The Brain Resting-State Functional Connectivity Underlying Violence Proneness: Is It a Reliable Marker for Neurocriminology? A Systematic Review
Abstract
1. Introduction
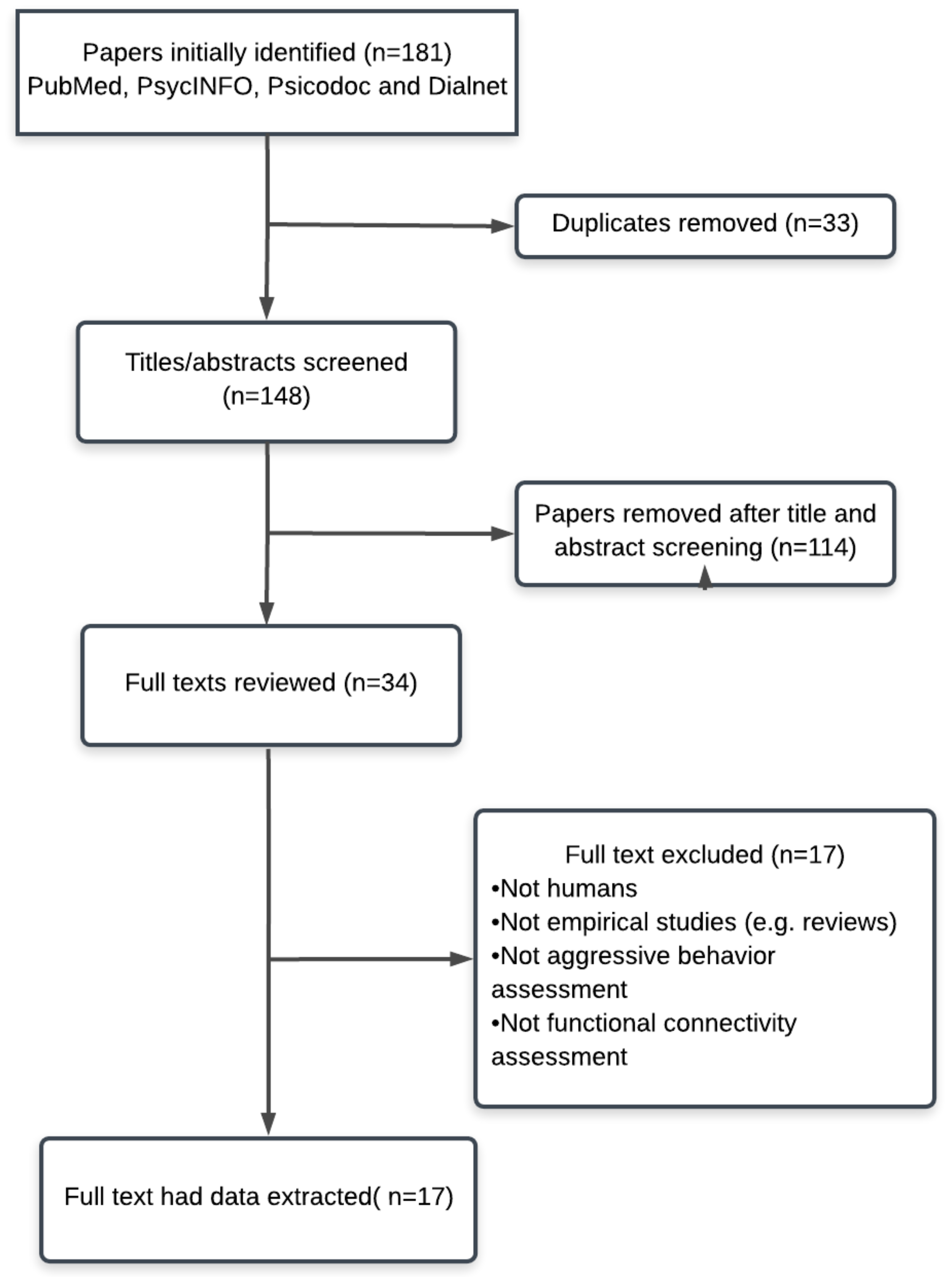
2. Methods
Search Strategy
3. Results
3.1. Normative Population (Self-Reported Aggression)
3.2. Self-Reported Aggression Mediated by Genetic Markers
3.3. Mental Disorders (Self-Reported Aggression)
3.4. Laboratory Assessment of Aggression
3.4.1. Non-Violent Groups
3.4.2. Violent Group
4. Violent Populations
5. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Glenn, A.L.; Raine, A. Neurocriminology: Implications for the punishment, prediction and prevention of criminal behaviour. Nat. Rev. Neurosci. 2014, 15, 54. [Google Scholar] [CrossRef] [PubMed]
- Moya-Albiol, L.; Sariñana-González, S.; Vitoria-Estruch, S.; Romero-Martínez, Á. La neurocriminología como disciplina aplicada emergente. Vox Juris 2017, 33, 6. [Google Scholar] [CrossRef]
- Nordstrom, B.R.; Gao, Y.; Glenn, A.L.; Peskin, M.; Rudo-Hutt, A.S.; Schug, R.A.; Yang, Y.; Raine, A. Neurocriminology. Adv. Genet. 2011, 75, 255–283. [Google Scholar] [PubMed]
- Menon, R.S.; Gati, J.S.; Goodyear, B.G.; Luknowsky, D.C.; Thomas, C.G. Spatial and temporal resolution of functional magnetic resonance imaging. Biochem. Cell Biol. 1998, 76, 560–571. [Google Scholar] [CrossRef] [PubMed]
- Zani, A.; Biella, G.; Proverbio, A.M. Brain imaging techniques: Invasiveness and spatial and temporal resolution. In The Cognitive Electrophysiology of Mind and Brain; Elsevier: Amsterdam, The Netherlands, 2003; pp. 417–422. [Google Scholar]
- Hare, R.D.; Smith, A.M.; Forster, B.B.; MacKay, A.L.; Whittall, K.P.; Kiehl, K.A.; Smith, A.M.; Hare, R.D.; Liddle, P.F. Functional magnetic resonance imaging: The basics of blood-oxygen-level dependent (BOLD) imaging. Can. Assoc. Radiol. J. 1998, 49, 320–329. [Google Scholar]
- Ekstrom, A. How and when the fMRI BOLD signal relates to underlying neural activity: The danger in dissociation. Brain Res. Rev. 2010, 62, 233–244. [Google Scholar] [CrossRef] [PubMed]
- Raine, A.; Yang, Y. Neural foundations to moral reasoning and antisocial behavior. Soc. Cogn. Affect. Neurosci. 2006, 1, 203–213. [Google Scholar] [CrossRef]
- Leutgeb, V.; Leitner, M.; Wabnegger, A.; Klug, D.; Scharmüller, W.; Zussner, T.; Schienle, A. Brain abnormalities in high-risk violent offenders and their association with psychopathic traits and criminal recidivism. Neuroscience 2015, 308, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Romero-Martínez, Á.; Moya-Albiol, L. Neuropsychology of perpetrators of domestic violence: The role of traumatic brain injury and alcohol abuse and/or dependence. Rev. Neurol. 2013, 57, 515–522. [Google Scholar]
- Romero-Martínez, A.; Moya-Albiol, L. Neuropsychological impairments associated with the relation between cocaine abuse and violence: Neurological facilitation mechanisms. Adicciones 2015, 27, 64–74. [Google Scholar] [CrossRef]
- Lundwall, R.A.; Stephenson, K.G.; Neeley-Tass, E.S.; Cox, J.C.; South, M.; Bigler, E.D.; Anderberg, E.; Prigge, M.D.; Hansen, B.D.; Lainhart, J.E.; et al. Relationship between brain stem volume and aggression in children diagnosed with autism spectrum disorder. Res. Autism Spectr. Disord. 2017, 34, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Glenn, A.L.; Raine, A.; Yaralian, P.S.; Yang, Y. Increased volume of the striatum in psychopathic individuals. Biol. Psychiatry 2010, 67, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Romero-Martínez, Á.; Moya-Albiol, L. ¿Facilitan los esteroides anabolizantes-androgénicos la expresión de la violencia? Rev. Esp. Drogodepend. 2015, 40, 12–26. [Google Scholar]
- Siever, L.J. Neurobiology of aggression and violence. Am. J. Psychiatry 2008, 165, 429–442. [Google Scholar] [CrossRef]
- Glenn, A.L.; Raine, A. Psychopathy and instrumental aggression: Evolutionary, neurobiological, and legal perspectives. Int. J. Law Psychiatry 2009, 32, 253–258. [Google Scholar] [CrossRef]
- Raine, A.; Meloy, J.R.; Bihrle, S.; Stoddard, J.; LaCasse, L.; Buchsbaum, M.S. Reduced prefrontal and increased subcortical brain functioning assessed using positron emission tomography in predatory and affective murderers. Behav. Sci. Law 1998, 16, 319–332. [Google Scholar] [CrossRef]
- Blair, R.J.R. The amygdala and ventromedial prefrontal cortex in morality and psychopathy. Trends Cogn. Sci. 2007, 11, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Blair, R. Dysfunctions of medial and lateral orbitofrontal cortex in psychopathy. Ann. N. Y. Acad. Sci. 2007, 1121, 461–479. [Google Scholar] [CrossRef]
- Lee, M.H.; Smyser, C.D.; Shimony, J.S. Resting-state fMRI: A review of methods and clinical applications. Am. J. Neuroradiol. 2013, 34, 1866–1872. [Google Scholar] [CrossRef]
- Di, X.; Biswal, B.B. Dynamic brain functional connectivity modulated by resting-state networks. Brain Struct. Funct. 2015, 220, 37–46. [Google Scholar] [CrossRef]
- Smitha, K.; Akhil Raja, K.; Arun, K.; Rajesh, P.; Thomas, B.; Kapilamoorthy, T.; Kesavadas, C. Resting state fMRI: A review on methods in resting state connectivity analysis and resting state networks. Neuroradiol. J. 2017, 30, 305–317. [Google Scholar] [CrossRef] [PubMed]
- Binder, J.R.; Frost, J.A.; Hammeke, T.A.; Bellgowan, P.; Rao, S.M.; Cox, R.W. Conceptual processing during the conscious resting state: A functional MRI study. J. Cogn. Neurosci. 1999, 11, 80–93. [Google Scholar] [CrossRef] [PubMed]
- Mazoyer, B.; Zago, L.; Mellet, E.; Bricogne, S.; Etard, O.; Houdé, O.; Crivello, F.; Joliot, M.; Petit, L.; Tzourio-Mazoyer, N. Cortical networks for working memory and executive functions sustain the conscious resting state in man. Brain Res. Bull. 2001, 54, 287–298. [Google Scholar] [CrossRef]
- Shulman, G.L.; Fiez, J.A.; Corbetta, M.; Buckner, R.L.; Miezin, F.M.; Raichle, M.E.; Petersen, S.E. Common blood flow changes across visual tasks: II. Decreases in cerebral cortex. J. Cogn. Neurosci. 1997, 9, 648–663. [Google Scholar] [CrossRef] [PubMed]
- Vatansever, D.; Menon, D.K.; Manktelow, A.E.; Sahakian, B.J.; Stamatakis, E.A. Default mode network connectivity during task execution. Neuroimage 2015, 122, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Greicius, M.D.; Krasnow, B.; Reiss, A.L.; Menon, V. Functional connectivity in the resting brain: A network analysis of the default mode hypothesis. Proc. Natl. Acad. Sci. USA 2003, 100, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Mars, R.B.; Neubert, F.; Noonan, M.P.; Sallet, J.; Toni, I.; Rushworth, M.F. On the relationship between the “default mode network” and the “social brain”. Front. Hum. Neurosci. 2012, 6, 189. [Google Scholar] [CrossRef] [PubMed]
- Smith, V.; Mitchell, D.J.; Duncan, J. Role of the default mode network in cognitive transitions. bioRxiv 2018, 295683. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Kim, S.; Kim, H.E.; Han, K.; Jeong, B.; Kim, J.; Namkoong, K.; Kim, J.W. Altered Functional Connectivity of the Default Mode Network in Low-Empathy Subjects. Yonsei Med. J. 2017, 58, 1061–1065. [Google Scholar] [CrossRef]
- Beaty, R.E.; Kaufman, S.B.; Benedek, M.; Jung, R.E.; Kenett, Y.N.; Jauk, E.; Neubauer, A.C.; Silvia, P.J. Personality and complex brain networks: The role of openness to experience in default network efficiency. Hum. Brain Mapp. 2016, 37, 773–779. [Google Scholar] [CrossRef]
- Zhao, J.; Tomasi, D.; Wiers, C.E.; Shokri-Kojori, E.; Demiral, Ş.B.; Zhang, Y.; Volkow, N.D.; Wang, G. Correlation between Traits of Emotion-Based Impulsivity and Intrinsic Default-Mode Network Activity. Neural Plast. 2017, 2017, 9297621. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef]
- Park, A.T.; Leonard, J.A.; Saxler, P.; Cyr, A.B.; Gabrieli, J.D.E.; Mackey, A.P. Amygdala-medial prefrontal connectivity relates to stress and mental health in early childhood. Soc. Cogn. Affect. Neurosci. 2018. [Google Scholar] [CrossRef]
- Fulwiler, C.E.; King, J.A.; Zhang, N. Amygdala-orbitofrontal resting-state functional connectivity is associated with trait anger. Neuroreport 2012, 23, 606–610. [Google Scholar] [CrossRef] [PubMed]
- Abram, S.V.; Wisner, K.M.; Grazioplene, R.G.; Krueger, R.F.; MacDonald, A.W.; DeYoung, C.G. Functional coherence of insula networks is associated with externalizing behavior. J. Abnorm. Psychol. 2015, 124, 1079–1091. [Google Scholar] [CrossRef] [PubMed]
- Klasen, M.; Wolf, D.; Eisner, P.D.; Habel, U.; Repple, J.; Vernaleken, I.; Schlüter, T.; Eggermann, T.; Zerres, K.; Zepf, F.D.; et al. Neural networks underlying trait aggression depend on MAOA gene alleles. Brain Struct. Funct. 2018, 223, 873–881. [Google Scholar] [CrossRef] [PubMed]
- Kolla, N.J.; Dunlop, K.; Meyer, J.H.; Downar, J. Corticostriatal connectivity in antisocial personality disorder by MAO-A genotype and its relationship to aggressive behavior. Int. J. Neuropsychopharmacol. 2018. [Google Scholar] [CrossRef]
- Hoptman, M.J.; D’Angelo, D.; Catalano, D.; Mauro, C.J.; Shehzad, Z.E.; Kelly, A.M.; Castellanos, F.X.; Javitt, D.C.; Milham, M.P. Amygdalofrontal functional disconnectivity and aggression in schizophrenia. Schizophr. Bull. 2010, 36, 1020–1028. [Google Scholar] [CrossRef] [PubMed]
- Wagner, G.; Krause-Utz, A.; de la Cruz, F.; Schumann, A.; Schmahl, C.; Bar, K.J. Resting-state functional connectivity of neurotransmitter producing sites in female patients with borderline personality disorder. Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 83, 118–126. [Google Scholar] [CrossRef]
- Hasler, R.; Preti, M.; Meskaldji, D.; Prados, J.; Adouan, W.; Rodriguez, C.; Toma, S.; Hiller, N.; Ismaili, T.; Hofmeister, J.; et al. Inter-hemispherical asymmetry in default-mode functional connectivity and BAIAP2 gene are associated with anger expression in ADHD adults. Psychiatry Res. Neuroimaging 2017, 269, 54–61. [Google Scholar] [CrossRef]
- McGlade, E.; Rogowska, J.; Yurgelun-Todd, D. Sex differences in orbitofrontal connectivity in male and female veterans with TBI. Brain Imaging Behav. 2015, 9, 535–549. [Google Scholar] [CrossRef] [PubMed]
- Goswami, R.; Dufort, P.; Tartaglia, M.C.; Green, R.E.; Crawley, A.; Tator, C.H.; Wennberg, R.; Mikulis, D.J.; Keightley, M.; Davis, K.D.; et al. Frontotemporal correlates of impulsivity and machine learning in retired professional athletes with a history of multiple concussions. Brain Struct. Funct. 2016, 221, 1911–1925. [Google Scholar] [CrossRef] [PubMed]
- Dailey, N.S.; Smith, R.; Vanuk, J.R.; Raikes, A.C.; Killgore, W.D.S. Resting-state functional connectivity as a biomarker of aggression in mild traumatic brain injury. Neuroreport 2018, 29, 1413–1417. [Google Scholar] [CrossRef] [PubMed]
- Gilam, G.; Maron-Katz, A.; Kliper, E.; Lin, T.; Fruchter, E.; Shamir, R.; Hendler, T.; et al. Tracing the Neural Carryover Effects of Interpersonal Anger on Resting-State fMRI in Men and Their Relation to Traumatic Stress Symptoms in a Subsample of Soldiers. Front. Behav. Neurosci. 2017, 11, 252. [Google Scholar] [CrossRef] [PubMed]
- Buades-Rotger, M.; Engelke, C.; Kramer, U.M. Trait and state patterns of basolateral amygdala connectivity at rest are related to endogenous testosterone and aggression in healthy young women. Brain Imaging Behav. 2018. [Google Scholar] [CrossRef] [PubMed]
- Siep, N.; Tonnaer, F.; van de Ven, V.; Arntz, A.; Raine, A.; Cima, M. Anger provocation increases limbic and decreases medial prefrontal cortex connectivity with the left amygdala in reactive aggressive violent offenders. Brain Imaging Behav. 2018, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Zhou, J.; Liu, C.; Witt, K.; Zhang, Y.; Jing, B.; Li, C.; Wang, X.; Li, L. Regional homogeneity of resting-state brain abnormalities in violent juvenile offenders: A biomarker of brain immaturity? J. Neuropsychiatry Clin. Neurosci. 2015, 27, 27–32. [Google Scholar] [CrossRef]
- Leutgeb, V.; Wabnegger, A.; Leitner, M.; Zussner, T.; Scharmüller, W.; Klug, D.; Schienle, A. Altered cerebellar-amygdala connectivity in violent offenders: A resting-state fMRI study. Neurosci. Lett. 2016, 610, 160–164. [Google Scholar] [CrossRef]
- Varkevisser, T.; Gladwin, T.E.; Heesink, L.; van Honk, J.; Geuze, E. Resting-state functional connectivity in combat veterans suffering from impulsive aggression. Soc. Cogn. Affect. Neurosci. 2017, 12, 1881–1889. [Google Scholar] [CrossRef]
- Tikasz, A.; Potvin, S.; Lungu, O.; Joyal, C.C.; Hodgins, S.; Mendrek, A.; Dumais, A. Anterior cingulate hyperactivations during negative emotion processing among men with schizophrenia and a history of violent behavior. Neuropsychiatr. Dis. Treat. 2016, 12, 1397–1410. [Google Scholar]
- Hoptman, M.J.; Antonius, D.; Mauro, C.J.; Parker, E.M.; Javitt, D.C. Cortical thinning, functional connectivity, and mood-related impulsivity in schizophrenia: Relationship to aggressive attitudes and behavior. Am. J. Psychiatry 2014, 171, 939–948. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhu, W.; Xiao, M.; Zhang, Q.; Zhao, Y.; Zhang, H.; et al. Hostile attribution bias mediates the relationship between structural variations in the left middle frontal gyrus and trait angry rumination. Front. Psychol. 2018, 9, 526. [Google Scholar] [CrossRef] [PubMed]
- Anestis, M.D.; Anestis, J.C.; Selby, E.A.; Joiner, T.E. Anger rumination across forms of aggression. Personal. Individ. Differ. 2009, 46, 192–196. [Google Scholar] [CrossRef]
- Wang, X.; Yang, L.; Yang, J.; Gao, L.; Zhao, F.; Xie, X.; Lei, L. Trait anger and aggression: A moderated mediation model of anger rumination and moral disengagement. Personal. Individ. Differ. 2018, 125, 44–49. [Google Scholar] [CrossRef]
- Wrangham, R.W. Two types of aggression in human evolution. Proc. Natl. Acad. Sci. USA 2018, 115, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Moya-Albiol, L.; Herrero, N.; Bernal, M.C. The neural bases of empathy. Rev. Neurol. 2010, 50, 89–100. [Google Scholar] [PubMed]
- Takeuchi, H.; Taki, Y.; Nouchi, R.; Sekiguchi, A.; Hashizume, H.; Sassa, Y.; Kotozaki, Y.; Miyauchi, C.M.; Yokoyama, R.; Iizuka, K.; et al. Association between resting-state functional connectivity and empathizing/systemizing. Neuroimage 2014, 99, 312–322. [Google Scholar] [CrossRef]
- Cox, C.L.; Uddin, L.Q.; Di Martino, A.; Castellanos, F.X.; Milham, M.P.; Kelly, C. The balance between feeling and knowing: Affective and cognitive empathy are reflected in the brain’s intrinsic functional dynamics. Soc. Cogn. Affect. Neurosci. 2011, 7, 727–737. [Google Scholar] [CrossRef] [PubMed]
- Romero-Martínez, Á.; Lila, M.; Martínez, M.; Pedrón-Rico, V.; Moya-Albiol, L. Improvements in empathy and cognitive flexibility after court-mandated intervention program in intimate partner violence perpetrators: The role of alcohol abuse. Int. J. Environ. Res. Public Health 2016, 13, 394. [Google Scholar] [CrossRef]
| Authors | Sample Characteristics | Age | Gender | Education | Drug Use | Handedness (Right/Left) | Violent Behavior Assessment | Methods of Analysis |
|---|---|---|---|---|---|---|---|---|
| Park et al. (2018) [34] | Healthy young children (n = 79) | 6.06 ± 0.96 | 49% ♂ 51% ♀ | - | - | - | Child Behavior Checklist | Seed-based |
| Fulwiler et al. (2012) [35] | Healthy males (n = 16) | 34 ± 14.42 | ♂ | - | No current drug use | Right-handed | Spielberger State–Trait Anger Expression Inventory-2 | ROI |
| Abram et al. (2015) [36] | Psychiatry healthy sample (n = 244) | 26 | 50% ♂ 50% ♀ | - | No current drug use | Right-handed | Externalizing Spectrum Inventory, brief form | ICA |
| Klasen et al. (2018) [37] | Healthy young adults (n = 83) | 23.8 ± 3.6 | ♂ | - | - | Right-handed | Buss–Perry Aggression Questionnaire | ROI |
| Kolla et al. (2018) [38] | Antisocial personality disorder subjects (n = 21) Controls (n = 19) | 36.2 ± 8.7 34.2 ± 7.7 | ♂ | - | No current drug use | - | Buss–Perry Aggression Questionnaire Reactive–Proactive Aggression Questionnaire | ROI |
| Hoptman et al. (2010) [39] | Patients with schizophrenia or schizoaffective disorder (n = 25) Controls (n = 21) | 36.7 ± 10.5 40.4 ± 10.8 | 88% ♂ 12% ♀ 76% ♂ 24% ♀ | 12.3 ± 2.1 (years) 15.5 ± 3.0 (years) | CPZ equivalents | - | Buss Perry Aggression Questionnaire Life history of aggression Number total of arrests | ROI |
| Wagner et al. (2018) [40] | Unmedicated female patients with BPD (n = 33) Controls (n = 33) | 26.7 ± 6.4 26.4 ± 6.2 | ♀ | 12.1 ± 1.6 (years) 11.8 ± 1.5 (years) | No current drug use | . | Buss–Perry Aggression Questionnaire | ROI |
| Hasler et al. (2017) [41] | ADHD (n = 30) Controls (n = 15) | 38.7 ± 9.9 32.2 ± 5.5 | 70% ♂ 30% ♀ 26% ♂ 73% ♀ | - | 48h free of methylphenidate before fMRI | Right handed | Spielberger State–Trait Anger Expression Inventory-2 | CO2 challenge regressor |
| McGlade et al. (2015) [42] | Veterans males with TBI (n = 24) Veterans females with TBI (n = 17) | 37.75 ± 9.59 40.0 ±11.15 | ♂ ♀ | 14.33 ± 2.10 (years) 15.06 ± 2.51 (years) | - | - | Buss–Perry Aggression QuestionnaireDisplaced Aggression Questionnaire | Seed-based |
| Goswami et al. (2016) [43] | Retired athletes with a history of multiple concussions (n = 19); Controls (n = 17) | 50 ± 12 46 ± 10 | ♂ | 17 ± 1.8 16 ± 1.9 | No current drug use | - | Personality Assessment Inventory (aggression scale) | Seed-based |
| Dailey et al. (2018) [44] | Adults with TBI (n = 17) Healthy controls (n = 17) | 21.86 ± 2.79 23.88 ± 3.26 | 26% ♂ 73% ♀ 29% ♂ 71% ♀ | - | - | - | Buss–Perry Aggression Questionnaire | ROI |
| Gilam et al. (2017) [45] | Soldiers (n = 60) | 18.62 ± 0.88 | ♂ | > secondary education | No current drug use | Right-handed | Spielberger State–Trait Anger Expression Inventory Geneva Emotion Wheel | Brain functional parcellation |
| Buades-Rotger et al. (2018) [46] | Healthy young women (n = 39) | 23.22 ± 3.2 | ♀ | - | No current drug use | Right-handed | Social Threat Aggression Paradigm | ROI |
| Siep et al. (2018) [47] | Violent offenders (n = 18) Non-offender controls (n = 18) | 35.17 ± 7.12 37.06 ± 15.24 | ♂ | Current alcohol use No current drug use | - | - | Seed-based | |
| Chen et al. (2015) [48] | Young violent offenders (n = 30) Controls (n = 29) | 16.06 ± 0.7 16.06 ± 0.4 | ♂ | 7.76 ± 2.2 10.06 ± 0.0 | - | Right-handed | - | ROI |
| Leutgeb et al. (2016) [49] | Violent inmates of maximum security prison (n = 31) Controls (n = 30) | 36.8 ±12.0 35.1 ± 9.0 | ♂ | 11.3 ± 1.7 (years) 11.6 ± 1.0 (years) | Non-medicated | Right-handed | - | Seed-based |
| Varkevisser et al. (2017) [50] | Impulsive and violent soldiers (n = 28) Controls (n = 30) | 36.54 ± 6.27 34.53 ± 7.59 | ♂ | 67,9% middle 53,3% middle | - | - | Interviews and criminal records | ROI |
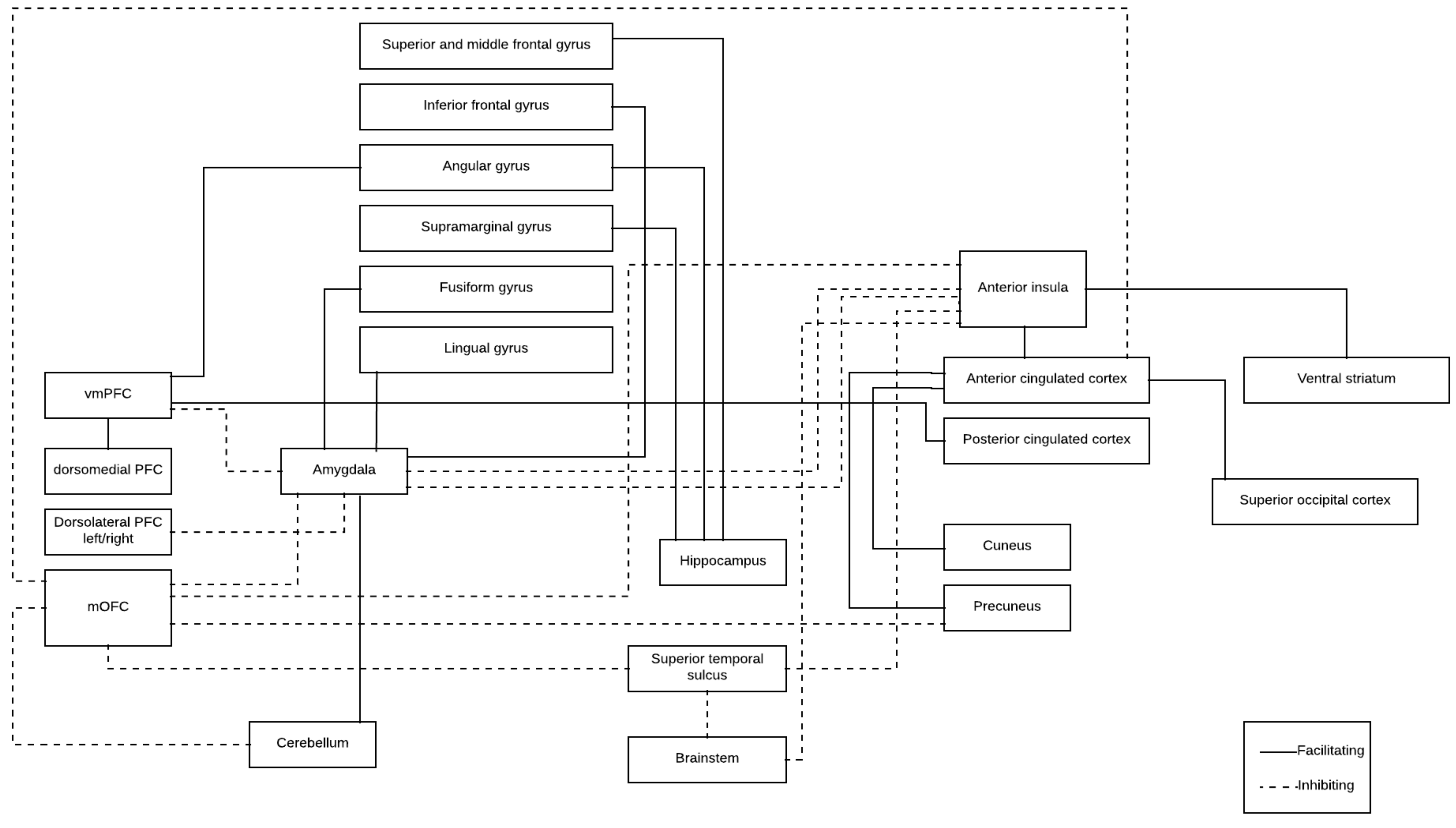
| Brain Structure (From) | Brain Structure (To) | Functional Connectivity | Aggression Assessment |
|---|---|---|---|
| Trait aggression | |||
| Amygdala (bilateral) | Left mOFC | ⬇ | ⬆ Trait aggression |
| vmPFC | ⬇ | ⬆ Trait aggression | |
| Nucleus centralis superior (median raphe nucleus) | Frontopolar cortex (Brodmann area 10) | ⬇ | ⬆ Trait aggression |
| vmPFC | Bilateral supramarginal gyrus | ⬇ | ⬆ Trait aggression |
| Amygdala (right) | Inferior frontal gyrus (right) | ⬆ | ⬆ Trait aggression |
| vmPFC | Angular gyrus (right) Posterior cingulated cortex Dorsomedial PFC | ⬆ | ⬆ Trait aggression |
| Anger expression | |||
| Amygdala (bilateral) | Left mOFC | ⬇ | ⬇ Anger control-out |
| Hippocampus (right) | Parietal (supramarginal and angular gyrus) Frontal lobe (superior and middle frontal gyrus) | ⬆ | ⬆ Anger expression-out |
| Proactive aggression | |||
| Ventral striatal | Angular gyrus | ⬆ | ⬆ Proactive aggression |
| Physical aggression | |||
| OFC (left) | Left angular region | ⬇ | ⬆ Physical aggression |
| Anterior insula | OFC | ⬇ | ⬆ Physical aggression ⬆ Destructive aggression |
| Anterior insula | Ventral striatumAnterior cingulate cortex | ⬆ | ⬆ Physical aggression |
| Revenge feelings | |||
| Right OFC | Right cerebellum Right angular gyrus | ⬆ | ⬆ Revenge feelings |
| Right OFC | Right midoccipital cortex | ⬆ | ⬇ Revenge feelings |
| Laboratory task | |||
| Amygdala basolateral | Left superior temporal gyrus | ⬆ | ⬆ Aggressive strategies (laboratory task) |
| mOFC Superior parietal lobule | ⬇ | ⬆ Aggressive strategies (laboratory task) | |
| Left mOFC | Left amygdala | ⬆ | Before emotional induction task (violent offenders) |
| Left amygdala | Left uncus/amygdala Posterior insula | ⬇ | Before emotional induction task (violent offenders) |
| Left mOFC | Left amygdala | ⬇ | After emotional induction task (violent offenders) |
| Left amygdala | Right posterior insula Right superior temporal gyrus | ⬆ | After emotional induction task (violent offenders) |
| Violent populations (no-self reported) | |||
| Caudate nucleus (right) mPFC (right) Precuneus (left) | Adjacent structures | ⬇ | Young violent offenders |
| Supramarginal gyrus (right) | Adjacent structures | ⬆ | Young violent offenders |
| Right cerebellar hemisphere | Left amygdala | ⬆ | Violent inmates |
| Bilateral cerebellar hemisphere | Bilateral OFC | ⬇ | Violent inmates |
| Cerebellar vermis | Left OFC | ⬇ | Violent inmates |
| Left dorsolateral PFC | Right dorsolateral PFC | ⬆ | Violent inmates |
| Amygdala basolateral (bilateral) | Left dorsolateral PFC | ⬇ | Impulsive and aggressive group |
| Left centromedial amygdala | Left fusiform gyrus Lingual gyrus | ⬆ | Impulsive and aggressive group |
| Left anterior cingulate cortex | Left cuneus Left calcarine cortex Left superior occipital cortex | ⬆ | Impulsive and aggressive soldiers |
| Right anterior cingulated cortex | Left cuneus Left precuneus Left calcarine cortex Left superior occipital cortex | ⬆ | Impulsive and aggressive soldiers |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romero-Martínez, Á.; González, M.; Lila, M.; Gracia, E.; Martí-Bonmatí, L.; Alberich-Bayarri, Á.; Maldonado-Puig, R.; Ten-Esteve, A.; Moya-Albiol, L. The Brain Resting-State Functional Connectivity Underlying Violence Proneness: Is It a Reliable Marker for Neurocriminology? A Systematic Review. Behav. Sci. 2019, 9, 11. https://doi.org/10.3390/bs9010011
Romero-Martínez Á, González M, Lila M, Gracia E, Martí-Bonmatí L, Alberich-Bayarri Á, Maldonado-Puig R, Ten-Esteve A, Moya-Albiol L. The Brain Resting-State Functional Connectivity Underlying Violence Proneness: Is It a Reliable Marker for Neurocriminology? A Systematic Review. Behavioral Sciences. 2019; 9(1):11. https://doi.org/10.3390/bs9010011
Chicago/Turabian StyleRomero-Martínez, Ángel, Macarena González, Marisol Lila, Enrique Gracia, Luis Martí-Bonmatí, Ángel Alberich-Bayarri, Rebeca Maldonado-Puig, Amadeo Ten-Esteve, and Luis Moya-Albiol. 2019. "The Brain Resting-State Functional Connectivity Underlying Violence Proneness: Is It a Reliable Marker for Neurocriminology? A Systematic Review" Behavioral Sciences 9, no. 1: 11. https://doi.org/10.3390/bs9010011
APA StyleRomero-Martínez, Á., González, M., Lila, M., Gracia, E., Martí-Bonmatí, L., Alberich-Bayarri, Á., Maldonado-Puig, R., Ten-Esteve, A., & Moya-Albiol, L. (2019). The Brain Resting-State Functional Connectivity Underlying Violence Proneness: Is It a Reliable Marker for Neurocriminology? A Systematic Review. Behavioral Sciences, 9(1), 11. https://doi.org/10.3390/bs9010011







