Seasonal Emergence and Historical Contaminant Exposure of Cave Myotis (Myotis velifer) in Central Texas and Current Status of the Population
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cave Descriptions
2.2. Colony Size and Emergence Patterns
2.3. Guano Samples
2.4. Bat Samples
2.5. Chemical Analyses
3. Results
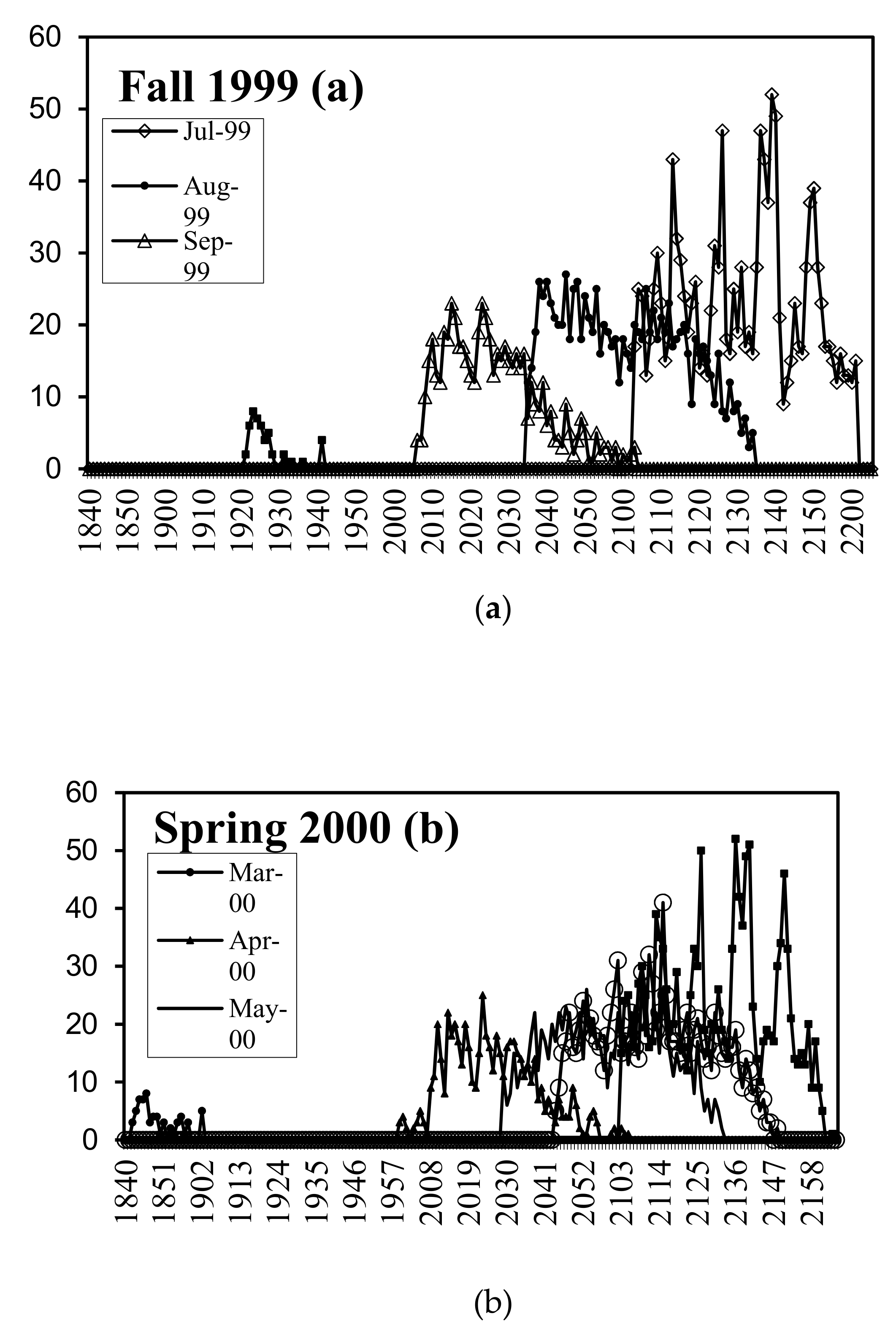
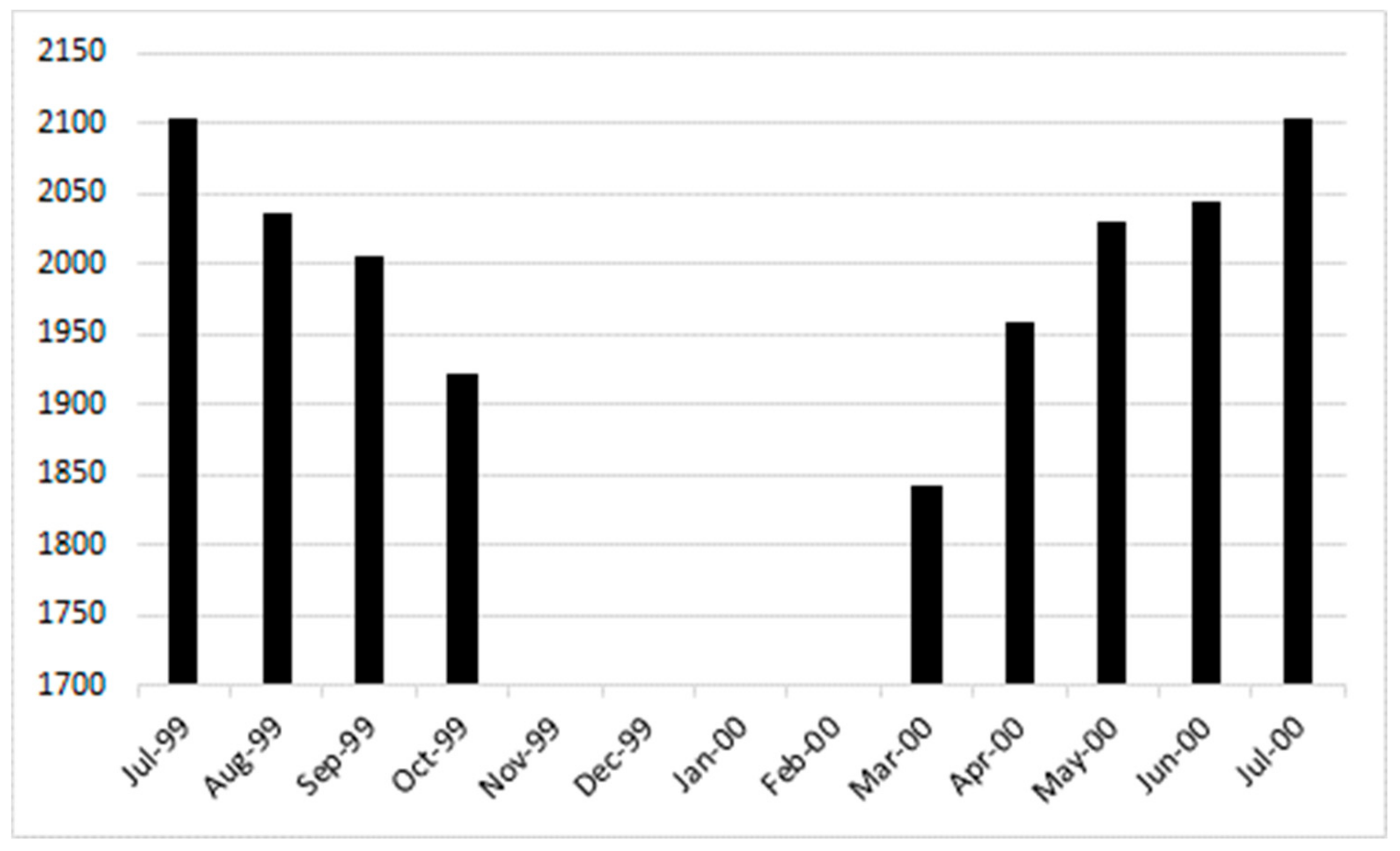
3.1. Emergence Patterns during Crop Season
3.2. Organochlorines in Guano
3.3. Organophosphates in Guano
3.4. Metals in Guano
3.5. Organochlorines in Carcasses
3.6. Carbamates and Pyrethroids
4. Discussion
4.1. Organochlorine Residues
4.2. Organophosphate Residues
4.3. Heavy Metal Residues
4.4. Patterns of Accumulation and Ecological Effects
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Costanza, R.; d’Arge, R.; de Groot, R.; Farber, S.; Grasso, M.; Hannon, B.; Limburg, K.; Naeem, S.; O’Neill, R.V.; Paruelo, J.; et al. The value of the world’s ecosystem services and natural capital. Nature 1997, 387, 253–260. [Google Scholar] [CrossRef]
- Hooper, D.U.; Chapin, F.S., III; Ewel, J.J.; Hector, A.; Inchausti, P.; Lavorel, S.; Lawton, J.H.; Lodge, D.M.; Loreau, M.; Naeem, S.; et al. Effects of biodiversity on ecosystem functioning: A consensus of current knowledge. Ecol. Monog. 2005, 75, 3–35. [Google Scholar] [CrossRef]
- Lacher, T.E., Jr.; Davidson, A.; Fleming, T.; Gomez-Ruiz, E.P.; McCracken, G.; Owen-Smith, N.; Peres, C.; Vander Wall, S. The functional role of mammals in ecosystems. J. Mamm. 2019, 100, 942–964. [Google Scholar] [CrossRef]
- Kunz, T.H.; de Torrez, E.B.; Bauer, D.; Lobova, T.; Fleming, T.H. Ecosystem services provided by bats. Ann. N. Y. Acad. Sci. 2011, 1223, 1–38. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.; Jacobs, D.S.; Kunz, T.H.; Willig, M.R.; Racey, P.A. Carpe noctem: The importance of bats as bioindicators. Endanger. Sp. Res. 2009, 8, 93–115. [Google Scholar] [CrossRef]
- Cleveland, C.J.; Betke, M.; Federico, P.; Frank, J.D.; Hallam, T.G.; Horn, J.; López, J.D., Jr.; McCracken, G.F.; Medellín, R.A.; Moreno-Valdez, A.; et al. Economic value of the pest control service provided by Brazilian free-tailed bats in south-central Texas. Front. Ecol. Environ. 2006, 4, 238–243. [Google Scholar] [CrossRef]
- Willig, M.R. A bat monitoring network for global change in the Anthropocene: Now or never. In Proceedings of the International Symposium on the Importance of Bats as Bioindicators; Flaquer, C., Puig-Montserrat, X., Eds.; Museum of Natural Sciences Edicions: Granollers, Catalonia, Spain, 2012; pp. 20–25. [Google Scholar]
- Hill, E.F. Organophosphorus and carbamate pesticides. In Handbook of Ecotoxicology; Hoffman, D.J., Rattner, B.A., Burton, G.A., Jr., Cairns, J., Jr., Eds.; Lewis Publishers: Boca Raton, FL, USA, 1995; pp. 243–273. [Google Scholar]
- Smith, G.J. Pesticide Use and Toxicology in Relation to Wildlife: Organophosphorus and Carbamate Compounds; Resource Publication, 170; U.S. Fish and Wildlife Service: Washington, DC, USA, 1987.
- Grue, C.E.; Hart, A.D.M.; Mineau, P. Biological consequences of depressed brain cholinesterase in wildlife. In Cholinesterase–Inhibiting Insecticides: Their Impact on Wildlife and the Environment; Mineau, P., Ed.; Elsevier Science Publishers: Amsterdam, The Netherlands, 1991; pp. 151–209. [Google Scholar]
- Eto, M. Organophosphorus Pesticides: Organic and Biological Chemistry; CRC Press: Cleveland, OH, USA, 1974. [Google Scholar]
- Kuhr, R.J.; Dorough, H.W. Carbamate Insecticides: Chemistry, Biochemistry, and Toxicology; CRC Press: Cleveland, OH, USA, 1976. [Google Scholar]
- Grue, C.E.; Fleming, W.J.; Busby, D.G.; Hill, E.F. Assessing hazards of organophosphate pesticides to wildlife. T. N. Am. Wildl. Nat. Res. 1983, 48, 200–220. [Google Scholar]
- Stromborg, K.L.; Grue, C.E.; Nichols, J.D.; Hepp, G.H.; Hines, J.E.; Bourne, H.C. Postfledgling survival of European starlings exposed to an organophosphorus insecticide. Ecology 1988, 50, 590–595. [Google Scholar] [CrossRef]
- Peterle, T.J. Wildlife Toxicology; Van Nostrand Reinhold: New York, NY, USA, 1991. [Google Scholar]
- Smith, A.G. Chlorinated hydrocarbon insecticides. In Handbook of Pesticide Toxicology; Hayes, W.J., Laws, E.R., Jr., Eds.; Academic Press: San Diego, CA, USA, 1991; pp. 816–850. [Google Scholar]
- Clark, D.R., Jr.; Bunck, C.M.; Cromartie, E.; LaVal, R.K. Year and age effects on residues of dieldrin and heptachlor in dead gray bats, Franklin County, Missouri–1976,1977, and 1978. Environ. Toxicol. Chem. 1983, 2, 387–391. [Google Scholar]
- Clark, D.R., Jr.; Clawson, R.L.; Stafford, C.J. Gray bats killed by dieldrin at two additional Missouri caves: Aquatic macroinvertebrates found dead. B. Environ. Contam. Tox. 1983, 30, 214–218. [Google Scholar] [CrossRef]
- Ammerman, L.K.; Hice, C.L.; Schmidly, D.J. Bats of Texas; Texas A&M University Press: College Station, TX, USA, 2012. [Google Scholar]
- Fitch, J.H.; Shump, K.A., Jr.; Shump, A.U. Myotis velifer. Mamm. Species 1981, 149, 1–5. [Google Scholar] [CrossRef]
- Shump, K.A. Cave myotis (Myotis velifer). In The Smithsonian Book of North American Mammals; Wilson, D.E., Ruff, S., Eds.; Smithsonian Institution Press: Washington, DC, USA, 1999; pp. 100–101. [Google Scholar]
- Warton, M. Status of work on cave mapping and gating on Fort Hood Military Reservation. In The Nature Conservancy Summary of 1997 Research Activities; Texas Data Center, The Nature Conservancy: Fort Hood, TX, USA, 1997; pp. 229–256. [Google Scholar]
- Reddell, J.R. Summary of past work on the cave biology of Ft. Hood, Bell and Coryell Counties, Texas. In The Nature Conservancy Summary of 1997 Research Activities; Texas Data Center, The Nature Conservancy: Fort Hood, TX, USA, 1997; pp. 257–314. [Google Scholar]
- Land, T.A. Population Size and Contaminant Exposure of Bats Using Caves on Fort Hood Military Base. Master’s Thesis, Texas A&M University, College Station, TX, USA, 2001. [Google Scholar]
- Land, T.A.; Lacher, T.E., Jr.; Clark, D.R., Jr. Emergence patterns of cave Myotis (Myotis velifer) on Fort Hood, in Central Texas. Bat. Res. News. 2003, 44, 92–94. [Google Scholar]
- Sikes, R.S.; Gannon, W.L. Animal Care and Use Committee of the American Society of Mammalogists. Guidelines of the American Society of Mammalogists for the use of wild mammals in research. J. Mamm. 2011, 92, 235–253. [Google Scholar] [CrossRef]
- Wade, T.L.; Atlas, E.L.; Brooks, J.M.; Kennicutt, M.C., II; Fox, R.G.; Sericano, J.L.; Garcia-Roero, B.; Defreitas, D.A. Gulf of Mexico status and trends program: Trace organic contaminant distribution in sediments and oysters. Estuaries 1988, 11, 1171–1179. [Google Scholar]
- Sabala, A.; Portillo, J.L.; Broto-Puig, F.; Commellas, L. Development of a new high-performance liquid chromatography method to analyze N-methylcarbamate insecticides by a simple post column derivatization system and fluorescence detection. J. Chromatogr. A 1997, 778, 103–110. [Google Scholar] [CrossRef]
- Pang, G.F.; Can, Y.Z.; Fan, C.L.; Zhang, J.J.; Li, X.M.; Mu, J.; Wang, D.; Liu, S.M.; Song, W.B.; Li, H.P.; et al. Interlaboratory study of identification and quantitation of multiresidue pyrethroids in agricultural products by gas chromatography-mass spectrometry. J. Chromatogr. A 2000, 882, 231–238. [Google Scholar] [CrossRef]
- Clark, D.R., Jr.; LaVal, R.K.; Tuttle, M.D. Estimating pesticide burdens of bats from guano analyses. B. Environ. Contam. Tox. 1982, 24, 214–220. [Google Scholar] [CrossRef]
- Kannan, K.; Yun, S.H.; Rudd, R.J.; Behr, M. High concentrations of persistent organic pollutants including PCBs, DDT, PBDEs and PFOS in little brown bats with white-nose syndrome in New York, USA. Chemosphere 2010, 80, 613–618. [Google Scholar] [CrossRef]
- United States Public Health Service. Toxicology Profile for Aldrin and Dieldrin; Agency for Toxic Substances and Disease Registry: Atlanta, GA, USA, 2002.
- Clark, D.R., Jr. DDT and the decline of free-tailed bats (Tadarida brasiliensis) at Carlsbad Caverns, New Mexico. Arch. Environ. Con. Tox. 2001, 40, 537–543. [Google Scholar]
- Clark, D.R., Jr.; Moreno-Valdez, A.; Mora, M.A. Organochlorine residues in bat guano from nine Mexican caves, 1991. Ecotoxicology 1995, 4, 258–265. [Google Scholar] [CrossRef]
- O’Shea, T.J.; Clark, D.R., Jr. An overview of contaminants and bats, with special reference to insecticides and the Indiana bat. In The Indiana bat: Biology and Management of an Endangered Species; Kurta, A., Kennedy, J., Eds.; Bat Conservation International: Austin, TX, USA, 2002; pp. 237–253. [Google Scholar]
- Mispagel, C.; Allinson, M.; Allinson, G.; Iseki, N.; Grant, C.; Morita, M. DDT and metabolites residues in the southern bent-wing bat (Miniopterus schreibersii bassanii) of south-eastern Australia. Chemosphere 2004, 55, 997–1003. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, D.J.; Rattner, B.A.; Burton, G., Jr.; Cairns, J., Jr. Introduction. In Handbook of Ecotoxicology; Lewis Publishers: Boca Raton, FL, USA, 1995; pp. 1–12. [Google Scholar]
- Wolfe, M.F.; Kendall, R.J. Age-dependent toxicity of diazinon and terbufos in European starlings (Sturnus vulgaris) and red-winged blackbirds (Agelaius phoeniceus). Environ. Toxicol. Chem. 1998, 17, 1300–1312. [Google Scholar] [CrossRef]
- Agosta, S.J. Habitat use, diet and roost selection by the Big Brown Bat (Eptesicus fuscus) in North America: A case for conserving an abundant species. Mamm. Rev. 2002, 32, 179–198. [Google Scholar] [CrossRef]
- Sandel, J.K. Insecticides and Bridge-Roosting Colonies of Mexican Free-Tailed Bats (Tadarida brasiliensis) in Texas. Master’s Thesis, Texas A&M University, College Station, TX, USA, 1999. [Google Scholar]
- Pain, D.J. Lead in the environment. In Handbook of Ecotoxicology; Hoffman, D.J., Rattner, B.A., Burton, G.A., Jr., Cairns, J., Jr., Eds.; Lewis Publishers: Boca Raton, FL, USA, 1995; pp. 356–430. [Google Scholar]
- Clark, D.R., Jr.; Shore, R.F. Chiroptera. In Ecotoxicology of Wild Mammals; Shore, R.F., Rattner, B.A., Eds.; John Wiley & Sons: New York, NY, USA, 2001; pp. 159–214. [Google Scholar]
- Theis, M.L.; Theis, K.M. Organochlorine residues in bats from Eckert James River Cave, Texas. Bull. Environ. Contam. Toxicol. 1997, 58, 673–680. [Google Scholar] [CrossRef]
- Desforges, J.-P.W.; Ross, P.S.; Loseto, L.L. Transplacental transfer of polychlorinated biphenyls and polybrominated diphenyl ethers in arctic beluga whales (Delphinapterus leucas). Environ. Toxicol. Chem. 2012, 31, 296–300. [Google Scholar] [CrossRef]
- Eidels, R.R.; Sparks, D.W.; Whitaker, J.O., Jr.; Sprague, C.A. Sub-lethal effects of chlorpyriphos on big brown bats (Eptesicus fuscus). Arch. Environ. Contam. Toxicol. 2016, 71, 322–335. [Google Scholar] [CrossRef]
- Stahlschmidt, P.; Brühl, C.A. Bats at risk? Bat activity and insecticide residue analysis of food items in an apple orchard. Environ. Toxicol. Chem. 2012, 31, 1556–1563. [Google Scholar] [CrossRef]
- Bernard, E.; Aguiar, L.M.S.; Machado, R.B. Discovering the Brazilian bat fauna: A task for two centuries? Mamm. Rev. 2010, 41, 23–39. [Google Scholar] [CrossRef]
- Oliveira, J.M.; Losano, N.F.; Condessa, S.S.; Pereira de Freitas, R.M.; Cardoso, S.A.; Freitas, M.B.; de Oliveira, L.L. Exposure to deltamethrin induces oxidative stress and decreases of energy reserve in tissues of the Neotropical fruit bat Artibeus lituratus. Ecotoxicol. Environ. Saf. 2018, 148, 684–692. [Google Scholar] [CrossRef]
- De Lange, H.J.; Lahr, J.; Van der Pol, J.J.C.; Wessels, Y.; Faber, J.H. Ecological vulnerability in wildlife: An expert judgment and multicriteria analysis tool using ecological traits to assess relative impact of pollutants. Environ. Toxicol. Chem. 2009, 28, 2233–2240. [Google Scholar] [CrossRef]
- Corso, A.E.; Woolley, J.B.; Lacher, T.E., Jr. Using digital photography and image analysis software to estimate the emergence of bats at Tou Santi Cave, Dominica, West Indies. Caribbean J. Sci. 2010, 46, 169–175. [Google Scholar] [CrossRef]
- Vaughan, T.A. Functional morphology of three bats: Eumops, Myotis, Macrotus. Univ. Kansas Publ. Mus. Nat. Hist. 1959, 12, 1–153. [Google Scholar]
- Kunz, T.H. Population studies of the cave bat (Myotis velifer): Reproduction, growth and development. Occas. Pap. Univ. Kansas Mus. Nat. Hist. 1973, 15, 1–43. [Google Scholar] [CrossRef]
- Kunz, T.H. Feeding ecology of a temperate insectivorous bat (Myotis velifer). Ecology 1974, 55, 693–711. [Google Scholar] [CrossRef]
- Lacher, T.E., Jr.; Roach, N. The status of biodiversity in the Anthropocene: Trends, threats, and actions. In Volume 3 (Biodiversity), The Encyclopedia of the Anthropocene; Lacher, T.E., Jr., Pyare, P., Eds.; Elsevier: Oxford, UK, 2018; pp. 1–8. [Google Scholar]
- Voigt, C.C.; Kingston, T. (Eds.) Bats in the Anthropocene: Conservation of Bats in a Changing World; Springer Open: Heidelberg, Germany, 2016. [Google Scholar]
- Williams-Guillén, K.; Olimpi, E.; Maas, B.; Taylor, P.J.; Arlettaz, R. Bats in the anthropogenic matrix: Challenges and opportunities for the conservation of Chiroptera and their ecosystem services in agricultural landscapes. In Bats in the Anthropocene: Conservation of Bats in a Changing World; Voigt, C.C., Kingston, T., Eds.; Springer Open: Heidelberg, Germany, 2016; pp. 151–186. [Google Scholar]
- Bayat, S.; Geiser, F.; Kristiansen, P.; Wilson, S.C. Organic contaminants in bats: Trends and new issues. Environ. Internat. 2014, 63, 40–52. [Google Scholar] [CrossRef] [PubMed]
| Date | Sample Number | Carcass Weight | Age | Sex | Reproduction |
|---|---|---|---|---|---|
| 09/23/99 | 11 | 5.82 g | adult | male | - |
| 09/23/99 | 13 | 4.68 g | adult | female | - |
| 03/13/00 | 12 | 5.93 g | adult | female | - |
| 05/14/00 | 07 | 0.92 g | fetal | female | - |
| 05/14/00 | 08 | 2.92 g | juvenile | female | - |
| 05/14/00 | 09 | 6.21 g | adult | female | lactating |
| 05/14/00 | 10 | 3.81 g | juvenile | male | - |
| Compound | Sample 1 Egypt | Sample 2 Tippit | Sample 3 SM (F1998) | Sample 4 SM (S1999) | Sample 5 SM (F1999) | Sample 6 SM (S2000) |
|---|---|---|---|---|---|---|
| Total PCB | 94.7 | 91.2 | 52.2 | 48.7 | 40.4 | 1114 |
| HCB | 2.49 | 2 | 40.2 | 44.9 | 38.6 | 66.1 |
| Lindane | 1.55 | 0.81 | 0.71 | 0.42 | 2.38 | 0.81 |
| Total Chlordanes | 193 | 163 | 91 | 121 | 25.4 | 95.6 |
| Dieldrin | 98.5 | 84.6 | 27.6 | 40.2 | 4.7 | 15.3 |
| Endrin | 6.87 | 2.95 | • | 1.48 | • | 1.85 |
| Mirex | 11 | 8.84 | 3.06 | 2.79 | • | 0.89 |
| Endosulfan II | 9.93 | 8.04 | 3.58 | 4.27 | • | 8.7 |
| p,p’-DDE | 240 | 186 | 63.1 | 63.3 | 24.5 | 92.4 |
| o,p’-DDT | 7.93 | 6.03 | 3.58 | 4.69 | • | 1.29 |
| Compound | Sample 1 Egypt | Sample 2 Tippit | Sample 3 SM (F1998) | Sample 4 SM (S1999) | Sample 5 SM (F1999) | Sample 6 SM (S2000) |
|---|---|---|---|---|---|---|
| Dichlorvos | • | • | • | 0.7 | 1.7 | 5 |
| Naled | 0.7 | • | • | 2.9 | 9.4 | 8.3 |
| Monochrotophos | • | • | • | • | 0.6 | 2.4 |
| Demetron-S | 1.5 | 0.6 | 7.4 | 2.4 | 12 | 14 |
| Dimethoate | • | • | • | • | 20.2 | 0.5 |
| Terbufos | 5 | 1.9 | 2 | 33.7 | 25.7 | 36 |
| Diazinon | 4.2 | 0.7 | • | 1.1 | 2 | 1.1 |
| EPN | 0.6 | • | 0.5 | 0.5 | 8 | 2.8 |
| Disulfoton | 2.2 | 0.5 | 1.3 | 2.5 | 13.5 | 13.9 |
| M parathion | 1.3 | • | 0.7 | 3.3 | 16.6 | 5.9 |
| Parathion | • | • | • | • | 1.3 | • |
| Trichloronate | • | • | • | • | • | 0.8 |
| Stiriphos | 1.6 | • | • | • | 2.4 | 2.2 |
| DEF (merphos) | 11.2 | 9.8 | 2.1 | 10.8 | 26.3 | 2.3 |
| Fensulfothion | • | • | • | • | 0.9 | • |
| Carbophenothion | 1.1 | • | 0.5 | 0.6 | 170 | 200 |
| Tokuthion | 0.8 | • | 1.7 | 18.4 | 481 | 138 |
| Methylazinphos | 0.9 | 1.2 | • | 1.7 | • | 2.9 |
| Compound | Sample 1 Egypt | Sample 2 Tippit | Sample 3 SM (F1998) | Sample 4 SM (S1999) | Sample 5 SM (F1999) | Sample 6 SM (S2000) |
|---|---|---|---|---|---|---|
| Arsenic | 7.17 | 8.64 | 6.87 | 7.31 | 6.17 | 2.54 |
| Cadmium | 1.07 | 1.21 | 1.0 | 1.13 | 0.83 | 1.7 |
| Lead | 4.33 | 4.47 | 4.14 | 4.53 | 3.26 | 75.6 |
| Chromium | 1.28 | 1.27 | 1.09 | 0.97 | 0.99 | 1.82 |
| Nickel | 2.45 | 2.93 | 1.96 | 2.07 | 1.94 | 1.26 |
| Mercury | 0.41 | 0.21 | 0.22 | 0.21 | 0.19 | 0.19 |
| Compound | Sample 7 Female-Fetal | Sample 8 Female Juvenile | Sample 9 Female Adult (Lactating) | Sample 10 Male Juvenile | Sample 11 Male Adult | Sample 12 Female Adult | Sample 13 Female Adult |
|---|---|---|---|---|---|---|---|
| Total PCBs | 48.8 | 76.7 | 31.1 | 77.2 | 192 | 38.6 | 28.4 |
| HCB | 4.44 | 14.9 | 2.41 | 20.4 | 6.29 | 6.38 | 8.84 |
| Lindane | • | • | • | 0.428 | • | 0.444 | • |
| Total Chlordanes | 16.2 | 42.6 | 7.29 | 49.3 | 66.1 | 23.2 | 20.2 |
| Dieldrin | 2.51 | 18.6 | 1.26 | 9.02 | 8.35 | 7.84 | 10.3 |
| Endrin | • | • | • | • | • | 1.29 | • |
| Mirex | • | 1.5 | 1.99 | 13.4 | 2.83 | 1.84 | • |
| Endosulfan II | • | 0.8 | • | 0.747 | • | 0.395 | • |
| p,p’-DDE | 119 | 753 | 99.8 | 294 | 266 | 172 | 106 |
| o,p’-DDT | • | 0.867 | • | • | 1.55 | • | • |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Land, T.A.; Clark, D.R., Jr.; Pekins, C.E.; Lacher, T.E., Jr. Seasonal Emergence and Historical Contaminant Exposure of Cave Myotis (Myotis velifer) in Central Texas and Current Status of the Population. Environments 2019, 6, 121. https://doi.org/10.3390/environments6120121
Land TA, Clark DR Jr., Pekins CE, Lacher TE Jr. Seasonal Emergence and Historical Contaminant Exposure of Cave Myotis (Myotis velifer) in Central Texas and Current Status of the Population. Environments. 2019; 6(12):121. https://doi.org/10.3390/environments6120121
Chicago/Turabian StyleLand, Tarisha A., Donald R. Clark, Jr., Charles E. Pekins, and Thomas E. Lacher, Jr. 2019. "Seasonal Emergence and Historical Contaminant Exposure of Cave Myotis (Myotis velifer) in Central Texas and Current Status of the Population" Environments 6, no. 12: 121. https://doi.org/10.3390/environments6120121
APA StyleLand, T. A., Clark, D. R., Jr., Pekins, C. E., & Lacher, T. E., Jr. (2019). Seasonal Emergence and Historical Contaminant Exposure of Cave Myotis (Myotis velifer) in Central Texas and Current Status of the Population. Environments, 6(12), 121. https://doi.org/10.3390/environments6120121





