 10 | 1.0 × 10−4 | 0.02 | 1.35 | 1.64 | 2.2 × 10−4 | 0.04 | 4.10 | 3.37 |
| Depths (mm) | Phantom A—Inclusion | |||||||
| Hybrid DOI | Topographic DOI | |||||||
| Mean% Change from True Over all Wavelengths (%) | Mean% Change from True Over all Wavelengths (%) | |||||||
| 2 | 4.0 × 10−4 | 0.03 | 3.65 | 4.99 | 3.3 × 10−3 | 0.03 | 25.19 | 4.89 |
| 7 | 1.0 × 10−3 | 0.09 | 8.37 | 15.34 | 5.1 × 10−3 | 0.03 | 38.43 | 3.52 |
| Phantom A—Substrate | ||||||||
| Hybrid DOI | Topographic DOI | |||||||
| Mean% change from true over all wavelengths (%) | Mean% change from true over all wavelengths (%) | |||||||
| 2 | 4.6 × 10−5 | 0.008 | 1.01 | 0.78 | 3.31 × 10−5 | 0.0018 | 0.75 | 0.32 |
| 7 | 8.1 × 10−5 | 0.010 | 1.78 | 1.32 | 3.32 × 10−5 | 0.002 | 0.76 | 0.31 |
| Depths (mm) | Phantom B—Inclusion | |||||||
| Hybrid DOI | Topographic DOI | |||||||
| Mean% Change from True over All Wavelengths (%) | Mean% Change from True over All Wavelengths (%) | |||||||
| 2 | 1.8 × 10−3 | 0.14 | 13.89 | 13.67 | 1.8 × 10−3 | 0.25 | 13.88 | 24.12 |
| 9 | 1.5 × 10−3 | 0.49 | 10.64 | 51.99 | 4.3 × 10−3 | 0.43 | 32.05 | 43.49 |
| Phantom B—Substrate | ||||||||
| Hybrid DOI | Topographic DOI | |||||||
| Mean% change from true over all wavelengths (%) | Mean% change from true over all wavelengths (%) | |||||||
| 2 | 1.8 × 10−4 | 0.03 | 2.62 | 5.58 | 1.7 × 10−5 | 0.005 | 0.22 | 0.90 |
| 9 | 1.9 × 10−4 | 0.004 | 2.64 | 0.67 | 1.66 × 10−5 | 0.0052 | 0.23 | 0.91 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vasudevan, S.; Forghani, F.; Campbell, C.; Bedford, S.; O’Sullivan, T.D. Method for Quantitative Broadband Diffuse Optical Spectroscopy of Tumor-Like Inclusions. Appl. Sci. 2020, 10, 1419. https://doi.org/10.3390/app10041419
Vasudevan S, Forghani F, Campbell C, Bedford S, O’Sullivan TD. Method for Quantitative Broadband Diffuse Optical Spectroscopy of Tumor-Like Inclusions. Applied Sciences. 2020; 10(4):1419. https://doi.org/10.3390/app10041419
Chicago/Turabian StyleVasudevan, Sandhya, Farnoush Forghani, Chris Campbell, Savannah Bedford, and Thomas D. O’Sullivan. 2020. "Method for Quantitative Broadband Diffuse Optical Spectroscopy of Tumor-Like Inclusions" Applied Sciences 10, no. 4: 1419. https://doi.org/10.3390/app10041419
APA StyleVasudevan, S., Forghani, F., Campbell, C., Bedford, S., & O’Sullivan, T. D. (2020). Method for Quantitative Broadband Diffuse Optical Spectroscopy of Tumor-Like Inclusions. Applied Sciences, 10(4), 1419. https://doi.org/10.3390/app10041419