The Relationship Between Emotion Processing Assessed by an Affect Rating Task and Depression Symptoms Following the Accelerated Sequential Dorsolateral–Dorsomedial Prefrontal rTMS Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Randomization and Blinding of Treatment
2.3. Transcranial Magnetic Stimulation
2.4. Assessment
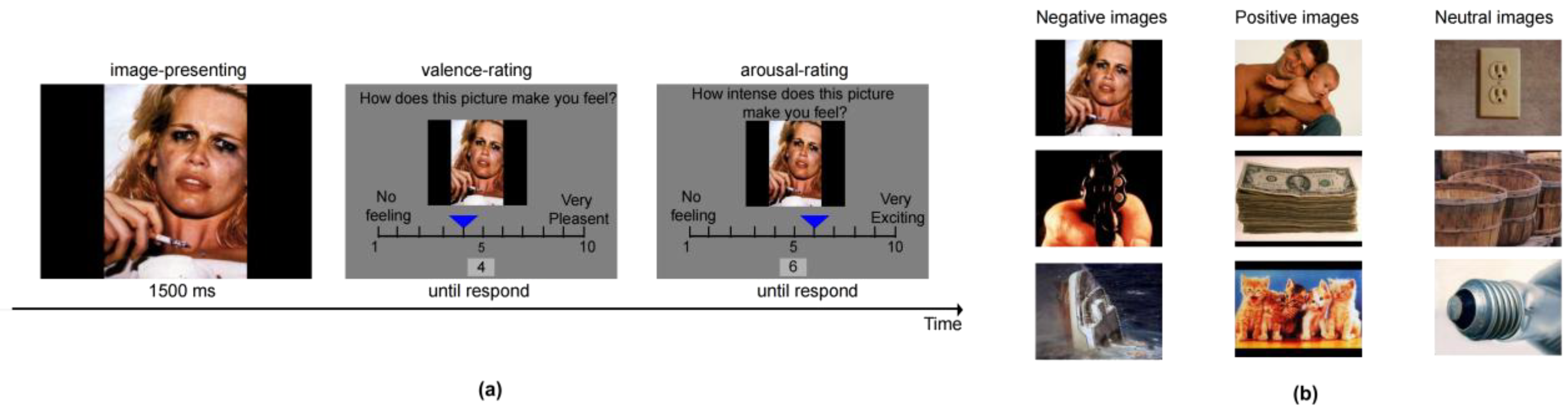
2.5. Affect Rating Task (ART)
2.6. Statistical Analysis
3. Results
3.1. Demographic and Clinical Data
3.2. Affect Rating Task
3.2.1. Analysis of Arousal and Valence Ratings
3.2.2. Correlations Between Baseline Ratings and Clinical Scales
3.2.3. Predictive Effects of Emotional Arousal Change on Symptom Improvement
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| rTMS | Repetitive Transcranial Magnetic Stimulation |
| dlPFC | Dorsolateral Prefrontal Cortex |
| dmPFC | Dorsomedial Prefrontal Cortex |
| ART | Affect Rating Task |
| MDD | Major Depressive Disorder |
| ECN | Executive Control Network |
| DMN | Default Mode Network |
| MADRS | Montgomery-Åsberg Depression Rating Scale |
| SHAPS | Hamilton Pleasure Scale |
| FSS | Fatigue Severity Scale |
Appendix A
References
- André-Obadia, N., Magnin, M., & Garcia-Larrea, L. (2021). Theta-burst versus 20 Hz repetitive transcranial magnetic stimulation in neuropathic pain: A head-to-head comparison. Clinical Neurophysiology, 132(10), 2702–2710. [Google Scholar] [CrossRef] [PubMed]
- Bermpohl, F., Fregni, F., Boggio, P. S., Thut, G., Northoff, G., Otachi, P. T. M., Rigonatti, S. P., Marcolin, M. A., & Pascual-Leone, A. (2006). Effect of low-frequency transcranial magnetic stimulation on an affective go/no-go task in patients with major depression: Role of stimulation site and depression severity. Psychiatry Research, 141(1), 1–13. [Google Scholar] [CrossRef]
- Bora, E., Fornito, A., Pantelis, C., & Yucel, M. (2012). Gray matter abnormalities in major depressive disorder: A meta-analysis of voxel based morphometry studies. Journal of Affective Disorders, 138(1–2), 9–18. [Google Scholar] [CrossRef]
- Bourke, C., Douglas, K., & Porter, R. (2010). Processing of facial emotion expression in major depression: A review. Australian and New Zealand Journal of Psychiatry, 44(8), 681–696. [Google Scholar] [CrossRef]
- Cash, R. F. H., Dar, A., Hui, J., De Ruiter, L., Baarbé, J., Fettes, P., Peters, S., Fitzgerald, P. B., Downar, J., & Chen, R. (2017). Influence of inter-train interval on the plastic effects of rTMS. Brain Stimulation, 10(3), 630–636. [Google Scholar] [CrossRef]
- Cole, E. J., Phillips, A. L., Bentzley, B. S., Stimpson, K. H., Nejad, R., Barmak, F., Veerapal, C., Khan, N., Cherian, K., Felber, E., Brown, R., Choi, E., King, S., Pankow, H., Bishop, J. H., Azeez, A., Coetzee, J., Rapier, R., Odenwald, N., … Williams, N. R. (2022). Stanford Neuromodulation Therapy (SNT): A double-blind randomized controlled trial. American Journal of Psychiatry, 179(2), 132–141. [Google Scholar] [CrossRef]
- Csukly, G., Czobor, P., Szily, E., Takács, B., & Simon, L. (2009). Facial expression recognition in depressed subjects: The impact of intensity level and arousal dimension. The Journal of Nervous and Mental Disease, 197(2), 98–103. [Google Scholar] [CrossRef]
- Cutler, A. J., Mattingly, G. W., & Maletic, V. (2023). Understanding the mechanism of action and clinical effects of neuroactive steroids and GABAergic compounds in major depressive disorder. Translational Psychiatry, 13(1), 228. [Google Scholar] [CrossRef] [PubMed]
- Dalili, M. N., Penton-Voak, I. S., Harmer, C. J., & Munafò, M. R. (2014). Meta-analysis of emotion recognition deficits in major depressive disorder. Psychological Medicine, 45(6), 1135–1144. [Google Scholar] [CrossRef] [PubMed]
- Downar, J., & Daskalakis, Z. J. (2013). New targets for rTMS in depression: A review of convergent evidence. Brain Stimulation, 6(3), 231–240. [Google Scholar] [CrossRef]
- Downar, J., Geraci, J., Salomons, T. V., Dunlop, K., Wheeler, S., McAndrews, M. P., Bakker, N., Blumberger, D. M., Daskalakis, Z. J., Kennedy, S. H., Flint, A. J., & Giacobbe, P. (2014). Anhedonia and reward-circuit connectivity distinguish nonresponders from responders to dorsomedial prefrontal repetitive transcranial magnetic stimulation in major depression. Biological Psychiatry, 76(3), 176–185. [Google Scholar] [CrossRef]
- Dritschel, B., & Agren, T. (2023). Physiological and subjective arousal to prospective mental imagery: A mechanism for behavioral change? PLoS ONE, 18(12), e0294629. [Google Scholar] [CrossRef]
- Drysdale, A. T., Grosenick, L., Downar, J., Dunlop, K., Mansouri, F., Meng, Y., Fetcho, R. N., Zebley, B., Oathes, D. J., Etkin, A., Schatzberg, A. F., Sudheimer, K., Keller, J., Mayberg, H. S., Gunning, F. M., Alexopoulos, G. S., Fox, M. D., Pascual-Leone, A., Voss, H. U., … Liston, C. (2017). Resting-state connectivity biomarkers define neurophysiological subtypes of depression. Nature Medicine, 23(1), 28–38. [Google Scholar] [CrossRef] [PubMed Central]
- Eldaief, M. C., Halko, M. A., Buckner, R. L., & Pascual-Leone, A. (2011). Transcranial magnetic stimulation modulates the brain’s intrinsic activity in a frequency-dependent manner. Proceedings of the National Academy of Sciences of the United States of America, 108(52), 21229–21234. [Google Scholar] [CrossRef] [PubMed]
- Geschwind, N., Nicolson, N. A., Peeters, F., van Os, J., Barge-Schaapveld, D., & Wichers, M. (2011). Early improvement in positive rather than negative emotion predicts remission from depression after pharmacotherapy. European Neuropsychopharmacology, 21(3), 241–247. [Google Scholar] [CrossRef] [PubMed]
- Gollan, J. K., Pane, H. T., McCloskey, M. S., & Coccaro, E. F. (2008). Identifying differences in biased affective information processing in major depression. Psychiatry Research, 159(1–2), 18–24. [Google Scholar] [CrossRef] [PubMed]
- Gotlib, I. H., & Joormann, J. (2010). Cognition and depression: Current status and future directions. Annual Review of Clinical Psychology, 6(1), 285–312. [Google Scholar] [CrossRef]
- Greicius, M. D., Flores, B. H., Menon, V., Glover, G. H., Solvason, H. B., Kenna, H., Reiss, A. L., & Schatzberg, A. F. (2007). Resting-state functional connectivity in major depression: Abnormally increased contributions from subgenual cingulate cortex and thalamus. Biological Psychiatry, 62(5), 429–437. [Google Scholar] [CrossRef] [PubMed Central]
- Grzelak, L. N., Glazer, J. E., Klemballa, D. M., Nabb, C. B., Mittal, V. A., Walther, S., Shankman, S. A., & Letkiewicz, A. M. (2025). Depressive symptoms are associated with impaired body affect recognition for neutral expressions. Journal of Affective Disorders, 387, 119531. [Google Scholar] [CrossRef]
- Hewlett, S., Dures, E., & Almeida, C. (2011). Measures of fatigue: Bristol Rheumatoid Arthritis Fatigue Multi-Dimensional Questionnaire (BRAF MDQ), Bristol Rheumatoid Arthritis Fatigue Numerical Rating Scales (BRAF NRS) for severity, effect, and coping, Chalder Fatigue Questionnaire (CFQ), Checklist Individual Strength (CIS20R and CIS8R), Fatigue Severity Scale (FSS), Functional Assessment Chronic Illness Therapy (Fatigue) (FACIT-F), Multi-Dimensional Assessment of Fatigue (MAF), Multi-Dimensional Fatigue Inventory (MFI), Pediatric Quality Of Life (PedsQL) Multi-Dimensional Fatigue Scale, Profile of Fatigue (ProF), Short Form 36 Vitality Subscale (SF-36 VT), and Visual Analog Scales (VAS). Arthritis Care & Research, 63, S263–S286. [Google Scholar] [CrossRef]
- Höflich, A., Michenthaler, P., Kasper, S., & Lanzenberger, R. (2019). Circuit mechanisms of reward, anhedonia, and depression. International Journal of Neuropsychopharmacology, 22(2), 105–118. [Google Scholar] [CrossRef] [PubMed Central]
- Krause, F. C., Linardatos, E., Fresco, D. M., & Moore, M. T. (2021). Facial emotion recognition in major depressive disorder: A meta-analytic review. Journal of Affective Disorders, 293, 320–328. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lambert, C., Da Silva, S., Ceniti, A. K., Rizvi, S. J., Foussias, G., & Kennedy, S. H. (2018). Anhedonia in depression and schizophrenia: A transdiagnostic challenge. CNS Neuroscience & Therapeutics, 24(7), 615–623. [Google Scholar] [CrossRef] [PubMed]
- Lang, P. J., Bradley, M. M., & Cuthbert, B. N. (2008). International affective picture system (IAPS): Affective ratings of pictures and instruction manual (Rep. No. A-8). University of Florida.
- Lantrip, C., Gunning, F. M., Flashman, L., Roth, R. M., & Holtzheimer, P. E. (2017). Effects of transcranial magnetic stimulation on the cognitive control of emotion. The Journal of ECT, 33(2), 73–80. [Google Scholar] [CrossRef] [PubMed]
- Lazarov, A., Ben-Zion, Z., Shamai, D., Pine, D. S., & Bar-Haim, Y. (2018). Free viewing of sad and happy faces in depression: A potential target for attention bias modification. Journal of Affective Disorders, 238, 94–100. [Google Scholar] [CrossRef]
- Leppänen, J. M., Milders, M., Bell, J. S., Terriere, E., & Hietanen, J. K. (2004). Depression biases the recognition of emotionally neutral faces. Psychiatry Research, 128(2), 123–133. [Google Scholar] [CrossRef]
- Leyman, L., De Raedt, R., Vanderhasselt, M.-A., & Baeken, C. (2011). Effects of repetitive transcranial magnetic stimulation of the dorsolateral prefrontal cortex on the attentional processing of emotional information in major depression: A pilot study. Psychiatry Research, 185(1–2), 102–107. [Google Scholar] [CrossRef]
- Li, X., Man, I. S. C., Shao, R., Zhao, X., Wu, S., Liu, Z., & Lin, K. (2025). The effect of non-invasive brain stimulation in reducing anhedonia and apathy in major depressive disorder and schizophrenia: A meta-analysis. Journal of Affective Disorders, 388, 119561. [Google Scholar] [CrossRef]
- Ma, J., Yang, H., Li, R., Yuan, X., Pan, C., Li, F., Yang, D., Bie, Z., Zheng, J., & Li, Y. (2025). Effects of SNRIs on emotion regulation and electrophysiological profiles in depressive patients: A correlational study with symptom remission. Psychiatry Research, 351, 116576. [Google Scholar] [CrossRef]
- Maniglio, R., Gusciglio, F., Lofrese, V., Belvederi Murri, M., Tamburello, A., & Innamorati, M. (2014). Biased processing of neutral facial expressions is associated with depressive symptoms and suicide ideation in individuals at risk for major depression due to affective temperaments. Comprehensive Psychiatry, 55(3), 518–525. [Google Scholar] [CrossRef]
- Milders, M., Bell, S., Platt, J., Serrano, R., & Runcie, O. (2010). Stable expression recognition abnormalities in unipolar depression. Psychiatry Research, 179(1), 38–42. [Google Scholar] [CrossRef]
- Millan, M. J., Agid, Y., Brune, M., Bullmore, E. T., Carter, C. S., Clayton, N. S., Connor, R., Davis, S., Deakin, B., DeRubeis, R. J., Dubois, B., Geyer, M. A., Goodwin, G. M., Gorwood, P., Jay, T. M., Joels, M., Mansuy, I. M., Meyer-Lindenberg, A., Murphy, D., … Young, L. J. (2012). Cognitive dysfunction in psychiatric disorders: Characteristics, causes and the quest for improved therapy. Nature Reviews Drug Discovery, 11(2), 141–168. [Google Scholar] [CrossRef]
- Miron, J.-P., Feffer, K., Cash, R. F. H., Derakhshan, D., Kim, J. M. S., Fettes, P., Giacobbe, P., Blumberger, D. M., Daskalakis, Z. J., & Downar, J. (2019). Safety, tolerability and effectiveness of a novel 20 Hz rTMS protocol targeting dorsomedial prefrontal cortex in major depression: An open-label case series. Brain Stimulation, 12(5), 1319–1321. [Google Scholar] [CrossRef]
- Nejati, V., Khorrami, A. S., & Fonoudi, M. (2022). Neuromodulation of facial emotion recognition in health and disease: A systematic review. Neurophysiologie Clinique, 52(3), 183–201. [Google Scholar] [CrossRef]
- Ochsner, K. N., & Gross, J. J. (2005). The cognitive control of emotion. Trends in Cognitive Sciences, 9(5), 242–249. [Google Scholar] [CrossRef]
- Park, C.-h., Lee, H.-K., Kweon, Y.-S., Lee, C. T., Kim, K.-T., Kim, Y.-J., & Lee, K.-U. (2015). Emotion-induced topological changes in functional brain networks. Brain Topography, 29(1), 108–117. [Google Scholar] [CrossRef] [PubMed]
- Rizeq, J. (2024). Affective forecasting and psychopathology: A scoping review. Clinical Psychology Review, 108, 102392. [Google Scholar] [CrossRef]
- Roiser, J. P., Elliott, R., & Sahakian, B. J. (2011). Cognitive mechanisms of treatment in depression. Neuropsychopharmacology, 37(1), 117–136. [Google Scholar] [CrossRef] [PubMed]
- Sheline, Y. I., Price, J. L., Yan, Z., & Mintun, M. A. (2010). Resting-state functional MRI in depression unmasks increased connectivity between networks via the dorsal nexus. Proceedings of the National Academy of Sciences of the United States of America, 107(24), 11020–11025. [Google Scholar] [CrossRef] [PubMed Central]
- Sterzer, P., Hilgenfeldt, T., Freudenberg, P., Bermpohl, F., & Adli, M. (2011). Access of emotional information to visual awareness in patients with major depressive disorder. Psychological Medicine, 41(8), 1615–1624. [Google Scholar] [CrossRef]
- Sun, S., Yu, H., Yu, R., & Wang, S. (2023). Functional connectivity between the amygdala and prefrontal cortex underlies processing of emotion ambiguity. Translational Psychiatry, 13(1), 334. [Google Scholar] [CrossRef]
- Trettin, M., Dvořák, J., Hilke, M., Wenzler, S., Hagen, M., Ghirmai, N., Stäblein, M., Matura, S., Huthmacher, A. C., Kraft, D., Balaban, C., Ciaramidaro, A., Prvulovic, D., Knöchel, C., Reif, A., & Oertel, V. (2022). Neuronal response to high negative affective stimuli in major depressive disorder: An fMRI study. Journal of Affective Disorders, 298, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Trøstheim, M., Eikemo, M., Meir, R., Hansen, I., Paul, E., Kroll, S. L., Garland, E. L., & Leknes, S. (2020). Assessment of anhedonia in adults with and without mental illness: A systematic review and meta-analysis. JAMA Network Open, 3(8), e2013233. [Google Scholar] [CrossRef] [PubMed Central]
- Vinograd, M., Stout, D. M., & Risbrough, V. B. (2022). Anhedonia in posttraumatic stress disorder: Prevalence, phenotypes, and neural circuitry. Current Topics in Behavioral Neurosciences, 58, 185–199. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y., Qin, C., Chen, H., Liang, W., Liu, M., & Liu, J. (2025). Global, regional, and national burden of major depressive disorders in adults aged 60 years and older from 1990 to 2021, with projections of prevalence to 2050: Analyses from the Global Burden of Disease Study 2021. Journal of Affective Disorders, 374, 486–494. [Google Scholar] [CrossRef]
- Wen, A., Fischer, E. R., Watson, D., & Yoon, K. L. (2023). Biased cognitive control of emotional information in remitted depression: A meta-analytic review. Journal of Psychopathology and Clinical Science, 132(8), 921–936. [Google Scholar] [CrossRef]
- Whitton, A. E., & Pizzagalli, D. A. (2022). Anhedonia in depression and bipolar disorder. Current Topics in Behavioral Neurosciences, 58, 111–127. [Google Scholar] [CrossRef]
- Wong, S., Le, G. H., Phan, L., Rhee, T. G., Ho, R., Meshkat, S., Teopiz, K. M., Kwan, A. T. H., Mansur, R. B., Rosenblat, J. D., & McIntyre, R. S. (2024). Effects of anhedonia on health-related quality of life and functional outcomes in major depressive disorder: A systematic review and meta-analysis. Journal of Affective Disorders, 356, 684–698. [Google Scholar] [CrossRef]
- Wu, C., Mu, Q., Gao, W., & Lu, S. (2025). The characteristics of anhedonia in depression: A review from a clinically oriented perspective. Translational Psychiatry, 15(1), 90. [Google Scholar] [CrossRef]
- Zhao, Y.-J., Xiang, S., Chen, R., Ding, Q., Geng, R., Wang, Y., Li, Y., Li, H., Wang, Y., Cui, H., Huang, Y., Feng, J., Liu, W., & Voon, V. (2025). A sequential dual-site repetitive transcranial magnetic stimulation for major depressive disorder: A randomized clinical trial. Cell Reports Medicine, 6(10), 102402. [Google Scholar] [CrossRef]



| Active (n = 26) | Sham (n = 25) | Statistics | ||
|---|---|---|---|---|
| t/χ2 | p | |||
| Sex (female/male) | 12/14 | 18/7 | 3.515 | 0.061 |
| Age (years, mean ± SD) | 31.46 ± 6.94 | 32.24 ± 11.11 | −0.301 | 0.764 |
| Education (year, mean ± SD) | 14.83 ± 3.56 | 14.88 ± 3.75 | −0.520 | 0.959 |
| Duration (month, mean ± SD) | 47.81 ± 40.63 | 67.64 ± 70.01 | −1.243 | 0.220 |
| Episodes (mean ± SD) | 1.50 ± 0.71 | 1.36 ± 0.49 | 0.819 | 0.417 |
| Baseline MADRS (mean ± SD) | 27.42 ± 3.82 | 26.24 ± 4.31 | 1.038 | 0.304 |
| Medication (on/off) | 26/0 | 24/1 | 1.061 | 0.303 |
| SSRIs (on/off) | 16/10 | 19/6 | 1.238 | 0.266 |
| SNRIs (on/off) | 8/18 | 8/17 | 0.009 | 0.925 |
| Augmentation (on/off) | 5/21 | 3/22 | 0.504 | 0.478 |
| Benzodiazepines (on/off) | 2/24 | 0/25 | 2.002 | 0.157 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Chen, R.; Dong, Z.; Geng, R.; Li, H.; Wang, Y.; Li, Y.; Ding, Q.; Zhang, Y.; Ding, X.; Huang, J.; et al. The Relationship Between Emotion Processing Assessed by an Affect Rating Task and Depression Symptoms Following the Accelerated Sequential Dorsolateral–Dorsomedial Prefrontal rTMS Treatment. Behav. Sci. 2026, 16, 178. https://doi.org/10.3390/bs16020178
Chen R, Dong Z, Geng R, Li H, Wang Y, Li Y, Ding Q, Zhang Y, Ding X, Huang J, et al. The Relationship Between Emotion Processing Assessed by an Affect Rating Task and Depression Symptoms Following the Accelerated Sequential Dorsolateral–Dorsomedial Prefrontal rTMS Treatment. Behavioral Sciences. 2026; 16(2):178. https://doi.org/10.3390/bs16020178
Chicago/Turabian StyleChen, Ruiqin, Zerun Dong, Ruijie Geng, Haibin Li, Yuan Wang, Yuanyuan Li, Qiong Ding, Yingying Zhang, Xuechen Ding, Jingjing Huang, and et al. 2026. "The Relationship Between Emotion Processing Assessed by an Affect Rating Task and Depression Symptoms Following the Accelerated Sequential Dorsolateral–Dorsomedial Prefrontal rTMS Treatment" Behavioral Sciences 16, no. 2: 178. https://doi.org/10.3390/bs16020178
APA StyleChen, R., Dong, Z., Geng, R., Li, H., Wang, Y., Li, Y., Ding, Q., Zhang, Y., Ding, X., Huang, J., Zhao, H., Liu, W., Voon, V., & Zhao, Y.-J. (2026). The Relationship Between Emotion Processing Assessed by an Affect Rating Task and Depression Symptoms Following the Accelerated Sequential Dorsolateral–Dorsomedial Prefrontal rTMS Treatment. Behavioral Sciences, 16(2), 178. https://doi.org/10.3390/bs16020178






