Adsorption Characteristics of Spent Coffee Grounds as an Alternative Adsorbent for Cadmium in Solution
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Adsorbents and Reagents
2.2. Characterization of the Adsorbents
2.3. Adsorption Study
2.4. Optimization of the Operating Parameters for the Cd Adsorption Study
2.5. Sorption Models
2.6. Data Analysis
3. Results and Discussion
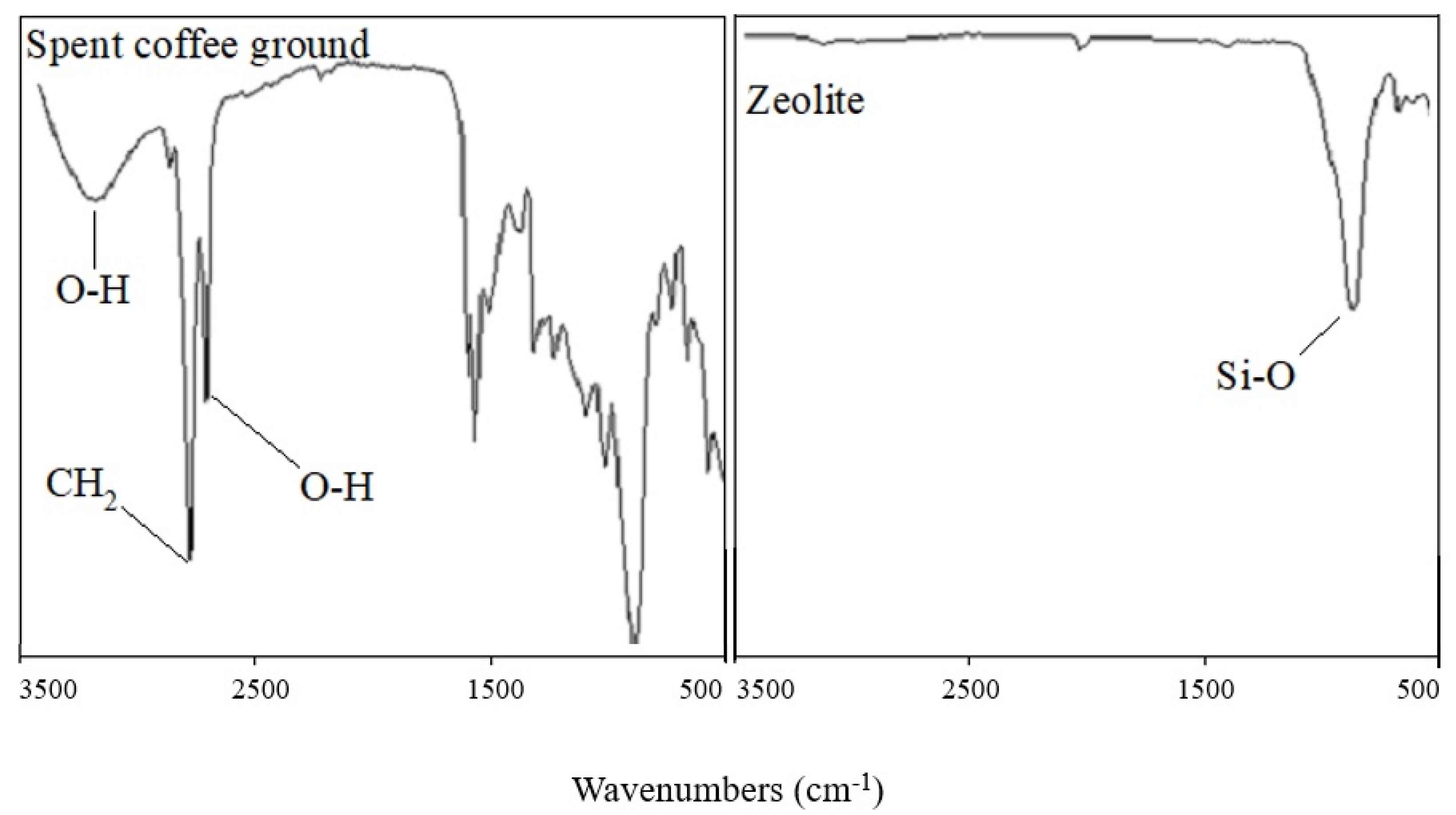
3.1. Characterization of Adsorbents
3.2. Sorption Dynamics
3.2.1. Effects of Ionic Strength on Cd Adsorption
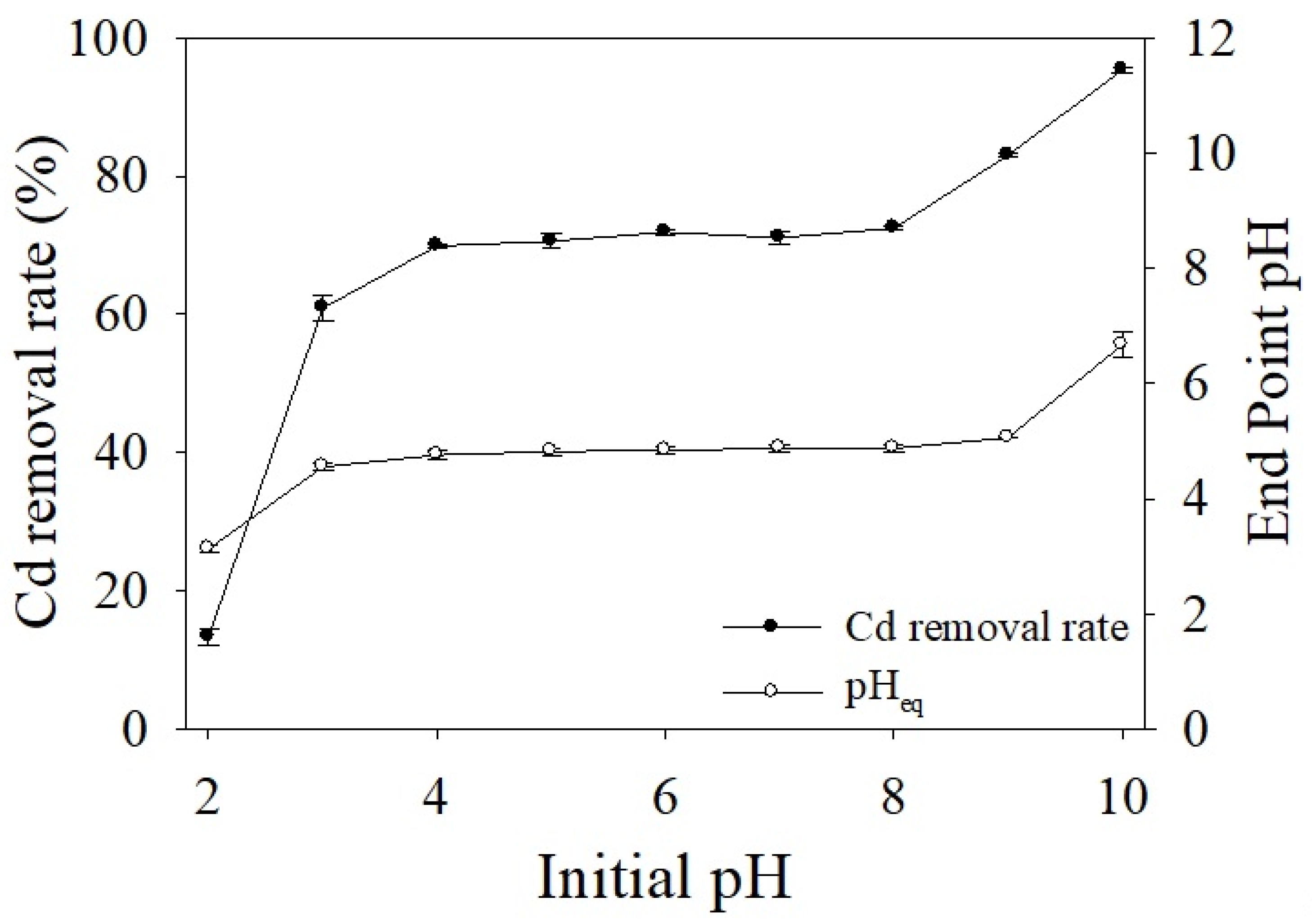
3.2.2. Effects of Solution pH on Cd Adsorption
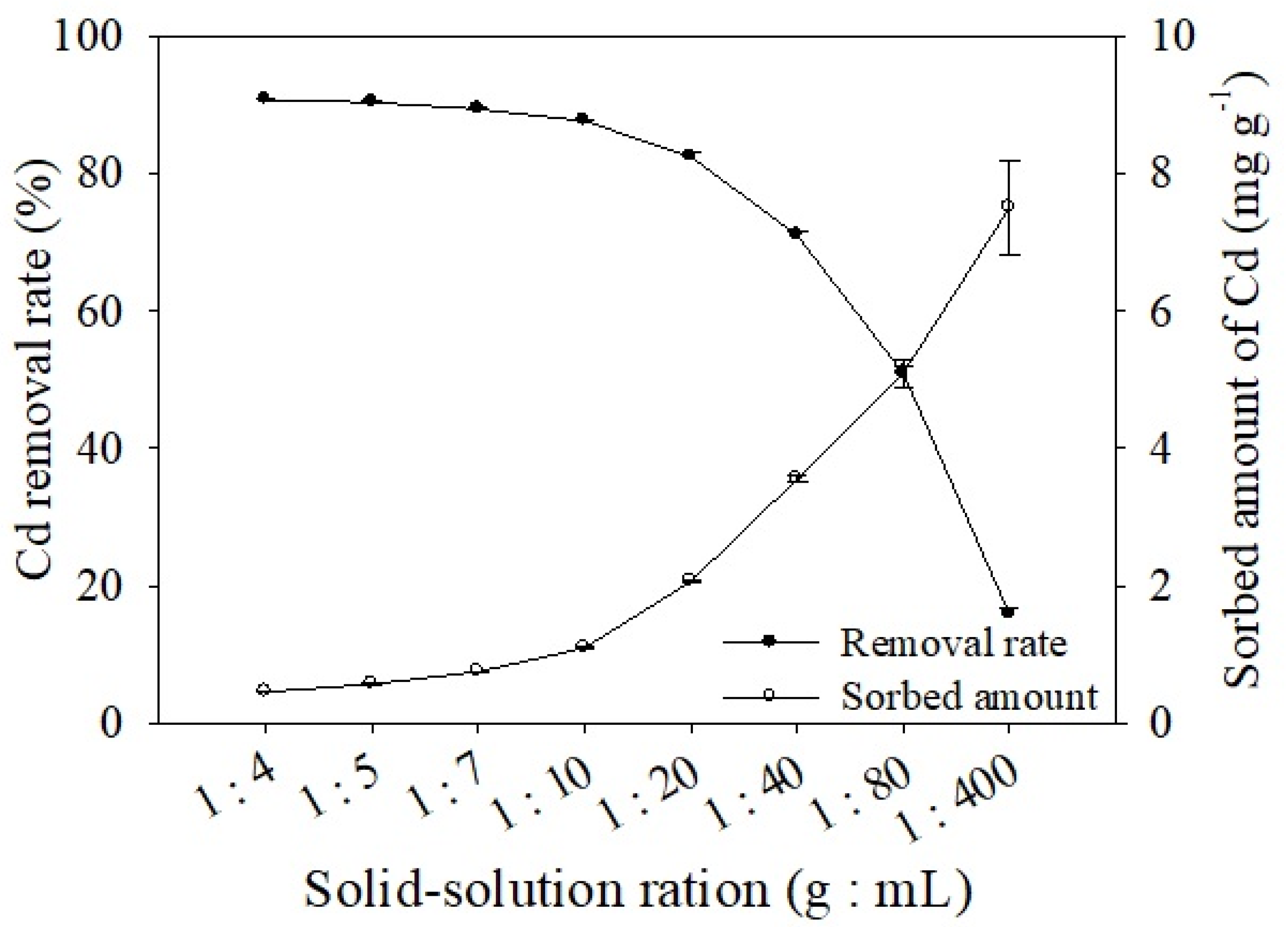
3.2.3. Effects of Solid/Solution Ratio on Cd Adsorption
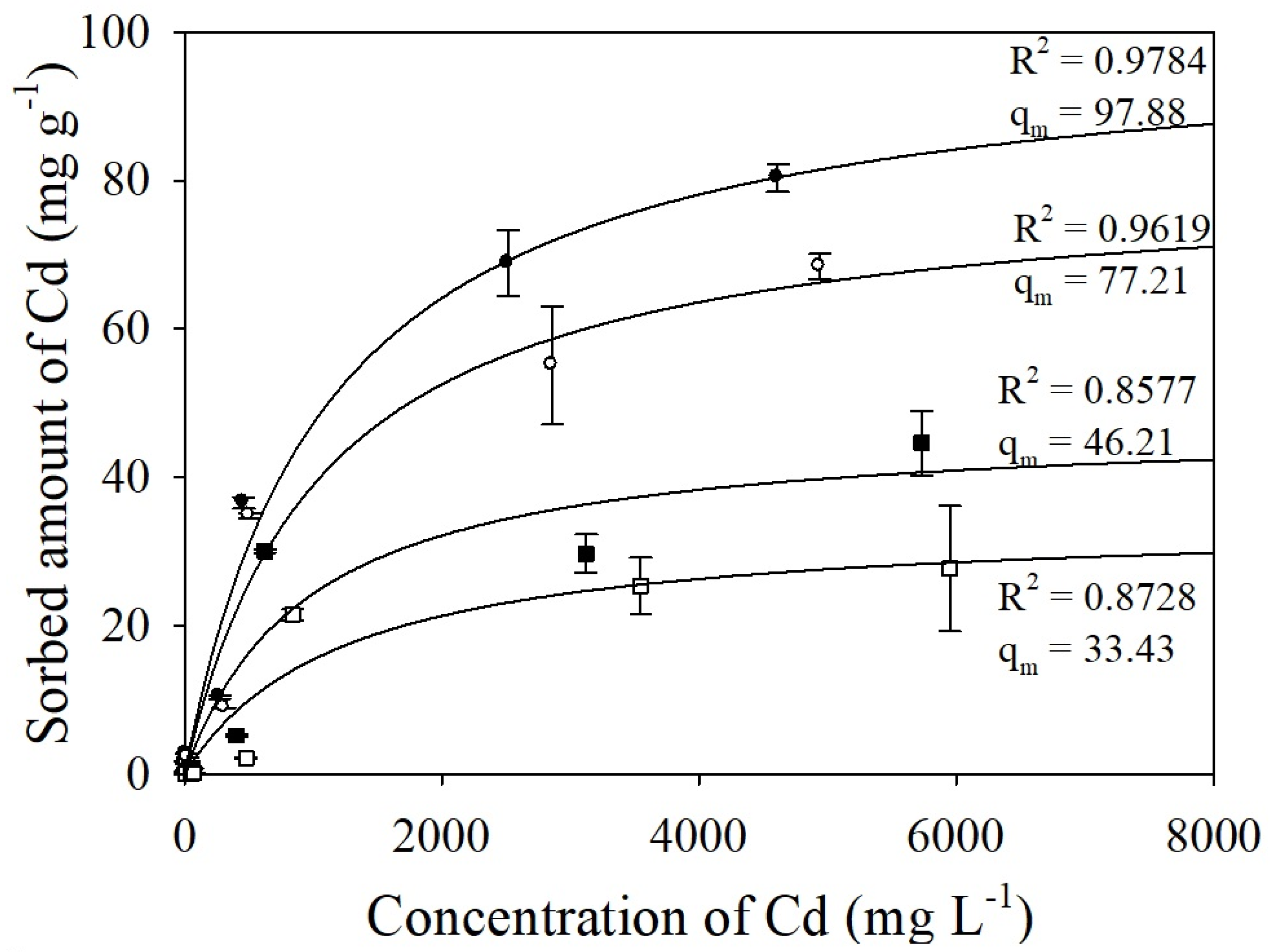
3.3. Adsorption Capacity of the Adsorbents
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Abollino, O.; Giacomino, A.; Malandrino, M.; Mentasti, E. The efficiency of vermiculite as natural sorbent for heavy metals. Application to a contaminated soil. Water Air Soil Pollut. 2007, 181, 149–160. [Google Scholar] [CrossRef]
- Mosa, A.A.; El-Ghamry, A.; Truby, P. Chemically modified crop residues as a low-cost technique for the removal of heavy metal ions from wastewater. Water Air Soil Pollut. 2011, 217, 637–647. [Google Scholar] [CrossRef]
- Martínez, M.; Miralles, N.; Hidalgo, S.; Fiol, N.; Villaescusa, I.; Poch, J. Removal of lead(II) and cadmium(II) from aqueous solutions using grape stalk waste. J. Hazard. Mater. 2006, 133, 203–211. [Google Scholar] [CrossRef]
- Kim, M.S.; Min, H.G.; Lee, S.H.; Kim, J.G. The effects of various amendments on trace element stabilization in acidic, neutral and alkali soil with similar pollution index. PLoS ONE 2016, 11, e0166335. [Google Scholar] [CrossRef] [PubMed]
- Minamisawa, M.; Minamisawa, H.; Yoshida, S.; Takai, N. Adsorption behavior of heavy metals on biomaterials. J. Agric. Food Chem. 2004, 52, 5606–5611. [Google Scholar] [CrossRef] [PubMed]
- Zuorro, A.; Lavecchia, R. Adsorption of Pb(II) on spent leaves of green and black tea. Am. J. Appl. Sci. 2010, 7, 153–159. [Google Scholar] [CrossRef]
- Babel, S.; Kurniawan, T.A. Low-cost adsorbents for heavy metals uptake from contaminated water: A review. J. Hazard. Mater. 2003, 97, 219–243. [Google Scholar] [CrossRef]
- Xu, X.; Liu, X.; Oh, M.; Park, J.B. Oyster shell as a low-cost adsorbent for removing heavy metal ions from wastewater. Pol. J. Environ. Stud. 2019, 28, 2949–2959. [Google Scholar] [CrossRef]
- Demirbas, A. Heavy metal adsorption onto agro-based waste materials: A review. J. Hazard. Mater. 2008, 157, 220–229. [Google Scholar] [CrossRef]
- Borreli, R.C.; Visconti, A.; Mennella, C.; Anese, M.; Fogliano, V. Chemical characterization and antioxidant properties of coffee melanoidins. J. Agric. Food Chem. 2002, 50, 6527–6533. [Google Scholar] [CrossRef] [PubMed]
- Vegro, C.L.R.; de Almeida, L.F. Global coffee market: Socio-economic and cultural dynamics. In Coffee Consumption and Industry Strategies in Brazil; Woodhead Publishing: Cambridge, UK, 2020; pp. 3–19. [Google Scholar] [CrossRef]
- McNutt, J.; He, Q.S. Spent coffee grounds: A review on current utilization. J. Ind. Eng. Chem. 2019, 71, 78–88. [Google Scholar] [CrossRef]
- Fiol, N.; Escudero, C.; Villaescusa, I. Re-use of exhausted ground coffee waste for Cr(VI) sorption. Sep. Sci. Technol. 2008, 43, 582–596. [Google Scholar] [CrossRef]
- Kondamudi, N.; Mohapatra, S.K.; Misra, M. Spent coffee grounds as a versatile source of green energy. J. Agric. Food Chem. 2008, 56, 11757–11760. [Google Scholar] [CrossRef] [PubMed]
- Krishnani, K.K.; Meng, X.; Christodoulatos, C.; Boddu, V.M. Biosorption mechanism of nine different heavy metals onto biomatrix from rice husk. J. Hazard. Mater. 2009, 163, 441–447. [Google Scholar] [CrossRef]
- Boonamnuayvitaya, V.; Chaiya, C.; Tanthapanichakoon, W.; Jarudilokkul, S. Removal of heavy metals by adsorbent prepared from pyrolyzed coffee residues and clay. Sep. Purif. Technol. 2004, 35, 11–22. [Google Scholar] [CrossRef]
- Kaikake, K.; Hoaki, K.; Sunada, H.; Dhakal, R.P.; Baba, Y. Removal characteristics of metal ions using degreased coffee beans: Adsorption equilibrium of cadmium. Biores. Technol. 2007, 98, 2787–2791. [Google Scholar] [CrossRef]
- Ministry of Environment. Standard Methods for Examination of Soil Pollution; Ministry of Environment: Gwacheon city, Gyeongi province, Korea, 2009.
- Mohammad, M.; Maitra, S.; Ahmad, N.; Bustam, A.; Sen, T.K.; Dutta, B.K. Metal ion removal from aqueous solution using physic seed hull. J. Hazard. Mater. 2010, 179, 363–372. [Google Scholar] [CrossRef]
- Brown, P.; Jefcoat, I.A.; Parrish, D.; Gill, S.; Graham, E. Evaluation of the adsorptive capacity of peanut hull pellets for heavy metals in solution. Adv. Environ. Res. 2000, 4, 19–29. [Google Scholar] [CrossRef]
- Azouaou, N.; Sadaoui, Z.; Djaafri, A.; Mokaddem, H. Adsorption of cadmium from aqueous solution onto untreated coffee grounds: Equilibrium, kinetics and thermodynamics. J. Hazard. Mater. 2010, 184, 126–134. [Google Scholar] [CrossRef]
- Chen, B.; Lee, Y.; Lin, W.; Lin, F.; Lin, K. Understanding the characteristics of L-ascorbic acid-montmorillonite nanocomposite: Chemical structure and biotoxicity. Biomed. Eng. Appl. Basis Commun. 2006, 18, 30–36. [Google Scholar] [CrossRef][Green Version]
- Nouri, L.; Ghodbane, I.; Hamdaoui, O.; Chiha, M. Batch sorption dynamics and equilibrium for the removal of cadmium ions from aqueous phase using wheat bran. J. Hazard. Mater. 2007, 149, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, G.; Singh, B.; Kookana, R.S. Cadmium adsorption and desorption behaviour on goethite at low equilibrium concentrations: Effects of pH and index cations. Chemosphere 2004, 57, 1325–1333. [Google Scholar] [CrossRef]
- Oliveira, W.E.; Franca, A.S.; Oliveira, L.S.; Rocha, S.D. Untreated coffee husks as biosorbents for the removal of heavy metals from aqueous solutions. J. Hazard. Mater. 2008, 152, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Mall, I.D.; Srivastava, V.C.; Kumar, G.V.A.; Mishra, I.M. Characterization and utilization of mesoporus fertilizer plant waste carbon for adsorptive removal of dyes from aqueous solution. Colloids Surf. A Physicochem. Eng. Asp. 2006, 278, 175–187. [Google Scholar] [CrossRef]
- Yang, C.; Huang, W.; Xiao, B.; Yu, Z.; Peng, P.; Fu, J.; Sheng, G. Intercorrelations among degree of geochemical alterations, physicochemical properties, and organic sorption equilibria of kerogen. Environ. Sci. Technol. 2004, 38, 4396–4408. [Google Scholar] [CrossRef]
- Bailey, S.E.; Olin, T.J.; Bricka, R.M.; Adrian, D.D. A review of potentially low-cost sorbents for heavy metals. Water Res. 1999, 33, 2469–2479. [Google Scholar] [CrossRef]
- Reddad, Z.; Gerente, C.; Andres, Y.; Cloirec, P.L. Adsorption of several metal ions onto a low-cost biosorbent: Kinetic and equilibrium studies. Environ. Sci. Technol. 2002, 36, 2067–2073. [Google Scholar] [CrossRef]
- Shin, E.W.; Rowell, R.M. Cadmium ion sorption onto lignocellulosic biosorbent modified by sulfonation: Origin of sorption capacity improvement. Chemosphere 2005, 60, 1054–1061. [Google Scholar] [CrossRef]
- Tiemann, K.J.; Gardea-Torresdey, J.L.; Gamez, G.; Dokken, K.; Sias, S.; Renner, M.W.; Furenlid, L.R. Use of X-ray absorption spectroscopy and esterification to investigate Cr(III) and Ni(II) ligands in alfalfa biomass. Environ. Sci. Technol. 1999, 33, 150–154. [Google Scholar] [CrossRef]
- Utomo, H.D.; Hunter, K.A. Adsorption of heavy metals by exhausted coffee grounds as a potential treatment method for waste waters. E-J. Surf. Sci. Nanotechnol. 2006, 4, 504–506. [Google Scholar] [CrossRef]
- Zhou, Y.F.; Haynes, R.J. Sorption of heavy metals by inorganic and organic components of solid wastes: Significance to use of wastes as low-cost adsorbents and immobilizing agents. Critic. Rev. Environ. Sci. Technol. 2010, 40, 909–977. [Google Scholar] [CrossRef]
- Vijayaraghavan, K.; Yun, Y.S. Bacterial biosorbents and biosorption. Biotechnol. Adv. 2008, 26, 266–291. [Google Scholar] [CrossRef] [PubMed]
- Prabhakaran, S.K.; Vijayaraghavan, K.; Balasubramanian, R. Removal of Cr(VI) ions by spent tea and coffee dust: Reduction to Cr(III) and biosorption. Indus. Eng. Chem. Res. 2009, 48, 2113–2117. [Google Scholar] [CrossRef]
- Saeed, A.; Iqbal, M. Bioremoval of cadmium from aqueous solution by black gram husk (Cicer arientinum). Water Res. 2003, 37, 3472–3480. [Google Scholar] [CrossRef]
- Ammari, T.G.; Al-Atiyat, M.; Abu-Nameh, E.S.; Ghrair, A.; Jaradat, D.; Abu-Romman, S. Bioremediation of cadmium-contaminated water systems using intact and alkaline-treated alga (Hydrodictypm reticulatum) naturally grown in an ecosystem. Int. J. Biorem. 2017, 19, 453–462. [Google Scholar] [CrossRef]
- Schiewer, S.; Patil, S.B. Pectin-rich fruit wastes as biosorbents for heavy metal removal: Equilibrium and kinetics. Biores. Technol. 2008, 99, 1896–1903. [Google Scholar] [CrossRef]
- Massimi, L.; Giuliano, A.; Astolfi, M.L.; Congedo, R.; Masotti, A.; Canepari, S. Efficiency evaluation of food waste materials for the removal of metals and metalloids from complex multi-element solutions. Materials 2018, 11, 334. [Google Scholar] [CrossRef]
- Patterer, M.S.; Bavasso, I.; Sambeth, J.E.; Medici, F. Cadmium removal from acqueous solution by adsorption on spent coffee grounds. Chem. Eng. Trans. 2017, 60, 157–162. [Google Scholar] [CrossRef]
| Adsorbent | pH | EC a | LOI b | Trace Elements (mg kg−1) | |||||
|---|---|---|---|---|---|---|---|---|---|
| (mS m−1) | (%) | As | Cd | Cu | Ni | Pb | Zn | ||
| SCGs | 5.2 ± 0.1 | 185 ± 11 | 94.7 ± 0.5 | ND c | 0.01 ± 0.00 | 12.6 ± 0.3 | 0.4 ± 0.05 | 1.1 ± 0.1 | 6.2 ± 0.2 |
| zeolite | 7.3 ± 0.1 | 74 ± 8 | 1.3 ± 0.2 | 4.3 ± 0.1 | 0.39 ± 0.02 | 19.9 ± 0.4 | 17.3 ± 0.7 | 15.6 ± 1.8 | 43.0 ± 2.3 |
| Freundlich | Langmuir | Dubinin-Radushkevich | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 1/n | Kf | R2 | qm (mg g−1) | k (L mg−1) | R2 | qm (mg g−1) | E (kJ mol−1) | R2 | |
| SCGs | 0.32 | 0.85 | 0.95 | 19.32 | <0.01 | 0.96 | 19.79 | 14.08 | 0.96 |
| Zeolite | 0.39 | 0.10 | 0.87 | 13.91 | <0.01 | 0.92 | 14.48 | 16.44 | 0.81 |
| Type | Biosorbents | Maximum Adsorption Capacity | References |
|---|---|---|---|
| Coffee residues | Spent coffee grounds | 19.32 | - a |
| Untreated coffee grounds | 15.65 | [21] | |
| Exhausted coffee ground | 11.60 | [32] | |
| Coffee husks | 6.85 | [25] | |
| Degreased coffee beans | 6.72 | [17] | |
| Coffee | 6.47 | [5] | |
| Spent coffee grounds | 4.34 | [40] | |
| Agro-waste | Rice husk | 14.40 | [15] |
| Grape stalk waste | 13.93 | [3] | |
| Physic seed hull | 11.89 | [19] | |
| Corn starch | 8.88 | [29] | |
| Peanut hull | 5.96 | [20] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, M.-S.; Kim, J.-G. Adsorption Characteristics of Spent Coffee Grounds as an Alternative Adsorbent for Cadmium in Solution. Environments 2020, 7, 24. https://doi.org/10.3390/environments7040024
Kim M-S, Kim J-G. Adsorption Characteristics of Spent Coffee Grounds as an Alternative Adsorbent for Cadmium in Solution. Environments. 2020; 7(4):24. https://doi.org/10.3390/environments7040024
Chicago/Turabian StyleKim, Min-Suk, and Jeong-Gyu Kim. 2020. "Adsorption Characteristics of Spent Coffee Grounds as an Alternative Adsorbent for Cadmium in Solution" Environments 7, no. 4: 24. https://doi.org/10.3390/environments7040024
APA StyleKim, M.-S., & Kim, J.-G. (2020). Adsorption Characteristics of Spent Coffee Grounds as an Alternative Adsorbent for Cadmium in Solution. Environments, 7(4), 24. https://doi.org/10.3390/environments7040024





