Humidity Impact on Air Quality in Straw- and Reed-Bale Houses
Abstract
1. Introduction
- Indoor climate indicators (relative humidity (RH%) and temperature) were measured from the indoor air of the houses (CO2 levels were also measured), as well as from two different heights (0.2 m and 1.2 m) in the exterior walls;
- Air samples were taken from the bedrooms of the studied houses as well as from the outdoor air, and material samples were collected from the building envelope. Moulds present in both indoor and outdoor air and in the envelope were identified to the genus level;
- The moisture load and mould risk of the exterior walls were assessed.
2. Materials and Methods
3. Results
3.1. Indoor Climate Indicators
3.2. Assessment of Moisture Load and Mould Risk in Exterior Structures
3.3. Abundance and Dynamics of Mould in Indoor and Outdoor Air by Season, and Mould Composition by Genera
3.4. Material Samples from Walls and Identified Mould Genera
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bakkour, A.; Ouldboukhitine, S.E.; Biwole, P.; Amziane, S. Modeling heat and moisture transfer in bio-based wall structures using the finite element method: Application to straw walls in varied climatic conditions. J. Build. Eng. 2025, 104, 112263. [Google Scholar] [CrossRef]
- Barreca, F.; Gabarron, A.M.; Yepes, J.A.F.; Pérez, J.J.P. Innovative use of giant reed and cork residues for panels of buildings in Mediterranean area. Resour. Conserv. Recycl. 2019, 140, 259–266. [Google Scholar] [CrossRef]
- Cicelsky, A.; Meir, I.A.; Peled, A. Novel insulating construction blocks of crop residue (straw) and natural binders. MRS Energy Sustain. 2024, 11, 92–106. [Google Scholar] [CrossRef]
- Shon, C.-S.; Mukashev, T.; Lee, D.; Zhang, D.; Kim, J. Can common reed fiber become an effective construction material? Physical, mechanical, and thermal properties of mortar mixture containing common reed fiber. Sustainability 2019, 11, 903. [Google Scholar] [CrossRef]
- Yang, L.; Wang, S.; Liu, Y.; Wu, Y.; Liu, Y. Towards net zero carbon buildings: Potential use of gypsum-based highland barley straw concrete in solar energy enrichment area of China. Build. Environ. 2025, 271, 112586. [Google Scholar] [CrossRef]
- González, A.D. Energy and carbon embodied in straw and clay wall blocks produced locally in the Andean Patagonia. Energy Build. 2014, 70, 15–22. [Google Scholar] [CrossRef]
- Mutani, G.; Azzolino, C.; Macrì, M.; Mancuso, S. Straw buildings: A good compromise between environmental sustainability and energy-economic savings. Appl. Sci. 2020, 10, 2858. [Google Scholar] [CrossRef]
- Marques, B.; Tadeu, A.; Almeida, J.; António, J.; de Brito, J. Characterisation of sustainable building walls made from rice straw bales. J. Build. Eng. 2020, 28, 101041. [Google Scholar] [CrossRef]
- Peng, H.; Walker, P.; Maskell, D.; Jones, B. Structural characteristics of load bearing straw bale Walls. Construct. Build. Mater. 2021, 287, 122911. [Google Scholar] [CrossRef]
- Bouasker, M.; Belayachi, N.; Hoxha, D.; Al-Mukhtar, M. Physical characterization of natural straw fibers as aggregates for construction materials applications. Materials 2014, 7, 3034–3038. [Google Scholar] [CrossRef]
- Henderson, K. Achieving legitimacy: Visual discourses in engineering design and green building code development. Build. Res. Inf. 2007, 35, 6–17. [Google Scholar] [CrossRef]
- King, B. Design of Straw Bale Buildings—The State of the Art; Green Building Press: San Rafael, CA, USA, 2006; p. 296. ISBN 0-9764911-1-7. [Google Scholar]
- D’Alessandro, F.; Bianchi, F.; Baldinelli, G.; Rotil, A.; Schiavoni, S. Straw bale constructions: Laboratory, in field and numerical assessment of energy and environmental performance. J. Build. Eng. 2017, 11, 56–68. [Google Scholar] [CrossRef]
- Peterková, J.; Zach, J.; Novák, V.; Korjenic, A.; Sulejmanovski, A.; Sesto, E. Optimizing indoor microclimate and thermal comfort through sorptive active elements: Stabilizing humidity for healthier living spaces. Buildings 2024, 14, 3836. [Google Scholar] [CrossRef]
- Laborel-Préneron, A.; Ouédraogo, K.; Simons, A.; Labat, M.; Bertron, A.; Magniont, C.; Roques, C.; Roux, C.; Aubert, J.-E. Laboratory test to assess sensitivity of bio-based earth materials to fungal growth. Build. Environ. 2018, 142, 11–21. [Google Scholar] [CrossRef]
- Meda, M.; Gentry, V.; Preece, E.; Nagy, C.; Kumari, P.; Wilson, P.; Hoffman, P. Assessment of mould remediation in a healthcare setting following extensive flooding. J. Hospital. Infect. 2024, 146, 1–9. [Google Scholar] [CrossRef]
- Lai, Q.; Liu, H.; Feng, C.; Gao, S. Comparison of mould experiments on building materials: A methodological review. Build. Environ. 2024, 261, 111725. [Google Scholar] [CrossRef]
- Quintero, O.; Allard, L.; Ho, D. Invasive mold infection of the gastrointestinal tract: A case series of 22 immunocompromised patients from a single academic center. Med. Mycol. 2022, 60, myac007. [Google Scholar] [CrossRef]
- Portnoy, J.M.; Kwak, K.; Dowling, P.; Vanosdol, T.; Barnes, C. Health effects of indoor fungi. Ann. Allergy Asthma Immunol. 2005, 94, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Al-Jumeily, D.; Hashim, K.; Alkaddar, R.; Al-Tufaily, M.; Lunn, J. Sustainable and environmental friendly ancient reed houses (inspired by the past to motivate the future). In Proceedings of the 11th International Conference on Developments in eSystems Engineering (DeSE), Cambridge, UK, 2–5 September 2018; pp. 214–219. [Google Scholar] [CrossRef]
- Raamets, J.; Kutti, S.; Ruus, A.; Ivask, M. Assessment of indoor air in Estonian straw bale and reed houses. WIT Trans. Ecol. Environ. 2017, 211, 193–196. [Google Scholar] [CrossRef][Green Version]
- Brischke, C.; Hanske, M. Durability of untreated and thermally modified reed (Phragmites australis) against brown, white and soft rot causing fungi. Ind. Crops Prod. 2016, 91, 49–55. [Google Scholar] [CrossRef]
- Johansson, P.; Ekstrand-Tobin, A.; Svensson, T.; Bok, G. Laboratory study to determine the critical moisture level for mould growth on building materials. Int. Biodeterior. Biodegrad. 2012, 73, 23–32. [Google Scholar] [CrossRef]
- ISO 16000-18:2011; Indoor Air, Part 18: Detection and Enumeration of Moulds—Sampling by Impaction. International Organization for Standardization: Geneva, Switzerland, 2011; Edition 1. Available online: https://www.iso.org/standard/44325.html (accessed on 10 August 2025).
- EVS-ISO 16000-17:2012; Indoor Air, Part 17: Detection and Enumeration of Moulds—Culture-Based Method (ISO 16000-17:2008). Estonian Centre for Standardisation and Accreditation: Tallinn, Estonia, 2012. Available online: https://www.evs.ee/en/evs-iso-16000-17-2012 (accessed on 10 August 2025).
- Domsch, K.H.; Gams, W.; Anderson, T.H. Compendium of Soil Fungi; Academic Press: London, UK, 1980; Volume 1, p. 860. [Google Scholar]
- Kilch, M.A. A Laboratory Guide to Common Aspergillus Species and Their Teleomorphs; Commonwealth Scientific and Industrial Research Organization, Division of Food Processing: North Ryde, Australia, 1988; p. 116. [Google Scholar]
- Bergey, D.H.; Holt, J.G. Bergey’s Manual of Determinative Bacteriology; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2000; p. 787. [Google Scholar]
- Larone, D.H. Medically Important Fungi. A Guide to Identification, 4th ed.; ASM Press, American Society for Microbiology: Washington, DC, USA, 2002; p. 409. [Google Scholar]
- Winn, W.C.; Koneman, E.W. Koneman’s Color Atlas and Textbook of Diagnostic Microbiology, 6th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2006; p. 1565. [Google Scholar]
- Watanabe, T. Pictorial Atlas of Soil and Seed Fungi, Morphologies of Cultured Fungi and Key to Species; CRC Press: Boca Raton, FL, USA, 2010; p. 426. Available online: https://doi.org/10.1201/EBK1439804193 (accessed on 10 August 2025).
- Caillaud, D.; Leynaert, B.; Keirsbulck, M.; Nadif, R. Indoor mould exposure, asthma and rhinitis: Findings from systematic reviews and recent longitudinal studies. Eur. Respir. Rev. 2018, 27, 170137. [Google Scholar] [CrossRef]
- Raamets, J.; Kutti, S.; Vettik, A.; Ilustrumm, K.; Rist, T.; Ivask, M. The antimicrobial effect of three different chemicals for the treatment of straw bales used in housing projects. In Proceedings of the International Conference on Sustainable Housing Planning, Management and Usability (ICSM), Porto, Portugal, 16–18 November 2016; Green Lines Institute for Sustainable Development: Barcelos, Portugal, 2016; pp. 537–545. [Google Scholar]
- Keskkonnaagentuur (Estonian Environment Agency). Ilmaülevaated (Weather Observations). Available online: http://www.ilmateenistus.ee/kliima/ulevaated/ (accessed on 30 January 2020). (In Estonian).
- EVS-EN ISO 13788:2012; Hygrothermal Performance of Building Components and Building Elements—Internal Surface Temperature to Avoid Critical Surface Humidity and Interstitial Condensation—Calculation Methods (ISO 13788:2012). Estonian Centre for Standardisation and Accreditation: Tallinn, Estonia, 2012. Available online: https://www.evs.ee/en/evs-en-iso-13788-2012 (accessed on 10 August 2025).
- Hukka, A.; Viitanen, H. A mathematical model of mould growth on wooden material. Wood Sci. Technol. 1999, 33, 475–485. [Google Scholar] [CrossRef]
- Lawrence, M.; Heath, A.; Walker, P. Determining moisture levels in straw bale. Constr. Const. Build. Mat. 2009, 23, 2763–2768. [Google Scholar] [CrossRef]
- EVS-EN 16798-1:2019; Energy Performance of Buildings—Ventilation for Buildings—Part 1: Indoor Environmental Input Parameters for Design and Assessment of Energy Performance of Buildings Addressing Indoor Air Quality, Thermal Environment, Lighting and Acoustics—Module M1-6. Eesti Standardimis-ja Akrediteerimiskeskus MTÜ (Estonian Centre for Standardisation and Accreditation): Tallinn, Estonia, 2019. Available online: https://www.evs.ee/en/evs-en-16798-1-2019 (accessed on 10 August 2025).
- Vecherin, S.; Joyner, M.; Smith, M.; Linkov, I. Risk assessment of mold growth across the US due to weather variations. Build. Environ. 2024, 256, 111498. [Google Scholar] [CrossRef]
- Jyotshna, M.; Helmut, B. Bioaerosols in indoor environment—A review with special reference to residential and occupational locations. Open Environ. Biol. Mon. J. 2011, 4, 83–96. [Google Scholar] [CrossRef]
- Nevalainen, A.; Morawaska, L. (Eds.) . Biological Agents in Indoor Environments. Assessment of Health Risks. Work Conducted by a WHO Expert Group Between 2000–2003; QUT: Brisbane, Australia, 2009; p. 201. Available online: https://s3-eu-west-1.amazonaws.com/awarticles/BIOLOGICAL_AGENTS_2009.pdf (accessed on 10 August 2025).
- Suomaa, L.; Pekuri, H.M. Kosteusvauriot Työpaikoilla. Kosteusvauriotyöryhmän Muistio; Sosiaali-ja Terveysministeriön Selvityksiä (Reports of the Ministry of Social Affairs and Health): Yliopistopaino, Helsinki, Finland, 2009; Volume 18, p. 82. (In Finnish) [Google Scholar]
- Hoseini, M.; Jabbari, H.; Naddafi, R.; Nabizadeh, M.; Rahbar, M.; Yunesian, M.; Jaafari, J. Concentration and distribution characteristics of airborne fungi in indoor and outdoor air of Tehran subway stations. Aerobiologia 2012, 29, 355–363. [Google Scholar] [CrossRef]
- Kalwasinska, A.; Burkowska, A.; Wilk, I. Microbial air contamination in indoor environment of a University library. Ann. Agric. Environ. Med. 2012, 19, 25–29. [Google Scholar] [PubMed]
- Raamets, J.; Ruus, A.; Ivask, M. Assessment of indoor air quality and hygrothermal conditions of boarders during autumn, winter and spring in two of Estonian straw-bale houses. In Cold Climate HVAC 2018, Proceedings of the CCC 2018, Kiruna, Sweden, 12–15 March 2018; Johansson, D., Bagge, H., Wahlström, Å., Eds.; Springer Proceedings in Energy; Springer: Cham, Switzerland, 2019; pp. 815–823. [Google Scholar] [CrossRef]
- Hameed, A.A.A.; Khoder, M.I.; Ibrahim, Y.H.; Saeed, Y.; Osman, M.E.; Ghanem, S. Study on some factors affecting survivability of airborne fungi. Sci. Total Environ. 2012, 414, 696–700. [Google Scholar] [CrossRef]
- Awad, A.H.; Saeed, Y.; Hassan, Y.; Fawzy, Y.; Osman, M. Air microbial quality in certain public buildings, Egypt: A comparative study. Atmos. Pollut. Res. 2018, 9, 617–626. [Google Scholar] [CrossRef]
- Medrela-Kuder, E. Seasonal variations in the occurrence of culturable airborne fungi in outdoor and indoor air in Craców. Int. Biodeterior. Biodegrad. 2003, 52, 203–205. [Google Scholar] [CrossRef]
- Haas, D.; Habib, J.; Galler, H.; Buzina, W.; Schlacher, R.; Marth, E.; Reinthaler, F. Assessment of indoor air in Austrian apartments with and without visible mould growth. Atmos. Environ. 2007, 41, 5192–5201. [Google Scholar] [CrossRef]
- Frankel, M.; Bekö, G.; Timm, M.; Gustavsen, S.; Hansen, E.W.; Madsen, A.M. Seasonal variations of indoor microbial exposures and their relation to temperature, relative humidity, and air exchange rate. Appl. Environ. Microbiol. 2012, 78, 8289–8297. [Google Scholar] [CrossRef] [PubMed]
- Kempe, M.; Haverinen-Shaughnessy, U.; Tuoresmäki, P.; Täubel, M. Effects of energy retrofits on the indoor microbiota in Northern European apartments. Int. J. Hyg. Environ. Health 2025, 265, 114538. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.; Yang, J.; Lou, H.; Luo, M.; Yang, Q. Potential respiratory hazards of fungal exposure in the residential indoor environment: A systematic review. Atmos. Pollut. Res. 2025, 16, 102634. [Google Scholar] [CrossRef]
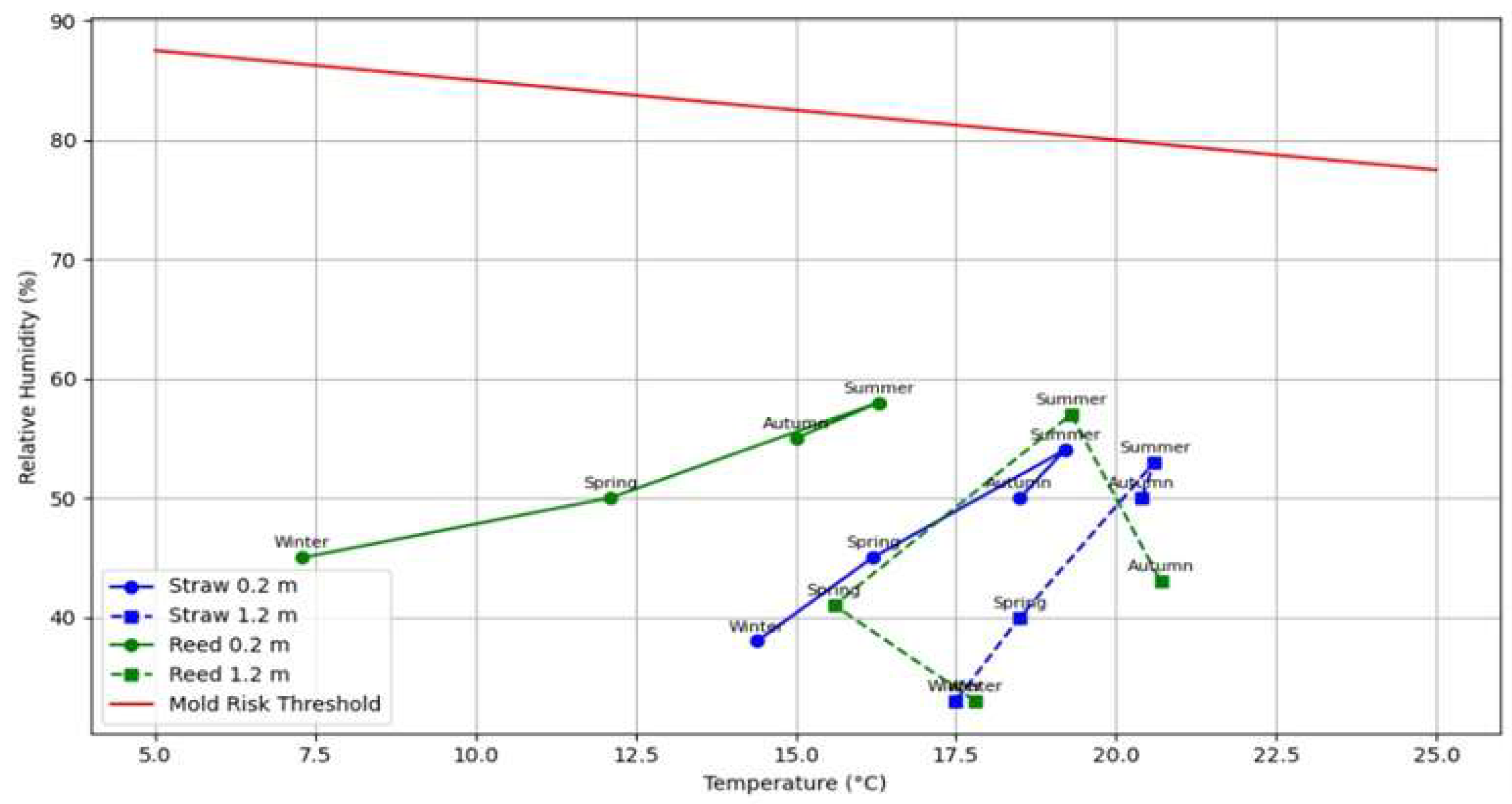
| Winter | Spring | Summer | Autumn | |||||
|---|---|---|---|---|---|---|---|---|
| Straw | Reed | Straw | Reed | Straw | Reed | Straw | Reed | |
| Temperature, °C at 1.2 m | 17 ± 3 | 16 ± 5 | 18 ± 2 | 16 ± 2 | 21 ± 1 | 19 ± 1 | 18 ± 1 | 17 ± 2 |
| Temperature (°C) at 0.2 m | 14 ± 3 | 7 ± 3 | 16 ± 2 | 11 ± 4 | 19 ± 1 | 16 ± 2 | 17 ± 2 | 14 ± 3 |
| RH (%) at 1.2 m | 33 ± 12 | 33 ± 14 | 41 ± 7 | 47 ± 2 | 53 ± 7 | 57 ± 1 | 41 ± 4 | 45 ± 4 |
| RH (%) at 0.2 m | 38 ± 4 | 45 ± 6 | 42 ± 3 | 47 ± 1 | 54 ± 2 | 58 ± 1 | 42 ± 2 | 51 ± 2 |
| Sample | Winter | Spring | Summer | Autumn | ||||
|---|---|---|---|---|---|---|---|---|
| Indoors | Outdoors | Indoors | Outdoors | Indoors | Outdoors | Indoors | Outdoors | |
| Straw (MEA) | 149 ± 29 | 94 ± 19 | 298 ± 100 | 197 ± 43 | 537 ± 102 | 289 ± 32 | 307 ± 99 | 168 ± 34 |
| Reed (MEA) | 380 ± 136 | 118 ± 15 | 518 ± 145 | 198 ± 48 | 858 ± 106 | 353 ± 41 | 548 ± 155 | 212 ± 34 |
| Straw (DG18) | 14 ± 8 | 18 ± 7 | 29 ± 9 | 30 ± 9 | 46 ± 12 | 50 ± 11 | 20 ± 11 | 22 ± 10 |
| Reed (DG18) | 22 ± 13 | 24 ± 9 | 23 ± 9 | 27 ± 11 | 36 ± 11 | 45 ± 12 | 22 ± 10 | 23 ± 9 |
| Winter | Spring | Summer | Autumn | |||||
|---|---|---|---|---|---|---|---|---|
| Straw | Reed | Straw | Reed | Straw | Reed | Straw | Reed | |
| Alternaria | 1 (1) | 4 (1) | 9 (3) | 16 (3) | 32 (6) | 43 (5) | 25 (8) | 49 (9) |
| Aspergillus | 25 (17) | 72 (19) | 24 (8) | 36 (7) | 11 (2) | 9 (1) | 71 (23) | 110 (20) |
| Cladosporium | 1 (1) | 23 (6) | 235 (79) | 420 (81) | 451 (84) | 738 (86) | 92 (30) | 142 (26) |
| Penicillium | 110 (74) | 266 (70) | 24 (8) | 36 (7) | 38 (7) | 51 (6) | 95 (31) | 203 (37) |
| Others | 10 (7) | 15 (4) | 6 (2) | 12 (2) | 5 (1) | 15 (2) | 25 (8) | 44 (8) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raamets, J.; Nei, L.; Ruus, A.; Ivask, M.; Muoni, K. Humidity Impact on Air Quality in Straw- and Reed-Bale Houses. Environments 2025, 12, 297. https://doi.org/10.3390/environments12090297
Raamets J, Nei L, Ruus A, Ivask M, Muoni K. Humidity Impact on Air Quality in Straw- and Reed-Bale Houses. Environments. 2025; 12(9):297. https://doi.org/10.3390/environments12090297
Chicago/Turabian StyleRaamets, Jane, Lembit Nei, Aime Ruus, Mari Ivask, and Karin Muoni. 2025. "Humidity Impact on Air Quality in Straw- and Reed-Bale Houses" Environments 12, no. 9: 297. https://doi.org/10.3390/environments12090297
APA StyleRaamets, J., Nei, L., Ruus, A., Ivask, M., & Muoni, K. (2025). Humidity Impact on Air Quality in Straw- and Reed-Bale Houses. Environments, 12(9), 297. https://doi.org/10.3390/environments12090297






