Infant Motor Competence Predicts Executive Functions in Preschoolers: The Role of Sleep
Abstract
1. Introduction
1.1. Infant Motor Competence and Later Cognitive Development
1.2. Infant Motor Competence and Executive Functions in Preschool Years
1.3. Sleep as a Moderator
1.4. The Present Study
2. Methods
2.1. Participants and Procedures
2.2. Measurements
2.2.1. Motor Competence and Cognitive Ability
2.2.2. Executive Functions
2.2.3. Sleep Quality and Quantity
2.2.4. Covariates
2.3. Statistical Analysis
3. Results
3.1. Preliminary Analysis and Descriptive Statistics
3.2. Measurement Model
3.3. Moderated Mediation Model
3.3.1. Mediation Analysis
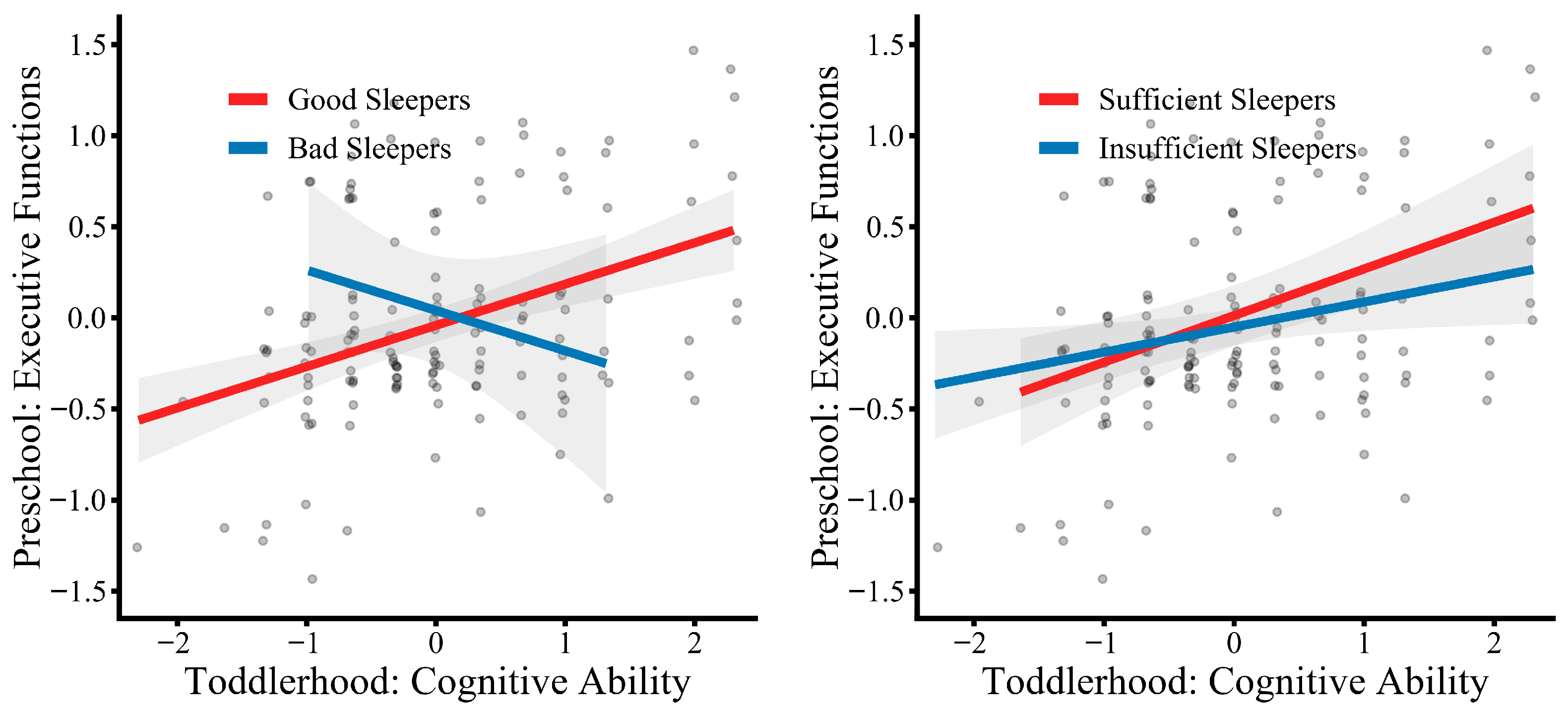
3.3.2. Moderated Mediation Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Variable | Mother | Father |
|---|---|---|
| Monthly income (Scored from 1~7) | % | % |
| 1. <1500 RMB | 3.5 | 0 |
| 2. 1500~3000 RMB | 3.5 | 0.4 |
| 3. 3000~6000 RMB | 23.9 | 12.2 |
| 4. 6000~10,000 RMB | 21.6 | 27.1 |
| 5. 10,000~15,000 RMB | 10.6 | 12.5 |
| 6. 15,000~20,000 RMB | 6.3 | 7.8 |
| 7. >20,000 RMB | 4.7 | 17.6 |
| Missing | 25.9 | 22.4 |
| Parental education (Scored from 1~4) | ||
| 1. Middle school or below | 0.8 | 0.4 |
| 2. High school or vocational school degree | 4.3 | 4.3 |
| 3. Bachelor’s degree or vocational college degree | 51.8 | 48.6 |
| 4. Master’s degree or above | 24.7 | 25.5 |
| Missing | 18.4 | 21.2 |
References
- Adolph, K. E., Berger, S. E., & Leo, A. J. (2011). Developmental continuity? Crawling, cruising, and walking. Developmental Science, 14(2), 306–318. [Google Scholar] [CrossRef] [PubMed]
- Adolph, K. E., & Hoch, J. E. (2020). The importance of motor skills for development. In Building future health and well-being of thriving toddlers and young children (Vol. 95, pp. 136–144). S. Karger AG. [Google Scholar] [CrossRef]
- Astill, R. G., van der Heijden, K. B., van IJzendoorn, M. H., & van Someren, E. J. W. (2012). Sleep, cognition, and behavioral problems in school-age children: A century of research meta-analyzed. Psychological Bulletin, 138(6), 1109–1138. [Google Scholar] [CrossRef]
- Bayley, N. (2006). Bayley scales of infant and toddler development, third edition: Administration manual. The Psychological Corporation. [Google Scholar]
- Berger, S. E., & Scher, A. (2017). Naps improve new walkers’ locomotor problem solving. Journal of Experimental Child Psychology, 162, 292–300. [Google Scholar] [CrossRef]
- Bernier, A., Cimon-Paquet, C., & Tétreault, É. (2021). Sleep development in preschool predicts executive functioning in early elementary school. In Advances in child development and behavior (Vol. 60, pp. 159–178). Elsevier. [Google Scholar] [CrossRef]
- Cameron, C. E., Brock, L. L., Murrah, W. M., Bell, L. H., Worzalla, S. L., Grissmer, D., & Morrison, F. J. (2012). Fine motor skills and executive function both contribute to kindergarten achievement. Child Development, 83(4), 1229–1244. [Google Scholar] [CrossRef]
- Campos, J. J., Anderson, D. I., Barbu-Roth, M. A., Hubbard, E. M., Hertenstein, M. J., & Witherington, D. (2000). Travel broadens the mind. Infancy, 1(2), 149–219. [Google Scholar] [CrossRef]
- Carlson, S. M. (2005). Developmentally sensitive measures of executive function in preschool children. Developmental Neuropsychology, 28(2), 595–616. [Google Scholar] [CrossRef]
- Cattuzzo, M. T., dos Santos Henrique, R., Nicolai Ré, A. H., de Oliveira, I. S., Melo, B. M., de Sousa Moura, S., de Araújo, R. C., & Stodden, D. (2016). Motor competence and health related physical fitness in youth: A systematic review. Journal of Science and Medicine in Sport, 19(2), 123–129. [Google Scholar] [CrossRef]
- Cook, C. J., Howard, S. J., Scerif, G., Twine, R., Kahn, K., Norris, S. A., & Draper, C. E. (2019). Associations of physical activity and gross motor skills with executive function in preschool children from low-income South African settings. Developmental Science, 22(5), e12820. [Google Scholar] [CrossRef] [PubMed]
- Davis, R., Phillips, R., Roscoe, J., & Roscoe, D. (2000). The nature and classification of skill. In Physical educational and study of sports (pp. 284–285). Harcourt Publishers. [Google Scholar]
- Dewald, J. F., Meijer, A. M., Oort, F. J., Kerkhof, G. A., & Bögels, S. M. (2010). The influence of sleep quality, sleep duration, and sleepiness on school performance in children and adolescents: A meta-analytic review. Sleep Medicine Reviews, 14(3), 179–189. [Google Scholar] [CrossRef]
- Diamond, A. (2013). Executive Functions. Annual Review of Psychology, 64(1), 135–168. [Google Scholar] [CrossRef] [PubMed]
- Diamond, A., Kirkham, N., & Amso, D. (2002). Conditions under which young children can hold two rules in mind and inhibit a prepotent response. Developmental Psychology, 38(3), 352–362. [Google Scholar] [CrossRef]
- Enders, C. K., & Bandalos, D. L. (2001). The relative performance of full information maximum likelihood estimation for missing data in structural equation models. Structural Equation Modeling: A Multidisciplinary Journal, 8(3), 430–457. [Google Scholar] [CrossRef]
- Fangupo, L. J., Haszard, J. J., Reynolds, A. N., Lucas, A. W., McIntosh, D. R., Richards, R., Camp, J., Galland, B. C., Smith, C., & Taylor, R. W. (2021). Do sleep interventions change sleep duration in children aged 0–5 years? A systematic review and meta-analysis of randomised controlled trials. Sleep Medicine Reviews, 59, 101498. [Google Scholar] [CrossRef] [PubMed]
- Flensborg-Madsen, T., & Mortensen, E. L. (2018). Developmental milestones during the first three years as precursors of adult intelligence. Developmental Psychology, 54(8), 1434–1444. [Google Scholar] [CrossRef] [PubMed]
- Floyer-Lea, A., & Matthews, P. M. (2004). Changing brain networks for visuomotor control with increased movement automaticity. Journal of Neurophysiology, 92(4), 2405–2412. [Google Scholar] [CrossRef]
- Gandotra, A., Csaba, S., Sattar, Y., Cserényi, V., Bizonics, R., Cserjesi, R., & Kotyuk, E. (2022). A meta-analysis of the relationship between motor skills and executive functions in typically-developing children. Journal of Cognition and Development, 23(1), 83–110. [Google Scholar] [CrossRef]
- Goodlin-Jones, B. L., Tang, K., Liu, J., & Anders, T. F. (2008). Sleep patterns in preschool-age children with autism, developmental delay, and typical development. Journal of the American Academy of Child & Adolescent Psychiatry, 47(8), 930–938. [Google Scholar] [CrossRef]
- Hayes, A. F. (2013). Introduction to mediation, moderation, and conditional process analysis: A regression-based approach. Guilford Press. [Google Scholar]
- Hongwanishkul, D., Happaney, K. R., Lee, W. S. C., & Zelazo, P. D. (2005). Assessment of hot and cool executive function in young children: Age-related changes and individual differences. Developmental Neuropsychology, 28(2), 617–644. [Google Scholar] [CrossRef]
- Hua, J., Lyu, J., & Du, W. (2022). Prevalence of sleep disorder in Chinese preschoolers: A national population-based study. Nature and Science of Sleep, 14, 2091–2095. [Google Scholar] [CrossRef]
- Ito, M. (1993). Movement and thought: Identical control mechanisms by the cerebellum. Trends in Neurosciences, 16(11), 448–450. [Google Scholar] [CrossRef]
- Jose, P. E. (2013). Doing statistical mediation and moderation. The Guilford Press. [Google Scholar]
- Karpinski, A. C., Scullin, M. H., & Montgomery-Downs, H. E. (2008). Risk for sleep-disordered breathing and executive function in preschoolers. Sleep Medicine, 9, 418–424. [Google Scholar] [CrossRef]
- Kim, H., Duran, C. A. K., Cameron, C. E., & Grissmer, D. (2018). Developmental relations among motor and cognitive processes and mathematics skills. Child Development, 89(2), 476–494. [Google Scholar] [CrossRef]
- Koziol, L. F., Budding, D. E., & Chidekel, D. (2012). From movement to thought: Executive function, embodied cognition, and the cerebellum. The Cerebellum, 11(2), 505–525. [Google Scholar] [CrossRef]
- Koziol, L. F., & Lutz, J. T. (2013). From movement to thought: The development of executive function. Applied Neuropsychology: Child, 2(2), 104–115. [Google Scholar] [CrossRef]
- Lawson, G. M., Hook, C. J., & Farah, M. J. (2018). A meta-analysis of the relationship between socioeconomic status and executive function performance among children. Developmental Science, 21(2), e12529. [Google Scholar] [CrossRef] [PubMed]
- Li, S., Jin, X., Shen, X., Wu, S., Jiang, F., Yan, C., Yu, X., & Qiu, Y. (2007). Development and psychometric properties of the Chinese version of children’s sleep habits questionnaire. Chinese Journal of Pediatrics, 45(3), 176–180. [Google Scholar] [PubMed]
- Liang, X., Zhang, X., Wang, Y., van IJzendoorn, M. H., & Wang, Z. (2022). Sleep problems and infant motor and cognitive development across the first two years of life: The Beijing longitudinal study. Infant Behavior and Development. [Google Scholar] [CrossRef]
- Little, R. J. A. (1988). A test of missing completely at random for multivariate data with missing values. Journal of the American Statistical Association, 83(404), 1198–1202. [Google Scholar] [CrossRef]
- Liu, J., Zhou, G., Wang, Y., Ai, Y., Pinto-Martin, J., & Liu, X. (2012). Sleep problems, fatigue, and cognitive performance in Chinese kindergarten children. The Journal of Pediatrics, 161(3), 520–525.e2. [Google Scholar] [CrossRef]
- Luo, Z., Jose, P. E., Huntsinger, C. S., & Pigott, T. D. (2007). Fine motor skills and mathematics achievement in East Asian American and European American kindergartners and first graders. British Journal of Developmental Psychology, 25(4), 595–614. [Google Scholar] [CrossRef]
- MacKinnon, D. P., Lockwood, C. M., & Williams, J. (2004). Confidence limits for the indirect effect: Distribution of the product and resampling methods. Multivariate Behavioral Research, 39(1), 99–128. [Google Scholar] [CrossRef] [PubMed]
- Marcinowski, E. C., Tripathi, T., Hsu, L., McCoy, S. W., & Dusing, S. C. (2019). Sitting skill and the emergence of arms-free sitting affects the frequency of object looking and exploration. Developmental Psychobiology, 61(7), 1035–1047. [Google Scholar] [CrossRef] [PubMed]
- Martzog, P., & Suggate, S. P. (2019). Fine motor skills and mental imagery: Is it all in the mind? Journal of Experimental Child Psychology, 186, 59–72. [Google Scholar] [CrossRef]
- Mason, G. M., Lokhandwala, S., Riggins, T., & Spencer, R. M. C. (2021). Sleep and human cognitive development. Sleep Medicine Reviews, 57, 101472. [Google Scholar] [CrossRef]
- Maurer, M. N., & Roebers, C. M. (2019). Towards a better understanding of the association between motor skills and executive functions in 5- to 6-year-olds: The impact of motor task difficulty. Human Movement Science, 66, 607–620. [Google Scholar] [CrossRef] [PubMed]
- Mäkelä, T. E., Peltola, M. J., Saarenpää-Heikkilä, O., Himanen, S., Paunio, T., Paavonen, E. J., & Kylliäinen, A. (2020). Night awakening and its association with executive functioning across the first two years of life. Child Development, 91(4), e937–e951. [Google Scholar] [CrossRef]
- McClelland, M. M., & Cameron, C. E. (2019). Developing together: The role of executive function and motor skills in children’s early academic lives. Early Childhood Research Quarterly, 46, 142–151. [Google Scholar] [CrossRef]
- Miyake, A., Friedman, N. P., Emerson, M. J., Witzki, A. H., Howerter, A., & Wager, T. D. (2000). The unity and diversity of executive functions and their contributions to complex “frontal lobe” tasks: A latent variable analysis. Cognitive Psychology, 41(1), 49–100. [Google Scholar] [CrossRef]
- Morales-Muñoz, I., Nolvi, S., Mäkelä, T., Eskola, E., Korja, R., Fernandes, M., Karlsson, H., Paavonen, E. J., & Karlsson, L. (2021). Sleep during infancy, inhibitory control and working memory in toddlers: Findings from the FinnBrain cohort study. Sleep Science and Practice, 5(1), 13. [Google Scholar] [CrossRef]
- Morgan, P. L., Farkas, G., Hillemeier, M. M., Pun, W. H., & Maczuga, S. (2019). Kindergarten children’s executive functions predict their second-grade academic achievement and behavior. Child Development, 90(5), 1802–1816. [Google Scholar] [CrossRef]
- Muthén, K. L., & Muthén, B. O. (2017). Mplus user’s guide (8th ed.). Muthén & Muthén. [Google Scholar]
- National Health and Family Planning Commission in China (NHFPCC). (2017). Guideline for sleep hygiene among children aged 0~5 years. Available online: https://www.nhc.gov.cn/ewebeditor/uploadfile/2017/10/20171026154305316.pdf (accessed on 11 June 2023).
- Nelson, T. D., Johnson, A. B., Ramsdell, E. L., & Brock, R. L. (2020). Individual differences in the effects of child sleep problems on early executive functioning. In Advances in child development and behavior (Vol. 60, pp. 179–195). Elsevier. [Google Scholar] [CrossRef]
- Nieto, M., Motos, B., Navarro, B., Jimeno, M. V., Fernández-Aguilar, L., Ros, L., Ricarte, J. J., & Latorre, J. M. (2022). Relation between nighttime sleep duration and executive functioning in a nonclinical sample of preschool children. Scandinavian Journal of Psychology, 63(3), 191–198. [Google Scholar] [CrossRef]
- Owens, J. A., Spirito, A., & McGuinn, M. (2000). The children’s sleep habits questionnaire (CSHQ): Psychometric properties of a survey instrument for school-aged children. Sleep, 23(8), 1–9. [Google Scholar] [CrossRef]
- Paruthi, S., Brooks, L. J., D’Ambrosio, C., Hall, W. A., Kotagal, S., Lloyd, R. M., Malow, B. A., Maski, K., Nichols, C., Quan, S. F., Rosen, C. L., Troester, M. M., & Wise, M. S. (2016). Recommended amount of sleep for pediatric populations: A consensus statement of the American Academy of Sleep Medicine. Journal of Clinical Sleep Medicine, 12(6), 785–786. [Google Scholar] [CrossRef] [PubMed]
- Perpétuo, C., Fernandes, M., & Veríssimo, M. (2020). Comparison between actigraphy records and parental reports of child’s sleep. Frontiers in Pediatrics, 8, 567390. [Google Scholar] [CrossRef] [PubMed]
- Petit, D., Touchette, É., Tremblay, R. E., Boivin, M., & Montplaisir, J. (2007). Dyssomnias and parasomnias in early childhood. Pediatrics, 119(5), e1016–e1025. [Google Scholar] [CrossRef]
- Philbrook, L. E., Becker, L. E., & Linde, J. (2022). Sleep disturbances moderate the association between effortful control and executive functioning in early childhood. Journal of Experimental Child Psychology, 220, 105421. [Google Scholar] [CrossRef] [PubMed]
- Piaget, J. (1936). Origins of intelligence in the child. Routledge & Kegan Paul. [Google Scholar]
- Ren, L., & Hu, B. Y. (2019). The relative importance of sleep duration and bedtime routines for the social-emotional functioning of Chinese children. Journal of Developmental & Behavioral Pediatrics, 40(8), 597–605. [Google Scholar] [CrossRef]
- Reynolds, A. M., Soke, G. N., Sabourin, K. R., Hepburn, S., Katz, T., Wiggins, L. D., Schieve, L. A., & Levy, S. E. (2019). Sleep problems in 2- to 5-year-olds with autism spectrum disorder and other developmental delays. Pediatrics, 143(3), e20180492. [Google Scholar] [CrossRef]
- Sadeh, A. (2007). Consequences of sleep loss or sleep disruption in children. Sleep Medicine Clinics, 2(3), 513–520. [Google Scholar] [CrossRef]
- Schumacher, A. M., Miller, A. L., Watamura, S. E., Kurth, S., Lassonde, J. M., & LeBourgeois, M. K. (2017). Sleep moderates the association between response inhibition and self-regulation in early childhood. Journal of Clinical Child & Adolescent Psychology, 46(2), 222–235. [Google Scholar] [CrossRef]
- Seger, C. A., & Spiering, B. J. (2011). A critical review of habit learning and the basal ganglia. Frontiers in Systems Neuroscience, 5, 66. [Google Scholar] [CrossRef] [PubMed]
- Song, H., Wang, J.-J., Zhang, B., Shi, L., & Lau, P. W. C. (2023). Do acute and chronic physical activity interventions affect the cognitive function of preschool children? A meta-analysis. Psychology of Sport and Exercise, 67, 102419. [Google Scholar] [CrossRef] [PubMed]
- Stride, C. B., Gardner, S., Catley, N., & Thomas, F. (2015). Mplus code for mediation, moderation, and moderated mediation models. Available online: http://www.offbeat.group.shef.ac.uk/FIO/mplusmedmod.htm (accessed on 12 June 2023).
- Suggate, S., & Stoeger, H. (2017). Fine motor skills enhance lexical processing of embodied vocabulary: A test of the nimble-hands, nimble-minds hypothesis. Quarterly Journal of Experimental Psychology, 70(10), 2169–2187. [Google Scholar] [CrossRef]
- Sun, X., Wang, G., Chen, M., Zhao, J., Zhang, Y., Jiang, Y., Zhu, Q., Rong, T., & Jiang, F. (2022). The effects of improvements of sleep disturbances throughout kindergarten on executive function: A latent change score analysis. Cognitive Development, 62, 101174. [Google Scholar] [CrossRef]
- Touchette, É., Petit, D., Séguin, J. R., Boivin, M., Tremblay, R. E., & Montplaisir, J. Y. (2007). Associations between sleep duration patterns and behavioral/cognitive functioning at school entry. Sleep, 30, 1213–1219. [Google Scholar] [CrossRef]
- Veer, G. V. D., Kamphorst, E., Cantell, M., Minnaert, A., & Houwen, S. (2020). Task-specific and latent relationships between motor skills and executive functions in preschool children. Frontiers in Psychology, 11, 2208. [Google Scholar] [CrossRef]
- Veldman, S. L. C., Santos, R., Jones, R. A., Sousa-Sá, E., & Okely, A. D. (2019). Associations between gross motor skills and cognitive development in toddlers. Early Human Development, 132, 39–44. [Google Scholar] [CrossRef]
- Voltaire, S. T., & Teti, D. M. (2018). Early nighttime parental interventions and infant sleep regulation across the first year. Sleep Medicine, 52, 107–115. [Google Scholar] [CrossRef]
- Weibley, H., Filippo, M. D., Liu, X., Lazenby, L., Goscha, J., Ferreira, A., Muscalu, L., & Rader, N. (2021). fNIRS monitoring of infant prefrontal cortex during crawling and an executive functioning task. Frontiers in Behavioral Neuroscience, 15, 675366. [Google Scholar] [CrossRef] [PubMed]
- Williams, K. E., Hayes, N., Berthelsen, D., & Quach, J. (2023). A longitudinal model of sleep problems and classroom self-regulation across elementary school. Journal of Applied Developmental Psychology, 89, 101596. [Google Scholar] [CrossRef]
- Wilson, M. (2002). Six views of embodied cognition. Psychonomic Bulletin & Review, 9(4), 625–636. [Google Scholar] [CrossRef]
- Woods, A. D., Jiao, J. L., Morgan, P. L., & Buxton, O. M. (2023). Is sleep longitudinally related to children’s achievement, executive function and classroom behaviour? Infant and Child Development, 33, e2426. [Google Scholar] [CrossRef]
- Wu, M., Liang, X., Lu, S., & Wang, Z. (2017). Infant motor and cognitive abilities and subsequent executive function. Infant Behavior and Development, 49, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Zelazo, P. D. (2006). The Dimensional Change Card Sort (DCCS): A method of assessing executive function in children. Nature Protocols, 1(1), 297–301. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z., Okely, A. D., Pereira, J. R., Sousa-Sá, E., Veldman, S. L. C., & Santos, R. (2022). Associations of sleep characteristics with cognitive and gross motor development in toddlers. Sleep Health, 8(4), 350–355. [Google Scholar] [CrossRef]
- Zuccarini, M., Guarini, A., Savini, S., Iverson, J. M., Aureli, T., Alessandroni, R., Faldella, G., & Sansavini, A. (2017). Object exploration in extremely preterm infants between 6 and 9 months and relation to cognitive and language development at 24 months. Research in Developmental Disabilities, 68, 140–152. [Google Scholar] [CrossRef] [PubMed]


| Variables | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. T1: Gross motor (n = 210) | 1 | ||||||||||
| 2. T1: Fine motor (n = 215) | 0.31 ** | 1 | |||||||||
| 3. T2: Gross motor (n = 140) | 0.24 ** | 0.28 ** | 1 | ||||||||
| 4. T2: Fine motor (n = 140) | 0.14 | 0.29 ** | 0.30 ** | 1 | |||||||
| 5. T3: Cognitive ability (n = 168) | 0.24 ** | 0.10 | 0.10 | 0.16 | 1 | ||||||
| 6. T4: Working memory (n = 176) | 0.14 | 0.02 | −0.01 | −0.09 | 0.22 ** | 1 | |||||
| 7. T4: Inhibition (n = 151) | 0.17 | 0.11 | 0.10 | 0.13 | 0.12 | −0.10 | 1 | ||||
| 8. T4: Cognitive flexibility (n = 178) | 0.20 * | 0.12 | −0.02 | −0.02 | 0.34 ** | 0.19 * | 0.15 | 1 | |||
| 9. T4: Sleep duration (n = 173) | 0.00 | 0.10 | −0.11 | −0.10 | 0.01 | 0.00 | 0.00 | 0.01 | 1 | ||
| 10. T4: Sleep disturbances (n = 173) | 0.04 | −0.02 | 0.04 | 0.12 | −0.09 | −0.01 | 0.06 | −0.04 | −0.05 | 1 | |
| 11. T1: family SES (n = 214) | 0.10 | 0.09 | 0.00 | −0.13 | 0.05 | 0.19 * | 0.08 | 0.07 | 0.10 | −0.21 ** | 1 |
| M | 9.65 | 11.78 | 11.30 | 12.86 | 12.00 | 4.44 | 6.44 | 1.21 | 9.76 | 47.00 | 0.01 |
| SD | 2.43 | 2.69 | 2.78 | 1.97 | 3.04 | 2.52 | 5.77 | 0.55 | 0.85 | 5.72 | 0.99 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Liu, C.; Zhang, Y.; Liang, X. Infant Motor Competence Predicts Executive Functions in Preschoolers: The Role of Sleep. Behav. Sci. 2026, 16, 288. https://doi.org/10.3390/bs16020288
Liu C, Zhang Y, Liang X. Infant Motor Competence Predicts Executive Functions in Preschoolers: The Role of Sleep. Behavioral Sciences. 2026; 16(2):288. https://doi.org/10.3390/bs16020288
Chicago/Turabian StyleLiu, Chao, Yuzhu Zhang, and Xi Liang. 2026. "Infant Motor Competence Predicts Executive Functions in Preschoolers: The Role of Sleep" Behavioral Sciences 16, no. 2: 288. https://doi.org/10.3390/bs16020288
APA StyleLiu, C., Zhang, Y., & Liang, X. (2026). Infant Motor Competence Predicts Executive Functions in Preschoolers: The Role of Sleep. Behavioral Sciences, 16(2), 288. https://doi.org/10.3390/bs16020288




