Study of the Water Quality of a Tropical Reservoir
Abstract
1. Introduction
2. Materials and Methods
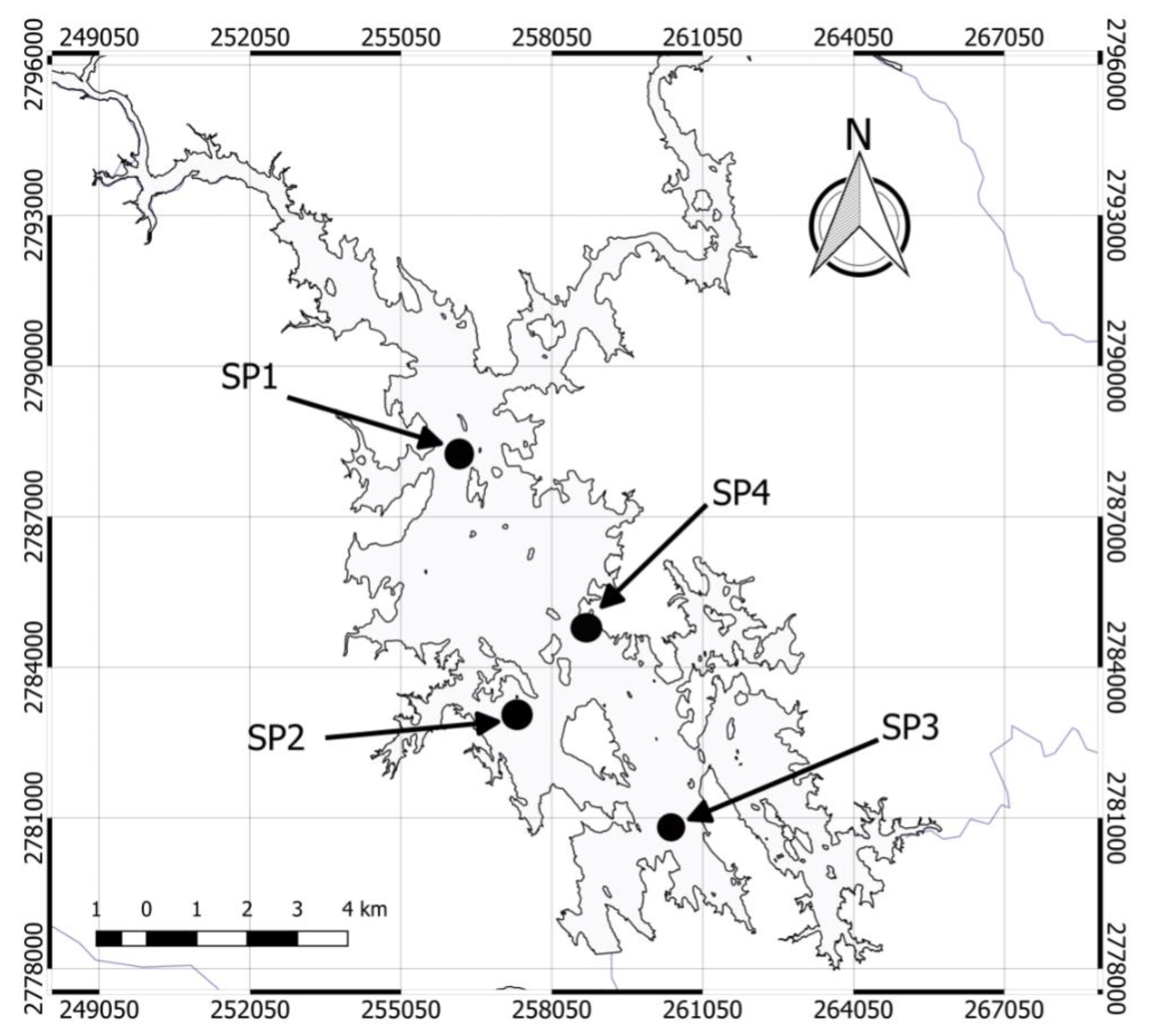
2.1. Study Area
2.2. Water Quality Index
2.3. Trophic State Index
2.4. Ecological Risk Index
3. Results and Discussion
3.1. Selection of Parameters
3.2. Water Quality Index
3.3. Trophic State Index
3.4. Ecological Risk Index
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ashinyani, N.; Suryanarayana, T.M. Assement of groundwater quality using GWQI method—A case study of Nadiad Taluka, Gujarat, India. In Proceedings of the Hydro 2015 International, 20th International Conference on Hydraulics, Water Resources and River Engineering, Hydro, Roorkee, India, 17–19 December 2015. [Google Scholar]
- Yu, S.; Xu, Z.; Wu, W.; Zuo, D. Effect of land use types on stream water quality under seasonalvariation and topographic characteristics in the Wei River basin, China. Ecol. Indic. 2016, 60, 202–212. [Google Scholar] [CrossRef]
- Vidhya, V.; John, M.; Mario, S. Does river restoration affect diurnal and seasonal changes to surface water quality? A study along the Thur River, Switzerland. Sci. Total Environ. 2015, 532, 91–102. [Google Scholar]
- Metcalfe, C.D.; Metcalfe, T.L.; Beddows, P.A.; Bouchot, G.G.; Li, H.; Van Lavieren, H. Contaminants in the coastal karst aquifer system along the Caribbean coast of the Yucatan peninsula, Mexico. Environ. Pollut. 2011, 159, 991–997. [Google Scholar] [CrossRef] [PubMed]
- Grill, G.; Ouellet Dallaire, C.; Fluet Chouinard, E.; Sindorf, N.; Lehner, B. Development of new indicators to evaluate river fragmentation and flow regulation at large scales: A case study for the Mekong river basin. Ecol. Indic. 2014, 45, 148–159. [Google Scholar] [CrossRef]
- Dimas, A.; Hinojosa, A.; Oller, P. Prefacio. In Boletín de la Sociedad Geológica Mexicana, Abril; Sociedad Geológica Mexican: Distrito Federa, Mexico, 2011; Volume 63, pp. i–ii. [Google Scholar]
- Cuthbertson, A.; Newkirk, C.; Llardo, J.; Loveridge, S.; Skidmore, M. Angry, Scared, and Unsure: Mental Health Consequences of Contaminated Water in Flint. J. Urban Health 2016, 93, 899–908. [Google Scholar] [CrossRef]
- Guillén, V.; Teck, H.; Kohlmann, B.; Yeomans, J. Microorganismos como bioindicadores de la Calidad del Agua. Tierra Trop. Sostenibilidad Ambiente y Sociedad 2012, 8, 65–93. [Google Scholar]
- Wu, Z.; Zhang, D.; Cai, Y.; Wang, X.; Zhang, L.; Chen, Y. Water quality assessment based on the water quality index method in Lake Poyang: The largest freshwater lake in China. Sci. Rep. 2017, 7, 17999. [Google Scholar] [CrossRef]
- Sharma, S.; Chhipa, R. Evaluation and Optimization of Water Quality Index for Ground Water Sourse of North West Jaipur and Agglomerates. Int. J. Chem. Sci. 2012, 10, 2297–2305. [Google Scholar]
- Li, W.; Gao, L.; Shi, Y.; Wang, Y.; Liu, J.; Cai, Y. Spatial distribution, temporal variation and risks of parabens and their chlorinated derivatives in urban surface water in Beijing, China. Sci. Total Environ. 2016, 539, 262–270. [Google Scholar] [CrossRef]
- Misaghi, F.; Delgosha, F.; Razzaghmanesh, M.; Myers, B. Introducing a water quality index for assessing water for irrigation purposes: A case study of the ghezel ozan river. Sci. Total Environ. 2017, 589, 107–116. [Google Scholar] [CrossRef]
- Bhadra, A.K.; Sahu, B.; Rout, S.P. Evaluation of the water quality index in river brahmani, odisha in the light of national sanitation foundation (NSF) standards. Asian J. Res. Chem. 2014, 7, 586–592. [Google Scholar]
- Simões, F.; Moreira, A.B.; Bisinoti, M.C.; Gimenez, S.M.; Yabe, M.J. Water quality index as a simple indicator of aquaculture effects on aquatic bodies. Ecol. Indic. 2008, 8, 476–484. [Google Scholar] [CrossRef]
- Behar, R.; Zúñiga de Cardozo, M.; Rojas, O. Análisis y Valoración del Índice de Calidad de Agua (ICA) de la NSF: Caso Ríos Cali y Meléndez. Ingeniería y Competitividad 1997, 1, 17–27. [Google Scholar]
- Liu, W.; Zhao, E.; Kuo, Y.; Jang, C. Identifying the relationships between trophic states and their driving factors in the Shihmen reservoir, Taiwan. Limnologica 2017, 64, 38–45. [Google Scholar] [CrossRef]
- De Freitas, P.; Reis Sousa, M.; Porfírio, A.F.; Almeida, B.S.; Furtado, R.; Santaella, S.T. Preliminary analysis on the use of trophic state indexes in a brazilian semiarid reservoir. Biol. Sci. 2015, 37, 309–318. [Google Scholar]
- Cañedo-Argüelles, M.; Rieradevall, M.; Farrés-Corell, R.; Newton, A. Annual characterisation of four mediterranean coastal lagoons subjected to intense human activity. Estuar. Coast. Shelf Sci. 2012, 114, 59–69. [Google Scholar] [CrossRef]
- Håkanson, L. An ecological risk index for aquatic pollution control. A sedimentological approach. Water Res. 1979, 14, 975–1001. [Google Scholar] [CrossRef]
- Wang, N.; Wang, A.; Kong, L.; He, M. Calculation and application of sb toxicity coefficient for potential ecological risk assessment. Sci. Total Environ. 2018, 610–611, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Jin, X.; Liu, D.; Lang, C.; Shan, B. Temporal and spatial variation of nitrogen and phosphorus and eutrophication assessment for a typical arid river—Fuyang river in northern China. J. Environ. Sci. 2017, 55, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yongqiang, Z.; Kun, S.; Qin, B.; Yao, X.; Zhang, Y. Optical properties and composition changes in chromophoric dissolved organic matter along trophic gradients: Implications for monitoring and assessing lake eutrophication. Water Res. 2018, 131, 255–263. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, M.; Wang, Q.; Zhuang, W.; Yuan, Y.; Yuan, Y.; Jiao, K.; Chen, Q. Calculation of Thallium’s toxicity coefficient in the potential ecological risk index: A case study. Chemosphere 2018, 194, 562–569. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Liu, J. Relationships between ecological risk indices for metals and benthic communities metrics in a macrophyte-dominated lake. Ecol. Indic. 2014, 40, 162–174. [Google Scholar] [CrossRef]
- Comisión Nacional del Agua (CONAGUA). Estadísticas del agua en México. Secretaria de Medio Ambiente y Recursos Naturales (SEMARNAT), México; Comisión Nacional del Agua (CONAGUA): Mexico City, Mexico, 2016.
- CONABIO. Cuenca Alta de los Ríos Culiacán y Humaya. Available online: http://www.conabio.gob.mx/conocimiento/regionalizacion/doctos/rhp_020.html (accessed on 15 March 2017).
- APHA. Standard Methods for the Examination of Water and Wastewater; APHA: Washington, DC, USA, 1992. [Google Scholar]
- Brown, R.M.; Mcclelland, N.; Deininger, R.A.; Tozer, R. A Water Quality Index—Do We Dare. In Proceedings of the National Symposium on Data and Instrumentation for Water Quality Management, Conference of State Sanitary Engineers and Wisconsin University, Madison, WI, USA, 21–23 July 1970; pp. 364–383. [Google Scholar]
- Khan, F.A.; Ansari, A. Eutrophication: An ecological vision. Bot. Rev. 2005, 71, 449–482. [Google Scholar] [CrossRef]
- López, M.; Madroñero, S. Estado trófico de un lago tropical de alta montaña: Caso laguna de La Cocha. Ciencia e Ingeniería Neogranadina 2015, 25, 21–42. [Google Scholar] [CrossRef]
- Wurzburger, N.; Wright, S.J. Fine-root responses to fertilization reveal multiple nutrient limitation in a lowland tropical forest. Ecology 2015, 96, 2137–2146. [Google Scholar] [CrossRef] [PubMed]
- Maavara, T.; Parsons, C.T.; Ridenour, C.; Stojanovic, S.; Dürr, H.H.; Powley, H.R.; Cappellen, P.V. Global phosphorus retention by river damming. Proc. Natl. Acad. Sci. USA 2015, 112, 15603–15608. [Google Scholar] [CrossRef] [PubMed]
- Munyati, C. A spatial analysis of eutrophication in dam reservoir water on the Molopo River at Mafikeng, South Africa. Sustain. Water Qual. Ecol. 2015, 6, 31–39. [Google Scholar] [CrossRef]
- Carlson, R.E. A trophic state index for lakes. Limnol. Oceanogr. 1974, 23, 361–369. [Google Scholar]
- Akin, B.S.; Kırmızıgül, O. Heavy metal contamination in surface sediments of Gökçekaya dam lake, Eskişehir, Turkey. Environ. Earth Sci. 2017, 76, 402. [Google Scholar] [CrossRef]
- López, E.; Barceló, I.; Solís, H.; Laure, A.; Avila, P.; Martínez-Delgadillo, S. Calculation of the Ecological Risk Index in the José Antonio Alzate Dam, State of Mexico, Mexico. Biol. Trace Elem. Res. 2009, 135, 121–135. [Google Scholar] [CrossRef]
- Kutlu, B. Dissemination of heavy-metal contamination in surface sediments of the uzunçayır dam lake, tunceli, Turkey. Hum. Ecol. Risk Assess. 2018, 24, 2182–2194. [Google Scholar] [CrossRef]
- Kükrer, S.; Erginal, A.E.; Şeker, S.; Karabıyıkoğlu, M. Distribution and environmental risk evaluation of heavy metal in core sediments from lake çıldır (NE Turkey). Environ. Monit. Assess. 2015, 187, 453. [Google Scholar] [CrossRef] [PubMed]
- USEPA. United States Environmental Protection Agency Understanding Variation in Partitioncoefficient, Kd, Values. Volume I: The Kd Model, Methods of Measurement, and Application of Chemical Reaction Codes; EPA: Washington, DC, USA, 1999.
- Cheng, Y.; Li, Y.; Ji, F.; Wang, Y. Global sensitivity analysis of a water quality model in the three gorges reservoir. Water. 2018, 10, 153. [Google Scholar] [CrossRef]
- Mahato, M.K.; Singh, G.; Singh, P.K.; Singh, A.K.; Tiwari, A.K. Assessment of mine water quality using heavy metal pollution index in a coal mining area of Damodar river basin, India. Bull. Environ. Contam. Toxicol. 2017, 99, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Akköz, C. The determination of some pollution parameters, water quality and heavy metal concentrations of Aci lake (Karapinar/Konya, Turkey). Transylv. Rev. Syst. Ecol. Res. 2017, 19, 1–18. [Google Scholar] [CrossRef]
- Wu, Z.; Wang, X.; Chen, Y.; Cai, Y.; Deng, J. Assessing river water quality using water quality index in lake taihu basin, China. Sci. Total Env. 2018, 612, 914–922. [Google Scholar] [CrossRef] [PubMed]
- Bao, K.; Liu, J.; You, X.; Shi, X.; Meng, B. A new comprehensive ecological risk index for risk assessment on luanhe river, china. Environ. Geochem. Hlth. 2018, 40, 1965–1978. [Google Scholar] [CrossRef]
- Dereli, H.; Dinçtürk, E. Population structure and some growth properties of Gibel carp (Carassius Gibelio) in a mesotrophic lake. Not. Sci. Biol. 2016, 8, 286–291. [Google Scholar] [CrossRef]
- Ewaid, S.H.; Abed, S.A. Water Quality Index for Al-Gharraf River, Southern Iraq. Egypt. J. Aquat. Res. 2017, 43, 117–122. [Google Scholar] [CrossRef]
- Jazzar, S.; Berrejeb, N.; Messaoud, C.; Marzouki, M.N.; Smaali, I. Growth parameters, photosynthetic performance, and biochemical characterization of newly isolated green microalgae in response to culture condition variations. Appl. Biochem. Biotechnol. 2016, 179, 1290–1308. [Google Scholar] [CrossRef]
- Carbajal-Hernández, J.J.; Sánchez-Fernández, L.P.; Villa-Vargas, L.A.; Carrasco-Ochoa, J.A.; Martínez-Trinidad, J.F. Water quality assessment in shrimp culture using an analytical hierarchical process. Ecol. Indic. 2013, 29, 148–158. [Google Scholar] [CrossRef]
- Chapman, D. Water Quality Assessments. A Guide to the Use of Biota, Sediments and Water in Environmental Monitoring, 2nd ed.; Chapman and Hall: London, UK, 1996. [Google Scholar]
- Thongtha, K.; Kasemsuwan, J. Numerical simulations of water quality measurement model in an opened-closed reservoir with contaminant removal mechanism. Int. J. Differ. Equ. 2018, 2018, 1343541. [Google Scholar] [CrossRef]
- Moreno, A.; Ramírez, J.J. Variación temporal de la condición trófica del lago Parque Norte, Medellín (Antioquia), Colombia. Actual. Biol. 2010, 32, 75–87. [Google Scholar]
- Santos, S.A.; dos Santos, T.R.; Furtado, M.S.; Henry, R.; Ferragut, C. Periphyton nutrient content, biomass and algal community on artificial substrate: Response to experimental nutrient enrichment and the effect of its interruption in a tropical reservoir. Limnology 2018, 19, 209–218. [Google Scholar] [CrossRef]
- Reimers, J.R.; Cai, Z.; Kobayashi, R.; Rätsep, M.; Freiberg, A.; Krausz, E. Formation of water-chlorophyll clusters in dilute samples of chlorophyll-a in ether at low temperature. Phys. Chem. Chem. Phys. Pccp. 2014, 16, 2323–2330. [Google Scholar] [CrossRef] [PubMed]
- Bachmann, R.W.; Hoyer, M.V.; Croteau, A.C.; Canfield, D.E., Jr. Factors related to secchi depths and their stability over time as determined from a probability sample of US lakes. Environ. Monit. Assess. 2017, 189, 206. [Google Scholar] [CrossRef]
- Rodriguez, S.; Gauna, L.; Martinez, G.; Acevedo, H.; Romero, C. Nitrate and Bacterial Contamination in Water. Tierra Latinoam. 2012, 30, 2018120335. [Google Scholar]
| Parameter | Units | Weight (wi) |
|---|---|---|
| FC | * CFU/100 mL | 0.15 |
| pH | pH units | 0.12 |
| BOD5 | mg/L | 0.1 |
| NO3− | mg/L | 0.1 |
| PO4− | mg/L | 0.1 |
| Tem | °C | 0.1 |
| Tur | ** NTU | 0.08 |
| TDS | mg/L | 0.08 |
| DO | mg/L | 0.17 |
| Eri | Potential Ecological Risk for Substance |
| <39 | Low |
| 40–79 | Moderate |
| 80–159 | Considerable |
| 160–319 | High |
| >320 | Very high |
| RIHAKANSON | Ecological Risk for Lake/Basin |
| <149 | Low |
| 150–299 | Moderate |
| 300–599 | Considerable |
| >600 | Very high |
| Ecological Indexes | Parameter | Units | Temporal Variation Average | Spatial Variation Average | Mean | Max Value | Min Value | Standard Deviation | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | SP1 | SP2 | SP3 | SP4 | |||||||
| WQINSF–BROWN | FC | CFU/100 mL | 206 | 2127 | 221 | 598 | 127 | 992 | 729 | 661 | 851 | 759 | 834 | 5794 | 1 | 1275.5 |
| pH | pH units | 7.58 | 7.37 | 7.76 | 7.71 | 8.26 | 9.25 | 8.05 | 7.98 | 8.07 | 8.01 | 7.91 | 9.5 | 7.07 | 0.62 | |
| BOD5 | mg/L | 6.02 | 2.66 | 5.38 | 3.03 | 2.0 | 3.46 | 3.63 | 3.64 | 3.53 | 3.43 | 3.45 | 9.7 | 2 | 2.3 | |
| NO3− | mg/L | 0.01 | 0.24 | 0.04 | 0.02 | 0.01 | 0.02 | 0.07 | 0.07 | 0.04 | 0.07 | 0.06 | 0.59 | 0.001 | 0.15 | |
| PO4− | mg/L | 0.04 | 0.03 | 0.02 | 0.03 | 0.01 | 0.02 | 0.03 | 0.03 | 0.02 | 0.02 | 0.02 | 0.09 | 0.001 | 0.02 | |
| Tem | °C | 32.4 | 31.2 | 29.1 | 28.8 | 30.7 | 29.2 | 29.9 | 30.1 | 29.8 | 30.1 | 29.69 | 33.8 | 24.5 | 2.73 | |
| Tur | NTU | 2.1 | 22.1 | 1.8 | 1.6 | 2.2 | 3.3 | 6.5 | 8.9 | 2.5 | 5.4 | 6.31 | 74 | 0.88 | 13.10 | |
| TDS | mg/L | 119 | 117 | 147 | 97 | 109 | 101 | 119 | 116 | 111 | 115 | 115 | 156 | 82 | 21.45 | |
| DO | mg/L | 3.65 | 5.07 | 7.28 | 6.77 | 9.63 | 9.90 | 7.50 | 7.20 | 7.54 | 7.22 | 7.3 | 12.1 | 2.93 | 2.62 | |
| TSICARLSON | Chla | mg/m3 | 2.4 | 18.1 | 4.03 | 12.7 | 4.2 | 3.5 | 8.5 | 7.62 | 11.3 | 6.07 | 9.2 | 37.3 | 0.05 | 9.5 |
| Tra | m | 2.4 | 0.63 | 1.8 | 2.7 | 1.12 | 1.39 | 1.51 | 1.65 | 1.71 | 1.62 | 1.6 | 3.4 | 0.4 | 0.86 | |
| TP | mg/L | 0.07 | 0.25 | 0.06 | 0.08 | 0.10 | 0.04 | 0.10 | 0.11 | 0.09 | 0.11 | 0.11 | 0.52 | 0.02 | 0.12 | |
| RIHÅKANSON | As | mg/L | 0.0014 | 0.0028 | 0.0023 | 0.0014 | 0.0014 | 0.0036 | 0.0014 | 0.0032 | 0.0014 | 0.0022 | 0.0020 | 0.0127 | 0.0014 | 0.0024 |
| Cd | mg/L | 0.0002 | 0.0002 | 0.0002 | 0.0002 | 0.0002 | 0.0002 | 0.0002 | 0.0002 | 0.0002 | 0.0002 | 0.0002 | 0.0002 | 0.0002 | 1.37 × 10−19 | |
| Cr | mg/L | 0.0003 | 0.0067 | 0.0003 | 0.0003 | 0.0011 | 0.0003 | 0.0014 | 0.00153 | 0.00181 | 0.0022 | 0.0017 | 0.0154 | 0.0003 | 0.0037 | |
| Hg | mg/L | 0.0001 | 0.0005 | 0.0001 | 0.0002 | 0.0001 | 0.0001 | 0.00016 | 0.00016 | 0.0003 | 0.00015 | 0.00019 | 0.0013 | 0.0001 | 0.00025 | |
| Pb | mg/L | 0.0015 | 0.0015 | 0.0015 | 0.0015 | 0.0015 | 0.0015 | 0.0015 | 0.0015 | 0.0015 | 0.0015 | 0.0015 | 0.0015 | 0.0015 | 8.78 × 10−19 | |
| Parameter | Chla | Tra | TP | FC | BOD5 | NO3− | pH | TDS | DO | PO4− | Tur |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Tra | −0.0714 | ||||||||||
| TP | 0.1035 | −0.3557 | |||||||||
| FC | 0.1865 | −0.4641 | 0.5241 | ||||||||
| BOD5 | −0.0096 | 0.3272 | −0.1678 | −0.1757 | |||||||
| NO3− | 0.1508 | −0.3848 | 0.89 | 0.6818 | −0.0806 | ||||||
| pH | −0.1622 | −0.2232 | −0.3966 | 0.0392 | −0.213 | −0.3271 | |||||
| TDS | −0.2484 | −0.0968 | −0.031 | −0.1798 | 0.2839 | 0.0908 | −0.3352 | ||||
| DO | −0.2632 | −0.1937 | −0.174 | −0.1482 | −0.1767 | −0.2293 | 0.7597 | −0.3385 | |||
| PO4− | 0.3632 | 0.3133 | 0.3395 | 0.1128 | 0.2151 | 0.4045 | −0.2635 | 0.0561 | −0.3643 | ||
| Tur | 0.1846 | −0.4621 | 0.7583 | 0.826 | −0.0798 | 0.8493 | −0.2597 | 0.0237 | −0.2985 | 0.2702 | |
| Tem | −0.1783 | −0.2145 | 0.1391 | −0.0985 | −0.3134 | 0.0526 | −0.3628 | 0.4309 | −0.4706 | −0.0406 | 0.0617 |
| Year | Sub | Parameters | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| FC | pH | BOD5 | NO3− | PO4− | Tem | Tur | TDS | DO | ||
| 2012 | Sub2012 | 83.89 | 100 | 30.12 | 100 | 100 | 10 | 94.48 | 65.47 | 17.42 |
| Sub2012 * wi | 12.58 | 12 | 3.01 | 10 | 10 | 1 | 7.56 | 5.24 | 2.96 | |
| 2013 | Sub2013 | 12.49 | 100 | 58.85 | 100 | 100 | 10 | 62.21 | 66.24 | 45.34 |
| Sub2013 * wi | 1.87 | 12 | 5.89 | 10 | 10 | 1 | 4.98 | 5.30 | 7.71 | |
| 2014 | Sub2014 | 82.65 | 100 | 34.22 | 100 | 100 | 10 | 96.8 | 56.65 | 77.56 |
| Sub2014 * wi | 12.40 | 12 | 3.42 | 10 | 10 | 1 | 7.74 | 4.53 | 13.19 | |
| 2015 | Sub2015 | 56.90 | 100 | 54.67 | 100 | 100 | 12.14 | 99.22 | 73.68 | 71.34 |
| Sub2015 * wi | 8.54 | 12 | 5.47 | 10 | 10 | 1.21 | 7.94 | 5.89 | 12.13 | |
| 2016 | Sub2016 | 90.78 | 87.38 | 67.12 | 100 | 100 | 10 | 93.26 | 69.08 | 96.81 |
| Sub2016 * wi | 13.62 | 10.49 | 6.71 | 10 | 10 | 1 | 7.46 | 5.53 | 16.46 | |
| 2017 | Sub2017 | 38.5 | 52.28 | 50.14 | 100 | 100 | 10 | 87.15 | 71.99 | 98.04 |
| Sub2017 * wi | 5.78 | 6.27 | 5.01 | 10 | 10 | 1 | 6.97 | 5.76 | 16.67 | |
| SP | Parameters | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| FC | pH | BOD5 | NO3− | PO4− | Tem | Tur | TDS | DO | ||
| SP1 | SubSP1 | 49.97 | 97.44 | 48.50 | 100.00 | 100.00 | 10.00 | 77.31 | 65.79 | 80.02 |
| SubSP1 * wi | 7.50 | 11.69 | 4.85 | 10.00 | 10.00 | 1.00 | 6.19 | 5.26 | 13.6 | |
| SP2 | SubSP2 | 53.42 | 100.00 | 48.50 | 100.00 | 100.00 | 10.00 | 73.11 | 66.81 | 76.64 |
| SubSP2 * wi | 8.01 | 12.00 | 4.85 | 10.00 | 10.00 | 1.00 | 5.85 | 5.35 | 13.03 | |
| SP3 | SubSP3 | 44.26 | 96.43 | 49.48 | 100.00 | 100.00 | 10.00 | 91.49 | 68.46 | 80.46 |
| SubSP3 * wi | 6.64 | 11.57 | 4.95 | 10.00 | 10.00 | 1.00 | 7.32 | 5.48 | 13.68 | |
| SP4 | SubSP4 | 48.51 | 100 | 52.32 | 100.00 | 100.00 | 10.00 | 78.77 | 67.12 | 76.05 |
| SubSP4 * wi | 7.28 | 12.00 | 5.23 | 10.00 | 10.00 | 1.00 | 6.30 | 5.37 | 12.93 | |
| Parameters | Temporal Variation | Spatial Variation | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | SP1 | SP2 | SP3 | SP4 | |
| TSIChla | 39.53 | 59.01 | 44.27 | 55.60 | 44.69 | 42.93 | 51.65 | 50.53 | 54.44 | 48.29 |
| TSITra | 47.31 | 66.48 | 51.53 | 45.23 | 58.30 | 55.24 | 54.02 | 52.73 | 52.26 | 53.00 |
| TSITP | 22.38 | 40.31 | 19.68 | 24.53 | 26.88 | 14.54 | 27.46 | 28.52 | 26.37 | 28.01 |
| TSICARLSON | 36.41 | 55.27 | 38.50 | 41.79 | 43.29 | 37.57 | 44.37 | 43.92 | 44.36 | 43.10 |
| TSISPATIAL-AVERAGE | 43.94 | |||||||||
| TSITEMPORAL-AVERAGE | 42.14 | |||||||||
| TSIGENERAL-AVERAGE | 43.04 | |||||||||
| Parameters | Temporal Variation | Spatial Variation | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | SP1 | SP2 | SP3 | SP4 | |
| FC | 12.58 | 1.87 | 12.40 | 8.54 | 13.62 | 5.78 | 7.50 | 8.01 | 6.64 | 7.28 |
| pH | 12 | 12 | 12 | 12 | 10.49 | 6.27 | 11.69 | 12 | 11.57 | 12 |
| BOD5 | 3.01 | 5.89 | 3.42 | 5.47 | 6.71 | 5.01 | 4.85 | 4.85 | 4.95 | 5.23 |
| NO3− | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
| PO4− | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
| Tem | 1 | 1 | 1 | 1.21 | 1 | 1 | 1 | 1 | 1 | 1 |
| Tur | 7.56 | 4.98 | 7.74 | 7.94 | 7.46 | 6.97 | 6.19 | 5.85 | 7.32 | 6.3 |
| TDS | 5.24 | 5.3 | 4.53 | 5.89 | 5.53 | 5.76 | 5.26 | 5.35 | 5.48 | 5.37 |
| DO | 2.96 | 7.71 | 13.19 | 12.13 | 16.46 | 16.67 | 13.60 | 13.03 | 13.68 | 12.93 |
| WQINSF–BROWN | 64 | 59 | 74 | 73 | 81 | 67 | 70 | 70 | 71 | 70 |
| WQISPATIAL-AVERAGE | 70 | |||||||||
| WQITEMPORAL-AVERAGE | 70 | |||||||||
| WQIGENERAL-AVERAGE | 70 | |||||||||
| Parameter | Tri | Degree of Contamination (Cd) | ||
|---|---|---|---|---|
| Temporal (2012–2017) | Spatial (SP1-SP4) | General | ||
| As | 15 | 0.00086 | 0.00054 | 0.00140 |
| Cd | 1 | 0.0012 | 0.0008 | 0.0020 |
| Cr | 90 | 0.0001 | 0.00007 | 0.00017 |
| Hg | 0.25 | 0.0044 | 0.0030 | 0.0074 |
| Pb | 70 | 0.00012 | 0.00008 | 0.00021 |
| Parameter (Tri * Cif) | Temporal Variation | Spatial Variation | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | SP1 | SP2 | SP3 | SP4 | |
| As | 0.0009 | 0.0018 | 0.0015 | 0.0009 | 0.0009 | 0.0024 | 0.0009 | 0.0021 | 0.0009 | 0.0014 |
| Cd | 0.0060 | 0.0060 | 0.0060 | 0.0060 | 0.0060 | 0.0060 | 0.0060 | 0.0060 | 0.0060 | 0.0060 |
| Cr | 0.000007 | 0.00014 | 0.000007 | 0.000007 | 0.00002 | 0.000007 | 0.00003 | 0.00001 | 0.00004 | 0.00005 |
| Hg | 0.016 | 0.08 | 0.016 | 0.032 | 0.016 | 0.016 | 0.0256 | 0.0256 | 0.048 | 0.024 |
| Pb | 0.0001 | 0.0001 | 0.0001 | 0.0001 | 0.0001 | 0.0001 | 0.0001 | 0.0001 | 0.0001 | 0.0001 |
| ∑Eri | 0.0230 | 0.0881 | 0.0236 | 0.0390 | 0.0230 | 0.0245 | 0.0326 | 0.0338 | 0.0550 | 0.0316 |
| RISPATIAL | 0.1530 | |||||||||
| RITEMPORAL | 0.2212 | |||||||||
| RIGENERAL | 0.3742 | |||||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quevedo-Castro, A.; Lopez, J.L.; Rangel-Peraza, J.G.; Bandala, E.; Bustos-Terrones, Y. Study of the Water Quality of a Tropical Reservoir. Environments 2019, 6, 7. https://doi.org/10.3390/environments6010007
Quevedo-Castro A, Lopez JL, Rangel-Peraza JG, Bandala E, Bustos-Terrones Y. Study of the Water Quality of a Tropical Reservoir. Environments. 2019; 6(1):7. https://doi.org/10.3390/environments6010007
Chicago/Turabian StyleQuevedo-Castro, Alberto, Jesús L. Lopez, Jesús Gabriel Rangel-Peraza, Erick Bandala, and Yaneth Bustos-Terrones. 2019. "Study of the Water Quality of a Tropical Reservoir" Environments 6, no. 1: 7. https://doi.org/10.3390/environments6010007
APA StyleQuevedo-Castro, A., Lopez, J. L., Rangel-Peraza, J. G., Bandala, E., & Bustos-Terrones, Y. (2019). Study of the Water Quality of a Tropical Reservoir. Environments, 6(1), 7. https://doi.org/10.3390/environments6010007







