Influence of Soil Characteristics and Land Use Type on Existing Fractions of Radioactive 137Cs in Fukushima Soils
Abstract
1. Introduction
2. Materials and Methods
2.1. Soil Sampling and Preparation
2.2. Soil Analysis
2.2.1. Soil Physical and Chemical Analysis
2.2.2. Measurement of 137Cs Activity
2.2.3. Sequential Extraction of 137Cs
2.3. Statistical Analysis
3. Results and Discussion
3.1. Soil Physiochemical Properties
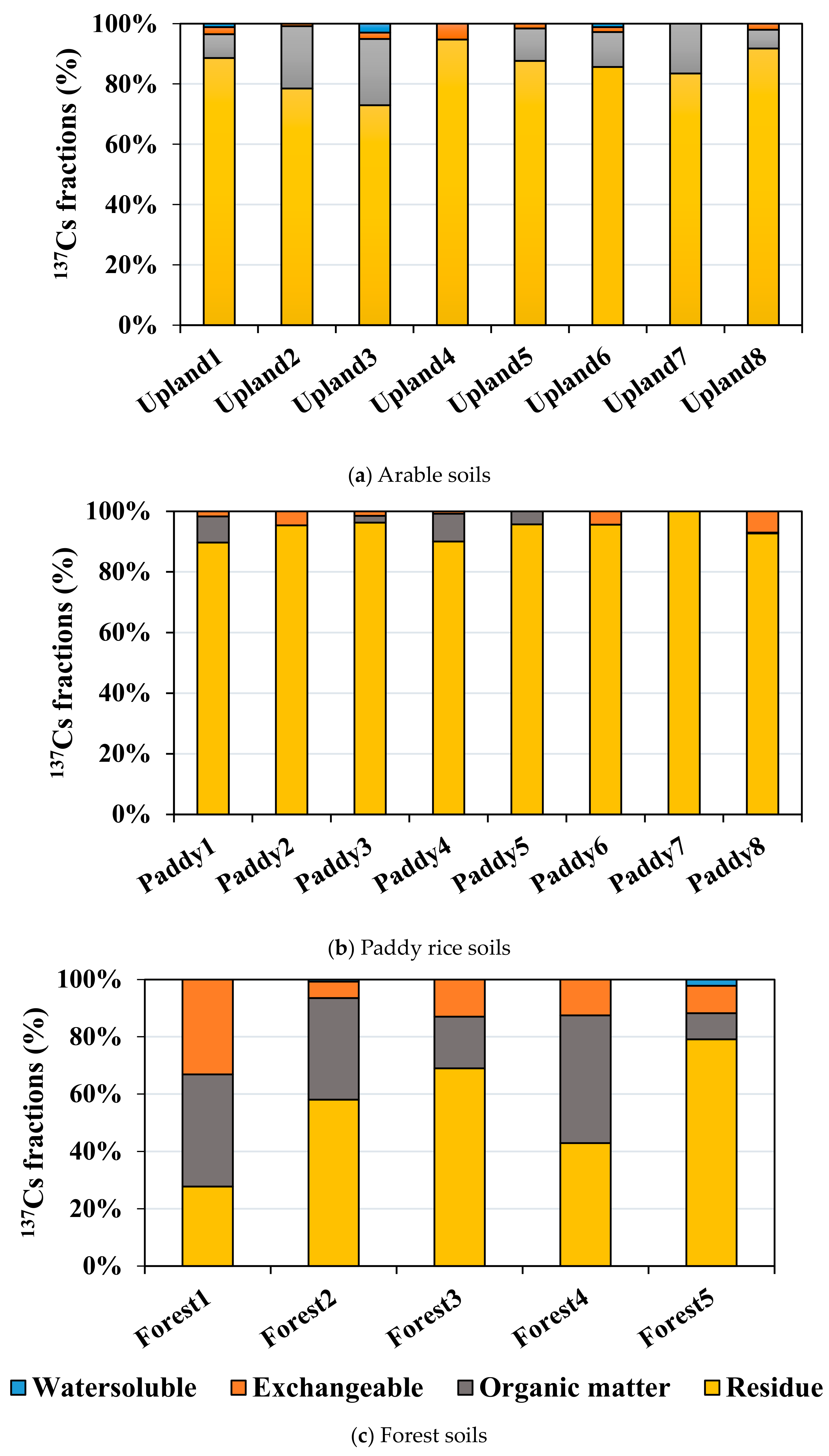
3.2. 137Cs Concentrations and Sequential Extraction
3.3. Correlation of 137Cs Fractions and Soil Characteristics
3.4. Influence of Soil Characteristics on 137Cs Availability
3.5. Influence of Land Use Types on 137Cs Availability in Soils
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Fujii, K.; Ikeda, S.; Akama, A.; Komatsu, M.; Takahashi, M.; Kaneko, S. Vertical Migration of Radiocesium and Clay Mineral Composition in Five Forest Soils Contaminated by the Fukushima Nuclear Accident. Soil Sci. Plant Nutr. 2014, 60, 751–764. [Google Scholar] [CrossRef]
- Teramage, M.T.; Onda, Y.; Patin, J.; Kato, H.; Gomi, T.; Nam, S. Vertical Distribution of Radiocesium in Coniferous Forest Soil after the Fukushima Nuclear Power Plant Accident. J. Environ. Radioact. 2014, 137, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.G.; Smolders, E. Plant Uptake of Radiocaesium: A Review of Mechanisms, Regulation and Application. J. Exp. Bot. 2000, 51, 1635–1645. [Google Scholar] [CrossRef] [PubMed]
- Krouglov, S.V.; Kurinov, A.D.; Alexakhin, R.M. Chemical Fractionation of 90Sr, 106Ru, 137Cs, and 144Ce in Chernobyl-Contaminated Soils: An Evolution in the Course of Time. J. Environ. Radioact. 1998, 38, 59–76. [Google Scholar] [CrossRef]
- Nakano, M.; Yong, R.N. Overview of Rehabilitation Schemes for Farmlands Contaminated with Radioactive Cesium Released from Fukushima Power Plant. Eng. Geol. 2013, 155, 87–93. [Google Scholar] [CrossRef]
- Cermers, A.; Elsaen, A.; De Preter, P.; Maes, A. Quantitatieve Analysis of Radio Caesium Adsorption in Soils- Japanese. Nature 1988, 335, 247–249. [Google Scholar] [CrossRef]
- Gerzabek, M.H.; Mohamad, S.A.; Muck, K. Cesium-137 in Soil Texture Fractions and Its Impact on Cesium-137 Soil-To-Plant Transfer. Commun. Soil Sci. Plant Anal. 1992, 23, 321–330. [Google Scholar] [CrossRef]
- Koarashi, J.; Atarashi-Andoh, M.; Matsunaga, T.; Sato, T.; Nagao, S.; Nagai, H. Factors Affecting Vertical Distribution of Fukushima Accident-Derived Radiocesium in Soil under Different Land-Use Conditions. Sci. Total Environ. 2012, 431, 392–401. [Google Scholar] [CrossRef]
- Konopleva, I.; Klemt, E.; Konoplev, A.; Zibold, G. Migration and Bioavailability of 137Cs in Forest Soil of Southern Germany. J. Environ. Radioact. 2009, 100, 315–321. [Google Scholar] [CrossRef]
- Cornell, R.M. Adsorption of Cesium on Minerals: A Review. J. Radioanal. Nucl. Chem. Artic. 1993, 171, 483–500. [Google Scholar] [CrossRef]
- Shand, C.A.; Rosén, K.; Thored, K.; Wendler, R.; Hillier, S. Downward Migration of Radiocaesium in Organic Soils across a Transect in Scotland. J. Environ. Radioact. 2013, 115, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Dumat, C.; Cheshire, M.V.; Fraser, A.R.; Shand, C.A.; Staunton, S. The Effect of Removal of Soil Organic Matter and Iron on the Adsorption of Radiocaesium. Eur. J. Soil Sci. 2008, 48, 675–683. [Google Scholar] [CrossRef]
- Choi, Y.J.; Tomás-Barberán, F.A.; Saltveit, M.E. Wound-Induced Phenolic Accumulation and Browning in Lettuce (Lactuca Sativa L.) Leaf Tissue Is Reduced by Exposure to n-Alcohols. Postharvest Biol. Technol. 2005, 37, 47–55. [Google Scholar] [CrossRef]
- Van Bergeijk, K.E.; Noordijk, H.; Lembrechts, J.; Frissel, M.J. Influence of pH, Soil Type and Soil Organic Matter Content on Soil-to-Plant Transfer of Radiocesium and -Strontium as Analyzed by a Nonparametric Method. J. Environ. Radioact. 1992, 15, 265–276. [Google Scholar] [CrossRef]
- Yamaguchi, N.; Takata, Y.; Hayashi, K. Behavior of Radiocaesium in Soil-Plant Systems and Its Controlling Factor. Bull Natl Inst Agro-Environ 2012, 31, 75–129. [Google Scholar] [CrossRef]
- Staunton, S.; Dumat, C.; Zsolnay, A. Possible Role of Organic Matter in Radiocaesium Adsorption in Soils. J. Environ. Radioact. 2002, 58, 163–173. [Google Scholar] [CrossRef]
- Dumat, C.; Staunton, S. Reduced Adsorption of Caesium on Clay Minerals Caused by Various Humic Substances. J. Environ. Radioact. 1999, 46, 187–200. [Google Scholar] [CrossRef]
- Staunton, S.; Roubaud, M. Adsorption of 137Cs on Montmorillonite and Illite: Effect of Charge Compensating Cation, Ionic Strength, Concentration of Cs, K and Fulvic Acid. Clays Clay Miner. 1997, 45, 251–260. [Google Scholar] [CrossRef]
- Brad, S.W.; Miles, P.D.; Perry, C.H.; Pugh, S.A. Forest Resources of the United States, 2007; Gen. Tech. Rept. WO-78; U.S. Department of Agriculture, Forest Service: Washington, DC, USA, 2009.
- Forsberg, S.; Strandmark, M. Migration and Chemical Availability of 137Cs and 90 Sr in Swedish Long-Term Experimental Pastures. Water. Air. Soil Pollut. 2001, 127, 157–171. [Google Scholar] [CrossRef]
- World Reference Base for Soil Resources. World Soil Resources Reports 103; Food and Agriculture Organization: Rome, Italy; Cambridge University Press: Cambridge, UK, 2006; p. 132. [CrossRef]
- Gee, G.W.; Bauder, J.W. Particle-Size Analysis·. In Methods of Soil Analysis, Part 1. Physical and Mineralogical Methods, Agronomy Monograph; Klute, A., Ed.; American Society of Agronomy/Soil Science Society of America: Madison, WI, USA, 1986; Volume 9, p. 53711. [Google Scholar]
- Committee of Soil Standard Methods for Analyses and Measurements: Soil Standard Methods for Analyses and Measurements; Hakuyusha: Tokyo, Japan, 1986; p. 354. (in Japanese)
- Schumacher, B.A. Methods for the Determination of Total Organic Carbon (TOC) in Soils and Sediments, NCEA-C-1282; U.S. Environmental Protection Agency, National Exposure Research Laboratory: Washington, DC, USA, 2002.
- Schollenberger, C.J.; Simon, R.H. Determination of Exchange Capacity and Exchangeable Bases in Soil—Ammonium Acetate Method. Soil Sci. 1945, 59, 13–24. [Google Scholar] [CrossRef]
- Forsberg, S. Behavior of 137Cs and 90Sr in agricultural soils influence of ageing and soil type on availability, migration and plant uptake. Acta Universitatis Agriculturae Sueciae - Agraria 2000, 212, 46. [Google Scholar]
- Puhakainen, M.; Riekkinen, I.; Heikkinen, T.; Jaakkola, T.; Steinnes, E.; Rissanen, K.; Suomela, M.; Thørring, H. Effect of Chemical Pollution on Forms of 137Cs, 90Sr and 239,240Pu in Arctic Soil Studied by Sequential Extraction. J. Environ. Radioact. 2001, 52, 17–29. [Google Scholar] [CrossRef]
- Saito, T.; Makino, H.; Tanaka, S. Geochemical and Grain-Size Distribution of Radioactive and Stable Cesium in Fukushima Soils: Implications for Their Long-Term Behavior. J. Environ. Radioact. 2014, 138, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Roig, M.; Vidal, M.; Rauret, G.; Rigol, A. Prediction of Radionuclide Aging in Soils from the Chernobyl and Mediterranean Areas. J. Environ. Qual. 2007, 36, 943–952. [Google Scholar] [CrossRef] [PubMed]
- Takeda, A.; Tsukada, H.; Takaku, Y.; Akata, N.; Hisamatsu, S. Plant Induced Changes in Concentrations of Caesium, Strontium and Uranium in Soil Solution with Reference to Major Ions and Dissolved Organic Matter. J. Environ. Radioact. 2008, 99, 900–911. [Google Scholar] [CrossRef] [PubMed]
- Riise, G.; Bjørnstad, H.E.; Lien, H.N.; Oughton, D.H.; Salbu, B. A Study on Radionuclide Association with Soil Components Using a Sequential Extraction Procedure. J. Radioanal. Nucl. Chem. Artic. 1990, 142, 531–538. [Google Scholar] [CrossRef]
- Fawaris, B.H.; Johanson, K.J. Fractionation of Caesium (137Cs) in Coniferous Forest Soil in Central Sweden. Sci. Total Environ. 1995, 170, 221–228. [Google Scholar] [CrossRef]
- Amano, H.; Matsunaga, T.; Nagao, S.; Hanzawa, Y.; Watanabe, M.; Ueno, T.; Onuma, Y. The Transfer Capability of Long-Lived Chernobyl Radionuclides from Surface Soil to River Water in Dissolved Forms. Org. Geochem. 1999, 30, 437–442. [Google Scholar] [CrossRef]
- Rigol, A.; Roig, M.; Vidal, M.; Rauret, G. Sequential Extractions for the Study of Radiocesium and Radiostrontium Dynamics in Mineral and Organic Soils from Western Europe and Chernobyl Areas. Environ. Sci. Technol. 1999, 33, 887–895. [Google Scholar] [CrossRef]
- Dumat, C.; Quiquampoix, H.; Staunton, S. Adsorption of Cesium by Synthetic Clay—Organic Matter Complexes: Effect of the Nature of Organic Polymers. Environ. Sci. Technol. 2000, 34, 2985–2989. [Google Scholar] [CrossRef]
- Matsunaga, T.; Koarashi, J.; Atarashi-Andoh, M.; Nagao, S.; Sato, T.; Nagai, H. Comparison of the Vertical Distributions of Fukushima Nuclear Accident Radiocesium in Soil before and after the First Rainy Season, with Physicochemical and Mineralogical Interpretations. Sci. Total Environ. 2013, 447, 301–314. [Google Scholar] [CrossRef]
- Wakahara, T.; Onda, Y.; Kato, H.; Sakaguchi, A.; Yoshimura, K. Radiocesium Discharge from Paddy Fields with Different Initial Scrapings for Decontamination after the Fukushima Dai-Ichi Nuclear Power Plant Accident. Environ. Sci. Process. Impacts 2014, 16, 2580–2591. [Google Scholar] [CrossRef] [PubMed]
- Sakai, M.; Gomi, T.; Nunokawa, M.; Wakahara, T.; Onda, Y. Soil Removal as a Decontamination Practice and Radiocesium Accumulation in Tadpoles in Rice Paddies at Fukushima. Environ. Pollut. 2014, 187, 112–115. [Google Scholar] [CrossRef] [PubMed]
- Tsumura, A.; Komamura, M.; Kobayashi, H. Behavior of Radioactive Sr and Cs in Soils and Soil-Plant Systems. Bull. Natl. Inst. Agric.Sci. 1984, 36, 57–113. [Google Scholar]
- Vandenhove, H.; Cremers, A.; Smolders, E.; Van Hees, M. Effect of K and Bentonite Additions on Cs-Transfer to Ryegrass. J. Environ. Radioact. 2005, 81, 233–253. [Google Scholar] [CrossRef]
- Wang, G.; Staunton, S. Dynamics of Caesium in Aerated and Flooded Soils: Experimental Assessment of Ongoing Adsorption and Fixation. Eur. J. Soil Sci. 2010, 61, 1005–1013. [Google Scholar] [CrossRef]
- Koarashi, J.; Atarashi-Andoh, M.; Ishizuka, S.; Miura, S.; Saito, T.; Hirai, K. Quantitative Aspects of Heterogeneity in Soil Organic Matter Dynamics in a Cool-Temperate Japanese Beech Forest: A Radiocarbon-Based Approach. Glob. Chang. Biol. 2009, 15, 631–642. [Google Scholar] [CrossRef]
- Ono, K.; Hiradate, S.; Morita, S.; Ohse, K.; Hirai, K. Humification Processes of Needle Litters on Forest Floors in Japanese Cedar (Cryptomeria Japonica) and Hinoki Cypress (Chamaecyparis Obtusa) Plantations in Japan. Plant Soil 2011, 338, 171–181. [Google Scholar] [CrossRef]
- Fesenko, S.V.; Soukhova, N.V.; Sanzharova, N.I.; Avila, R.; Spiridonov, S.I.; Klein, D.; Badot, P.M. 137Cs Availability for Soil to Understory Transfer in Different Types of Forest Ecosystems. Sci. Total Environ. 2001, 269, 87–103. [Google Scholar] [CrossRef]
- Fukuyama, T.; Takenaka, C. Upward Mobilization Of137Cs in Surface Soils of Chamaecyparis Obtusa Sieb. et Zucc. (Hinoki) Plantation in Japan. Sci. Total Environ. 2004, 318, 187–195. [Google Scholar] [CrossRef]
- Rigol, A.; Vidal, M.; Rauret, G. An Overview of the Effect of Organic Matter on Soil-Radiocaesium Interaction: Implications in Root Uptake. J. Environ. Radioact. 2002, 58, 191–216. [Google Scholar] [CrossRef]
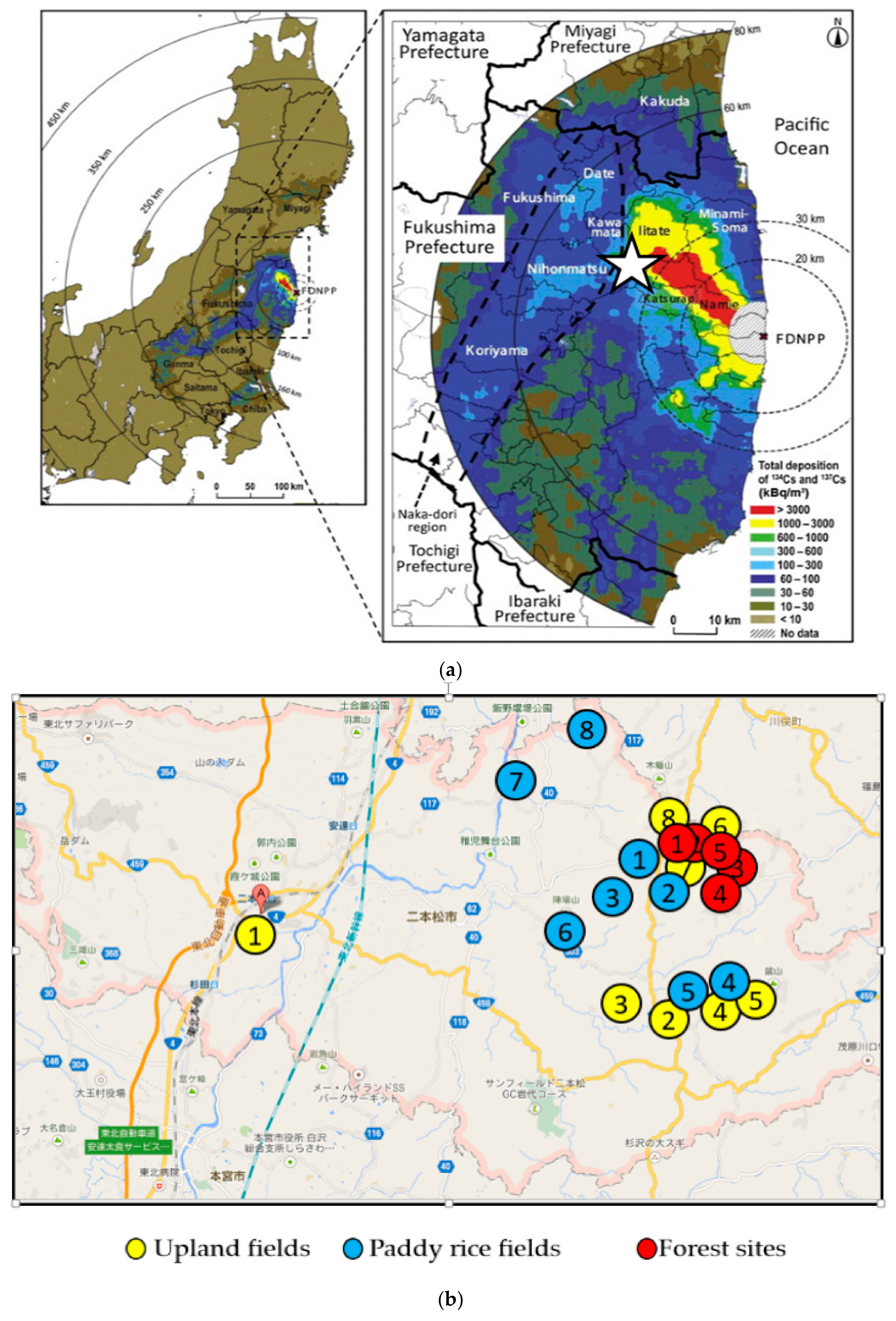
| Field | Location | Soil Group (World Reference Base, 2006) | Surface Geology | Sampling Date | |
|---|---|---|---|---|---|
| N | E | ||||
| Arable 1 | 37.34.41 | 140.25.43 | Haplic Fluvisol | Granite | 6 August 2013 |
| 2 | 37.33.26 | 140.35.22 | Gleyic Fluvisol | Granite | 5 August 2013 |
| 3 | 37.33.26 | 140.34.05 | Cambisol | Granite | 5 August 2013 |
| 4 | 37.33.32 | 140.36.21 | Andosol | Granite | 5 August 2013 |
| 5 | 37.33.36 | 140.36.40 | Andosol | Granite | 5 August 2013 |
| 6 | 37.36.28 | 140.35.03 | Andosol | Granite | 6 August 2013 |
| 7 | 37.36.22 | 140.35.02 | Cambisol | Granite | 6 August 2013 |
| 8 | 37.36.09 | 140.34.38 | Cambisol | Granite | 8 December 2013 |
| Paddy 1 | 37.36.37 | 140.35.03 | Haplic Fluvisol | Mud, sand, gravel | 8 December 2013 |
| 2 | 37.36.10 | 140.34.55 | Gleyic Fluvisol | Mud, sand, gravel | 8 December 2013 |
| 3 | 37.35.24 | 140.33.46 | Gleyic Fluvisol | Granite | 8 December 2013 |
| 4 | 37.36.14 | 140.34.43 | Andosol | Granite | 9 December 2013 |
| 5 | 37.33.41 | 140.36.30 | Gleyic Fluvisol | Granite | 9 December 2013 |
| 6 | 37.34.52 | 140.32.45 | Gleyic Fluvisol | Granite | 9 December 2013 |
| 7 | 37.37.31 | 140.31.41 | Gleyic Fluvisol | Granite | 9 December 2013 |
| 8 | 37.38.32 | 140.33.11 | Gleyic Fluvisol | Granite | 9 December 2013 |
| Forest 1 | 37.35.58 | 140.35.20 | Cambisol | Granite | 27 November 2012 |
| 2 | 37.35.59 | 140.35.24 | Cambisol | Granite | 27 November 2012 |
| 3 | 37.35.54 | 140.36.52 | Cambisol | Granite | 31 October 2012 |
| 4 | 37.35.38 | 140.36.52 | Cambisol | Granite | 28 November 2012 |
| 5 | 37.36.05 | 140.36.29 | Cambisol | Granite | 31 October 2012 |
| Fraction | Extractant | Treatment | Reference |
|---|---|---|---|
| W, Water-soluble | 200 mL distilled H2O | Shaken for 1 min at room temperature | Forsberg and Strandmark (2001) |
| E, Exchangeable | 200 mL 1 M NH4OAc (pH 7) | Shaken for 2 h at 20 °C | Forsberg and Strandmark (2001) |
| O, Organic matter | 10 mL 30% H2O2 (pH 2) | Shaken for 5 h at 80 °C | Puhakainen et al. (2001) |
| R, residual fixation | Residue | Remaining 137Cs activity content after organic matter fraction extraction | Forsberg and Strandmark (2001) |
| Field | 137Cs a | pH (H2O) | CEC b | TC c | TN d | Soil Texture e | ||
|---|---|---|---|---|---|---|---|---|
| (Bq kg−1) | (cmol 100 g−1) | (g kg−1) | (g kg−1) | Clay (%) | Silt (%) | Sand (%) | ||
| Arable 1 | 1718 | 5.7 | 6.9 | 15.6 | 1.4 | 34.0 | 19.8 | 46.3 |
| 2 | 1068 | 5.6 | 8.8 | 16.3 | 1.5 | 12.2 | 19.9 | 67.9 |
| 3 | 895 | 5.7 | 17.8 | 12.9 | 1.3 | 27.6 | 28.1 | 44.3 |
| 4 | 3019 | 7.1 | 11.4 | 64.3 | 6.0 | 22.5 | 25.4 | 52.1 |
| 5 | 2341 | 6.3 | 10.5 | 20.6 | 1.9 | 31.6 | 29.9 | 38.5 |
| 6 | 569 | 5.0 | 9.5 | 18.8 | 2.0 | 19.9 | 17.6 | 62.5 |
| 7 | 605 | 7.1 | 9.3 | 18.0 | 1.9 | 9.8 | 12.0 | 78.2 |
| 8 | 1167 | 7.5 | 15.8 | 44.5 | 4.3 | 20.5 | 27.5 | 51.9 |
| Paddy 1 | 792 | 5.9 | 9.3 | 9.6 | 0.9 | 21.5 | 26.3 | 52.2 |
| 2 | 1083 | 6.2 | 9.4 | 25.7 | 2.1 | 17.3 | 18.4 | 64.3 |
| 3 | 1963 | 6.8 | 6.3 | 13.2 | 1.1 | 5.8 | 7.1 | 87.1 |
| 4 | 1270 | 6.4 | 5.7 | 8.7 | 1.0 | 1.2 | 16.2 | 82.6 |
| 5 | 1200 | 6.2 | 7.0 | 14.4 | 1.3 | 2.9 | 24.1 | 72.9 |
| 6 | 1708 | 5.8 | 9.3 | 17.7 | 1.6 | 9.9 | 21.0 | 69.1 |
| 7 | 1842 | 6.0 | 7.7 | 16.4 | 1.6 | 9.0 | 18.6 | 72.5 |
| 8 | 1664 | 6.1 | 7.7 | 15.8 | 1.4 | 14.9 | 26.1 | 60.0 |
| Forest 1 | 114 | 5.5 | 15.7 | 77.7 | 4.1 | 11.0 | 41.7 | 47.2 |
| 2 | 229 | 5.0 | 15.8 | 77.6 | 3.8 | 43.1 | 24.2 | 32.7 |
| 3 | 690 | 5.1 | 12.7 | 80.5 | 4.9 | 6.5 | 27.8 | 65.7 |
| 4 | 5268 | 5.4 | 20.7 | 116.0 | 7.1 | 26.9 | 22.6 | 50.5 |
| 5 | 461 | 4.6 | 18.9 | 124.0 | 6.7 | 28.9 | 18.9 | 52.2 |
| E | O | R | ||
|---|---|---|---|---|
| Arable | W | 0.02 | 0.38 | −0.50 |
| E | −0.46 | 0.54 | ||
| O | −0.97 * | |||
| Paddy | W | NA | NA | NA |
| E | −0.49 | −0.18 | ||
| O | −0.96 * | |||
| Forest | W | −0.45 | −0.69 | 0.69 |
| E | 0.34 | −0.75 | ||
| O | −0.88 ** |
| pH | CEC | TC | TN | Clay | Silt | Sand | TC/Clay | TN/Clay | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Arable | W | −0.55 | −0.38 | −0.45 | −0.44 | 0.42 | 0.17 | −0.35 | −0.62 | −0.61 |
| E | 0.23 | 0.76 * | 0.80 * | 0.78 * | 0.42 | 0.49 | −0.50 | 0.44 | 0.39 | |
| O | −0.30 | −0.74 | −0.61 | −0.59 | −0.41 | −0.13 | 0.33 | −0.15 | −0.13 | |
| R | 0.58 | 0.84 * | 0.73 * | 0.72 * | 0.28 | 0.27 | −0.31 | 0.50 | 0.48 | |
| Paddy | W | NA | NA | NA | NA | NA | NA | NA | NA | NA |
| E | −0.25 | 0.48 | 0.43 | 0.49 | 0.53 | 0.30 | −0.49 | −0.49 | −0.51 | |
| O | 0.14 | −0.35 | −0.83 * | −0.80 * | −0.14 | 0.10 | 0.03 | 0.55 | 0.51 | |
| R | 0.03 | 0.04 | 0.62 | 0.54 | −0.24 | −0.33 | 0.33 | −0.26 | −0.19 | |
| Forest | W | −0.91 * | 0.33 | 0.57 | 0.34 | 0.48 | −0.60 | −0.15 | −0.47 | −0.46 |
| E | 0.67 | −0.17 | −0.34 | −0.30 | −0.66 | 0.92 * | 0.13 | 0.33 | 0.42 | |
| O | 0.84 ** | 0.25 | −0.27 | −0.21 | 0.15 | 0.41 | −0.49 | −0.29 | −0.28 | |
| R | −0.92 * | −0.11 | 0.35 | 0.30 | 0.21 | −0.75 | 0.30 | 0.07 | 0.01 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mensah, A.D.; Terasaki, A.; Aung, H.P.; Toda, H.; Suzuki, S.; Tanaka, H.; Onwona-Agyeman, S.; Omari, R.A.; Bellingrath-Kimura, S.D. Influence of Soil Characteristics and Land Use Type on Existing Fractions of Radioactive 137Cs in Fukushima Soils. Environments 2020, 7, 16. https://doi.org/10.3390/environments7020016
Mensah AD, Terasaki A, Aung HP, Toda H, Suzuki S, Tanaka H, Onwona-Agyeman S, Omari RA, Bellingrath-Kimura SD. Influence of Soil Characteristics and Land Use Type on Existing Fractions of Radioactive 137Cs in Fukushima Soils. Environments. 2020; 7(2):16. https://doi.org/10.3390/environments7020016
Chicago/Turabian StyleMensah, Akwasi Dwira, Akimi Terasaki, Han Phyo Aung, Hiroto Toda, Sohzoh Suzuki, Haruo Tanaka, Siaw Onwona-Agyeman, Richard Ansong Omari, and Sonoko Dorothea Bellingrath-Kimura. 2020. "Influence of Soil Characteristics and Land Use Type on Existing Fractions of Radioactive 137Cs in Fukushima Soils" Environments 7, no. 2: 16. https://doi.org/10.3390/environments7020016
APA StyleMensah, A. D., Terasaki, A., Aung, H. P., Toda, H., Suzuki, S., Tanaka, H., Onwona-Agyeman, S., Omari, R. A., & Bellingrath-Kimura, S. D. (2020). Influence of Soil Characteristics and Land Use Type on Existing Fractions of Radioactive 137Cs in Fukushima Soils. Environments, 7(2), 16. https://doi.org/10.3390/environments7020016





