Removal of Synthetic Dyes by Dried Biomass of Freshwater Moss Vesicularia Dubyana: A Batch Biosorption Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Moss Biomass
2.2. Reagents and Instruments
2.3. Sorption Kinetics in Single and Binary Solutions in Batch System
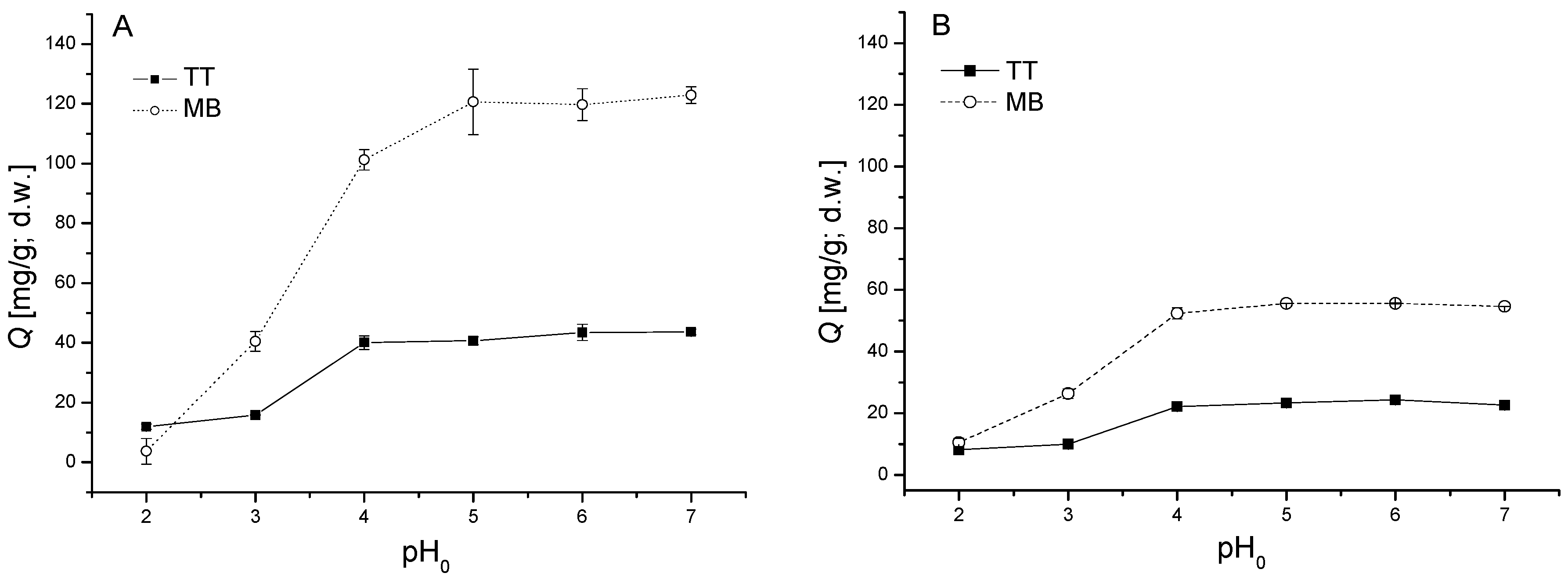
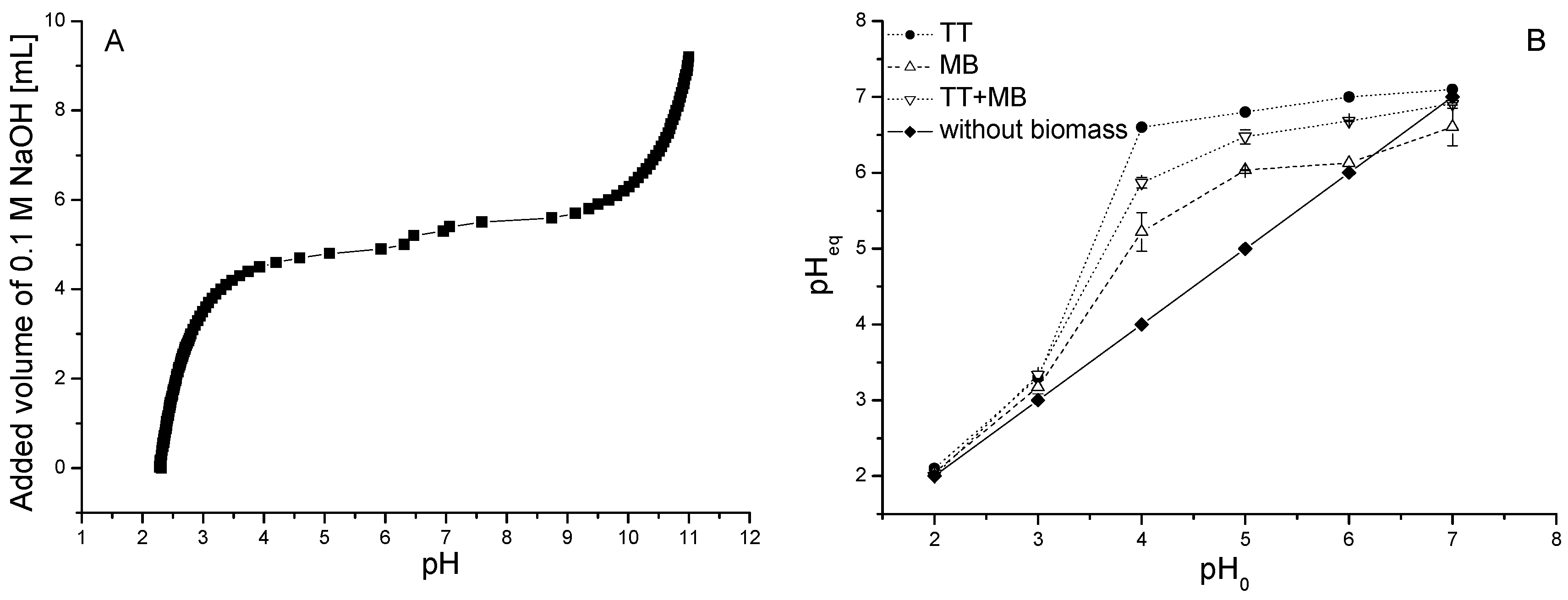
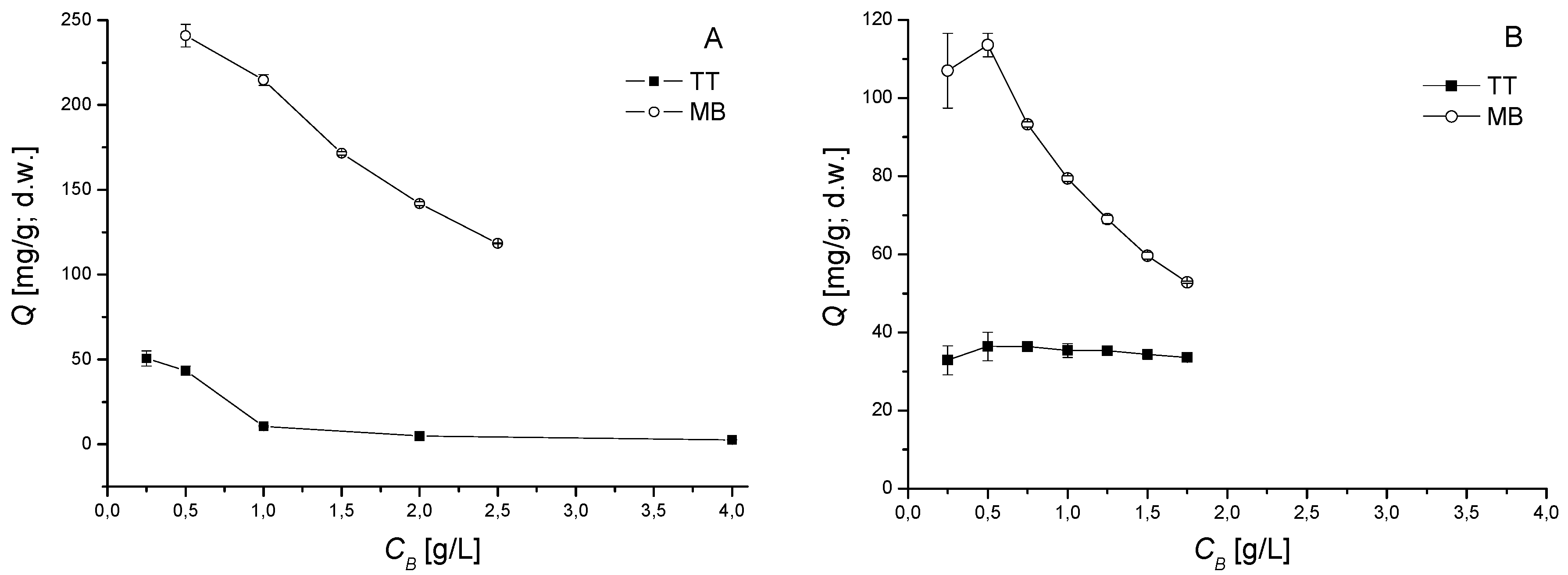
2.4. Influence of pH and Biomass Dosage
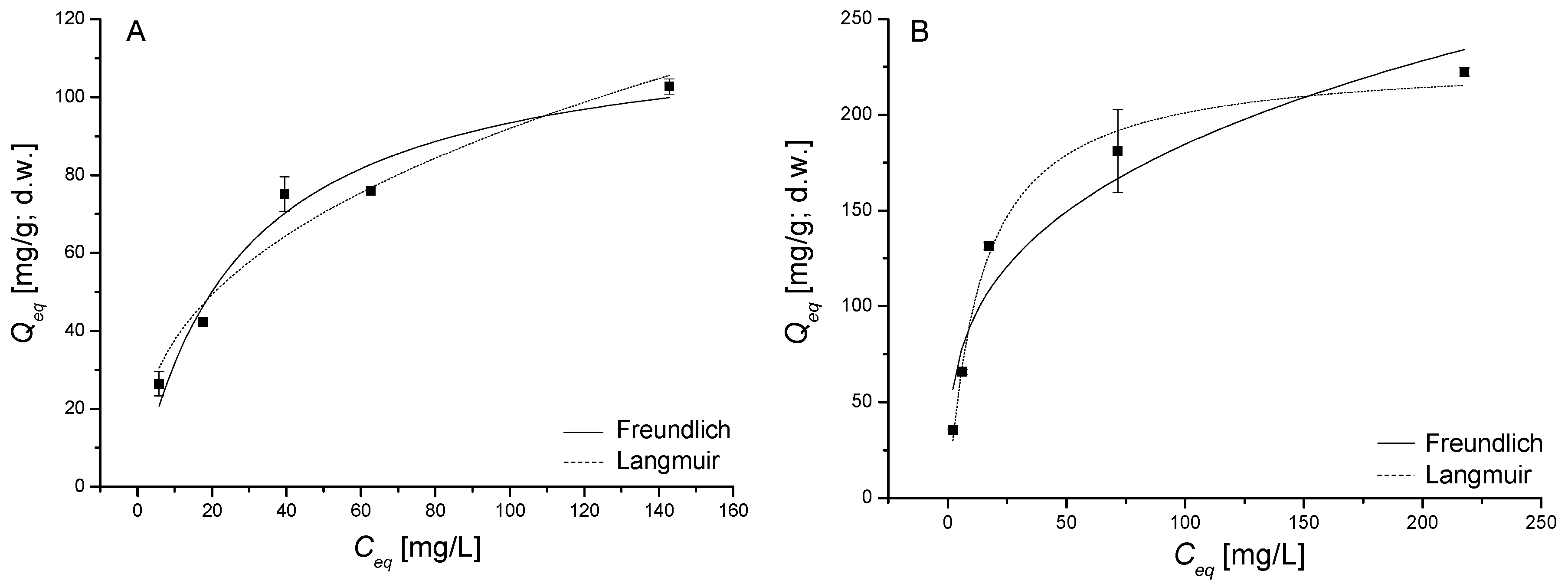
2.5. Sorption Equilibrium in Single and Binary Solutions in Batch System
2.6. Potentiometric Titration
3. Results and Discussion
3.1. Biosorption Kinetics in Single and Binary Systems
3.2. Influence of pH
3.3. Influence of Biomass Dosage
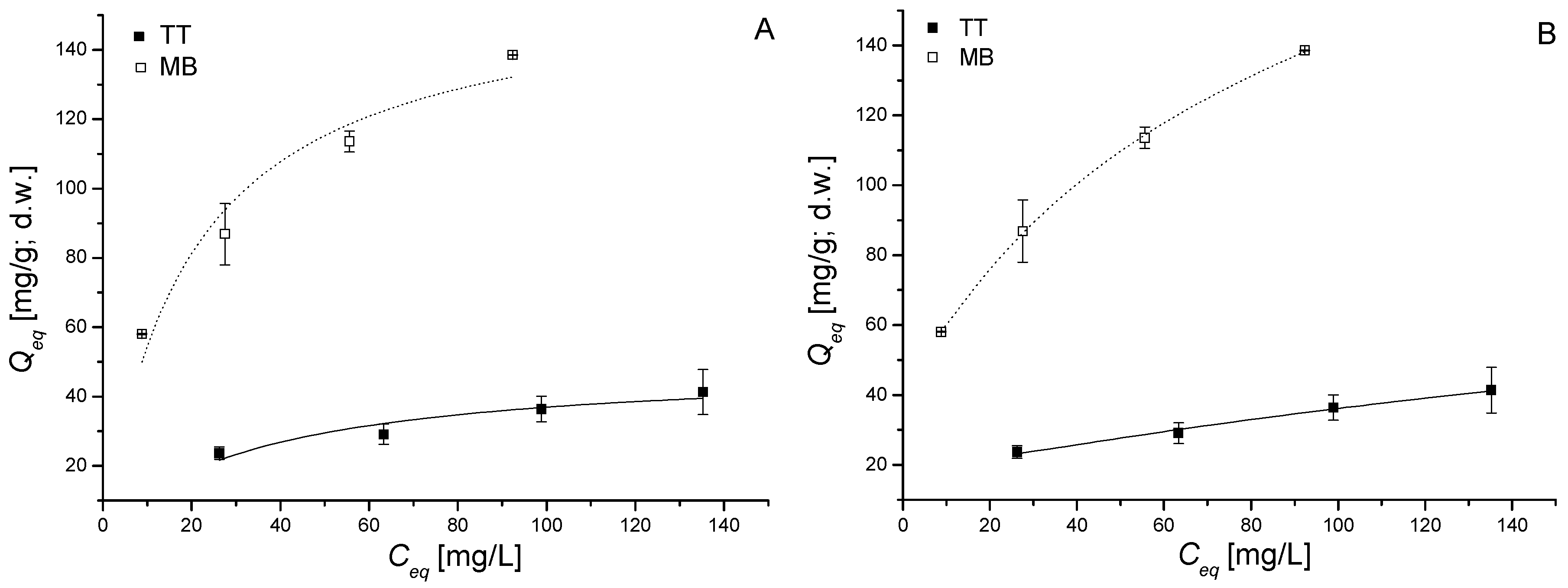
3.4. Sorption Equilibrium in Single and Binary Systems
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ghaly, A.E.; Ananthasankhar, R.; Alhattab, M.; Ramakrishnan, V.V. Production, characterization and treatment of textile effluents: A critical review. J. Chem. Eng. Process Technol. 2014, 5, 182. [Google Scholar] [CrossRef]
- Market Research Future. Global Textile Dyes Market Research Report—Forecast to 2023; ID: MRFR/CnM/2238-HCRR; Market Research Future: Maharashtra, India, 2017. [Google Scholar]
- GosReports. Global Textile Dyes Industry 2015 Market Research Report. 2015. Available online: https://www.prnewswire.com/news-releases/global-textile-dyes-industry-report-2015---forecasts-to-2020-498532981.html (accessed on 2 April 2015).
- Pereira, L.; Alves, M. Dyes—Environmental impact and remediation. In Environmental Protection Strategies for Sustainable Development; Malik, A., Grohmann, E., Eds.; Springer Science + Business Media B.V.: Dordrecht, The Netherlands, 2012; pp. 111–162. ISBN 978-94-007-1591-2. [Google Scholar]
- Dias, A.A.; Sampaio, A.; Bezerra, R.M. Environmental Applications of Fungal and Plant Systems: Decolourisation of Textile Wastewater and Related Dyestuffs. In Environmental Bioremediation Technologies; Singh, S.N., Tripathi, R.D., Eds.; Springer: Berlin/Heidelberg, Germany, 2007; pp. 445–463. ISBN 978-3-540-34790-3. [Google Scholar]
- Sing, K.; Arora, S. Removal of synthetic textile dyes from wastewaters: A critical review on present treatment technologies. Crit. Rev. Environ. Sci. Technol. 2011, 41, 807–878. [Google Scholar] [CrossRef]
- Robinson, T.; McMullan, G.; Marchant, R.; Nigam, P. Remediation of dyes in textile effluent: A critical review on current treatment technologies with a proposed alternative. Bioresour. Technol. 2001, 77, 247–255. [Google Scholar] [CrossRef]
- Paşka, O.M.; Pəcurariu, C.; Muntean, S.G. Kinetic and thermodynamic studies on methylene blue biosorption using corn-husk. RSC Adv. 2014, 4, 62621–62630. [Google Scholar] [CrossRef]
- Daneshvar, E.; Vazirzadeh, A.; Niazi, A.; Sillanpää, M.; Bhatnagar, A. A comparative study of methylene blue biosorption using different modified brown, red and green macroalgae—Effect of pretreatment. Chem. Eng. J. 2017, 307, 435–446. [Google Scholar] [CrossRef]
- Liang, J.; Xia, J.; Long, J. Biosorption of methylene blue by nonliving biomass of the brown macroalga Sargassum hemiphyllum. Water Sci. Technol. 2017, 76, 1574–1583. [Google Scholar] [CrossRef] [PubMed]
- Horník, M.; Šuňovská, A.; Partelová, D.; Pipíška, M.; Augustín, J. Continuous sorption of synthetic dyes on dried biomass of microalga Chlorella pyrenoidosa. Chem. Pap. 2013, 67, 254–264. [Google Scholar] [CrossRef]
- Zhao, S.; Zhou, T. Biosorption of methylene blue from wastewater by an extraction residue of Salvia miltiorrhiza Bge. Bioresour. Technol. 2017, 219, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Li, E.; You, X.; Hu, C.; Hu, Q. Adsorption of methylene blue on an agro-waste oiltea shell with and without fungal treatment. Sci. Rep. 2016, 6, 38450. [Google Scholar] [CrossRef] [PubMed]
- Maurya, N.S.; Mittal, A.K. Biosorptive uptake of cationic dyes from aqueous phase using immobilised dead macro fungal biomass. Int. J. Environ. Technol. Manag. 2011, 14, 282–293. [Google Scholar] [CrossRef]
- Rizzi, V.; D’Agostino, F.; Fini, P.; Semeraro, P.; Cosma, P. An interesting environmental friendly cleanup: The excellent potential of olive pomace for disperse blue adsorption/desorption from wastewater. Dyes Pigment. 2017, 140, 480–490. [Google Scholar] [CrossRef]
- Rizzi, V.; D’Agostino, F.; Gubitosa, J.; Fini, P.; Petrella, A.; Agostiano, A.; Semeraro, P.; Cosma, P. An Alternative use of olive pomace as a wide-ranging bioremediation strategy to adsorb and recover disperse orange and disperse red industrial dyes from wastewater. Separations 2017, 4, 29. [Google Scholar] [CrossRef]
- Semeraro, P.; Rizzi, V.; Fini, P.; Matera, S.; Cosma, P.; Franco, E.; García, R.; Ferrándiz, M.; Núñez, E.; Gabaldón, J.A.; et al. Interaction between industrial textile dyes and cyclodextrins. Dyes Pigment. 2015, 119, 84–94. [Google Scholar] [CrossRef]
- Albadarin, A.B.; Mangwandi, C. Mechanism of Alizarin red S and Methylene blue biosorption onto a olive stone by-product: Isotherm study in single and binary systems. J. Environ. Manag. 2015, 164, 86–93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Remenárová, L.; Pipíška, M.; Horník, M.; Augustín, J. Biosorption of cationic dyes BY1, BY2 and BG4 by moss Rhytidiadelphus squarrosus from binary solutions. Nova Biotechnol. 2009, 9, 239–247. [Google Scholar]
- Giwa, A.R.A.; Abdulsalam, K.A.; Wewers, F.; Oladipo, M.A. Biosorption of acid dye in single and multidye systems onto sawdust of locust bean (Parkia biglobosa) tree. J. Chem. 2016, 2016, 6436039. [Google Scholar] [CrossRef]
- Hoagland, D.R. Optimum nutrient solution for plants. Science 1920, 52, 562–564. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, W.; Xu, M.; Zheng, F.; Zhao, M. Study of the mechanisms of Cu2+ biosorption by ethanol/caustic-pretreated baker’s yeast biomass. J. Hazard. Mater. 2010, 178, 1085–1093. [Google Scholar] [CrossRef] [PubMed]
- Ncibi, M.C.; Ben Hamissa, A.M.; Fathallah, A.; Kortas, M.H.; Baklouti, T.; Mahjoub, B.; Seffen, M. Biosorptive uptake of methylene blue using Mediterranean green alga Enteromorpha spp. J. Hazard. Mater. 2009, 170, 1050–1055. [Google Scholar] [CrossRef] [PubMed]
- Hameed, B.H. Grass waste: A novel sorbent for the removal of basic dye from aqueous solution. J. Hazard. Mater. 2009, 166, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, M.E.; Nunell, G.V.; Bonelli, P.R.; Cukierman, A.L. Batch and dynamic biosorption of basic dyes from binary solutions by alkaline-treated cypress cone chips. Bioresour. Technol. 2012, 106, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Salazar-Rabago, J.J.; Leyva-Ramos, R.; Rivera-Utrilla, J.; Ocampo-Perez, R.; Cerino-Cordova, F.J. Biosorption mechanism of Methylene Blue from aqueous solution onto White Pine (Pinus durangensis) sawdust: Effect of operating conditions. Sustain. Environ. Res. 2017, 27, 32–40. [Google Scholar] [CrossRef]
- Partelová, D.; Šuňovská, A.; Marešová, J.; Horník, M.; Pipíška, S.; Hostin, S. Removal of contaminats from aqueous solutions using hop (Humulus lupulus L.) agricultural by-products. Nova Biotechnol. Chim. 2015, 14, 212–227. [Google Scholar] [CrossRef]
- Tabaraki, R.; Sadeghinejad, N. Biosorption of six basic and acidic dyes on brown alga Sargassum ilicifolium: Optimization, kinetics and isotherm studies. Water Sci. Technol. 2017, 75, 2631–2638. [Google Scholar] [CrossRef] [PubMed]
- Tural, B.; Ertaş, E.; Enez, B.; Fincan, S.A.; Tural, S. Preparation and characterization of a novel magnetic biosorbent functionalized with biomass of Bacillus subtilis: Kinetic and isotherm studies of biosorption processes in the removal of Methylene Blue. J. Environ. Chem. Eng. 2017, 5, 4795–4802. [Google Scholar] [CrossRef]
- Kumar, K.V.; Porkodi, K. Mass transfer, kinetics and equilibrium studies for the biosorption of methylene blue using Paspalum notatum. J. Hazard. Mater. 2007, 146, 214–226. [Google Scholar] [CrossRef] [PubMed]
- Uddin, N.; Islam, T.; Das, S. A novel biosorbent, water-hyacinth, uptaking methylene blue from aqueous solution: Kinetics and equilibrium studies. Int. J. Chem. Eng. 2014, 2014, 819536. [Google Scholar] [CrossRef]
- Zhu, L.; Wang, Y.; Zhu, F.; You, L.; Shen, X. Evaluation of the biosorption characteristics of Tremella fuciformis for the decolorization of cationic dye from aqueous solution. J. Polym. Environ. 2017, in press. [Google Scholar] [CrossRef]
- Hamitouche, A.; Haffas, M.; Boudjemaa, A.; Benammar, S.; Sehailia, M.; Bachari, K. Efficient biosorption of methylene blue, malachite green and methyl violet organic pollutants on biomass derived from Anethum graveolens: An eco-benign approach for wastewater treatment. Desalin. Water Treat. 2017, 5, 225–236. [Google Scholar] [CrossRef]
- Remenárová, L.; Pipíška, M.; Horník, M.; Augustín, J. Sorption of cationic dyes from aqueous solution by moss Rhytidiadelphus squarrosus: Kinetics and nequilibrium study. Nova Biotechnol. 2009, 9, 75–84. [Google Scholar]
- Shin, W.S. Competitive sorption of anionic and cationic dyes onto cetylpyridinium-modified montmorillonite. J. Environ. Sci. Health A Toxic Hazard. Subst. Environ. Eng. 2008, 43, 1459–1470. [Google Scholar] [CrossRef] [PubMed]
- Kuppusamy, S.; Venkateswarlu, K.; Thavamani, P.; Lee, Y.B.; Naidu, R.; Megharaj, M. Quercus robur acorn peel as a novel coagulating adsorbent for cationic dye removal from aquatic ecosystems. Ecol. Eng. 2017, 101, 3–8. [Google Scholar] [CrossRef]
- Afroze, S.; Sen, T.K.; Ang, M.; Nishioka, H. Adsorption of methylene blue dye from aqueous solution by novel biomass Eucalyptus sheathiana bark: Equilibrium, kinetics, thermodynamics and mechanism. Desalin. Water Treat. 2016, 57, 5858–5878. [Google Scholar] [CrossRef]
- Kong, L.; Gong, L.; Wang, J. Removal of methylene blue from wastewater using fallen leaves as an adsorbent. Desalin. Water Treat. 2015, 53, 2489–2500. [Google Scholar] [CrossRef]
- Hameed, B.H.; Mahmoud, D.K.; Ahmad, A.L. Sorption equilibrium and kinetics of basic dye from aqueous solution using banana stalk waste. J. Hazard. Mater. 2008, 158, 499–506. [Google Scholar] [CrossRef] [PubMed]

| Functional Group | pKa | CAn (mmol/g) |
|---|---|---|
| COOH | 2.34–6.25 | 0.98 |
| PO3H2 | 6.36–8.72 | 0.69 |
| NH2 | 10.7–11.5 | 0.61 |
| OH | 8.36–12.0 | 0.64 |
| Dye | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| Qmax (mg/g) | b (L/mg) | R2 | K (L/g) | 1/n | R2 | |
| TT | 119 ± 11 | 0.04 ± 0.01 | 0.965 | 15.5 ± 3.8 | 2.58 ± 0.38 | 0.955 |
| MB | 229 ± 9 | 0.07 ± 0.01 | 0.990 | 45.2 ± 11.5 | 3.27 ± 0.59 | 0.938 |
| Sorbent | Qmax MB (mg/g) | Qmax TT (mg/g) | pH | T (°C) | Reference |
|---|---|---|---|---|---|
| V. dubyana | 229 | 119 | 6.0 | 25 | present study |
| hop leaf biomass | 184 | 77.6 | 6.0 | 25 | [27] |
| Rhytidiadelphus squarrosus | - | 98.9 | 4.0 | 25 | [34] |
| montmorillonite * | - | 95.2 | 6.0 | 25 | [35] |
| Fomitopsis carnea | - | 21.9 | - | 30 | [14] |
| Corn husk | 47.95 | - | 6.0 | 25 | [8] |
| Oak acorn peel | 120.5 | - | 7.0 | 24 | [36] |
| Salvia miltiorrhiza | 100 | - | 7.0 | [12] | |
| Eucalyptus bark | 204.8 | - | 9.9 | 30 | [37] |
| Platanus leaf | 99.1 | - | 7.0 | 30 | [38] |
| Banana stalk | 243.9 | - | - | 30 | [39] |
| Dye | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| Qmax (mg/g) | b (L/mg) | R2 | K (L/g) | 1/n | R2 | |
| TT 1 | 49.3 ± 6.2 | 0.03 ± 0.01 | 0.905 | 6.92 ± 1.46 | 2.77 ± 0.36 | 0.972 |
| MB 1 | 160 ± 17 | 0.05 ± 0.02 | 0.950 | 25.4 ± 0.72 | 2.67 ± 0.05 | 0.999 |
| TT + MB 2 | 232 ± 29 | 0.013 ± 0.004 | 0.955 | 16.7 ± 1.9 | 2.29 ± 0.12 | 0.839 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pipíška, M.; Valica, M.; Partelová, D.; Horník, M.; Lesný, J.; Hostin, S. Removal of Synthetic Dyes by Dried Biomass of Freshwater Moss Vesicularia Dubyana: A Batch Biosorption Study. Environments 2018, 5, 10. https://doi.org/10.3390/environments5010010
Pipíška M, Valica M, Partelová D, Horník M, Lesný J, Hostin S. Removal of Synthetic Dyes by Dried Biomass of Freshwater Moss Vesicularia Dubyana: A Batch Biosorption Study. Environments. 2018; 5(1):10. https://doi.org/10.3390/environments5010010
Chicago/Turabian StylePipíška, Martin, Martin Valica, Denisa Partelová, Miroslav Horník, Juraj Lesný, and Stanislav Hostin. 2018. "Removal of Synthetic Dyes by Dried Biomass of Freshwater Moss Vesicularia Dubyana: A Batch Biosorption Study" Environments 5, no. 1: 10. https://doi.org/10.3390/environments5010010
APA StylePipíška, M., Valica, M., Partelová, D., Horník, M., Lesný, J., & Hostin, S. (2018). Removal of Synthetic Dyes by Dried Biomass of Freshwater Moss Vesicularia Dubyana: A Batch Biosorption Study. Environments, 5(1), 10. https://doi.org/10.3390/environments5010010





