A Biopsychosocial Approach to Grief, Depression, and the Role of Emotional Regulation
Abstract
1. Introduction
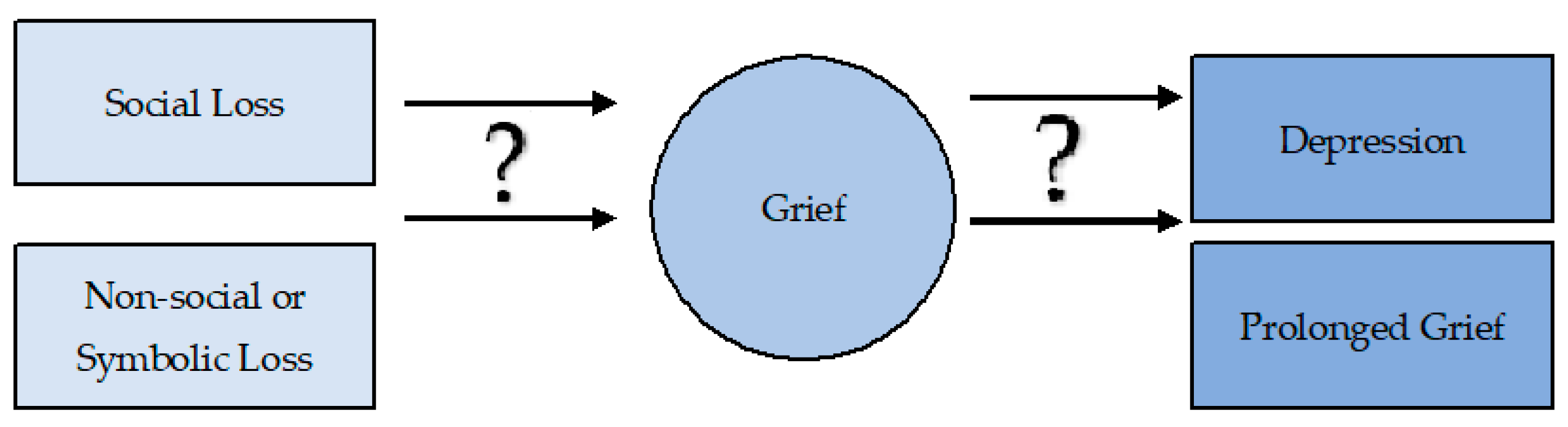
2. Loss and Grief: How Are They Associated with Prolonged Grief and Depression?
3. Neuro-Affective Pathways and Animal Models of Depression Linked to Grief
4. Loss, Grief, Depression, and Inflammatory Markers
5. Grief, Emotional Regulation, and Inflammatory Biomarkers
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- MedlinePlus. Grief. Available online: https://medlineplus.gov/ency/article/001530.htm (accessed on 1 February 2021).
- Shear, M.K. Complicated grief. N. Engl. J. Med. 2015, 372, 153–160. [Google Scholar] [CrossRef]
- Koblenz, J. Growing from grief: Qualitative experiences of parental loss. OMEGA J. Death Dying 2016, 73, 203–230. [Google Scholar] [CrossRef]
- Bagbey, C.D.D. A New Mourning: Synthesizing an Interactive Model of Adaptive Grieving Dynamics. Illn. Crisis Loss 2014, 22, 195–235. [Google Scholar]
- Coenen, V.A.; Schlaepfer, T.E.; Maedler, B.; Panksepp, J. Cross-species affective functions of the medial forebrain bundle—Implications for the treatment of affective pain and depression in humans. Neurosci. Biobehav. Rev. 2011, 35, 1971–1981. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.Y.; Haley, W.E.; Small, B.J.; Schonwetter, R.S.; McMillan, S.C. Bereavement among hospice caregivers of cancer patients one year following loss: Predictors of grief, complicated grief, and symptoms of depression. J. Palliat. Med. 2013, 16, 745–751. [Google Scholar] [CrossRef] [PubMed]
- Latham, A.E.; Prigerson, H.G. Suicidality and bereavement: Complicated grief as psychiatric disorder presenting greatest risk for suicidality. Suicide Life-Threat. Behav. 2004, 34, 350–362. [Google Scholar] [CrossRef]
- Kübler-Ross, E. Questions and Answers on Death and Dying; Macmillan: New York, NY, USA, 1974. [Google Scholar]
- Stroebe, M.; Schut, H.; Boerner, K. Cautioning health-care professionals: Bereaved persons are misguided through the stages of grief. OMEGA J. Death Dying 2017, 74, 455–473. [Google Scholar] [CrossRef]
- Friedman, R.; James, J.W. The Myth of the Stages of Dying, Death, and Grief; The Skeptics Society: Altadena, CA, USA, 2008; Volume 14, pp. 37–42. [Google Scholar]
- Corr, C.A. Let’s stop “staging” persons who are coping with loss. Illn. Crisis Loss 2015, 23, 226–241. [Google Scholar] [CrossRef]
- Corr, C.A. The ‘five stages’ in coping with dying and bereavement: Strengths, weaknesses and some alternatives. Mortality 2019, 24, 405–417. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- World Health Organization. International Statistical Classification of Diseases and Related Health Problems, 11th ed.; World Health Organization: Geneva, Switzerladnd, 2020. [Google Scholar]
- Djelantik, A.M.J.; Smid, G.E.; Kleber, R.J.; Boelen, P.A. Symptoms of prolonged grief, post-traumatic stress, and depression after loss in a Dutch community sample: A latent class analysis. Psychiatry Res. 2017, 247, 276–281. [Google Scholar] [CrossRef]
- Boelen, P.A.; Lenferink, L.I. Symptoms of prolonged grief, posttraumatic stress, and depression in recently bereaved people: Symptom profiles, predictive value, and cognitive behavioral correlates. Soc. Psychiatry Psychiatr. Epidemiol. 2019, 55, 1–13. [Google Scholar] [CrossRef]
- Lenferink, L.I.; de Keijser, J.; Smid, G.E.; Djelantik, A.M.J.; Boelen, P.A. Prolonged grief, depression, and posttraumatic stress in disaster-bereaved individuals: Latent class analysis. Eur. J. Psychotraumatol. 2017, 8, 1298311. [Google Scholar] [CrossRef] [PubMed]
- Lenferink, L.I.; Nickerson, A.; de Keijser, J.; Smid, G.E.; Boelen, P.A. Trajectories of grief, depression, and posttraumatic stress in disaster-bereaved people. Depress. Anxiety 2020, 37, 35–44. [Google Scholar] [CrossRef]
- Malgaroli, M.; Maccallum, F.; Bonanno, G.A. Symptoms of persistent complex bereavement disorder, depression, and PTSD in a conjugally bereaved sample: A network analysis. Psychol. Med. 2018, 48, 2439–2448. [Google Scholar] [CrossRef]
- Papa, A.; Lancaster, N.G.; Kahler, J. Commonalities in grief responding across bereavement and non-bereavement losses. J. Affect. Disord. 2014, 161, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Schaal, S.; Dusingizemungu, J.P.; Jacob, N.; Neuner, F.; Elbert, T. Associations between prolonged grief disorder, depression, posttraumatic stress disorder, and anxiety in Rwandan genocide survivors. Death Stud. 2012, 36, 97–117. [Google Scholar] [CrossRef]
- Mitchell, M.B. “No one acknowledged my loss and hurt”: Non-death loss, grief, and trauma in foster care. Child Adolesc. Soc. Work J. 2018, 35, 1–9. [Google Scholar] [CrossRef]
- Gitterman, A.; Knight, C. Non-death loss: Grieving for the loss of familiar place and for precious time and associated opportunities. Clin. Soc. Work J. 2019, 47, 147–155. [Google Scholar] [CrossRef]
- Cooley, E.; Toray, T.; Roscoe, L. Reactions to loss scale: Assessing grief in college students. OMEGA J. Death Dying 2010, 61, 25–51. [Google Scholar] [CrossRef]
- Burns, V.F.; Sussman, T.; Bourgeois-Guerin, V. Later-life homelessness as disenfranchised grief. Can. J. Aging 2018, 37, 171–184. [Google Scholar] [CrossRef]
- Neimeyer, R.A. The changing face of grief: Contemporary directions in theory, research, and practice. Prog. Palliat. Care 2014, 22, 125–130. [Google Scholar] [CrossRef]
- O’Connor, M.F. Grief: A Brief History of Research on How Body, Mind, and Brain Adapt. Psychosom. Med. 2019, 81, 731–738. [Google Scholar] [CrossRef]
- Seiler, A.; von Känel, R.; Slavich, G.M. The Psychobiology of Bereavement and Health: A Conceptual Review from the Perspective of Social Signal Transduction Theory of Depression. Front. Psychiatry 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Lopez, R.B.; Denny, B.T.; Fagundes, C.P. Neural mechanisms of emotion regulation and their role in endocrine and immune functioning: A review with implications for treatment of affective disorders. Neurosci. Biobehav. Rev. 2018, 95, 508–514. [Google Scholar] [CrossRef] [PubMed]
- Walter, C.A.; McCoyd, J.L. Grief and Loss Across the Lifespan: A Biopsychosocial Perspective; Springer Publishing: New York, NY, USA, 2015. [Google Scholar]
- Slavich, G.M.; Irwin, M.R. From stress to inflammation and major depressive disorder: A social signal transduction theory of depression. Psychol. Bull. 2014, 140, 774. [Google Scholar] [CrossRef]
- Panksepp, J. The basic emotional circuits of mammalian brains: Do animals have affective lives? Neurosci. Biobehav. Rev. 2011, 35, 1791–1804. [Google Scholar] [CrossRef] [PubMed]
- Panksepp, J.; Watt, D. Why does depression hurt? Ancestral primary-process separation-distress (PANIC/GRIEF) and diminished brain reward (SEEKING) processes in the genesis of depressive affect. Psychiatry Interpers. Biol. Process. 2011, 74, 5–13. [Google Scholar] [CrossRef]
- Mason, T.M.; Tofthagen, C.S.; Buck, H.G. Complicated grief: Risk factors, protective factors, and interventions. J. Soc. Work End Life Palliat. Care 2020, 16, 151–174. [Google Scholar] [CrossRef]
- Smid, G.E.; Groen, S.; de la Rie, S.M.; Kooper, S.; Boelen, P.A. Toward cultural assessment of grief and grief-related psychopathology. Psychiatr. Serv. 2018, 69, 1050–1052. [Google Scholar] [CrossRef]
- Lee, J.S.; Kim, E.Y.; Choi, Y.; Koo, J.H. Cultural variances in composition of biological and supernatural concepts of death: A content analysis of children’s literature. Death Stud. 2014, 38, 538–545. [Google Scholar] [CrossRef]
- Rosenblatt, P.C. Researching grief: Cultural, relational, and individual possibilities. J. Loss Trauma 2017, 22, 617–630. [Google Scholar] [CrossRef]
- Walsh, F.; McGoldrick, M. Bereavement: A family life cycle perspective. Fam. Sci. 2013, 4, 20–27. [Google Scholar] [CrossRef]
- Biank, N.M.; Werner-Lin, A. Growing up with grief: Revisiting the death of a parent over the life course. OMEGA J. Death Dying 2011, 63, 271–290. [Google Scholar] [CrossRef]
- Bonoti, F.; Leondari, A.; Mastora, A. Exploring children’s understanding of death: Through drawings and the death concept questionnaire. Death Stud. 2013, 37, 47–60. [Google Scholar] [CrossRef] [PubMed]
- Coelho, A.; Barbosa, A. Family anticipatory grief: An integrative literature review. Am. J. Hosp. Palliat. Med. 2017, 34, 774–785. [Google Scholar] [CrossRef] [PubMed]
- Gaudio, F.D.; Hichenberg, S.; Eisenberg, M.; Kerr, E.; Zaider, T.I.; Kissane, D.W. Latino values in the context of palliative care: Illustrative cases from the family focused grief therapy trial. Am. J. Hosp. Palliat. Med. 2013, 30, 271–278. [Google Scholar] [CrossRef]
- Mondia, S.; Hichenberg, S.; Kerr, E.; Eisenberg, M.; Kissane, D.W. The impact of Asian American value systems on palliative care: Illustrative cases from the family-focused grief therapy trial. Am. J. Hosp. Palliat. Med. 2012, 29, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Panagiotaki, G.; Hopkins, M.; Nobes, G.; Ward, E.; Griffiths, D. Children’s and adults’ understanding of death: Cognitive, parental, and experiential influences. J. Exp. Child Psychol. 2018, 166, 96–115. [Google Scholar] [CrossRef] [PubMed]
- Dyregrov, A.; Dyregrov, K. Complicated grief in children—the perspectives of experienced professionals. OMEGA J. Death Dying 2013, 67, 291–303. [Google Scholar] [CrossRef] [PubMed]
- Ridaura, I.; Penelo, E.; Raich, R.M. Depressive symptomatology and grief in Spanish women who have suffered a perinatal loss. Psicothema 2017, 29, 43–48. [Google Scholar] [PubMed]
- Boelen, P.A.; Reijntjes, A.; Djelantik, A.M.; Smid, G.E. Prolonged grief and depression after unnatural loss: Latent class analyses and cognitive correlates. Psychiatry Res. 2016, 240, 358–363. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.K.; Neergaard, M.A.; Jensen, A.B.; Vedsted, P.; Bro, F.; Guldin, M.B. Predictors of complicated grief and depression in bereaved caregivers: A nationwide prospective cohort study. J. Pain Symptom Manag. 2017, 53, 540–550. [Google Scholar] [CrossRef]
- Onrust, S.; Cuijpers, P.; Smit, F.; Bohlmeijer, E. Predictors of psychological adjustment after bereavement. Int. Psychogeriatr. 2007, 19, 921–934. [Google Scholar] [CrossRef]
- Al-Gamal, E.; Saeed, S.B.; Victor, A.; Long, T. Prolonged grief disorder and its relationship with perceived social support and depression among university students. J. Psychosoc. Nurs. Ment. Health Serv. 2018, 57, 44–51. [Google Scholar] [CrossRef]
- Morris, C.E.; Reiber, C. Frequency, intensity, and expression of post-relationship grief. EvoS J. J. Evol. Stud. Consort. 2011, 3, 1–11. [Google Scholar]
- Chen, R. Social support as a protective factor against the effect of grief reactions on depression for bereaved single older adults. Death Stud. 2020, 1–8. [Google Scholar] [CrossRef]
- Von Cheong, E.; Sinnott, C.; Dahly, D.; Kearney, P.M. Adverse childhood experiences (ACEs) and later-life depression: Perceived social support as a potential protective factor. BMJ Open 2017, 7, e013228. [Google Scholar] [CrossRef]
- Reid, K.M.; Taylor, M.G. Social support, stress, and maternal postpartum depression: A comparison of supportive relationships. Soc. Sci. Res. 2015, 54, 246–262. [Google Scholar] [CrossRef]
- McGuire, A.P.; Gauthier, J.M.; Anderson, L.M.; Hollingsworth, D.W.; Tracy, M.; Galea, S.; Coffey, S.F. Social support moderates effects of natural disaster exposure on depression and posttraumatic stress disorder symptoms: Effects for displaced and nondisplaced residents. J. Trauma. Stress 2018, 31, 223–233. [Google Scholar] [CrossRef]
- Ye, M.; DeMaris, A.; Longmore, M.A. Role of marital quality in explaining depressive symptoms after marital termination among older adults. Marriage Fam. Rev. 2018, 54, 34–49. [Google Scholar] [CrossRef]
- Gariepy, G.; Honkaniemi, H.; Quesnel-Vallee, A. Social support and protection from depression: Systematic review of current findings in Western countries. Br. J. Psychiatry 2016, 209, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Zamani, C. Ambiguous Loss & Friendship: The Meaning of Friendship. Ph.D. Thesis, Alliant International University, Alhambra, CA, USA, 2020. [Google Scholar]
- O’Connor, M.F.; Sussman, T.J. Developing the yearning in situations of loss scale: Convergent and discriminant validity for bereavement, romantic breakup, and homesickness. Death Stud. 2014, 38, 450–458. [Google Scholar] [CrossRef]
- Reimer, J.E.; Estrada, A.R. College students’ grief over a breakup. J. Loss Trauma 2021, 26, 179–191. [Google Scholar] [CrossRef]
- Yárnoz-Yaben, S. Grief due to divorce: Relationship with attachment style and effects on subjective well-being and co-parenting/El duelo ante el divorcio: Relación con el estilo de apego y efectos en el bienestar subjetivo y el ejercicio de la co-parentalidad. Estud. Psicol. 2017, 38, 667–688. [Google Scholar] [CrossRef]
- Morris, C.E.; Reiber, C.; Roman, E. Quantitative sex differences in response to the dissolution of a romantic relationship. Evol. Behav. Sci. 2015, 9, 270. [Google Scholar] [CrossRef]
- Brewington, J.O.; Nassar-McMillan, S.C.; Flowers, C.P.; Furr, S.R. A preliminary investigation of factors associated with job loss grief. Career Dev. Q. 2004, 53, 78–83. [Google Scholar] [CrossRef]
- Archer, J.; Rhodes, V. The grief process and job loss: A cross-sectional study. Br. J. Psychol. 1993, 84, 395–410. [Google Scholar] [CrossRef] [PubMed]
- Mandal, B.; Ayyagari, P.; Gallo, W.T. Job loss and depression: The role of subjective expectations. Soc. Sci. Med. 2011, 72, 576–583. [Google Scholar] [CrossRef]
- Papa, A.; Maitoza, R. The Role of Loss in the Experience of Grief: The Case of Job Loss. J. Loss Trauma 2013, 18, 152–169. [Google Scholar] [CrossRef]
- Stolove, C.A.; Galatzer-Levy, I.R.; Bonanno, G.A. Emergence of depression following job loss prospectively predicts lower rates of reemployment. Psychiatry Res. 2017, 253, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Price, R.H.; Choi, J.N.; Vinokur, A.D. Links in the chain of adversity following job loss: How financial strain and loss of personal control lead to depression, impaired functioning, and poor health. J. Occup. Health Psychol. 2002, 7, 302. [Google Scholar] [CrossRef] [PubMed]
- Carrillo-Alarcón, L.C.; López-López, E.; López-Carbajal, M.J.; Ortiz, M.I.; Ponce-Montern, H.E. Level of knowledge in patients with type 2 diabetes mellitus and its relationship with glycemic levels and stages of grief according to Kübler-Ross. J. Diabetes Metab. 2015, 6, 2. [Google Scholar] [CrossRef]
- Bergeron, C.M.; Wanet-Defalque, M.C. Psychological adaptation to visual impairment: The traditional grief process revised. Br. J. Vis. Impair. 2013, 31, 20–31. [Google Scholar] [CrossRef]
- Trusson, D.; Pilnick, A. The role of hair loss in cancer identity: Perceptions of chemotherapy-induced alopecia among women treated for early-stage breast cancer or ductal carcinoma in situ. Cancer Nurs. 2017, 40, E9–E16. [Google Scholar] [CrossRef]
- Danemalm Jägervall, C.; Brüggemann, J.; Johnson, E. Gay men’s experiences of sexual changes after prostate cancer treatment—A qualitative study in Sweden. Scand. J. Urol. 2019, 53, 40–44. [Google Scholar] [CrossRef]
- Schmidt, H.D.; Shelton, R.C.; Duman, R.S. Functional biomarkers of depression: Diagnosis, treatment, and pathophysiology. Neuropsychopharmacology 2011, 36, 2375–2394. [Google Scholar] [CrossRef] [PubMed]
- Budge, S.L.; Adelson, J.L.; Howard, K.A. Anxiety and depression in transgender individuals: The roles of transition status, loss, social support, and coping. J. Consult. Clin. Psychol. 2013, 81, 545. [Google Scholar] [CrossRef] [PubMed]
- Panksepp, J.; Watt, D. What is basic about basic emotions? Lasting lessons from affective neuroscience. Emot. Rev. 2011, 3, 387–396. [Google Scholar] [CrossRef]
- Ikemoto, S.; Panksepp, J. The effects of early social isolation on the motivation for social play in juvenile rats. Dev. Psychobiol. J. Int. Soc. Dev. Psychobiol. 1992, 25, 261–274. [Google Scholar] [CrossRef]
- Yates, G.; Panksepp, J.; Ikemoto, S.; Nelson, E.; Conner, R. Social isolation effects on the “behavioral despair” forced swimming test: Effect of age and duration of testing. Physiol. Behav. 1991, 49, 347–353. [Google Scholar] [CrossRef]
- Panksepp, J. Affective neuroscience of the emotional BrainMind: Evolutionary perspectives and implications for understanding depression. Dialogues Clin. Neurosci. 2010, 12, 533. [Google Scholar] [PubMed]
- Rilling, J.K.; Young, L.J. The biology of mammalian parenting and its effect on offspring social development. Science 2014, 345, 771–776. [Google Scholar] [CrossRef] [PubMed]
- Solms, M.; Panksepp, J. Why depression feels bad. New Horiz. Neurosci. Conscious. 2010, 79, 169–179. [Google Scholar]
- Watt, D.F.; Panksepp, J. Depression: An evolutionarily conserved mechanism to terminate separation distress? A review of aminergic, peptidergic, and neural network perspectives. Neuropsychoanalysis 2009, 11, 7–51. [Google Scholar] [CrossRef]
- Wright, J.S.; Panksepp, J. An evolutionary framework to understand foraging, wanting, and desire: The neuropsychology of the SEEKING system. Neuropsychoanalysis 2012, 14, 5–39. [Google Scholar] [CrossRef]
- Fischer, C.W.; Liebenberg, N.; Elfving, B.; Lund, S.; Wegener, G. Isolation-induced behavioral changes in a genetic animal model of depression. Behav. Brain Res. 2012, 230, 85–91. [Google Scholar] [CrossRef]
- Lapiz, M.D.; Fulford, A.; Muchimapura, S.; Mason, R.; Parker, T.; Marsden, C.A. Influence of postweaning social isolation in the rat on brain development, conditioned behavior, and neurotransmission. Neurosci. Behav. Physiol. 2003, 33, 13–29. [Google Scholar] [CrossRef]
- Graeff, F.G.; Guimarães, F.S.; De Andrade, T.G.; Deakin, J.F. Role of 5-HT in stress, anxiety, and depression. Pharmacol. Biochem. Behav. 1996, 54, 129–141. [Google Scholar] [CrossRef]
- Yohn, C.N.; Gergues, M.M.; Samuels, B.A. The role of 5-HT receptors in depression. Mol. Brain 2017, 10, 1–12. [Google Scholar] [CrossRef]
- Baik, J.H. Dopamine signaling in reward-related behaviors. Front. Neural Circuits 2013, 7, 152. [Google Scholar] [CrossRef] [PubMed]
- Nocjar, C.; Zhang, J.; Feng, P.; Panksepp, J. The social defeat animal model of depression shows diminished levels of orexin in mesocortical regions of the dopamine system, and of dynorphin and orexin in the hypothalamus. Neuroscience 2012, 218, 138–153. [Google Scholar] [CrossRef]
- Panksepp, J. What is an emotional feeling? Lessons about affective origins from cross-species neuroscience. Motiv. Emot. 2012, 36, 4–15. [Google Scholar] [CrossRef]
- Tanaka, M.; Tóth, F.; Polyák, H.; Szabó, Á.; Mándi, Y.; Vécsei, L. Immune Influencers in Action: Metabolites and Enzymes of the Tryptophan-Kynurenine Metabolic Pathway. Biomedicines 2021, 9, 734. [Google Scholar] [CrossRef]
- Cohen, M.; Granger, S.; Fuller-Thomson, E. The association between bereavement and biomarkers of inflammation. Behav. Med. 2015, 41, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Fagundes, C.P.; Murdock, K.W.; LeRoy, A.; Baameur, F.; Thayer, J.F.; Heijnen, C. Spousal bereavement is associated with more pronounced ex vivo cytokine production and lower heart rate variability: Mechanisms underlying cardiovascular risk? Psychoneuroendocrinology 2018, 93, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Uchino, B.N.; Trettevik, R.; Kent de Grey, R.G.; Cronan, S.; Hogan, J.; Baucom, B.R. Social support, social integration, and inflammatory cytokines: A meta-analysis. Health Psychol. 2018, 37, 462. [Google Scholar] [CrossRef]
- Hughes, S.; Jaremka, L.M.; Alfano, C.M.; Glaser, R.; Povoski, S.P.; Lipari, A.M.; Agnese, D.M.; Farrar, W.B.; Yee, L.D.; Carson, W.E., III; et al. Social support predicts inflammation, pain, and depressive symptoms: Longitudinal relationships among breast cancer survivors. Psychoneuroendocrinology 2014, 42, 38–44. [Google Scholar] [CrossRef]
- Fagundes, C.P.; Brown, R.L.; Chen, M.A.; Murdock, K.W.; Saucedo, L.; LeRoy, A.; Wu, E.L.; Garcini, L.M.; Shahane, A.D.; Baameur, F.; et al. Grief, depressive symptoms, and inflammation in the spousally bereaved. Psychoneuroendocrinology 2019, 100, 190–197. [Google Scholar] [CrossRef]
- Field, T. Romantic breakups, heartbreak and bereavement—Romantic breakups. Psychology 2011, 2, 382. [Google Scholar] [CrossRef]
- Das, A. Psychosocial distress and inflammation: Which way does causality flow? Soc. Sci. Med. 2016, 170, 1–8. [Google Scholar] [CrossRef]
- Liu, R.H.; Pan, J.Q.; Tang, X.E.; Li, B.; Liu, S.F.; Ma, W.L. The role of immune abnormality in depression and cardiovascular disease. J. Geriatr. Cardiol. JGC 2017, 14, 703–710. [Google Scholar] [PubMed]
- Troy, A.S.; Mauss, I.B. Resilience in the face of stress: Emotion regulation as a protective factor. Resil. Ment. Health Chall. Across Lifesp. 2011, 1, 30–44. [Google Scholar]
- Gross, J.J. The emerging field of emotion regulation: An integrative review. Rev. Gen. Psychol. 1998, 2, 271–299. [Google Scholar] [CrossRef]
- Lenferink, L.I.; Eisma, M.C.; de Keijser, J.; Boelen, P.A. Grief rumination mediates the association between self-compassion and psychopathology in relatives of missing persons. Eur. J. Psychotraumatol. 2017, 8, 1378052. [Google Scholar] [CrossRef]
- Gegieckaite, G.; Kazlauskas, E. Do emotion regulation difficulties mediate the association between neuroticism, insecure attachment, and prolonged grief? Death Stud. 2020, 3, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.J.; Jaremka, L.M.; Fagundes, C.P.; Andridge, R.; Peng, J.; Malarkey, W.B.; Habash, D.; Belury, M.A.; Kiecolt-Glaser, J.K. Shortened sleep fuels inflammatory responses to marital conflict: Emotion regulation matters. Psychoneuroendocrinology 2017, 79, 74–83. [Google Scholar] [CrossRef]
- Lopez, R.B.; Brown, R.L.; Wu, E.L.L.; Murdock, K.W.; Denny, B.T.; Heijnen, C.; Fagundes, C. Emotion Regulation and Immune Functioning During Grief: Testing the Role of Expressive Suppression and Cognitive Reappraisal in Inflammation Among Recently Bereaved Spouses. Psychosom. Med. 2020, 82, 2–9. [Google Scholar] [CrossRef]
- Appleton, A.A.; Buka, S.L.; Loucks, E.B.; Gilman, S.E.; Kubzansky, L.D. Divergent associations of adaptive and maladaptive emotion regulation strategies with inflammation. Health Psychol. Off. J. Div. Health Psychol. Am. Psychol. Assoc. 2013, 32, 748. [Google Scholar] [CrossRef] [PubMed]
| Model | Authors | Theory |
|---|---|---|
| Affective neuroscience approach to depression | Jaak Panksepp and Douglas Watt | Sustained and prolonged activation of the GRIEF system leads (by a yet-unknown mechanism) to inhibition of the reward-seeking system. Inhibition of the reward-seeking system in rats manifests as surrender and defeat in different tasks (used to measured depression phenotype), which Panksepp and Watt conceptualized as anhedonia—one of the primary symptoms of major depressive disorder (MDD). |
| Social signal transduction theory | George Slavish and Michael Irwin | Prolonged early stress (such as loss) is a predictor of MDD and dysregulation of inflammatory processes in chronic health conditions. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peña-Vargas, C.; Armaiz-Peña, G.; Castro-Figueroa, E. A Biopsychosocial Approach to Grief, Depression, and the Role of Emotional Regulation. Behav. Sci. 2021, 11, 110. https://doi.org/10.3390/bs11080110
Peña-Vargas C, Armaiz-Peña G, Castro-Figueroa E. A Biopsychosocial Approach to Grief, Depression, and the Role of Emotional Regulation. Behavioral Sciences. 2021; 11(8):110. https://doi.org/10.3390/bs11080110
Chicago/Turabian StylePeña-Vargas, Cristina, Guillermo Armaiz-Peña, and Eida Castro-Figueroa. 2021. "A Biopsychosocial Approach to Grief, Depression, and the Role of Emotional Regulation" Behavioral Sciences 11, no. 8: 110. https://doi.org/10.3390/bs11080110
APA StylePeña-Vargas, C., Armaiz-Peña, G., & Castro-Figueroa, E. (2021). A Biopsychosocial Approach to Grief, Depression, and the Role of Emotional Regulation. Behavioral Sciences, 11(8), 110. https://doi.org/10.3390/bs11080110







