Impact of Coal-Fired Power Plant Activities on the Ecological Status of River Ecosystems: Case Study of Sokolitsa River, Bulgaria
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
- Site So1: Sokolitsa River near Vladimirovo village, upstream of the discharges from the facilities of CFPP “Contour Global Maritsa East 3”, located within SWB BG3MA200R018 (reference site, not affected by CFPP activities);
- Site So2: Sokolitsa River near Obruchishte village, downstream of the discharges of the CFPP “Contour Global Maritsa East 3” facilities, located within SWB BG3MA200R017 (impacted site, affected by CFPP activities).
2.2. Data Collection and Analysis
2.3. Statistical Analyses
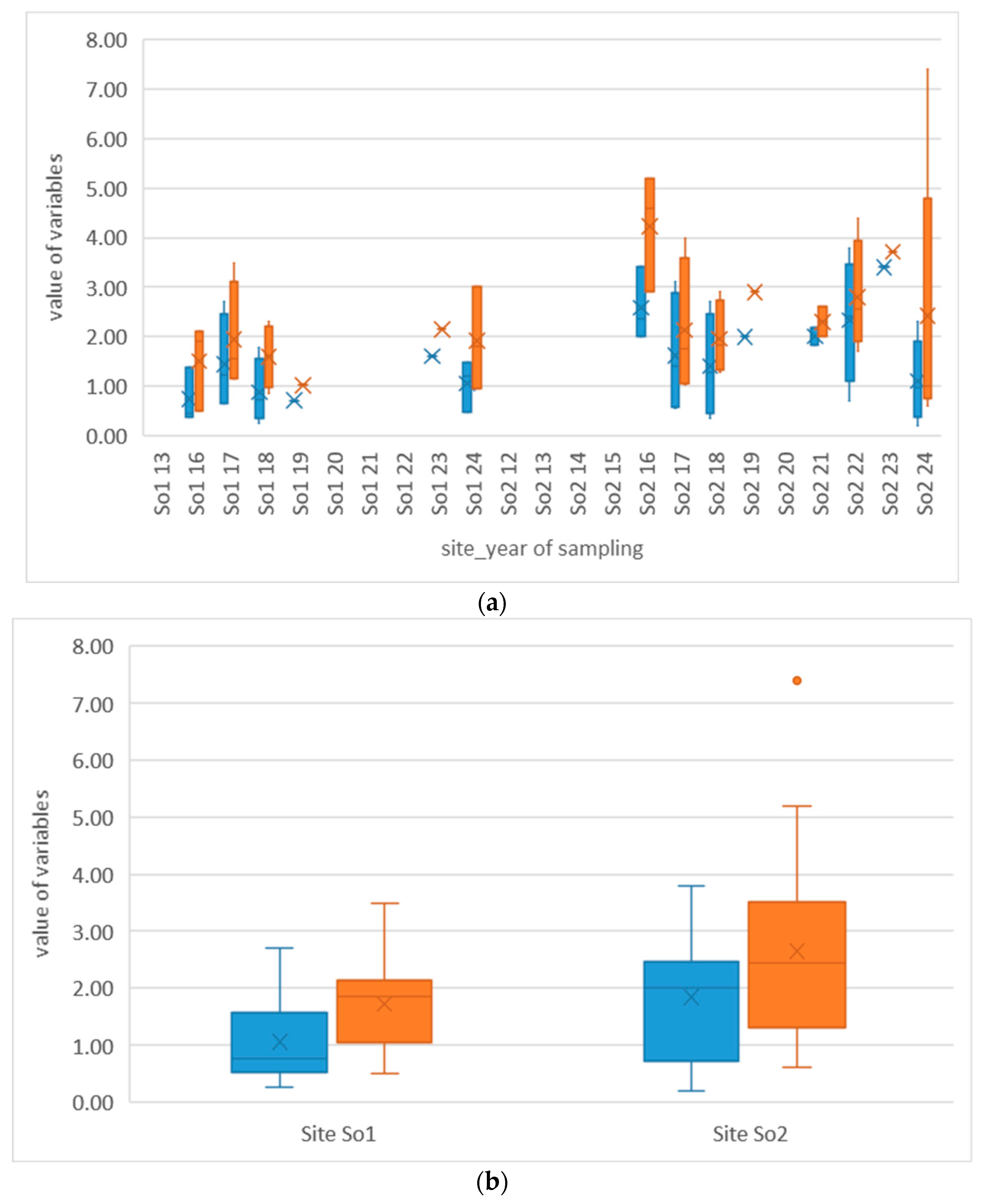
3. Results
3.1. Physicochemical Variables
3.2. Results for BQE Phytobenthos
3.3. Results for BQE Macrozoobenthos
4. Discussion
4.1. Environmental Variables
4.2. BQEs
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- DRAX. The Turbulent History of Coal. Available online: https://www.drax.com/power-generation/the-turbulent-history-of-coal/ (accessed on 23 March 2026).
- EBSCO; Bailey, E.; Morley, D.C. Coal-Burning Power Plants: Overview. 2024. Available online: https://www.ebsco.com/research-starters/power-and-energy/coal-burning-power-plants-overview#research-starter-title4 (accessed on 25 February 2026).
- Institute for Global Sustainability. Visualizing Energy. The History of Global Coal Production. Available online: https://visualizingenergy.org/the-history-of-global-coal-production/ (accessed on 25 February 2026).
- Brown, B.; Spiegel, S.J. Coal, climate justice, and the cultural politics of energy transition. Glob. Environ. Politics 2019, 19, 149–168. [Google Scholar] [CrossRef]
- Hendryx, M.; Zullig, K.J.; Luo, J. Impacts of coal use on health. Annu. Rev. Publ. Health 2020, 41, 397–415. [Google Scholar] [CrossRef]
- Finkelman, R.B.; Wolfe, A.; Hendryx, M.S. The future environmental and health impacts of coal. Energy Geosci. 2021, 2, 99–112. [Google Scholar] [CrossRef]
- Public Service Commission of Wisconsin. Environmental Impacts of Power Plants. Available online: https://psc.wi.gov/Documents/Brochures/Enviromental%20Impacts%20of%20PP.pdf (accessed on 25 February 2026).
- United States Environmental Protection Agency. Human Health & Environmental Impacts of the Electric Power Sector. Available online: https://www.epa.gov/power-sector/human-health-environmental-impacts-electric-power-sector (accessed on 25 February 2026).
- International Energy Agency (IEA). 21st Century Coal. Advanced Technology and Global Energy Solution; OECD Publishing: Paris, France; IEA: Paris, France, 2013; Available online: https://www.oecd.org/content/dam/oecd/en/publications/reports/2013/04/21st-century-coal_g17a23ae/5k3wb8f6tt5f-en.pdf (accessed on 25 February 2026).
- Hendryx, M.; Islam, M.S.; Dong, G.H.; Paul, G. Air pollution emissions 2008–2018 from Australian coal mining, Implications for public and occupational health. Int. J. Environ. Res. Public Health 2020, 17, 1570. [Google Scholar] [CrossRef]
- Yan, J.; Jia, B.; Liu, B.; Zhang, J. Molecular simulation of molecular diffusion properties of coal in Qicheng coal mine. Molecules 2023, 28, 6933. [Google Scholar] [CrossRef] [PubMed]
- Global Energy Monitor. Global Coal Plant Tracker 2026. Coal-Fired Power Stations by Country. Available online: https://globalenergymonitor.org/projects/global-coal-plant-tracker/summary-tables/ (accessed on 25 February 2026).
- Rosslowe, C.; Petrovich, B. European Electricity Review; EMBER: London, UK, 2025; Available online: https://ember-energy.org/latest-insights/european-electricity-review-2025/ (accessed on 25 February 2026).
- Energy and Water Regulatory Commission (EWRC). Annual Report to the European Commission. 2025. Available online: https://www.dker.bg/uploads/2025/ewrc-annual-rep-ec-jul2025-en.pdf (accessed on 23 March 2026).
- European Commission. The European Green Deal. Brussels 2019. COM, 640, Final, 4. Available online: https://eur-lex.europa.eu/resource.html?uri=cellar:b828d165-1c22-11ea-8c1f-01aa75ed71a1.0002.02/DOC_1&format=PDF (accessed on 25 February 2026).
- Council of Ministers of the Republic of Bulgaria. Updated Recovery and Resilience Plan 2023, p. 113. Available online: https://commission.europa.eu/business-economy-euro/economic-recovery/recovery-and-resilience-facility/country-pages/bulgarias-recovery-and-resilience-plan_en (accessed on 25 February 2026).
- European Environment Agency. Greenhouse Gas Emission Intensity of Electricity Generation in Europe 2023. Available online: https://www.eea.europa.eu/en/analysis/indicators/greenhouse-gas-emission-intensity-of-1 (accessed on 25 February 2026).
- Electric Power Research Institute (EPRI). Water and Sustainability (Volume 3): U.S. Water Use for Electricity Generation—The Next Half Century. EPRI Technical Report 2002. Available online: http://www.circleofblue.org/waternews/wp-content/uploads/2010/08/EPRI-Volume-3.pdf (accessed on 25 February 2026).
- Widiawaty, M.; Nurhanifah, N.; Ismail, A.; Dede, M. The impact of Cirebon coal-fired power plants on water quality in Mundu Bay, Cirebon Regency. Sustinere J. Environ. Sustain. 2020, 4, 189–204. [Google Scholar] [CrossRef]
- Ngamlana, Z.; Malherbe, W.; Gericke, G. Association of coal fired power plants with river water quality in South Africa. Limnologica 2024, 104, 126140. [Google Scholar] [CrossRef]
- Mardi, N.H.; Ean, L.W.; Malek, M.; Chua, K.H.; Ahmed, A.N. Water impact analysis due to coal-electricity generation using the life cycle assessment method: A case study in Malaysia. Water Sci. Technol. 2025, 91, 219–234. [Google Scholar] [CrossRef]
- Union of Concerned Scientists. Coal and Water Pollution. Available online: https://www.ucs.org/resources/coal-and-water-pollution (accessed on 25 February 2026).
- Saletnik, A.; Saletnik, B.; Puchalski, C. Coal as the world’s dominant energy source and its role in the energy transformation and regulations of European Green Deal. J. Environ. Manag. 2025, 392, 126815. [Google Scholar] [CrossRef]
- European Commission. LIFE21-GIC-BG-LIFE WatClima on LIFE Public Database. Bulgaria’s Water Strategies for Transition to a Climate Neutral and Resilient Society. Available online: https://webgate.ec.europa.eu/life/publicWebsite/project/LIFE21-GIC-BG-LIFE-WatClima-101074157/bulgarias-water-strategies-for-transition-to-a-climate-neutral-and-resilient-society (accessed on 23 March 2026).
- Ministry of Environment and Water. Water-Related Risks Arising from Climate Change Must Be Managed 2024. Available online: https://www.moew.government.bg/en/water-related-risks-from-climate-change-need-to-be-managed/ (accessed on 25 February 2026).
- De Castro-Català, N.; Dolédec, S.; Kalogianni, E.; Skoulikidis, N.T.; Paunovic, M.; Vasiljević, B.; Sabater, S.; Tornés, E.; Muñoz, I. Unravelling the effects of multiple stressors on diatom and macroinvertebrate communities in European river basins using structural and functional approaches. Sci. Total Environ. 2020, 742, 140543. [Google Scholar] [PubMed]
- Markert, N.; Guhl, B.; Feld, C.K. Water quality deterioration remains a major stressor for macroinvertebrate, diatom and fish communities in German rivers. Sci. Total Environ. 2023, 907, 167994. [Google Scholar] [CrossRef]
- Global Energy Monitor. Maritsa Iztok Complex. Available online: https://www.gem.wiki/Maritsa_Iztok_Complex (accessed on 25 February 2026).
- Rivers in Bulgaria. Available online: https://rekite.bg-schools.com/inforiv.php?id=71 (accessed on 25 February 2026).
- Register of Protected Areas and Protected Zones in Bulgaria. Available online: https://eea.government.bg/zpo/bg/area.jsp?NEM_Partition=2&categoryID=2&areaID=440 (accessed on 22 February 2026).
- Council Directive 92/43/EEC of 21 May 1992 on the Conservation of Natural Habitats and of Wild Fauna and Flora. Available online: https://data.europa.eu/eli/dir/1992/43/oj (accessed on 22 February 2026).
- River Basin Management Plan 2022–2027 of East Aegean River Basin District (RBMP of EARBD), 2025. Available online: https://earbd.bg/indexdetails.php?menu_id=966 (accessed on 25 February 2026).
- Ministry of Environment and Water of Bulgaria (MOEW). Ordinance No. N-4 of 14 September 2012 on the Characterization of Surface Waters. State Gazette, Issue 22, 5 March 2013 (Amended SG No. 79/2014, 85/2020, 13/2021). Available online: www.moew.government.bg (accessed on 23 March 2026).
- EN ISO 5667-3:2018; Water Quality—Sampling—Part 3: Preservation and Handling of Water Samples, Amended with ISO 5667-3:2024. International Organization for Standardization (ISO): Geneva, Switzerland, 2008. Available online: https://www.iso.org/standard/82273.html (accessed on 25 February 2026).
- BDS EN ISO 10523:2012; Water Quality—Determination of pH (ISO 10523:2008). Bulgarian Institute for Standardization: Sofia, Bulgaria, 2012. Available online: https://bds-bg.org/en/project/show/bds:proj:85125 (accessed on 25 February 2026).
- BDS 17.1.4.01:1977; Water Temperature. Bulgarian Institute for Standardization: Sofia, Bulgaria, 1977. Available online: https://bds-bg.org/en/project/show/bds:proj:21353 (accessed on 25 February 2026).
- BDS EN 27888:2000; Water Quality—Determination of Electrical Conductivity (ISO 7888:1985). Bulgarian Institute for Standardization: Sofia, Bulgaria, 2000. Available online: https://bds-bg.org/en/project/show/bds:proj:669 (accessed on 25 February 2026).
- BDS EN ISO 5814:2012; Water Quality—Determination of Dissolved Oxygen—Electrochemical Probe Method (ISO 5814:2012). Bulgarian Institute for Standardization: Sofia, Bulgaria, 2012. Available online: https://bds-bg.org/en/project/show/bds:proj:82365 (accessed on 25 February 2026).
- BDS EN ISO 6878:2005; Water Quality—Determination of Phosphorus—Ammonium Molybdate Spectrometric Method (ISO 6878:2004). Bulgarian Institute for Standardization: Sofia, Bulgaria, 2005. Available online: https://bds-bg.org/en/project/show/bds:proj:49392 (accessed on 25 February 2026).
- BDS ISO 7150-1:2002; Water Quality—Determination of Ammonium—Part 1: Manual Spectrometric Method. Bulgarian Institute for Standardization: Sofia, Bulgaria, 2002. Available online: https://bds-bg.org/en/project/show/bds:proj:29986 (accessed on 25 February 2026).
- BDS EN ISO 10304-1:2009; Water Quality—Determination of Dissolved Anions by Liquid Chromatography of Ions—Part 1: Determination of Bromide, Chloride, Fluoride, Nitrate, Nitrite, Phosphate and Sulfate ISO 10304-1:2007. Bulgarian Institute for Standardization: Sofia, Bulgaria, 2009. Available online: https://bds-bg.org/en/project/show/bds:proj:79923 (accessed on 25 February 2026).
- BDS EN ISO 20236:2021; Water Quality—Determination of Total Organic Carbon (TOC), Dissolved Organic Carbon (DOC), Total Bound Nitrogen (TNb) and Dissolved Bound Nitrogen (DNb) After High Temperature Catalytic Oxidative Combustion (ISO 20236:2018), Revised by BDS EN ISO 20236:2025 Water Quality—Determination of total Organic Carbon (TOC), Dissolved Organic Carbon (DOC), Total Bound Nitrogen (TNb) and Dissolved Bound Nitrogen (DNb) After High Temperature Catalytic Oxidative Combustion (ISO 20236:2024). Bulgarian Institute for Standardization: Sofia, Bulgaria, 2025. Available online: https://bds-bg.org/en/project/show/bds:proj:122388 (accessed on 23 February 2026).
- BDS EN ISO 14911:2002; Water Quality—Determination of Dissolved Li+, Na+, NH4+, K+, Mn2+, Ca2+, Mg2+, Sr2+ and Ba2+ Using Ion Chromatography—Method for Water and Waste Water (ISO 14911:1998). Bulgarian Institute for Standardization: Sofia, Bulgaria, 2002. Available online: https://bds-bg.org/en/project/show/bds:proj:30707 (accessed on 23 February 2026).
- BDS ISO 6059:2002; Water Quality—Determination of the Sum of Calcium and Magnesium—EDTA Titrimetric Method. Bulgarian Institute for Standardization: Sofia, Bulgaria, 2002. Available online: https://bds-bg.org/en/project/show/bds:proj:30123 (accessed on 23 February 2026).
- EN 13946:2014; Water Quality—Guidance Standard for the Routine Sampling and Preparation of Benthic Diatoms from Rivers and Lakes (amended with EN 13946:2024). European Committee for Standardization: Brussels, Belgium, 2014.
- EN 14407:2014; Water Quality—Guidance Standard for the Identification and Enumeration of Benthic Diatom Samples from Rivers and Lakes. European Committee for Standardization: Brussels, Belgium, 2014.
- CEMAGREF. Etude des Methods Biologiques D’appréciation Quantitative de la Qualité des Eaux. Rapport QE Lyon Bassin Rhône-Méditerranée-Corse; CEMAGREF: Antony, France, 1982. [Google Scholar]
- Lecointe, C.; Coste, M.; Prygiel, J. Omnidia. Diatom index software including diatom database with taxonomic names, references and codes of 11645 diatom taxa. Hydrobiologia 2003, 269, 509–513. [Google Scholar]
- Mertens, A.; van der Wal, J.; Verweij, G.; Pex, B.; van Dulmen, A.; van Dam, H. A revised list of diatom ecological indicator values in The Netherlands. Ecol. Indic. 2025, 172, 113219. [Google Scholar] [CrossRef]
- Cheshmedjiev, S.; Soufi, R.; Vidinova, Y.; Tyufekchieva, V.; Yaneva, I.; Uzunov, Y.; Varadinova, E. Multi-habitat sampling method for benthic macroinvertebrate communities in different river types in Bulgaria. Water Res. Manag. 2011, 1, 55–58. [Google Scholar]
- BDS EN ISO 10870:2012; Water Quality—Guidelines for the Selection of Sampling Methods and Devices for Benthic Macroinvertebrates in Fresh Waters. Bulgarian Institute for Standardization: Sofia, Bulgaria, 2012. Available online: https://bds-bg.org/en/project/show/bds:proj:81883 (accessed on 23 February 2026).
- BDS EN 16150:2012; Water Quality—Guidance on Pro-Rata Multi-Habitat Sampling of Benthic Macro-Invertebrates from Wadeable Rivers. Bulgarian Institute for Standardization: Sofia, Bulgaria, 2012. Available online: https://bds-bg.org/en/project/show/bds:proj:82773 (accessed on 23 February 2026).
- Nagel, P. Bildbestimmungsschlüssel der Saprobien. Macrozoobenthon; Gustav Fischer Verlag: Stuttgard, Germany, 1989; p. 180. [Google Scholar]
- Cham, S. Field Guide to the Larvae and Exuviae of British Dragonflies; Dragonflies (Anisoptera); The British Dragonfly Society: Telford, UK, 2007; Volume 1, p. 75. [Google Scholar]
- Cham, S. Field Guide to the Larvae and Exuviae of British Dragonflies; Damselflies (Zygoptera); The British Dragonfly Society: Telford, UK, 2007; Volume 2, p. 75. [Google Scholar]
- Schmedtje, U.; Kohmann, F. Bestimmungsschlüssel Für Die Saprobier-DIN-Arten (Makroorganismen); Informationsberichte des Bayerischen Landesamtes für Wasserwirtschaft; Bayern, Landesamt für Wasserwirtschaft: Augsburg, Germany, 1992; p. 274. [Google Scholar]
- Rozkošny, R. Řád Dvoukřídlí—Diptera. In Klíč Vodních Larev Hmyzu; Jeżek, J., Knoz, J., Kramář, J., Krampl, F., Kubiċek, F., Lellák, J., Minář, J., Pokorný, P., Raušer, J., Rozkošny, R., et al., Eds.; Československá Akademie Vĕd.: Praha, Czech Republic, 1980; pp. 226–457. [Google Scholar]
- Waringer, J.; Graf, W. Key and bibliography of the genera of European Trichoptera larvae. Zootaxa 2013, 3640, 101–151. [Google Scholar] [CrossRef]
- Ellliott, J.M.; Humpesch, U.H. Mayfly Larvae (Ephemeroptera) of Britain and Ireland: Keys and Review of Their Ecology; Freshwater Biological Association: Ambleside, UK, 2010; p. 152. [Google Scholar]
- Flanagan, P.J.; Toner, P.F. The National Survey of Irish Rivers: A Report on Water Quality; An Foras Forbartha: Dublin, Ireland, 1972; p. 213. [Google Scholar]
- Clabby, K.J. The National Survey of Irish Rivers: River Quality Investigations—Biological Results of the 1980 & 1981 Investigations: Summary Report; Water Resources Division 1982; An Foras Forbartha: Dublin, Ireland, 1982. [Google Scholar]
- Clabby, K.J.; Bowman, J.J. Report of Irish participants. In 3rd Technical Seminar on Biological Water Assessment Method; Ghetti, P.F., Ed.; Commission of the European Communities: Parma, Italy, 1979; Volume 1. [Google Scholar]
- Shannon, C.E.; Weaver, W. The Mathematical Theory of Communication; University of Illinois Press: Champaign, IL, USA, 1949. [Google Scholar]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological Statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]
- Legendre, P.; Anderson, M.J. Distance-based redundancy analysis: Testing multispecies responses in multifactorial ecological experiments. Ecol. Monogr. 1999, 69, 1–24. [Google Scholar] [PubMed]
- Clarke, K.R.; Gorley, R.N. Primer v6: User Manual/Tutorial; PRIMER-E Ltd.: Auckland, New Zealand, 2006. [Google Scholar]
- Standard Methods for the Examination of Water and Wastewater. Standard Methods Committee of the American Public Health Association, American Water Works Association, and Water Environment Federation. part 1000 analytical and data quality systems. In Standard Methods for the Examination of Water and Wastewater; Lipps, W.C., Baxter, T.E., Braun-Howland, E., Eds.; APHA Press: Washington, DC, USA, 2023; Available online: https://www.standardmethods.org/action/showTopic?taxonomyUri=part&topicCode=part1000 (accessed on 23 March 2026).
- Punde, S.; Kulkarni, M. A Critical Review on Physicochemical Parameters in Water Quality Management: Key Indicators for Sustainability and Pollution Mitigation. Int. J. Creat. Res. Thoughts 2025, 13, 380–393. Available online: https://www.researchgate.net/publication/394412530_A_Critical_Review_On_Physicochemical_Parameters_In_Water_Qualiy_Management_Key_Indicators_For_Sustainability_And_Pollution_Mitigation (accessed on 23 March 2026).
- Khandagale, D.K.; Pradhan, V. Importance of water pH and hardness on fish biological processes. Int. J. Sci. Dev. Res. 2021, 6, 8–11. Available online: https://www.ijsdr.org/papers/IJSDR2111003.pdf (accessed on 23 March 2026).
- Wetzel, R.G. Limnology: Lake and River Ecosystems; Gulf Professional Publishing: Houston, TX, USA, 2001. [Google Scholar]
- Zak, D.; Hupfer, M.; Cabezas, A.; Jurasinski, G.; Audet, J.; Kleeberg, A.; McInnes, R.; Kristiansen, S.M.; Petersen, R.J.; Liu, H.; et al. Sulphate in freshwater ecosystems: A review of sources, biogeochemical cycles, ecotoxicological effects and bioremediation. Earth-Sci. Rev. 2021, 212, 103446. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, Q. Research Advances in Identifying Sulfate Contamination Sources of Water Environment by Using Stable Isotopes. Int. J. Environ. Res. Public Health 2019, 16, 1914. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Elphick, J.; Davies, M.; Gilron, G.; Canaria, E.; Lo, B.; Bailey, H. An aquatic toxicological evaluation of sulfate: The case for considering hardness as a modifying factor in setting water quality guidelines. Environ. Toxicol. Chem. 2011, 30, 247–253. [Google Scholar] [CrossRef]
- Davies, T.D.; Hall, K.J. Importance of calcium in modifying the acute toxicity of sodium sulfate to Hyella 21zteca and Daphnia magna. Environ. Toxicol. Chem. 2007, 26, 1243. [Google Scholar] [CrossRef] [PubMed]
- Griffith, M.B. Toxicological perspective on the osmoregulation and ionoregulation physiology of major ions by freshwater animals: Teleost fish, crustacea, aquatic insects, and mollusca. Environ. Toxicol. Chem. 2017, 36, 576–600. [Google Scholar] [CrossRef] [PubMed]
- WISE Freshwater Information System. List of the River Basin-Specific Pollutants Identified as Locally Important and Used as a Supporting Quality Component for the Assessment of Ecological Status or Potential of Surface Water Bodies, for the 2nd and 3rd Cycle of the Water Framework Directive (WFD)—River Basin Management Plan (RBMP). Dashboard Modified 20 November 2024. Available online: https://water.europa.eu/freshwater/resources/metadata/wfd-dashboards/surface-water-bodies-river-basin-specific-pollutants-2nd-3rd-rbmp-table (accessed on 23 February 2026).
- Karjalainen, J.; Hu, X.; Mäkinen, M.; Karjalainen, A.; Järvistö, J.; Järvenpää, K.; Sepponen, M.; Leppänen, M.T. Sulfate sensitivity of aquatic organism in soft freshwaters explored by toxicity tests and species sensitivity distribution. Ecotoxicol. Environ. Saf. 2023, 258, 114984. [Google Scholar] [CrossRef]
- European Commission. Technical Guidance for Deriving Environmental Quality Standards; Guidance Document No: 27; European Commission: Brussels, Belgium, 2018.
- U.S. Environmental Protection Agency (EPA). Proposed Rule, Effluent Limitations Guidelines and Standards for the Steam Electric Power Generating Point Source Category; U.S. Environmental Protection Agency (EPA): Washington, DC, USA, 2013.
- Brigden, K.; Labunska, I.; Santillo, D. Metals, Metalloids and Organic Contaminants in Wastewater, River Water, Sediment and Ash Samples Associated with the Bobov Dol Coal Fired Power Plant, Golemo Selo, Bulgaria; Greenpeace Research Laboratories Technical Report; The Greenpeace Research Laboratories: Exeter, UK, 2019; Available online: https://www.greenpeace.to/greenpeace/?p=3225 (accessed on 23 March 2026).
- Alewell, C.; Ringeval, B.; Ballabio, C.; Robinson, D.A.; Panagos, P.; Borrelli, P. Global phosphorus shortage will be aggravated by soil erosion. Nat. Commun. 2020, 11, 4546. Available online: www.nature.com/naturecommunications (accessed on 23 March 2026). [CrossRef]
- Executive Environmental Agency 2024. National Report on the State and Protection of the Environment in the Republic of Bulgaria for 2023. Available online: https://eea.government.bg/bg/soer/2023/soer-bg-2023.pdf (accessed on 20 February 2026).
- Schulz, G.; van Beusekom, J.E.E.; Jacob, J.; Bold, S.; Schöl, A.; Ankele, M.; Sanders, T.; Dähnke, K. Low discharge intensifies nitrogen retention in rivers—A case study in the Elbe River. Sci. Total Environ. 2023, 904, 166740. [Google Scholar] [CrossRef]
- Harvey, J.W.; Schmadel, N.M. The River Corridor’s Evolving Connectivity of Lotic and Lentic Waters. Front. Water 2021, 2, 580727. [Google Scholar] [CrossRef]
- Environmental Protection Agency USA (EPA USA). Sources and Solutions: Fossil Fuels. Available online: https://www.epa.gov/nutrientpollution/sources-and-solutions-fossil-fuels (accessed on 19 December 2025).
- European Environment Agency (EEA). European Zero Pollution Dashboards. Emissions of Major Pollutants into the Air by Extractive Industries (Indicator). Available online: https://www.eea.europa.eu/en/european-zero-pollution-dashboards/indicators/emissions-of-major-pollutants-into-the-air-by-extractive-industries-indicator (accessed on 19 December 2025).
- US Department of Energy. Kemper Country Integrated Gasification Combined-Cycle (IGCC) Project, Draft Environmental Impact Statement (EIS) (DOE/EIS-0409D); Kemper County, Mississippi; US Department of Energy: Washington, DC, USA, 2009.
- Russell, M.C.; Belle, J.H.; Liu, Y. The impact of three recent coal-fired power plant closings on Pittsburgh air quality: A natural experiment. J. Air Waste Manag. Assoc. 2017, 67, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Crawford, J.; Cohen, D.D.; Atanacio, A. The impact of closure of coal-fired power stations on aerosol concentrations in the Sydney Basin Atmos. Pollut. Res. 2018, 9, 1167–1176. [Google Scholar] [CrossRef]
- Schröder, M.; King, R.S.; Pannard, A. Effects of salinity gradients on benthic invertebrate and diatom communities in a German lowland river. Ecol. Indic. 2015, 57, 462–474. [Google Scholar] [CrossRef]
- Potapova, M.; Charles, D.F. Distribution of benthic diatoms in U.S. rivers in relation to conductivity and ionic composition. Freshw. Biol. 2003, 48, 1311–1328. [Google Scholar] [CrossRef]
- Smucker, N.J.; Vis, M.L. Use of diatoms to assess agricultural and coal mining impacts on streams and a multiassemblage case study. J. N. Am. Benthol. Soc. 2009, 28, 659–675. [Google Scholar] [CrossRef]
- Bąk, M.; Halabowski, D.; Kryk, A.; Lewin, I.; Sowa, A. Mining salinisation of rivers: Its impact on diatom (Bacillariophyta) assemblages. Fottea 2020, 20, 1–16. [Google Scholar] [CrossRef]
- Stenger-Kovács, C.; Béres, V.B.; Buczkó, K.; Tapolczai, K.; Padisák, J.; Selmeczy, G.B.; Lengyel, E. Diatom community response to inland water salinization: A review. Hydrobiologia 2023, 850, 2345–2364. [Google Scholar] [CrossRef]
- Berger, E.; Frör, O.; Schäfer, R.B. Salinity impacts on river ecosystem processes: A critical mini-review. Philos. Trans. R. Soc. B Biol. Sci. 2019, 374, 20180010. [Google Scholar] [CrossRef]
- Rico-Sánchez, A.E.; Rodríguez-Romero, A.J.; Sedeño-Díaz, J.E.; López-López, E.; Sundermann, A. Aquatic macroinvertebrate assemblages in rivers influenced by mining activities. Sci. Rep. 2022, 12, 3209. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Guo, F.; Zhang, Y.; Yang, Z.; Ma, S. Effects of secondary salinisation on macroinvertebrate functional traits in surface mining-contaminated streams, and recovery potential. Sci. Total Environ. 2018, 640–641, 1088–1097. [Google Scholar] [CrossRef] [PubMed]
- Kuchapski, K.A.; Rasmussen, J.B. Surface coal mining influences on macroinvertebrate assemblages in streams of the Canadian Rocky Mountains. Environ. Toxicol. Chem. 2015, 34, 2138–2148. [Google Scholar] [CrossRef]
- Halabowski, D.; Lewin, I.; Buczyński, P.; Krodkiewska, M.; Płaska, W.; Sowa, A.; Buczyńska, E. Impact of the Discharge of Salinised Coal Mine Waters on the Structure of the Macroinvertebrate Communities in an Urban River (Central Europe). Water Air Soil Pollut. 2020, 231, 5. [Google Scholar] [CrossRef]
- Aal-e, A.; Sloane, D.R.; Strezov, V. Assessment of Impacts of Coal Mining in the Region of Sydney, Australia on the Aquatic Environment Using Macroinvertebrates and Chlorophyll as Indicators. Int. J. Environ. Res. Public Health 2018, 15, 1556. [Google Scholar] [CrossRef]
- Durance, I.; Ormerod, S.J. Climate change effects on upland stream macroinvertebrates over a 25-year period. Glob. Change Biol. 2007, 13, 942–957. [Google Scholar] [CrossRef]
- Salmah, R.C.; Al-shami, S.A.; Hassan, A.A.; Madrus, M.R.; Huda, A.N. Distribution of detritivores in tropical forest streams of Peninsular Malaysia: Role of temperature, canopy cover and altitude variability. Int. J. Biometeorol. 2014, 58, 679–690. [Google Scholar] [CrossRef] [PubMed]
- West, A.; Penk, M.R.; Larney, R.; Piggott, J.J. Response of macroinvertebrates to industrial warm discharges: The River Shannon case study (Ireland). Inland Waters 2021, 11, 381–395. [Google Scholar] [CrossRef]
- Williams, W.D.; Boulton, A.J.; Taaffe, R.G. Salinity as a determinant of salt lake fauna: A question of scale. Hydrobiologia 1990, 197, 257–266. [Google Scholar] [CrossRef]
- Bäthe, J.; Coring, E. Biological effects of anthropogenic salt—Load on the aquatic fauna: A synthesis of 17 years of biological survey on the rivers Werra and Weser. Limnologica 2011, 41, 125–133. [Google Scholar] [CrossRef]
- Scheibler, E.E.; Ciocco, N.F. Distribution of macroinvertebrate assemblages along a saline wetland in harsh environmental conditions from Central-West Argentina. Limnologica 2011, 41, 37–47. [Google Scholar] [CrossRef]
- Kang, S.R.; King, S.L. Influence of salinity and prey presence on the survival of aquatic macroinvertebrates of a freshwater marsh. Aquat. Ecol. 2012, 46, 411–420. [Google Scholar] [CrossRef]
- Arle, J.; Wagner, F. Effects of anthropogenic salinisation on the ecological status of macroinvertebrate assemblages in the Werra River (Thuringia, Germany). Hydrobiologia 2013, 701, 129–148. [Google Scholar] [CrossRef]
- Pond, G.J.; Passmore, M.E.; Borsuk, F.A.; Reynolds, L.; Rose, C.J. Downstream effects of mountaintop coal mining: Comparing biological conditions using family- and genus-level macroinvertebrate bioassessment tools. J. N. Am. Benthol. Soc. 2008, 27, 717–737. [Google Scholar] [CrossRef]
- Herbst, D.B. Salinity controls on trophic interactions among invertebrates and algae of solar evaporation ponds in the Majove Desert and ralation to shorebird foraging and selenium risk. Wetlands 2006, 26, 475–485. [Google Scholar] [CrossRef]
- Sowa, A.; Krodkiewska, M.; Halabowski, D. How Does Mining Salinisation Gradient Affect the Structure and Functioning of Macroinvertebrate Communities? Water Air Soil Pollut. 2020, 231, 453. [Google Scholar] [CrossRef]
- Piscart, C.; Moreteau, J.C.; Beisel, J.N. Biodiversity and structure of macroinvertebrate communities along a small permanent salinity gradient (Meurthe River, France). Hydrobiologia 2005, 551, 227–236. [Google Scholar] [CrossRef]
- Piscart, C.; Moretea, J.C.; Beisel, J.N. Monitoring changes in freshwater macroinvertebrate communities along a salinity gradient using artificial substrates. Environ. Monit. Assess. 2006, 116, 529–542. [Google Scholar] [CrossRef] [PubMed]
- Kefford, B.J.; Buchwalter, D.; Cañedo-Argüelles, M.; Davis, J.; Duncan, R.P.; Hoffmann, A.; Thompson, R. Salinized rivers: Degraded systems or new habitats for salt-tolerant faunas? Biol. Lett. 2016, 12, 20151072. [Google Scholar] [CrossRef]
- Savic, A.; Dmitrovic, D.; Pesic, V. Ephemeroptera, Plecoptera, and Trichoptera assemblages of karst springs in relation to some environmental factors: A case study in central Bosnia and Herzegovina. Turk. J. Zool. 2017, 41, 119–129. [Google Scholar] [CrossRef]
- Graça, M.A.S.; Pinto, P.; Cortes, R.; Coimbra, N.; Oliveira, S.; Morais, M.; Carvalho, M.J.; Malo, J. Factors Affecting Macroinvertebrate Richness and Diversity in Portuguese Streams: A Two-Scale Analysis. Internat. Rev. Hydrobiol. 2004, 89, 151–164. [Google Scholar] [CrossRef]
- Croijmans, L.; De Jong, J.F.; Prins, H.H.T. Oxygen is a better predictor of macroinvertebrate richness than temperature—A systematic review. Environ. Res. Lett. 2021, 16, 023002. [Google Scholar] [CrossRef]
- Kefford, B.J.; Marchant, R.; Schäfer, R.B.; Metzeling, L.; Dunlop, J.E. The definition of species richness used by species sensitivity distributions approximates observed effects of salinity on stream macroinvertebrates. Environ. Pollut. 2011, 159, 302–310. [Google Scholar] [CrossRef]
- Cañedo-Argüelles, M.; Kefford, B.; Piscart, C.; Prat, N.; Schäfer, R.B.; Schulz, C.J. Salinisation of rivers: An urgent ecological issue. Environ. Pollut. 2013, 173, 157–167. [Google Scholar] [CrossRef]
- Bray, J.P.; Reich, J.; Nichols, S.J.; Kon Kam King, G.; Mac Nally, R.; Thompson, R.; O’Reilly-Nugent, A.; Kefford, B.J. Biological interactions mediate context and species-specific sensitivities to salinity. Philos. Trans. R. Soc. B Biol. Sci. 2018, 374, 20180020. [Google Scholar] [CrossRef] [PubMed]










| № | Name of the Studied Site | Coordinates | Code and Name of the Water Body | Environmental Variables and BQEs |
|---|---|---|---|---|
| 1 | So1: Sokolitsa River near Vladimirovo village, upstream of discharges from CFPP Contour Global Maritsa East 3 facilities and the landfill “Embankment Mednikarovo” | 26°8′11.76″ E, 42°7′38.64″ N | BG3MA200R018, Sokolitsa River upper stream | t° C, pH, dissolved oxygen (mg/L), conductivity (µS/cm), ammonium nitrogen (mg/L), nitrite nitrogen (mg/L), nitrate nitrogen (mg/L), total nitrogen (mg/L), orthophosphates as phosphorus (mg/L), total phosphorus (mg/L), calcium carbonate hardness (mg/L), calcium (mg/L), sulfates (mg/L), BQE phytobenthos, BQE macrozoobenthos. |
| 2 | So2: Sokolitsa River near Obruchishte village, downstream of discharges from CFPP Contour Global Maritsa East 3 facilities and the landfill “Embankment Mednikarovo” | 25°55′8.4″ E, 42°8′5.64″ N | BG3MA200R017, Sokolitsa River middle reaches to the Rozov Kladenets Dam |
| Variable | Units | Limit of Quantification (LoQ) | Measurement/Analytical Standards |
|---|---|---|---|
| pH | 0.02 | BDS EN ISO 10523:2012 [35] | |
| Water temperature | °C | BDS 17.1.4.01:1977 [36] | |
| Conductivity | µS/cm | 1.3 | BDS EN 27888:2000 [37] |
| Dissolved oxygen | mg/L | 0.3 | BDS EN ISO 5814:2012 [38] |
| Total phosphorus | mg/L | 0.008 | BDS EN ISO 6878:2005 [39] |
| Ammonium nitrogen | mg/L | 0.01 | BDS ISO 7150-1:2002[40] |
| Nitrite nitrogen | mg/L | 0.015 | BDS EN ISO 10304-1:2009 [41] |
| Nitrate nitrogen | mg/L | 0.02 | BDS EN ISO 10304-1:2009 [41] |
| Total nitrogen | mg/L | 0.2 | BDS EN ISO 20236:2021, point. 5.3 (amended with BDS EN ISO 20236:2025) [42] |
| Orthophosphates (as phosphorus) | mg/L | 0.006 | BDS EN ISO 6878:2005 [39] |
| Sulfates | mg/L | 1 | BDS EN ISO 10304-1:2009 [41] |
| Calcium | mg/L | 1 | BDS EN ISO 14911:2002 [43] |
| Calcium carbonate hardness | mg CaCO3/L | 20 | BDS ISO 6059:2002 [44] |
| Monitoring Site | - | pH | Conductivity | Dissolved Oxygen | Ammonium Nitrogen N-NH4 | Nitrite Nitrogen N-NO2 | Nitrate Nitrogen N-NO3 | Total Nitrogen—N-tot | Orthophosphates (as P)—PO4-P | Total Phosphorus (as P)—P-tot |
|---|---|---|---|---|---|---|---|---|---|---|
| - | - | µS/cm | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | |
| Monitoring site So1 | min for the period 2013–2022 | 7 | 160 | 4.2 | 0.005 * | 0.003 * | 0.26 | 0.5 * | 0.0165 * | 0.097 |
| max for the period 2013–2022 | 8.4 | 1354 | 14.8 | 0.46 | 0.057 | 2.7 | 3.5 | 0.05 | 0.21 | |
| mean for the period 2013–2022 | 7.79 | 637 | 7.84 | 0.1 | 0.0176 | 1.06 | 1.72 | 0.06 | 0.092 | |
| measured in 2023 | 7.74 | 237 | 5.79 | 0.13 | 0.053 | 1.6 | 2.15 | 0.0276 | 0.0417 | |
| measured in 2024 | 7.51 | 824 | 6.03 | 0.06 | 0.006 | 1.2 | 1.8 | 0.068 | 0.1 | |
| Monitoring site So2 | min for the period 2013–2022 | 6.8 | 297 | 2.2 | 0.005 * | 0.005 | 0.204 | 0.6 | 0.003 * | 0.004 * |
| max for the period 2013–2022 | 8.6 | 4610 | 15.3 | 0.81 | 0.52 | 3.8 | 7.4 | 0.112 | 0.24 | |
| mean for the period 2013–2022 | 7.8 | 2282 | 8.47 | 0.21 | 0.095 | 1.84 | 2.65 | 0.0324 | 0.0725 | |
| measured in 2023 | 7.26 | 297 | 7.93 | 0.2 | 0.128 | 3.4 | 3.72 | 0.0947 | 0.103 | |
| measured in 2024 | 7.61 | 1636 | 7.4 | 0.005 | 0.005 | 1.52 | 2.21 | 0.091 | 0.14 |
| Monitoring Site | So1 2019 | So1 2023 | So1 2024 | So2 2022 | So2 2023 | So2 2024 |
|---|---|---|---|---|---|---|
| IPS/20 | 12.8 | 12.6 | 7.9 | 10.8 | 9.7 | 3.9 |
| nEQR | 0.64 | 0.63 | 0.395 | 0.54 | 0.485 | 0.195 |
| Ecological status (R13): | Moderate | Moderate | Poor | Moderate | Moderate | Bad |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Mitseva, V.; Isheva, T.; Ihtimanska, M.; Varadinova, E. Impact of Coal-Fired Power Plant Activities on the Ecological Status of River Ecosystems: Case Study of Sokolitsa River, Bulgaria. Environments 2026, 13, 191. https://doi.org/10.3390/environments13040191
Mitseva V, Isheva T, Ihtimanska M, Varadinova E. Impact of Coal-Fired Power Plant Activities on the Ecological Status of River Ecosystems: Case Study of Sokolitsa River, Bulgaria. Environments. 2026; 13(4):191. https://doi.org/10.3390/environments13040191
Chicago/Turabian StyleMitseva, Vanina, Tsvetelina Isheva, Mila Ihtimanska, and Emilia Varadinova. 2026. "Impact of Coal-Fired Power Plant Activities on the Ecological Status of River Ecosystems: Case Study of Sokolitsa River, Bulgaria" Environments 13, no. 4: 191. https://doi.org/10.3390/environments13040191
APA StyleMitseva, V., Isheva, T., Ihtimanska, M., & Varadinova, E. (2026). Impact of Coal-Fired Power Plant Activities on the Ecological Status of River Ecosystems: Case Study of Sokolitsa River, Bulgaria. Environments, 13(4), 191. https://doi.org/10.3390/environments13040191







