Sensitivity to Social Contingency in Adults with High-Functioning Autism during Computer-Mediated Embodied Interaction
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
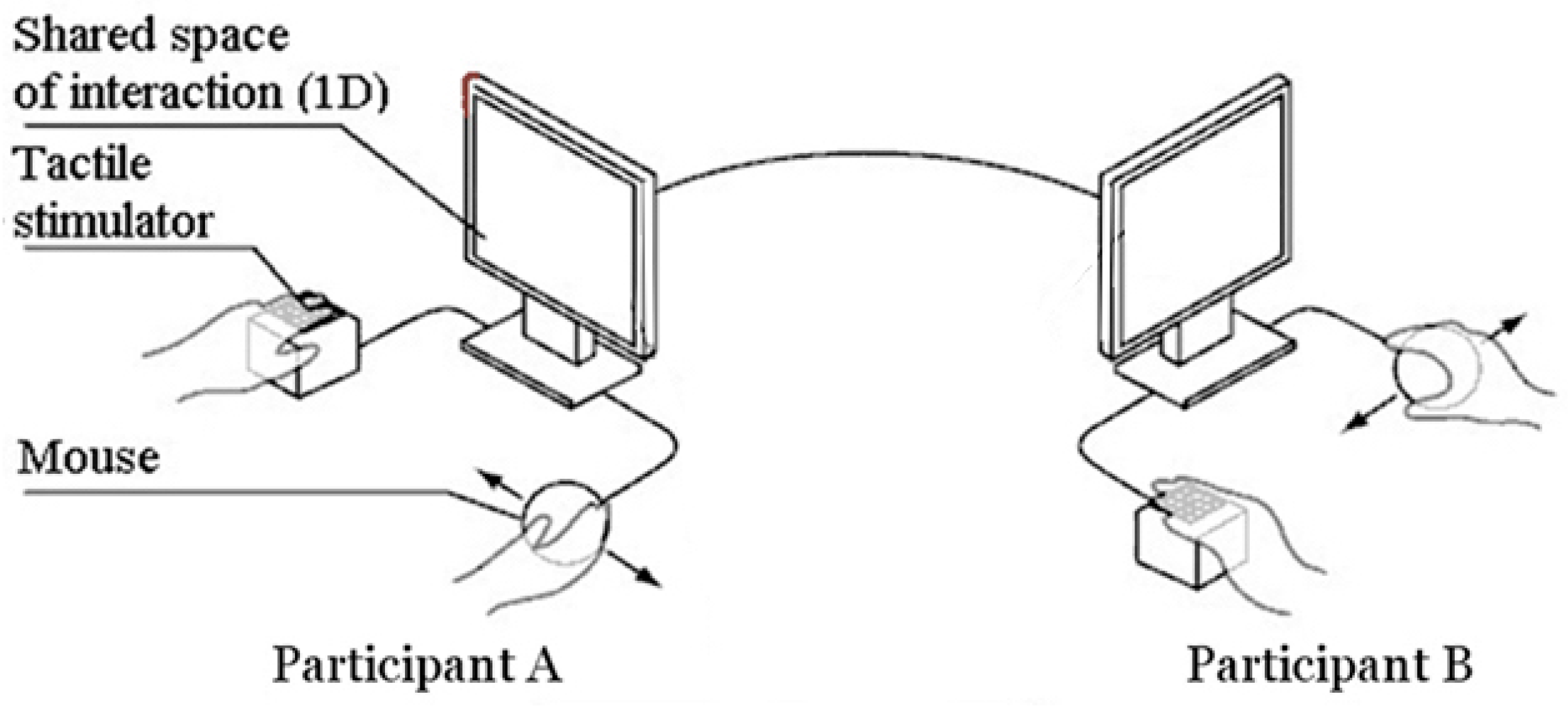
2.2. Apparatus
2.3. Procedure
2.4. Analysis
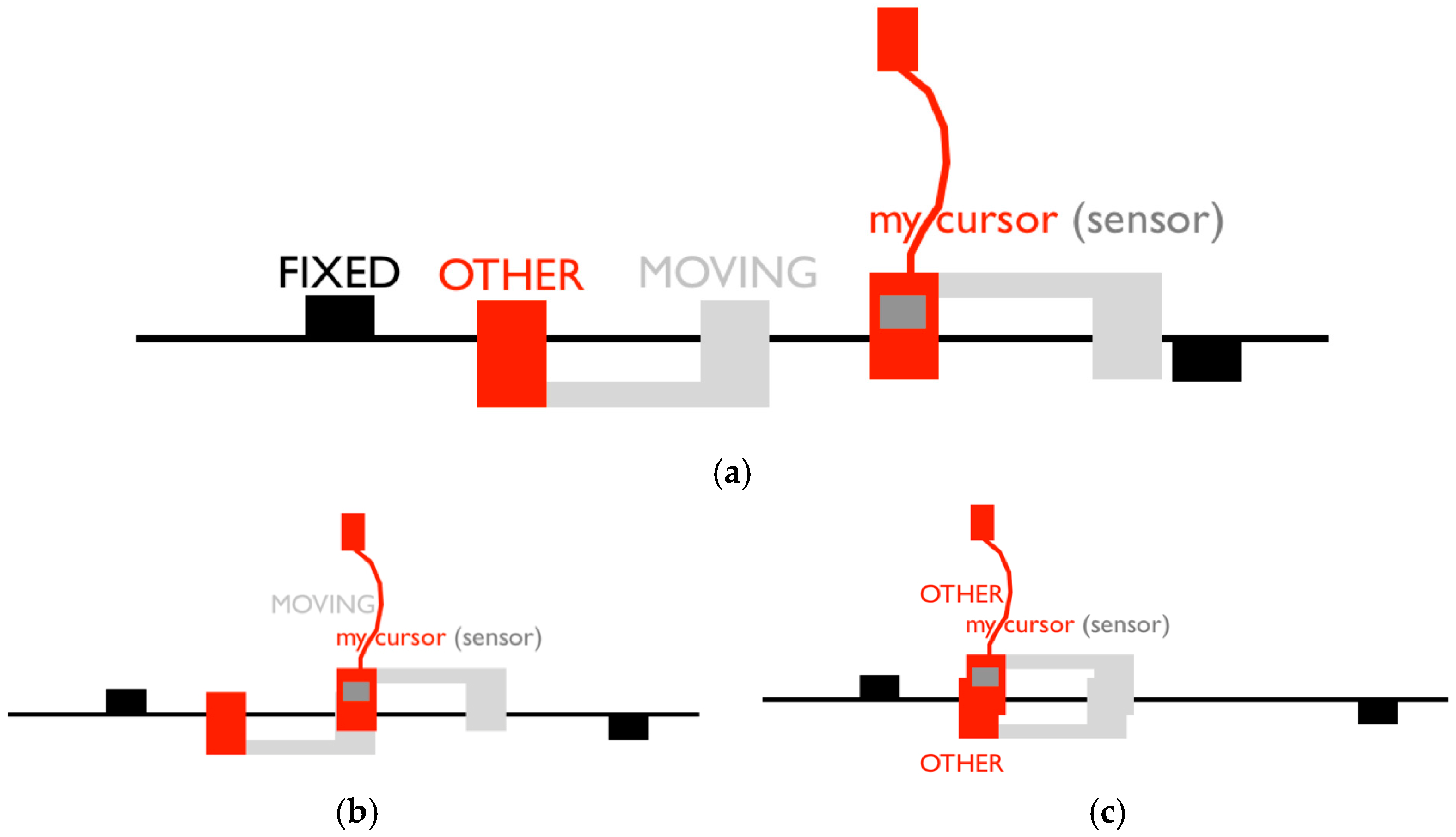
2.4.1. Visual Inspection
2.4.2. Classification of Encounters and Clicks
- (1)
- Frequency of discrete encounters, which reflects interaction dynamics;
- (2)
- Frequency of discrete clicks, which reflects conscious identification;
- (3)
- Ratios of clicks to encounters according to ObjectType, which essentially indicates the propensity to click on a specific type of encountered object.
2.4.3. Statistical Analysis
3. Results
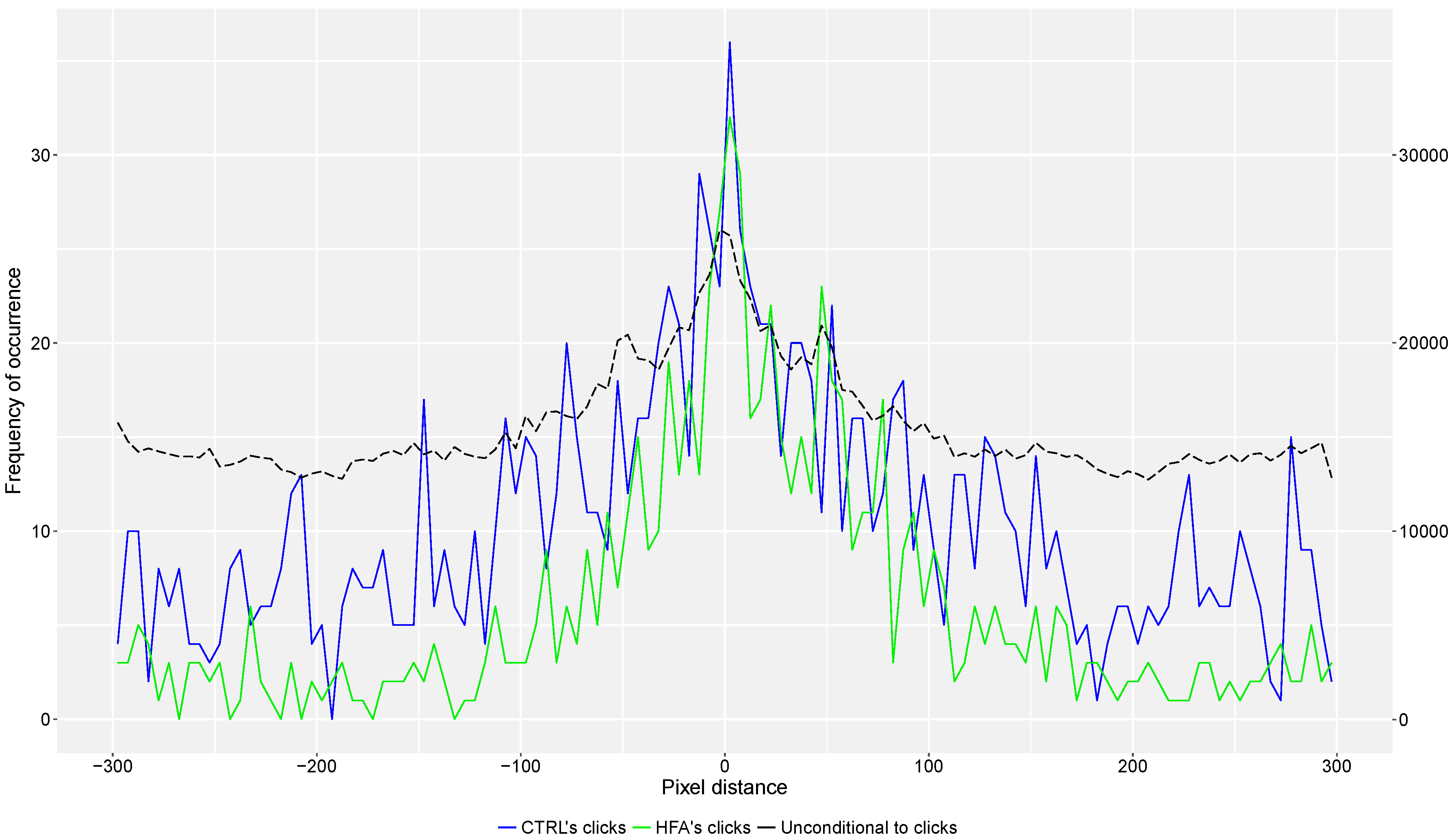
3.1. Frequency Distributions as a Function of the Distance between Participants
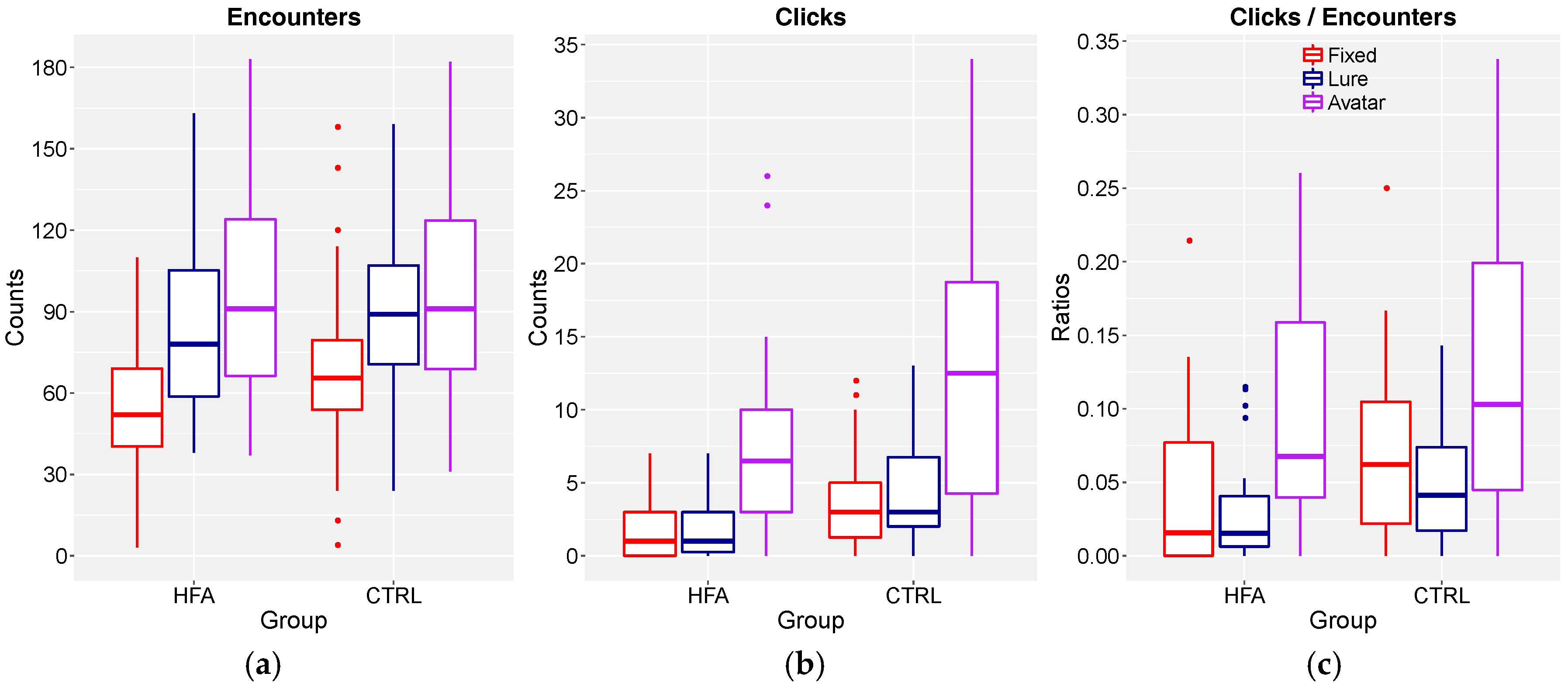
3.2. Number of Stimulations Per Object Type and Case of Clicks
3.3. Ratio of Clicks to Encounters
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- American Psychiatric Association, American Psychiatric Association DSM-5 Task Force. Diagnostic and Statistical Manual of Mental Disorders: DSM-5, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Baron-Cohen, S.; Wheelwright, S.; Skinner, R.; Martin, J.; Clubley, E. The autism-spectrum quotient (AQ): Evidence from Asperger syndrome/high-functioning autism, males and females, scientists and mathematicians. J. Autism Dev. Disord. 2001, 31, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Baron-Cohen, S.; Leslie, A.M.; Frith, U. Does the autistic child have a “theory of mind”? Cognition 1985, 21, 37–46. [Google Scholar] [CrossRef]
- Frith, U.; Happe, F. Theory of Mind and Self-Consciousness: What Is It Like to Be Autistic? Mind Lang. 1999, 14, 82–89. [Google Scholar] [CrossRef]
- Frith, U.; Frith, C.D. Development and neurophysiology of mentalizing. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2003, 358, 459–473. [Google Scholar] [CrossRef] [PubMed]
- Senju, A.; Southgate, V.; White, S.; Frith, U. Mindblind eyes: An absence of spontaneous theory of mind in Asperger syndrome. Science 2009, 325, 883–885. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, S. Understanding interpersonal problems in autism: Interaction theory as an alternative to theory of mind. Philos. Psychiatry Psychol. 2004, 11, 199–217. [Google Scholar] [CrossRef]
- Froese, T.; Stanghellini, G.; Bertelli, M.O. Is it normal to be a principal mindreader? Revising theories of social cognition on the basis of schizophrenia and high functioning autism-spectrum disorders. Res. Dev. Disabil. 2013, 34, 1376–1387. [Google Scholar] [CrossRef] [PubMed]
- Schilbach, L.; Eickhoff, S.B.; Cieslik, E.C.; Kuzmanovic, B.; Vogeley, K. Shall we do this together? Social gaze influences action control in a comparison group, but not in individuals with high-functioning autism. Autism 2012, 16, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Reed, T.; Peterson, C. A comparative study of autistic subjects’ performance at two levels of visual and cognitive perspective taking. J. Autism Dev. Disord. 1990, 20, 555–567. [Google Scholar] [CrossRef] [PubMed]
- Prior, M.; Dahlstrom, B.; Squires, T.-L. Autistic children’s knowledge of thinking and feeling states in other people. J. Child Psychol. Psychiatry 1990, 31, 587–601. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, T. Pathologies of intersubjectivity in autism and schizophrenia. J. Conscious. Stud. 2015, 22, 191–214. [Google Scholar]
- Schilbach, L. Towards a second-person neuropsychiatry. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2016, 371. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, T. Phenomenology and Psychopathology. In Handbook of Phenomenology and Cognitive Science; Schmicking, D., Gallagher, S., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 546–573. [Google Scholar]
- De Jaegher, H. Embodiment and sense-making in autism. Front. Integr. Neurosci. 2013, 7, 15. [Google Scholar] [CrossRef] [PubMed]
- Schilbach, L. A second-person approach to other minds. Nat. Rev. Neurosci. 2010, 11, 449. [Google Scholar] [CrossRef] [PubMed]
- Dumas, G.; Laroche, J.; Lehmann, A. Your body, my body, our coupling moves our bodies. Front. Hum. Neurosci. 2014, 8, 1004. [Google Scholar] [CrossRef] [PubMed]
- Newen, A.; de Bruin, L.; Shaun, G. Oxford Handbook of 4E Cognition; Oxford University Press: Oxford, UK, 2016. [Google Scholar]
- Auvray, M.; Rohde, M. Perceptual crossing: The simplest online paradigm. Front. Hum. Neurosci. 2012, 6, 181. [Google Scholar] [CrossRef] [PubMed]
- Schilbach, L.; Timmermans, B.; Reddy, V.; Costall, A.; Bente, G.; Schlicht, T.; Vogeley, K. Toward a second-person neuroscience. Behav. Brain Sci. 2013, 36, 393–414. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, P.; Frazier, J.A.; Cochran, D.M.; Mitchell, T.; Coleman, C.; Schmidt, R.C. Impairments of social motor synchrony evident in autism spectrum disorder. Front. Psychol. 2016, 7, 1323. [Google Scholar] [CrossRef] [PubMed]
- Curioni, A.; Minio-Paluello, I.; Sacheli, L.M.; Candidi, M.; Aglioti, S.M. Autistic traits affect interpersonal motor coordination by modulating strategic use of role-based behavior. Mol. Autism 2017, 8, 23. [Google Scholar] [CrossRef] [PubMed]
- Peper, C.L.E.; van der Wal, S.J.; Begeer, S. Autism in Action: Reduced Bodily Connectedness during Social Interactions? Front. Psychol. 2016, 7, 1862. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, P.; Diorio, R.; Richardson, M.J.; Schmidt, R.C. Dynamical methods for evaluating the time-dependent unfolding of social coordination in children with autism. Front. Integr. Neurosci. 2013, 7, 21. [Google Scholar] [CrossRef] [PubMed]
- Froese, T.; Iizuka, H.; Ikegami, T. Embodied social interaction constitutes social cognition in pairs of humans: A minimalist virtual reality experiment. Sci. Rep. 2014, 4, 3672. [Google Scholar] [CrossRef] [PubMed]
- Kyselo, M.; Tschacher, W. An enactive and dynamical systems theory account of dyadic relationships. Front. Psychol. 2014, 5, 452. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, T. Intercorporeality and interaffectivity. Phenomenol. Mind 2016, 194–209. [Google Scholar] [CrossRef]
- De Jaegher, H.; Di Paolo, E.; Gallagher, S. Can social interaction constitute social cognition? Trends Cogn. Sci. 2010, 14, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Zapf, A.C.; Glindemann, L.A.; Vogeley, K.; Falter, C.M. Sex differences in mental rotation and how they add to the understanding of autism. PLoS ONE 2015, 10, e0124628. [Google Scholar] [CrossRef] [PubMed]
- Schwarzkopf, S.; Schilbach, L.; Vogeley, K.; Timmermans, B. “Making it explicit” makes a difference: Evidence for a dissociation of spontaneous and intentional level 1 perspective taking in high-functioning autism. Cognition 2014, 131, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Böckler, A.; Timmermans, B.; Sebanz, N.; Vogeley, K.; Schilbach, L. Effects of observing eye contact on gaze following in high-functioning autism. J. Autism Dev. Disord. 2014, 44, 1651–1658. [Google Scholar] [CrossRef] [PubMed]
- Colombetti, G. The Feeling Body: Affective Science Meets the Enactive Mind; MIT Press: Cambridge, MA, USA, 2014. [Google Scholar]
- Kojima, H.; Froese, T.; Oka, M.; Iizuka, H.; Ikegami, T. A Sensorimotor Signature of the Transition to Conscious Social Perception: Co-regulation of Active and Passive Touch. Front. Psychol. 2017, 8, 1778. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, S.; Varga, S. Social cognition and psychopathology: A critical overview. World Psychiatry 2015, 14, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, M.K.; Koch, S.C.; Fuchs, T. “We dance and find each other”1: Effects of dance/movement therapy on negative symptoms in autism spectrum disorder. Behav. Sci. (Basel) 2016, 6, 24. [Google Scholar] [CrossRef] [PubMed]
- De Jaegher, H.; Pieper, B.; Clénin, D.; Fuchs, T. Grasping intersubjectivity: An invitation to embody social interaction research. Phenomenol. Cogn. Sci. 2017, 16, 491–523. [Google Scholar] [CrossRef]
- Ciaunica, A. The ‘meeting of bodies’: Empathy and basic forms of shared experiences. Topoi 2017, 1–11. [Google Scholar] [CrossRef]
- Lenay, C. Médiations techniques des interactions perceptives: Rencontres tactiles dans les environnements numériques partagés. Soc. Sci. Inf. 2008, 47, 331–352. [Google Scholar] [CrossRef]
- Auvray, M.; Lenay, C.; Stewart, J. Perceptual interactions in a minimalist virtual environment. New Ideas Psychol. 2009, 27, 32–47. [Google Scholar] [CrossRef]
- Di Paolo, E.A.; Rohde, M.; Iizuka, H. Sensitivity to social contingency or stability of interaction? Modelling the dynamics of perceptual crossing. New Ideas Psychol. 2008, 26, 278–294. [Google Scholar] [CrossRef]
- Froese, T.; Lenay, C.; Ikegami, T. Imitation by social interaction? Analysis of a minimal agent-based model of the correspondence problem. Front. Hum. Neurosci. 2012, 6, 202. [Google Scholar] [CrossRef] [PubMed]
- Deschamps, L.; Lenay, C.; Rovira, K.; Le Bihan, G.; Aubert, D. Joint perception of a shared object: A minimalist perceptual crossing experiment. Front. Psychol. 2016, 7, 1059. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, K.; Metzler, P. Wortschatztest (WST); Beltz Test GmbH: Weinheim, Germany, 1992. [Google Scholar]
- Lehrl, S.; Triebig, G.; Fischer, B. Multiple choice vocabulary test MWT as a valid and short test to estimate premorbid intelligence. Acta Neurol. Scand. 1995, 91, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Satzger, W.; Fessmann, H.; Engel, R.R. Liefern HAWIE-R, WST und MWT-B vergleichbare IQ-Werte? Z. Differ. Diagn. Psychol. 2002, 23, 159–170. [Google Scholar] [CrossRef]
- David, N.; Aumann, C.; Bewernick, B.H.; Santos, N.S.; Lehnhardt, F.-G.; Vogeley, K. Investigation of mentalizing and visuospatial perspective taking for self and other in Asperger syndrome. J. Autism Dev. Disord. 2010, 40, 290–299. [Google Scholar] [CrossRef] [PubMed]
- Scheel, C.; Rotarska-Jagiela, A.; Schilbach, L.; Lehnhardt, F.G.; Krug, B.; Vogeley, K.; Tepest, R. Imaging derived cortical thickness reduction in high-functioning autism: Key regions and temporal slope. Neuroimage 2011, 58, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Kuzmanovic, B.; Schilbach, L.; Lehnhardt, F.-G.; Bente, G.; Vogeley, K. A matter of words: Impact of verbal and nonverbal information on impression formation in high-functioning autism. Res. Autism Spectr. Disord. 2011, 5, 604–613. [Google Scholar] [CrossRef]
- Beck, A.T.; Steer, R.A.; Brown, G.K. Beck depression inventory-II. San Antonio 1996, 78, 490–498. [Google Scholar]
- Stewart, M.E.; Barnard, L.; Pearson, J.; Hasan, R.; O’Brien, G. Presentation of depression in autism and Asperger syndrome: A review. Autism 2006, 10, 103–116. [Google Scholar] [CrossRef] [PubMed]
- Lehnhardt, F.G.; Gawronski, A.; Volpert, K.; Schilbach, L.; Tepest, R.; Huff, W.; Vogeley, K. Autism spectrum disorders in adulthood: Clinical and neuropsychological findings of Aspergers syndrome diagnosed late in life. Fortschr. Neurol. Psychiatr. 2011, 79, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Grapenne, O.; Rovira, K.; Ammar, A.; Lenay, C. Tactos: Special computer interface for the reading and writing of 2D forms in blind people. In Universal Access in HCI, Inclusive Design in the Information Society; Stephanidis, S., Ed.; Lawrence Erlbaum Associates: London, UK, 2003; pp. 1270–1274. [Google Scholar]
- Lenay, C.; Stewart, J. Minimalist approach to perceptual interactions. Front. Hum. Neurosci. 2012, 6, 98. [Google Scholar] [CrossRef] [PubMed]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Softw. 2015, 67. [Google Scholar] [CrossRef]
- Neumann, N.; Braun, C.; Dubischar-Krivec, A.M.; Bölte, S. Recognition Memory for High and Low Associative Stimuli in Autistic Individuals with Outstanding Memory Skill. Scand. J. Child Adolesc. Psychiatry Psychol. 2013, 1, 43–50. [Google Scholar] [CrossRef]
- Ragland, J.D.; Moelter, S.T.; McGrath, C.; Hill, S.K.; Gur, R.E.; Bilker, W.B.; Siegel, S.J.; Gur, R.C. Levels-of-processing effect on word recognition in schizophrenia. Biol. Psychiatry 2003, 54, 1154–1161. [Google Scholar] [CrossRef]
- Murphy, F.C.; Rubinsztein, J.S.; Michael, A.; Rogers, R.D.; Robbins, T.W.; Paykel, E.S.; Sahakian, B.J. Decision-making cognition in mania and depression. Psychol. Med. 2001, 31, 679–693. [Google Scholar] [CrossRef] [PubMed]
- Forbes, E.E.; Shaw, D.S.; Dahl, R.E. Alterations in reward-related decision making in boys with recent and future depression. Biol. Psychiatry 2007, 61, 633–639. [Google Scholar] [CrossRef] [PubMed]
- Froese, T.; Iizuka, H.; Ikegami, T. Using minimal human-computer interfaces for studying the interactive development of social awareness. Front. Psychol. 2014, 5, 1061. [Google Scholar] [CrossRef] [PubMed]
- Collier, J. Self-organization, individuation and identity. Rev. Int. Philos. 2004, 2, 151–172. [Google Scholar]
- Noy, L.; Dekel, E.; Alon, U. The mirror game as a paradigm for studying the dynamics of two people improvising motion together. Proc. Natl. Acad. Sci. USA 2011, 108, 20947–20952. [Google Scholar] [CrossRef] [PubMed]
- Brezis, R.-S.; Noy, L.; Alony, T.; Gotlieb, R.; Cohen, R.; Golland, Y.; Levit-Binnun, N. Patterns of Joint Improvisation in Adults with Autism Spectrum Disorder. Front. Psychol. 2017, 8, 1790. [Google Scholar] [CrossRef] [PubMed]
- Mengoni, S.E.; Irvine, K.; Thakur, D.; Barton, G.; Dautenhahn, K.; Guldberg, K.; Robins, B.; Wellsted, D.; Sharma, S. Feasibility study of a randomised controlled trial to investigate the effectiveness of using a humanoid robot to improve the social skills of children with autism spectrum disorder (Kaspar RCT): A study protocol. BMJ Open 2017, 7, e017376. [Google Scholar] [CrossRef] [PubMed]
- Costa, S.; Lehmann, H.; Dautenhahn, K.; Robins, B.; Soares, F. Using a Humanoid Robot to Elicit Body Awareness and Appropriate Physical Interaction in Children with Autism. Int. J. Soc. Robot. 2015, 7, 265–278. [Google Scholar] [CrossRef]
- Cusack, J.P.; Williams, J.H.G.; Neri, P. Action perception is intact in autism spectrum disorder. J. Neurosci. 2015, 35, 1849–1857. [Google Scholar] [CrossRef] [PubMed]
- Von der Lühe, T.; Manera, V.; Barisic, I.; Becchio, C.; Vogeley, K.; Schilbach, L. Interpersonal predictive coding, not action perception, is impaired in autism. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2016, 371. [Google Scholar] [CrossRef] [PubMed]
- Von Mohr, M.; Kirsch, L.P.; Fotopoulou, A. The soothing function of touch: Affective touch reduces feelings of social exclusion. Sci. Rep. 2017, 7, 13516. [Google Scholar] [CrossRef] [PubMed]
- Chatel-Goldman, J.; Congedo, M.; Jutten, C.; Schwartz, J.-L. Touch increases autonomic coupling between romantic partners. Front. Behav. Neurosci. 2014, 8, 95. [Google Scholar] [CrossRef] [PubMed]
- Torres, E.B.; Brincker, M.; Isenhower, R.W.; Yanovich, P.; Stigler, K.A.; Nurnberger, J.I.; Metaxas, D.N.; José, J.V. Autism: The micro-movement perspective. Front. Integr. Neurosci. 2013, 7, 32. [Google Scholar] [CrossRef] [PubMed]
- Torres, E.B.; Mistry, S.; Caballero, C.; Whyatt, C.P. Stochastic Signatures of Involuntary Head Micro-movements Can Be Used to Classify Females of ABIDE into Different Subtypes of Neurodevelopmental Disorders. Front. Integr. Neurosci. 2017, 11, 10. [Google Scholar] [CrossRef] [PubMed]
- Zapata-Fonseca, L.; Dotov, D.; Fossion, R.; Froese, T. Time-Series Analysis of Embodied Interaction: Movement Variability and Complexity Matching as Dyadic Properties. Front. Psychol. 2016, 7, 1940. [Google Scholar] [CrossRef] [PubMed]
- Bedia, M.G.; Aguilera, M.; Gómez, T.; Larrode, D.G.; Seron, F. Quantifying long-range correlations and 1/f patterns in a minimal experiment of social interaction. Front. Psychol. 2014, 5, 1281. [Google Scholar] [CrossRef] [PubMed]
| HFA N = 10 (5F/5M) | CTRL N = 10 (5F/5M) | Cohen’s d | |
|---|---|---|---|
| Age | 42.32 (9.20) | 43.00 (9.27) | d = 0.074 |
| WST | 113.11 (20.13) | 109.22 (12.98) | d = 0.235 |
| BDI | 14.11 (9.83) | 4.44 (2.65) | d = 1.55 |
| AQ | 40.56 (4.45) | 15.63 (3.78) | d = 6.06 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zapata-Fonseca, L.; Froese, T.; Schilbach, L.; Vogeley, K.; Timmermans, B. Sensitivity to Social Contingency in Adults with High-Functioning Autism during Computer-Mediated Embodied Interaction. Behav. Sci. 2018, 8, 22. https://doi.org/10.3390/bs8020022
Zapata-Fonseca L, Froese T, Schilbach L, Vogeley K, Timmermans B. Sensitivity to Social Contingency in Adults with High-Functioning Autism during Computer-Mediated Embodied Interaction. Behavioral Sciences. 2018; 8(2):22. https://doi.org/10.3390/bs8020022
Chicago/Turabian StyleZapata-Fonseca, Leonardo, Tom Froese, Leonhard Schilbach, Kai Vogeley, and Bert Timmermans. 2018. "Sensitivity to Social Contingency in Adults with High-Functioning Autism during Computer-Mediated Embodied Interaction" Behavioral Sciences 8, no. 2: 22. https://doi.org/10.3390/bs8020022
APA StyleZapata-Fonseca, L., Froese, T., Schilbach, L., Vogeley, K., & Timmermans, B. (2018). Sensitivity to Social Contingency in Adults with High-Functioning Autism during Computer-Mediated Embodied Interaction. Behavioral Sciences, 8(2), 22. https://doi.org/10.3390/bs8020022





