Emotional Education in Early Onset Schizophrenia and Asperger’s Syndrome
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Procedures and Tasks
2.3. Procedures
2.4. Methods
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; American Psychiatric Association: Washington, DC, USA, 2014. [Google Scholar]
- Frith, C.D. The Cognitive Neuropsychology of Schizophrenia, 1st ed.; Psychology Press: New York, NY, USA, 1992; p. 184. [Google Scholar]
- Baron-Cohen, S.; Leslie, A.M.; Frith, U. Does the autistic child have a “Theory of Mind”? Cognition 1985, 21, 37–46. [Google Scholar] [CrossRef]
- Baron-Cohen, S. Mindblindness: An Essay on Autism and Theory of Mind; MIT Press: Cambridge, MA, USA, 1995; p. 2412. [Google Scholar]
- Auyeung, B.; Allison, C.; Wheelwright, S.; Baron-Cohen, S. Brief report: Development of the adolescent empathy and systemizing quotients. J. Autism Dev. Disord. 2012, 42, 2225–2235. [Google Scholar] [CrossRef] [PubMed]
- Corcoran, R.; Mercer, G.; Frith, C.D. Schizophrenia, symptomatology and social inference: Investigating ‘theory of mind’ in people with schizophrenia. Schizophr. Res. 1995, 17, 5–13. [Google Scholar] [CrossRef]
- Frajo-Apor, B.; Pardeller, S.; Kemmler, G.; Welte, A.; Hofer, A. Emotional Intelligence deficits in Schizophrenia: The impact of non-social cognition. Schizophr. Res. 2016, 172, 131–136. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Frolli, A.; La Penna, I.; Cavallaro, A.; Ricci, M.C. Theory of Mind: Autism and Typical Development. Acad. J. Ped. Neonatol. 2019, 8, 555799. [Google Scholar] [CrossRef]
- Grossi, D.; Trojano, L. Neuropsicologia dei Lobi Frontali; Il Mulino: Bologna, Italy, 2005; p. 296. [Google Scholar]
- Penn, D.L.; Corrigan, P.W.; Bentall, R.P.; Racenstein, J.M.; Newman, L. Social cognition in schizophrenia. Psychol Bull. 1997, 121, 114–132. [Google Scholar] [CrossRef]
- Pickup, G.J.; Frith, C.D. Theory of mind impairments in schizophrenia: Symptomatology, severity and specificity. Psychol. Med. 2001, 31, 207–220. [Google Scholar] [CrossRef]
- Remschmidt, H.E.; Schulz, E.; Martin, M.; Warnke, A.; Trott, G.E. Childhood-onset schizophrenia: History of the concept and recent studies. Schizophr. Bull. 1994, 20, 727–745. [Google Scholar] [CrossRef]
- Ritsner, M.S. Handbook of Schizofrenia Spectrum Disorders, Therapeutic Approaches, Comorbidity and Outcomes; Springer Science+Business Media: Berlin, Germany, 2001; pp. 195–205. [Google Scholar]
- Fletcher, P.C.; Happ, F.; Frith, U.; Baker, S.C.; Dolan, R.J.; Frackowiak, R.S.J. Other minds in the brain: A functional imaging study of “theory of mind” in story comprehension. Cognition 1995, 57, 109–128. [Google Scholar] [CrossRef]
- Stuss, D.T.; Gallup, G.G., Jr.; Alexander, M.P. The frontal lobes are necessary for ‘theory of mind’. Brain 2001, 124, 279–286. [Google Scholar] [CrossRef]
- Paloscia, C.; Rosa, C.; Fagiolo, D.; Pompili, E.; Sciarretta, A.; Curatolo, P.; Pasini, A. Early-onset schizophrenia: Psychopathology and clinical features. Trad. Schizofrenia ad esordio precoce: Psicopatologia e clinica. Giorn Ital. Psicopat. 2008, 14, 425–447. [Google Scholar]
- Ellis, A. Changing rational-emotive therapy (RET) to rational emotive behavior therapy (REBT). J. Ration. Emot. Cogn. Behav. Ther. 1995, 13, 85–89. [Google Scholar] [CrossRef]
- Di Pietro, M. L’ABC delle Mie Emozioni. Corso di Alfabetizzazione Socio-Affettiva; Manuale per l’alunno; Erickson: Trento, Italy, 1999; p. 128. [Google Scholar]
- Serafin, M.; Surian, L. Il test degli occhi: Uno strumento per valutare la “Teoria Della Mente”. G. Ital. Psicol. 2004, 4, 839–862. [Google Scholar]
- Baron-Cohen, S.; Wheelwright, S. The Empathy Quotient: An Investigation of Adults with Asperger Syndrome or High Functioning Autism, and Normal Sex Differences. J. Autism Dev. Disord. 2004, 34, 163–175. [Google Scholar] [CrossRef]
- Fonagy, P.; Luyten, P.; Moulton-Perkins, A.; Lee, Y.W.; Warren, F.; Howard, S.; Ghinai, R.; Fearon, P.; Lowyck, B. Sviluppo e validazione di una misura self-report di mentalizzazione: Il Questionario sul funzionamento riflessivo. PLoS ONE 2016, 11, e0158678. [Google Scholar] [CrossRef]
- Lord, C.; Rutter, M.; DiLavore, P.C.; Risi, S.; Luyster, R.J.; Gotham, K.; Bishop, S.L.; Guthrie, W. ADOS–Autism Diagnostic Observation Schedule-Second Edition; Hogrefe; Western Psychological Services: Los Angeles, CA, USA, 2012. [Google Scholar]
- Krug, D.A.; Arick, J.R. KADI-Krug Asperger’s Disorder Index; Erikson: Trento, Italy, 2007. [Google Scholar]
- Le Couter, A.; Lord, C.; Rutter, M. Autism Diagnostic Interview-Revised (ADI-R); University of Michigan Autism & Communication Disorders Research Center; Western Psychological Services: Los Angeles, CA, USA, 2007. [Google Scholar]
- Kaufman, J.; Birmaher, B.; Brent, D.; Rao, U.; Ryan, N. K-SADS-PL–Intervista Diagnostica per la Valutazione Dei Disturbi Psicopatologici in Bambini e Adolescenti; Erikson: Trento, Italy, 2014. [Google Scholar]
- Raven, J.C. SPM–Standard Progressive Matrices; Giunti: Florence, Italy, 1997. [Google Scholar]
- Wechsler, D. WAIS–IV: Wechsler Adult Intelligence Scale; Pearson: San Antonio, TX, USA, 2008. [Google Scholar]
- Di Pietro, M. ABC delle Mie Emozioni; Erickson: Trento, Italy, 2014; p. 160. [Google Scholar]
- IBM to Acquire SPSS Inc. to Provide Clients Predictive Analytics Capabilities. Available online: ibm.com (accessed on 8 August 2017).
- Cervi, M.A.; Bonesso, C. Emozioni per Crescere. Come Educare l’Emotività; Armando Editore: Roma, Italy, 2008; p. 368. [Google Scholar]
- First, M.B.; Williams, J.B.W.; Karg, R.S.; Spitzer, R.L. SCID-5-CV, Intervista Clinica Strutturata per i Disturbi del DSM-5–Versione per il Clinico-Starter Kit; Raffaello Cortina: Milano, Italy, 2017; p. 682. [Google Scholar]
- Goleman, D. Intelligenza Emotiva; Rizzoli: Milano, Italy, 1996. [Google Scholar]
- Hurkens, E.; Loth, F.L.; Minderaa, R.B. Theory of mind in children with “lesser variants” of autism: A longitudinal study. J. Child. Psychol. Psychiatry 2002, 43, 885–900. [Google Scholar]
- Marchi, S.; Prior, M.; Sartori, G. Cognizione Sociale e Comportamento; Domenighini Editore: Padua, Italy, 2003. [Google Scholar]
- Cohen, A.S.; Docherty, N.M. Affective reactivity of speech and emotional experience in patients with schizophrenia. Schizophr. Res. 2004, 69, 7–14. [Google Scholar] [CrossRef]
- Crawford, L.; Rowe, D.; Rudkin, A. Cerebral dominance and schizophrenia-spectrum disorders in adults with intellectual disability. J. Intellect Disabil. Res. 2000, 44, 638–643. [Google Scholar]
- Flowers, K.A.; Laws, K.R.; Mortimer, A.M. Cognitive function and social abilities in patient with schizophrenia: Relationship with atypical antipsychotics. Psychiatry Clin. Neurosci. 2006, 60, 473–479. [Google Scholar]
- Adenzato, M.; Endici, I. Comprendere le menti altrui. Meccanismi neuro-cognitivi dell’interazione sociale. Quad. Psicoter. Cogn. 2005, 16, 14–28. [Google Scholar]
- Brune, M. Social cognition and psychopathology in an evolutionary perspective—Current status and proposals for research. Psychopathology 2001, 34, 3485–3494. [Google Scholar] [CrossRef] [PubMed]
- Gabbard, G.O. Psichiatria Psicodinamica; Raffaello Cortina Editore: Milano, Italy, 2008; p. 638. [Google Scholar]
- Seiferth, N.; Pauly, K.; Kellermann, T.; Shah, N.J.; Ott, G.; Herpertz-Dahlmann, B.; Kikcher, T.; Schneider, F.; Habel, U. Neuronal Correlates of Facial Emotion Discrimination in Early Onset Schizophrenia. Neuropsychopharmacology 2009, 34, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Ballerini, A. Autismo e schizofrenia: Una proposta. In Patologia di un Eremitaggio. Uno Studio Sull’Autismo Schizofrenico; Bollati Boringhieri: Torino, Italy, 2002; pp. 136–154. [Google Scholar]
- Ellis, A. Ragione ed Emozione in Psicoterapia; Astrolabio: Roma, Italy, 1989; p. 272. [Google Scholar]
- Leahy, R.L.; Tirch, D.; Napolitano, L.A. La Regolazione delle Emozioni in Psicoterapia. Guida Pratica Per il Professionista; Edizioni Eclipsi: Trento, Italy, 2013; p. 358. [Google Scholar]
- Beck, A.T. A Cognitive Model of Schizophrenia. J. Cogn. Psychother. 2004, 18, 281–288. [Google Scholar] [CrossRef]
- Rocca, P.; Pulvirenti, L.; Giugiario, M.; Bogetto, F. Compliance in the treatment of schizophrenia. Riv. Psichiatr. 2006, 41, 301–306. [Google Scholar]
| ASe | EOSe | ASc | EOSc | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mage | SD | Gerder | Mage | SD | Gerder | Mage | SD | Gerder | Mage | SD | Gerder |
| 16, 49 | 0, 23 | M/F 19/6 | 16, 33 | 0, 46 | M/F 18/7 | 16, 53 | 0, 17 | M/F 19/9 | 16, 33 | 0, 46 | M/F 17/8 |
| Subgroup (N = 25) | Total Sample (N = 100) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RFQ_C | RFQ_U | ET | EQ | |||||||||||||
| Means | SD | F | p | Means | SD | F | p | Means | SD | F | p | Means | SD | F | p | |
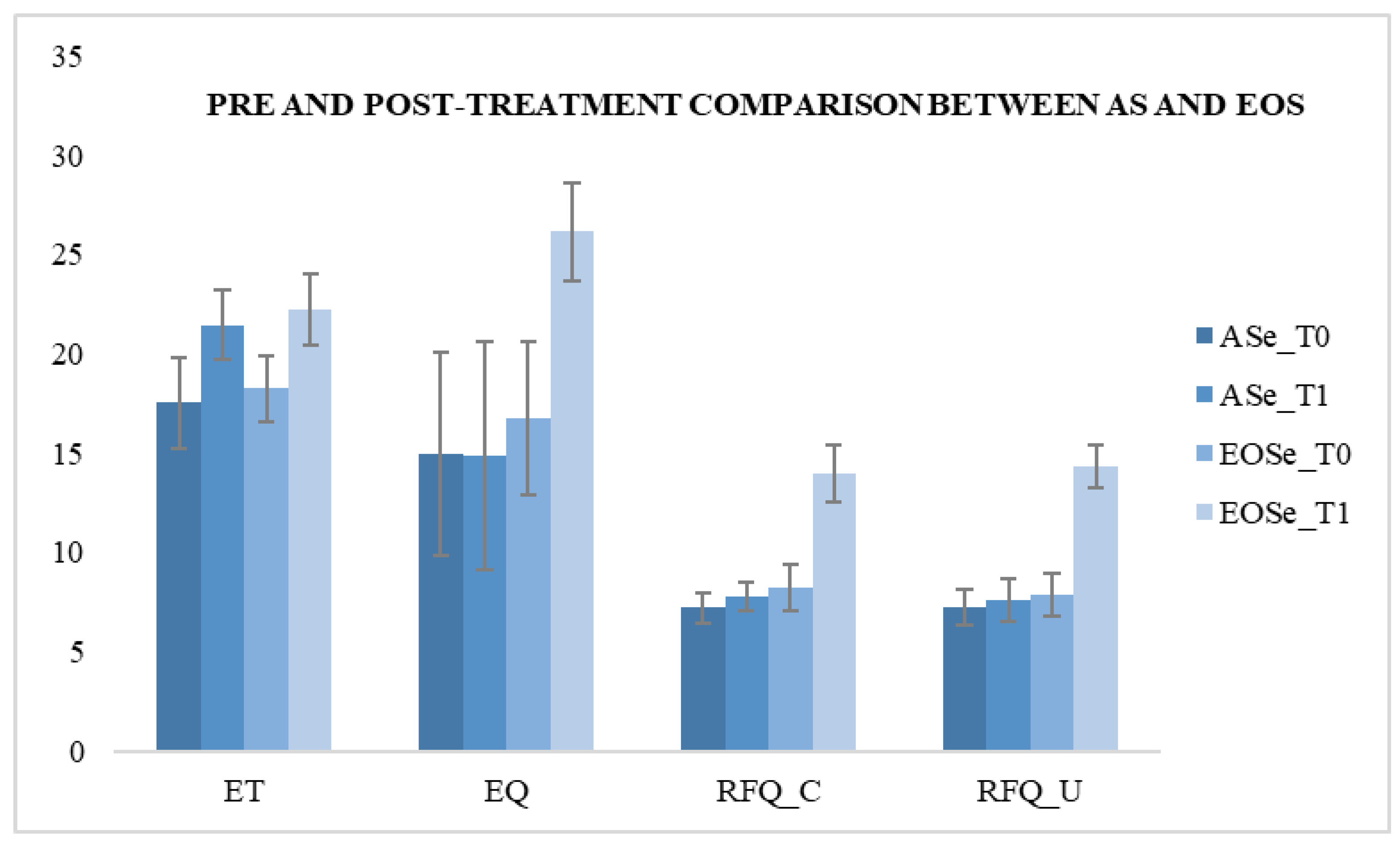
| ASe_T0 | 7.2 | 0.866 | 7.28 | 0.891 | 17.52 | 2.33 | 14.96 | 5.16 | ||||||||
| Asc_T0 | 7.96 | 0.676 | 7.76 | 0.723 | 16.6 | 2.43 | 11.56 | 2.5 | ||||||||
| EOSe_T0 | 8.24 | 1.16 | 2.982 | 0.091 | 7.88 | 1.09 | 1.887 | 0.176 | 18.24 | 1.64 | 1.597 | 0.212 | 16.76 | 3.88 | 1.93 | 0.170 |
| EOSc_T0 | 8.4 | 1.08 | 8.12 | 1.09 | 18.12 | 1.81 | 15.68 | 3.54 | ||||||||
| Subgroup (N = 25) | Total Sample (N = 100) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RFQ_C | RFQ_U | ET | EQ | |||||||||||||
| Means | SD | F | p | Means | SD | F | p | Means | SD | F | p | Means | SD | F | p | |
| ASe_T0 | 7.2 | 0.866 | 7.28 | 0.891 | 17.52 | 2.33 | 14.96 | 5.16 | ||||||||
| ASe_T1 | 7.76 | 0.723 | 6.157 | 0.017 | 7.6 | 1.08 | 1.306 | 0.259 | 21.44 | 1.758 | 45.107 | 0.000 * | 14.88 | 5.71 | 0.003 | 0.959 |
| EOSe_T0 | 8.24 | 1.16 | 7.88 | 1.09 | 18.24 | 1.64 | 16.76 | 3.88 | ||||||||
| EOSe_T1 | 13.96 | 1.42 | 240.813 | 0.000 * | 14.32 | 1.1 | 428.446 | 0.000 * | 22.2 | 1.8 | 66 | 0.000 * | 26.16 | 2.49 | 103.563 | 0.000 * |
| Subgroup (N = 25) | Total Sample (N = 100) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RFQ_C | RFQ_U | ET | EQ | |||||||||||||
| Means | SD | F | p | Means | SD | F | p | Means | SD | F | p | Means | SD | F | p | |
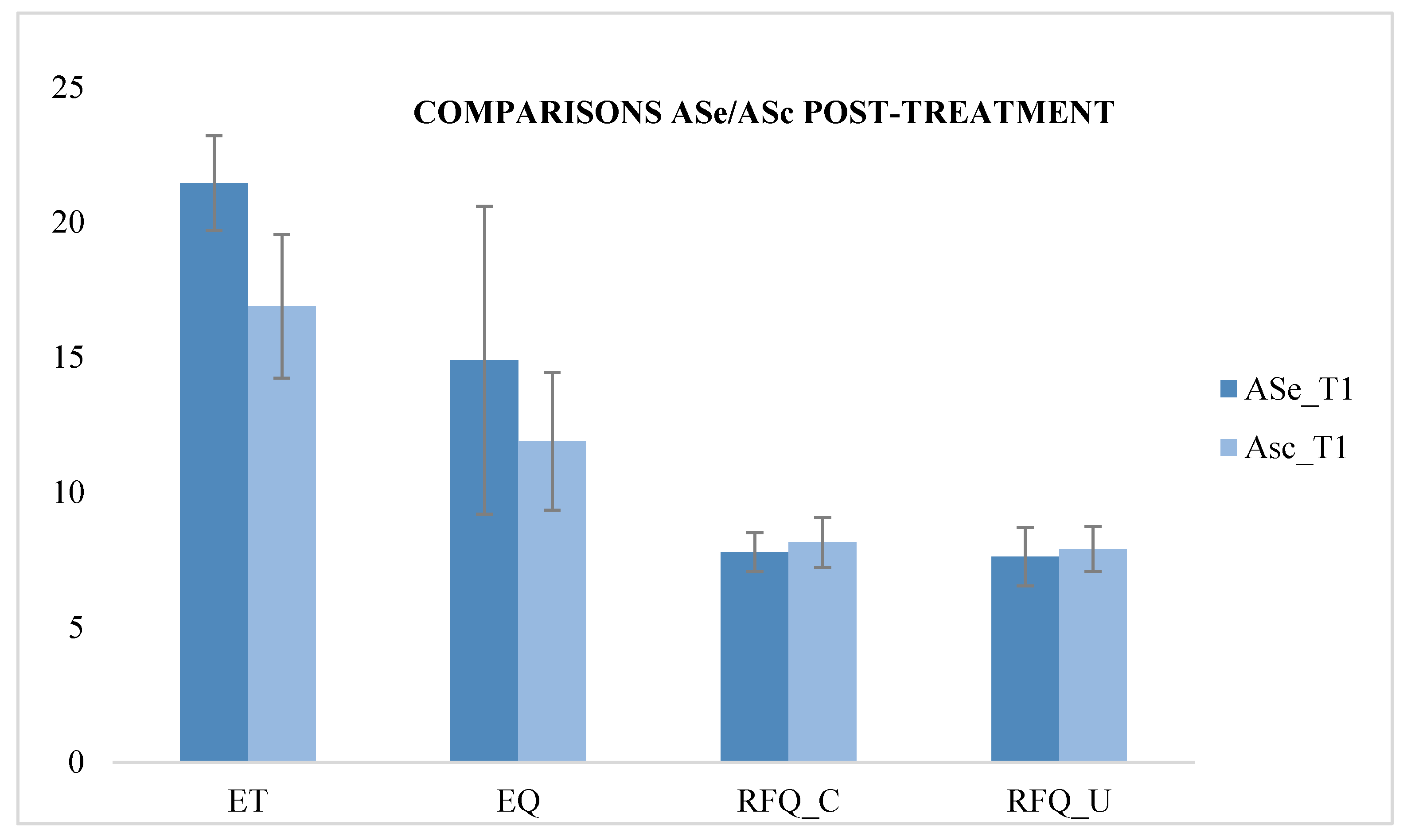
| ASe_T1 | 7.76 | 0.723 | 2.342 | 0.132 | 7.6 | 1.08 | 1.054 | 0.310 | 21.44 | 1.758 | 50.965 | 0.000 * | 14.88 | 5.718 | 5.737 | 0.021 |
| Asc_T1 | 8.12 | 0.927 | 7.88 | 0.833 | 16.88 | 2.666 | 11.88 | 2.555 | ||||||||
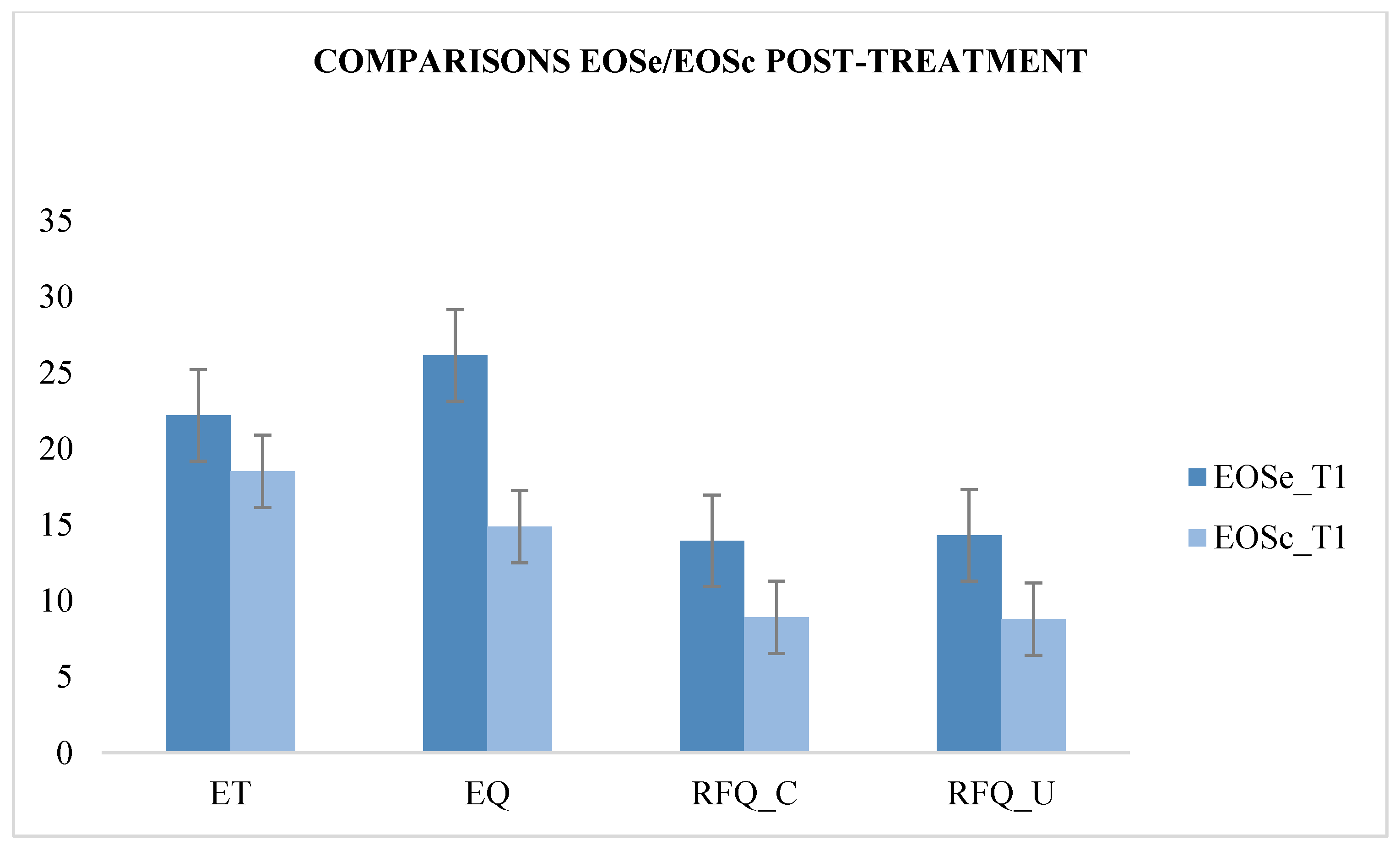
| EOSe_T1 | 13.96 | 1.428 | 179.728 | 0.000 * | 14.32 | 1.108 | 279.374 | 0.000 * | 22.2 | 1.803 | 50.708 | 0.000 * | 26.16 | 2.495 | 147.60 | 0.000 * |
| EOSc_T1 | 8.92 | 1.222 | 8.8 | 1.225 | 18.52 | 1.851 | 16.04 | 3.335 | ||||||||
| Subgroup (N = 25) | Total Sample (N = 100) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RFQ_C | RFQ_U | ET | EQ | |||||||||||||
| Means | SD | F | p | Means | SD | F | p | Means | SD | F | p | Means | SD | F | p | |
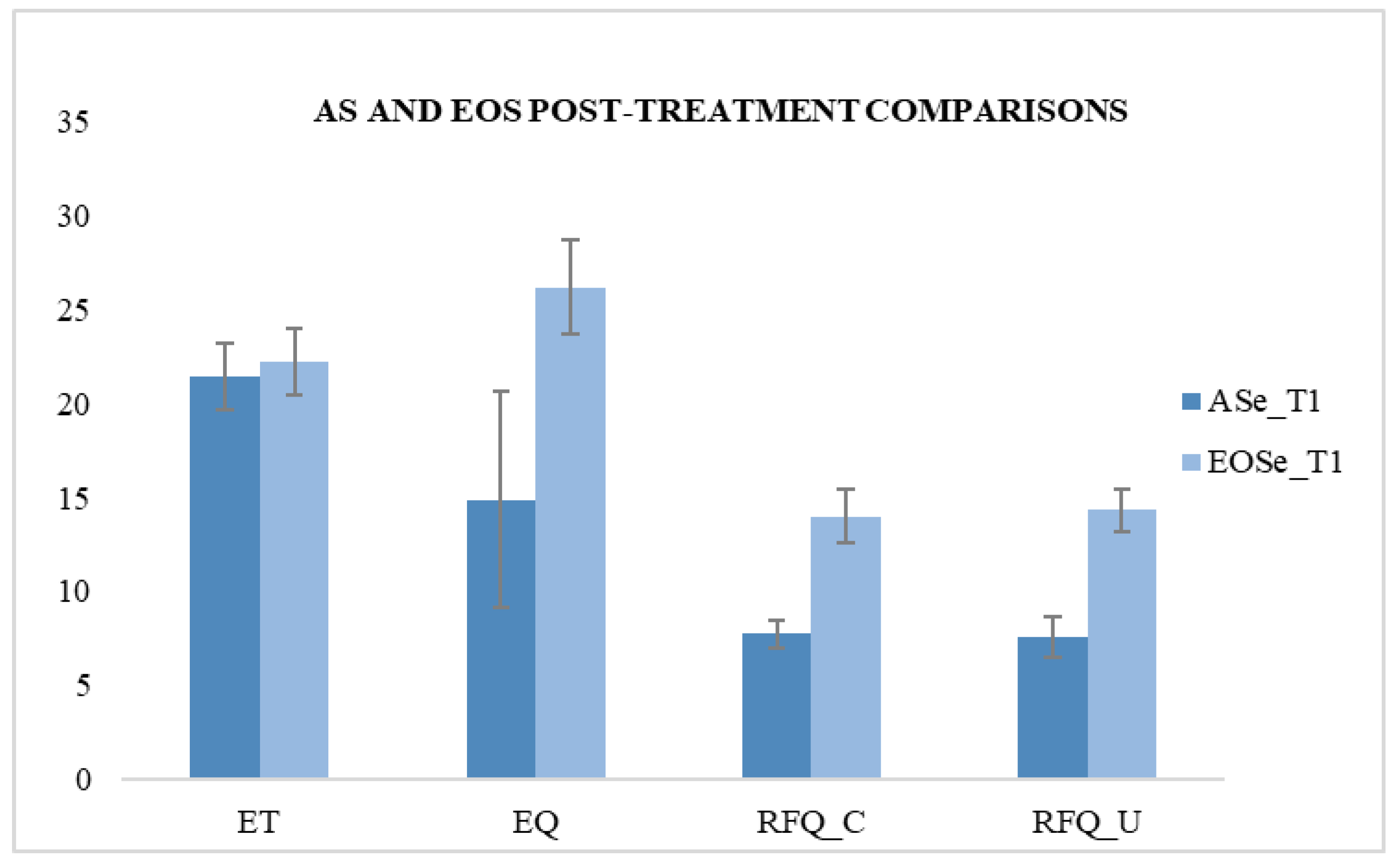
| ASe_T1 | 7.76 | 0.723 | 7.6 | 1.08 | 21.44 | 1.758 | 14.88 | 5.718 | ||||||||
| EOSe_T1 | 13.96 | 1.428 | 374.902 | 0.000 * | 14.32 | 1.108 | 471.71 | 0.000 * | 22.2 | 1.803 | 2.278 | 0.138 | 26.16 | 2.495 | 81.738 | 0.000 * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frolli, A.; Ricci, M.C.; Tortorelli, F.A.; Cavallaro, A.; Valenzano, L.; Rega, A.; Operto, F.F.; Corrivetti, G. Emotional Education in Early Onset Schizophrenia and Asperger’s Syndrome. Behav. Sci. 2020, 10, 131. https://doi.org/10.3390/bs10090131
Frolli A, Ricci MC, Tortorelli FA, Cavallaro A, Valenzano L, Rega A, Operto FF, Corrivetti G. Emotional Education in Early Onset Schizophrenia and Asperger’s Syndrome. Behavioral Sciences. 2020; 10(9):131. https://doi.org/10.3390/bs10090131
Chicago/Turabian StyleFrolli, Alessandro, Maria Carla Ricci, Francesco Alberto Tortorelli, Antonella Cavallaro, Luana Valenzano, Angelo Rega, Francesca Felicia Operto, and Giulio Corrivetti. 2020. "Emotional Education in Early Onset Schizophrenia and Asperger’s Syndrome" Behavioral Sciences 10, no. 9: 131. https://doi.org/10.3390/bs10090131
APA StyleFrolli, A., Ricci, M. C., Tortorelli, F. A., Cavallaro, A., Valenzano, L., Rega, A., Operto, F. F., & Corrivetti, G. (2020). Emotional Education in Early Onset Schizophrenia and Asperger’s Syndrome. Behavioral Sciences, 10(9), 131. https://doi.org/10.3390/bs10090131







