Simple Ventilators for Emergency Use Based on Bag-Valve Pressing Systems: Lessons Learned and Future Steps
Abstract
1. Introduction
- Have not been approved for use by any health regulatory agency or homologous institution.
- Have not been tested on humans or other animals, and that all tests have been carried out with mechanical and electronic equipment only. No clinical or preclinical trials have been conducted or approved up to the date of this publication.
- We strongly discourage any attempt to use the devices presented here on humans. Its replication should only be for the purpose of further investigation and development.
2. Design Considerations
3. Results
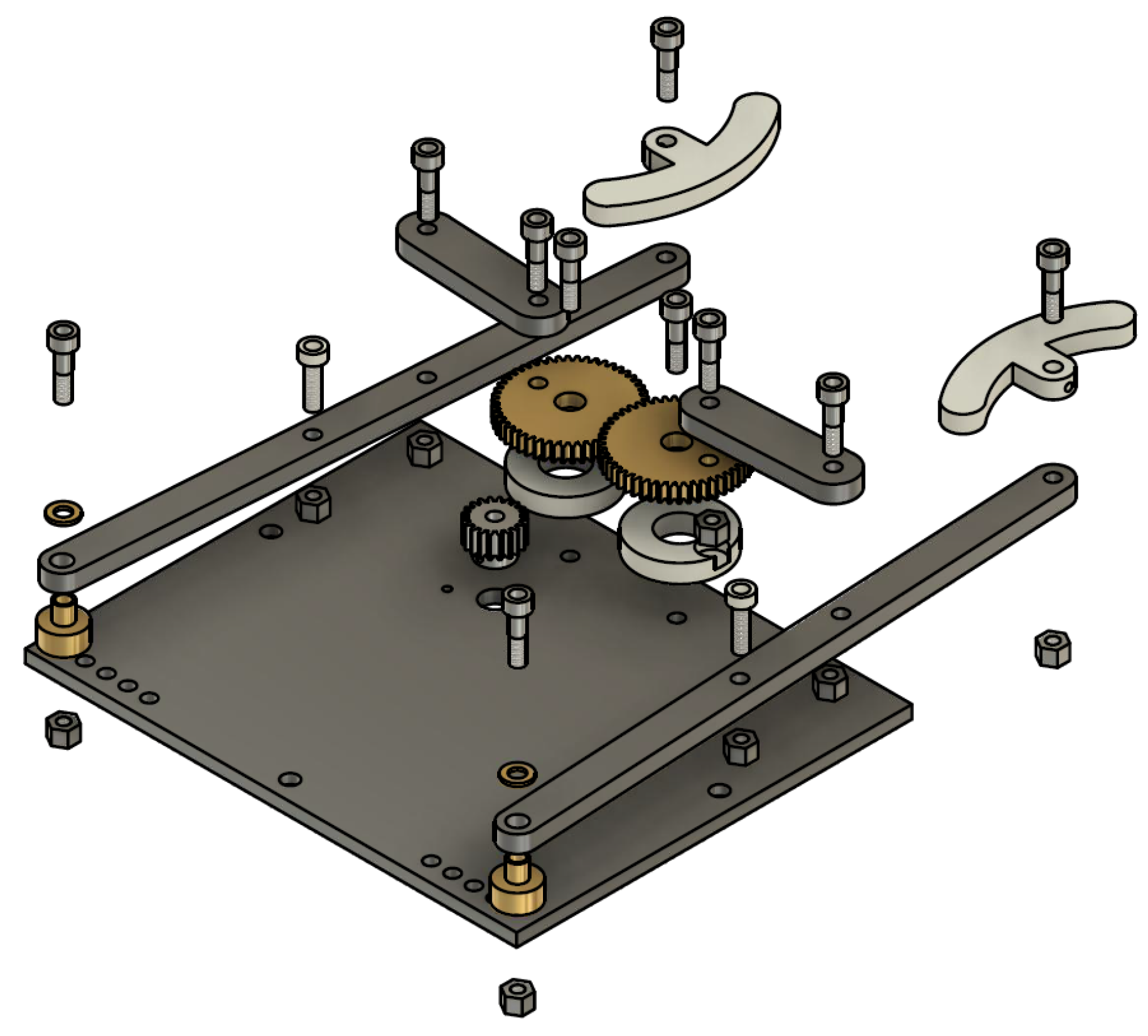
3.1. Prototype 1 (Contingence Life-Support Air Pressure Emergency Respirator—CLAPER)
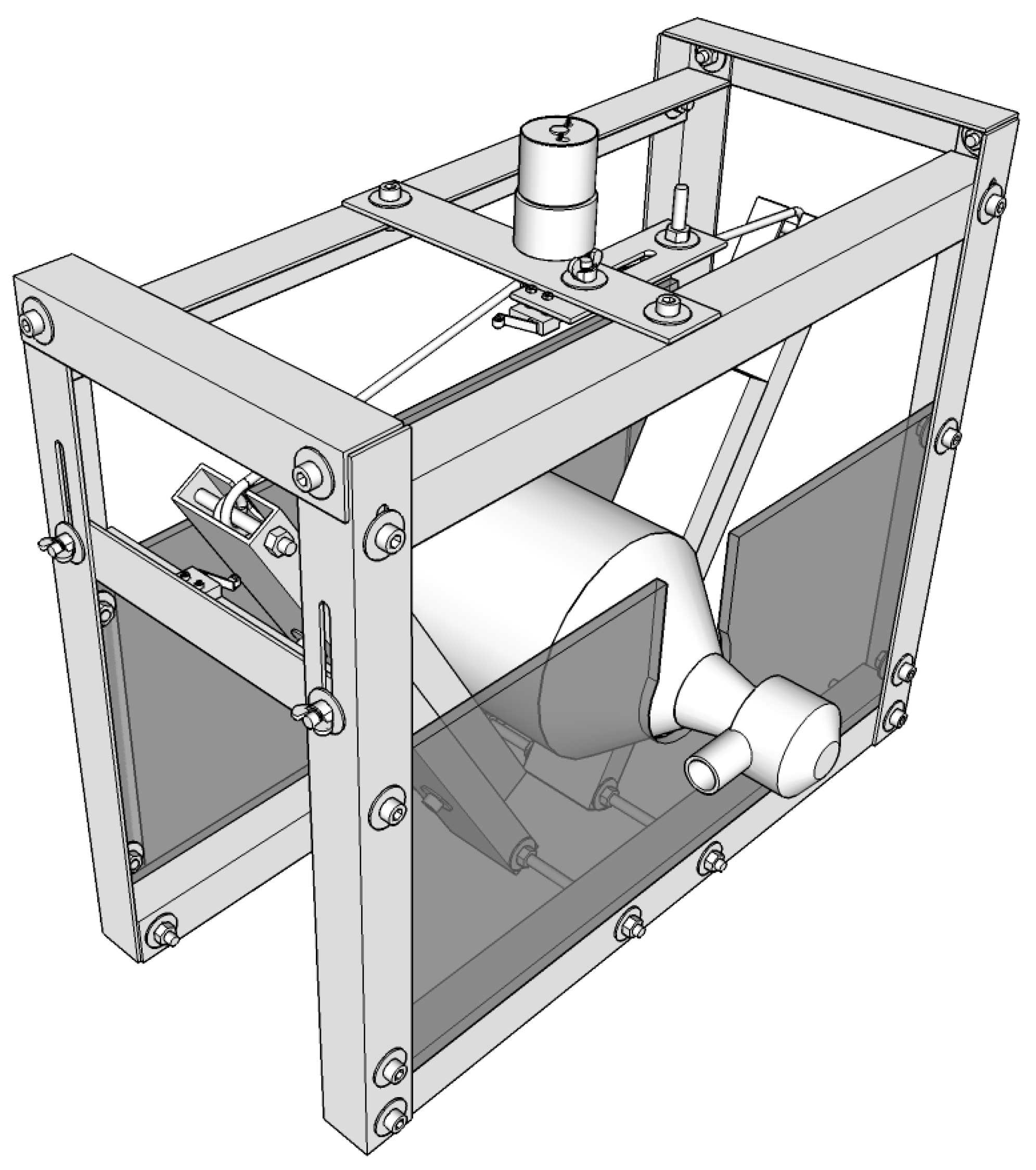
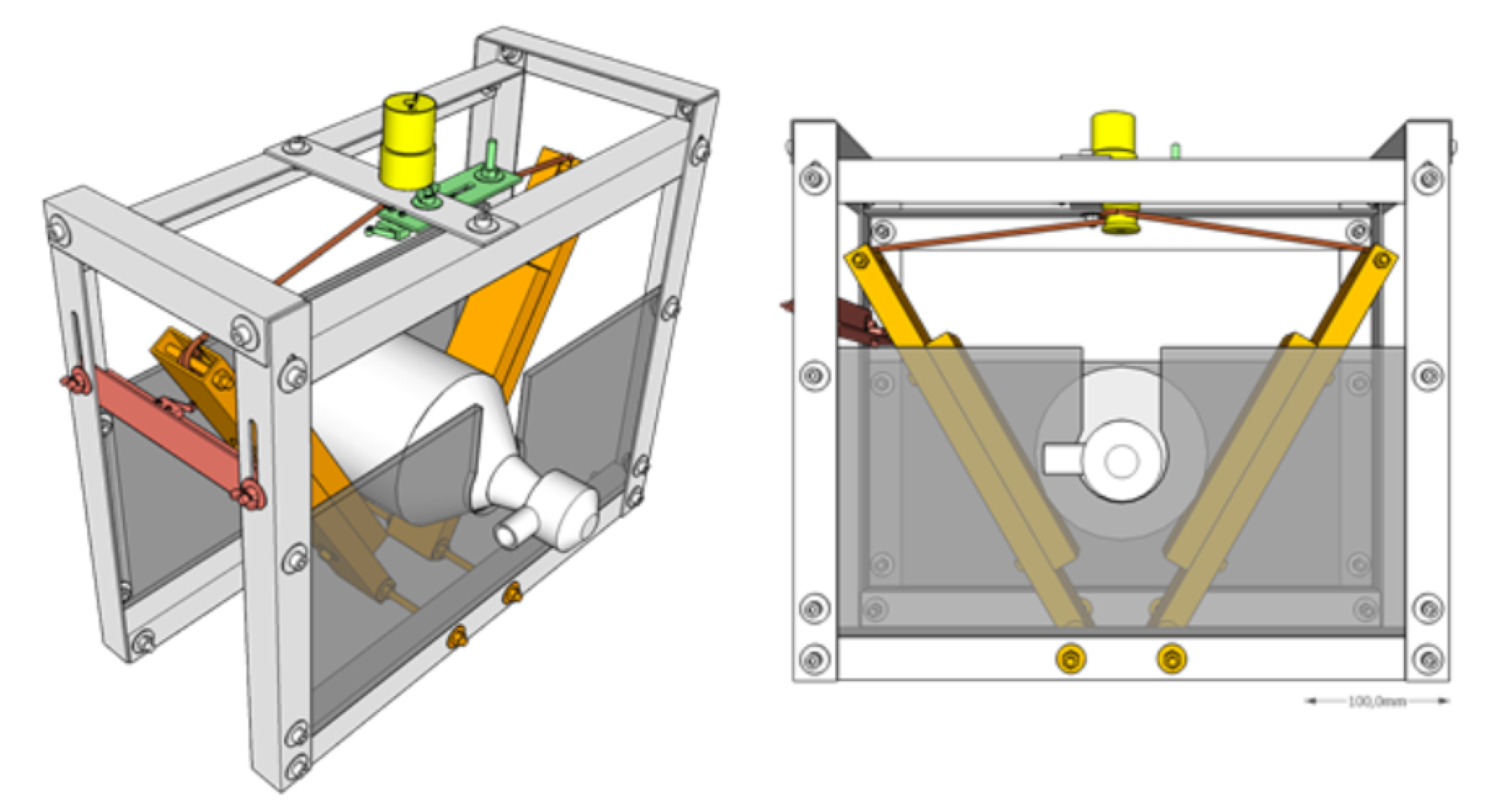
3.2. Prototype 2 (Experimental Actuated Resuscitator Machine—eARM)
4. Discussion and Conclusions
- Reliability of the mechanisms, software, etc.: Since these were fast track developments, there has not been enough time to undergo appropriate long term reliability tests on these pieces of equipment. Although we did run tests for several days on both prototypes, appropriate tests for weeks or months under realistic operation conditions are still needed and will require a significant amount of time, incompatible with the urgent requirement of ventilators to deal with the ongoing international crisis.
- Mass production with appropriate standards: Given that the concept of the devices presented here with the necessary adjustments for industrial production are expected to become medical equipment, the fabrication has to be performed in appropriate industrial environments with the necessary manufacture equipment and by trained personnel. In addition appropriate quality control has to be implemented. Once again, it is not clear how this could be done in a timely manner to face the COVID-19 pandemic.
- Regulatory approval or exemption: Being medical equipment, the use of the devices presented here, or variations around them, requires appropriate approval by the health regulatory agencies of the different countries that intend to use them. Such a process is, under normal circumstances, time consuming, involves a significant amount of bureaucratic paperwork and financial investment, not to mention extensive preclinical and clinical trials. While some countries, such as the USA that have issued a declaration of “liability immunity for activities related to medical countermeasures against COVID-19” [16], most countries have not taken similar measures, therefore, the use of these devices depends either on the creation of similar policies of general application, or the expedited approval of specific devices.
- Additional features: In addition to the central characteristics such as volumes, pressures, etc. required from a professional mechanical ventilator, there are a number of other characteristics that need to be met. For instance, the system has to be enclosed in an easy-to-clean fluid spill resistant casing, it requires battery support for the case of a power cut or for patient transportation, among other characteristics. The possibility to provide real-time pressure, flow and volume plots on a screen is also a characteristic that would be desirable as well as the possibility to program other inhalation/exhalation sequences that are of use for clinicians as diagnostic tools or for specific ventilation strategies such as pressure support ventilation.
5. Methods
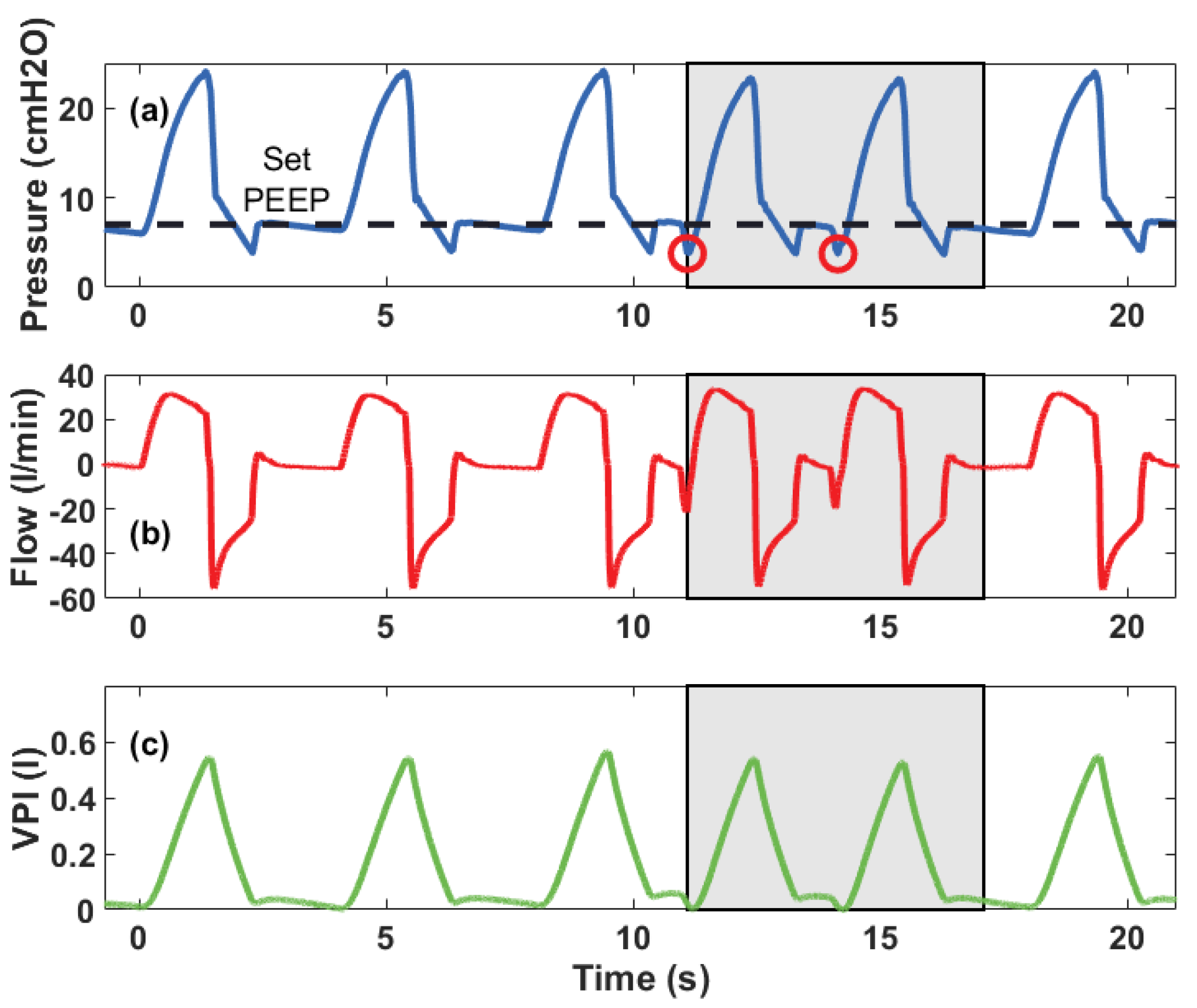
5.1. Pressure and Volume Measurements
5.2. CLAPER Prototype
5.3. eARM Prototype
- A flexible and strong string is required (e.g., braided nylon cord), which should be able to carry enough weight, depending on the pressure which is to be achieved.
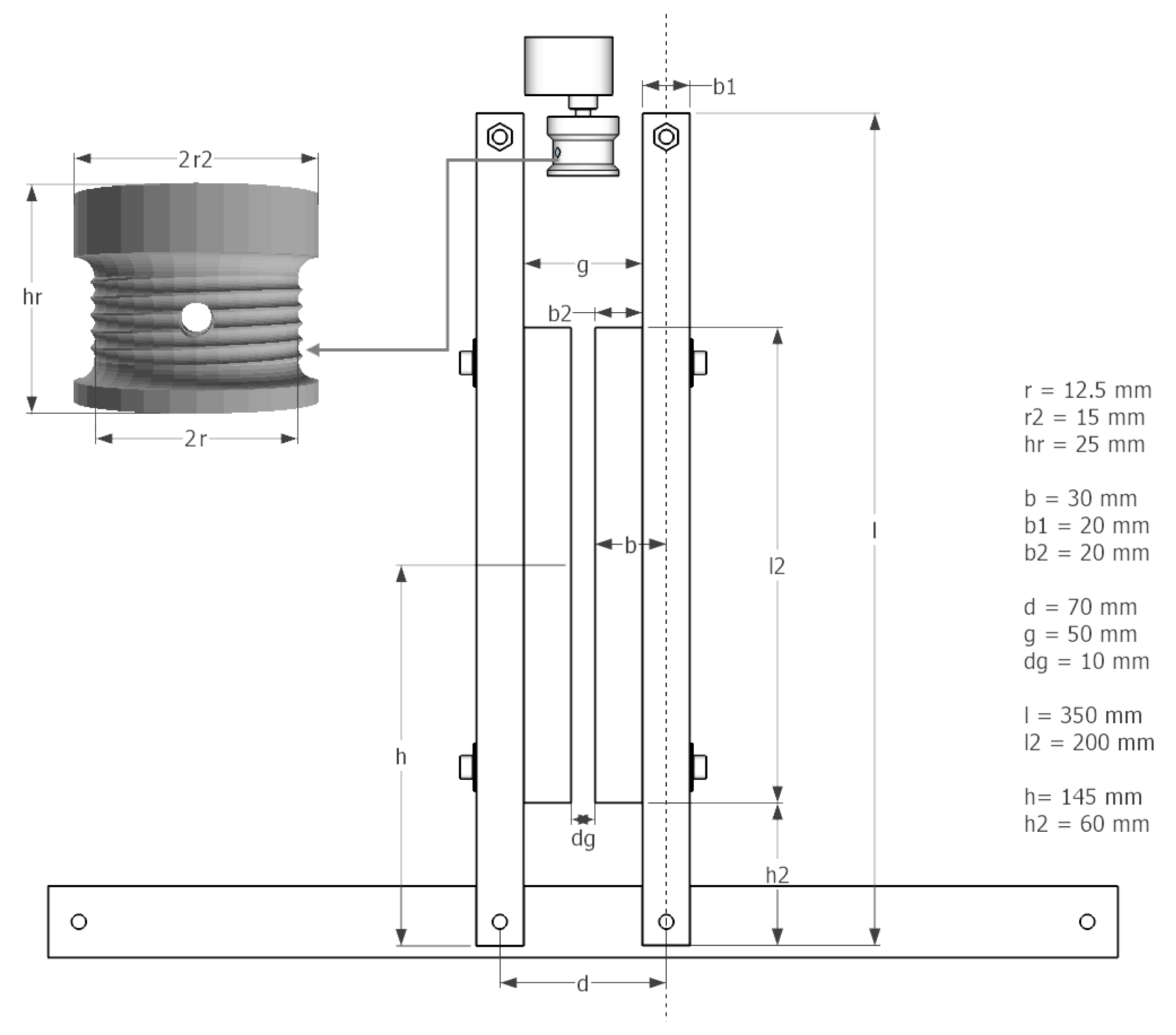
- A spiral form in the string roll (see Figure 16) reduces jitter from string rolling up badly. Choosing a sufficient r2, in a way that squeezing is achieved in only a few turns, also helps avoiding jitter.
- The string roll should enable easy installation/attachment of the string (e.g., twice through a hole in the string role).
- An attachment on the arms (b2) helps pressing the bag-valve resuscitator while leaving space for the string roll (g > 2r2).
- To avoid potential damages of the bag-valve resuscitator at the maximum squeezing, an additional space between the arms (dg) should be left.
- Movable microswitches make setting up the device easier. The tunable position of the upper microswitch is essential for a manual volume and pressure control.
- Two microswitches are needed to monitor and set respiration rate and inspiration to expiration ratio. Also if 1:1 ratio is the goal, keep in mind that opening and closing times differ.
- Since bag-valve resuscitators are not designed for continuous long-term use, their characteristics and material can deteriorate with time. Regular rechecking and, if necessary, replacement are advised. Therefore, a design enabling relatively easy and quick replacement of it should be realized.
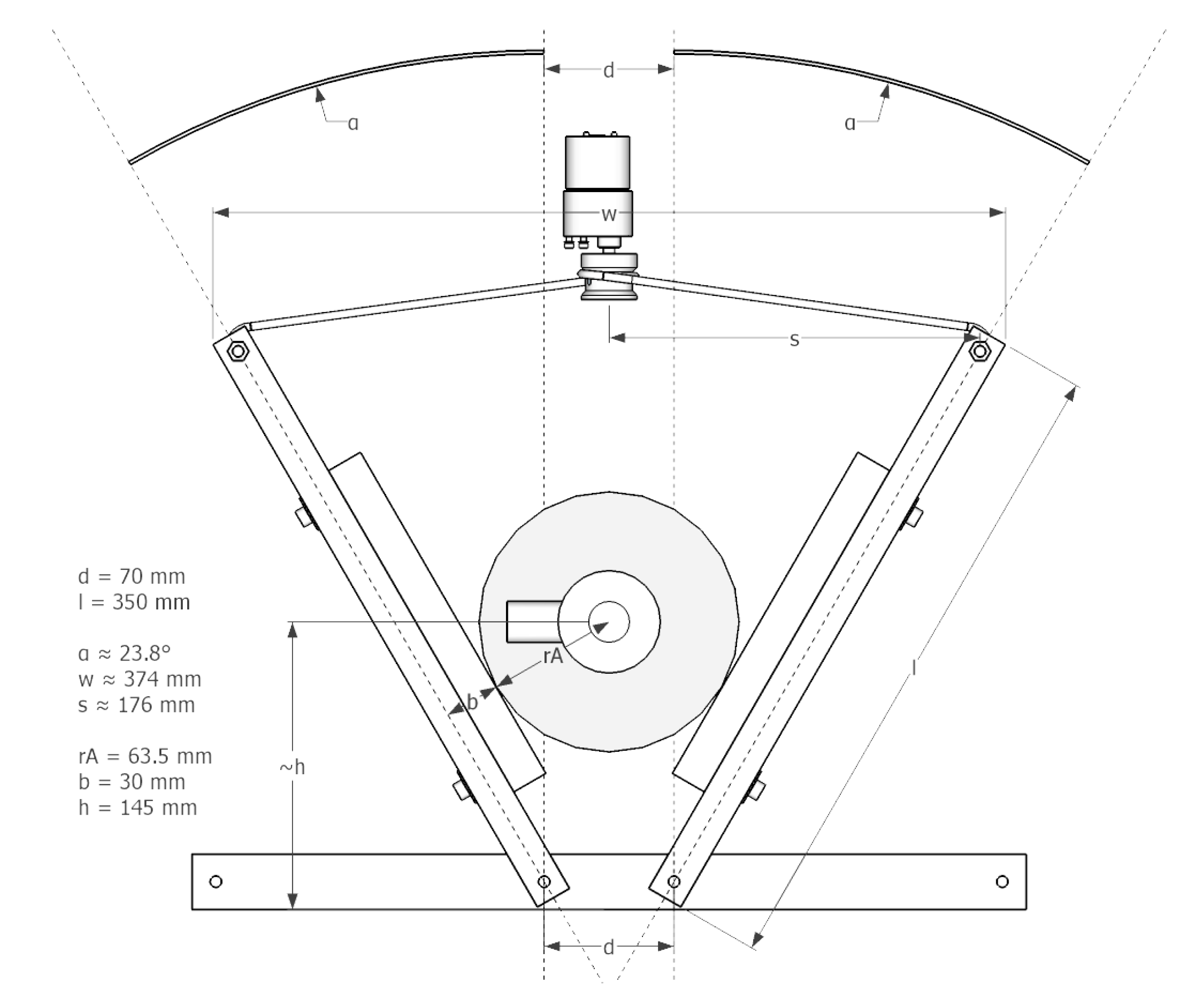
- In Figure 17, the arm the length (l), spacing between the two rotational axis (d), bag-valve resuscitator position (h) and string roll radius (r) are shown. Accordingly a motor with appropriate characteristics (torque, rpm) have to be chosen in a way, that the squeezing process runs smoothly with the desired pace while reaching the desired pressure and volume.
- By increasing l, the motor’s leverage also increases, if h stays unchanged. This means that a motor with less torque could perform the squeezing. However, in this case the end points of the arms have to travel a greater distance from released to squeezed state, meaning that the motor has to rotate faster at the same r2 of the string role to maintain the same rate of squeezing (BPM). By increasing the r2, the speed of the motor could be kept unchanged, however, this would cancel out the leverage gained by increasing l. Nevertheless, these relations allow for some tunability of the design according to the needs.
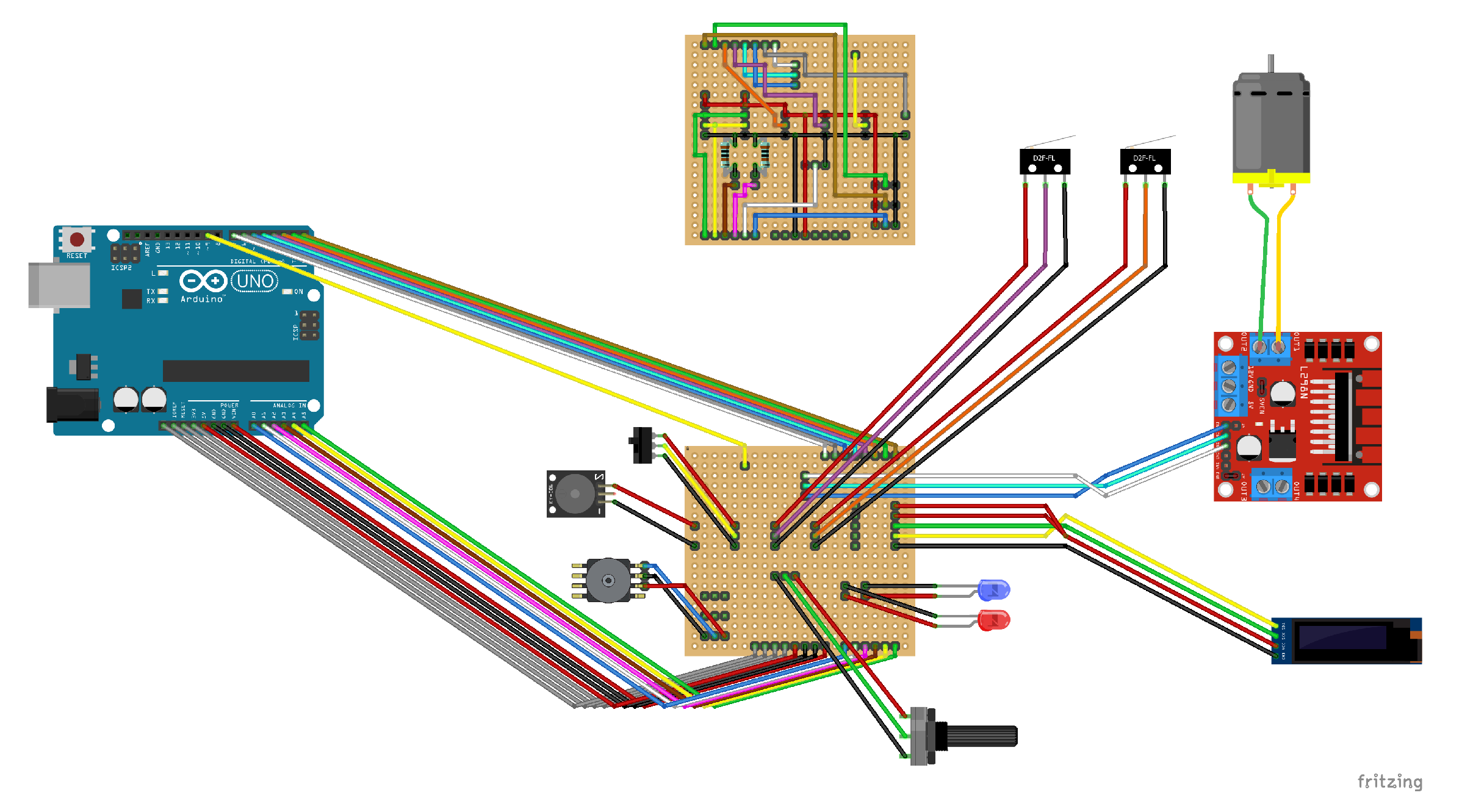
- The systems requires active control from a microcontroller and we propose the connection schematics shown in Figure 18. The microcontroller used was the ArduinoUNO platform and the code can be found in the supplementary information.
Author Contributions
Funding
Conflicts of Interest
References
- Ranney, M.L.; Griffeth, V.; Jha, A.K. Critical Supply Shortages—The Need for Ventilators and Personal Protective Equipment during the Covid-19 Pandemic. N. Engl. J. Med. 2020, 382, e41. [Google Scholar] [CrossRef] [PubMed]
- Young, J.; Sykes, M. Assisted ventilation. 1. Artificial ventilation: History, equipment and techniques. Thorax 1990, 45, 753. [Google Scholar] [CrossRef] [PubMed]
- Powelson, S.K. Design and Prototyping of a Low-Cost Portable Mechanical Ventilator. Ph.D. Thesis, Massachusetts Institute of Technology, Cambridge, MA, USA, 2010. [Google Scholar]
- Malya, R.; De Santiago, C.; Dickman, N.; Nasteff, M.; Nonet, T.; Sundaramraj, A.; Vasquez, K.; Abidi, S.; Oden, M. A Novel Augmentation Device for Ventilation Utilizing the Bag Valve Mask in EMS and Resource Limited Settings. Circulation 2019, 140, A14208. [Google Scholar]
- Vicente, V.C.; Padilla, J.N.; Tanguilig III, B.T. Portable automated bag-valve mask with android technology. Int. J. Adv. Technol. Eng. Explor. 2016, 3, 28. [Google Scholar] [CrossRef]
- MIT Emergency Ventilator (E-Vent). Available online: https://e-vent.mit.edu/ (accessed on 12 August 2020).
- Kwon, A.H.; Slocum, A.H.; Varelmann, D.; Nabzdyk, C.G. Rapidly scalable mechanical ventilator for the COVID-19 pandemic. Intensive Care Med. 2020, 46, 1642–1644. [Google Scholar] [CrossRef] [PubMed]
- PROJEKT DIY-BEATMUNGSGERÄT. Available online: http://diy-beatmungsgerat.de/ (accessed on 27 September 2020).
- Emergency Ventilator Response: Spiro Wave. 2020. Available online: https://ventilatorresponse.com/ (accessed on 27 September 2020).
- Buytaert, J.; Abud, A.A.; Akiba, K.; Bay, A.; Bertella, C.; Bowcock, T.; Byczynski, W.; Coco, V.; Collins, P.; Francisco, O.; et al. The HEV Ventilator Proposal ventilator proposal. arXiv 2020, arXiv:2004.00534. [Google Scholar]
- Williams, L.M.; Sharma, S. Ventilator Safety. In StatPearls [Internet]; StatPearls Publishing: Tampa/St. Petersburg, FL, USA, 2019. [Google Scholar]
- Bezzant, T.B.; Mortensen, J. Risks and hazards of mechanical ventilation: A collective review of published literature. Disease-a-Month 1994, 40, 585–638. [Google Scholar] [CrossRef]
- Ates, A.; Arikan, M.; Ozgok, A. Use of Bag Valve Device Resulting in Pneumothorax. J. Case Rep. 2017, 6, 595–597. [Google Scholar] [CrossRef]
- Whipp, B. Pulmonary ventilation. In Comprehensive Human Physiology; Springer: Berlin, Germany, 1996; pp. 2015–2036. [Google Scholar]
- Overview of Initiating Invasive Mechanical Ventilation in Adults in the Intensive Care Unit. Available online: https://www.uptodate.com/contents/overview-of-initiating-invasive-mechanical-ventilation-in-adults-in-the-intensive-care-unit (accessed on 5 April 2020).
- Health and Human Services Department. Declaration Under the Public Readiness and Emergency Preparedness Act for Medical Countermeasures Against COVID-19, 17 March 2020. Fed. Regist. 2020, 85, 15198. [Google Scholar]
- Evita V800/V600 Intensive Care Ventilator: Instructions for Use. Available online: www.draeger.com/Products/Content/evita-v800-v600-sw-1n-ifu-9055601-en.pdf (accessed on 28 September 2020).




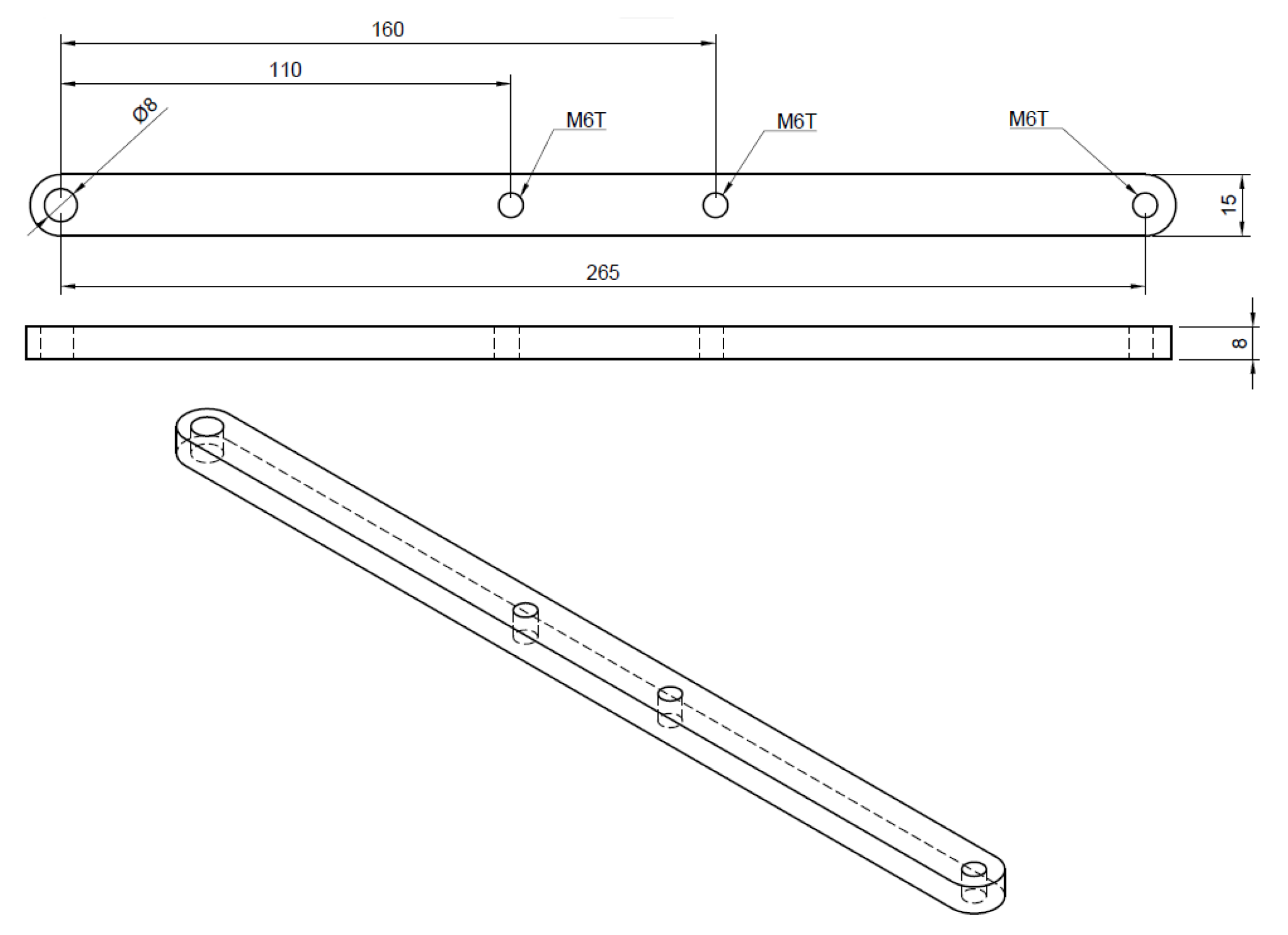
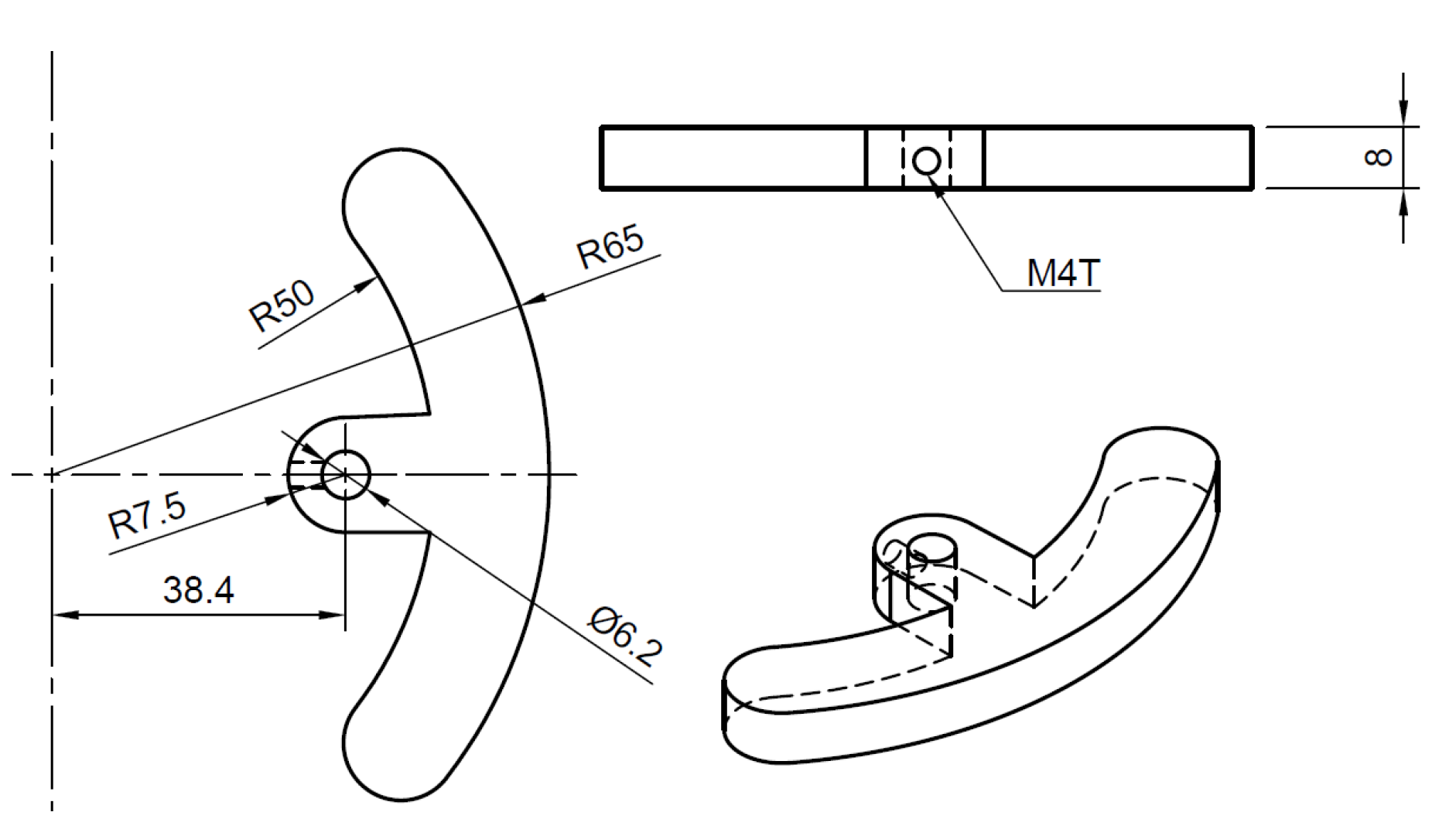

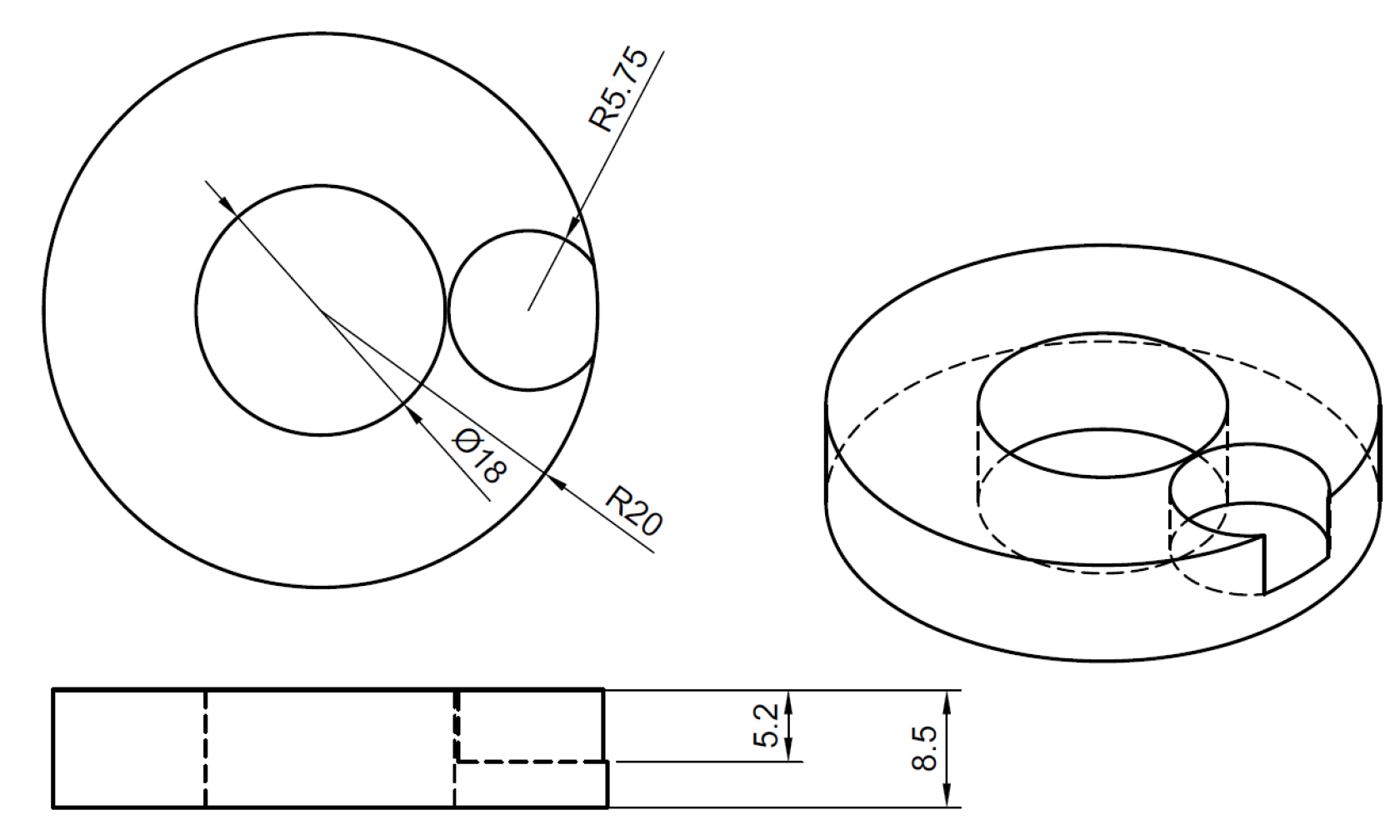
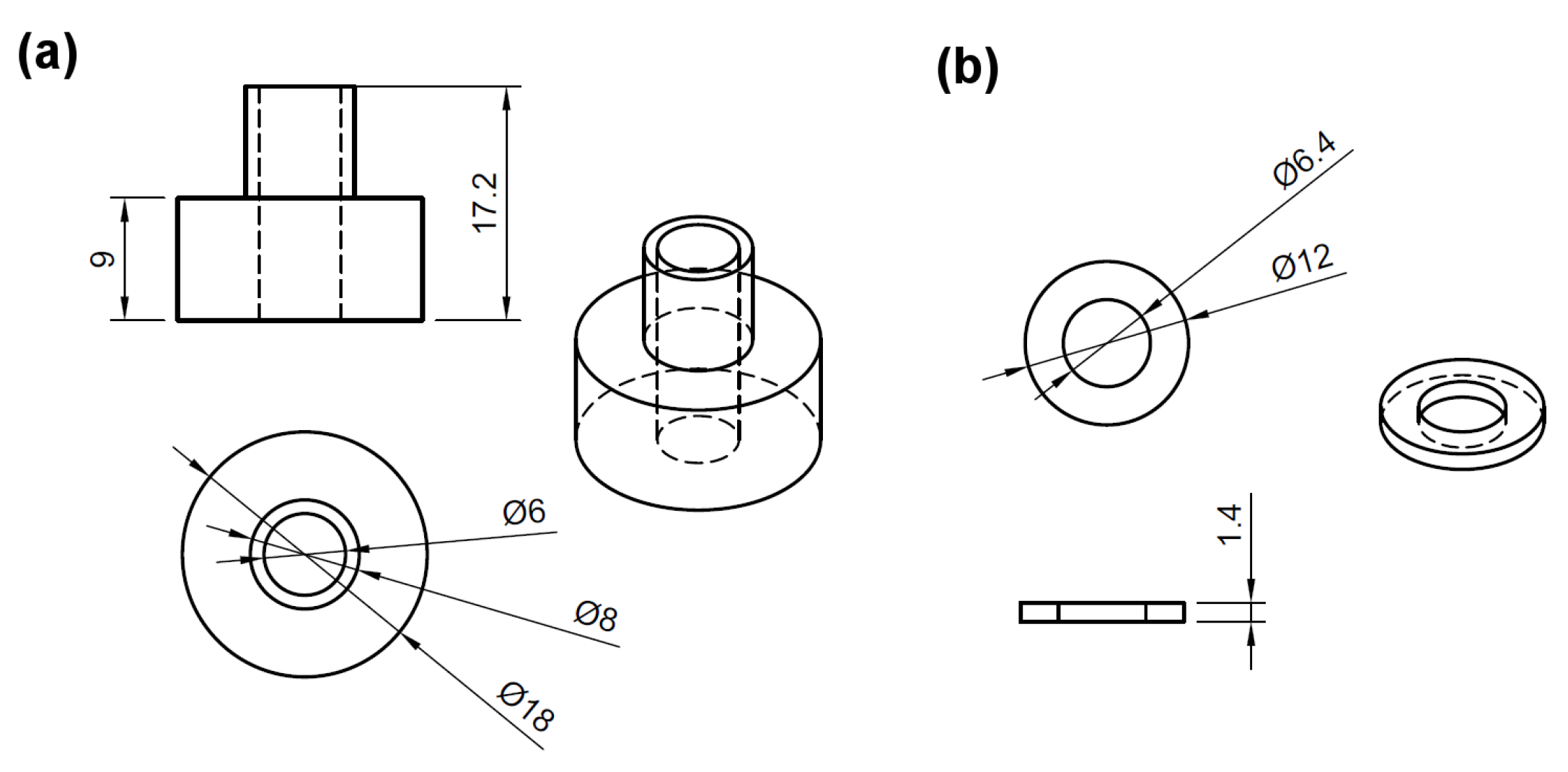
| Parameter | Value |
|---|---|
| Tidal volume | 6–8 mL/kg PBW |
| Rate | 2–16 BPM *, for ARDS ** up to 35 BPM * |
| PEEP | 5–10 cmHO |
| FiO2 | Adjusted to obtain SpO2 > 90% |
| Inspiratory flow | 40–60 L/min |
| Insp:exp ratio | 1:1–1:3 |
| PSV | 5–10 cmHO |
| Insp. pressure | 12–25 cmHO (typ.) |
| Trigger pressure | −2 to −1 cmHO |
| Trigger flow | 2 L/min |
| Parameter | Typical (ARDS) | Evita | CLAPER | eARM |
|---|---|---|---|---|
| Tidal volume (mL) | 100–800 | 2–3000 | 227, 338, 460, 595, 722 | 0–800 |
| Rate (BPM) | 2–35 | 0.5–98 | 15–24 | 8–30 |
| PEEP (cm HO) | 5–10 | 0–50 | 0–40 | 0–40 |
| FiO2 (%) | 0–90 | 21–100 | E/O | E/O |
| Inspiratory flow (L/min) | 40–60 | 2–120 | 30–56 | 21–49 |
| Insp:exp ratio | 1:1–1:3 | 600:1–1:300 | 1:1 | 1:1, 1:2 |
| PSV (cm HO) | 5–10 | 0–95 | 0–51 | 0–44 |
| Insp. pressure (cm HO) | 12–25 | 0–95 | 10–45 | 10–100 |
| Trigger pressure (cm HO) | −2–−1 | N/A | N/A | −2–0 |
| Trigger flow (L/min) | 2 | 0.2–15 | N/A | N/A |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castro-Camus, E.; Ornik, J.; Mach, C.; Hernandez-Cardoso, G.; Savalia, B.; Taiber, J.; Ruiz-Marquez, A.; Kesper, K.; Konde, S.; Sommer, C.; et al. Simple Ventilators for Emergency Use Based on Bag-Valve Pressing Systems: Lessons Learned and Future Steps. Appl. Sci. 2020, 10, 7229. https://doi.org/10.3390/app10207229
Castro-Camus E, Ornik J, Mach C, Hernandez-Cardoso G, Savalia B, Taiber J, Ruiz-Marquez A, Kesper K, Konde S, Sommer C, et al. Simple Ventilators for Emergency Use Based on Bag-Valve Pressing Systems: Lessons Learned and Future Steps. Applied Sciences. 2020; 10(20):7229. https://doi.org/10.3390/app10207229
Chicago/Turabian StyleCastro-Camus, Enrique, Jan Ornik, Cornelius Mach, Goretti Hernandez-Cardoso, Bhushan Savalia, Jochen Taiber, Armando Ruiz-Marquez, Karl Kesper, Srumika Konde, Caroline Sommer, and et al. 2020. "Simple Ventilators for Emergency Use Based on Bag-Valve Pressing Systems: Lessons Learned and Future Steps" Applied Sciences 10, no. 20: 7229. https://doi.org/10.3390/app10207229
APA StyleCastro-Camus, E., Ornik, J., Mach, C., Hernandez-Cardoso, G., Savalia, B., Taiber, J., Ruiz-Marquez, A., Kesper, K., Konde, S., Sommer, C., Wiener, J., Geisel, D., Hüppe, F., Kräling, G., Mross, P., Nguyen, J., Wiesmann, T., Beutel, B., & Koch, M. (2020). Simple Ventilators for Emergency Use Based on Bag-Valve Pressing Systems: Lessons Learned and Future Steps. Applied Sciences, 10(20), 7229. https://doi.org/10.3390/app10207229





