Effects of Developmental Timing on Cognitive and Behavioral Profiles in Fetal Alcohol Spectrum Disorder: Considerations for Education
Abstract
1. Introduction
- It is observed that the contribution of families through the BRIEF-2 and SENA assessments allows for the development of a cognitive and behavioral functioning pattern for children and adolescents with FASD.
- It is contemplated that age is a factor that modulates significant differences in the neurocognitive and behavioral profiles of individuals affected by FASD.
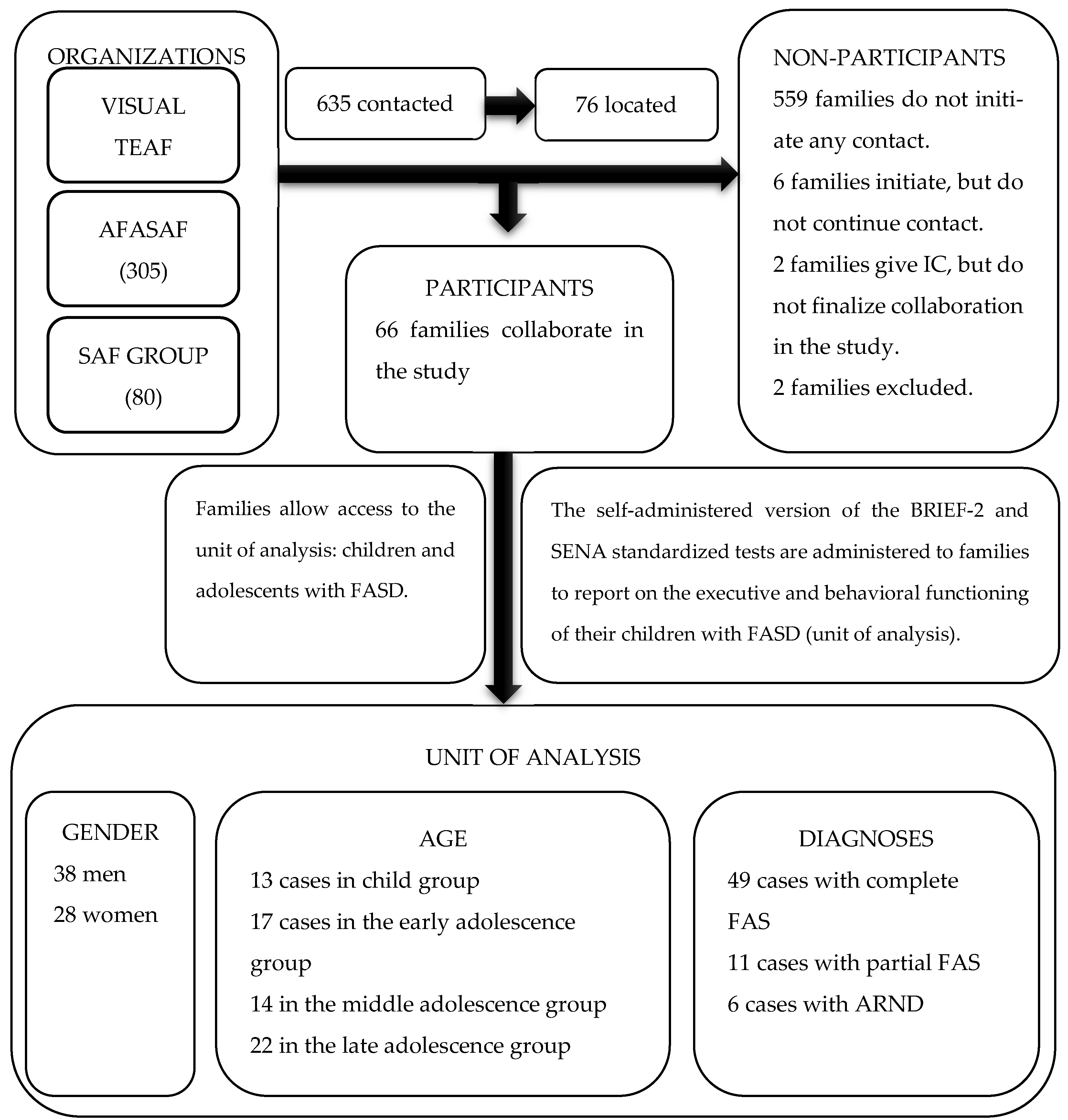
2. Materials and Methods
2.1. Study Design
2.2. Participants
2.3. Variables
2.4. Measurement
3. Statistical Methods and Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| PROFILE OF EXECUTIVE FUNCTIONING BY AGE | |
| CHILDHOOD (7–10 years) Inhibition: PCE Self-monitoring: ME Flexibility: PCE Emotional control: ME Initiative: CSE Working memory: CSE Planning and organization: PCE Task monitoring: PCE Material organization: EL. | EARLY ADOLESCENCE (11–14 years) Inhibition: PCE Self-monitoring: ME Flexibility: PCE Emotional control: ME Initiative: CSE Working memory: CSE Planning and organization: PCE Task monitoring: PCE Material organization: ME |
| MIDDLE ADOLESCENCE (15–17 years) Inhibition: PCE Self-monitoring: ME Flexibility: PCE Emotional control: ME Initiative: CSE Working memory: CSE Planning and organization: PCE Task monitoring: PCE Material organization: ME | LATE ADOLESCENCE (18–19 years) Inhibition: PCE Self-monitoring: ME Flexibility: EPC. Emotional control: ME Initiative: CSE Working memory: CSE Planning and organization: PCE Task monitoring: PCE Material organization: ME |
| PROFILE OF EXECUTIVE FUNCTIONING BY GENDER | |
| WOMEN Inhibition: CSE Self-monitoring: CSE Flexibility: CSE Emotional control: PCE Initiative: CSE Working memory: CSE Planning and organization: CSE Task monitoring: CSE Material organization CSE | MEN Inhibition: CSE Self-monitoring: CSE Flexibility: CSE Emotional control: PCE Initiative: CSE Working memory: CSE Planning and organization: CSE Task monitoring: CSE Material organization: CSE |
| PROFILE OF EXECUTIVE FUNCTIONING BY DIAGNOSIS | |
| Complete FAS Inhibition: CSE Self-monitoring: CSE Flexibility: CSE Emotional control: CSE Initiative: CSE Working memory: CSE Planning and organization: PCE Task monitoring: CSE Material organization: CSE | Partial FAS Inhibition: CSE Self-monitoring: CSE Flexibility: CSE Emotional control: PCE Initiative: CSE Working memory: CSE Planning and organization: CSE Task monitoring: CSE Material organization: CSE |
| ARND Inhibition: CSE Self-monitoring: CSE Flexibility: CSE Emotional control: CSE Initiative: CSE Working memory: CSE Planning and organization: CSE Task monitoring: PCE Material organization: ME | |
Appendix B
| PROFILE OF BEHAVIORAL FUNCTIONING BY AGE | |
| CHILDHOOD (7–10 years) Depression: 58.7 Anxiety: 56.9 Social anxiety: 53.9 Somatic complaints: 47 Attention problems: 67.7 Hyperactivity-impulsivity: 67.2 Anger control problems: 66.9 Aggression: 65.7 Challenging behavior: 66.5 Antisocial behavior: 0 Substance use: 0 Eating behavior problems: 0 Unusual behavior: 70.9 Emotional regulation problems: 59.5 Rigidity: 61.5 Isolation: 67.6 Social integration and competence: 29 Emotional intelligence: 38.1 Study disposition: 32.2 | EARLY ADOLESCENCE (11–14 years) Depression: 63.5 Anxiety: 65.6 Social anxiety: 62.4 Somatic complaints: 57.1 Attention problems: 72.7 Hyperactivity-impulsivity: 66.7 Anger control problems: 68.4 Aggression: 63.8 Challenging behavior: 63.8 Antisocial behavior: 50.7 Substance use: 36.1 Eating behavior problems: 41.9 Unusual behavior: 80.1 Emotional regulation problems: 66.7 Rigidity: 69.4 Isolation: 78.7 Social integration and competence: 30.4 Emotional intelligence: 34.4 Study disposition: 28.1 |
| MIDDLE ADOLESCENCE (15–17 years) Depression: 66.3 Anxiety: 75.8 Social anxiety: 66.5 Somatic complaints: 60.7 Attention problems: 78.6 Hyperactivity-impulsivity: 76.8 Anger control problems: 71.7 Aggression: 76.3 Challenging behavior: 69.1 Antisocial behavior: 86.5 Substance use: 51.1 Eating behavior problems: 59.9 Unusual behavior: 93.2 Emotional regulation problems: 72.9 Rigidity: 74.6 Isolation: 85.4 Social integration and competence: 24.3 Emotional intelligence: 35 Study disposition: 28.3 | LATE ADOLESCENCE (18–19 years) Depression: 63.4 Anxiety: 66.5 Social anxiety: 56.1 Somatic complaints: 49.9 Attention problems: 74.6 Hyperactivity-impulsivity: 73.6 Anger control problems: 75.6 Aggression: 80 Challenging behavior: 76.8 Antisocial behavior: 99.5 Substance use: 79 Eating behavior problems: 59.2 Unusual behavior: 96 Emotional regulation problems: 73.3 Rigidity: 73.2 Isolation: 82.3 Social integration and competence: 27.2 Emotional intelligence: 32.8 Study disposition: 28.1 |
| PROFILE OF BEHAVIORAL FUNCTIONING BY GENDER | |
| WOMEN Depression: 60.5 Anxiety: 65.5 Social anxiety: 63.4 Somatic complaints: 50.1 Attention problems: 75.9 Hyperactivity-impulsivity: 68.9 Anger control problems: 69.9 Aggression: 73 Challenging behavior: 71.3 Antisocial behavior: 68.6 Substance use: 46.2 Eating behavior problems: 41.4 Unusual behavior: 82.8 Emotional regulation problems: 68.3 Rigidity: 68.4 Isolation: 85.5 Social integration and competence: 25.5 Emotional intelligence: 33.9 Study disposition: 26.3 | MEN Depression: 65.1 Anxiety: 66.9 Social anxiety: 56.6 Somatic complaints: 56 Attention problems: 71.9 Hyperactivity-impulsivity: 73 Anger control problems: 72.2 Aggression: 71.6 Challenging behavior: 68.7 Antisocial behavior: 61.6 Substance use: 46.7 Eating behavior problems: 44.6 Unusual behavior: 89 Emotional regulation problems: 69.2 Rigidity: 71.5 Isolation: 74.5 Social integration and competence: 29.4 Emotional intelligence: 35.3 Study disposition: 31 |
| PROFILE OF BEHAVIORAL FUNCTIONING BY DIAGNOSIS | |
| Complete FAS Depression: 62.6 Anxiety: 67.6 Social anxiety: 59.1 Somatic complaints: 54.2 Attention problems: 75 Hyperactivity-impulsivity: 73.3 Anger control problems: 69.8 Aggression: 71.5 Challenging behavior: 69.9 Antisocial behavior: 67.1 Substance use: 44.4 Eating behavior problems: 44.5 Unusual behavior: 90.3 Emotional regulation problems: 68.6 Rigidity: 71 Isolation: 81.9 Social integration and competence: 27.2 Emotional intelligence: 35.4 Study disposition: 28 | Partial FAS Depression: 67.3 Anxiety: 61.7 Social anxiety: 60.8 Somatic complaints: 52.6 Attention problems: 70.8 Hyperactivity-impulsivity: 65 Anger control problems: 69.7 Aggression: 67.3 Challenging behavior: 66.8 Antisocial behavior: 57.5 Substance use: 52.9 Eating behavior problems: 37.5 Unusual behavior: 76.6 Emotional regulation problems: 66 Rigidity: 66.7 Isolation: 74.2 Social integration and competence: 30.4 Emotional intelligence: 31.8 Study disposition: 29.7 |
| ARND Depression: 60.3 Anxiety: 64.7 Social anxiety: 60.3 Somatic complaints: 48.7 Attention problems: 67.7 Hyperactivity-impulsivity: 65.7 Anger control problems: 85.3 Aggression: 86.7 Challenging behavior: 74.3 Antisocial behavior: 57.3 Substance use: 51.7 Eating behavior problems: 43.7 Unusual behavior: 72.3 Emotional regulation problems: 76.3 Rigidity: 69.7 Isolation: 66 Social integration and competence: 27 Emotional intelligence: 34.7 Study disposition: 36 | |
References
- Hoyme, E.H.; Kalberg, W.; Elliott, A.; Blankenship, J.; Buckley, D.; Marais, A.S.; Nursing, C.; Manning, M.; Robinson, L.; Adam, M.; et al. Updated clinical guidelines for diagnosing fetal alcohol spectrum disorders. Pediatrics 2016, 138, e20154256. [Google Scholar] [CrossRef] [PubMed]
- Maya-Enero, S.; Ramis-Fernández, S.; Astals-Vizcaino, M.; García-Algar, O. Perfil neurocognitivo y conductual del trastorno del espectro alcohólico fetal. An. Pediatría 2021, 95, e1–e208. [Google Scholar] [CrossRef]
- Popova, S.; Dozet, D.; Burd, L. Fetal Alcohol Spectrum Disorder: Can We Change the Future? Alcohol Clin. Exp. Res. 2020, 44, 815–819. [Google Scholar] [CrossRef] [PubMed]
- Popova, S.; Lange, S.; Probst, C.; Gmel, C.; Rehm, J. Estimation of national, regional, and global prevalence of alcohol use during pregnancy and fetal alcohol syndrome: A systematic review and meta-analysis. Lancet Glob. 2017, 5, e290–e299. [Google Scholar] [CrossRef]
- Bombardelli, O. Inclusive Education and Its Implementation: International Practices. Educ. Self Dev. 2020, 15, 37–46. [Google Scholar] [CrossRef]
- Coates, J.; Harris, J.; Waring, M. The effectiveness of a special school experience for improving preservice teachers’ efficacy to teach children with special educational needs and disabilities. Br. Educ. Res. J. 2020, 46, 909–928. [Google Scholar] [CrossRef]
- Emmers, E.; Baeyens, D.; Petry, K. Attitudes and self-efficacy of teachers towards inclusion in higher education. Eur. J. Spec. Needs Educ. 2019, 35, 139–153. [Google Scholar] [CrossRef]
- Lees, B.; Riches, J.; Mewton, L.; Elliot, E.; Allsop, S.; Newton, N.; Thomas, S.; Rice, L.; Nepal, S.; Teeson, M.; et al. Fetal alcohol spectrum disorder resources for educators: A scoping review. Health Promot. J. Austr. 2022, 33, 797–809. [Google Scholar] [CrossRef] [PubMed]
- Taresh, S.; Aniza, N.; Roslan, A.S.; Ma’rof, A.M.; Zaid, S. Pre-School Teachers’ Knowledge, Belief, Identification Skills, and Self-Efficacy in Identifying Autism Spectrum Disorder (ASD): A Conceptual Framework to Identify Children with ASD. Brain Sci. 2020, 10, 165. [Google Scholar] [CrossRef] [PubMed]
- Taylor, T.; Enns, L.N. Factors predictive of a fetal alcohol spectrum disorder diagnosis: Parent and teacher ratings. Child Neuropsychol. 2018, 25, 507–527. [Google Scholar] [CrossRef] [PubMed]
- Reid, N.; White, C.; Hawkins, E.; Crawford, A.; Liu, W.; Shanley, D. Outcomes and needs of health and education professionals following fetal alcohol spectrum disorder-specific training. J. Paediatr. Child Health 2019, 56, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Zameer, M.; Carlisle, A.C.S.; Livesey, A.C.; Mukherjee, R.A.S. Comparisons of the BRIEF parental report and neuropsychological clinical tests of executive function in Fetal Alcohol Spectrum Disorders: Data from the UK national specialist clinic. Child Neuropsychol. 2020, 25, 648–663. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Pinto, I.; Santamaría, P.; Sánchez-Sánchez, F.; Carrasco, M.; del Barrio, V. SENA. Sistema de Evaluación de Niños y Adolescentes. Manual de Aplicación, Corrección e Interpretación; TEA Ediciones: Madrid, Spain, 2015. [Google Scholar]
- Gioia, G.; Isquith, P.; Guy, S.; Kenworthy, K. BRIEF-2. Evaluación Conductual de la Función Ejecutiva; Maldonado, M.J., Fournier, M.C., Martínez-Arias, R., González-Marqués, J., Santamaría, J.M.E.-S.Y.P., Eds.; TEA Ediciones: Madrid, Spain, 2017. [Google Scholar]
- Baddeley, A. Working memory: Theories, models, and controversies. Annu. Rev. Psychol. 2012, 63, 1–29. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Herman, L.E.; Acosta, M.; Chang, P.N. Gender and attention deficits in children diagnosed with a fetal alcohol spectrum disorder. Can. J. Clin. Pharmacol. 2008, 15, e411–e419. Available online: https://jptcp.com/index.php/jptcp/article/view/234 (accessed on 21 September 2023). [PubMed]
- Rai, J.; Abecassis, M.; Casey, J.; Flaro, L.; Erdodi, L.; Roth, R. Parent rating of executive function in fetal alcohol spectrum disorder: A review of the literature and new data on Aboriginal Canadian children. Child Neuropsychol. 2017, 23, 713–732. [Google Scholar] [CrossRef] [PubMed]
- Mattson, S.; Bernes, G.; Doyle, L. Fetal Alcohol Spectrum Disorders: A review of the neurobehavioral deficits associated with prenatal alcohol exposure. Alcohol Clin. Exp. Res. 2019, 43, 1046–1062. [Google Scholar] [CrossRef] [PubMed]
- Rangmar, J.; Sandberg, A.D.; Aronson, M.; Fahlke, C. Cognitive and executive functions, social cognition, and sense of coherence in adults with fetal alcohol syndrome. Nord. J. Psychiatry 2015, 69, 1754–1760. [Google Scholar] [CrossRef]
- Sakano, M.; Mukherjee, R.A.S.; Turk, J. Behaviour and adaptive functioning in children and young people with fetal alcohol spectrum disorders: A UK study. Adv. Dual Diagn. 2019, 12, 62–72. [Google Scholar] [CrossRef]
- Rasmussen, C.; Andrew, G.; Zwaigenbaum, L.; Tough, S. Neurobehavioral outcomes of children with Fetal Alcohol Spectrum Disorders: A Canadian perspective. Paediatr. Child Health 2008, 13, 185–191. [Google Scholar] [CrossRef] [PubMed]
- McLachlan, K.; Paolozza, A.; Kully-Martens, K.; Portales-Casamar, E.; Pavlidis, P.; Andrew, G.; Hanlon-Dearman, A.; Loock, C.; McFarlane, A.; Nikkel, S.; et al. Unpacking the heterogeneity of cognitive functioning in children and adolescents with fetal alcohol spectrum disorder: Determining the role of moderators and strengths. Adv. Neurodev. Disord. 2017, 1, 271–282. [Google Scholar] [CrossRef]
- Rasmussen, C.; Wyper, K.; Talwar, V. The relation between theory of mind and executive functions in children with fetal alcohol spectrum disorders. J. Popl. Ther. Clin. Pharmacol. 2018, 16, e370–e380. Available online: https://jptcp.com/index.php/jptcp/article/view/561 (accessed on 30 January 2024).
- Rasmussen, C.; Tamana, S.; Baugh, L.; Andrew, G.; Tough, S.; Zwaigenbaum, L. Neuropsychological impairments on the NEPSYII among children with FASD. Child Neuropsichol. 2013, 19, 337–349. [Google Scholar] [CrossRef]
- Khoury, J.E.; Milligan, K. Comparing Executive Functioning in Children and Adolescents with Fetal Alcohol Spectrum Disorders and ADHD: A Meta-Analysis. J. Atten. Disord. 2019, 23, 1801–1815. [Google Scholar] [CrossRef] [PubMed]
- Brown, N.; Connor, P.; Adler, R. Conduct-disordered adolescents with fetal alcohol spectrum disorder. Intervention in secure treatment settings. Crim. Justice Behav. 2012, 39, 770–793. [Google Scholar] [CrossRef]
- Nash, K.; Stevens, S.; Clairman, H.; Rovet, J. Preliminary Findings that a Targeted Intervention Leads to Altered Brain Function in Children with Fetal Alcohol Spectrum Disorder. Brain Sci. 2018, 8, 7. [Google Scholar] [CrossRef] [PubMed]
- Xiang, X.; Cho, J.; Sun, Y.; Wang, X. Childhood adversity and cognitive impairment in later life. Front. Psychol. 2022, 13, 935254. [Google Scholar] [CrossRef]
- Kingdon, D.; Cardoso, C.; McGrath, J.J. Research Review: Executive function deficits in fetal alcohol spectrum disorders and attention-deficit/hyperactivity disorder—A meta-analysis. J. Child Psychol. Psychiatry Allied Discip. 2016, 57, 116–131. [Google Scholar] [CrossRef] [PubMed]
- Chudley, A.E.; Conry, J.; Cook, J.L.; Loock, C.; Rosales, T.; Le Blanc, N. Fetal alcohol spectrum disorder: Canadian guidelines for diagnosis. Can. Med. Assoc. J. 2005, 1, s1–s21. [Google Scholar] [CrossRef] [PubMed]
- Kully-Martens, K.; Denys, K.; Treit, S.; Tamana, S.; Rasmussen, C. A Review of Social Skills Deficits in Individuals with Fetal Alcohol Spectrum Disorders and Prenatal Alcohol Exposure: Profiles, Mechanisms, and Interventions. Alcohol Clin. Exp. Res. 2012, 36, 568–576. [Google Scholar] [CrossRef]
- Johnson, M.E.; Robinson, R.V.; Corey, S.; Dewane, S.L.; Brems, C.; Casto, L.D. Knowledge, attitudes, and behaviors of health, education, and service professionals as related to fetal alcohol spectrum disorders. Int. J. Public Health 2010, 55, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Westland, J.C. Information loss and bias in Likert survey responses. PLoS ONE 2022, 17, e0271949. [Google Scholar] [CrossRef]
| Variable EF | FE Statistics | Variable BF | BF Statistics | ||
|---|---|---|---|---|---|
| D | p | D | p | ||
| INH | 0.076 | 0.435 | DEP | 0.810 | 0.331 |
| SMO | 0.156 | 0.000 | ANS | 0.089 | 0.212 |
| FLE | 0.118 | 0.021 | SCA | 0.103 | 0.075 |
| EMC | 0.121 | 0.014 | SOM | 0.161 | 0.000 |
| INI | 0.094 | 0.150 | ATE | 0.079 | 0.376 |
| WM | 0.073 | 0.503 | HIP | 0.102 | 0.079 |
| PLA | 0.181 | 0.000 | ANG | 0.092 | 0.170 |
| TAS | 0.116 | 0.026 | AGG | 0.123 | 0.013 |
| ORG | 0.094 | 0.151 | CHA | 0.113 | 0.032 |
| ANT | 0.191 | 0.000 | |||
| SUB | 0.362 | 0.000 | |||
| EAT | 0.166 | 0.001 | |||
| UNU | 0.103 | 0.077 | |||
| REG | 0.124 | 0.011 | |||
| RIG | 0.110 | 0.043 | |||
| ISO | 0.106 | 0.062 | |||
| SOC | 0.133 | 0.005 | |||
| EMI | 0.091 | 0.191 | |||
| STU | 0.135 | 0.004 | |||
| Variable EF | t-Test Results EF | Variable BF | t-Test Results BF | ||
|---|---|---|---|---|---|
| T | p | T | p | ||
| INH ** | 15.22 | 0.000 | DEP ** | 8.38 | 0.000 |
| SMO * | 17.71 | 0.000 | ANS ** | 11.37 | 0.000 |
| FLE * | 17.52 | 0.000 | SCA ** | 5.79 | 0.000 |
| EMC * | 11.86 | 0.000 | SOM * | 2.02 | 0.047 |
| INI ** | 23.33 | 0.000 | ATE ** | 20.22 | 0.000 |
| WM ** | 22.38 | 0.000 | HIP ** | 13.04 | 0.000 |
| PLA * | 26.81 | 0.000 | ANG ** | 10.7 | 0.000 |
| TAS * | 20.13 | 0.000 | AGG * | 8.45 | 0.000 |
| ORG ** | 12.87 | 0.000 | CHA * | 10.6 | 0.000 |
| ANT * | 9.32 | 0.000 | |||
| SUB * | 3.46 | 0.001 | |||
| EAT ** | 3.72 | 0.001 | |||
| UNU ** | 11.83 | 0.000 | |||
| REG * | 14.05 | 0.000 | |||
| RIG * | 16.77 | 0.000 | |||
| ISO ** | 13.66 | 0.000 | |||
| SOC * | −15.37 | 0.000 | |||
| EMI ** | −13.79 | 0.000 | |||
| STU * | −19.36 | 0.000 | |||
| Variable EF | Pearson | Spearman’s Rho | pEF | Variable BF | Pearson | Spearman’s Rho | pBF |
|---|---|---|---|---|---|---|---|
| INH | 0.456 ** | 0.000 ** | DEP | 0.105 | 0.401 | ||
| SMO | 0.529 ** | 0.000 ** | ANS | 0.274 * | 0.026 * | ||
| FLE | 0.487 ** | 0.000 ** | SCA | 0.019 | 0.877 | ||
| EMC | 0.473 ** | 0.000 ** | SOM | −0.015 | 0.907 | ||
| INI | 0.153 | 0.219 | ATE | 0.290 * | 0.018 * | ||
| WM | 0.275 * | 0.026 * | HIP | 0.251 * | 0.042 * | ||
| PLA | 0.335 ** | 0.006 ** | ANG | 0.263 * | 0.033 * | ||
| TAS | 0.427 ** | 0.000 ** | AGG | 0.379 ** | 0.002 ** | ||
| ORG | 0.163 | 0.183 | CHA | 0.352 ** | 0.004 ** | ||
| ANT | 0.566 ** | 0.000 ** | |||||
| SUB | 0.258 | 0.070 | |||||
| EAT | 0.100 | 0.491 | |||||
| UNU | 0.340 ** | 0.005 ** | |||||
| REG | 0.434 ** | 0.000 ** | |||||
| RIG | 410 ** | 0.001 ** | |||||
| ISO | 0.263 * | 0.033 * | |||||
| SOC | 0.004 | 0.976 | |||||
| EMI | −243 * | 0.049 * | |||||
| STU | −0.152 | 0.222 |
| Variable EF | Pearson | Spearman’s Rho | pEF | Variable BF | Pearson | Spearman’s Rho | pBF |
|---|---|---|---|---|---|---|---|
| INH | −0.034 | 0.786 | DEP | 0.021 | 0.434 | ||
| SMO | 0.030 | 0.813 | ANS | −0.144 | 0.124 | ||
| FLE | −0.085 | 0.497 | SCA | 0.045 | 0.359 | ||
| EMC | 0.044 | 0.726 | SOM | −0.043 | 0.366 | ||
| INI | −0.122 | 0.329 | ATE | −0.257 * | 0.019 | ||
| WM | −0.090 | 0.471 | HIP | −0.262 * | 0.017 | ||
| PLA | 0.054 | 0.668 | ANG | 0.228 | 0.033 | ||
| TAS | 0.001 | 0.996 | AGG | 0.097 | 0.220 | ||
| ORG | −0.240 | 0.052 | CHA | −0.010 | 0.468 | ||
| ANT | −0.075 | 0.275 | |||||
| SUB | 0.110 | 0.190 | |||||
| EAT | −0.062 | 0.312 | |||||
| UNU | −0.264 * | 0.016 | |||||
| REG | 0.108 | 0.195 | |||||
| RIG | −0.188 | 0.066 | |||||
| ISO | 0.025 | 0.422 | |||||
| SOC | −0.293 ** | 0.008 | |||||
| EMI | −0.085 | 0.249 | |||||
| STU | 0.215 * | 0.041 |
| ANOVA EF | ANOVA BF | ||||
|---|---|---|---|---|---|
| EF | F | p | F | p | BF |
| INI | 5.703 | 0.002 | ANS | 7.845 | 0.000 |
| SMO | 9.274 | 0.000 | ATE | 3.511 | 0.020 |
| FLE | 7.518 | 0.000 | HIP | 2.278 | 0.088 |
| EMC | 3.907 | 0.013 | ANG | 1.039 | 0.381 |
| WM | 4.430 | 0.007 | AGG | 2.610 | 0.059 |
| PLA | 3.534 | 0.020 | CHA | 2.911 | 0.041 |
| TAS | 6.075 | 0.001 | ANT | 5.695 | 0.002 |
| UNU | 3.955 | 0.012 | |||
| REG | 6.693 | 0.001 | |||
| RIG | 6.434 | 0.001 | |||
| SOC | 3.045 | 0.035 | |||
| EMI | 0.954 | 0.420 | |||
| ANOVA BF | ||
|---|---|---|
| F | p | BF |
| ATE | 2.232 | 0.116 |
| HIP | 2.450 | 0.094 |
| UNU | 2.509 | 0.089 |
| SOC | .322 | 0.726 |
| STU | 2.365 | 0.102 |
| Variables EF | Average Range | Z | p | ||
|---|---|---|---|---|---|
| FAS | pFAS | ARND | |||
| INH | 33.97 | 34.41 | 28 | 0.548 | 0.760 |
| SMO | 33.28 | 32.23 | 37.67 | 0.341 | 0.843 |
| FLE | 34.43 | 31.36 | 29.83 | 0.474 | 0.789 |
| EMC | 33.18 | 31.27 | 40.17 | 0.889 | 0.641 |
| INI | 35.14 | 25.73 | 34.33 | 2.186 | 0.335 |
| WM | 34.07 | 35.86 | 24.50 | 1.536 | 0.464 |
| PLA | 33.20 | 29 | 44.17 | 2.504 | 0.286 |
| TAS | 33 | 43.45 | 19.33 | 6.310 | 0.043 |
| ORG | 36.01 | 29.86 | 19.67 | 4.373 | 0.112 |
| Variables BF | Average Range | Z | p | ||
|---|---|---|---|---|---|
| FAS | pFAS | ARND | |||
| DEP | 32.99 | 37.77 | 29.83 | 0.801 | 0.670 |
| ANS | 35.41 | 27.73 | 28.50 | 1.894 | 0.388 |
| SCA | 32.92 | 35.27 | 35.00 | 0.176 | 0.916 |
| SOM | 33.97 | 32.41 | 31.67 | 0.120 | 0.942 |
| ATE | 36.30 | 29.41 | 18.17 | 5.388 | 0.068 |
| HIP | 36.51 | 24.00 | 26.33 | 4.750 | 0.093 |
| ANG | 31.70 | 31.50 | 51.83 | 6.031 | 0.049 |
| AGG | 32.71 | 30.45 | 45.50 | 2.713 | 0.258 |
| CHA | 33.78 | 29.91 | 37.83 | 0.702 | 0.704 |
| ANT | 34.30 | 31.95 | 29.83 | 0.384 | 0.825 |
| SUB | 32.32 | 36.14 | 38.33 | 0.795 | 0.672 |
| EAT | 34.39 | 27.55 | 37.17 | 1.411 | 0.494 |
| UNU | 36.45 | 26.36 | 22.50 | 4.674 | 0.097 |
| REG | 32.65 | 29.55 | 47.67 | 3.845 | 0.146 |
| RIG | 35.71 | 25.45 | 30.17 | 2.792 | 0.248 |
| ISO | 35.81 | 31.50 | 18.33 | 4.581 | 0.101 |
| SOC | 33.20 | 34.64 | 33.83 | 0.052 | 0.974 |
| EMI | 34.71 | 28.82 | 32.17 | 0.884 | 0.643 |
| STU | 31.32 | 35.41 | 47.83 | 4.105 | 0.128 |
| Variables EF | Z | p | Variables BF | Z | p |
|---|---|---|---|---|---|
| INH | 1.155 | 0.139 | DEP | 1.079 | 0.194 |
| SMO | 0.936 | 0.345 | ANS | 0.906 | 0.385 |
| FLE | 0.936 | 0.345 | SCA | 1.260 | 0.083 |
| EMC | 0.762 | 0.607 | SOM | 1.268 | 0.080 |
| INI | 0.694 | 0.721 | ATE | 1.743 | 0.005 |
| WM | 1.977 | 0.001 | HIP | 1.004 | 0.266 |
| PLA | 0.770 | 0.594 | ANG | 0.687 | 0.733 |
| TAS | 1.592 | 0.013 | AGG | 0.611 | 0.849 |
| ORG | 0.868 | 0.438 | CHA | 1.162 | 0.134 |
| ANT | 0.574 | 0.897 | |||
| SUB | 1.253 | 0.087 | |||
| EAT | 1.004 | 0.266 | |||
| UNU | 0.732 | 0.657 | |||
| REG | 0.634 | 0.816 | |||
| RIG | 1.230 | 0.097 | |||
| ISO | 1.608 | 0.011 | |||
| SOC | 0.845 | 0.473 | |||
| EMI | 0.981 | 0.291 | |||
| STU | 1.208 | 0.108 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Felgueras, N.; López-Díaz, J.M.; Garrote, I. Effects of Developmental Timing on Cognitive and Behavioral Profiles in Fetal Alcohol Spectrum Disorder: Considerations for Education. Behav. Sci. 2024, 14, 431. https://doi.org/10.3390/bs14060431
Felgueras N, López-Díaz JM, Garrote I. Effects of Developmental Timing on Cognitive and Behavioral Profiles in Fetal Alcohol Spectrum Disorder: Considerations for Education. Behavioral Sciences. 2024; 14(6):431. https://doi.org/10.3390/bs14060431
Chicago/Turabian StyleFelgueras, Nerea, José María López-Díaz, and Inmaculada Garrote. 2024. "Effects of Developmental Timing on Cognitive and Behavioral Profiles in Fetal Alcohol Spectrum Disorder: Considerations for Education" Behavioral Sciences 14, no. 6: 431. https://doi.org/10.3390/bs14060431
APA StyleFelgueras, N., López-Díaz, J. M., & Garrote, I. (2024). Effects of Developmental Timing on Cognitive and Behavioral Profiles in Fetal Alcohol Spectrum Disorder: Considerations for Education. Behavioral Sciences, 14(6), 431. https://doi.org/10.3390/bs14060431





