Age-Related Changes in the Behavior of Apolipoprotein E Knockout Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Behavioral Tests
2.2.1. Gross Neurologic Function
2.2.2. Specific Behavioral Test
2.3. Learning and Memory Tests
2.3.1. Forced Alternation Test (Y-Maze)
2.3.2. Novel Object Recognition Test
2.3.3. Passive Avoidance (PA) Conditioning
2.4. Statistical Analysis
3. Results
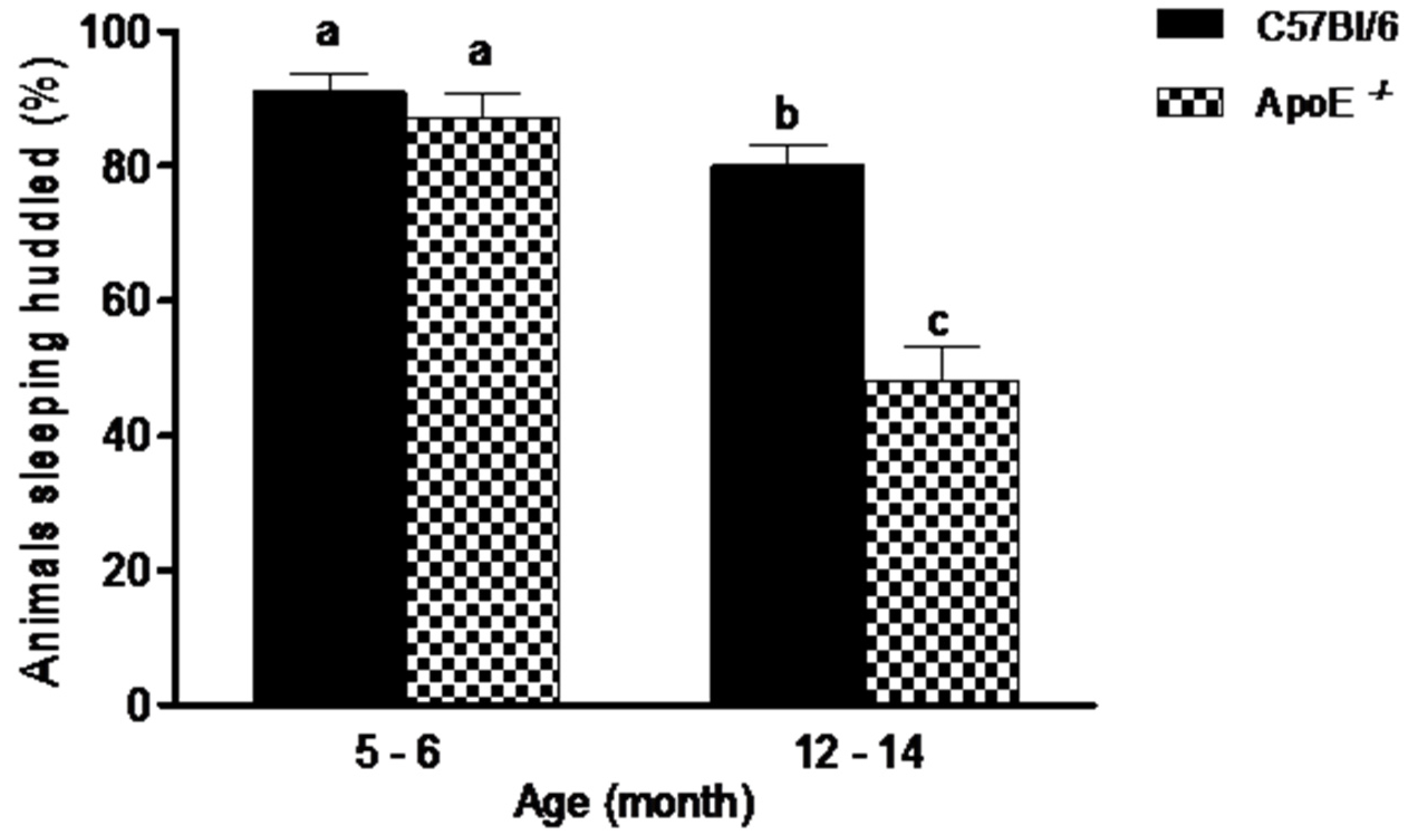
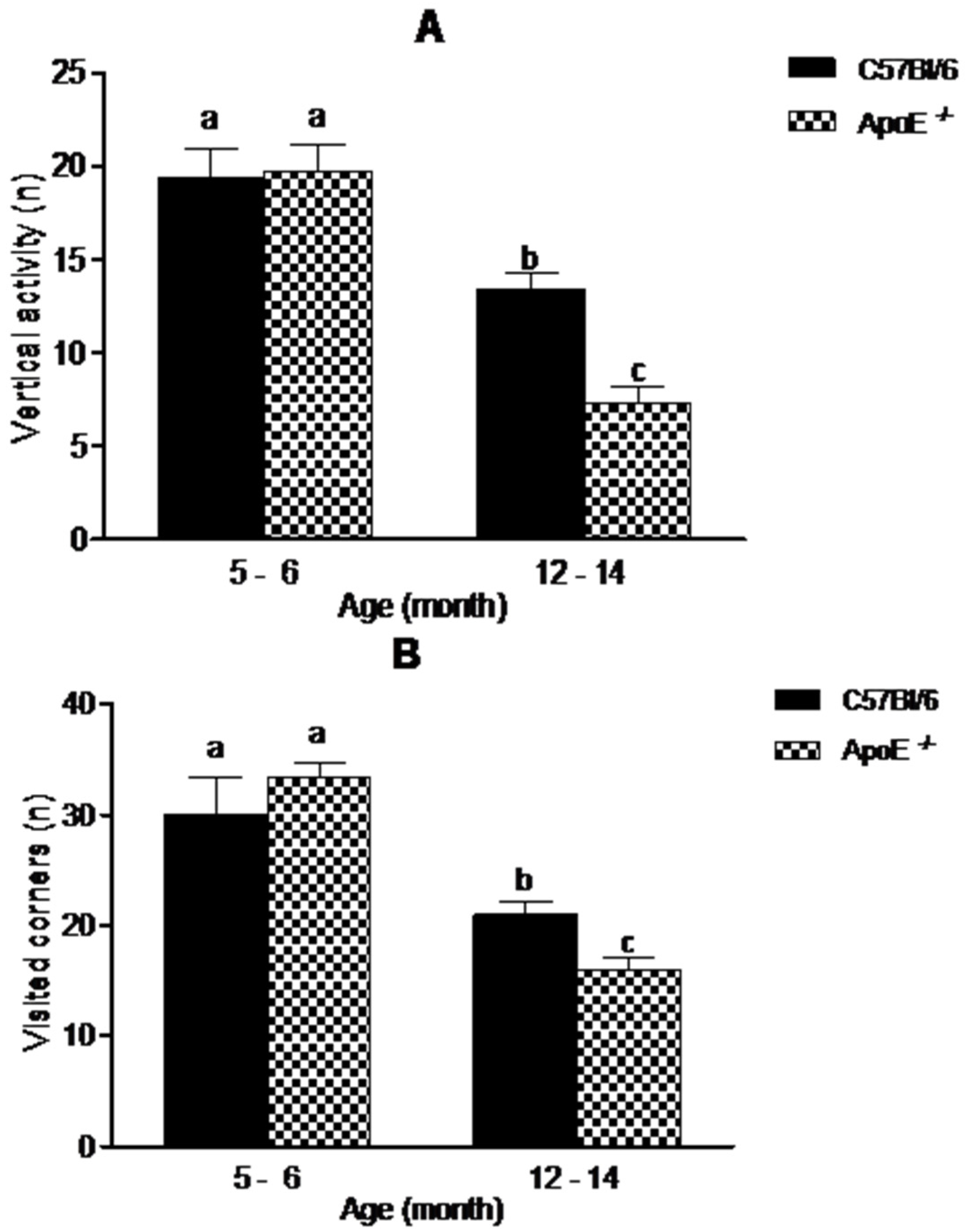
3.1. Gross Neurologic Function
3.2. Specific Behavioral Test
3.3. Learning and Memory Tests
3.3.1. Forced Alternation Test (Y-Maze)
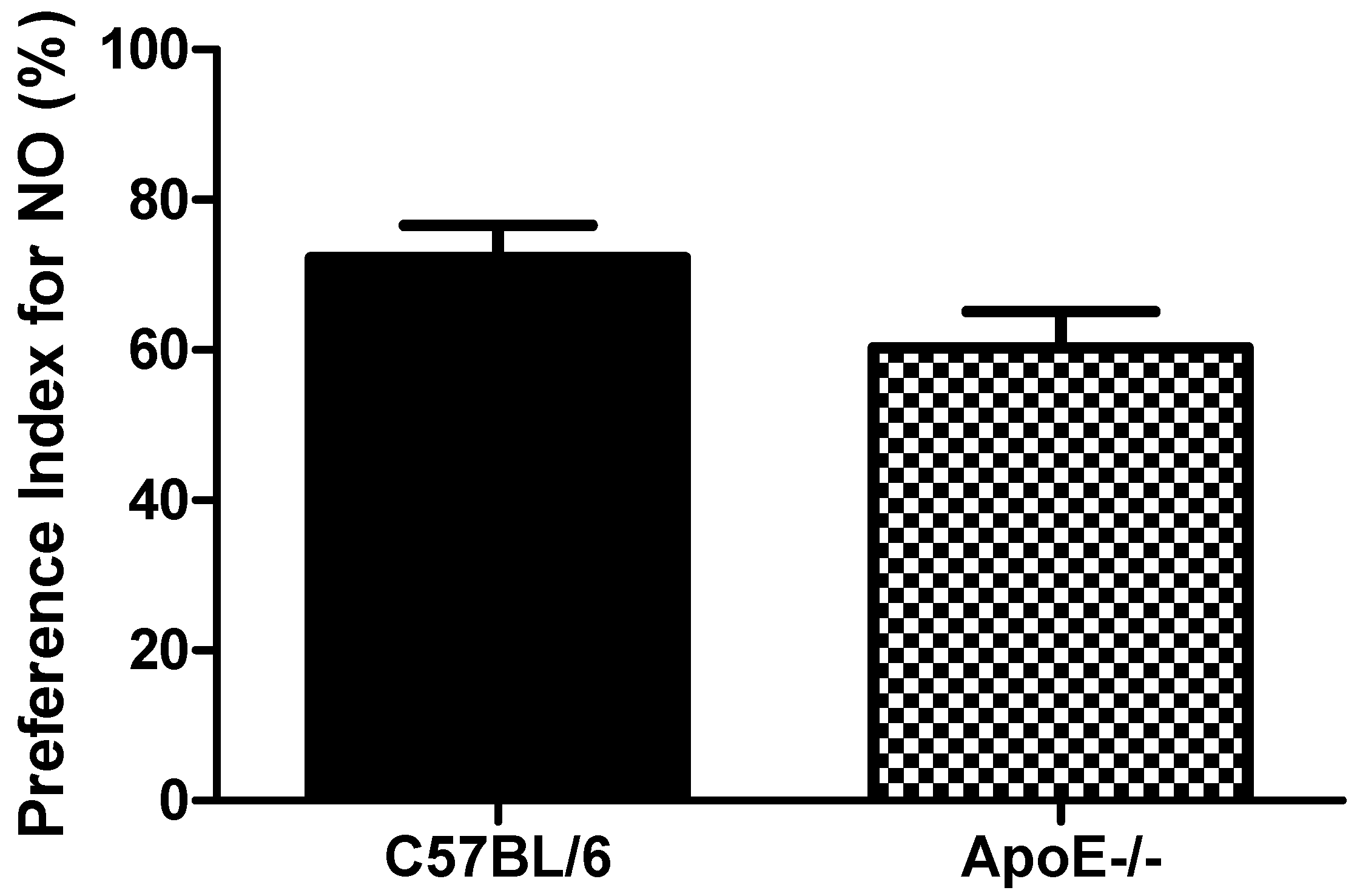
3.3.2. Novel Object Recognition Test
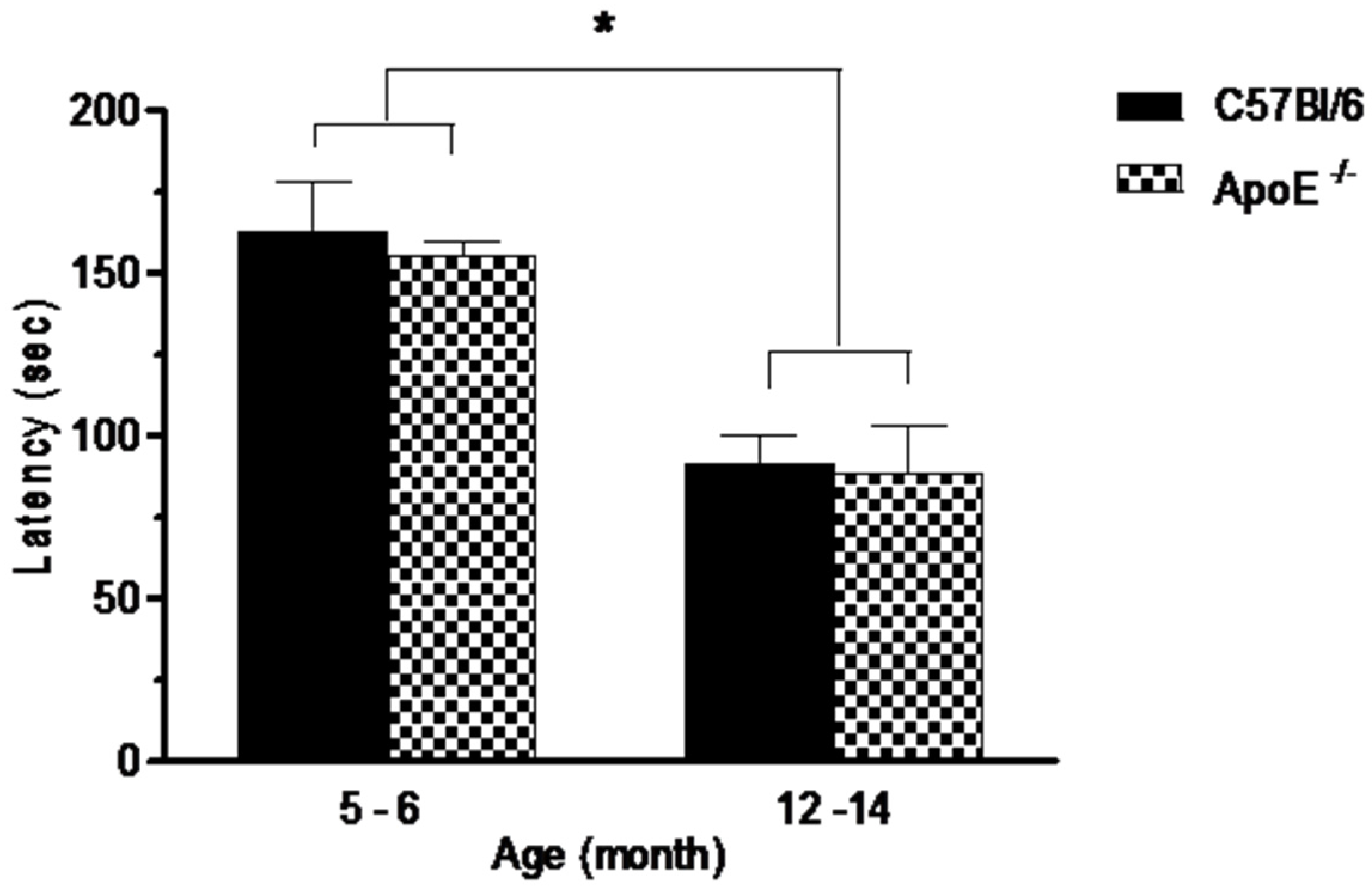
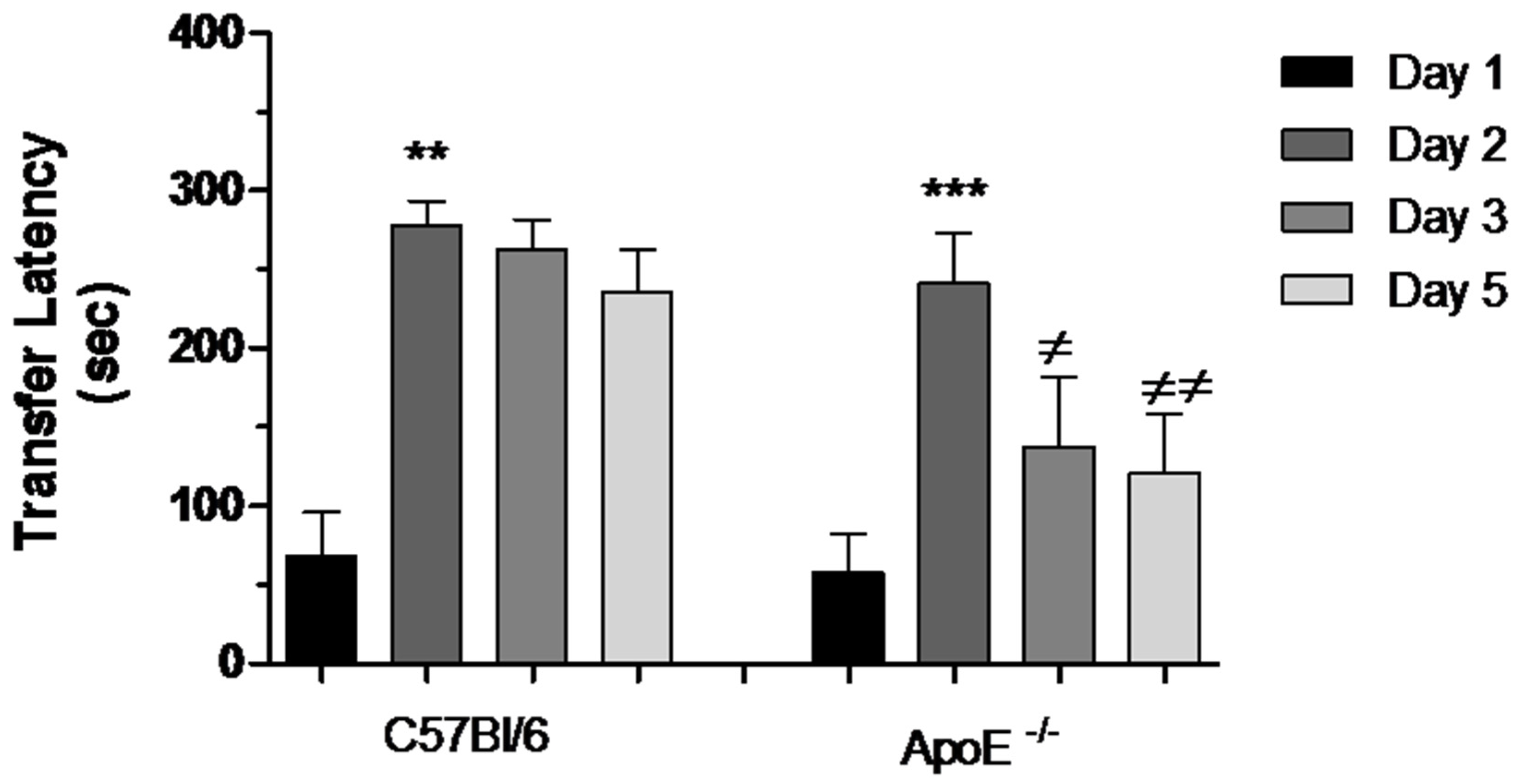
3.3.3. Passive Avoidance (PA) Conditioning
4. Discussion
Author Contributions
Conflicts of Interest
References
- Mahley, R.W. Apolipoprotein E: Cholesterol transport protein with expanding role in cell biology. Science 1988, 240, 622–630. [Google Scholar] [CrossRef] [PubMed]
- Mahley, R.W.; Rall, S.C. Apolipoprotein E: Far more than a lipid transport protein. Annu. Rev. Genom. Hum. Genet. 2000, 1, 507–537. [Google Scholar] [CrossRef] [PubMed]
- Boyles, J.K.; Pitas, R.E.; Wilson, E.; Mahley, R.W.; Taylor, J.M. Apolipoprotein E associated with astrocytic glia of the central nervous system and with non myelinating glia of the peripheral nervous system. J. Clin. Investig. 1985, 76, 1501–1513. [Google Scholar] [CrossRef] [PubMed]
- Hauser, P.S.; Narayanaswami, V.; Ryan, R.O. Apolipoprotein E: From lipid transport to neurobiology. Prog. Lipid Res. 2011, 50, 62–74. [Google Scholar] [CrossRef] [PubMed]
- Boyles, J.K.; Zoellner, C.D.; Anderson, L.J.; Kosik, L.M.; Pitas, R.E.; Weisgraber, K.H.; Hui, D.Y.; Mahley, R.W.; Gebicke-Haerter, P.J.; Ignatious, M.J.; et al. A role for apolipoprotein E, apolipoprotein A-1, and low density lipoprotein receptors in cholesterol transport during regeneration and remyelination of rat sciatic nerve. J. Clin. Investig. 1989, 83, 1015–1031. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, E.J.; Gregg, R.E.; Ghiselli, G.; Forte, T.M.; Ordovas, J.M.; Zech, L.A.; Brewer, H.B., Jr. Familial apolipoprotein E deficiency. J. Clin. Investig. 1986, 78, 1206–1219. [Google Scholar] [CrossRef] [PubMed]
- Piedrahita, J.A.; Zhang, S.H.; Hagaman, J.R.; Oliver, P.M.; Maeda, N. Generation of mice carrying a mutant apolipoprotein E gene inactivated by gene targeting in embryonic stem cells. Proc. Natl. Acad. Sci. USA 1992, 89, 4471–4475. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Lomnitski, L.; Michaelson, D.M.; Shohami, E. Motor and cognitive deficits in apolipoprotein E-deficient mice after closed head injury. Neuroscience 1997, 80, 1255–1262. [Google Scholar] [CrossRef]
- Masliah, E.; Mallory, M.; Ge, N.; Alford, M.; Veinbergs, I.; Roses, A.D. Neurodegeneration in the central nervous system of apoE-deficient mice. Exp. Neurol. 1995, 136, 107–122. [Google Scholar] [CrossRef] [PubMed]
- Buttini, M.; Orth, M.; Bellosta, S.; Akeefe, H.; Pitas, R.E.; Wyss-Coray, T.; Mucke, L.; Mahley, R.W. Expression of human apolipoprotein E3 or E4 in the brains of Apoe−/− mice: Isoform-specific effects on neurodegeneration. J. Neurosci 1999, 19, 4867–4880. [Google Scholar] [PubMed]
- Gordon, I.; Grauer, E.; Genis, I.; Sehayek, E.; Michaelson, D.M. Memory deficits and cholinergic impairments in apolipoprotein E.-deficient mice. Neurosci. Lett. 1995, 199, 1–4. [Google Scholar] [CrossRef]
- Oitzl, M.S.; Mulder, M.; Lucassen, P.J.; Havekes, L.M.; Grootendorst, J.; de Kloet, E.R. Severe learning deficits in apolipoprotein E-knockout mice in a water maze task. Brain Res. 1997, 752, 189–196. [Google Scholar] [CrossRef]
- Veinbergs, I.; Mallory, M.; Mante, M.; Rockenstein, E.; Gilbert, J.R.; Masliah, E. Differential neurotrophic effects of apolipoprotein E in aged transgenic mice. Neurosci. Lett. 1999, 265, 218–222. [Google Scholar] [CrossRef]
- Veinbergs, I.; Mallory, M.; Sagara, Y.; Masliah, E. Vitamin E supplementation prevents spatial learning deficits and dendritic alterations in aged apolipoprotein E-deficient mice. Eur. J. Neurosci. 2000, 12, 4541–4546. [Google Scholar] [CrossRef] [PubMed]
- Shea, T.B.; Rogers, E.; Ashline, D.; Ortiz, D.; Sheu, M.S. Apolipoprotein Edeficiency promotes increased oxidative stress and compensatory increases in antioxidants in brain tissue. Free Radic. Biol. Med. 2002, 33, 1115–1120. [Google Scholar] [CrossRef]
- Fullerton, S.M.; Shirman, G.A.; Strittmatter, W.J.; Matthew, W.D. Impairment of the blood-nerve and blood-brain barriers in apolipoprotein e knockout mice. Exp. Neurol. 2001, 169, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Gordon, I.; Genis, I.; Grauer, E.; Sehayek, E.; Michaelson, D.M. Biochemical and cognitive studies of apolipoprotein-E-deficient mice. Mol. Chem. Neuropathol. 1996, 28, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Masliah, E.; Samuel, W.; Veinbergs, I.; Mallory, M.; Mante, M.; Saitoh, T. Neurodegeneration and cognitive impairment in apoE-deficient mice is ameliorated by infusion of recombinant apoE. Brain Res. 1997, 751, 307–314. [Google Scholar] [CrossRef]
- Mauch, D.H.; Nagler, K.; Schumacher, S.; Goritz, C.; Muller, E.C.; Otto, A.; Pfrieger, F.W. CNS synaptogenesis promoted by glia-derived cholesterol. Science 2001, 294, 1354–1357. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.; Barnes, J.C.; Bliss, T.V.P.; Cain, D.P.; Cambon, K.; Davies, H.A.; Errington, M.L.; Fellows, L.A.; Gray, R.A.; Hoh, T.; et al. Behavioural, physiological and morphological analysis of a line of apolipoprotein E knockout mouse. Neuroscience 1998, 85, 93–110. [Google Scholar] [CrossRef]
- Hartman, R.E.; Wozniak, D.F.; Nardi, A.; Olney, J.W.; Sartorius, L.; Holtzman, D.M. Behavioral phenotyping of GFAP-apoE3 and -apoE4 transgenic mice: ApoE4 mice show profound working memory impairments in the absence of Alzheimer’s-like neuropathology. Exp. Neurol. 2001, 170, 326–344. [Google Scholar] [CrossRef] [PubMed]
- Bour, A.; Grootendorst, J.; Vogel, E.; Kelche, C.; Dodart, J.-C.; Bales, K.; Moreau, P.H.; Sullivan, P.M.; Mathis, C. Middle-aged human apoE4 targeted-replacement mice show retention deficits on a wide range of spatial memory tasks. Behav. Brain Res. 2008, 193, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Raber, J.; Wong, D.; Buttini, M.; Orth, M.; Bellosta, S.; Pitas, R.E.; Mahley, R.W.; Mucke, L. Isoform specific effects of human apolipoprotein E on brain function revealed in ApoE knockout mice: Increased susceptibility of females. Proc. Natl. Acad. Sci. USA 1998, 95, 10914–10919. [Google Scholar] [CrossRef] [PubMed]
- Grootendorst, J.; de Kloet, E.R.; Vossen, C.; Dalm, S.; Oitzl, M.S. Repeated exposure to rats has persistent genotype-dependent effects on learning and locomotor activity of apolipoprotein E knockout and C57Bl/6 mice. Behav. Brain Res. 2001, 125, 249–259. [Google Scholar] [CrossRef]
- Crawley, J.N.; Paylor, R. A proposed test battery and constellations of specific behavioral paradigms to investigate the behavioral phenotypes of transgenic and knockout mice. Horm. Behav. 1997, 31, 197–211. [Google Scholar] [CrossRef] [PubMed]
- Lijam, N.; Paylor, R.; McDonald, M.P.; Crawley, J.N.; Deng, C.X.; Herrup, K.; Stevens, K.E.; Maccaferri, G.; McBain, C.J.; Sussman, D.J.; et al. Social interaction and sensorimotor gating abnormalities in mice lacking Dvl1. Cell 1997, 90, 895–905. [Google Scholar] [CrossRef]
- Schumm, S.; Sebban, C.; Cohen-Salmon, C.; Callebert, J.; Launay, J.M.; Golmard, J.L.; Boussicault, L.; Petropoulos, I.; Hild, A.; Rousselet, E.; et al. Aging of the dopaminergic system and motor behavior in mice intoxicated with the parkinsonian toxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. J. Neurochem. 2012, 122, 1032–1046. [Google Scholar] [CrossRef] [PubMed]
- Prut, L.; Belzung, C. The open field as a paradigm to measure the effects of drugs on anxiety-like behaviors: A review. Eur. J. Pharmacol. 2003, 463, 3–33. [Google Scholar] [CrossRef]
- Hughes, R.N. The value of spontaneous alternation behavior (SAB) as a test of retention in pharmacological investigations of memory. Neurosci. Biobehav. Rev. 2004, 28, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Antunes, M.; Biala, G. The novel object recognition memory: Neurobiology, test procedure, and its modifications. Cogn. Process. 2012, 13, 93–110. [Google Scholar] [CrossRef] [PubMed]
- Caston, J.; Hilber, P.; Chianale, C.; Mariani, J. Effect on training on motor abilities of heterozygous staggerer mutant (Rora(+)/ Rora(sg)) mice during aging. Behav. Brain Res. 2003, 141, 35–42. [Google Scholar] [CrossRef]
- Deacon, R.M.; Rawlins, J.N. T-maze alternation in the rodent. Nat. Protoc. 2006, 1, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Dietschy, J.M.; Turley, S.D. Thematic review series: Brain Lipids. Cholesterol metabolism in the central nervous system during early development and in the mature animal. J. Lipid Res. 2004, 45, 1375–1397. [Google Scholar] [CrossRef] [PubMed]
- Saher, G.; Brügger, B.; Lappe-Siefke, C.; Möbius, W.; Tozawa, R.; Wehr, M.C.; Wieland, F.; Ishibashi, S.; Nave, K.A. High cholesterol level is essential for myelin membrane growth. Nat. Neurosci. 2005, 8, 468–475. [Google Scholar] [CrossRef] [PubMed]
- Pfrieger, F.W. Cholesterol homeostasis and function in neurons of the central nervous system. Cell Mol. Life Sci. 2003, 60, 1158–1171. [Google Scholar] [CrossRef] [PubMed]
- Fisher, A.; Brandeis, R.; Chapman, S.; Pittel, Z.; Michaelson, D.M. M1 Muscarinic Agonist Treatment Reverses Cognitive and Cholinergic Impairments of Apolipoprotein E-Deficient Mice. J. Neurochem. 1998, 70, 1991–1997. [Google Scholar] [CrossRef] [PubMed]
- Alzheimer’s Association. 2013 Alzheimer’s disease facts and figures. Alzheimers Dement. 2013, 9, 208–245. [Google Scholar] [CrossRef]
- Shoji, H.; Takao, K.; Hattori, S.; Miyakawa, T. Age-related changes in behavior in C57BL/6J mice from young adulthood to middle age. Mol. Brain 2016, 9, 11. [Google Scholar] [CrossRef] [PubMed]
- Lamberty, I.; Gower, A.J. Age-Related Changes in Spontaneous Behavior and Learning in NMRI Mice From Maturity to Middle Age. Physiol. Behav. 1990, 47, 1137–1144. [Google Scholar] [CrossRef]
- Fagan, A.M.; Murphy, B.A.; Patel, S.N.; Kilbridge, J.F.; Mobley, W.C.; Bu, G.; Holtzman, D.M. Evidence for normal aging of the septo-hippocampal cholinergic system in apoE (−/−) mice but impaired clearance of axonal degeneration products following injury. Exp. Neurol. 1998, 151, 314–325. [Google Scholar] [CrossRef] [PubMed]
- Dutta, S.; Sengupta, P. Men and mice: Relating their ages. Life Sci. 2015, 152, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Raber, J.; Wong, D.; Yu, G.Q.; Buttini, M.; Mahley, R.W.; Pitas, R.E.; Mucke, L. Apolipoprotein E and cognitive performance. Nature 2000, 404, 352–354. [Google Scholar] [CrossRef] [PubMed]
- Lane-Donovan, C.; Wong, W.M.; Durakoglugil, M.S.; Wasser, C.R.; Jiang, S.; Xian, X.; Herz, J. Genetic Restoration of Plasma ApoE Improves Cognition and Partially Restores Synaptic Defects in ApoE-Deficient Mice. J. Neurosci. 2016, 36, 10141–10150. [Google Scholar] [CrossRef] [PubMed]
- Ruitenberg, A.; Ott, A.; van Swieten, J.C.; Hofman, A.; Breteler, M.M. Incidence of dementia: Does gender make a difference? Neurobiol. Aging 2001, 22, 575–580. [Google Scholar] [CrossRef]
- Hebert, L.E.; Scherr, P.A.; Bienias, J.L.; Bennett, D.A.; Evans, D.A. Alzheimer disease in the US population: Prevalence estimates using the 2000 census. Arch Neurol. 2003, 60, 1119–1122. [Google Scholar] [CrossRef] [PubMed]
- Nomura, Y.; Nishiyama, N.; Saito, H.; Matsuki, N. Role of cholinergic neurotransmission in the amygdala on performances of passive avoidance learning in mice. Biol. Pharm. Bull. 1994, 17, 490–494. [Google Scholar] [CrossRef] [PubMed]
- Klinkenberg, I.; Blokland, A. The validity of scopolamine as a pharmacological model for cognitive impairment: A review of animal behavioral studies. Neurosci. Biobehav. Rev. 2010, 34, 1307–1350. [Google Scholar] [CrossRef] [PubMed]
- Raber, J.; Huang, Y.; Ashford, J.W. ApoE genotype accounts for the vast majority of AD risk and AD pathology. Neurobiol. Aging 2004, 25, 641–650. [Google Scholar] [CrossRef] [PubMed]
- Corder, E.H.; Saunders, A.M.; Strittmatter, W.J.; Schmechel, D.E.; Gaskell, P.C.; Small, G.W.; Roses, A.D.; Haines, J.L.; Pericak-Vance, M.A. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’s disease in late onset families. Science 1993, 261, 921–923. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.-C.; Kanekiyo, T.; Xu, H.; Bu, G. Apolipoprotein E and Alzheimer disease: Risk, mechanisms, and therapy, Nature reviews. Neurology 2013, 9, 106–118. [Google Scholar] [CrossRef] [PubMed]
- Izaks, G.J.; Gansevoort, R.T.; van der Knaap, A.M.; Navis, G.; Dullaart, R.P.; Slaets, J.P. The association of APOE genotype with cognitive function in persons aged 35 years or older. PLoS ONE 2011, 6, e27415. [Google Scholar] [CrossRef] [PubMed]
- Messier, C. Diabetes, Alzheimer’s disease and apolipoprotein genotype. Exp. Gerontol. 2003, 38, 941–946. [Google Scholar] [CrossRef]
- Kim, J.; Basak, J.M.; Holtzman, D.M. The role of apolipoprotein E in Alzheimer’s disease. Neuron 2009, 63, 287–303. [Google Scholar] [CrossRef] [PubMed]
- Drosatus, K.; Goldberg, I.J. Lipoproteins: A source of Cardiac Lipids. In Cardiac Energy Metabolism in Health and Disease, 2nd ed.; Lopaschuk, G.D., Dhalla, N.S., Eds.; Springer Science + Business Media: New York, NY, USA, 2014; ISBN 978-1-4939-1227-8. [Google Scholar]
- Plump, A.S.; Smith, J.D.; Hayek, T.; Aalto-Setälä, K.; Walsh, A.; Verstuyft, J.G.; Rubin, E.M.; Breslow, J.L. Severe hypercholesterolemia and atherosclerosis in apolipoprotein E-deficient mice created by homologous recombination in ES cells. Cell 1992, 71, 343–353. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.H.; Reddick, R.L.; Piedrahita, J.A.; Maeda, N. Spontaneous hypercholesterolemia and arterial lesions in mice lacking apolipoprotein E. Science 1992, 258, 468–471. [Google Scholar] [CrossRef] [PubMed]
- Anstey, K.J.; Lipnicki, D.M.; Low, L.F. Cholesterol as a risk factor for dementia and cognitive decline: A systematic review of prospective studies with meta-analysis. Am. J. Geriatr. Psychiatry 2008, 16, 343–354. [Google Scholar] [CrossRef] [PubMed]
- Farr, S.A.; Yamada, K.A.; Butterfield, D.A.; Abdul, H.M.; Xu, L.; Miller, N.E.; Banks, W.A.; Morley, J.E. Obesity and hypertriglyceridemia produce cognitive impairment. Endocrinology 2008, 149, 2628–2636. [Google Scholar] [CrossRef] [PubMed]
- Freeman, L.R.; Haley-Zitlin, V.; Rosenberger, D.S.; Granholm, A.C. Damaging effects of a high-fat diet to the brain and cognition: A review of proposed mechanisms. Nutr. Neurosci. 2014, 17, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Ayata, C.; Shin, H.K.; Dileko, Z.E.; Atochin, D.N.; Kashiwagi, S.; Eikermann-Haerter, K.; Huang, P.L. Hyperlipidemia disrupts cerebrovascular reflexes and worsens ischemic perfusion defect. J. Cereb. Blood. Flow Metab. 2013, 33, 954–962. [Google Scholar] [CrossRef] [PubMed]
- Mulder, M.; Blokland, A.; van den Berg, D.J.; Schulten, H.; Bakker, A.H.; Terwel, D.; Honig, W.; de Kloet, E.R.; Havekes, L.M.; Steinbusch, H.W.; et al. Apolipoprotein E protects against neuropathology induced by a high-fat diet and maintains the integrity of the blood-brain barrier during aging. Lab. Investig. 2001, 81, 953–960. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bell, R.D.; Winkler, E.A.; Singh, I.; Sagare, A.P.; Deane, R.; Wu, Z.; Holtzman, D.M.; Betsholtz, C.; Armulik, A.; Sallstrom, J.; et al. Apolipoprotein E controls cerebrovascular integrity via cyclophilin A. Nature 2012, 485, 512–516. [Google Scholar] [CrossRef] [PubMed]
- Panza, F.; D’Introno, A.; Colacicco, A.M.; Basile, A.M.; Capurso, C.; Kehoe, P.G.; Capurso, A.; Solfrizzi, V. Vascular risk and genetics of sporadic late-onset Alzheimer’s disease. J. Neural Transm. 2004, 111, 69–89. [Google Scholar] [CrossRef] [PubMed]
- Duron, E.; Hanon, O. Vascular risk factors, cognitve decline, and dementia. Vasc. Health Risk Manag. 2008, 4, 363–381. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fuentes, D.; Fernández, N.; García, Y.; García, T.; Morales, A.R.; Menéndez, R. Age-Related Changes in the Behavior of Apolipoprotein E Knockout Mice. Behav. Sci. 2018, 8, 33. https://doi.org/10.3390/bs8030033
Fuentes D, Fernández N, García Y, García T, Morales AR, Menéndez R. Age-Related Changes in the Behavior of Apolipoprotein E Knockout Mice. Behavioral Sciences. 2018; 8(3):33. https://doi.org/10.3390/bs8030033
Chicago/Turabian StyleFuentes, Dasha, Nidia Fernández, Yenela García, Teidy García, Ana Ruth Morales, and Roberto Menéndez. 2018. "Age-Related Changes in the Behavior of Apolipoprotein E Knockout Mice" Behavioral Sciences 8, no. 3: 33. https://doi.org/10.3390/bs8030033
APA StyleFuentes, D., Fernández, N., García, Y., García, T., Morales, A. R., & Menéndez, R. (2018). Age-Related Changes in the Behavior of Apolipoprotein E Knockout Mice. Behavioral Sciences, 8(3), 33. https://doi.org/10.3390/bs8030033




