10 Minutes Frontal 40 Hz tACS—Effects on Working Memory Tested by Luck-Vogel Task
Abstract
1. Introduction
1.1. Brain Oscillations and Working Memory
1.2. Transcranial Electrical Stimulation
1.3. Transcranial Alternating Current Stimulation and Working Memory
1.3.1. Theta-tACS
1.3.2. Gamma-tACS
1.3.3. Cross Frequency Coupling-tACS
2. Material and Methods
2.1. Participants
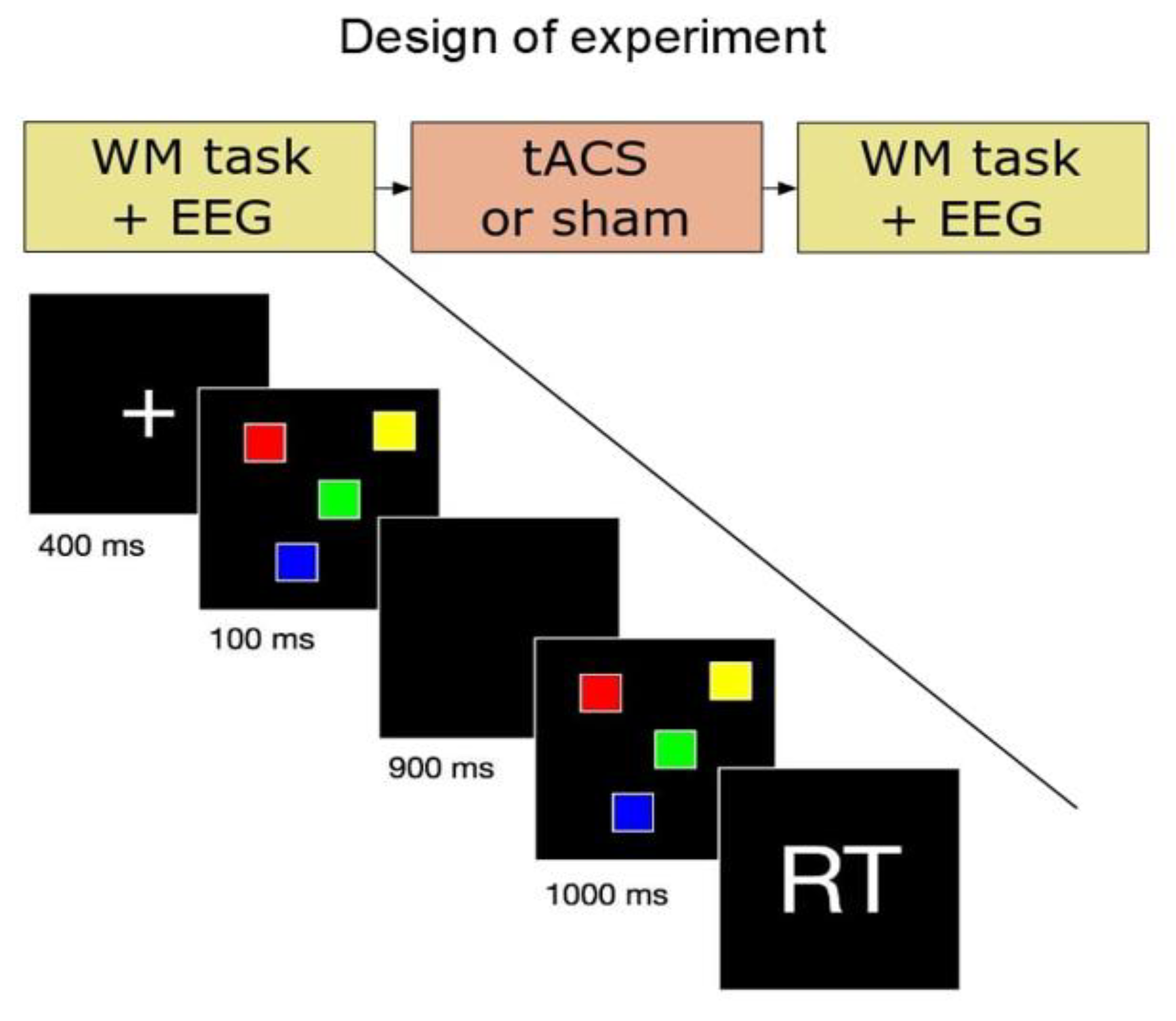
2.2. Experimental Procedure
2.3. Behavioral Task
2.4. Electrophysiology
2.5. Data Processing and Statistics
3. Results
3.1. Behavior
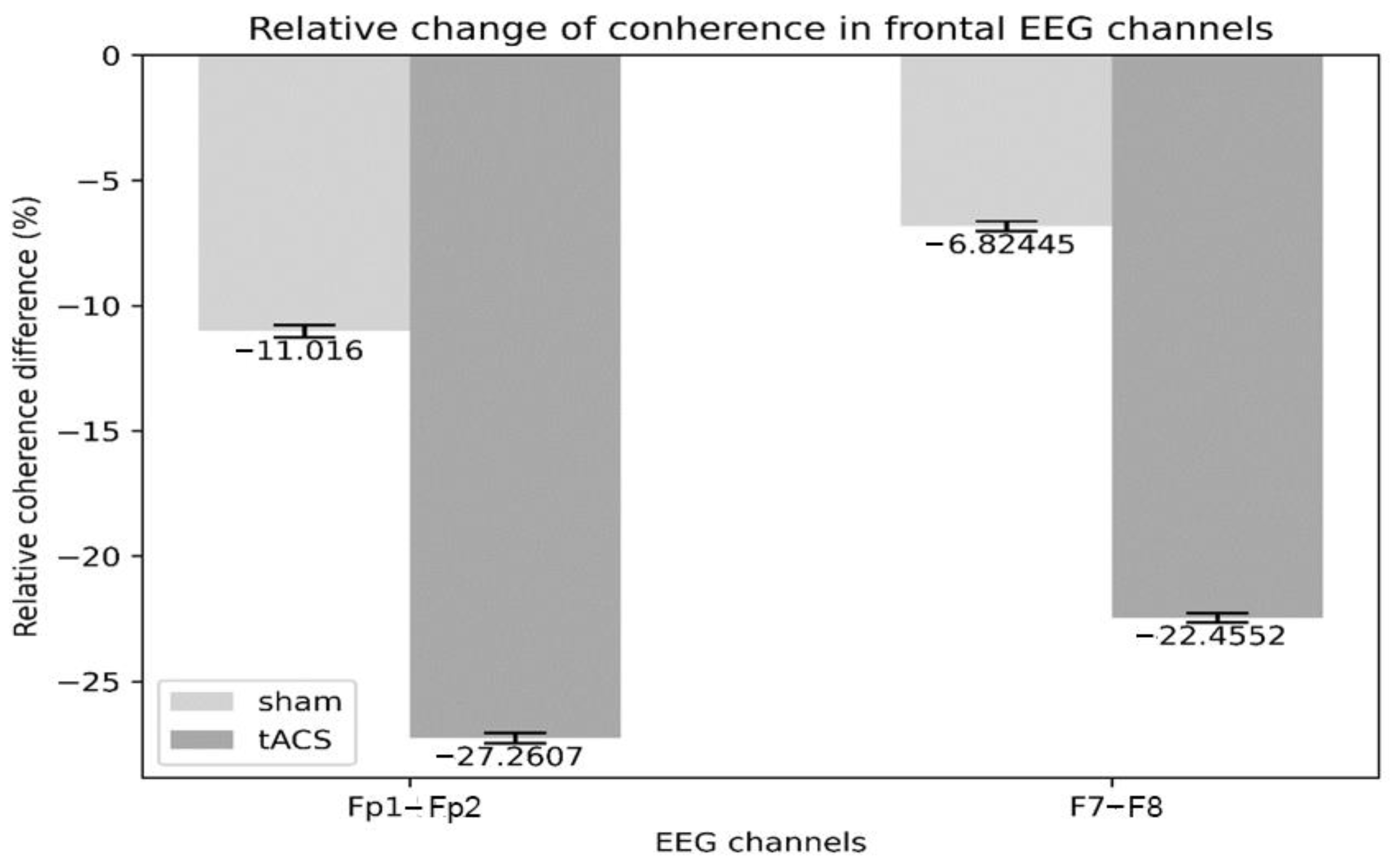
3.2. Electrophysiology
4. Discussion
4.1. Behavior
4.2. Electrophysiology
5. Conclusions, Limitations and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Başar, E. Brain oscillations in neuropsychiatric disease. Dialogues Clin. Neurosci. 2013, 15, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Düzel, E.; Penny, W.D.; Burgess, N. Brain oscillations and memory. Curr. Opin. Neurobiol. 2010, 20, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Merker, B. Cortical gamma oscillations: The functional key is activation, not cognition. Neurosci. Biobehav. Rev. 2013, 37, 401–417. [Google Scholar] [CrossRef]
- Symons, A.E.; El-Deredy, W.; Schwartze, M.; Kotz, S.A. The functional role of neural oscillations in non-verbal emotional communication. Front. Hum. Neurosci. 2016, 10, 239. [Google Scholar] [CrossRef] [PubMed]
- Marzetti, L.; Basti, A.; Chella, F.; D’Andrea, A.; Syrjälä, J.; Pizzella, V. Brain Functional Connectivity Through Phase Coupling of Neuronal Oscillations: A Perspective From Magnetoencephalography. Front. Neurosci. 2019, 13, 964. [Google Scholar] [CrossRef]
- Andersen, L.M.; Jerbi, K.; Dalal, S.S. Can EEG and MEG detect signals from the human cerebellum? Neuroimage 2020, 215, 116817. [Google Scholar] [CrossRef] [PubMed]
- Canolty, R.T.; Edwards, E.; Dalal, S.S.; Soltani, M.; Nagarajan, S.S.; Kirsch, H.E.; Berger, M.S.; Barbaro, N.M.; Knight, R.T. High gamma power is phase-locked to theta oscillations in human neocortex. Science 2006, 313, 1626–1628. [Google Scholar] [CrossRef]
- Lakatos, P.; Shah, A.S.; Knuth, K.H.; Ulbert, I.; Karmos, G.; Schroeder, C.E. An Oscillatory Hierarchy Controlling Neuronal Excitability and Stimulus Processing in the Auditory Cortex. J. Neurophysiol. 2005, 94, 1904–1911. [Google Scholar] [CrossRef]
- Canolty, R.T.; Knight, R.T. The functional role of cross-frequency coupling. Trends Cogn. Sci. 2010, 14, 506–515. [Google Scholar] [CrossRef]
- Siems, M.; Siegel, M. Dissociated neuronal phase- and amplitude-coupling patterns in the human brain. Neuroimage 2020, 209, 116538. [Google Scholar] [CrossRef]
- Jirsa, V.; Müller, V. Cross-frequency coupling in real and virtual brain networks. Front. Comput. Neurosci. 2013, 7, 78. [Google Scholar] [CrossRef] [PubMed]
- Jensen, O.; Colgin, L.L. Cross-frequency coupling between neuronal oscillations. Trends Cogn. Sci. 2007, 11, 267–269. [Google Scholar] [CrossRef] [PubMed]
- Lynn, P.A.; Sponheim, S.R. Disturbed theta and gamma coupling as a potential mechanism for visuospatial working memory dysfunction in people with schizophrenia. Neuropsychiatr. Electrophysiol. 2016, 2, 1–30. [Google Scholar] [CrossRef]
- Wang, J.; Fang, Y.; Wang, X.; Yang, H.; Yu, X.; Wang, H. Enhanced gamma activity and cross-frequency interaction of resting-state electroencephalographic oscillations in patients with Alzheimer’s disease. Front. Aging Neurosci. 2017, 9, 243. [Google Scholar] [CrossRef]
- Kiesel, A.; Steinhauser, M.; Wendt, M.; Falkenstein, M.; Jost, K.; Philipp, A.M.; Koch, I. Control and interference in task switching-a review. Psychol. Bull. 2010, 136, 849–874. [Google Scholar] [CrossRef]
- Pettigrew, C.; Martin, R.C. Cognitive declines in healthy aging: Evidence from multiple aspects of interference resolution. Psychol. Aging 2014, 29, 187–204. [Google Scholar] [CrossRef]
- Jeffries, S.; Everatt, J. Working memory: Its role in dyslexia and other specific learning difficulties. Dyslexia 2004, 10, 196–214. [Google Scholar] [CrossRef]
- Logie, R.H. The Functional Organization and Capacity Limits of Working Memory. Curr. Dir. Psychol. Sci. 2011, 20, 240–245. [Google Scholar] [CrossRef]
- Todd, J.J.; Marois, R. Capacity limit of visual short-term memory in human posterior parietal cortex. Nature 2004, 428, 751–754. [Google Scholar] [CrossRef]
- Curtis, C.E.; D’Esposito, M. Persistent activity in the prefrontal cortex during working memory. Trends Cogn. Sci. 2003, 7, 415–423. [Google Scholar] [CrossRef]
- Smith, E.E.; Jonides, J. Storage and Executive Processes in the Frontal Lobes. Science 1999, 283, 1657–1661. [Google Scholar] [CrossRef] [PubMed]
- Cowan, N. The magical number 4 in short-term memory: A reconsideration of mental storage capacity. Behav. Brain Sci. 2001, 24, 87–114. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, M.; Kitajo, K.; Yamaguchi, Y. Dynamic links between theta executive functions and alpha storage buffers in auditory and visual working memory. Eur. J. Neurosci. 2010, 31, 1683–1689. [Google Scholar] [CrossRef] [PubMed]
- Mizuhara, H.; Yamaguchi, Y. Human cortical circuits for central executive function emerge by theta phase synchronization. Neuroimage 2007, 36, 232–244. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Chen, X.; Li, Z.; Han, S.; Zhang, D. Binding of verbal and spatial information in human working memory involves large-scale neural synchronization at theta frequency. Neuroimage 2007, 35, 1654–1662. [Google Scholar] [CrossRef] [PubMed]
- Lisman, J.E.; Idiart, M.A.P. Storage of 7 ± 2 short-term memories in oscillatory subcycles. Science 1995, 267, 1512–1515. [Google Scholar] [CrossRef] [PubMed]
- Jensen, O.; Lisman, J.E. Novel lists of 7 ± 2 known items can be reliably stored in an oscillatory short-term memory network: Interaction with long-term memory. Learn Mem. 1996, 3, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Roux, F.; Uhlhaas, P.J. Working memory and neural oscillations: Alpha-gamma versus theta-gamma codes for distinct WM information? Trends Cogn. Sci. 2014, 18, 16–25. [Google Scholar] [CrossRef]
- Herrmann, C.S.; Rach, S.; Neuling, T.; Strüber, D. Transcranial alternating current stimulation: A review of the underlying mechanisms and modulation of cognitive processes. Front. Hum. Neurosci. 2013, 7, 279. [Google Scholar] [CrossRef]
- Van Vugt, M.K.; Chakravarthi, R.; Lachaux, J.-P. For whom the bell tolls: Periodic reactivation of sensory cortex in the gamma band as a substrate of visual working memory maintenance. Front. Hum. Neurosci. 2014, 8, 696. [Google Scholar] [CrossRef][Green Version]
- Smith, E.H.; Banks, G.P.; Mikell, C.; Cash, S.S.; Patel, S.R.; Eskandar, E.N.; Sheth, S.A. Frequency-Dependent Representation of Reinforcement-Related Information in the Human Medial and Lateral Prefrontal Cortex. J. Neurosci. 2015, 35, 15827. [Google Scholar] [CrossRef] [PubMed]
- Ruffini, G.; Wendling, F.; Merlet, I.; Molaee-Ardekani, B.; Mekonnen, A.; Salvador, R.; Soria-Frisch, A.; Grau, C.; Dunne, S.; Miranda, P.C. Transcranial current brain stimulation (tCS): Models and technologies. IEEE Trans. Neural. Syst. Rehabil. Eng. 2013, 21, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Terney, D.; Chaieb, L.; Moliadze, V.; Antal, A.; Paulus, W. Increasing Human Brain Excitability by Transcranial High-Frequency Random Noise Stimulation. J. Neurosci. 2008, 28, 14147. [Google Scholar] [CrossRef] [PubMed]
- Bikson, M.; Bulow, P.; Stiller, J.W.; Datta, A.; Battaglia, F.; Karnup, S.V.; Postolache, T.T. Transcranial Direct Current Transcranial Direct Current Stimulation for Major Depression: Stimulation for Major Depression: A General System for Quantifying A General System for Quantifying Transcranial Electrotherapy Dosage Transcranial Electrotherapy Dosage Opinion statement. Curr. Treat. Options Neurol. 2008, 10, 377–385. [Google Scholar]
- Auvichayapat, N.; Rotenberg, A.; Gersner, R.; Ngodklang, S.; Tiamkao, S.; Tassaneeyakul, W.; Auvichayapat, P. Transcranial direct current stimulation for treatment of refractory childhood focal epilepsy. Brain Stimul. 2013, 6, 696–700. [Google Scholar] [CrossRef]
- Frank, E.; Schecklmann, M.; Landgrebe, M.; Burger, J.; Kreuzer, P.; Poeppl, T.B.; Kleinjung, T.; Hajak, G.; Langguth, B. Treatment of chronic tinnitus with repeated sessions of prefrontal transcranial direct current stimulation: Outcomes from an open-label pilot study. J. Neurol. 2011, 259, 327–333. [Google Scholar] [CrossRef]
- Fregni, F.; Boggio, P.S.; Santos, M.C.; Lima, M.; Vieira, A.L.; Rigonatti, S.P.; Silva, M.T.A.; Barbosa, E.R.; Nitsche, M.A.; Pascual-Leone, A. Noninvasive cortical stimulation with transcranial direct current stimulation in Parkinson’s disease. Mov. Disord. 2006, 21, 1693–1702. [Google Scholar] [CrossRef]
- Fregni, F.; Gimenes, R.; Valle, A.C.; Ferreira, M.J.L.; Rocha, R.R.; Natalle, L.; Bravo, R.; Rigonatti, S.P.; Freedman, S.D.; Nitsche, M.A.; et al. A randomized, sham-controlled, proof of principle study of transcranial direct current stimulation for the treatment of pain in fibromyalgia. Arthritis Rheum. 2006, 54, 3988–3998. [Google Scholar] [CrossRef]
- Fregni, F.; Freedman, S.; Pascual-Leone, A. Recent advances in the treatment of chronic pain with non-invasive brain stimulation techniques. Lancet Neurol. 2007, 6, 188–191. [Google Scholar] [CrossRef]
- Schlaug, G.; Renga, V.; Nair, D. Transcranial Direct Current Stimulation in Stroke Recovery. Arch. Neurol. 2008, 65, 1571–1576. [Google Scholar] [CrossRef]
- Baker, J.M.; Rorden, C.; Fridriksson, J. Using transcranial direct current stimulation (tDCS) to treat stroke patients with aphasia. Stroke 2010, 41, 1229. [Google Scholar] [CrossRef]
- Jacobson, L.; Koslowsky, M.; Lavidor, M. tDCS polarity effects in motor and cognitive domains: A meta-analytical review. Exp. Brain Res. 2011, 216, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Nitsche, M.A.; Paulus, W. Transcranial direct current stimulation—Update 2011. Restor. Neurol. Neurosci. 2011, 29, 463–492. [Google Scholar] [CrossRef]
- Nitsche, M.A.; Paulus, W. Sustained excitability elevations induced by transcranial DC motor cortex stimulation in humans. Neurology 2001, 57, 1899–1901. [Google Scholar] [CrossRef] [PubMed]
- Nitsche, M.A.; Paulus, W. Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J. Physiol. 2000, 527, 633. [Google Scholar] [CrossRef]
- Nitsche, M.A.; Fricke, K.; Henschke, U.; Schlitterlau, A.; Liebetanz, D.; Lang, N.; Henning, S.; Tergau, F.; Paulus, W. Pharmacological Modulation of Cortical Excitability Shifts Induced by Transcranial Direct Current Stimulation in Humans. J. Physiol. 2003, 553, 293–301. [Google Scholar] [CrossRef]
- Bindman, L.J.; Lippold, O.C.J.; Redfearn, J.W.T. The action of brief polarizing currents on the cerebral cortex of the rat (1) during current flow and (2) in the production of long-lasting after-effects. J. Physiol. 1964, 172, 369–382. [Google Scholar] [CrossRef]
- Lee, S.B.; Youn, J.; Jang, W.; Yang, H.O. Neuroprotective effect of anodal transcranial direct current stimulation on 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced neurotoxicity in mice through modulating mitochondrial dynamics. Neurochem. Int. 2019, 129, 104491. [Google Scholar] [CrossRef]
- Lee, S.B.; Kim, H.T.; Yang, H.O.; Jang, W. Anodal transcranial direct current stimulation prevents methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced neurotoxicity by modulating autophagy in an in vivo mouse model of Parkinson’s disease. Sci. Rep. 2018, 8, 15165. [Google Scholar] [CrossRef]
- Scelzo, E.; Giannicola, G.; Rosa, M.; Ciocca, M.; Ardolino, G.; Cogiamanian, F.; Ferrucci, R.; Fumagalli, M.; Mameli, F.; Barbieri, S.; et al. Increased short latency afferent inhibition after anodal transcranial direct current stimulation. Neurosci. Lett. 2011, 498, 167–170. [Google Scholar] [CrossRef]
- Marshall, L.; Mölle, M.; Hallschmid, M.; Born, J. Transcranial Direct Current Stimulation during Sleep Improves Declarative Memory. J. Neurosci. 2004, 24, 9985. [Google Scholar] [CrossRef] [PubMed]
- Fregni, F.; Boggio, P.S.; Nitsche, M.; Bermpohl, F.; Antal, A.; Feredoes, E.; Marcolin, M.A.; Rigonatti, S.P.; Silva, M.T.; Paulus, W.; et al. Anodal transcranial direct current stimulation of prefrontal cortex enhances working memory. Exp. Brain. Res. 2005, 166, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Reis, J.; Fritsch, B. Modulation of motor performance and motor learning by transcranial direct current stimulation. Curr. Opin. Neurol. 2011, 24, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Pereira, J.B.; Junqué, C.; Bartrés-Faz, D.; Martí, M.J.; Sala-Llonch, R.; Compta, Y.; Falcón, C.; Vendrell, P.; Pascual-Leone, Á.; Valls-Solé, J.; et al. Modulation of verbal fluency networks by transcranial direct current stimulation (tDCS) in Parkinson’s disease. Brain Stimul. 2013, 6, 16–24. [Google Scholar] [CrossRef]
- Dockery, C.A.; Hueckel-Weng, R.; Birbaumer, N.; Plewnia, C. Enhancement of planning ability by transcranial direct current stimulation. J. Neurosci. 2009, 29, 7271–7277. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; A Liu, A.; Lafon, B.; Friedman, D.; Dayan, M.; Wang, X.; Bikson, M.; Doyle, W.K.; Devinsky, O.; Parra, L.C. Measurements and models of electric fields in the in vivo human brain during transcranial electric stimulation. Elife 2017, 6, e18834. [Google Scholar] [CrossRef]
- Kuo, M.F.; Nitsche, M.A. Effects of Transcranial Electrical Stimulation on Cognition. Clin. EEG Neurosci. 2012, 43, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Thut, G.; Miniussi, C. New insights into rhythmic brain activity from TMS-EEG studies. Trends Cogn. Sci. 2009, 13, 182–189. [Google Scholar] [CrossRef]
- Kirov, R.; Weiss, C.; Siebner, H.R.; Born, J.; Marshall, L. Slow oscillation electrical brain stimulation during waking promotes EEG theta activity and memory encoding. Proc. Natl. Acad. Sci. USA 2009, 106, 15460–15465. [Google Scholar] [CrossRef]
- Zaehle, T.; Rach, S.; Herrmann, C.S. Transcranial Alternating Current Stimulation Enhances Individual Alpha Activity in Human EEG. PLoS ONE 2010, 5, e13766. [Google Scholar] [CrossRef]
- Zaghi, S.; Rezende, L.D.F.; Oliveira, L.; El-Nazer, R.; Menning, S.; Tadini, L.; Fregni, F. Inhibition of motor cortex excitability with 15 Hz transcranial alternating current stimulation (tACS). Neurosci. Lett. 2010, 479, 211–214. [Google Scholar] [CrossRef]
- Wach, C.; Krause, V.; Moliadze, V.; Paulus, W.; Schnitzler, A.; Pollok, B. Effects of 10 Hz and 20 Hz transcranial alternating current stimulation (tACS) on motor functions and motor cortical excitability. Behav. Brain Res. 2013, 241, 1–6. [Google Scholar] [CrossRef]
- Schutter, D.J.L.G.; Hortensius, R. Brain oscillations and frequency-dependent modulation of cortical excitability. Brain Stimul. 2011, 4, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Veniero, D.; Vossen, A.; Gross, J.; Thut, G. Lasting EEG/MEG aftereffects of rhythmic transcranial brain stimulation: Level of control over oscillatory network activity. Front. Cell. Neurosci. 2015, 9, 477. [Google Scholar] [CrossRef] [PubMed]
- Bland, N.S.; Sale, M.V. Current challenges: The ups and downs of tACS. Exp. Brain Res. 2019, 237, 3071–3088. [Google Scholar] [CrossRef]
- Kanai, R.; Chaieb, L.; Antal, A.; Walsh, V.; Paulus, W. Frequency-Dependent Electrical Stimulation of the Visual Cortex. Curr. Biol. 2008, 18, 1839–1843. [Google Scholar] [CrossRef] [PubMed]
- Laczó, B.; Antal, A.; Niebergall, R.; Treue, S.; Paulus, W. Transcranial alternating stimulation in a high gamma frequency range applied over V1 improves contrast perception but does not modulate spatial attention. Brain Stimul. 2012, 5, 484–491. [Google Scholar] [CrossRef]
- Antal, A.; Boros, K.; Poreisz, C.; Chaieb, L.; Terney, D.; Paulus, W. Comparatively weak after-effects of transcranial alternating current stimulation (tACS) on cortical excitability in humans. Brain Stimul. 2008, 1, 97–105. [Google Scholar] [CrossRef]
- Pogosyan, A.; Gaynor, L.D.; Eusebio, A.; Brown, P. Boosting Cortical Activity at Beta-Band Frequencies Slows Movement in Humans. Curr. Biol. 2009, 19, 1637. [Google Scholar] [CrossRef]
- Feurra, M.; Bianco, G.; Santarnecchi, E.; del Testa, M.; Rossi, A.; Rossi, S. Frequency-Dependent Tuning of the Human Motor System Induced by Transcranial Oscillatory Potentials. J. Neurosci. 2011, 31, 12165–12170. [Google Scholar] [CrossRef]
- Brignani, D.; Ruzzoli, M.; Mauri, P.; Miniussi, C. Is Transcranial Alternating Current Stimulation Effective in Modulating Brain Oscillations? PLoS ONE 2013, 8, e56589. [Google Scholar] [CrossRef]
- Wach, C.; Krause, V.; Moliadze, V.; Paulus, W.; Schnitzler, A.; Pollok, B. The effect of 10 Hz transcranial alternating current stimulation (tACS) on corticomuscular coherence. Front. Hum. Neurosci. 2013, 7, 511. [Google Scholar] [CrossRef] [PubMed]
- Mulquiney, P.G.; Hoy, K.E.; Daskalakis, Z.J.; Fitzgerald, P.B. Improving working memory: Exploring the effect of transcranial random noise stimulation and transcranial direct current stimulation on the dorsolateral prefrontal cortex. Clin. Neurophysiol. 2011, 122, 2384–2389. [Google Scholar] [CrossRef] [PubMed]
- Boggio, P.S.; Rigonatti, S.P.; Ribeiro, R.; Myczkowski, M.L.; Nitsche, M.A.; Pascual-Leone, A.; Fregni, F. A randomized, double-blind clinical trial on the efficacy of cortical direct current stimulation for the treatment of major depression. Int. J. Neuropsychopharmacol. 2008, 11, 249–254. [Google Scholar] [CrossRef]
- Polanía, R.; Nitsche, M.A.; Korman, C.; Batsikadze, G.; Paulus, W. The importance of timing in segregated theta phase-coupling for cognitive performance. Curr. Biol. 2012, 22, 1314–1318. [Google Scholar] [CrossRef] [PubMed]
- Cappelletti, M.; Gessaroli, E.; Hithersay, R.; Mitolo, M.; Didino, D.; Kanai, R.; Kadosh, R.C.; Walsh, V. Transfer of Cognitive Training across Magnitude Dimensions Achieved with Concurrent Brain Stimulation of the Parietal Lobe. J. Neurosci. 2013, 33, 14899. [Google Scholar] [CrossRef]
- Grabner, R.H.; Krenn, J.; Fink, A.; Arendasy, M.; Benedek, M. Effects of alpha and gamma transcranial alternating current stimulation (tACS) on verbal creativity and intelligence test performance. Neuropsychologia 2018, 118, 91–98. [Google Scholar] [CrossRef]
- Santarnecchi, E.; Polizzotto, N.R.; Godone, M.; Giovannelli, F.; Feurra, M.; Matzen, L.; Rossi, A.; Rossi, S. Frequency-dependent enhancement of fluid intelligence induced by transcranial oscillatory potentials. Curr. Biol. 2013, 23, 1449–1453. [Google Scholar] [CrossRef]
- Sauseng, P.; Griesmayr, B.; Freunberger, R.; Klimesch, W. Control mechanisms in working memory: A possible function of EEG theta oscillations. Neurosci. Biobehav. Rev. 2010, 34, 1015–1022. [Google Scholar] [CrossRef]
- Jaušovec, N.; Jaušovec, K. Increasing working memory capacity with theta transcranial alternating current stimulation (tACS). Biol. Psychol. 2014, 96, 42–47. [Google Scholar] [CrossRef]
- Jaušovec, N.; Jaušovec, K.; Pahor, A. The influence of theta transcranial alternating current stimulation (tACS) on working memory storage and processing functions. Acta Psychol. 2014, 146, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Pahor, A.; Jaušovec, N. The effects of theta transcranial alternating current stimulation (tACS) on fluid intelligence. Int. J. Psychophysiol. 2014, 93, 322–331. [Google Scholar] [CrossRef] [PubMed]
- Neuling, T.; Rach, S.; Herrmann, C.S. Orchestrating neuronal networks: Sustained after-effects of transcranial alternating current stimulation depend upon brain states. Front. Hum. Neurosci. 2013, 7, 161. [Google Scholar] [CrossRef] [PubMed]
- Klimesch, W. Alpha-band oscillations, attention, and controlled access to stored information. Trends Cogn. Sci. 2012, 16, 606–617. [Google Scholar] [CrossRef]
- Voss, U.; Holzmann, R.; Hobson, A.; Paulus, W.; Koppehele-Gossel, J.; Klimke, A.; A Nitsche, M. Induction of self awareness in dreams through frontal low current stimulation of gamma activity. Nat. Neurosci. 2014, 17, 810–812. [Google Scholar] [CrossRef]
- Klink, K.; Paßmann, S.; Kasten, F.H.; Peter, J. The modulation of cognitive performance with transcranial alternating current stimulation: A systematic review of frequency-specific effects. Brain Sci. 2020, 10, 932. [Google Scholar] [CrossRef]
- Schutter, D.J.L.G.; Wischnewski, M. A meta-analytic study of exogenous oscillatory electric potentials in neuroenhancement. Neuropsychologia 2016, 86, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Figee, M.; Mayberg, H. The future of personalized brain stimulation. Nat. Med. 2021, 27, 196–197. [Google Scholar] [CrossRef]
- Frohlich, F.; Riddle, J. Conducting double-blind placebo-controlled clinical trials of transcranial alternating current stimulation (tACS). Transl. Psychiatry 2021, 11, 1–12. [Google Scholar] [CrossRef]
- Van Driel, J.; Sligte, I.G.; Linders, J.; Elport, D.; Cohen, M.X. Frequency Band-Specific Electrical Brain Stimulation Modulates Cognitive Control Processes. PLoS ONE 2015, 10, e0138984. [Google Scholar] [CrossRef]
- Onslow, A.C.E.; Bogacz, R.; Jones, M.W. Quantifying phase–amplitude coupling in neuronal network oscillations. Prog. Biophys. Mol. Biol. 2011, 105, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Meiron, O.; Lavidor, M. Prefrontal oscillatory stimulation modulates access to cognitive control references in retrospective metacognitive commentary. Clin. Neurophysiol. 2014, 125, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Perez, M.; Wakui, E.; Thoma, V.; Nitsche, M.A.; Rivolta, D. Transcranial alternating current stimulation (tACS) at 40 Hz enhances face and object perception. Neuropsychologia 2019, 135, 107237. [Google Scholar] [CrossRef] [PubMed]
- Abellaneda-Pérez, K.; Vaqué-Alcázar, L.; Perellón-Alfonso, R.; Bargalló, N.; Kuo, M.F.; Pascual-Leone, A.; Nitsche, M.A.; Bartrés-Faz, D. Differential tDCS and tACS Effects on Working Memory-Related Neural Activity and Resting-State Connectivity. Front. Neurosci. 2020, 13, 1440. [Google Scholar] [CrossRef]
- Sahu, P.P.; Tseng, P. Frontoparietal theta tACS nonselectively enhances encoding, maintenance, and retrieval stages in visuospatial working memory. Neurosci. Res. 2021, 172, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Chander, B.S.; Witkowski, M.; Braun, C.; Robinson, S.E.; Born, J.; Cohen, L.G.; Birbaumer, N.; Soekadar, S.R. tACS Phase Locking of Frontal Midline Theta Oscillations Disrupts Working Memory Performance. Front. Cell. Neurosci. 2016, 10, 120. [Google Scholar] [CrossRef] [PubMed]
- Vosskuhl, J.; Huster, R.J.; Herrmann, C.S. Increase in short-term memory capacity induced by down-regulating individual theta frequency via transcranial alternating current stimulation. Front. Hum. Neurosci. 2015, 9, 257. [Google Scholar] [CrossRef] [PubMed]
- Alekseichuk, I.; Pabel, S.C.; Antal, A.; Paulus, W. Intrahemispheric theta rhythm desynchronization impairs working memory. Restor. Neurol. Neurosci. 2017, 35, 147–158. [Google Scholar] [CrossRef]
- Tseng, P.; Iu, K.C.; Juan, C.H. The critical role of phase difference in theta oscillation between bilateral parietal cortices for visuospatial working memory. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef]
- Violante, I.R.; Li, L.M.; Carmichael, D.W.; Lorenz, R.; Leech, R.; Hampshire, A.; Rothwell, J.C.; Sharp, D.J. Externally induced frontoparietal synchronization modulates network dynamics and enhances working memory performance. Elife 2017, 6, e22001. [Google Scholar] [CrossRef]
- Kleinert, M.L.; Szymanski, C.; Müller, V. Frequency-unspecific effects of θ-tACS related to a visuospatial working memory task. Front. Hum. Neurosci. 2017, 11, 367. [Google Scholar] [CrossRef]
- Becher, A.K.; Höhne, M.; Axmacher, N.; Chaieb, L.; Elger, C.E.; Fell, J. Intracranial electroencephalography power and phase synchronization changes during monaural and binaural beat stimulation. Eur. J. Neurosci. 2015, 41, 254–263. [Google Scholar] [CrossRef] [PubMed]
- Fries, P. Neuronal gamma-band synchronization as a fundamental process in cortical computation. Annu. Rev. Neurosci. 2009, 32, 209–224. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Kohn, A. Gamma Rhythms in the Brain. PLoS Biol. 2011, 9, e1001045. [Google Scholar] [CrossRef] [PubMed]
- Hoy, K.E.; Bailey, N.; Arnold, S.; Windsor, K.; John, J.; Daskalakis, Z.J.; Fitzgerald, P.B. The effect of γ-tACS on working memory performance in healthy controls. Brain. Cogn. 2015, 101, 51–56. [Google Scholar] [CrossRef]
- Tseng, P.; Chang, Y.T.; Chang, C.F.; Liang, W.K.; Juan, C.H. The critical role of phase difference in gamma oscillation within the temporoparietal network for binding visual working memory. Sci. Rep. 2016, 6, 32138. [Google Scholar] [CrossRef]
- Möller, A.; Nemmi, F.; Karlsson, K.; Klingberg, T. Transcranial Electric Stimulation Can Impair Gains during Working Memory Training and Affects the Resting State Connectivity. Front. Hum. Neurosci. 2017, 11, 364. [Google Scholar] [CrossRef]
- Misselhorn, J.; Göschl, F.; Higgen, F.L.; Hummel, F.C.; Gerloff, C.; Engel, A.K. Sensory capability and information integration independently explain the cognitive status of healthy older adults. Sci. Rep. 2020, 10, 22437. [Google Scholar] [CrossRef]
- Santarnecchi, E.; Muller, T.; Rossi, S.; Sarkar, A.; Polizzotto, N.; Rossi, A.; Kadosh, R.C. Individual differences and specificity of prefrontal gamma frequency-tACS on fluid intelligence capabilities. Cortex 2016, 75, 33–43. [Google Scholar] [CrossRef]
- Pahor, A.; Jaušovec, N. The effects of theta and gamma tacs on working memory and electrophysiology. Front. Hum. Neurosci. 2018, 11, 651. [Google Scholar] [CrossRef]
- Lisman, J.E.; Jensen, O. The Theta-Gamma Neural Code. Neuron 2013, 77, 1002–1016. [Google Scholar] [CrossRef] [PubMed]
- de Lara, G.A.; Alekseichuk, I.; Turi, Z.; Lehr, A.; Antal, A.; Paulus, W. Perturbation of theta-gamma coupling at the temporal lobe hinders verbal declarative memory. Brain Stimul. 2018, 11, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Turi, Z.; Mittner, M.; Lehr, A.; Bürger, H.; Antal, A.; Paulus, W. θ-γ Cross-Frequency Transcranial Alternating Current Stimulation over the Trough Impairs Cognitive Control. ENeuro 2020, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Riddle, J.; McFerren, A.; Frohlich, F. Causal role of cross-frequency coupling in distinct components of cognitive control. Prog. Neurobiol. 2021, 202, 102033. [Google Scholar] [CrossRef]
- Alekseichuk, I.; Turi, Z.; Amador de Lara, G.; Antal, A.; Paulus, W. Spatial Working Memory in Humans Depends on Theta and High Gamma Synchronization in the Prefrontal Cortex. Curr. Biol. 2016, 26, 1513–1521. [Google Scholar] [CrossRef]
- Delorme, A.; Makeig, S. EEGLAB: An open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods 2004, 134, 9–21. [Google Scholar] [CrossRef]
- Benesty, J.; Chen, J.; Huang, Y. A generalized MVDR spectrum. IEEE Signal. Process. Lett. 2005, 12, 827–830. [Google Scholar] [CrossRef]
- Grover, S.; Wen, W.; Viswanathan, V.; Gill, C.T.; Reinhart, R.M.G. Long-lasting, dissociable improvements in working memory and long-term memory in older adults with repetitive neuromodulation. Nat. Neurosci. 2022, 25, 1237–1246. [Google Scholar] [CrossRef]
- Pittman-polletta, B.; Hsieh, W.H.; Kaur, S.; Lo, M.T.; Hu, K. Detecting phase-amplitude coupling with high frequency resolution using adaptive decompositions. J. Neurosci. Methods 2014, 226, 15–32. [Google Scholar] [CrossRef]
- Joundi, R.A.; Jenkinson, N.; Brittain, J.S.; Aziz, T.Z.; Brown, P. Driving Oscillatory Activity in the Human Cortex Enhances Motor Performance. Curr. Biol. 2012, 22, 403. [Google Scholar] [CrossRef]
- Kasten, F.H.; Dowsett, J.; Herrmann, C.S. Sustained aftereffect of α-tACS lasts up to 70 min after stimulation. Front. Hum. Neurosci. 2016, 10, 245. [Google Scholar] [CrossRef] [PubMed]
- Elyamany, O.; Leicht, G.; Herrmann, C.S.; Mulert, C. Transcranial alternating current stimulation (tACS): From basic mechanisms towards first applications in psychiatry. Eur. Arch. Psychiatry Clin. Neurosci. 2021, 271, 135–156. [Google Scholar] [CrossRef] [PubMed]
- Moussavi, Z.; Kimura, K.; Kehler, L.; de Oliveira Francisco, C.; Lithgow, B. A Novel Program to Improve Cognitive Function in Individuals With Dementia Using Transcranial Alternating Current Stimulation (tACS) and Tutored Cognitive Exercises. Front. Aging 2021, 2, 632545. [Google Scholar] [CrossRef]
- Kehler, L.; Francisco, C.O.; Uehara, M.A.; Moussavi, Z. The effect of transcranial alternating current stimulation (tACS) on cognitive function in older adults with dementia. In Proceedings of the 2020 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Montreal, QC, Canada, 20–24 July 2020; Volume 2020, pp. 3649–3653. [Google Scholar] [CrossRef]
- Haller, N.; Hasan, A.; Padberg, F.; Brunelin, J.; da Costa Lane Valiengo, L.; Palm, U. Gamma transcranial alternating current stimulation in patients with negative symptoms in schizophrenia: A case series. Neurophysiol. Clin. 2020, 50, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Bréchet, L.; Yu, W.; Biagi, M.C.; Ruffini, G.; Gagnon, M.; Manor, B.; Pascual-Leone, A. Patient-Tailored, Home-Based Non-invasive Brain Stimulation for Memory Deficits in Dementia Due to Alzheimer’s Disease. Front. Neurol. 2021, 12, 775. [Google Scholar] [CrossRef] [PubMed]


| Condition | Median RT (ms) | SD | SEM | |||
|---|---|---|---|---|---|---|
| Before | After | Before | After | Before | After | |
| Sham | 587 | 378 | 244 | 139 | 39.6 | 22.5 |
| tACS | 605 | 355 | 255 | 162 | 40.3 | 25.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kvašňák, E.; Magyarová, E.; Domankuš, M.; Tesař, M.; Kymplová, J.; Fetissov, V.; Abubaker, M.; Al Qasem, W. 10 Minutes Frontal 40 Hz tACS—Effects on Working Memory Tested by Luck-Vogel Task. Behav. Sci. 2023, 13, 39. https://doi.org/10.3390/bs13010039
Kvašňák E, Magyarová E, Domankuš M, Tesař M, Kymplová J, Fetissov V, Abubaker M, Al Qasem W. 10 Minutes Frontal 40 Hz tACS—Effects on Working Memory Tested by Luck-Vogel Task. Behavioral Sciences. 2023; 13(1):39. https://doi.org/10.3390/bs13010039
Chicago/Turabian StyleKvašňák, Eugen, Eva Magyarová, Miroslav Domankuš, Michael Tesař, Jaroslava Kymplová, Vitaly Fetissov, Mohammed Abubaker, and Wiam Al Qasem. 2023. "10 Minutes Frontal 40 Hz tACS—Effects on Working Memory Tested by Luck-Vogel Task" Behavioral Sciences 13, no. 1: 39. https://doi.org/10.3390/bs13010039
APA StyleKvašňák, E., Magyarová, E., Domankuš, M., Tesař, M., Kymplová, J., Fetissov, V., Abubaker, M., & Al Qasem, W. (2023). 10 Minutes Frontal 40 Hz tACS—Effects on Working Memory Tested by Luck-Vogel Task. Behavioral Sciences, 13(1), 39. https://doi.org/10.3390/bs13010039





