Formulation Strategies to Improve Oral Bioavailability of Ellagic Acid
Featured Application
Abstract
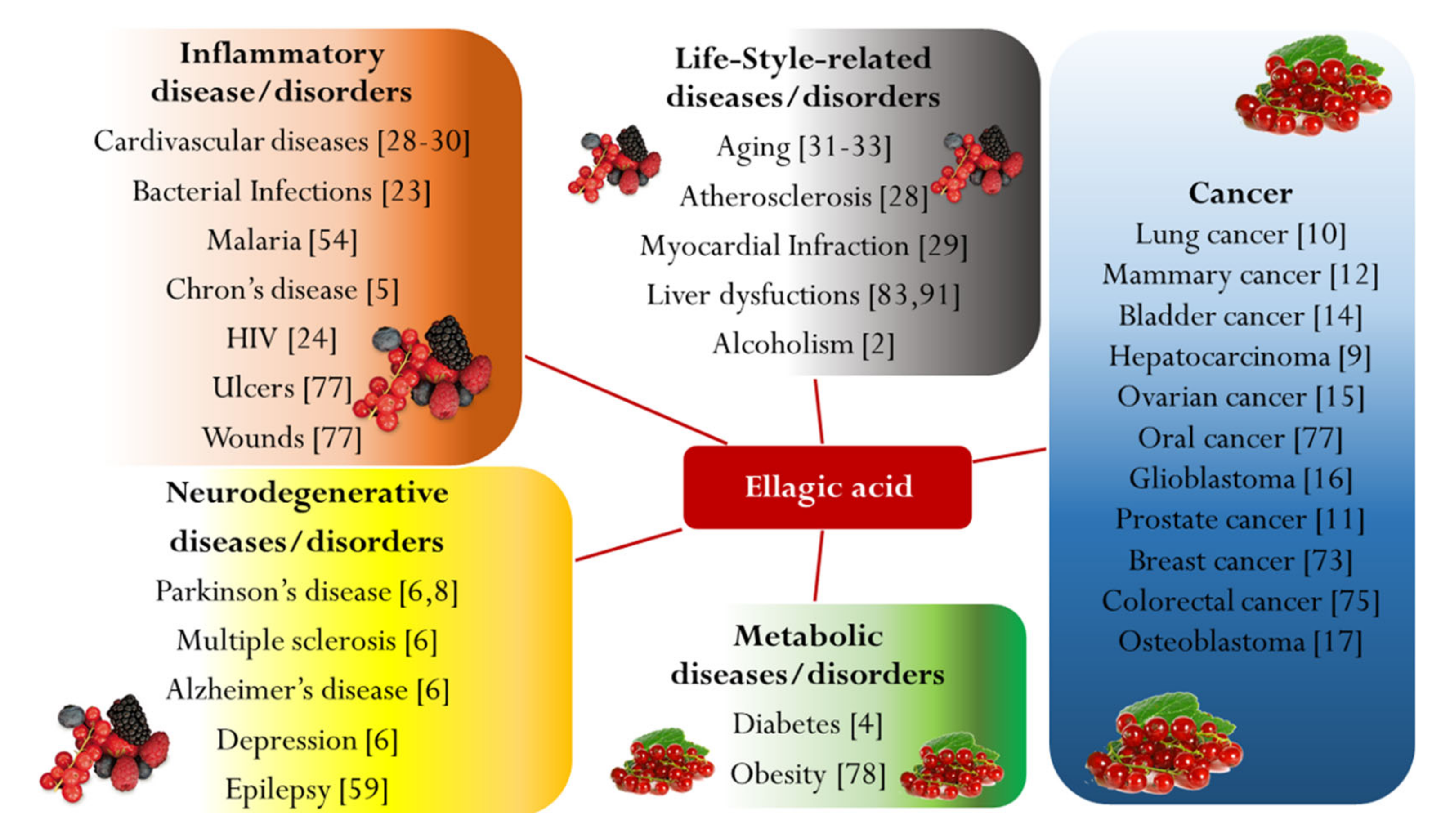
1. Introduction
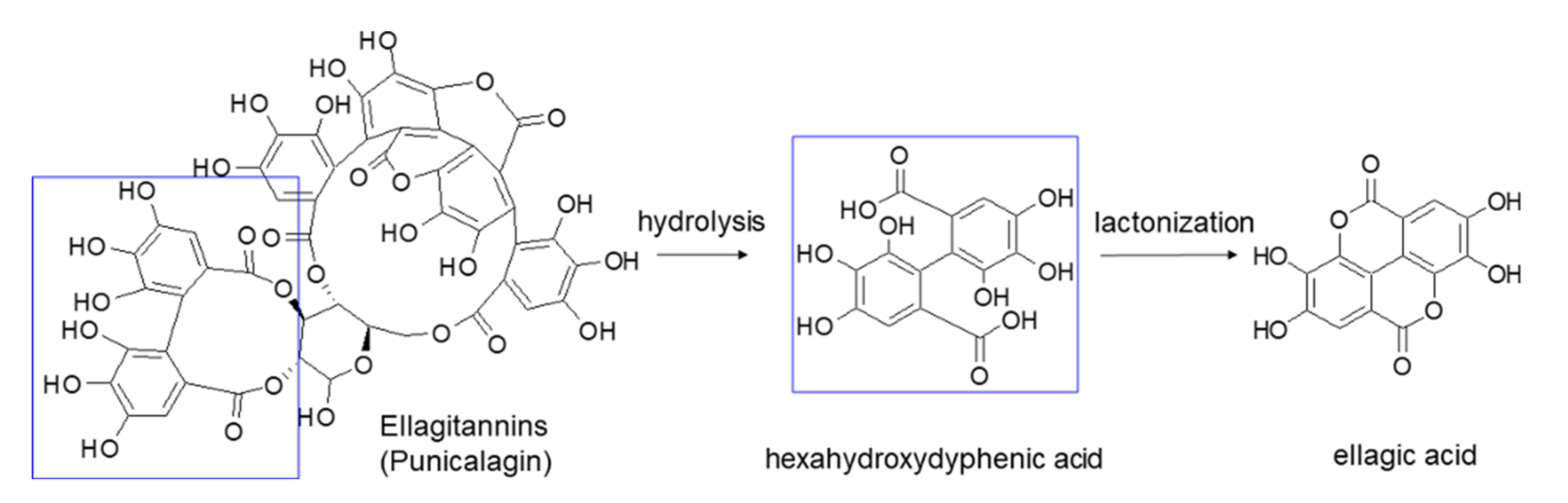
2. EA Chemical Structure and Solubility
3. EA Dietary Assumption
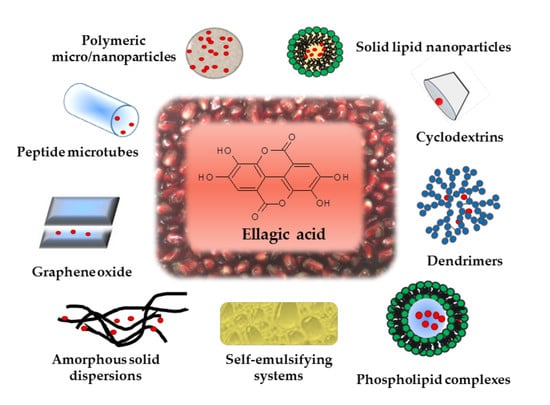
4. Formulation Strategies for Improving EA Oral Bioavailability
4.1. Micronized EA (m-EA)
4.2. EA in Spray Dried and Lyophilized Powders
4.3. Inclusion Complexes
4.3.1. EA Inclusion in Cyclodextrins (CDs)
4.3.2. EA Inclusion in Metalla-Cages
4.4. EA Encapsulated in Polymeric Carriers
4.4.1. Eudragit® Microspheres
4.4.2. Poly (Lactic-Co-Glycolic Acid) (PLGA) and Poly (ε-Caprolactone) (PCL) Nanospheres
4.4.3. Chitosan Micro/Nanospheres
4.4.4. Zein Nanocapsules
4.5. Dendrimers
4.6. Peptide Microtubes
4.7. Functionalized Graphene Oxide (GO) Carriers
4.8. Lipid-Based Carriers
4.8.1. Solid Lipid Nanoparticles (SLNs)
4.8.2. Liposomes (LPs)
4.8.3. Self-Emulsifying Delivery Systems (SNEDDS)
4.9. EA Formulations in Fixed Combination with Other Bioactive Molecules
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Miguel, M.G.; Neves, M.A.; Antunes, M.D. Pomegranate (Punica granatum L.): A medicinal plant with myriad biological properties—A short review. J. Med. Plants Res. 2010, 4, 2836–2847. [Google Scholar]
- Ríos, J.L.; Giner, R.M.; Marín, M.; Recio, M.C. A Pharmacological Update of Ellagic Acid. Planta Med. 2018, 84, 1068–1093. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Zhang, J.; Chen, N.G.; Shi, Z.; Qiu, J.; He, C.; Chen, M. Recent Advances in Anticancer Activities and Drug Delivery Systems of Tannins. Med. Res. Rev. 2017, 37, 665–701. [Google Scholar] [CrossRef] [PubMed]
- Ahangarpour, A.; Sayahi, M.; Sayahi, M. The antidiabetic and antioxidant properties of some phenolic phytochemicals: A review study. Diabetes Metab. Syndr. Clin. Res. 2019, 13, 854–857. [Google Scholar] [CrossRef]
- Baradaran Rahimi, V.; Ghadiri, M.; Ramezani, M.; Askari, V.R. Anti-inflammatory and anti-cancer activities of pomegranate and its constituent, ellagic acid: Evidence from cellular, animal, and clinical studies. Phytother. Res. 2020, in press. [Google Scholar] [CrossRef]
- Alfei, S.; Turrini, F.; Catena, S.; Zunin, P.; Grilli, M.; Pittaluga, A.M.; Boggia, R. Ellagic acid a multi-target bioactive compound for drug discovery in CNS? A narrative review. Eur. J. Med. Chem. 2019, 183, 111724. [Google Scholar] [CrossRef]
- Kang, I.; Buckner, T.; Shay, N.F.; Gu, L.; Chung, S. Improvements in Metabolic Health with Consumption of Ellagic Acid and Subsequent Conversion into Urolithins: Evidence and Mechanisms. Adv. Nutr. 2016, 7, 961–972. [Google Scholar] [CrossRef]
- Sarkaki, A.; Farbood, Y.; Dolatshahi, M.; Mansouri, S.M.; Khodadadi, A. Neuroprotective Effects of Ellagic Acid in a Rat Model of Parkinson’s Disease. Acta Med. Iran. 2016, 54, 494–502. [Google Scholar]
- Das, U.; Biswas, S.; Chattopadhyay, S.; Chakraborty, A.; Dey Sharma, R.; Banerji, A.; Dey, S. Radiosensitizing effect of ellagic acid on growth of Hepatocellular carcinoma cells: An in vitro study. Sci. Rep. 2017, 7, 14043. [Google Scholar] [CrossRef]
- Jeong, H.; Phan, A.N.H.; Choi, J.W. Anti-cancer Effects of Polyphenolic Compounds in Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitor-resistant Non-small Cell Lung Cancer. Pharmacogn. Mag. 2017, 13, 595–599. [Google Scholar] [CrossRef]
- Vanella, L.; Di Giacomo, C.; Acquaviva, R.; Barbagallo, I.; Li Volti, G.; Cardile, V.; Abraham, N.; Sorrenti, V. Effects of Ellagic Acid on Angiogenic Factors in Prostate Cancer Cells. Cancers 2013, 5, 726–738. [Google Scholar] [CrossRef] [PubMed]
- Dai, Z.; Nair, V.; Khan, M.; Ciolino, H.P. Pomegranate extract inhibits the proliferation and viability of MMTV-Wnt-1 mouse mammary cancer stem cells in vitro. Oncol. Rep. 2010, 24, 1087–1091. [Google Scholar] [CrossRef] [PubMed]
- Kowshik, J.; Giri, H.; Kishore, T.; Kesavan, R.; Vankudavath, R.; Reddy, G.; Dixit, M.; Nagini, S. Ellagic Acid Inhibits VEGF/VEGFR2, PI3K/Akt and MAPK Signaling Cascades in the Hamster Cheek Pouch Carcinogenesis Model. Anticancer Agents Med. Chem. 2014, 14, 1249–1260. [Google Scholar] [CrossRef] [PubMed]
- Ceci, C.; Tentori, L.; Atzori, M.; Lacal, P.; Bonanno, E.; Scimeca, M.; Cicconi, R.; Mattei, M.; de Martino, M.; Vespasiani, G.; et al. Ellagic Acid Inhibits Bladder Cancer Invasiveness and In Vivo Tumor Growth. Nutrients 2016, 8, 744. [Google Scholar] [CrossRef]
- Liu, H.; Zeng, Z.; Wang, S.; Li, T.; Mastriani, E.; Li, Q.H.; Bao, H.X.; Zhou, Y.J.; Wang, X.; Liu, Y.; et al. Main components of pomegranate, ellagic acid and luteolin, inhibit metastasis of ovarian cancer by down-regulating MMP2 and MMP9. Cancer Biol. Ther. 2017, 18, 990–999. [Google Scholar] [CrossRef]
- Wang, D.; Chen, Q.; Tan, Y.; Liu, B.; Liu, C. Ellagic acid inhibits human glioblastoma growth in vitro and in vivo. Oncol. Rep. 2017, 37, 1084–1092. [Google Scholar] [CrossRef]
- Xu, W.; Xu, J.; Wang, T.; Liu, W.; Wei, H.; Yang, X.; Yan, W.; Zhou, W.; Xiao, J. Ellagic acid and Sennoside B inhibit osteosarcoma cell migration, invasion and growth by repressing the expression of c-Jun. Oncol. Lett. 2018, 16, 898–904. [Google Scholar] [CrossRef]
- Ceci, C.; Lacal, P.; Tentori, L.; De Martino, M.; Miano, R.; Graziani, G. Experimental Evidence of the Antitumor, Antimetastatic and Antiangiogenic Activity of Ellagic Acid. Nutrients 2018, 10, 1756. [Google Scholar] [CrossRef]
- Umesalma, S.; Nagendraprabhu, P.; Sudhandiran, G. Ellagic acid inhibits proliferation and induced apoptosis via the Akt signaling pathway in HCT-15 colon adenocarcinoma cells. Mol. Cell. Biochem. 2015, 399, 303–313. [Google Scholar] [CrossRef]
- Goyal, Y.; Koul, A.; Ranawat, P. Ellagic acid ameliorates cisplatin induced hepatotoxicity in colon carcinogenesis. Environ. Toxicol. 2019, 34, 804–813. [Google Scholar] [CrossRef]
- Lin, M.; Yin, M. Preventive Effects of Ellagic Acid Against Doxorubicin-Induced Cardio-Toxicity in Mice. Cardiovasc. Toxicol. 2013, 13, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Sonaje, K.; Italia, J.L.; Sharma, G.; Bhardwaj, V.; Tikoo, K.; Kumar, M.N.V.R. Development of Biodegradable Nanoparticles for Oral Delivery of Ellagic Acid and Evaluation of Their Antioxidant Efficacy Against Cyclosporine A-Induced Nephrotoxicity in Rats. Pharm. Res. 2007, 24, 899–908. [Google Scholar] [CrossRef] [PubMed]
- Sepúlveda, L.; Ascacio, A.; Rodríguez-Herrera, R.; Aguilera-Carbó, A.; Aguilar, C.N. Ellagic acid biological properties and biotechnological development. Afr. J. Biotechnol. 2011, 10, 4518–4523. [Google Scholar] [CrossRef]
- Notka, F.; Meier, G.; Wagner, R. Concerted inhibitory activities of Phyllanthus amarus on HIV replication in vitro and ex vivo. Antivir. Res. 2004, 64, 93–102. [Google Scholar] [CrossRef]
- Govindarajan, R.; Vijayakumar, M.; Rao, C.V.; Shirwaikar, A.; Mehrotra, S.; Pushpangadan, P. Healing potential of Anogeissus latifolia for dermal wounds in rats. Acta Pharm. 2004, 54, 331–338. [Google Scholar]
- Giménez-Bastida, J.A.; González-Sarrías, A.; Larrosa, M.; Tomás-Barberán, F.; Espín, J.C.; García-Conesa, M.T. Intestinal ellagitannin metabolites ameliorate cytokine-induced inflammation and associated molecular markers in human colon fibroblasts. J. Agric. Food Chem. 2012, 60, 8866–8876. [Google Scholar] [CrossRef]
- Prabha, B.; Sini, S.; Priyadarshini, T.S.; Sasikumar, P.; Gopalan, G.; Jayesh, P.J.; Jithin, M.M.; Sivan, V.V.; Jayamurthy, P.; Radhakrishnan, K.V. Anti-inflammatory effect and mechanism of action of ellagic acid-3,3’,4-trimethoxy-4’-O-α-L-rhamnopyranoside isolated from Hopea parviflora in lipopolysaccharide-stimulated RAW 264.7 macrophages. Nat. Prod. Res. 2019, 12, 1–5. [Google Scholar] [CrossRef]
- Mele, L.; Mena, P.; Piemontese, A.; Marino, V.; López-Gutiérrez, N.; Bernini, F.; Brighenti, F.; Zanotti, I.; Del Rio, D. Antiatherogenic effects of ellagic acid and urolithins in vitro. Arch. Biochem. Biophys. 2016, 599, 42–50. [Google Scholar] [CrossRef]
- Jordão, J.B.R.; Porto, H.K.P.; Lopes, F.M.; Batista, A.C.; Rocha, M.L. Protective Effects of Ellagic Acid on Cardiovascular Injuries Caused by Hypertension in Rats. Planta Med. 2017, 83, 830–836. [Google Scholar] [CrossRef]
- Turrini, F.; Boggia, R.; Donno, D.; Parodi, B.; Beccaro, G.; Baldassari, S.; Signorello, M.G.; Catena, S.; Alfei, S.; Zunin, P. From pomegranate marcs to a potential bioactive ingredient: A recycling proposal for pomegranate squeezed-marcs. Eur. Food Res. Technol. 2019, 246, 273–285. [Google Scholar] [CrossRef]
- Kim, Y.H.; Kim, K.H.; Han, C.S.; Yang, H.C.; Park, S.H.; Jang, H.-I.; Kim, J.-W.; Choi, Y.-S.; Lee, N.H. Anti-wrinkle activity of Platycarya strobilacea extract and its application as a cosmeceutical ingredient. J. Cosmet. Sci. 2018, 14, 211–223. [Google Scholar]
- Liu, R.; Li, J.; Cheng, Y.; Huo, T.; Xue, J.; Liu, Y.; Liu, J.; Chen, X. Effects of ellagic acid-rich extract of pomegranates peel on regulation of cholesterol metabolism and its molecular mechanism in hamsters. Food Funct. 2015, 6, 780–787. [Google Scholar] [CrossRef]
- Boggia, R.; Turrini, F.; Villa, C.; Lacapra, C.; Zunin, P.; Parodi, B. Green extraction from pomegranate marcs for the production of functional foods and cosmetics. Pharmaceuticals 2016, 9, 63. [Google Scholar] [CrossRef] [PubMed]
- Núñez-Sánchez, M.A.; García-Villalba, R.; Monedero-Saiz, T.; GarcíaTalavera, N.V.; Gómez-Sánchez, M.B.; Sánchez-Álvarez, C.; García-Albert, A.M.; Rodríguez-Gil, F.J.; Ruiz-Marín, M.; Pastor-Quirante, F.A.; et al. Targeted metabolic profiling of pomegranate polyphenols and urolithins in plasma, urine and colon tissues from colorectal cancer patients. Mol. Nutr. Food Res. 2014, 58, 1199–1211. [Google Scholar] [CrossRef] [PubMed]
- Bellone, J.A.; Murray, J.R.; Jorge, P.; Fogel, T.G.; Kim, M.; Wallace, D.R.; Hartman, R.E. Pomegranate supplementation improves cognitive and functional recovery following ischemic stroke: A randomized trial. Nutr. Neurosci. 2019, 22, 738–743. [Google Scholar] [CrossRef] [PubMed]
- Bala, I.; Bhardwaj, V.; Hariharan, S.; Kumar, M.N. Analytical methods for assay of ellagic acid and its solubility studies. J. Pharm. Biomed. Anal. 2006, 40, 206–210. [Google Scholar] [CrossRef]
- Muzolf, M.; Szymusiak, H.; Gliszczynska-Swiglo, A.; Rietjens, I.M.C.M.; Tyrakowska, B. pH-Dependent radical scavenging capacity of green tea catechins. J. Agric. Food Chem. 2008, 56, 816–823. [Google Scholar] [CrossRef]
- Panichayupakaranant, P.; Itsuriya, A.; Sirikatitham, A. Preparation method and stability of ellagic acid-rich pomegranate fruit peel extract. Pharm. Biol. 2010, 48, 201–205. [Google Scholar] [CrossRef]
- Mao, X.; Wu, L.-F.; Zhao, H.; Liang, W.-Y.; Chen, W.-J.; Han, S.-X.; Qi, Q.; Cui, Y.-P.; Li, S.; Yang, G.-H.; et al. Transport of Corilagin, Gallic Acid, and Ellagic Acid from Fructus Phyllanthi Tannin Fraction in Caco-2 Cell Monolayers. Evid. Based Complementary Altern. Med. 2016, 2016, 9205379. [Google Scholar] [CrossRef] [PubMed]
- Mertens-talcot, S.U.; Jilma-Stohlawetz, P.; Rios, J.; Hingorani, L.; Derendorf, H. Absorption metabolism and antioxidant effects of pomegranate. J. Agric. Food Chem. 2006, 54, 8956–8961. [Google Scholar] [CrossRef]
- Seeram, N.P.; Henning, S.M.; Zhang, Y.; Suchard, M.; Li, Z.; Heber, D. Pomegranate juice ellagitannin metabolites are present in human plasma and some persist in urine for up to 48 hours. J. Nutr. 2006, 136, 2481–2485. [Google Scholar] [CrossRef] [PubMed]
- Tomás-Barberán, F.A.; García-Villalba, R.; González-Sarrías, A.; Selma, M.V.; Espín, J.C. Ellagic acid metabolism by human gut microbiota: Consistent observation of three urolithin phenotypes in intervention trials, independent of food source, age, and health status. J. Agric. Food Chem. 2014, 62, 6535–6538. [Google Scholar] [CrossRef] [PubMed]
- Tomás-Barberán, F.A.; González-Sarrias, A.; Garcıa-Villalba, R.; Nunez-Sanchez, M.A.; Selma, M.V.; Garcıa-Conesa, M.T.; Espın, J.C. Urolithins, the rescue of “old” metabolites to understand a “new” concept: Metabotypes as a nexus among phenolic metabolism, microbiota dysbiosis, and host health status. Mol. Nutr. Food Res. 2017, 61, 1500901. [Google Scholar] [CrossRef] [PubMed]
- Cortés-Martín, A.; García-Villalba, R.; González-Sarrías, A.; Romo-Vaquero, M.; Loria-Kohen, V.; Ramírez-de-Molina, A.; Tomás-Barberán, F.A.; Selma, M.V.; Espín, J.C. The gut microbiota urolithin metabotypes revisited: The human metabolism of ellagicacid is mainly determined by aging. Food Funct. 2018, 9, 4100–4106. [Google Scholar] [CrossRef] [PubMed]
- González-Sarrías, A.; García-Villalba, R.; Núñez-Sánchez, M.Á.; Tomé-Carneiro, J.; Zafrilla, P.; Mulero, J.; Tomás-Barberán, F.A.; Espín, J.C. Identifying the limits for ellagic acid bioavailability: A crossover pharmacokinetic study in healthy volunteers after consumption of pomegranate extracts. J. Funct. Foods 2015, 19, 225–235. [Google Scholar] [CrossRef]
- Tennant, D.R.; Davidson, J.; Day, A.J. Phytonutrient intakes in relation to European fruit and vegetable consumption patterns observed in different food surveys. Br. J. Nutr. 2014, 112, 1214–1225. [Google Scholar] [CrossRef]
- Whitley, A.C.; Stoner, G.D.; Darby, M.V.; Walle, T. Intestinal epithelial cell accumulation of the cancer preventive polyphenol ellagic acid—Extensive binding to protein and DNA. Biochem. Pharmacol. 2003, 66, 907–915. [Google Scholar] [CrossRef]
- Srivastava, A.K.; Bhatnagar, P.; Singh, M.; Mishra, S.; Kumar, P.; Shukla, Y.; Gupta, K.C. Synthesis of PLGA nanoparticles of tea polyphenols and their strong in vivo protective effect against chemically induced DNA damage. Int. J. Nanomed. 2019, 14, 7001–7002. [Google Scholar] [CrossRef]
- Xie, X.; Tao, Q.; Zou, Y.; Zhang, F.; Guo, M.; Wang, Y.; Wang, H.; Zhou, Q.; Yu, S. PLGA Nanoparticles Improve the Oral Bioavailability of Curcumin in Rats: Characterizations and Mechanisms. J. Agric. Food Chem. 2011, 59, 9280–9289. [Google Scholar] [CrossRef]
- Sanna, V.; Siddiqui, I.A.; Sechi, M.; Mukhtar, H. Resveratrol-Loaded Nanoparticles Based on Poly (epsilon-caprolactone) and Poly (d l-lactic-co-glycolic acid)–Poly (ethylene glycol) Blend for Prostate Cancer Treatment. Mol. Pharmaceut. 2013, 10, 3871–3881. [Google Scholar] [CrossRef]
- Noyes, A.A.; Whitney, W.R. The rate of solution of solid substances in their own solutions. J. Am. Chem. Soc. 1987, 19, 930–934. [Google Scholar] [CrossRef]
- Li, Y.; Zhao, X.; Zu, Y.; Zhang, Y.; Ge, Y.; Zhong, C.; Wu, W. Preparation and characterization of micronized ellagic acid using antisolvent precipitation for oral delivery. Int. J. Pharmaceut. 2015, 486, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Committee for Human Medicinal Products ICH guideline Q3C (R6) on impurities: Guideline for residual solvents. EMA/CHMP/ICH/82260/ 2006, 2006, 1–39.
- Beshbishy, A.M.; Batiha, G.E.-S.; Yokoyama, N.; Igarashi, I. Ellagic acid microspheres restrict the growth of Babesia and Theileria in vitro and Babesia microti in vivo. Parasites Vectors 2019, 12, 269. [Google Scholar] [CrossRef] [PubMed]
- Montes, A.; Wehner, L.; Pereyra, C.; Martínez de la Ossa, E.J. Generation of microparticles of ellagic acid by supercritical antisolvent process. J. Supercrit. Fluids 2016, 116, 101–110. [Google Scholar] [CrossRef]
- Laine, P.; Kylli, P.; Heinonen, M.; Jouppila, K. Storage Stability of Microencapsulated Cloudberry (Rubus chamaemorus) Phenolics. J. Agric. Food Chem. 2008, 56, 11251–11261. [Google Scholar] [CrossRef]
- Li, B.; Harich, K.; Wegiel, L.; Taylor, L.S.; Edgar, K.J. Stability and solubility enhancement of ellagic acid in cellulose ester solid dispersions. Carbohyd. Polym. 2013, 92, 1443–1450. [Google Scholar] [CrossRef]
- Alfei, S.; Turrini, F.; Catena, S.; Zunin, P.; Parodi, B.; Zuccari, G.; Pittaluga, A.M.; Boggia, R. Preparation of ellagic acid micro and nano formulations with amazingly increased water solubility by its entrapment in pectin or non-PAMAM dendrimers suitable for clinical applications. New J. Chem. 2019, 43, 2438–2448. [Google Scholar] [CrossRef]
- Food Additive Database. Available online: https://webgate.ec.europa.eu/foods_system/main/?sector=FAD&auth=SANCAS (accessed on 2 February 2017).
- El-Missiry, M.A.; Othman, A.I.; Amer, M.A.; Sedki, M.; Ali, S.M.; El-Sherbiny, I.M. Nanoformulated ellagic acid ameliorates pentylenetetrazol-induced experimental epileptic seizures by modulating oxidative stress, inflammatory cytokines and apoptosis in the brains of male mice. Metab. Brain Dis. 2020, 35, 385–399. [Google Scholar] [CrossRef]
- Bulani, V.D.; Kothavade, P.S.; Nagmoti, D.M.; Kundaikar, H.S.; Degani, M.S.; Juvekar, A.R. Characterisation and anti-inflammatory evaluation of the inclusion complex of ellagic acid with hydroxypropyl-β-cyclodextrin. J. Incl. Phenom. Macrocycl. Chem. 2015, 82, 361–372. [Google Scholar] [CrossRef]
- Bulani, V.D.; Kothavade, P.S.; Kundaikar, H.S.; Gawali, N.B.; Chowdhury, A.A.; Degani, M.S.; Juvekar, A.R. Inclusion complex of ellagic acid with β-cyclodextrin: Characterization and in vitro anti-inflammatory evaluation. J. Mol. Struct. 2016, 1105, 308–315. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, Y.; Tian, Z.; Ma, J.; Kang, M.; Ding, C.; Ming, D. Preparation of β-CD-Ellagic Acid Microspheres and Their Effects on HepG2 Cell Proliferation. Molecules 2017, 22, 2175. [Google Scholar] [CrossRef] [PubMed]
- Mady, F.M.; Ibrahim, S.R.M. Cyclodextrin-based nanosponge for improvement of solubility and oral bioavailability of Ellagic acid. Pak. J. Pharm. Sci. 2018, 31, 2069–2076. [Google Scholar] [PubMed]
- Joseph, E.; Singhvi, G. Nanomaterials for Drug Delivery and Therapy; Elsevier: Amsterdam, The Netherlands, 2019; pp. 91–111. [Google Scholar]
- Lu, G.W.; Gao, P. Handbook of Non-Invasive Drug Delivery Systems; Elsevier: Amsterdam, The Netherlands, 2010; pp. 59–94. [Google Scholar]
- Fan, G.; Cai, Y.; Fu, E.; Yuan, X.; Tang, J.; Sheng, H.; Gong, J. Preparation and process optimization of pomegranate ellagic acid-hydroxypropyl-β-cyclodextrin inclusion complex and its antibacterial activity in vitro. Acta Medica. Mediterr. 2019, 35, 383–389. [Google Scholar] [CrossRef]
- Gontijo, A.V.G.; Sampaio, A.; Da, G.; Koga-Ito, C.Y.; Salvador, M.J. Biopharmaceutical and antifungal properties of ellagic acid-cyclodextrin using an in vitro model of invasive candidiasis. Future Microbiol. 2019, 14, 957–967. [Google Scholar] [CrossRef]
- An, S.S.; Chi, K.-W.; Kang, S.C.; Dubey, A.; Park, D.W.; Kwon, J.E.; Jeong, Y.J.; Kim, T.; Kim, I. Investigation of the biological and anti-cancer properties of ellagic acid-encapsulated nano-sized metalla-cages. Int. J. Nanomed. 2015, 10, 227–240. [Google Scholar] [CrossRef]
- Jeong, Y.-I.; Yv, R.P.; Ohno, T.; Yoshikawa, Y.; Shibata, N.; Kato, S.; Takeuchi, K.; Takada, K. Application of Eudragit P-4135F for the delivery of ellagic acid to the rat lower small intestine. J. Phar. Pharmacol. 2001, 53, 1079–1085. [Google Scholar] [CrossRef]
- Danhier, F.; Ansorena, E.; Silva, J.M.; Coco, R.; Le Breton, A.; Préat, V. PLGA-based nanoparticles: An overview of biomedical applications. J. Control. Release 2012, 161, 505–522. [Google Scholar] [CrossRef]
- Bala, I.; Bhardwaj, V.; Hariharan, S.; Kharade, S.V.; Roy, N.; Ravi Kumar, M.N.V. Sustained release nanoparticulate formulation containing antioxidant-ellagic acid as potential prophylaxis system for oral administration. J. Drug Target. 2006, 14, 27–34. [Google Scholar] [CrossRef]
- Reliene, R.; Shirode, A.; Coon, J.; Nallanthighal, S.; Bharali, D.; Mousa, S. Nanoencapsulation of pomegranate bioactive compounds for breast cancer chemoprevention. Int. J. Nanomed. 2015, 10, 475–484. [Google Scholar] [CrossRef]
- Abd-Rabou, A.A.; Ahmed, H.H. CS-PEG decorated PLGA nano-prototype for delivery of bioactive compounds: A novel approach for induction of apoptosis in HepG2 cell line. Adv. Med. Sci. 2017, 62, 357–367. [Google Scholar] [CrossRef] [PubMed]
- Mady, F.; Shaker, M. Enhanced anticancer activity and oral bioavailability of ellagic acid through encapsulation in biodegradable polymeric nanoparticles. Int. J. Nanomed. 2017, 12, 7405–7417. [Google Scholar] [CrossRef]
- Arulmozhi, V.; Pandian, K.; Mirunalini, S. Ellagic acid encapsulated chitosan nanoparticles for drug delivery system in human oral cancer cell line (KB). Colloids Surf. B 2013, 110, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Gopalakrishnan, L.; Ramana, L.N.; Sethuraman, S.; Krishnan, U.M. Ellagic acid encapsulated chitosan nanoparticles as anti-hemorrhagic agent. Carbohydr. Polym. 2014, 111, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Ding, C.; Bi, H.; Wang, D.; Kang, M.; Tian, Z.; Zhang, Y.; Wang, H.; Zhu, T.; Ma, J. Preparation of Chitosan/Alginate-ellagic acid sustained-release microspheres and their Inhibition of preadipocyte adipogenic differentiation. Curr. Pharm. Biotechnol. 2019, 20, 1213–1222. [Google Scholar] [CrossRef] [PubMed]
- Ruan, J.; Yang, Y.; Yang, F.; Wan, K.; Fan, D.; Wang, D. Novel oral administrated ellagic acid nanoparticles for enhancing oral bioavailability and anti-inflammatory efficacy. J. Drug Deliv. Sci. Technol. 2018, 46, 215–222. [Google Scholar] [CrossRef]
- Padilla De Jesus, O.L.; Ihre, H.R.; Gagne, L.; Frechet, J.M.J.; Szoka, F.C. Polyester dendritic systems for drug delivery applications: In vitro and in vivo evaluation. Bioconjug. Chem. 2002, 13, 453–461. [Google Scholar] [CrossRef]
- Alfei, S.; Marengo, B.; Domenicotti, C. Polyester-Based Dendrimer Nanoparticles Combined with Etoposide Have an Improved Cytotoxic andmPro-Oxidant Effect on Human Neuroblastoma Cells. Antioxidants 2020, 9, 50. [Google Scholar] [CrossRef]
- Tesauro, D.; Accardo, A.; Diaferia, C.; Milano, V.; Guillon, J.; Ronga, L.; Rossi, F. Peptide based drug delivery systems in biotechnological applications. Molecules 2019, 24, 351. [Google Scholar] [CrossRef]
- Barnaby, S.N.; Fath, K.R.; Tsiola, A.; Banerjee, I.A. Fabrication of ellagic acid incorporated self-assembled peptide microtubes and their applications. Colloid Surf. B 2012, 95, 154–161. [Google Scholar] [CrossRef]
- Rahmanian, N.; Hamishehkar, H.; Dolatabadi, J.E.; Arsalani, N. Nano graphene oxide: A novel carrier for oral delivery of flavonoids. Colloids Surf. B 2014, 123, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Kakran, M.G.; Sahoo, N.; Bao, H.; Pan, Y.; Li, L. Functionalized graphene oxide as nanocarrier for loading and delivery of ellagic acid. Curr. Med. Chem. 2011, 18, 4503–4512. [Google Scholar] [CrossRef]
- Ghasemiyeh, P.; Mohammadi-Samani, S. Solid lipid nanoparticles and nanostructured lipid carriers as novel drug delivery systems: Applications, advantages and disadvantages. Res. Pharm. Sci. 2018, 13, 288–303. [Google Scholar] [PubMed]
- Yoon, G.; Park, J.W.; Yoon, I.S. Solid lipid nanoparticles (SLNs) and nanostructured lipid carriers (NLCs): Recent advances in drug delivery. J. Pharm. Investig. 2013, 43, 353–362. [Google Scholar] [CrossRef]
- Makwana, V.; Jain, R.; Patel, K.; Nivsarkar, M.; Joshi, A. Solid lipid nanoparticles (SLN) of Efavirenz as lymph targeting drug delivery system: Elucidation of mechanism of uptake using chylomicron flow blocking approach. Int. J. Pharm. 2015, 495, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Hajipour, H.; Hamishehkar, H.; Rahmati-yamchi, M.; Shanehbandi, D.; Nazari Soltan Ahmad, S.; Hasani, A. Enhanced anti-cancer capability of ellagic acid using solid lipid nanoparticles (SLNs). Int. J. Cancer Manag. 2018, 11, e9402. [Google Scholar] [CrossRef]
- Murugan, V.; Mukherjee, K.; Maiti, K.; Mukherjee, P.K. Enhanced oral bioavailability and antioxidant profile of ellagic acid by phospholipids. J. Agric. Food Chem. 2009, 57, 4559–4565. [Google Scholar] [CrossRef]
- Stojiljković, N.; Ilić, S.; Stojanović, N.; Janković-Veličković, L.; Stojnev, S.; Kocić, G.; Radenković, G.; Arsić, I.; Stojanović, M.; Petković, M. Nanoliposome-encapsulated ellagic acid prevents cyclophosphamide-induced rat liver damage. Mol. Cell. Biochem. 2019, 458, 185–195. [Google Scholar] [CrossRef]
- Wang, X.; Jiang, Y.; Wang, Y.-W.; Huang, M.-T.; Ho, C.-T.; Huang, Q. Enhancing anti-inflammation activity of curcumin through O/W nanoemulsions. Food Chem. 2008, 108, 419–424. [Google Scholar] [CrossRef]
- Avachat, A.M.; Patel, V.G. Self nanoemulsifying drug delivery system of stabilized ellagic acid–phospholipid complex with improved dissolution and permeability. Saudi Pharm. J. 2015, 23, 276–289. [Google Scholar] [CrossRef]
- Wang, S.-T.; Chou, C.-T.; Su, N.-W. A food-grade self-nanoemulsifying delivery system for enhancing oral bioavailability of ellagic acid. J. Funct. Foods 2017, 34, 207–215. [Google Scholar] [CrossRef]
- Dokania, S.; Joshi, A.K. Self-microemulsifying drug delivery system (SMEDDS)—Challenges and road ahead. Drug Deliv. 2015, 22, 675–690. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.; Lv, C.; Sun, X.; Wang, J.; Zhao, Z. Preparation of a supersaturatable self-microemulsion as drug delivery system for ellagic acid and evaluation of its antioxidant activities. J. Drug Deliv. Sci. Technol. 2019, 53, 101209. [Google Scholar] [CrossRef]
- Tavano, L.; Muzzalupo, R.; Picci, N.; de Cindio, B. Co-encapsulation of antioxidants into niosomal carriers: Gastrointestinal release studies for nutraceutical applications. Colloid Surf. B 2014, 114, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Ratnam, D.V.; Chandraiah, G.; Meena, A.K.; Ramarao, P.; Kumar, M.N. The co-encapsulated antioxidant nanoparticles of ellagic acid and coenzyme Q10 ameliorates hyperlipidemia in high fat diet fed rats. Nanosci. Nanotechnol. 2009, 9, 6741–6746. [Google Scholar] [CrossRef] [PubMed]
- Suri, S.; Mirza Mohd, A.; Anwer, M.K.; Alshetaili, A.S.; Alshahrani, S.M.; Ahmed, F.J.; Iqbal, Z. Development of NIPAAm-PEG acrylate polymeric nanoparticles for co-delivery of paclitaxel with ellagic acid for the treatment of breast cancer. J. Polym. Eng. 2019, 39, 271–278. [Google Scholar] [CrossRef]
- Fahmy, U.A. Augmentation of fluvastatin cytotoxicity against prostate carcinoma PC3 cell line utilizing alpha lipoic–ellagic acid nanostructured lipid carrier formula. AAPS PharmSciTech 2018, 19, 3454–3461. [Google Scholar] [CrossRef]
- Tamjidi, F.; Shahedi, M.; Varshosaz, J.; Nasirpour, A. Nanostructured lipid carriers (NLC): A potential delivery system for bioactive food molecules. Innov. Food Sci. Emerg. Technol. 2013, 19, 23–43. [Google Scholar] [CrossRef]
- Abd Elwakil, M.M.; Mabrouk, M.T.; Helmy, M.W.; Abdelfattah, E.-Z.A.; Khiste, S.K.; Elkhodairy, K.A.; Elzoghby, A.O. Inhalable lactoferrin–chondroitin nanocomposites for combined delivery of doxorubicin and ellagic acid to lung carcinoma. Nanomedicine 2018, 13, 2015–2035. [Google Scholar] [CrossRef]
| Vehicles | Solubility (mg/mL) | Temperature | Ref. | |
|---|---|---|---|---|
| Solvents | N-methyl-2-pyrrolidone | 25 | 37 °C | [36] |
| DMSO | 2.5 | 37 °C | [36] | |
| Pyridine | 2.0 | 37 °C | [36] | |
| Methanol | (671 ± 17) × 10−3 | 37 °C | [36] | |
| Ethanol | 1.02 ± 0.04 | 25 °C | [96] | |
| Cosolvents | PEG 200 | 4.178 | 25 °C | [94] |
| PEG 400 | 11.0 ± 0.5 | 25 °C | [96] | |
| Propylene glycol | 2.1 ± 0.1 | 25 °C | [96] | |
| Oils | Palmester 3575 1 | 0.030 | 25 °C | [94] |
| Cottonseed oil | 0.005 | 25 °C | [94] | |
| Soybean oil | 0.29 ± 0.01 | 25 °C | [95] | |
| Castor oil | 1.63 ± 0.07 | 25 °C | [96] | |
| Oleic acid | 0.29 ± 0.01 | 25 °C | [96] | |
| Ethyl oleate | 2.34 ± 0.06 | 25 °C | [96] | |
| Surfactants | Tween 20 | 1.605 | 25 °C | [94] |
| Sucrose esters | 0.115 | 25 °C | [94] | |
| Isopropyl myristate | 1.94 ± 0.07 | 25 °C | [96] | |
| Cremophor RH40 2 | 2.5 ± 0.1 | 25 °C | [96] | |
| Tween 80 | 3.5 ± 0.1 | 25 °C | [96] | |
| Lecithin | 0.085 ± 0.004 | 25 °C | [96] | |
| Poloxamer F68 | 0.036 ± 0.002 | 25 °C | [96] | |
| Aqueous Solutions | Phosphate buffer pH 7.4 | (33 ± 16) × 10−3 | 37 °C | [36] |
| Phosphate buffer pH 6.8 | (11.1 ± 0.4) × 10−3 | 25 °C | [96] | |
| Acetate buffer pH 4.5 | (6.9 ± 0.3) × 10−3 | 25 °C | [96] | |
| Distilled water | (8.2 ± 0.4) × 10−3 | 25 °C | [96] | |
| HCl 0.1 M in water | (1.03 ± 0.06) × 10−3 | 25 °C | [96] |
| Formulation Type | Fabrication Method | Excipients | Mean Size | Remarkable Features | Highlights | Ref. |
|---|---|---|---|---|---|---|
| Micro-sized EA | Anti-solvent precipitation | - | n.a. | Use of a syringe pump Good dispersion | In vitro and in vivo inhibition of blood parasites | [54] |
| Micro-sized EA | Supercritical anti-solvent process | Eudragit® L 100 | 3.73 µm | Co-precipitate product EA content 49% Residual NMP 148 ppm | Increased EA dissolution rate | [55] |
| Amorphous solid dispersion | Freeze drying | Maltodextrin | n.a. | Use of cloudberry extract | Higher storage stability up to 32 days Food supplement formulation | [56] |
| Amorphous solid dispersion | Spray drying | Hydroxypropyl-methyl cellulose acetate succinate | n.a. | EA solubility 280 µg/mL EA content 25%Stable supersaturated EA solution at pH 6.8 | pH-sensitive polymer Minimal release in the stomach, quite fast at pH 6.8 (35% after 0.5 h) | [57] |
| Amorphous solid dispersion | Spray drying | Pectin | 10 µm | EA solubility 63 µg/mL EA content 21% No organic solvent used | High biocompatibility Suitable to formulate antioxidant-rich functional food | [58] |
| Amorphous solid dispersion | Spray drying | Alginic acid | 670 nm | EA solubilized in basic solution Crosslinking with CaCl2 Complete release after 8 h | Highly biocompatible formulation Improved in vivo neuroprotective and anticonvulsant effect in orally treated mice | [60] |
| Polymeric microspheres | Emulsion-evaporation technique in oil phase (acetone/light liquid paraffin) | Eudragit® P-4135F | 113 µm | EA content 35% EE 81% Total release after 6 h | pH responsive release Ileocaecal targeting | [70] |
| Polymeric microspheres | Ionotropic gelation by sodium alginate | Chitosan | 4.36 µm | EE 29% Sustained release | Obesity prevention and treatment | [78] |
| Peptide microtubes | Self-assembling | bis(N-α-amido threonine)-1, 5-pentane dicarboxylate | 0.5–1 µm | EE 80% Rate release dependent on the EA deprotonation process at different pH | High biocompatibility Enhanced antibacterial activity | [83] |
| Phospholipidvesicles | Anti-solvent precipitation | Hydrogenated soy phosphatidyl-choline | 1–3 µm | EE 29% | High biocompatibility Liver protection Relative AUC increase by2.8-fold | [83] |
| Formulation Type | Fabrication Method | Excipients | Mean Size | Remarkable Features | Highlights | Ref. |
|---|---|---|---|---|---|---|
| Nano-sized EA | Anti-solvent precipitation | - | 428 nm | EA water solubility 11.67 µg/mL Lyophilized product with maltodextrin as diluent Residual NMP 405 ppm | Higher radical scavenging activity Enhanced relative AUC by 2 times | [52] |
| Polymer nanospheres | Emulsion-diffusion-evaporation | PLGA or PCL | 125 nm | EA content 62% Slow EA release (about 24% after 6 days) | Sustained release for 20 days Good stability Potential prophylaxis system Higher in situ uptake and greater in vivo nephron-protection in CyA-treated rats | [22,72] |
| Dendrimer | Self-assembling | Aminoacid-modifed hetero dendrimer | 70 nm | EA solubility 9 mg/mL EA content 53% | EA solubility increase | [58] |
| Inclusion complex | Freeze drying | HP-β-CD | n.a. | Formation of 1:2 EA:HP-β-CD complex Increased total EA solubility up to 54.40 µg/mL 60% EA released after 0.5 h | Enhanced in vivo anti-inflammatory effect | [61] |
| Inclusion complex | Freeze drying | β-CD | n.a. | Formation of 1:2 EA:β-CD complex Increased EA solubility up to 39.14 µg/mL Less than 30% EA released after 0.5 h | Enhanced in vivo anti-inflammatory effect | [62,63] |
| Inclusion complex | Overnight shaking | β-CD nanosponge | 423 nm | EA solubility up to 49.79 µg/mL EA content 69% Prolonged release | High biocompatibility Increased relative AUC by 2.2-fold | [64] |
| Inclusion complex | Stirring-ultrasonic and final freeze drying | HP-β-CD | n.a. | Optimized production process EE 84% | Antibacterial activity | [67] |
| Inclusion complex | Precipitation | Arene-Ru metalla-prisms | n.a. | Yield 92% High complex stability | Enhanced antitumor activity against A549 cells | [69] |
| Polymeric nanospheres | Double emulsion-evaporation (w/o/w) | PLGA-PEG | 175 nm | Opsonization avoided Prolonged circulation time in blood Suitable for i.v. administration | Inhibition of breast cancer cell growth More than 2-fold IC50 reduction in MCF-7 cells | [73] |
| Polymeric nanospheres | Emulsion-evaporation in aqueous phase | PLGA coated with chitosan and PEG | 255 nm | Opsonization avoided Sustained release Suitable for i.v. administration | 3-fold IC50 reduction in HepG2 and HCT 116 cells | [74] |
| Polymeric nanospheres | Emulsion-diffusion-evaporation | PCL | 193 nm | EA content 58% EE 66% Slow EA release: approx. 48% after 8 days | Long-term release 6.9-fold cytotoxicity increase against HCT 116 cells Improved relative AUC by 3.6 times | [75] |
| Polymeric nanospheres | Ionic gelation by sodium tripolyphosphate | Chitosan | 176 nm | EA content 33% Rapid release up to 8 h, then more controlled up to 48 h | More than 3-fold IC50 reduction in KB cells Faster clotting time | [76,77] |
| Polymeric nanocapsules | Anti-solvent coprecipitation | Zein | 72 nm | Shell thickness of 20 nm 326 mg EA loaded per 1 g of excipient Sustained release up to 6 days | High biocompatibility Enhanced in vivo anti-inflammatory effect Relative AUC increased by 8.7-fold | [79] |
| Adsorption complex | Overnight shaking | Functionalized graphene oxide | Sheets ranged from 20 to 120 nm | Easy EA loading by physisorption EA solubility up to 610 µg/mL 1.22 g EA loaded per 1 g of excipient 23% EA released at pH 4 38% at pH 10 after 72 h | Enhanced in vitro cytotoxicity against MCF7 and HT29 cells | [85] |
| Solid lipid nanoparticle | Hot homogenization method | Precirol® Poloxamer 407 Tween 80 | 100 nm | EE 89% EA content 36% Initial burst effect (>40%) followed by a sustained release | Enhanced antitumor activity against PC3 cells | [89] |
| Liposome | Overnight mixing | Phospholipid nanoparticles solution (10%) in form of nanospheres | n.a. | EE 60% Good EA protection in different pH buffers and metal ion containing solutions | For nutritional supplements as adjuvant therapy in cancer | [91] |
| Self-nanoemulsifying delivery systems | Preliminary EA and soy lecithin complex obtained by anti-solvent precipitation | Soy lecithin Captex® 500 Cremophor® RH40 PEG 400 Tocopherol | 106 nm | EE 95% Fast release of nearly 95% after 1 h | Improved ex vivo intestinal permeability | [93] |
| Self-nanoemulsifying delivery systems | Vortex mixing | Palmester® 3575 Tween 80 PEG 400 | 120 nm | 2.5 mg/mL EA loading into SNEDDS Fast release | Food-grade components Increased relative AUC by 6.6-fold | [94] |
| Self-nanoemulsifying delivery systems | Vortex mixing | Ethyl oleate Tween 80 PEG 400 | 45 nm | 4 mg/g EA loading into SNEDDS Adjunct of PVP 0.5% as stabilizer Less fast release | Enhanced in vitro and in vivo radical scavenging activity | [96] |
| Polymeric nanospheres | Emulsion-diffusion-evaporation | PLGA | 259 nm | Fixed formulation with CoQ10 EE of 70% for EA and of 72% for CoQ10 | Prolonged control on cholesterol levels in rats | [98] |
| Polymeric nanospheres | Self-assembling and dialysis method for drug loading | Poly (N-iso-propylacrylamide-PEG acrylate) | 200 nm | Combined formulation with paclitaxel Controlled release (only 8% after 2 h) EE 98% for EA and 92% for paclitaxel | Enhanced in vitro cytotoxicity against MCF-7 cells | [99] |
| Nanostructured lipid carrier | Hot emulsification-ultrasonication | Almond oil Compritol® ATO Phosphatidylcholine Gelucire® | 85 nm | Co-delivery of EA, fluvastatin, alpha lipoic acid Total drug released within 3 h | Enhanced in vitro cytotoxicity against PC3 cells | [100] |
| Polymeric nanoparticles | EA nanocrystallization by anti-solvent precipitation + polyelectrolyte electrostatic complexation | Lactoferrin Chondroitin sulfate | 192 nm | Inhalable combined formulation with doxorubicin | Low cost of production Easy scalability Excipients usable in food products | [102] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zuccari, G.; Baldassari, S.; Ailuno, G.; Turrini, F.; Alfei, S.; Caviglioli, G. Formulation Strategies to Improve Oral Bioavailability of Ellagic Acid. Appl. Sci. 2020, 10, 3353. https://doi.org/10.3390/app10103353
Zuccari G, Baldassari S, Ailuno G, Turrini F, Alfei S, Caviglioli G. Formulation Strategies to Improve Oral Bioavailability of Ellagic Acid. Applied Sciences. 2020; 10(10):3353. https://doi.org/10.3390/app10103353
Chicago/Turabian StyleZuccari, Guendalina, Sara Baldassari, Giorgia Ailuno, Federica Turrini, Silvana Alfei, and Gabriele Caviglioli. 2020. "Formulation Strategies to Improve Oral Bioavailability of Ellagic Acid" Applied Sciences 10, no. 10: 3353. https://doi.org/10.3390/app10103353
APA StyleZuccari, G., Baldassari, S., Ailuno, G., Turrini, F., Alfei, S., & Caviglioli, G. (2020). Formulation Strategies to Improve Oral Bioavailability of Ellagic Acid. Applied Sciences, 10(10), 3353. https://doi.org/10.3390/app10103353