Cytotoxicity and UV Light Absorption in Biopolymeric Membranes from Native Vegetation of Mexico
Abstract
1. Introduction
2. Materials and Methods
2.1. Agavaceous Materials for the Conformation of the Membranes
2.1.1. Obtaining Fibrous Material
2.1.2. Obtaining Opuntia ficus-indica Mucilage
2.1.3. Obtaining Aloe barbadensis Miller Mucilage
2.1.4. Obtaining Fructans
2.2. Obtaining Lignocellulosic Biopolymer Membranes
2.3. Cytotoxicity Test by ISO: 10993-5: 2009
2.3.1. Cell Culture
2.3.2. Cytotoxicity Test
2.3.3. Staining with Sulphordamine B (SRB)
2.3.4. Viability
- is the average OD of the respective groups that were in contact with different LBMs.
- is the average OD of all the wells of the control group. All values are final ODs after subtraction of background absorbance.
2.4. Assessment of Cytotoxicity According to ISO: 10993-5: 2009
2.4.1. Quantitative Evaluation
2.4.2. Qualitative Evaluation
2.4.3. Environmental Scanning Electron Microscopy
2.4.4. Fourier Transform Infrared Spectroscopy
2.4.5. Adsorption of Ultraviolet Radiation
2.4.6. Image Texture Analysis
3. Results
3.1. Assessment of Cytotoxicity According to ISO: 10993-5: 2009
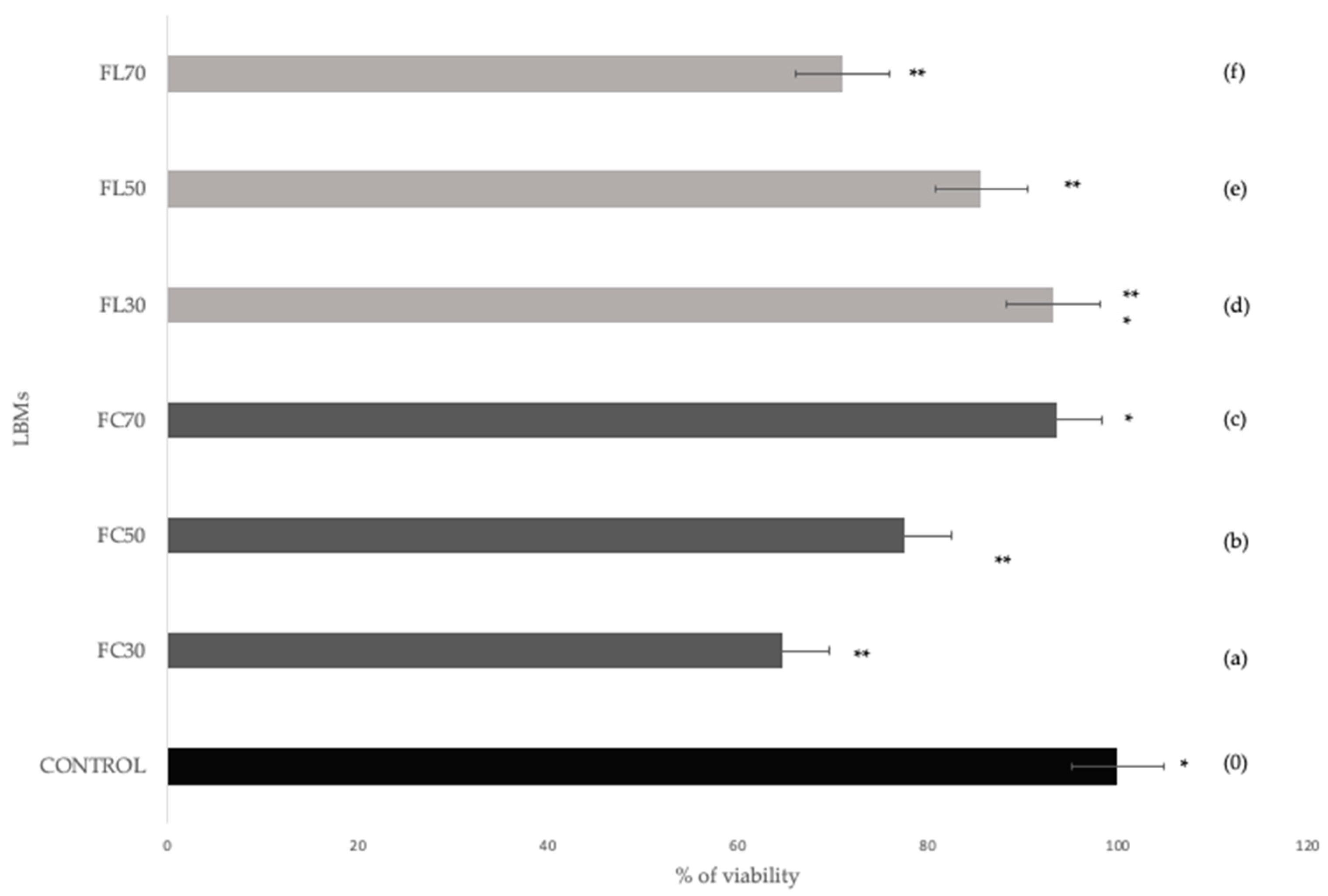
3.1.1. Quantitative Evaluation
3.1.2. Qualitative Evaluation
3.2. Environmental Scanning Electron Microscopy
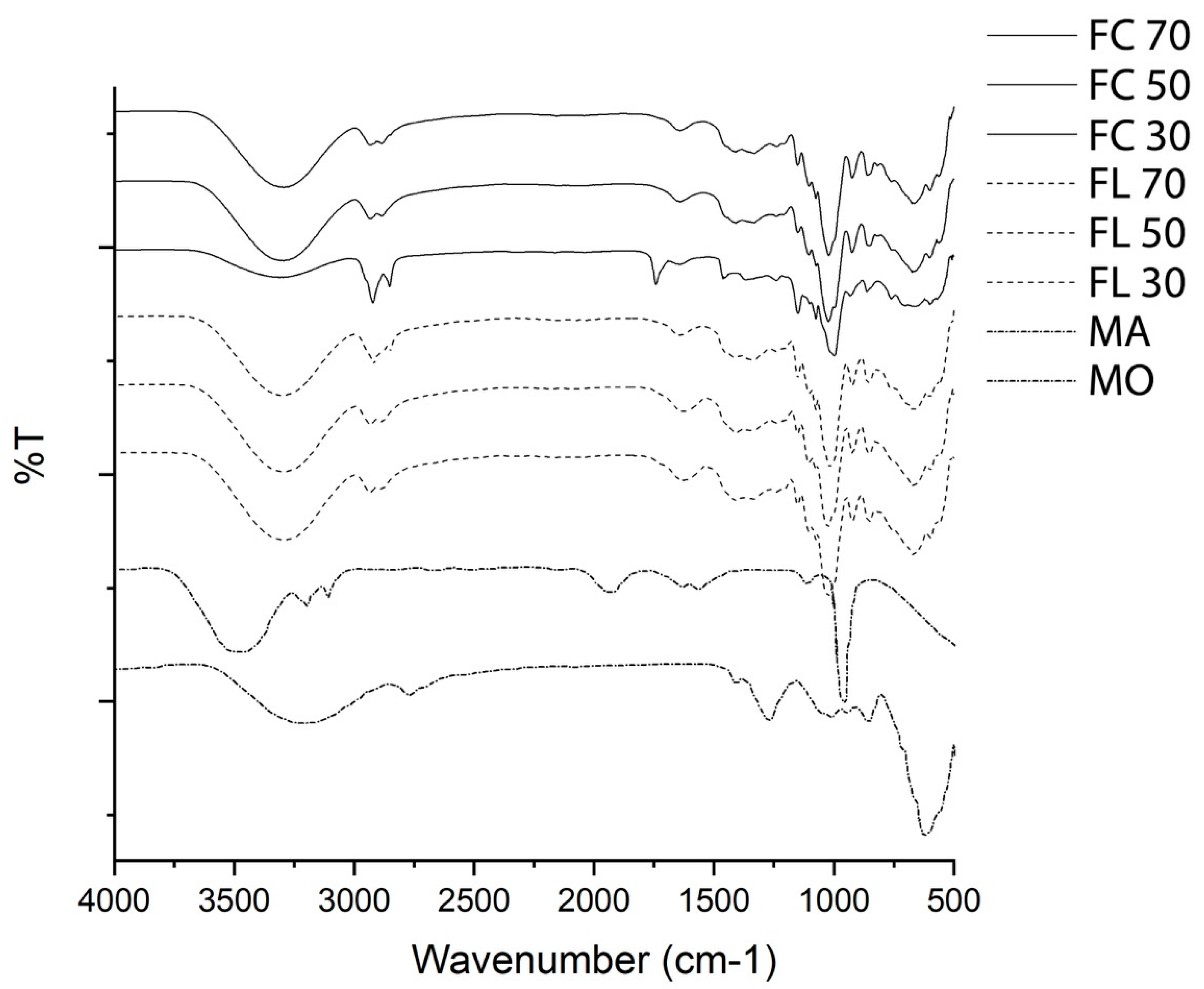
3.3. Fourier Transform Infrared Spectroscopy
3.4. Adsorption of Ultraviolet Radiation
3.5. Image Texture Analysis
4. Discussion
4.1. Assessment of Cytotoxicity According to ISO: 10993-5: 2009
Quantitative and Qualitative Evaluation
4.2. Environmental Scanning Electron Microscopy
4.3. Fourier Transform Infrared Spectroscopy (FTIR)
4.4. Adsorption of Ultraviolet Radiation
4.5. Analysis of Image Texture
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Oviedo Prieto, R.; Herrera Oliver, P.; Caluff, M.G. National list of invasive and potentially invasive plants in the Republic of Cuba 2011. Bissea Boletín Sobre Conservación Plantas Jardín Botánico Nacional Cuba 2012, 6, 22–96. [Google Scholar]
- De Rzedowski, G.C.; Rzedowski y colaboradores, J. Flora fanerogámica del Valle de México, 2nd ed.; y Comisión Nacional para el Conocimiento y Uso de la Biodiversidad: Pátzcuaro, Michoacán, México, 2005; p. 1406. [Google Scholar]
- Olivera, A.; Caballero Caballero, M.; Alavez Ramírez, R. Biocomposite tepexil cement reinforced with fibers of Agave angustifolia Haw as a light mortar. Rev. Mex. Cienc Agric. 2018, 21, 4406–4415. [Google Scholar] [CrossRef]
- Agrawal, C.; Ong, J.; Appleford, M.; Mani, G. Introduction to Biomaterials: Basic Theory with Engineering Applications; Cambridge University Press: Cambridge, UK, 2019; pp. 19–47. [Google Scholar]
- Pignatello, R. PLGA-Alendronate Conjugate as a New Biomaterial to Produce Osteotropic Drug Nanocarriers; Biomaterials Applications for Nanomedicine; InTech: Milan, Italy, 2011; pp. 165–184. [Google Scholar] [CrossRef]
- Sunita, V.; Shivaram, S.; Sharma, C. Recent Advances in Biomaterials Science and Engineering Research in India: A Minireview. ACS Biomater. Sci. Eng. 2019, 5, 3–18. [Google Scholar] [CrossRef]
- Parida, P.; Behera, A.; Chandra Mishra, S. Classification of Biomaterials used in Medicine. Int. J. Adv. Eng. Sci. Appl. Math. 2012, 1, 125–129. [Google Scholar] [CrossRef]
- Vert, M. Biopolymers and Artificial Biopolymers in Biomedical Applications, an Overview. In Biorelated Polymers; Chiellini, E., Gil, H., Braunegg, G., Buchert, J., Gatenholm, P., Van Der Zeem, M., Eds.; Springer: Boston, MA, USA, 2001; pp. 63–79. [Google Scholar]
- Velasco, M.; Narvaez-Tovar, C.; Garzon-Alvarado, D. Design, Materials, and Mechanobiology of Biodegradable Scaffolds for Bone Tissue Engineering. Biomed. Res. Int. 2015, 2015, 1–21. [Google Scholar] [CrossRef]
- Momoh, F.U.; Boateng, J.S.; Richardson, S.C.W.; Chowdhry, B.Z.; Mitchell, J.C. Development and functional characterization of alginate dressing as potential protein delivery system for wound healing. Int. J. Biol. Macromol. 2015, 81, 137–150. [Google Scholar] [CrossRef]
- Pina, S.; Ribeiro, V.; Marques, C.F.; Maia, F.R.; Silva, T.H.; Reis, R.L.; Oliveira, J.M. Scaffolding Strategies for Tissue Engineering and Regenerative Medicine Applications. Materials 2019, 12, 1824. [Google Scholar] [CrossRef]
- Sikareepaisan, P.; Ruktanonchai, U.; Supaphol, P. Preparation and characterization of asiaticoside-loaded alginate films and their potential for use as effectual wound dressings. Carbohydr. Polym. 2011, 83, 1457–1469. [Google Scholar] [CrossRef]
- Vainio, H.; Bianchini, F. Cancer-preventive effects of sunscreens are uncertain. Scand. J. Work Environ. Health 2000, 26, 529–531. [Google Scholar] [CrossRef]
- Ebrahimzad, M.A.; Enayatifard, R.; Khalili, M.; Ghaffarloo, M.; Saeedi, M.; Charati, J.Y. Correlation between Sun Protection Factor and Antioxidant Activity, Phenol and Flavonoid Contents of some Medicinal Plants. Iran. J. Pharm. Res. 2014, 13, 1041–1047. [Google Scholar] [CrossRef]
- Gonzalez-Pumariega, M.; Vernhes Tamayo, M.; Sanchez-Lamar, A. Ultraviolet radiation and its incidence in the human health. Theoria 2009, 18, 69–80. [Google Scholar]
- Sordo, C.; Gutierrez, C. Skin cancer and sun radiation: Peruvian experience in the prevention and early detection of skin cancer and melanoma. Rev. Peru. Med. Exp. Salud Publica 2013, 30, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Yao, J.; Chen, H.; Chen, K.; Trebše, P.; Zaray, G. Comparative toxicity of chlorpyrifos and its oxon derivatives to soil microbial activity by combined methods. Chemosphere 2010, 78, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.O.; Etheridge, J.M.; Thompson, J.A.; Vorwald, C.E.; Dean, D.; Fisher, J.P. Evaluation of the In Vitro Cytotoxicity of Cross-Linked Biomaterials. Biomacromolecules 2013, 14, 1321–1329. [Google Scholar] [CrossRef]
- Loyzaga, G. Cultivo de Células Animales y Humanas: Aplicaciones En Medicina Regenerativa; Editorial Visión Libros: Madrid, Spain, 2011; Available online: https://books.google.com.mx/books?id=Z3t4R39FByYC (accessed on 28 February 2019).
- Willershausen, B.; Marroquin, B.B.; Schafer, D.; Schulze, R. Cytotoxicity of Root Canal Filling Materials to Three Different Human Cell Lines. J. Endod. 2000, 26, 703–707. [Google Scholar] [CrossRef]
- Liu, X.; Rodeheaver, D.P.; White, J.C.; Wright, A.M.; Walker, L.M.; Zhang, F.; Shannon, S. A comparison of in vitro cytotoxicity assays in medical device regulatory studies. Regul. Toxicol. Pharmacol. 2018, 27, 24–32. [Google Scholar] [CrossRef] [PubMed]
- International Standard ISO 10993-5. Biological Evaluation of Medical Devices—Part 5: Tests for In Vitro Cytotoxicity, 3rd ed.; ISO: Geneva, Switzerland, 2009; ISSN 0273-2300. [Google Scholar]
- Ghavami, K.; Toledo Filho, R.D.; Barbosa, N.P. Behaviour of composite soil reinforced with natural fibres. Cem. Concr. Compos. 1999, 21, 39–48. [Google Scholar] [CrossRef]
- Mohammed, L.; Ansari, M.N.M.; Pua, G.; Jawaid, M.; Islam, M.S. A Review on Natural Fiber Reinforced Polymer Composite and Its Applications. Int. J. Polym. Sci. 2015, 2015, 1–15. [Google Scholar] [CrossRef]
- Lopez, K.M.; Ravula, S.; Pérez, R.L.; Ayala, C.E.; Losso, J.N.; Janes, M.E.; Warner, I.M. Hyaluronic Acid–Cellulose Composites as Patches for Minimizing Bacterial Infections. ACS Omega 2020, 5, 4125–4132. [Google Scholar] [CrossRef]
- Anton-Sales, I.; Beekmann, U.; Laromaine, A.; Roig, A.; Kralisch, D. Opportunities of Bacterial Cellulose to Treat Epithelial Tissues. Curr. Drug Targets 2019, 20, 808–822. [Google Scholar] [CrossRef]
- Vargas-Rodríguez, L.; Arroyo Figueroa, G.; Herrera Méndez, C.H.; Pérez Nieto, A.; García Vieyra, M.I.; Rodríguez Núñez, J.R. Propiedades físicas del mucílago de nopal. Acta Univ. 2016, 26, 8–11. [Google Scholar] [CrossRef]
- Espinosa-Andrews, H.; Urias-Silvas, J.E. Thermal properties of agave fructans (Agave tequilana Weber var. Azul). Carbohydr. Polym. 2012, 87, 2671–2676. [Google Scholar] [CrossRef]
- Le Moigne, N.; Otazaghine, B.; Corn, S.; Angellier-Coussy, H.; Bergeret, A. Surfaces and Interfaces in Natural Fibre Reinforced Composites; Springer International Publishing: Cham, Switzerland, 2018. [Google Scholar] [CrossRef]
- Skehan, P.; Storeng, R.; Scudiero, D.; Monks, A.; McMahon, J.; Vistica, D.; Warren, J.T.; Bokesch, H.; Kenney, S.; Boyd, M.R. New Colorimetric Cytotoxicity Assay for Anticancer-Drug Screening. JNCI J. Natl. Cancer Inst. 1990, 82, 1107–1112. [Google Scholar] [CrossRef] [PubMed]
- Moya, L.; Zakeri, H.; Yamazaki, F.; Liu, W.; Mas, E.; Koshimura, S. 3D gray level co-occurrence matrix and its application to identifying collapsed buildings. ISPRS J. Photogramm. Remote Sens. 2019, 149, 14–28. [Google Scholar] [CrossRef]
- Arzate Vazquez, I. Application of Image Texture Analysis for the Quantitative Characterization of Biological Surfaces. Ph.D. Thesis, Instituto Politecnico Nacional, México City, México, 2011. [Google Scholar]
- Presutti, M. La matriz de co-ocurrencia en la clasificación multiespectral: Tutorial para la enseñanza de medidas texturales en cursos de grado universitario. In 4a Jornada de Educação Em Sensoriamento Remoto No Âmbito Do Mercosul; Recursos Didáticos para o Ensino de Sensoriamento Remoto: São Leopoldo, Brasil, 2004; pp. 1–9. [Google Scholar]
- Hall-Beyer, M. Practical guidelines for choosing GLCM textures to use in landscape classification tasks over a range of moderate spatial scales. Int. J. Remote Sens. 2017, 38, 1312–1338. [Google Scholar] [CrossRef]
- Batchelor, B.; Waltz, F. Intelligent Machine Vision—Techniques, Implementations and Applications; Springer: London, UK, 2001; pp. 61–62. [Google Scholar]
- Rodríguez, J.; Prieto, S.; Ortiz, L.; Wiesner, C.; Díaz, M.; Correa, C. Descripción matemática con dimensiones fractales de células normales y con anormalidades citológicas de cuello uterino. Rev. Cienc. Salud 2006, 4, 58–63. [Google Scholar]
- Jat, M.K.; Garg, P.K.; Khare, D. Monitoring and modelling of urban sprawl using remote sensing and GIS techniques. Int. J. Appl. Earth Obs. Geoinf. 2008, 10, 26–43. [Google Scholar] [CrossRef]
- Chen, W.-S.; Yuan, S.-Y.; Hsieh, C.-M. Two algorithms to estimate fractal dimension of gray-level images. Opt. Eng. 2003, 42, 2452–2465. [Google Scholar] [CrossRef]
- Rezwan, K.; Chen, Q.Z.; Blaker, J.J.; Boccaccini, A.R. Biodegradable and bioactive porous polymer/inorganic composite scaffolds for bone tissue engineering. Biomaterials 2006, 27, 3413–3431. [Google Scholar] [CrossRef]
- Haralick, R.M.; Dinstein, I.; Shanmugam, K. Textural Features for Image Classification. IEEE Trans. Syst. Man Cybern. 1973, 6, 610–621. [Google Scholar] [CrossRef]
- Korać, R.; Khambholja, K. Potential of herbs in skin protection from ultraviolet radiation. Pharmacogn. Rev. 2011, 5, 164. [Google Scholar] [CrossRef] [PubMed]
- Domíguez-Fernánde, R.N.; Arzate-Vázquez, I.; Chanona-Pérez, J.J.; Welti-Chanes, J.S.; Alvarado-González, J.S.; Calderon-Dominguez, G.; Garibay-Febles, V.; Gutierrez-Lopez, G.F. El gel de aloe vera: Estructura, composición quimica, procesameinto, actividad biológica e importancia en la industria farmacéutica y alimentaria. Rev. Mex. Ing. Qum. 2012, 11, 23–43. [Google Scholar]
- Rivero, R.; Alustiza, F.; Capella, V.; Liaudat, C.; Rodriguez, N.; Bosch, P.; Barbero, C.; Rivarola, C. Physicochemical properties of ionic and non-ionic biocompatible hydrogels in water and cell culture conditions: Relation with type of morphologies of bovine fetal fibroblasts in contact with the surfaces. Colloids Surf. B Biointerfaces 2017, 158, 488–497. [Google Scholar] [CrossRef] [PubMed]
- Howard, R.L.; Abotsi, E.; Jansen van, R.E.L.; Howard, S. Lignocellulose biotechnology: Issues of bioconversion and enzyme production. Afr. J. Biotechnol. 2003, 2, 602–619. [Google Scholar] [CrossRef]
- Grolik, M.; Szczubiałka, K.; Wowra, B.; Dobrowolski, D.; Orzechowska-Wylęgała, B.; Wylęgała, E.; Nowakowska, M. Hydrogel membranes based on genipin-cross-linked chitosan blends for corneal epithelium tissue engineering. J. Mater. Sci. Mater. Med. 2012, 23, 1991–2000. [Google Scholar] [CrossRef]
- Gao, L.; Gan, H.; Meng, Z.; Gu, R.; Wu, Z.; Zhang, L.; Zhu, X.; Sun, W.; Li, J.; Zheng, Y.; et al. Effects of genipin cross-linking of chitosan hydrogels on cellular adhesion and viability. Colloids Surf. B Biointerfaces 2014, 117, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Gürbay, A.; Gonthier, B.; Barret, L.; Favier, A.; Hıncal, F. Cytotoxic effect of ciprofloxacin in primary culture of rat astrocytes and protection by Vitamin E. Toxicology 2007, 229, 54–61. [Google Scholar] [CrossRef]
- Cisneros-López, E.O.; Anzaldo, J.; Fuentes-Talavera, F.J.; González-Núñez, R.; Robledo-Ortíz, J.R.; Rodrigue, D. Effect of agave fiber surface treatment on the properties of polyethylene composites produced by dry-blending and compression molding. Polym. Compos. 2017, 38, 96–104. [Google Scholar] [CrossRef]
- Cacicedo, M.L.; Islan, G.A.; Drachemberg, M.F.; Alvarez, V.A.; Bartel, L.C.; Bolzán, A.C.; Castro, G.R. Hybrid bacterial cellulose-pectin films for delivery of bioactive molecules. RSC New J. Chem. 2018, 42, 7457–7467. [Google Scholar] [CrossRef]
- Sampath, U.; Ching, Y.; Chuah, C.; Sabariah, J.; Lin, P.-C. Fabrication of Porous Materials from Natural/Synthetic Biopolymers and Their Composites. Materials 2016, 9, 991. [Google Scholar] [CrossRef]
- Miao, X.; Sun, D. Graded/Gradient Porous Biomaterials. Materials 2009, 3, 26–47. [Google Scholar] [CrossRef]
- Gavillon, R.; Budtova, T. Aerocellulose: New Highly Porous Cellulose Prepared from Cellulose−NaOH Aqueous Solutions. Biomacromolecules 2008, 9, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Capitani, D.; Crescenzi, V.; De Angelis, A.A.; Segre, A.L. Water in Hydrogels. An NMR Study of Water/Polymer Interactions in Weakly Cross-Linked Chitosan Networks. Macromolecules 2001, 34, 4136–4144. [Google Scholar] [CrossRef]
- Migneault, I.; Dartiguenave, C.; Bertrand, M.J.; Waldron, K.C. Glutaraldehyde: Behavior in Aqueous Solution, Reaction with Proteins, and Application to Enzyme Crosslinking. BioTechniques 2004, 37, 790–802. [Google Scholar] [CrossRef]
- Pereira, P.H.F.; Oliveira, T.Í.S.; Rosa, M.F.; Cavalcante, F.L.; Moates, G.K.; Wellner, N.; Waldron, K.W.; Azeredo, H.M. Pectin extraction from pomegranate peels with citric acid. Int. J. Biol. Macromol. 2016, 88, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Madera-Santana, T.J.; Vargas-Rodríguez, L.; Núñez-Colín, C.A.; González-García, G.; Peña-Caballero, V.; Núñez-Gastélum, J.A.; Gallegos-Vázquez, C.; Rodríguez-Núñez, J.R. Mucilage from cladodes of Opuntia spinulifera Salm-Dyck: Chemical, morphological, structural and thermal characterization. CyTA—J. Food 2018, 16, 650–657. [Google Scholar] [CrossRef]
- Femenia, A.; García-Pascual, P.; Simal, S.; Rosselló, C. Effects of heat treatment and dehydration on bioactive polysaccharide acemannan and cell wall polymers from Aloe barbadensis Miller. Carbohydr. Polym. 2003, 51, 397–405. [Google Scholar] [CrossRef]
- Camelo Caballero, L.R.; Wilches-Torres, A.; Cárdenas-Chaparro, A.; Gómez Castaño, J.A.; Otálora, M.C. Preparation and Physicochemical Characterization of Softgels Cross-Linked with Cactus Mucilage Extracted from Cladodes of Opuntia Ficus-Indica. Molecules 2019, 24, 2531. [Google Scholar] [CrossRef]
- Nizama, F.; Borja, N.; Rocha, G.; Infante, A.; Teixeira, B.; Casalino, A. Análisis por espectroscopía UV y FTIR de macerados acuosos y alcohólicos de Aloe vera L. y Aloe barbadensis Miller. Interacción con sales inorgánicas. Rev. Soc. Química Perú 2010, 76, 242–260. [Google Scholar]
- Muthukumaran, P.; Divya, R.; Indhumathi, E.; Keerthika, C. Total phenolic and flavonoid content of membrane processed Aloe vera extract: A comparative study. Int. Food Res. J. 2018, 25, 1450–1456. [Google Scholar]
- Ray, A.; Ghosh, S. Chemometrics for Functional Group Distribution, and UV Absorption Potential of Aloe vera L. Gel at Different Growth Periods. Mater. Today Proc. 2018, 5, 22245–22253. [Google Scholar] [CrossRef]
- Duhoranimana, E.; Karangwa, E.; Lai, L.; Xu, X.; Yu, J.; Xia, S.; Zhang, X.; Muhoza, B.; Habinshuti, I. Effect of sodium carboxymethyl cellulose on complex coacervates formation with gelatin: Coacervates characterization stabilization and formation mechanism. Food Hydrocoll. 2017, 69, 111–120. [Google Scholar] [CrossRef]
- Kumar, M.S.; Datta, P.K.; Dutta Gupta, S. In vitro evaluation of UV opacity potential of Aloe vera L. gel from different germplasms. J. Nat. Med. 2009, 63, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Landry, L.G.; Chapple, C.; Last, R.L. Arabidopsis Mutants Lacking Phenolic Sunscreens Exhibit Enhanced Ultraviolet-B Injury and Oxidative Damage. Plant Physiol. 1995, 109, 1159–1166. [Google Scholar] [CrossRef] [PubMed]
- Maddocks-Jennings, W.; Wilkinson, J.M.; Shillington, D. Novel approaches to radiotherapy-induced skin reactions: A literature review. Complement. Ther. Clin. Pract. 2005, 11, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Rabe, J.H.; Mamelak, A.J.; McElgunn, P.J.S.; Morison, W.L.; Sauder, D.N. Photoaging: Mechanisms and repair. J. Am. Acad. Dermatol. 2006, 55, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Marionnet, C.; Tricaud, C.; Bernerd, F. Exposure to non-extreme solar UV daylight: Spectral characterization, effects on skin and photoprotection. Int. J. Mol. Sci. 2014, 16, 68–90. [Google Scholar] [CrossRef]
- Sadeghifar, H.; Venditti, R.; Jur, J.; Gorga, R.E.; Pawlak, J.J. Cellulose-Lignin Biodegradable and Flexible UV Protection Film. ACS Sustain. Chem. Eng. 2017, 5, 625–631. [Google Scholar] [CrossRef]
- Teacă, C.-A.; Roşu, D.; Bodîrlău, R.; Roşu, L. Structural changes in wood under artificial UV light irradiation determined by FTIR spectroscopy and color measurements—A brief review. BioResources 2013, 8, 1478–1507. [Google Scholar] [CrossRef]
- Villalobos, R.; Chanona, J.; Hernández, P.; Gutiérrez, G.; Chiralt, A. Gloss and transparency of hydroxypropyl methylcellulose films containing surfactants as affected by their microstructure. Food Hydrocoll. 2005, 19, 53–61. [Google Scholar] [CrossRef]
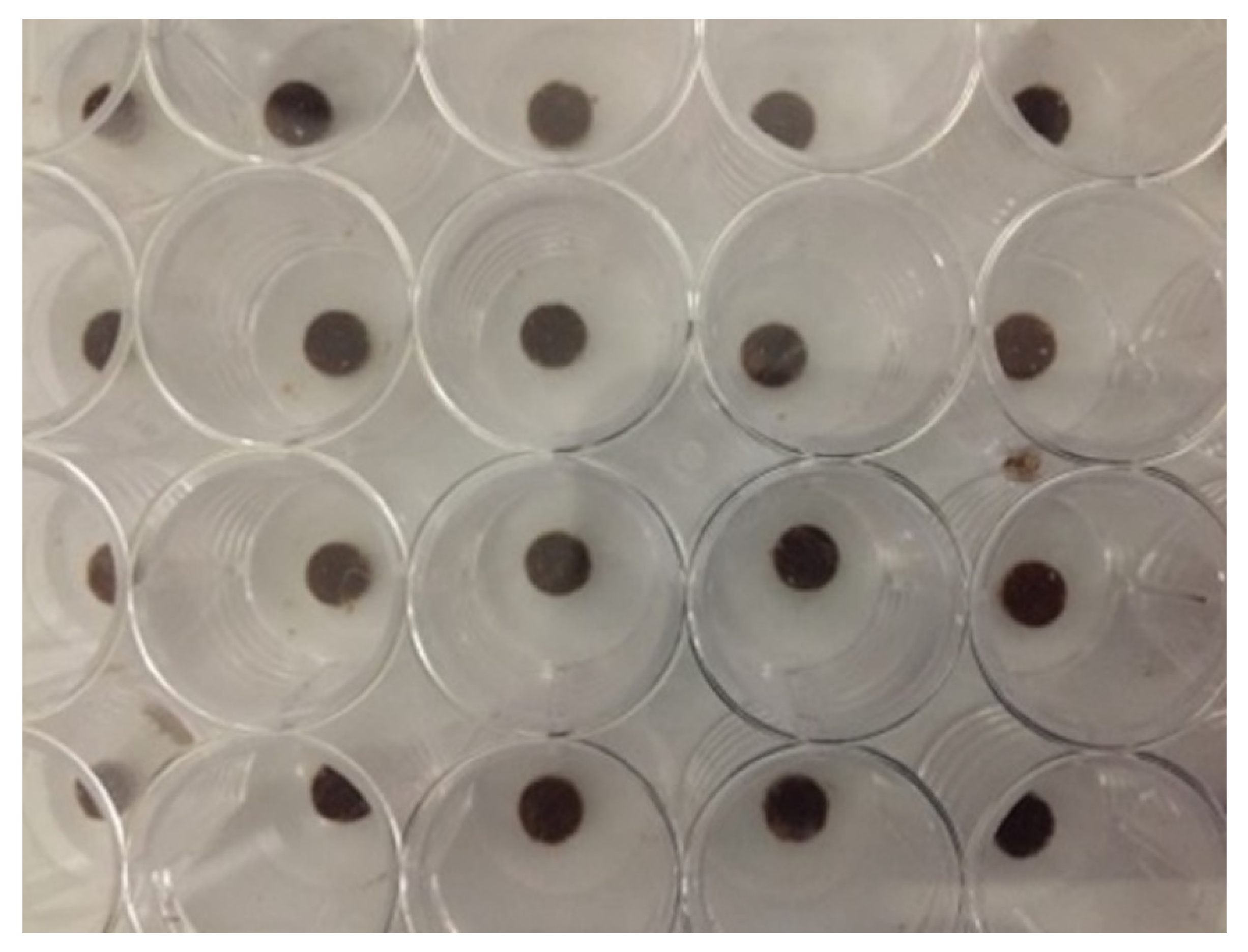


| Membranes | n | Composition | |||
|---|---|---|---|---|---|
| Fibrous Material (wt. %) | Mucilages (Opuntia ficus-indica and Aloe Barbadensis Miller) 1:1 (wt. %) | Fructans (wt. %) | Total Solids | ||
| FC30 | 3 | 30 | 39 | 31 | 100 |
| FC50 | 3 | 50 | 32 | 18 | 100 |
| FC70 | 3 | 70 | 25 | 5 | 100 |
| FL30 | 3 | 30 | 39 | 31 | 100 |
| FL50 | 3 | 50 | 32 | 18 | 100 |
| FL70 | 3 | 70 | 25 | 5 | 100 |
| Grade | Reactivity | Cell Conditions |
|---|---|---|
| 0 | None | Discrete intracytoplasmic granules, without cell lysis, without reduction of cell growth. |
| 1 | Slight | No more than 20% of the cells are round, freely bound and without intracytoplasmic granules, without changes in morphology; occasional lysed cells; a slight growth inhibition is observed. |
| 2 | Measured | No more than 50% of the cells are uniform, without intracytoplasmic granules, without extensive cell lysis; no more than 50% inhibition of observable growth. |
| 3 | Moderate | No more than 70% of the cell layers contain rounded cells or these are lysed; cell layers are not completely destroyed, but more than 50% growth inhibition is observed. |
| 4 | Severe | Almost complete or complete destruction of cell layers. |
| Membranes | Viability Media | Cytotoxicity |
|---|---|---|
| CONTROL | 100.00 a | Viable |
| FC30 | 62.12 b | Not viable |
| FC50 | 71.59 b | Viable |
| FC70 | 80.84 a | Viable |
| FL30 | 79.51 a,b | Viable |
| FL50 | 75.56 b | Viable |
| FL70 | 61.13 b | Not viable |
| Grade | Reactivity | Cell Conditions |
|---|---|---|
| 1 | Slight | No more than 20% of the cells are round, freely bound and without intracytoplasmic granules, without changes in morphology; occasional lysed cells; a slight growth inhibition is observed. |
| Wave Number (cm−1) | Vibration Mode | Functional Groups |
|---|---|---|
| 3600 and 3200 cm−1 | Stretching of the phenolic OH group | Specific phenols of aloe, including flavonoids, flavonols. |
| 2900–2936 cm−1 | C-H bond stretching of carbohydrates. Flexion belonging to C-O-H, C-O-C bonds | Oligosaccharides and polysaccharides. |
| 1608 cm−1 | Plane bending C = C bonds of lignin molecules. | Depending on the concentration and if it was LMB FC and FL. |
| 1870–1540 cm−1 | Ketones carbonyl group | Ketones with possible keto enolic balances. |
| 650–1580 cm−1 | Stretch = CO | Amino acids and polysaccharides. |
| 700–650 cm−1 | Acetyl groups | These grant biological activity |
| 1185–1045 cm−1 | Stretch (CO, ether bond) and the pyranoid rings (CC) stretch | Stretching of glycosidic bonds (CO, ether bond) and pyranoid rings (CC) attributed to polysaccharides that are part of pectins and mucilage. |
| 3298 cm−1 | Stretching of the hydroxyl groups, -OH | Carboxylic acids, amino acids, alcohols, and water molecules. |
| Wavelength | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 365 nm UV Long | 302 nm UV Middle | 254 nm UV Short | |||||||||
| % Fiber | % Fiber | % Fiber | |||||||||
| 30 | 50 | 70 | 30 | 50 | 70 | 30 | 50 | 70 | |||
| Media | Media | Media | Media | Media | Media | Media | Media | Media | |||
| UV Absorbance | Conformation | FC * | 47.89 a | 34.89 b | 29.43 c | 50.21 a | 45.12 b | 37.23 c | 51.79 a | 52.01 a | 45.35 b |
| FL ** | 70.56 a | 62.90 b | 51.56 c | 75.21 a | 70.24 b | 59.12 c | 82.45 a | 81.98 a | 74.99 b | ||
| DFT | Contrast | Entropy | SMA | Entropy/DFT Relation | ||
|---|---|---|---|---|---|---|
| FL30 | 2.56215 a,b,c | 187.93 a,b | 8.9805 a,b | 0.000245 a,b | 3.50506 |  |
| FC30 | 2.6462 a | 311.902 a | 9.393 a | 0.000569 a,b | 3.54961 |  |
| FL50 | 2.5922 b | 214.2285 a,b | 9.042 b | 0.000381 a,b | 3.48815 | 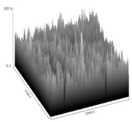 |
| FC50 | 2.5896 a,b,c | 215.95 a,b | 9.1265 a,b | 0.00025 a,b | 3.52428 | 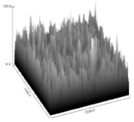 |
| FL70 | 2.5313 c | 183.5485 b | 8.996 b,c | 0.000182 a | 3.55390 | 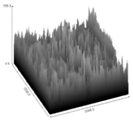 |
| FC70 | 2.5948 a,b,c | 222.6915 a,b | 9.157 a,b | 0.000238 b | 3.52898 |  |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
García de Alva Magos, M.A.; Santa-Olalla Tapia, J.; Ramos López, M.Á.; Molina Maturano, J.; Jiménez Aparicio, A.R.; Camacho Díaz, B.H.; García Serrano, L.A. Cytotoxicity and UV Light Absorption in Biopolymeric Membranes from Native Vegetation of Mexico. Appl. Sci. 2020, 10, 4995. https://doi.org/10.3390/app10144995
García de Alva Magos MA, Santa-Olalla Tapia J, Ramos López MÁ, Molina Maturano J, Jiménez Aparicio AR, Camacho Díaz BH, García Serrano LA. Cytotoxicity and UV Light Absorption in Biopolymeric Membranes from Native Vegetation of Mexico. Applied Sciences. 2020; 10(14):4995. https://doi.org/10.3390/app10144995
Chicago/Turabian StyleGarcía de Alva Magos, Mayela Alhelí, Jesús Santa-Olalla Tapia, Miguel Ángel Ramos López, Jessica Molina Maturano, Antonio Ruperto Jiménez Aparicio, Brenda Hildeliza Camacho Díaz, and Luz Arcelia García Serrano. 2020. "Cytotoxicity and UV Light Absorption in Biopolymeric Membranes from Native Vegetation of Mexico" Applied Sciences 10, no. 14: 4995. https://doi.org/10.3390/app10144995
APA StyleGarcía de Alva Magos, M. A., Santa-Olalla Tapia, J., Ramos López, M. Á., Molina Maturano, J., Jiménez Aparicio, A. R., Camacho Díaz, B. H., & García Serrano, L. A. (2020). Cytotoxicity and UV Light Absorption in Biopolymeric Membranes from Native Vegetation of Mexico. Applied Sciences, 10(14), 4995. https://doi.org/10.3390/app10144995








