The Use of Biosorbents in Water Treatment
Abstract
1. Introduction
1.1. Types of Pollutants
1.2. The Purpose of the Review
2. Biosorbents
2.1. Classification of Biosorbents
2.1.1. Natural Biosorbents
Biomass-Derived Biosorbents
Microbial Biosorbents
2.1.2. Modified/Synthetic Biosorbents
2.2. Characteristics of Biosorbents
2.2.1. Physical Characteristics
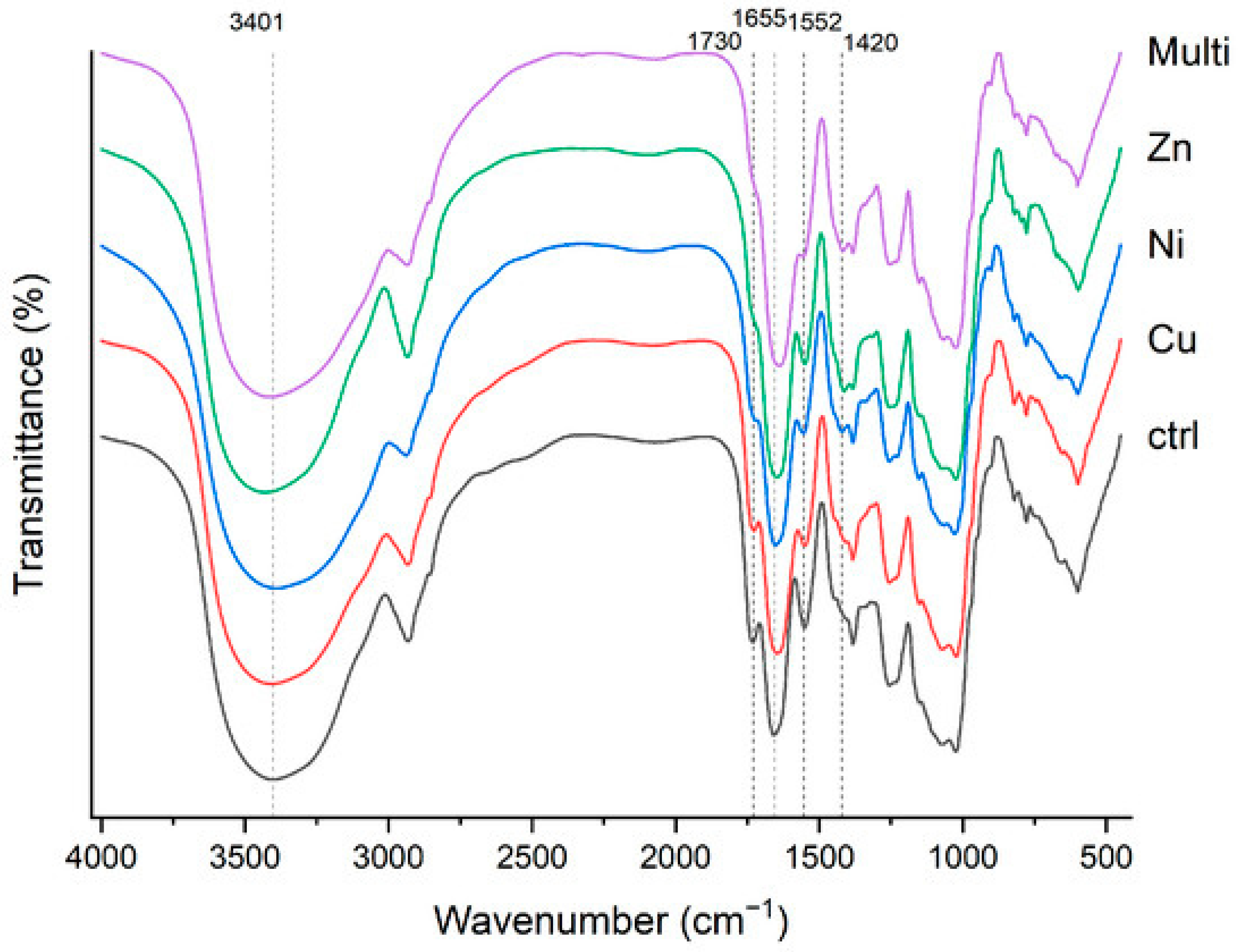
2.2.2. Chemical Characteristics
2.3. Synthesis of Biosorbents
2.3.1. Physical
2.3.2. Chemical
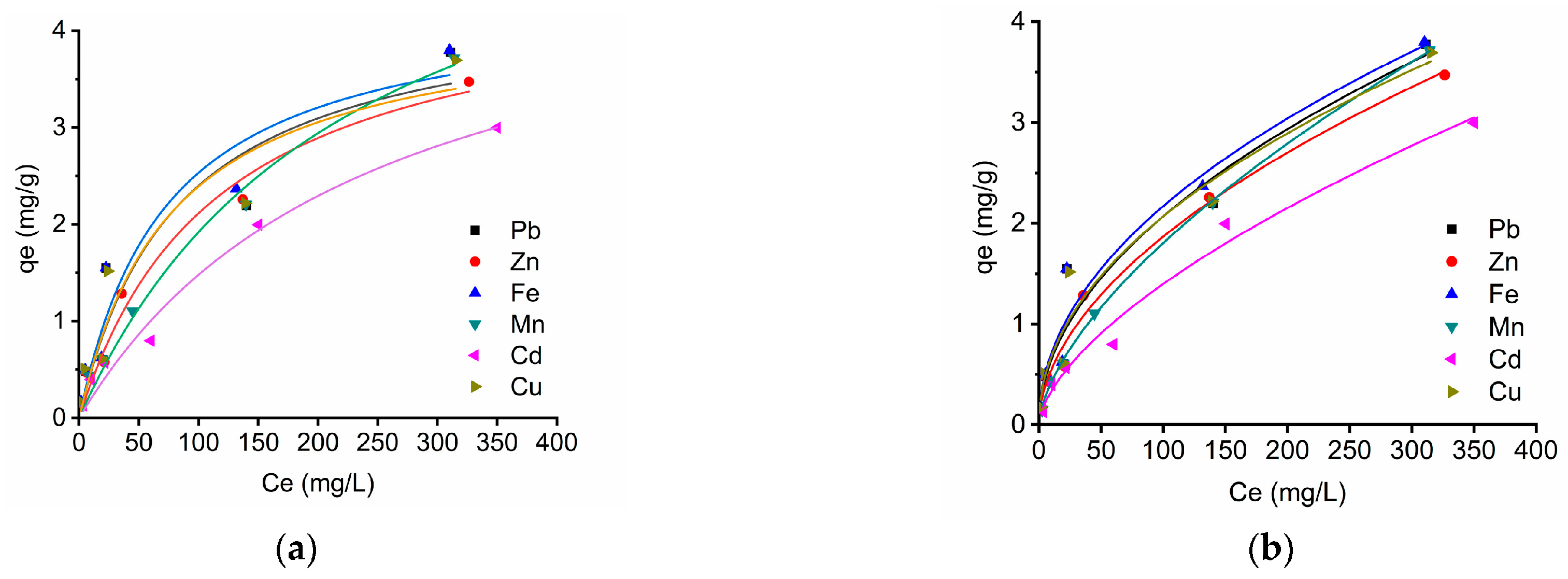
2.4. Equilibrium Isotherms: Langmuir and Freundlich Models
2.5. Biosorption Kinetics
3. Biosorption
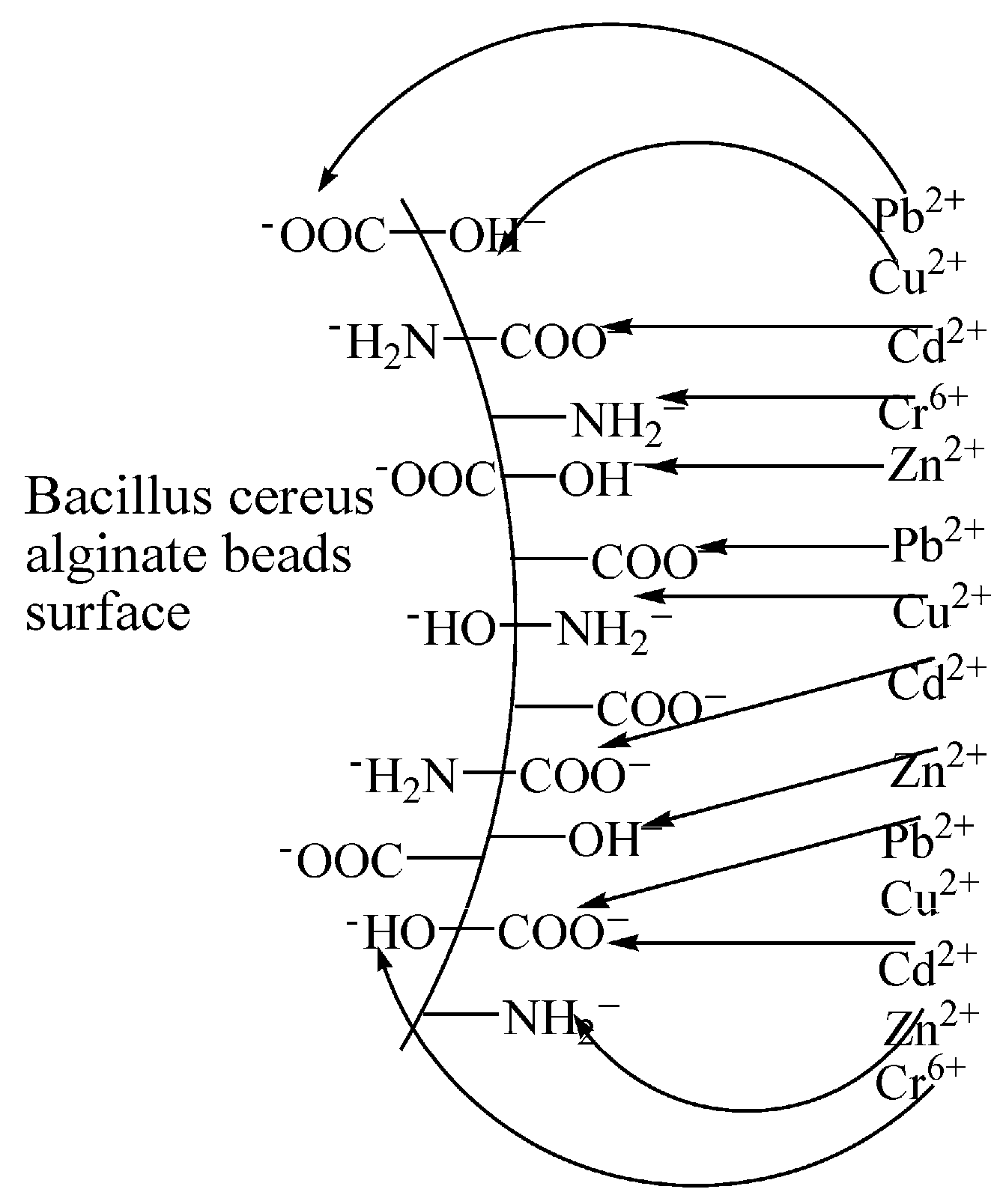
3.1. Mechanisms of Biosorption
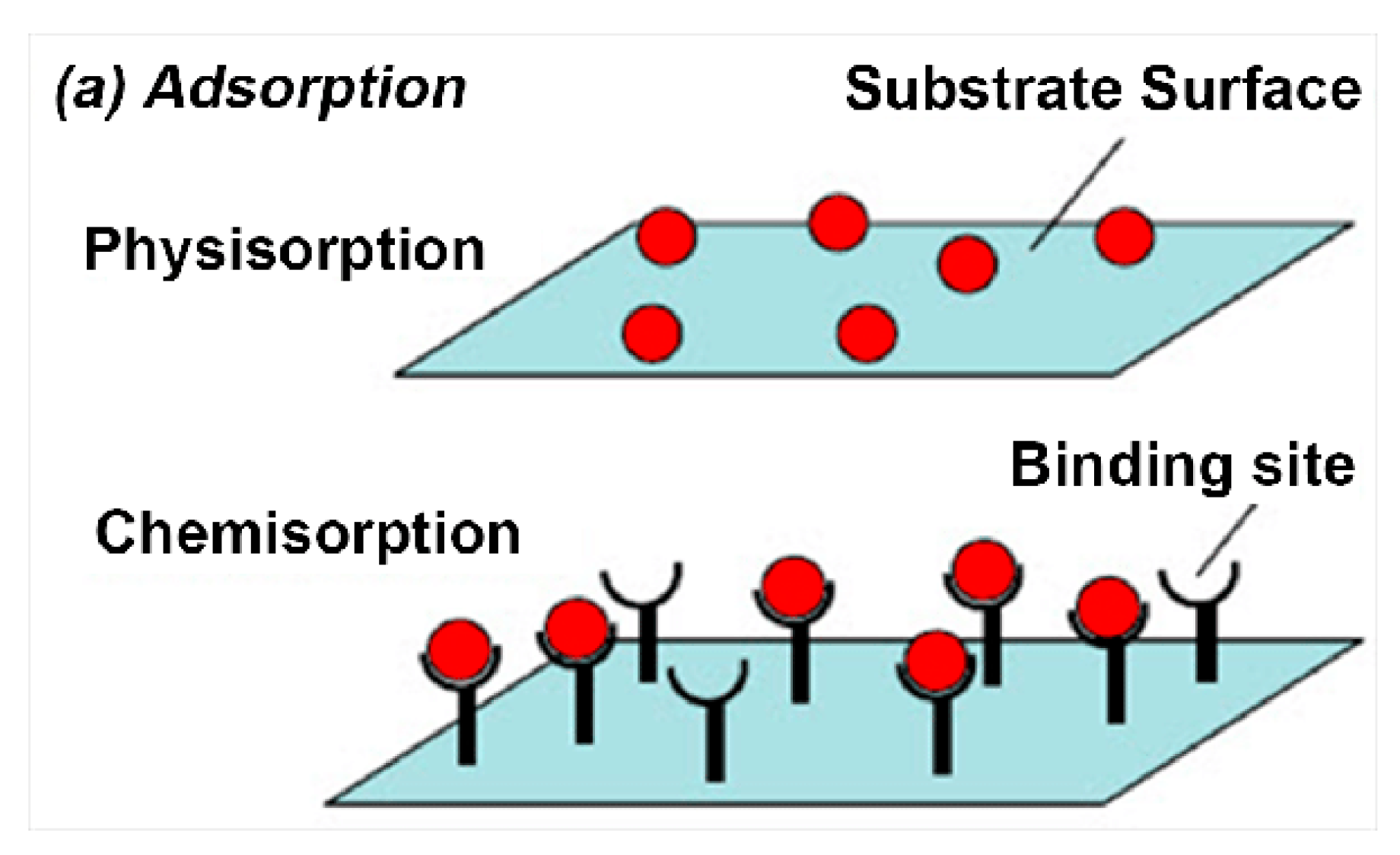
3.1.1. Physical Adsorption
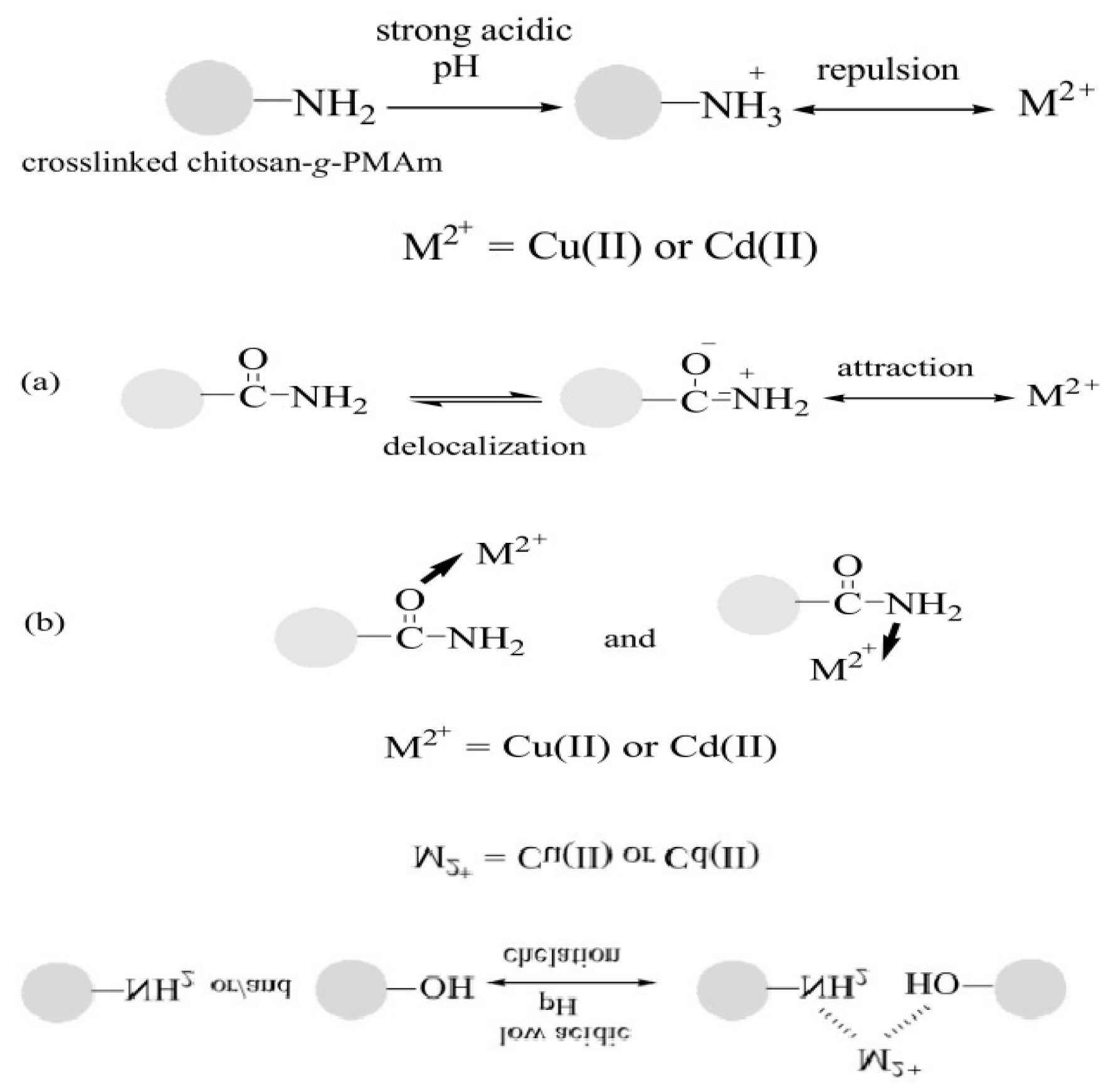
3.1.2. Chemical Adsorption
- I.
- Step 1: Cr6+ ions, either as chromate (CrO42−) or dichromate (Cr2O72−), are adsorbed onto the biosorbent surface: Biosorbent-NH2 + CrO42− → Biosorbent-Cr(VI);
- II.
- Step 2: Cr6+ undergoes reduction by receiving electrons from the biosorbent. The general reduction half-reaction is: Cr6+ + 3e− → Cr3+
- III.
- Step 3: Protonation of the reduced Cr3+ Species: CrO42− + 8H+ + 3e− → Cr3+ + 4H2O;
- IV.
- Step 4: Cr3+ ions are formed and remain adsorbed on the biosorbent: Cr3+ + 3NH2− → Cr(NH2)3 (S);
- V.
- Step 5: Cr3+ ions are then stabilized on the biosorbent surface through complexation- Biosorbent Cr3+ (complex)
Ion Exchange
Complexation
3.2. Factors Influencing Biosorption
3.2.1. pH
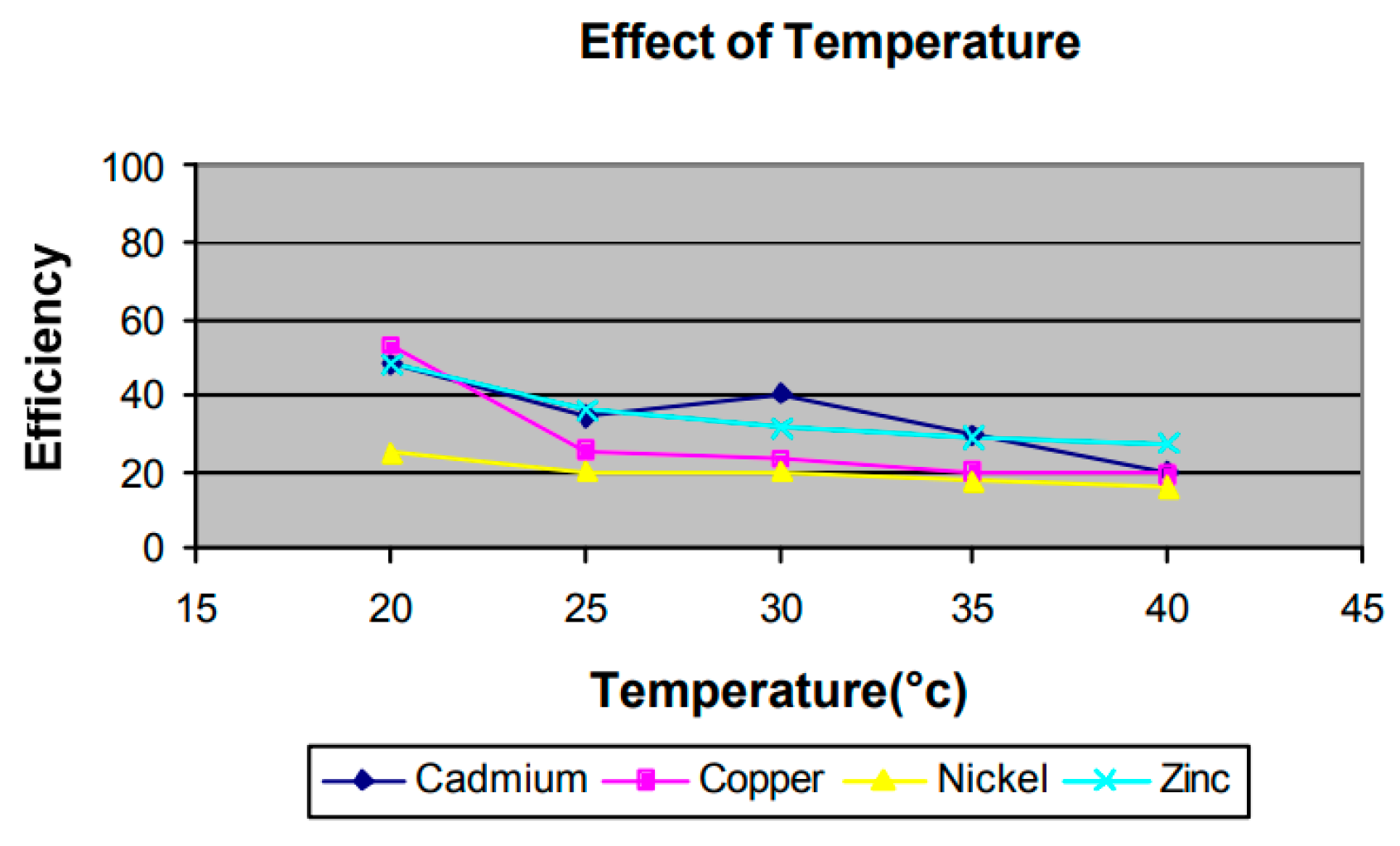
3.2.2. Temperature
3.2.3. Concentration
3.2.4. Particle Size
4. Current Status of Research on Biosorbents
5. Challenges and Future Directions
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Agrifood, M. Types of Agricultural Water Use. 2022. Available online: https://mazeros.com/types-of-agricultural-water-use/ (accessed on 31 July 2024).
- Beltecno. Why Industries Have to Be Water Efficient in Coming Future? 2020. Available online: https://www.beltecno-global.com/blog/why-industries-have-to-be-water-efficient-in-coming-future- (accessed on 31 July 2024).
- Goodnet. Fun Without Limitations. 2017. Available online: https://www.goodnet.org/articles/this-water-park-haven-for-kids-special-needs (accessed on 31 July 2024).
- Setlhare, L. Assessment of Wastewater Management in Gaborone, Botswana; Department of Environmental Affairs: Gaborone, Botswana, 2021. [Google Scholar]
- United Nations Environment Programme (UNEP). Wastewater Management in Africa: A Review of the Current Status and Future Prospects; United Nations Environment Programme (UNEP): Nairobi, Kenya, 2021. [Google Scholar]
- World Health Organization (WHO). Water Pollution and Health. 2023. Available online: https://www.who.int/news-room/fact-sheets/detail/drinking-water (accessed on 17 April 2025).
- United Nations. Department of Economic and Social Affairs. Sustainable Development Goal 6. 2025. Available online: https://sdgs.un.org/goals/goal6 (accessed on 16 February 2025).
- Prüss-Ustün, A.; Wolf, J.; Bartram, J.; Clasen, T.; Cumming, O.; Freeman, M.C.; Johnston, R. Burden of disease from inadequate water, sanitation and hygiene for selected adverse health outcomes: An updated analysis with a focus on low- and middle-income countries. Int. J. Hyg. Environ. Health 2019, 222, 765–777. [Google Scholar] [CrossRef]
- United Nations. Department of Economic and Social Affairs. Sustainable Development Goal 3. 2025. Available online: https://sdgs.un.org/goals/goal3 (accessed on 1 March 2025).
- Silver, N. Why Is Water Important? 16 Reasons to Drink Up- Healthline. 2023. Available online: https://www.healthline.com/health/food-nutrition/why-is-water-important (accessed on 26 October 2023).
- World Bank. High and Dry: Climate Change, Water, and the Economy; World Bank: Washington, DC, USA, 2016. [Google Scholar]
- Food and Agriculture Organization of the United Nations (FAO). Water for Sustainable Food and Agriculture: A Report Produced for the G20 Presidency of Germany; FAO: Rome, Italy, 2017; Available online: https://www.fao.org/3/i7959e/i7959e.pdf (accessed on 16 March 2024).
- Redha, A.A. Removal of heavy metals from aqueous media by biosorption. J. Basic Appl. Sci. 2020, 9, 183–193. [Google Scholar] [CrossRef]
- Lu, F.; Astruc, D. Nanocatalysts and other nanomaterials for water remediation from organic pollutants. Coord. Chem. Rev. 2020, 408, 213180. [Google Scholar] [CrossRef]
- Guo, Z.; Chen, X.; Hang, J.; Li, Z.; Zhong, C.; Sun, A.; Li, J.; Xu, S. Oxidative magnetization of biochar at relatively low pyrolysis temperature for efficient removal of different types of pollutants. Bioresour. Technol. 2023, 374, 129572. [Google Scholar] [CrossRef]
- Rathi, B.S.; Kumar, P.S.; Vo, D.-V.N. Critical review on hazardous pollutants in water environment: Occurrence, monitoring, fate, removal technologies and risk assessment. Sci. Total Environ. 2021, 797, 149134. [Google Scholar] [CrossRef]
- Xu, R.; Su, M.; Liu, Y.; Chen, Z.; Ji, C.; Yang, M.; Chang, X.; Chen, D. Comparative study on the removal of different-type organic pollutants on hierarchical tetragonal bismutite microspheres: Adsorption, degradation and mechanism. J. Clean. Prod. 2020, 262, 121336. [Google Scholar] [CrossRef]
- Yefremova, S.; Kablanbekov, A.; Satbaev, B.; Zharmenov, A. Rice Husk-Based Adsorbents for Removal of Metals from Aqueous Solutions. Materials 2023, 16, 7353. [Google Scholar] [CrossRef]
- Yefremova, S.; Kablanbekov, A.; Satbaev, B.; Zharmenov, A. A comparative study of heavy metals removal using agricultural waste biosorbents. Bioresour. Technol. Rep. 2021, 15, 100742. [Google Scholar] [CrossRef]
- Madela, M.; Skuza, M. Towards a Circular Economy: Analysis of the Use of Biowaste as Biosorbent for the Removal of Heavy Metals. Energies 2021, 14, 6257. [Google Scholar] [CrossRef]
- Addisie, M.B. Evaluating Drinking Water Quality Using Water Quality Parameters and Esthetic Attributes. Air Soil Water Res. 2022, 15, 11786221221075005. [Google Scholar] [CrossRef]
- Lapworth, D.J.; Nkhuwa, D.C.W.; Okotto-Okotto, J.; Pedley, S.; Stuart, M.E.; Tijani, M.N.; Wright, J. Urban groundwater quality in sub-Saharan Africa: Current status and implications for water security and public health. Hydrogeol. J. 2017, 25, 1093–1116. [Google Scholar] [CrossRef]
- Howard, G.; Pedley, S.; Barrett, M.; Nalubega, M.; Johal, K. Risk factors contributing to microbiological contamination of shallow groundwater in Kampala, Uganda. Water Res. 2003, 37, 3421–3429. [Google Scholar] [CrossRef]
- Kanyerere, T.; Levy, J.; Xu, Y.; Saka, J. Assessment of Microbial Contamination of Groundwater in Upper Limphasa River Catchment, Located in a Rural Area of Northern Malawi. Water SA 2012, 38, 581–596. [Google Scholar] [CrossRef]
- Still, D.A.; Nash, S.R. Groundwater contamination due to pit latrines located in a sandy aquifer: A case from Maputaland. In Proceedings of the Water Institute of Southern Africa (WISA) Biennial Conference, Durban, South Africa, 19–23 May 2002. [Google Scholar]
- Alemaw, B.F.; Shemang, E.M.; Chaoka, T.R. Assessment of groundwater pollution vulnerability and modelling of the Kanye Wellfield in SE Botswana—A GIS approach. Phys. Chem. Earth 2004, 29, 1125–1128. [Google Scholar] [CrossRef]
- Yaashikaa, P.R.; Kumar, P.S.; Saravanan, A.; Vo, D.-V.N. Advances in biosorbents for removal of environmental pollutants: A review on pretreatment, removal mechanism and future outlook. J. Hazard. Mater. 2021, 420, 126596. [Google Scholar] [CrossRef]
- Makoś-Chełstowska, P.; Słupek, E.; Gębicki, J. Agri-food waste biosorbents for volatile organic compounds removal from air and industrial gases—A review. Sci. Total Environ. 2024, 927, 173910. [Google Scholar] [CrossRef]
- Martín-García, B.; Longo, E.; Ceci, A.T.; Pii, Y.; Romero-Gozalez, R.; Frenich, A.G.; Boselli, E. Pesticides and winemaking: A comprehensive review of conventional and emerging approaches. Compr. Rev. Food Sci. Food Saf. 2024, 23, e13419. [Google Scholar] [CrossRef]
- Gao, P.; Chen, D.; Chen, W.; Sun, J.; Wang, G.; Zhou, L. Facile synthesis of amine-crosslinked starch as an efficient biosorbent for adsorptive removal of anionic organic pollutants from water. Int. J. Biol. Macromol. 2021, 189, 1240–1248. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Vilar, V.J.; Botelho, C.M.; Boaventura, R.A. Coconut-based biosorbents for water treatment—a review of the recent literature. Adv. Colloid Interface Sci. 2010, 160, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Aranda-García, E.; Chávez-Camarillo, G.M.; Cristiani-Urbina, E. Effect of Ionic Strength and Coexisting Ions on the Biosorption of Divalent Nickel by the Acorn Shell of the Oak Quercus crassipes Humb. & Bonpl. Processes 2020, 8, 1439. [Google Scholar] [CrossRef]
- Sharma, S.; Bhattacharya, A. Drinking water contamination and treatment techniques. Appl. Water Sci. 2017, 7, 1043–1067. [Google Scholar] [CrossRef]
- Landrigan, P.J.; Stegeman, J.J.; Fleming, L.E. Human health and ocean pollution. Ann. Glob. Health 2020, 86, 151. [Google Scholar] [CrossRef]
- Richards, M.; Dawson, J.; Stutter, M. The potential use of natural vs commercial biosorbent material to remediate stream waters by removing heavy metal contaminants. J. Environ. Manag. 2019, 231, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Bulgariu, L.; Bulgariu, D. Potential use of alkaline treated algae waste biomass as sustainable biosorbent for clean recovery of cadmium (II) from aqueous media: Batch and column studies. J. Clean. Prod. 2016, 112, 4525–4533. [Google Scholar] [CrossRef]
- Hossain, M.S.; Hossain, M.M.; Khatun, M.K.; Hossain, K.R. Hydrogel-based superadsorbents for efficient removal of heavy metals in industrial wastewater treatment and environmental conservation. Environ. Funct. Mater. 2024, 3, 100001. [Google Scholar] [CrossRef]
- Wu, J.; Wang, T.; Wang, J.; Zhang, Y.; Pan, W. A novel modified method for the efficient removal of Pb and Cd from wastewater by biochar: Enhanced the ion exchange and precipitation capacity. Sci. Total Environ. 2021, 754, 142150. [Google Scholar] [CrossRef]
- Mushtaq, F.; Zahid, M.; Bhatti, A.A.; Nasir, S.; Hussain, T. Possible applications of coal fly ash in wastewater treatment. J. Environ. Manag. 2019, 240, 27–46. [Google Scholar] [CrossRef]
- Nasrollahzadeh, M.; Sajjadi, M.; Iravani, S.; Varma, R.S. Carbon-based sustainable nanomaterials for water treatment: State-of-art and future perspectives. Chemosphere 2021, 270, 128005. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, J.; Wang, Y.; Chen, H. Research and application of water treatment technologies for emerging pollutants. Water 2024, 16, 1837. [Google Scholar] [CrossRef]
- Tee, G.T.; Gok, X.Y.; Yong, W.F. Adsorption of pollutants in wastewater via biosorbents, nanoparticles and magnetic biosorbents: A review. Environ. Res. 2022, 212, 113248. [Google Scholar] [CrossRef]
- Gahlot, S.; Kulshrestha, V. Fluoropolymer nanocomposite membranes for fuel cell applications. In Advanced Fluoropolymer Nanocomposites: Fabrication, Processing, Characterization and Applications; Woodhead Publishing Series in Composites Science and Engineering: Cambridge, UK, 2023; pp. 597–643. [Google Scholar] [CrossRef]
- Fernandes, J.; Ramísio, P.; Puga, H.A. Comprehensive Review on Various Phases of Wastewater Technologies: Trends and Future Perspectives. Eng 2024, 5, 2633–2661. [Google Scholar] [CrossRef]
- Gupta, V.K.; Pathania, D. Biosorbents for water purification: A review. Environ. Chem. Lett. 2021, 19, 2351–2381. [Google Scholar] [CrossRef]
- Dong, H.; Chen, X.; Wu, Y. Bacteria biosorbents for heavy metals removal: A review. J. Hazard. Mater. 2021, 418, 126336. [Google Scholar] [CrossRef]
- Li, M.; Liu, X.; Xie, J. Enhanced cadmium removal from aqueous solution using surface-modified peanut shell biochar. Environ. Pollut. 2021, 269, 116178. [Google Scholar] [CrossRef]
- Ayach, J.; El Malti, W.; Duma, L.; Lalevée, J.; Al Ajami, M.; Hamad, H.; Hijazi, A. Comparing conventional and advanced approaches for heavy metal removal in wastewater treatment: An in-depth review. Polymers 2024, 16, 1959. [Google Scholar] [CrossRef]
- Osman, A.I.; Abd El-Monaem, E.M.; Elgarahy, A.M.; Aniagor, C.O.; Hosny, M.; Farghali, M.; Rashad, E.; Ejimofor, M.I.; López-Maldonado, E.A.; Ihara, I.; et al. Methods to prepare biosorbents and magnetic sorbents for water treatment: A review. Environ. Chem. Lett. 2023, 21, 2337–2398. [Google Scholar] [CrossRef]
- Elgarahy, A.M.; Elwakeel, K.Z.; Mohammad, S.H.; Elshoubaky, G.A. A critical review of biosorption of dyes, heavy metals and metalloids from wastewater as an efficient and green process. Clean. Eng. Technol. 2021, 4, 100209. [Google Scholar] [CrossRef]
- Zamora-Ledezma, C. Heavy metal water pollution: A fresh look about hazards, novel and conventional remediation methods. Environ. Technol. 2021, 42, 101504. [Google Scholar] [CrossRef]
- Chowdhury, S.M.M. Recent trends and emerging strategies of biosorbent-based heavy metals removal from wastewater: A review. J. Environ. Chem. Eng. 2021, 9, 105861. [Google Scholar] [CrossRef]
- Banch, T.J.H.; Hanafiah, M.M.; Amr, S.S.A.; Alkarkhi, A.F.M.; Hasan, M. Treatment of landfill leachate using palm oil mill effluent. Processes 2020, 8, 601. [Google Scholar] [CrossRef]
- Okoro, H.K.; Pandey, S.; Ogunkunle, C.O.; Ngila, C.J.; Zvinowanda, C.; Jimoh, I.; Lawal, I.A.; Orosun, M.M.; Adeniyi, A.G. Nanomaterial-based biosorbents: Adsorbent for efficient removal of selected organic pollutants from industrial wastewater. Emerg. Contam. 2022, 8, 46–58. [Google Scholar] [CrossRef]
- Halysh, V.; Trus, I.; Radovenchyk, V.; Gomelya, M. Efficient biosorbents for wastewater treatment: Preparation, characterization, utilization. J. Chem. Technol. Metall. 2022, 57, 302–309. [Google Scholar]
- Plazinski, W. Binding of heavy metals by algal biosorbents. Theoretical models of kinetics, equilibria and thermodynamics. Adv. Colloid Interface Sci. 2013, 197–198, 58–67. [Google Scholar] [CrossRef]
- Shrestha, R.; Ban, S.; Devkota, S.; Sharma, S.; Joshi, R.; Tiwari, A.R.; Kim, H.Y.; Joshi, M.K. Technological trends in heavy metals removal from industrial wastewater: A review. J. Environ. Chem. Eng. 2021, 9, 105688. [Google Scholar] [CrossRef]
- Fan, Q.; Lu, T.; Deng, Y.; Zhang, Y.; Ma, W.; Xiong, R.; Huang, C. Bio-based materials with special wettability for oil-water separation. Sep. Purif. Technol. 2022, 297, 121445. [Google Scholar] [CrossRef]
- Quintas, P.Y.; Fiorentini, E.F.; Oviedo, M.N.; Escudero, L.B. Magnetic Biosorbents and Their Applications in Water Remediation. In Advanced Magnetic Adsorbents for Water Treatment; Springer: Cham, Switzerland, 2021; pp. 59–104. [Google Scholar] [CrossRef]
- Kumar, R.T.A. A comprehensive review on activated carbon for heavy metal removal from aqueous solution. J. Environ. Chem. Eng. 2020, 8, 104257. [Google Scholar] [CrossRef]
- Wang, L.; Lin, J.; Zhang, M.; Xu, L. Chitosan-based biosorbents: Modification and application for heavy metal removal. Carbohydr. Polym. 2021, 251, 117118. [Google Scholar] [CrossRef]
- Gautam, K.; Gupta, N.; Patel, D.K.; Singh, R.P.; Singh, A. Comparative study of simple and modified rice husk biochar for cadmium removal: Adsorption performance and possible mechanisms. Aqua Water Infrastruct. Ecosyst. Soc. 2023, 72, 1269–1287. [Google Scholar] [CrossRef]
- Saleem, J.; Moghal, Z.K.B.; Pradhan, S.; McKay, G. High-performance activated carbon from coconut shells for dye removal: Study of isotherm and thermodynamics. RSC Adv. 2024, 14, 33797–33808. [Google Scholar] [CrossRef]
- Li, M.; Xiao, X.; Wang, S.; Zhang, X.; Li, J.; Pavlostathis, S.G.; Luo, X.; Luo, S.; Zeng, G. Synergistic removal of cadmium and organic matter by a microalgae-endophyte symbiotic system (MESS): An approach to improve the application potential of plant-derived biosorbents. Environ. Pollut. 2020, 265, 114177. [Google Scholar] [CrossRef]
- Pham, V.H.T.; Kim, J.; Chang, S.; Chung, W. Bacterial Biosorbents, an Efficient Heavy Metals Green Clean-Up Strategy: Prospects, Challenges, and Opportunities. Microorganisms 2022, 10, 610. [Google Scholar] [CrossRef]
- Li, Y.; Yu, H. Algae-based biosorbents for the removal of heavy metals. Algal Res. 2020, 51, 102035. [Google Scholar] [CrossRef]
- Saravanan, A.; Karishma, S.; Kumar, P.S.; Thamarai, P.; Yaashikaa, P.R. Recent insights into mechanism of modified bio-adsorbents for the remediation of environmental pollutants. Environ. Pollut. 2023, 335, 122720. [Google Scholar] [CrossRef]
- Adewuyi, A. Chemically Modified Biosorbents and Their Role in the Removal of Emerging Pharmaceutical Waste in the Water System. Water 2020, 12, 1551. [Google Scholar] [CrossRef]
- Majiya, H.; Clegg, F.; Sammon, C. Bentonite-Chitosan composites or beads for lead (Pb) adsorption: Design, preparation, and characterisation. Appl. Clay Sci. 2023, 246, 107180. [Google Scholar] [CrossRef]
- El-Nahas, S.; Osman, A.I.; Arafat, A.S.; Al-Muhtaseb, A.H.; Salman, H.M. Facile and affordable synthetic route of nano powder zeolite and its application in fast softening of water hardness. J. Water Process Eng. 2020, 33, 101104. [Google Scholar] [CrossRef]
- Mambetova, M.; Dossumov, K.; Baikhamurova, M.; Yergaziyeva, G. Sorbents based on natural zeolites for carbon dioxide capture and removal of heavy metals from wastewater: Current progress and future opportunities. Processes 2024, 12, 2071. [Google Scholar] [CrossRef]
- Lahiri, S.K.; Liu, L. Fabrication of a nanoporous silica hydrogel by cross-linking of SiO2–H3BO3–hexadecyltrimethoxysilane for excellent adsorption of azo dyes from wastewater. Langmuir 2021, 37, 8753–8764. [Google Scholar] [CrossRef]
- Vijaya, Y.; Venkata Subbaiah, M.; Subba Reddy, A.; Ajitha Priya, J.; Krishnaiah, A. Chitosan coated silica as biosorbent to remove Cr(VI) from aqueous solutions. Desalin. Water Treat. 2010, 20, 272–280. [Google Scholar]
- Thines, R.K.; Mubarak, N.M.; Nizammudin, S.; Sahu, J.N.; Abdullah, E.C.; Ganesan, P. Application potential of carbon nanomaterials in water and wastewater treatment: A review. J. Taiwan Inst. Chem. Eng. 2017, 72, 116–133. [Google Scholar] [CrossRef]
- Boer, D.G.; Langerak, J.; Pescarmona, P.P. Zeolites as Selective Adsorbents for CO2 Separation. ACS Appl. Energy Mater. 2023, 6, 3983–4002. [Google Scholar] [CrossRef]
- Duan, C.; Meng, X.; Liu, C.; Lu, W.; Liu, J.; Dai, L.; Wang, W.; Zhao, W.; Xiong, C.; Ni, Y. Carbohydrates-rich corncobs supported metal-organic frameworks as versatile biosorbents for dye removal and microbial inactivation. Carbohydr. Polym. 2019, 218, 115042. [Google Scholar] [CrossRef]
- Omidinasab, M.; Rahbar, N.; Ahmadi, M.; Kakavandi, B.; Ghanbari, F.; Kyzas, G.Z.; Martinez, S.S.; Jaafarzadeh, N. Removal of vanadium and palladium ions by adsorption onto magnetic chitosan nanoparticles. Environ. Sci. Pollut. Res. 2018, 25, 34262–34276. [Google Scholar] [CrossRef]
- Zubair, M.; Noreen, S.; Bukhari, S.I. Nano-modified feather keratin derived green and sustainable biosorbents for the remediation of heavy metals from synthetic wastewater. Environ. Technol. Innov. 2022, 28, 102639. [Google Scholar] [CrossRef]
- Tran, H.N. Modified natural biosorbent for dye removal from aqueous solution. J. Environ. Chem. Eng. 2010, 8, 104462. [Google Scholar] [CrossRef]
- Torres, E. Biosorption: A review of the latest advances. Processes 2020, 8, 1584. [Google Scholar] [CrossRef]
- Bai, Z.; Gao, B.; Yang, L. Adsorption of lead (Pb2+) from aqueous solutions by biochar derived from agricultural residues. Environ. Sci. Pollut. Res. 2021, 28, 3461–3471. [Google Scholar] [CrossRef]
- Ahmad, A.; Ghazi, Z.A.; Saeed, M.; Ilyas, M.; Ahmad, R.; Khattak, A.M.; Iqbal, A. A comparative study of the removal of Cr(VI) from synthetic solution using natural biosorbents. New J. Chem. 2017, 41, 10799–10807. [Google Scholar] [CrossRef]
- Park, J.; Wang, Y.; Chen, L.; Zhu, Y.; Fang, W.; Tan, Y.; He, Z.; Liao, H. Research status, trends, and mechanisms of biochar adsorption for wastewater treatment: A scientometric review. Environ. Sci. Eur. 2023, 35, 73. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Y.; Zhang, X.; Lu, S. Effective removal of heavy metals using thiourea modified biosorbents: A review. J. Hazard. Mater. 2018, 344, 362–370. [Google Scholar] [CrossRef]
- Dey, S.; Basha, S.R.; Babu, G.V.; Nagendra, T. Characteristic and biosorption capacities of orange peels biosorbents for removal of ammonia and nitrate from contaminated water. Clean. Mater. 2021, 2, 100001. [Google Scholar] [CrossRef]
- Khatri, A.; Singh, J. Functional groups in bio-based adsorbents: Roles in heavy metal removal. J. Environ. Chem. Eng. 2023, 11, 109257. [Google Scholar] [CrossRef]
- Wang, S.; Chen, Y. Surface functionalization of biosorbents for pollutant adsorption: A review. Chem. Eng. J. 2022, 428, 131187. [Google Scholar] [CrossRef]
- Liu, W.; Sun, Z. Influence of elemental ratios in biosorbents on their adsorption efficiency. J. Clean. Prod. 2021, 278, 123456. [Google Scholar] [CrossRef]
- Yang, T.; Fan, J. Stability and reusability of chemically modified biosorbents. Environ. Technol. Innov. 2023, 29, 102987. [Google Scholar] [CrossRef]
- Vallero, D.A. Control of hazardous air pollutants. In Fundamentals of Air Pollution; Academic Press: Cambridge, MA, USA, 2008; pp. 825–851. [Google Scholar]
- Venkata, A.; Kumar, A.; Darwish, N.A.; Hilal, N. Study of Various Parameters in the Biosorption of Heavy Metals on Activated Sludge. World Appl. Sci. J. 2009, 7, 32–40. [Google Scholar]
- Yasemin, B.; Tez, Z. Adsorption studies on ground shells of hazelnut and almond. J. Hazard. Mater. 2007, 149, 35–41. [Google Scholar] [CrossRef]
- Yang, X.; Wang, L.; Shao, X.; Tong, J.; Zhou, J.; Feng, Y.; Chen, R.; Yang, Q.; Han, Y.; Yang, X.; et al. Characteristics and aqueous dye removal ability of novel biosorbents derived from acidic and alkaline one-step ball milling of hickory wood. Chemosphere 2022, 307, 136610. [Google Scholar] [CrossRef]
- Masoumi, A.; Hemmati, K.; Ghaedi, M.; Khodadoust, S. Low-Cost Nanoparticles Sorbent from Modified Rice Husk and a Copolymer for Efficient Removal of Pb(II) and Crystal Violet from Water. Chemosphere 2016, 146, 253–262. [Google Scholar] [CrossRef]
- Supiyeva, Z.; Avchukir, K.; Pavlenko, V.; Yeleuov, M.; Taurbekov, A.; Smagulova, G.; Mansurov, Z. The Investigation of Electroreduction of AuCl4- in the Case of Gold Electrosorption Using Activated Carbon. Mater. Today Proc. 2020, 25, 33–38. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Gunjal, A. Langmuir Kinetics Study for Biosorption of Heavy Metals by Fungi. Micro Environ. 2023, 13, 7–9. [Google Scholar] [CrossRef]
- Moges, G.; Mulu, T.; Taddesse, A.M. Removal of cadmium from aqueous solution using acid-modified rice husk: Adsorption isotherms, kinetics, and thermodynamics. Environ. Syst. Res. 2022, 11, 23. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Sillanpää, M.; Witek-Krowiak, A.; Lima, E.C. Agricultural waste-derived biosorbents for water treatment: Advances and future prospects. J. Environ. Manag. 2022, 317, 115304. [Google Scholar] [CrossRef]
- Freundlich, H. Über die adsorption in lösungen. Z. Phys. Chem. 1906, 57, 385–470. [Google Scholar] [CrossRef]
- Mohammed, A.; Shartooh, S.; Trigui, M. Biosorption and Isotherm Modeling of Heavy Metals Using Phragmites australis. Sustainability 2025, 17, 5366. [Google Scholar] [CrossRef]
- Lagergren, S. Zur theorie der sogenannten adsorption gelöster stoffe. K. Sven. Vetenskapsakademiens Handl. 1898, 24, 1–39. [Google Scholar]
- Ho, Y.S.; Mckay, G.; Wase, D.A.J.; Forster, C.F. Removal of heavy metals from aqueous solutions using modified peat. J. Hazard. Mater. 2006, 137, 1805–1812. [Google Scholar] [CrossRef]
- Lodh, B.K. Bisorbents for heavy metal removal. In Microbial Ecology of Wastewater Treatment Plants; Elsevier: Amsterdam, The Netherlands, 2021; pp. 377–394. [Google Scholar]
- Karić, N.; Maia, A.S.; Teodorović, A.; Atanasova, N.; Langergraber, G.; Crini, G.; Đolić, M. Bio-waste valorisation: Agricultural wastes as biosorbents for removal of (in)organic pollutants in wastewater treatment. Chem. Eng. J. Adv. 2022, 9, 100239. [Google Scholar] [CrossRef]
- Muthamilselvi, P.; Sivasamy, B.; Ashish, K.; Sivaraman, P. Natural materials as adsorbents for water purification. In Green Sustainable Process for Chemical and Environmental Engineering and Science, Natural Materials Based Green Composites 1: Plant Fibers; Chapter 7; Elsevier: Amsterdam, The Netherlands, 2023; pp. 123–144. [Google Scholar] [CrossRef]
- Kennedy, K.K.; Maseka, K.J.; Mbulo, M. Selected Adsorbents for Removal of contaminants from wastewater: Towards engineering clay minerals. Open J. Appl. Sci. 2018, 8, 355–365. [Google Scholar] [CrossRef]
- Gusain, R.; Kumar, N.; Ray, S.S. Recent advances in carbon nanomaterial-based adsorbents for water purification. Coord. Chem. Rev. 2020, 405, 213111. [Google Scholar] [CrossRef]
- Saravanan, A.; Kumar, P.S.; Jeevanantham, S.; Karishma, S.; Tajsabreen, B.; Yaashikaa, P.R.; Reshma, B. Effective water/wastewater treatment methodologies for toxic pollutants removal: Processes and applications towards sustainable development. Chemosphere 2021, 280, 130595. [Google Scholar] [CrossRef]
- Qin, H.; Hu, T.; Zhai, Y.; Lu, N.; Aliyeva, J. The improved methods of heavy metals removal by biosorbents: A review. Environ. Pollut. 2020, 258, 113777. [Google Scholar] [CrossRef]
- Ho, S.H.; Yang, Z.K.; Nagarajan, D.; Chang, J.S.; Ren, N.Q. High-efficiency removal of lead from wastewater by biochar derived from anaerobic digestion sludge. Bioresour. Technol. 2017, 246, 142–149. [Google Scholar] [CrossRef]
- Ncibi, M.C.; Mahjoub, B.; Seffen, B. Optimization, equilibrium and kinetics study for lead and methylene blue sorption onto PAMAM/CNTs and its application in wastewater treatment. J. Mol. Liq. 2017, 248, 1049–1058. [Google Scholar] [CrossRef]
- Dey, S.; Veerendra, G.T.N.; Padavala, S.S.A.B.; Manoj, A.V.P. Evaluate the use of flower waste biosorbents for treatment of contaminated water. Water-Energy Nexus 2023, 6, 100001. [Google Scholar] [CrossRef]
- Aljamali, N.M.; Aldujaili, R.A.B.; Alfatlawi, I.O. Physical and chemical adsorption and its applications. Thermodyn. Chem. Kinet. 2021, 1, 1–15. [Google Scholar]
- Garg, R.; Garg, R.; Sillanpää, M.; Khan, A.M.A.; Mubarak, N.M.; Tan, Y.H. Rapid adsorptive removal of chromium from wastewater using walnut-derived biosorbents. Sci. Rep. 2023, 13, 6859. [Google Scholar] [CrossRef]
- Lakshmi, U.R.; Srivastava, V.C.; Mall, I.D.; Lataye, D.H. Adsorption of phenol from aqueous solutions using activated carbon prepared from waste wood. J. Hazard. Mater. 2008, 153, 620–627. [Google Scholar] [CrossRef]
- Zhang, M.; Song, Y.; Fang, Q. Recent progress in MOF-based materials for heavy metal removal from water. Coord. Chem. Rev. 2020, 423, 213482. [Google Scholar] [CrossRef]
- Mohan, D.; Pittman, C.U.; Steele, P.H. Biochar for the removal of heavy metals and organic pollutants from water: A review. Bioresour. Technol. 2014, 160, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Niu, L.; Zhang, B.; Tang, X.; Huang, X.; Zhang, Q.; Wang, Y. Chemisorption of Cr(VI) from aqueous solutions using modified orange peel biosorbent: Optimization, isotherm and kinetic studies. J. Environ. Chem. Eng. 2021, 9, 105687. [Google Scholar] [CrossRef]
- He, H.; Zhang, Z.; Cai, H.; Chen, W. Chemisorption of lead (II) on graphene oxide: Mechanism, structure and properties. Chem. Eng. J. 2016, 302, 509–515. [Google Scholar] [CrossRef]
- Liang, Z.; Marshall, M.; Chaffe, A.L. Selective chemisorption of mercury (II) by thiol-functionalised metal-organic frameworks. J. Hazard. Mater. 2015, 300, 371–377. [Google Scholar] [CrossRef]
- Mehta, S.K.; Gaur, J.P. Chemisorption of copper (II) by chlorella vulgaris and its applicability to treating metal-contaminated water. J. Appl. Phycol. 2001, 13, 335–342. [Google Scholar]
- Kumar, S.; Jain, S. History, Introduction, and Kinetics of Ion Exchange Materials. J. Chem. 2013, 2013, 957647. [Google Scholar] [CrossRef]
- Sutirman, Z.A.; Sanagi, M.M.; Karim, K.J.A.; Ibrahim, W.A.W.; Jume, B.H. Equilibrium, kinetic and mechanism studies of Cu(II) and Cd(II) ions adsorption by modified chitosan beads. Int. J. Biol. Macromol. 2018, 116, 255–263. [Google Scholar] [CrossRef]
- Aljamali, N.M. Reactions and Mechanisms; IJMRA Publishing: Baghdad, Iraq, 2018. [Google Scholar]
- Sulaymon, A.H.; Mohammed, A.A.; Al-Musawi, T.J. Competitive biosorption of lead, cadmium, copper, and arsenic ions using algae. Environ. Sci. Pollut. Res. 2013, 20, 3011–3023. [Google Scholar] [CrossRef] [PubMed]
- Nie, J.; Zhang, D.; Zhang, Y.; Yang, Z. Magnetic biochar-based composites for removal of recalcitrant pollutants in water. In Sorbents Materials for Controlling Environmental Pollution; Elsevier: Amsterdam, The Netherlands, 2021; pp. 163–187. [Google Scholar]
- Guibal, E. Interactions of metal ions with chitosan-based sorbents: A review. Sep. Purif. Technol. 2004, 38, 43–74. [Google Scholar] [CrossRef]
- Michalak, I.; Baśladyńska, S.; Mokrzycki, J.; Rutkowski, P. Biochar from a freshwater macroalga as a potential biosorbent for wastewater treatment. Water 2019, 11, 1390. [Google Scholar] [CrossRef]
- Ciani, M.; Lepore, G.; Puri, A.; Facchetti, G.; Adessi, A. Exploring Metal Interactions with Released Polysaccharides from Cyanothece sp. CE4: A Chemical and Spectroscopic Study on Biosorption Mechanism. Polymers 2025, 17, 371. [Google Scholar] [CrossRef]
- Ajaykumar, A.V.; Saleh, A.H.; Hilal, N. Investigation of kinetics and mechanisms involved in the biosorption of heavy metals on activated sludge. Int. J. Green Energy 2008, 5, 313–321. [Google Scholar] [CrossRef]
- Cheah, W.Y.; Lim, L.T.; Lee, Y.L. Hybrid biosorbents from exopolymeric substances immobilized in Ca-alginate and their biosorption mechanisms in single and multi-metal systems. Sep. Sci. Technol. 2023, 58, 1485–1498. [Google Scholar] [CrossRef]
- Das, N.; Vimala, R.; Karthika, P. Biosorption of Cr(VI) by dead fungal biomass of Aspergillus niger. Bioresour. Technol. 2006, 97, 1091–1099. [Google Scholar] [CrossRef]
- Brazesh, B.; Mousavi, S.M.; Zarei, M. Biosorption. In Adsorption: Fundamental Processes and Applications, Interface Science and Technology; Elsevier: Amsterdam, The Netherlands, 2021; pp. 587–628. [Google Scholar] [CrossRef]
- OECD/OCDE. Determination of pH, Acidity and Alkalinity; CIPAC MT Publications: Paris, France, 2013. [Google Scholar]
- Chojnacka, K. Biosorption and Bioaccumulation in Practice; Nova Science Publishers: New York, NY, USA, 2009. [Google Scholar]
- Dorris, K.L.; Yu, B.U.; Zhang, Y.; Shukla, A.; Shukla, S.S. The removal of heavy metal from aqueous solutions by sawdust adsorption-removal of copper. J. Hazard. Mater. 2000, 80, 33–42. [Google Scholar] [CrossRef]
- Osu, C.I.; Odoemelam, S.A. Studies on adsorbent dosage, particle sizes and pH constraints on biosorption of Pb(II) and Cd(II) ions from aqueous solution using modified and unmodified Crasstrotrea Gasar (Bivalve) biomass. Int. J. Adv. Sci. Res. 2010, 1, 62–68. [Google Scholar]
- Cuppett, J.D.; Duncan, S.E.; Dietrich, A.M. Evaluation of Copper Speciation and Water Quality Factors That Affect Aqueous Copper Tasting Response. Chem. Senses 2006, 31, 689–697. [Google Scholar] [CrossRef] [PubMed]
- Grimes, R.T.; Leginze, J.A.; Zochowski, R.; Bennett, J.W. Inorganic Chemistry. Inorg. Chem. 2021, 60, 1228–1240. [Google Scholar] [CrossRef]
- Abka-Khajouei, R.; Tounsi, L.; Shahabi, N.; Patel, A.K.; Abdelkafi, S.; Michaud, P. Structures, Properties and Applications of Alginates. Mar. Drugs 2022, 20, 364. [Google Scholar] [CrossRef]
- da Silva Alves, D.C.; Healy, B.; Pinto, L.A.d.A.; Cadaval, T.R.S., Jr.; Breslin, C.B. Recent Developments in Chitosan-Based Adsorbents for the Removal of Pollutants from Aqueous Environments. Molecules 2021, 26, 594. [Google Scholar] [CrossRef]
- Afolabi, F.O.; Musonge, P.; Bakare, B.F. Adsorption of Copper and Lead Ions in a Binary System onto Orange Peels: Optimization, Equilibrium and Kinetic Study. Environ. Nanotechnol. Monit. Manag. 2022, 18, 100709. [Google Scholar] [CrossRef]
- Al-Qodah, Z. Biosorption of heavy metal ions from aqueous solutions by activated sludge. Desalination 2006, 196, 164–176. [Google Scholar] [CrossRef]
- Wang, M.; Yaakop, A.S.; Ng, S.L. A comparative study of Cu(II) biosorption onto dried activated sludge of different sludge ages. Pure Appl. Chem. 2025. [Google Scholar] [CrossRef]
- Abiodun, O.-A.; Oluwaseun, O.; Oladayo, O.; Abayomi, O.; George, A.; Opatola, E.; Orah, R.; Isukuru, E.; Ede, I.; Oluwayomi, O.; et al. Remediation of Heavy Metals Using Biomass-Based Adsorbents: Adsorption Kinetics and Isotherm Models. Clean Technol. 2023, 5, 934–960. [Google Scholar] [CrossRef]
- Hassan, M.; Naidu, R.; Du, J.; Liu, Y.; Qi, F. Critical review of magnetic biosorbents: Their preparation, application, and regeneration for wastewater treatment. Sci. Total Environ. 2020, 702, 134893. [Google Scholar] [CrossRef]
- Puja; Kurniawati, D.; Nasra, E.; Bahrizal; Nizar, U.K.; Dewata, I. Effect of biosorbent particle size on biosorption of lead (II) from lengkeng seeds and shell (Euphoria logan Lour). Int. J. Prog. Sci. Technol. 2019, 15, 62–68.
- Shakoor, M.B.; Niazi, N.K.; Bibi, I.; Shahid, M.; Saqib, Z.A.; Nawaz, M.F.; Rinklebe, J. Exploring the arsenic removal potential of various biosorbents from water. Environ. Int. 2019, 123, 567–579. [Google Scholar] [CrossRef] [PubMed]

| Pollutant Class | Pollutant | Effects |
|---|---|---|
| Organics | Volatile organic compounds, for example, toluene, benzene, vinyl chloride, etc. | Exposure can be through ingestion or inhalation and causes chronic illnesses like cancer and reproductive disorders, and affects the central nervous system. Inhalation brings lung problems, and a mild problem associated with ingestion is ulcers [27,28]. |
| Persistent organic pollutants (POPs): Polychlorinated Biphenyls (PCBs), DDT (Dichloro diphenyl trichloroethane), | These can cause stroke, hypertension leading to heart failure, metabolic disease, reduced fetal growth, endocrine disruption (when hormones are interfered with), and cancer [8]. | |
| Pesticides, e.g., benzamide, found in Fluopyram | They cause skin irritation and allergic reactions [29]. | |
| Pharmaceuticals like amoxicillin, ciprofloxacin, ibuprofen, and diclofenac | Prolonged or even low-level exposure may lead to various organ dysfunctions. May also lead to hormonal imbalances to decreased fertility, as some of them are contraceptives, developmental abnormalities, and even congenital conditions in future generations [30]. | |
| Dyes: methylene blue, turmeric, congo red, crystal violet | Dye pollution in water brings allergies, cancer, dermatitis, and skin irritation [31]. | |
| Inorganics | Nickel (Ni) | It has harmful effects on human health. These include gastrointestinal complications, headache, anemia, dizziness, allergies, lung, and kidney failure [32]. |
| Total dissolved solids (TDS) Fluoride, F | Prolonged exposure to high amounts of dissolved salts leads to kidney stones. Fluoride consumption causes dental problems like teeth discoloration and weakening [33]. | |
| Arsenic, As | In addition to causing cancer, elevated levels of arsenic are associated with discoloration of the skin, stomach pain, nausea, diarrhea, and vomiting. | |
| Mercury, Hg | Causes neurological disorders and may lead to abortion in pregnant women. Methylmercury causes cardiovascular diseases and dementia in elderly people [34]. | |
| Copper, Cu Chromium, Cr | Nausea, diarrhea, liver, and kidney damage [19,35]. | |
| Cadmium, Cd Lead, Pb | Excessive amounts cause damage to the nervous system and destroy the blood circulation. Irritation of the respiratory system [36,37,38]. | |
| Biological pollutants | Viruses, protozoa, and bacteria | These produce harmful toxins and are cholera-causing organisms. Exposure causes liver damage and skin problems [33]. Mild problems are diarrhea, nausea, vomiting, headaches, and dehydration. |
| Type of Adsorbent | Synthesis Method and Active Functional Group | Pollutant Removed | The Amount Removed and the Mechanism of Biosorption |
|---|---|---|---|
| Natural-biomass-derived biosorbent: Modified walnut shells biosorbent comprising 41.2% cellulose | Chemical treatments were employed to enhance the adsorption efficiency. The adsorption capacity was due to the activation of hydroxyl and carboxyl functional groups. The experiment was carried out at pH 6 within 240 min of contact time, using a biosorbent dosage of 0.2 g. | Methylene blue | As the size of the walnut shell was reduced, the methylene sorption rate increased from 17.2% to 39.2% [55]. The mechanisms of biosorption were ion exchange and complexation. |
| Natural-biomass-derived biosorbent: Acorn shell of Quercus crassipes Humb. and Bonpl. (QCS) | Acorn shell biosorbent was cleaned, dried, and ground without chemical modification. The lignocellulosic components with hydroxyl, carboxyl, and phenolic groups contributed to metal ion binding. Biosorption was conducted at an optimal pH of 8.0, with a biosorbent dosage of 1 g/L, and contact times monitored for up to 120 h. | Chromium Nickel | Removed the hexavalent chromium. An increase in NaCl ionic strength increased the biosorption of Ni2+. The main adsorption mechanism is ion exchange, where divalent nickel ions exchange with cations on the acorn shell’s active sites [32]. |
| Natural-biomass-derived biosorbent: Coconut husk and tree sawdust | The material was modified with acids and bases to enhance its adsorption capability. Hydroxyl and carboxyl groups were active groups in binding the ions. | Cd(II), Cr(III) and Hg(II) ions Acid violet and acid brilliant blue Rhodamine B and methylene blue | The material removed about 84% to 96% of the metal ions. There was an 85% removal of rhodamine B at a lower pH of 3, while methylene blue was removed at about 97–100% at a pH of 6.9. The mechanisms of adsorption were chemical, that is, specifically ion exchange and surface complexation, since the positively charged ions were attaching to the negatively charged functional groups [31]. |
| Natural-biomass-derived biosorbent: Coconut coir | Both physical (particle size reduction) and chemical (use of acids) processing were employed. Hydroxyl and carboxyl groups were the binding surfaces for the adsorption of Zn and Pb ions. | Lead Zinc | 91% of Zn was removed at pH 4.5 and 97% at pH 2.5 for Pb. The mechanism of adsorption was ion exchange and complexation [31]. |
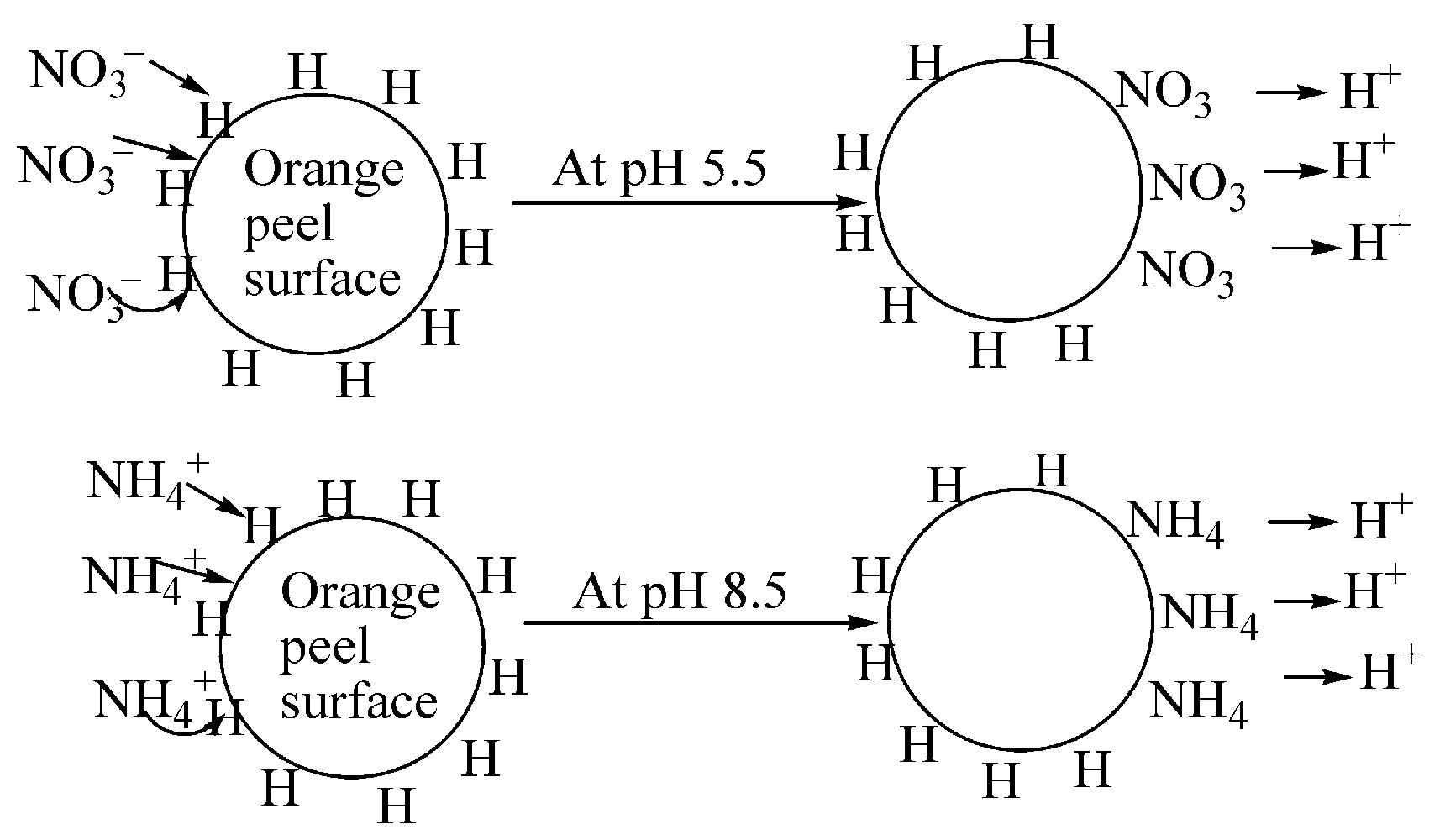
| Natural-biomass-derived biosorbent: Orange peels | Physical synthesis: The peels were washed, dried, and ground into powder. The amine group binds and traps the ammonia molecule, while a combination of carbonyl and hydroxyl groups has high affinities for pollutants. At pH 5.5, with an orange peel dosage of 4 g and a contact time of 60 min, the biosorbent showed optimal removal performance for both ammonia and nitrate. | Nitrate (NO3) Ammonia (NH) | 100% removal of nitrates and 70–100% of ammonia on the increased amount of orange peel. The mechanism of adsorption is ion exchange [85]. The work also included characterization of the material via XRD, FTIR, BET, and SEM-EDX to elucidate its structure and adsorption functionality. |
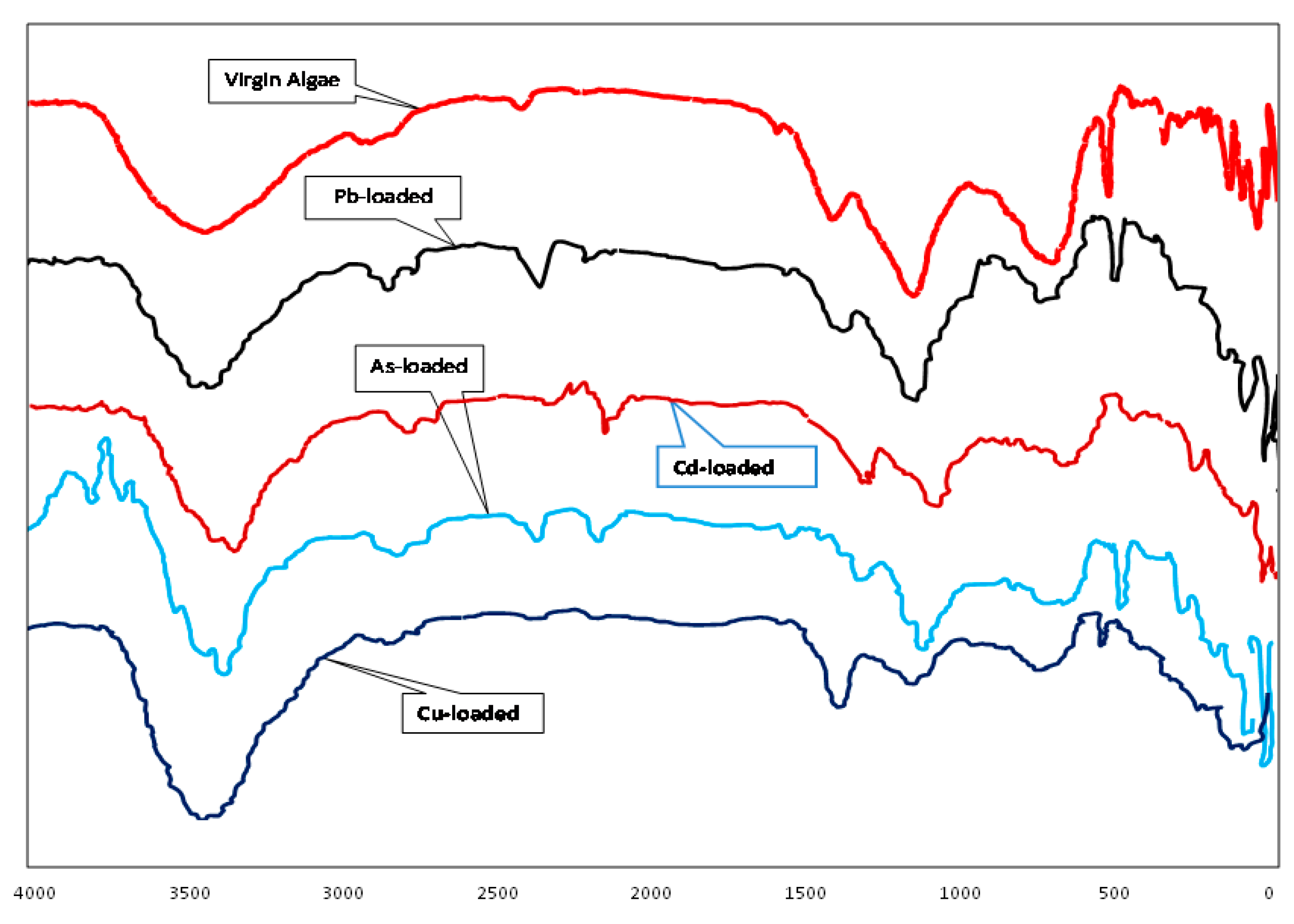
| Natural microbial biosorbent: Biochar from freshwater macroalga Cladophora glomerata | Chemical synthesis: biochar was tested at pH 5, using a dosage of 1 g/L, over a contact time of 3 h under agitation (150 rpm, ~20 °C), pyrolysed at a maximum temperature of 450 °C. The active groups are hydroxyl, silicate, aliphatic, and aromatic. | Chromium (III) Copper (II) Zinc (II) | The removal efficiency for Cr(III) was 89.9%, Cu(II) was 97.1%, and for Zn(II) ions was 93.7%. The adsorption mechanism is ion exchange [130]. |
| Natural-biomass-derived biosorbent: eggshell and Java plum seed | Physical preparation: drying and grinding of eggshells and seeds. The eggshells have calcium carbonate, but the surface is predominantly hydroxyl groups. | Arsenic (III) | As(III) elimination was 78–87% at a pH of 7 using ion exchange as an adsorption mechanism [150]. The pH values for the eggshell were 4.1 and 5.3 for Java plum seed. |
| Natural-biomass-derived biosorbent: Tea waste and pomegranate peel | Physical: drying and grinding. The active group is carboxyl and hydroxyl. Maximum arsenic biosorption occurred at 2 h, following kinetics best described by the Langmuir isotherm and pseudo-second-order model. | Arsenic | Arsenic removal by tea waste was 74%, and pomegranate peel yielded 65%, which shows tea waste to be more effective than pomegranate peel. The adsorption mechanism is physisorption [150]. |
| Natural microbial biosorbent: Marine green algae | Physical: drying and grinding. The active groups are carboxyl and hydroxyl. The alkaline-treated algae biomass was most effective at pH 6 with a contact time of 240 min and a biosorbent dose of 1 g/L. | Cadmium (II) | The biosorption capacity for Cd(II) ions was 53.68%. The mechanism of adsorption is ion exchange [36]. |
| Modified/synthetic biosorbent: Magnetic biochar | Physical through high-temperature pyrolysis. | 4-nitrotolouene nitrotoluene | 80% removal of nitrotoluene [149]. |
| Modified/synthetic Biosorbent: Amine-crosslinked starch (ACS) | The chemical synthesis method was used by crosslinking starch with amine-containing agents to introduce amine groups. | Dyes: Brilliant Blue (BB) and Amaranth (ART) Pharmaceutical: Diclofenac Sodium (DS) | Amount Removed (Adsorption Capacities): BB: 1287.7 mg/g ART: 724.6 mg/g DS: 595.2 mg/g The mechanism of adsorption is physical and ion exchange, where the protonated amine group interacts with negatively charged pollutants (e.g., anionic dyes and pharmaceuticals) [30]. |
| Modified/synthetic and biomass-derived biosorbent: Magnetic biochar prepared from chicken bones | Physical through low-temperature pyrolysis. | Methylene blue Rhodamine B dye Tetracycline | 70% removal of methylene blue and 75% removal of other organic contaminants [149] |
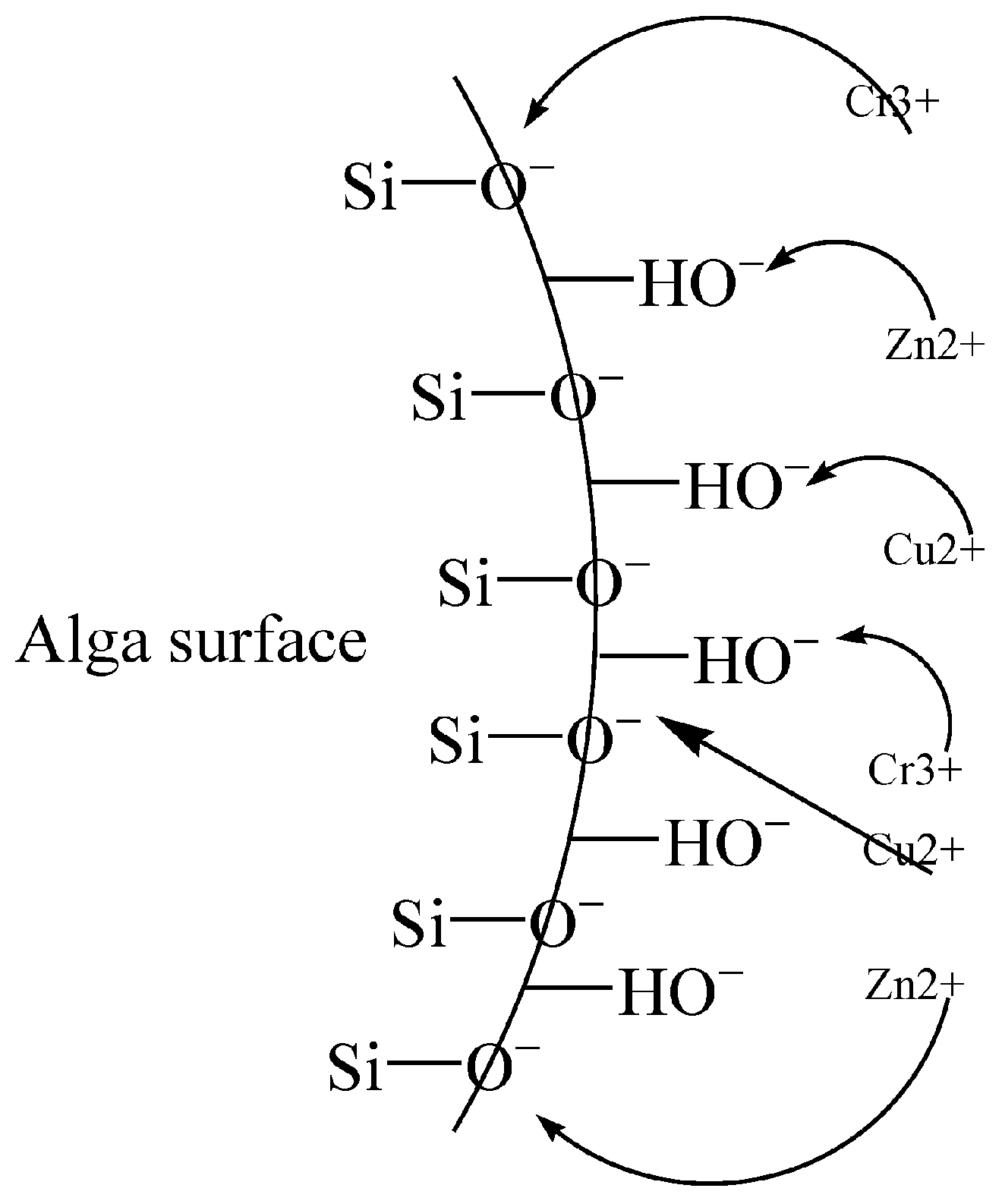
| Natural-microbial biosorbent: Macro algae (Fucus vesiculosus) Natural: Crab shells (Cancer pagurus) Natural-biomass-derived biosorbent: Wood chippings Natural: Iron-rich soil Modified/synthetic material: Biochar (commercial) Modified/synthetic material: Activated Carbon (commercial) | Physical: breaking the particles into smaller ones. Chemical by the use of modifiers and acids to expose chitin in the case of crab shells. Sulfonic groups on the surface of the algae. Amino and hydroxyl groups from the chitin. A phenolic group from the lignin of wood chippings. | Copper Zinc | Copper activated carbon (100%) and biochar (96–99%) removal. Iron-rich soils removed 96%. Crab shells removed 92%. The algae removed 59–67% while wood chipping had a low removal efficiency of 31%. Zinc The highest removal was achieved from activated carbon and biochar, with 100% removal. Crab shells removed 96% Algae removed 39–87% Iron soils had 66% removal, while wood chippings had the lowest removal of 57%. Ion exchange, chelation, precipitation, electrostatic attraction, complexation, and mechanism [35] |
| Modified/synthetic biosorbent: Magnetic activated carbon | Physical synthesis: pyrolysis | Lead (II) Cadmium (II) Chromium (VI) Copper (II) | The removal efficiencies were: Pb 99.4%, Cr 66.7%, Cu 99.1% [149]. |
| Parameter that Requires Further Investigation | Challenge | Proposed Future Direction |
|---|---|---|
| Enhancement of biosorption capacity through modification of the biosorbent | Low biosorption capacities: natural biosorbents show low removal efficiencies; therefore, there is a need for further enhancement to improve their capacity to adsorb pollutants effectively. | Modified/synthetic biosorbents show improved removal efficiencies; therefore, methods should be developed and optimized to modify biosorbents, such as chemical treatments, physical alterations, or the incorporation of functional groups, to significantly increase their biosorption capacity. |
| Assessment of biosorbents under multi-component pollutants | Limited information on biosorption of multi-component pollutants: most studies on natural biosorbents focus on single pollutants, which do not represent real-world scenarios. | Modified/synthetic biosorbents have shown efficiency in multi-component pollutants. These adsorbents have different functional groups, such as -OH, -COOH, -NH2, and -SO3H enhance interactions with pollutants via ion exchange, hydrogen bonding, or complexation. |
| Investigation with real industrial effluents | Limited reports on studies of real wastewater samples: laboratory studies often use synthetic wastewater, which does not accurately reflect industrial conditions. | Perform investigations using industrial effluents to validate the performance of biosorbents in practical applications and ensure their effectiveness in real-world conditions. |
| Regeneration studies | Limited information on regeneration studies of biosorbents: the studies that show their materials being regenerated do not report the removal efficiencies after regeneration. | More studies can be dedicated to the efficiency and cost-effectiveness of regeneration techniques to improve the recyclability of biosorbents. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molebatsi, M.; Nkoane, B.; Keroletswe, N.; Chigome, S.; Kabomo, M.T. The Use of Biosorbents in Water Treatment. Environments 2025, 12, 302. https://doi.org/10.3390/environments12090302
Molebatsi M, Nkoane B, Keroletswe N, Chigome S, Kabomo MT. The Use of Biosorbents in Water Treatment. Environments. 2025; 12(9):302. https://doi.org/10.3390/environments12090302
Chicago/Turabian StyleMolebatsi, Mothusi, Bonang Nkoane, Ngonye Keroletswe, Samuel Chigome, and Moses Tlhabologo Kabomo. 2025. "The Use of Biosorbents in Water Treatment" Environments 12, no. 9: 302. https://doi.org/10.3390/environments12090302
APA StyleMolebatsi, M., Nkoane, B., Keroletswe, N., Chigome, S., & Kabomo, M. T. (2025). The Use of Biosorbents in Water Treatment. Environments, 12(9), 302. https://doi.org/10.3390/environments12090302







