Assessing Treatment-Resistant Posttraumatic Stress Disorder: The Emory Treatment Resistance Interview for PTSD (E-TRIP)
Abstract
:1. Introduction
2. Methods
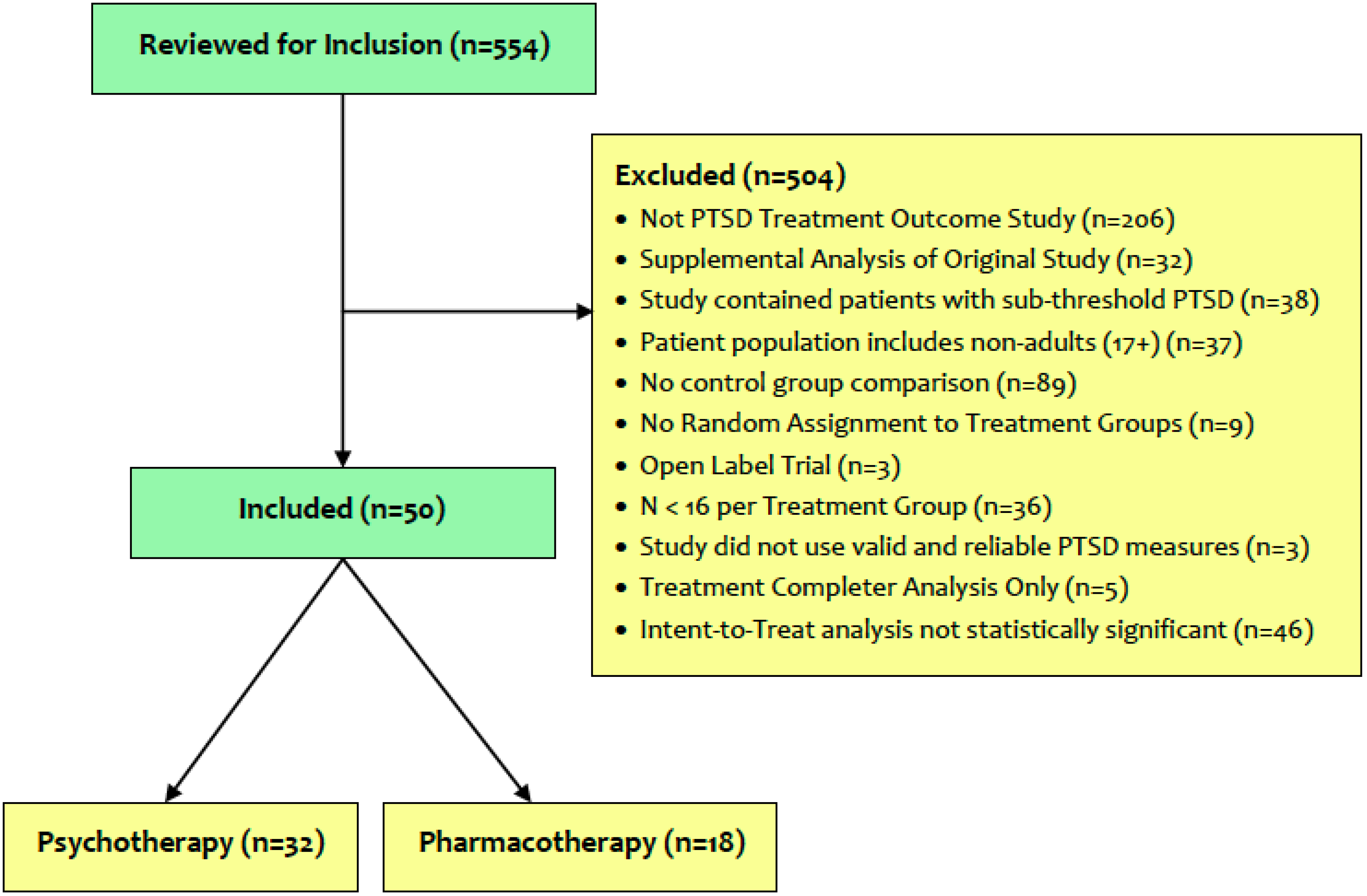
2.1. Systematic Review
| Adults (age ≥ 17) |
| Current PTSD diagnosis |
| Published in English in peer-reviewed journal |
| PTSD outcome measure with high reliability and validity |
| Randomized treatment allocation |
| If evaluating a single modality, includes a minimal control group (e.g., placebo, wait-list, or psychotherapy control condition) |
| If an augmentation treatment study (in which an experimental intervention was added to an established treatment), includes a control condition arm that did not employ the experimental intervention |
| Treatment group improves significantly more than the control group on the PTSD outcome measure at a 2-sided alpha < 0.05 for the intent-to-treat sample |
| Adequate sample size (see text) |
2.2. E-TRIP Development
2.2.1. Defining What Constitutes a Demonstrably Efficacious Treatment for PTSD
2.2.2. Differential Weighting of Treatments
- 3 points: Treatments with demonstrated efficacy in multiple (>1) RCTs.
- 2 points: Treatments with a single positive RCT.
- 1 point: Augmentation medications or combination psychotherapies with at least one positive RCT. For these treatments there are no additional weighting added for replications of efficacy.
- 0 points: Treatments without a positive RCT demonstrating its efficacy.
2.2.3. Defining the Minimum Levels of Dose and Duration that Constitute Adequate Exposure to a Treatment
2.2.4. Definition of Treatment Outcome
2.2.5. Lifetime Treatment Response versus Most Recent Episode Response
2.2.6. Distinguishing between Intolerance and Non-Response
2.2.7. Classification of Medications Individually or by Class
2.2.8. Clinical Features
2.2.9. Assessing Adherence to Prior Treatments
3. Results
3.1. Review of Available PTSD Treatment Outcome Literature
| Treatment | Points | Supporting Studies |
|---|---|---|
| SSRIs | ||
| Citalopram | 3 | |
| Escitalopram | 3 | |
| Fluoxetine | 3 | Connor et al. [36], Martenyi et al. [37] |
| Fluvoxamine | 3 | |
| Paroxetine | 3 | Marshall et al. [38,39], Tucker et al. [40], Schneier et al. [41] |
| Sertraline | 3 | Brady et al. [42], Davidson et al. [21], Panahi et al. [43] |
| Vilazodone | 3 | |
| SNRIs | ||
| Venlafaxine | 3 | Davidson et al. [22,44] |
| TCAs | ||
| Imipramine | 2 | Kosten et al. [45] |
| MAOIs | ||
| Phenelzine | 2 | Kosten et al. [45] |
| Other Antidepressants | ||
| Nefazodone | 2 | Davis et al. [46] |
| Atypical Antipsychotics | ||
| Risperidone | 1 | Bartzokis et al. [47] |
| Sedatives | ||
| Eszopiclone | 1 | Pollack et al. [18] |
| Other Medications | ||
| Prazosin | 1 | Raskind et al. [19,48] |
| Topiramate | 1 | Akuchekian and Amanant [49] |
| Treatment | Points | Supporting Studies |
|---|---|---|
| Trauma-Focused CBT | ||
| Prolonged Exposure (PE) | 3 | Cloitre et al. [50], Resick et al. [51], Foa et al. [52], Rothbaum et al. [53], Schnurr et al. [54], Mills et al. [55], Pacella et al. [56] |
| Cognitive Processing Therapy (CPT) | 3 | Resick et al. [51], Chard [57], Monson et al. [58], Forbes et al. [59] |
| Trauma-Focused Cognitive-Behavioral Therapy (TFCBT) | 3 | Ehlers et al. [60], Kubany et al. [61,62], Duffy et al. [63], Hollifield et al. [64], Cottraux et al. [65], Mueser et al. [66] |
| Internet-based Therapies | ||
| Internet-Based Cognitive-Behavioral Therapy | 3 | Knaevelsrud and Maercker [67], Litz et al. [68], Spence et al. [69] |
| Group Therapies | ||
| Group Interpersonal Therapy (IPT) | 3 | Krupnick et al. [70] |
| Cognitive-Behavioral Conjoint Therapy (CBCT) | 2 | Monson et al. [71] |
| Complementary and Alternative Medicine Therapies | ||
| Mindfulness | 2 | Niles et al. [72] |
| Acupuncture | 2 | Hollifield et al. [64] |
| Healing Touch with Guided Imagery | 2 | Jain et al. [73] |
| Other Therapies | ||
| Resiliency Intervention | 2 | Kent et al. [74] |
| Emotional Freedom Techniques (EFT) | 2 | Church et al. [75] |
| Mind-Body Bridging Program for sleep management | 2 | Nakamura et al. [76] |
| Combination Therapies | ||
| Acupoint Stimulation added to Cognitive-Behavioral Therapy | 1 | Zhang et al. [77] |
3.2. Using the E-TRIP
4. Discussions
5. Conclusions
Supplementary Files
Supplementary File 1Supplementary File 2Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Rodriguez, P.; Holowka, D.W.; Marx, B.P. Assessment of posttraumatic stress disorder-related functional impairment: A review. J. Rehabil. Res. Dev. 2012, 49, 649–666. [Google Scholar] [CrossRef] [PubMed]
- Katz, C.; Stein, M.; Richardson, J.D.; Seedat, S.; Sareen, J. A review of interventions for treatment-resistant posttraumatic stress disorder. In Different Views of Anxiety Disorders; Selek, S., Ed.; InTech: Rijeka, Croatia, 2011; pp. 251–270. [Google Scholar]
- Forbes, D.; Creamer, M.; Bisson, J.I.; Cohen, J.A.; Chow, B.E.; Foa, E.B.; Friedman, M.J.; Keane, T.M.; Kudler, H.S.; Ursano, R.J. A guide to guidelines for the treatment of PTSD and related conditions. J. Trauma. Stress 2010, 23, 537–552. [Google Scholar] [CrossRef] [PubMed]
- Foa, E.B.; Keane, T.M.; Friedman, M.J.; Cohen, J.A. Effective Treatments for PTSD: Practice Guidelines from the International Society for Traumatic Stress Studies, 2nd ed.; Guilford: New York, NY, USA, 2008. [Google Scholar]
- Lachin, J.M. Statistical considerations in the intent-to-treat principle. Control. Clin. Trials 2000, 21, 167–189. [Google Scholar] [CrossRef] [PubMed]
- Blake, D.D.; Weathers, F.W.; Nagy, L.M.; Kaloupek, D.G.; Gusman, F.D.; Charney, D.S.; Keane, T.M. The development of a clinician-administered PTSD scale. J. Trauma. Stress 1995, 8, 75–90. [Google Scholar] [PubMed]
- Schnurr, P.P.; Friedman, M.J.; Foy, D.W.; Shea, M.T.; Hsieh, F.Y.; Lavori, P.W.; Glynn, S.M.; Wattenberg, M.; Bernardy, N.C. Randomized trial of trauma-focused group therapy for posttraumatic stress disorder: Results from a Department of Veterans Affairs cooperative study. Arch. Gen. Psychiatry 2003, 60, 481–489. [Google Scholar] [CrossRef] [PubMed]
- Davidson, J.R.; Book, S.W.; Colket, J.T.; Tupler, L.A.; Roth, S.; David, D.; Hertzberg, M.; Mellman, T.; Beckham, J.C.; Smith, R.D. Assessment of a new self-rating scale for post-traumatic stress disorder. Psychol. Med. 1997, 27, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Weiss, D.S.; Marmar, C.R. The Impact of Event Scale Revised. In Assessing Psychological Trauma and PTSD–A Practitioners Handbook; Wilson, J.P., Keane, T.M., Eds.; Guilford Press: New York, NY, USA, 1997; pp. 168–169. [Google Scholar]
- Blanchard, E.B.; Jones-Alexander, J.; Buckley, T.C.; Forneris, C.A. Psychometric properties of the PTSD Checklist (PCL). Behav. Res. Ther. 1996, 34, 669–673. [Google Scholar] [CrossRef] [PubMed]
- Foa, E.B.; Cashman, L.; Jaycox, L.; Perry, K. The validation of a self-report measure of posttraumatic stress disorder: the Posttraumatic Diagnostic Scale. Psychol. Assess. 1997, 9, 445–451. [Google Scholar] [CrossRef]
- Foa, E.B.; Riggs, D.S.; Dancu, C.V.; Rothbaum, B.O. Reliability and validity of a brief instrument for assessing post-traumatic stress disorder. J. Trauma. Stress 1993, 6, 459–473. [Google Scholar] [CrossRef]
- Fava, M. Diagnosis and definition of treatment-resistant depression. Biol. Psychiatry 2003, 53, 649–659. [Google Scholar] [CrossRef] [PubMed]
- Fekadu, A.; Wooderson, S.; Donaldson, C.; Markopoulou, K.; Masterson, B.; Poon, L.; Cleare, A.J. A multidimensional tool to quantify treatment resistance in depression: The Maudsley staging method. J. Clin. Psychiatry 2009, 70, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Australian Centre for Posttraumatic Mental Health (ACPMH). Australian Guidelines for the Treatment of Adults with Acute Stress Disorder and Posttraumatic Stress Disorder; ACPMH: Melbourne, Australia, 2007. [Google Scholar]
- Friedman, M.J. Current and future drug treatment for posttraumatic stress disorder patients. Psychiatr. Ann. 1998, 28, 461–468. [Google Scholar] [CrossRef]
- Martenyi, F.; Brown, E.B.; Zhang, H.; Koke, S.C.; Prakash, A. Fluoxetine v. placebo in prevention of relapse in post-traumatic stress disorder. Br. J. Psychiatry 2002, 181, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Pollack, M.H.; Hoge, E.A.; Worthington, J.J.; Moshier, S.J.; Wechsler, R.S.; Brandes, M.; Simon, N.M. Eszopiclone for the treatment of posttraumatic stress disorder and associated insomnia: A randomized, double-blind, placebo-controlled trial. J. Clin. Psychiatry 2011, 72, 892–897. [Google Scholar] [CrossRef] [PubMed]
- Raskind, M.A.; Peterson, K.; Williams, T.; Hoff, D.J.; Hart, K.; Holmes, H.; Homas, D.; Hill, J.; Daniels, C.; Calohan, J. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am. J. Psychiatry 2013, 170, 1003–1010. [Google Scholar] [CrossRef] [PubMed]
- Benedek, D.M.; Friedman, M.J.; Zatzick, D.; Ursano, R.J. Guideline Watch (March 2009): Practice Guideline for the Treatment of Patients with Acute Stress Disorder and Posttraumatic Stress Disorder; American Psychiatric Association: Arlington, MA, USA, 2009. [Google Scholar]
- Davidson, J.R.; Rothbaum, B.O.; van der Kolk, B.A.; Sikes, C.R.; Farfel, G.M. Multicenter, double-blind comparison of sertraline and placebo in the treatment of posttraumatic stress disorder. Arch. Gen. Psychiatry 2001, 58, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Davidson, J.; Rothbaum, B.O.; Tucker, P.; Asnis, G.; Benattia, I.; Musgnung, J.J. Venlafaxine extended release in posttraumatic stress disorder: A sertraline-and placebo-controlled study. J. Clin. Psychopharmacol. 2006, 26, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Glenn, D.; Golinelli, D.; Rose, R.D.; Roy-Byrne, P.; Stein, M.B.; Sullivan, G.; Bystritksy, A.; Sherbourne, C.; Craske, M.G. Who gets the most out of cognitive behavioral therapy for anxiety disorders? The role of treatment dose and patient engagement. J. Consult. Clin. Psychol. 2013, 81, 639–649. [Google Scholar] [CrossRef] [PubMed]
- Foa, E.B.; Yusko, D.A.; McLean, C.P.; Suvak, M.K.; Bux, D.A., Jr.; Oslin, D.; O’Brien, C.P.; Imms, P.; Riggs, D.S.; Volpicelli, J. Concurrent naltrexone and prolonged exposure therapy for patients with comorbid alcohol dependence and PTSD: A randomized clinical trial. JAMA 2013, 310, 488–495. [Google Scholar] [CrossRef] [PubMed]
- Hamner, M.B.; Robert, S.; Frueh, B.C. Treatment-resistant posttraumatic stress disorder: Strategies for intervention. CNS Spectr. 2004, 9, 740–752. [Google Scholar] [PubMed]
- Guy, W. Clinical Global Impressions. In ECDEU Assessment Manual for Psychopharmacology, Revised; US Department of Health, Education and Welfare, National Institute of Mental Health: Bethesda, MD, USA, 1976; pp. 217–222. [Google Scholar]
- Foa, E.B.; Dancu, C.V.; Hembree, E.A.; Jaycox, L.H.; Meadows, E.A.; Street, G.P. A comparison of exposure therapy, stress inoculation training, and their combination for reducing posttraumatic stress disorder in female assault victims. J. Consult. Clin. Psychol. 1999, 67, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Simon, G.E.; Rutter, C.M.; Stewart, C.; Pabiniak, C.; Wehnes, L. Response to past depression treatments is not accurately recalled: comparison of structured recall and patient health questionnaire scores in medical records. J. Clin. Psychiatry 2012, 73, 1503–1508. [Google Scholar] [CrossRef] [PubMed]
- Desseilles, M.; Witte, J.; Chang, T.E.; Iovieno, N.; Dording, C.M.; Ashih, H.; Nyer, M.; Freeman, M.P.; Fava, M.; Mischoulon, D. Assessing the adequacy of past antidepressant trials: A clinician’s guide to the antidepressant treatment response questionnaire. J. Clin. Psychiatry 2011, 72, 1152–1154. [Google Scholar] [CrossRef] [PubMed]
- Ipser, J.C.; Stein, D.J. Evidence-based pharmacotherapy of post-traumatic stress disorder (PTSD). Int. J. Neuropsychopharmacol. 2012, 15, 825–840. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association (APA). Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; APA: Arlington, MA, USA, 2013. [Google Scholar]
- Williams, M.T.; Cahill, S.P.; Foa, E.B. Psychotherapy for posttraumatic stress disorder. In Textbook of Anxiety Disorders, 2nd ed.; Stein, D.J., Hollander, E., Rothbaum, B.O., Eds.; American Psychiatric Publishing: Arlington, MA, USA, 2009; pp. 603–628. [Google Scholar]
- Lockwood, A.; Steinke, D.T.; Botts, S.R. Medication adherence and its effect on relapse among patients discharged from a Veterans Affairs posttraumatic stress disorder treatment program. Ann. Pharmacother. 2009, 43, 1227–1232. [Google Scholar] [CrossRef] [PubMed]
- Bulloch, A.G.; Patten, S.B. Non-adherence with psychotropic medications in the general population. Soc. Psychiatry Psychiatr. Epidemiol. 2010, 45, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Osterberg, L.; Blaschke, T. Adherence to medication. N. Engl. J. Med. 2005, 353, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Connor, K.M.; Sutherland, S.M.; Tupler, L.A.; Malik, M.L.; Davidson, J.R. Fluoxetine in post-traumatic stress disorder: Randomised, double-blind study. Br. J. Psychiatry 1999, 175, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Martenyi, F.; Brown, E.B.; Zhang, H.; Prakash, A; Koke, S.C. Fluoxetine versus placebo in posttraumatic stress disorder. J. Clin. Psychiatry 2002, 63, 199–206. [Google Scholar] [CrossRef]
- Marshall, R.D.; Beebe, K.L.; Oldham, M.; Zaninelli, R. Efficacy and safety of paroxetine treatment for chronic PTSD: A fixed-dose, placebo-controlled study. Am. J. Psychiatry 2001, 158, 1982–1988. [Google Scholar] [CrossRef] [PubMed]
- Marshall, R.D.; Lewis‐Fernandez, R.; Blanco, C.; Simpson, H.B.; Lin, S.; Vermes, D.; Garcia, W.; Schneier, F.; Neria, Y.; Sanchez-Lacay, A.; et al. A controlled trial of paroxetine for chronic PTSD, dissociation, and interpersonal problems in mostly minority adults. Depress. Anxiety 2007, 24, 77–84. [Google Scholar] [CrossRef]
- Tucker, P.; Zaninelli, R.; Yehuda, R.; Ruggiero, L.; Dillingham, K.; Pitts, C.D. Paroxetine in the treatment of chronic posttraumatic stress disorder: Results of a placebo-controlled, flexible-dosage trial. J. Clin. Psychiatry 2001, 62, 860–868. [Google Scholar] [CrossRef] [PubMed]
- Schneier, F.R.; Neria, Y.; Pavlicova, M.; Hembree, E.; Suh, E.J.; Amsel, L.; Marshall, R.D. Combined prolonged exposure therapy and paroxetine for PTSD related to the World Trade Center attack: A randomized controlled trial. Am. J. Psychiatry 2012, 169, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Brady, K.; Pearlstein, T.; Asnis, G.M.; Baker, D.; Rothbaum, B.; Sikes, C.R.; Farfel, G.M. Efficacy and safety of sertraline treatment of posttraumatic stress disorder. JAMA 2000, 283, 1837–1844. [Google Scholar] [CrossRef] [PubMed]
- Panahi, Y.; Moghaddam, B.R.; Sahebkar, A.; Nazari, M.A.; Beiraghdar, F.; Karami, G.; Saadat, A.R. A randomized, double-blind, placebo-controlled trial on the efficacy and tolerability of sertraline in Iranian veterans with post-traumatic stress disorder. Psychol. Med. 2011, 41, 2159–2166. [Google Scholar] [CrossRef] [PubMed]
- Davidson, J.; Baldwin, D.; Stein, D.J.; Kuper, E.; Benattia, I.; Ahmed, S.; Pedersen, R.; Musgnung, J. Treatment of posttraumatic stress disorder with venlafaxine extended release: A 6-month randomized controlled trial. Arch. Gen. Psychiatry 2006, 63, 1158–1165. [Google Scholar] [CrossRef] [PubMed]
- Kosten, T.R.; Frank, J.B.; Dan, E.; McDougle, C.J.; Giller, E., Jr. Pharmacotherapy for posttraumatic stress disorder using phenelzine or imipramine. J. Nerv. Ment. Dis. 1991, 179, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Davis, L.L.; Jewell, M.E.; Ambrose, S.; Farley, J.; English, B.; Bartolucci, A.; Petty, F. A placebo-controlled study of nefazodone for the treatment of chronic posttraumatic stress disorder: A preliminary study. J. Clin. Psychopharmacol. 2004, 24, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Bartzokis, G.L.; Po, H.; Turner, J.; Mintz, J.; Saunders, C.S. Adjunctive risperidone in the treatment of chronic combat-related posttraumatic stress disorder. Biol. Psychiatry 2005, 57, 474–479. [Google Scholar] [CrossRef] [PubMed]
- Raskind, M.A.; Peskind, E.R.; Kanter, E.D.; Petrie, E.C.; Radant, A.; Thompson, C.E.; Dobie, D.J.; Hoff, D.; Rein, R.J.; Straits-Troster, K.; et al. Reduction of nightmares and other PTSD symptoms in combat veterans by prazosin: A placebo controlled study. Am. J. Psychiatry 2003, 160, 371–373. [Google Scholar] [CrossRef]
- Akuchekian, S.; Amanat, S. The comparison of topiramate and placebo in the treatment of posttraumatic stress disorder: A randomized, double-blind study. J. Res. Med. Sci. 2004, 9, 240–244. [Google Scholar]
- Cloitre, M.; Koenen, K.C.; Cohen, L.R.; Han, H. Skills training in affective and interpersonal regulation followed by exposure: A phase-based treatment for PTSD related to childhood abuse. J. Consult. Clin. Psychol. 2002, 70, 1067–1074. [Google Scholar] [CrossRef] [PubMed]
- Resick, P.A.; Nishith, P.; Weaver, T.L.; Astin, M.C.; Feuer, C.A. A comparison of cognitive-processing therapy with prolonged exposure and a waiting condition for the treatment of chronic posttraumatic stress disorder in female rape victims. J. Consult. Clin. Psychol. 2002, 70, 867–879. [Google Scholar] [CrossRef] [PubMed]
- Foa, E.B.; Hembree, E.A.; Cahill, S.P.; Rauch, S.A.M.; Riggs, D.S.; Feeny, N.C.; Yadin, E. Randomized trial of prolonged exposure for posttraumatic stress disorder with and without cognitive restructuring: Outcome at academic and community clinics. J. Consult. Clin. Psychol. 2005, 73, 953–964. [Google Scholar] [CrossRef] [PubMed]
- Rothbaum, B.O.; Astin, M.C.; Marsteller, F. Prolonged exposure versus eye movement desensitization and reprocessing (EMDR) for PTSD rape victims. J. Trauma. Stress 2005, 18, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Schnurr, P.P.; Friedman, M.J.; Engel, C.C.; Foa, E.B.; Shea, T.; Chow, B.K.; Resick, P.A.; Thurston, V.; Orsillo, S.M.; Haug, R.; et al. Cognitive behavioral therapy for posttraumatic stress disorder in women. JAMA 2007, 297, 820–830. [Google Scholar] [CrossRef] [PubMed]
- Mills, K.L.; Teesson, M.; Back, S.E.; Brady, K.T.; Baker, A.L.; Hopwood, S.; Sannibale, C.; Barrett, E.L.; Merz, S.; Rosenfeld, J.; et al. Integrated exposure-based therapy for co-occurring posttraumatic stress disorder and substance dependence: A randomized controlled trial: Exposure therapy for PTSD and substance dependence. JAMA 2012, 308, 690–699. [Google Scholar] [PubMed]
- Pacella, M.L.; Armelie, A.; Boarts, J.; Wagner, G.; Jones, T.; Feeny, N.; Delahanty, D.L. The impact of prolonged exposure on PTSD symptoms and associated psychopathology in people living with HIV: A randomized test of concept. AIDS Behav. 2012, 16, 1327–1340. [Google Scholar] [CrossRef] [PubMed]
- Chard, K.M. An evaluation of cognitive processing therapy for the treatment of posttraumatic stress disorder related to childhood sexual abuse. J. Consult. Clin. Psychol. 2005, 73, 965–971. [Google Scholar] [CrossRef] [PubMed]
- Monson, C.M.; Schnurr, P.P.; Resick, P.A.; Friedman, M.J.; Young-Xu, Y.; Stevens, S.P. Cognitive processing therapy for veterans with military-related posttraumatic stress disorder. J. Consult. Clin. Psychol. 2006, 74, 898–907. [Google Scholar] [CrossRef] [PubMed]
- Forbes, D.; Lloyd, D.; Nixon, R.D.V.; Elliott, P.; Varker, T.; Perry, D.; Bryant, R.A.; Creamer, M. A multisite randomized controlled effectiveness trial of cognitive processing therapy for military-related posttraumatic stress disorder. J. Anxiety Disord. 2012, 26, 442–452. [Google Scholar] [CrossRef] [PubMed]
- Ehlers, A.; Clark, D.M.; Hackmann, A.; McManus, F.; Fennell, M.; Herbert, C.; Mayou, R. A randomized controlled trial of cognitive therapy, a self-help booklet, and repeated assessments as early interventions for posttraumatic stress disorder. Arch. Gen. Psychiatry 2003, 60, 1024–1032. [Google Scholar] [CrossRef] [PubMed]
- Kubany, E.S.; Hill, E.E.; Owens, J.A. Cognitive trauma therapy for battered women with PTSD: Preliminary findings. J. Trauma. Stress 2003, 16, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Kubany, E.S.; Hill, E.E.; Owens, J.A.; Iannce-Spencer, C.; McCaig, M.A.; Tremayne, K.J.; Williams, P.L. Cognitive trauma therapy for battered women with PTSD (CTT-BW). J. Consult. Clin. Psychol. 2004, 72, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Duffy, M.; Gillespie, K.; Clark, D.M. Post-traumatic stress disorder in the context of terrorism and other civil conflict in Northern Ireland: randomised controlled trial. Br. Med. J. 2007, 334, 1147–1150. [Google Scholar] [CrossRef]
- Hollifield, M.; Sinclair-Lian, N.; Warner, T.D.; Hammerschlag, R. Acupuncture for posttraumatic stress disorder: A randomized controlled pilot trial. J. Nerv. Ment. Dis. 2007, 195, 504–513. [Google Scholar] [CrossRef] [PubMed]
- Cottraux, J.; Note, I.; Yao, S.N.; de Mey-Guillard, C.; Bonasse, F.; Djamoussian, D.; Mollard, E.; Note, B.; Chen, Y. Randomized controlled comparison of cognitive behavior therapy with Rogerian supportive therapy in chronic post-traumatic stress disorder: A 2-year follow-up. Psychother. Psychosom. 2008, 77, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Mueser, K.T.; Rosenberg, S.D.; Xie, H.; Jankowski, M.K.; Bolton, E.E.; Lu, W.; Hamblen, J.L.; Rosenberg, H.J.; McHugo, G.J.; Wolfe, R. A randomized controlled trial of cognitive-behavioral treatment for posttraumatic stress disorder in severe mental illness. J. Consult. Clin. Psychol. 2008, 76, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Knaevelsrud, C.; Maercker, A. Internet-based treatment for PTSD reduces distress and facilitates the development of a strong therapeutic alliance: A randomized controlled clinical trial. BMC Psychiatry 2007, 7. [Google Scholar] [CrossRef] [Green Version]
- Litz, B.; Engel, C.; Bryant, R.; Papa, A. A randomized, controlled proof-of-concept trial of an Internet-based, therapist-assisted self-management treatment for posttraumatic stress disorder. Am. J. Psychiatry 2007, 164, 1676–1684. [Google Scholar] [CrossRef] [PubMed]
- Spence, J.; Titov, N.; Dear, B.F.; Johnston, L.; Solley, K.; Lorian, C.; Wootton, B.; Zou, J.; Schwenke, G. Randomized controlled trial of Internet‐delivered cognitive behavioral therapy for posttraumatic stress disorder. Depress. Anxiety 2011, 28, 541–550. [Google Scholar] [CrossRef] [PubMed]
- Krupnick, J.L.; Green, B.L.; Stockton, P.; Miranda, J.; Krause, E.; Mete, M. Group interpersonal psychotherapy for low-income women with posttraumatic stress disorder. Psychother. Res. 2008, 18, 497–507. [Google Scholar] [CrossRef] [PubMed]
- Monson, C.M.; Fredman, S.J.; Macdonald, A.; Pukay-Martin, N.D.; Resick, P.A.; Schnurr, P.P. Effect of cognitive-behavioral couple therapy for PTSD: A randomized controlled trial: Cognitive-behavioral couple therapy for PTSD. JAMA 2012, 308, 700–709. [Google Scholar] [CrossRef] [PubMed]
- Niles, B.L.; Klunk-Gillis, J.; Ryngala, D.J.; Silberbogen, A.K.; Paysnick, A.; Wolf, E.J. Comparing mindfulness and psychoeducation treatments for combat-related PTSD using a telehealth approach. Psychol. Trauma 2012, 4, 538–547. [Google Scholar] [CrossRef]
- Jain, S.; McMahon, G.F.; Hasen, P.; Kozub, M.P.; Porter, V.; King, R.; Guarneri, E.M. Healing Touch with Guided Imagery for PTSD in returning active duty military: A randomized controlled trial. Mil. Med. 2012, 177, 1015–1021. [Google Scholar] [CrossRef] [PubMed]
- Kent, M.; Davis, M.C.; Stark, S.L.; Stewart, L.A. A resilience‐oriented treatment for posttraumatic stress disorder: Results of a preliminary randomized clinical trial. J. Trauma. Stress 2011, 24, 591–595. [Google Scholar] [CrossRef] [PubMed]
- Church, D.; Hawk, C.; Brooks, A.J.; Toukolehto, O.; Wren, M.; Dinter, I.; Stein, P. Psychological trauma symptom improvement in veterans using Emotional Freedom Techniques: A randomized controlled trial. J. Nerv. Ment. Dis. 2013, 201, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Lipschitz, D.L.; Landward, R.; Kuhn, R.; West, G. Two sessions of sleep-focused mind–body bridging improve self-reported symptoms of sleep and PTSD in veterans: A pilot randomized controlled trial. J. Psychosom. Res. 2011, 70, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Feng, B.; Xie, J.; Xu, F.; Chen, J. Clinical study on treatment of the earthquake-caused post-traumatic stress disorder by cognitive-behavior therapy and acupoint stimulation. J. Tradit. Chin. Med. 2011, 31, 60–63. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dunlop, B.W.; Kaye, J.L.; Youngner, C.; Rothbaum, B. Assessing Treatment-Resistant Posttraumatic Stress Disorder: The Emory Treatment Resistance Interview for PTSD (E-TRIP). Behav. Sci. 2014, 4, 511-527. https://doi.org/10.3390/bs4040511
Dunlop BW, Kaye JL, Youngner C, Rothbaum B. Assessing Treatment-Resistant Posttraumatic Stress Disorder: The Emory Treatment Resistance Interview for PTSD (E-TRIP). Behavioral Sciences. 2014; 4(4):511-527. https://doi.org/10.3390/bs4040511
Chicago/Turabian StyleDunlop, Boadie W., Joanna L. Kaye, Cole Youngner, and Barbara Rothbaum. 2014. "Assessing Treatment-Resistant Posttraumatic Stress Disorder: The Emory Treatment Resistance Interview for PTSD (E-TRIP)" Behavioral Sciences 4, no. 4: 511-527. https://doi.org/10.3390/bs4040511
APA StyleDunlop, B. W., Kaye, J. L., Youngner, C., & Rothbaum, B. (2014). Assessing Treatment-Resistant Posttraumatic Stress Disorder: The Emory Treatment Resistance Interview for PTSD (E-TRIP). Behavioral Sciences, 4(4), 511-527. https://doi.org/10.3390/bs4040511





