CDK4/6 Inhibitors and Arthralgia: A Single Institution Experience
Abstract
1. Introduction
2. Materials and Methods
Statistics
3. Results
3.1. Patients Characteristics
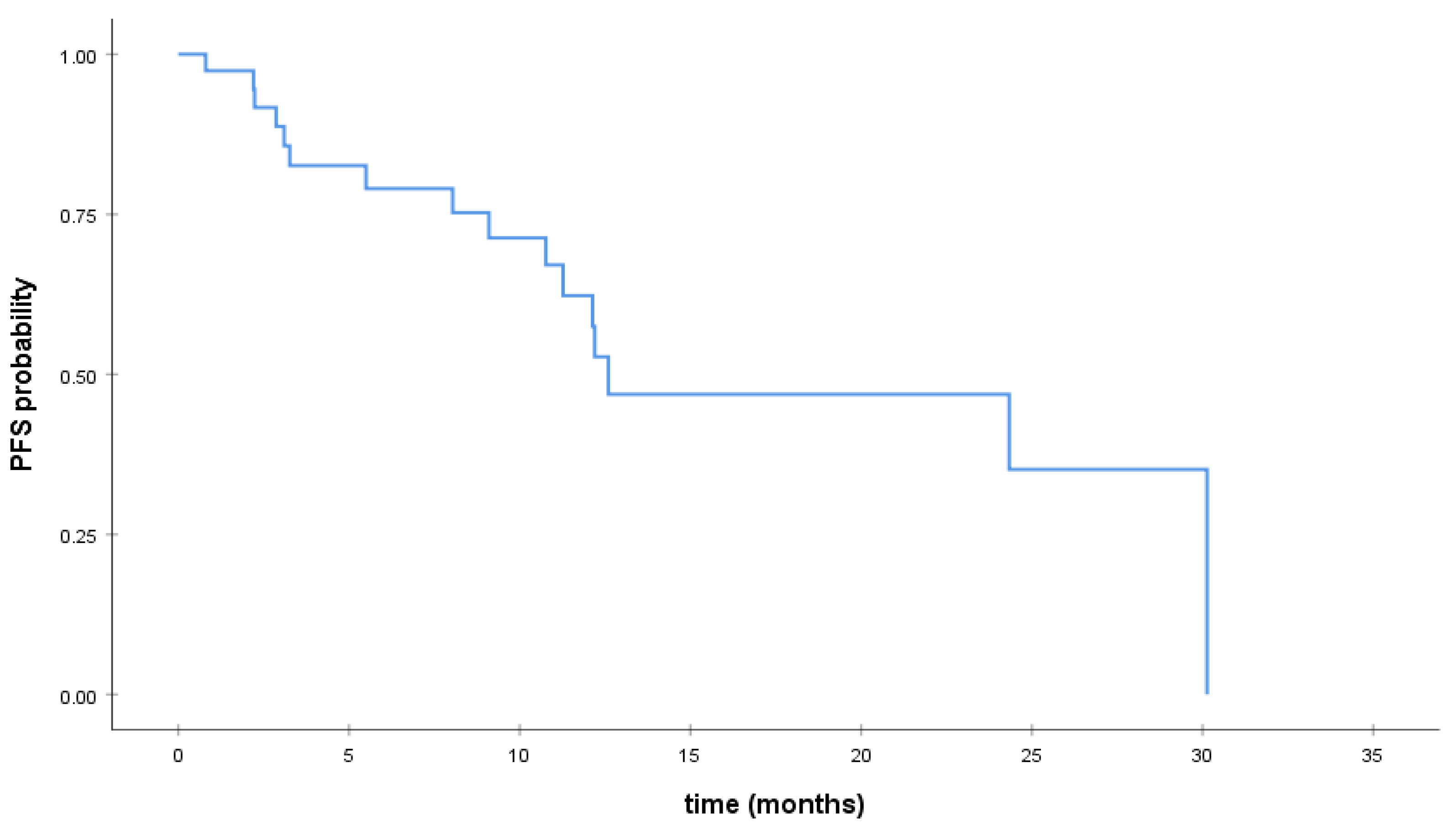
3.2. Survival Analysis
3.3. Association of CDK4/6 Inhibitors with Arthralgia
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Clinical Trial | Trial Number | Treatment Arms | Sample Size (Size per Arm) | Median Age | Median Follow-Up | PFS | OS | Arthralgia (%) |
|---|---|---|---|---|---|---|---|---|
| PALOMA-1 | NCT00721409 | palbociclib plus letrozole vs. letrozole plus placebo | 165 (84/81) | 63/64 | 64.7 | 20.2/10.2 (HR:0.488; p = 0.0004) | 37.5/34.5 (HR: 0.897; p = 0.281) | 27.7/18.2 |
| PALOMA-2 | NCT01740427 | palbociclib plus letrozole vs. letrozole plus placebo | 666 (444/222) | 62/61 | 23 | 24.8/14.5 (H: 0.58; p < 0.001) | 42.1/34.7 (p = 0.06) | 33.3/33.8 |
| PALOMA-3 | NCT01942135 | palbociclib plus fulvestrant vs. fulvestrant plus placebo | 517 (345/172) | 57/56 | 44.8 | 9.2/3.8 (HR: 0.422; p < 0.0001) | 34.9/28 (HR: 0.81; p = 0.09) | 13/16.3 |
| MONALEESA-2 | NCT01958021 | ribociclib plus letrozole vs. placebo plus letrozole | 668 (334/334) | 62/63 | 26.4 | 25.3/16 (HR: 0.568) | 42.5/28.7; H: 0.746 | 27.5/28.8 |
| MONALEESA-3 | NCT02422615 | ribociclib plus fulvestrant vs. placebo plus fulvestrant | 726 (484/242) | 63.4/62.8 | 39.4 | 20.5/12.8 (HR: 0.593; p < 0.001) | 57.8%/45.9% at 42 months | 23.8/26.6 |
| MONALEESA-7 | NCT02278120 | ribociclib plus tamoxifen/NSAI plus goserelin vs. placebo plus tamoxifen/NSAI plus goserelin | 672 (335/337) | 42.6/43.7 | 19.2 | 23.8 vs. 13 (H: 0·55, p < 0·0001) | NR | 29.85/27.3 |
| MONARCHE-2 | NCT02107703 | abemaciclib plus fulvestrant vs. placebo plus fulvestrant | 669 (446/223) | 59.3/61.1 | 19.5 | 16.4/9.3 (HR: 0.553; p < 0.001) | NR | 11.6/14.3 |
| MONARCHE-3 | CT02246621 | abemaciclib plus anastrozole or letrozole vs. placebo plus anastrozole or letrozole | 493 (328/165) | 63/63 | 26.8 | 28.2 vs. 14.7; (H: 0.54; p < 0.001) | NR | 17.43/20.5 |
| MONARCHE PLUS | NCT02763566 | abemaciclib plus NSAI vs. placebo plus NSAI or abemaciclib plus fulvestrant vs. placebo plus fulvestrant | 463 (207/99/104/53) | 56/59/55/58) | 26 | Not reached vs. 14.7; H: 0.49 | 56/30 (p < 0.0001) | 5.8/13.1 6.7/5.6 |
References
- Crew, K.D.; Greenlee, H.; Capodice, J.; Raptis, G.; Brafman, L.; Fuentes, D.; Sierra, A.; Hershman, D.L. Prevalence of joint symptoms in postmenopausal women taking aromatase inhibitors for early-stage breast cancer. J. Clin. Oncol. 2007, 25, 3877–3883. [Google Scholar] [CrossRef]
- Henry, N.L.; Giles, J.T.; Stearns, V. Aromatase inhibitor-associated musculoskeletal symptoms: Etiology and strategies for management. Oncology 2008, 22, 1401–1408. [Google Scholar]
- Henry, N.L.; Azzouz, F.; Desta, Z.; Li, L.; Nguyen, A.T.; Lemler, S.; Hayden, J.; Tarpinian, K.; Yakim, E.; Flockhart, D.A.; et al. Predictors of aromatase inhibitor discontinuation as a result of treatment-emergent symptoms in early-stage breast cancer. J. Clin. Oncol. 2012, 30, 936–942. [Google Scholar] [CrossRef]
- Coleman, R.E.; Bolten, W.W.; Lansdown, M.; Dale, S.; Jackisch, C.; Merkel, D.; Maass, N.; Hadji, P. Aromatase inhibitor-induced arthralgia: Clinical experience and treatment recommendations. Cancer Treat. Rev. 2008, 34, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Buzdar, A.U. Clinical features of joint symptoms observed in the ‘Arimidex’, Tamoxifen, Alone or in Combination (ATAC) trial. J. Clin. Oncol. 2006, 24, 551. [Google Scholar] [CrossRef]
- Henry, N.L.; Giles, J.T.; Ang, D.; Mohan, M.; Dadabhoy, D.; Robarge, J.; Hayden, J.; Lemler, S.; Shahverdi, K.; Powers, P.; et al. Prospective characterization of musculoskeletal symptoms in early stage breast cancer patients treated with aromatase inhibitors. Breast Cancer Res. Treat. 2008, 111, 365–372. [Google Scholar] [CrossRef]
- FDA. Palbociclib (IBRANCE). Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/palbociclib-ibrance (accessed on 24 December 2020).
- FDA. Ribociclib (Kisqali). Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/ribociclib-kisqali (accessed on 24 December 2020).
- Lee, K.A.; Shepherd, S.T.C.; Johnston, S.R.D. Abemaciclib, a potent cyclin-dependent kinase 4 and 6 inhibitor, for treatment of ER-positive metastatic breast cancer. Future Oncol. 2019, 15, 3309–3326. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.G.; Dickler, M.N. The Role of CDK4/6 Inhibition in Breast Cancer. Oncologist 2015, 20, 483–490. [Google Scholar] [CrossRef]
- de Melo Gagliato, D.; CBuzaid, A.; Perez-Garcia, J.M.; Llombart, A.; Cortes, J. Cdk4/6 inhibitors in hormone receptor-positive metastatic breast cancer: Current practice and knowledge. Cancers 2020, 12, 2480. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Wu, J.; Wang, C.; Zhuang, S.; Chen, J.; Ye, F. Combination cyclin-dependent kinase 4/6 inhibitors and endocrine therapy versus endocrine monotherapy for hormonal receptor-positive, human epidermal growth factor receptor 2-negative advanced breast cancer: A systematic review and meta-analysis. PLoS ONE 2020, 15, e0233571. [Google Scholar] [CrossRef]
- Onesti, C.E.; Jerusalem, G. CDK4/6 inhibitors in breast cancer: Differences in toxicity profiles and impact on agent choice. A systematic review and meta-analysis. Expert Rev. Anticancer Ther. 2020, 21, 283–298. [Google Scholar] [CrossRef]
- Diéras, V.; Rugo, H.S.; Schnell, P.; Gelmon, K.; Cristofanilli, M.; Loi, S.; Colleoni, M.; Lu, D.R.; Mori, A.; Gauthier, E.; et al. Long-term pooled safety analysis of palbociclib in combination with endocrine therapy for HR1/HER2- Advanced breast cancer. J. Natl. Cancer Inst. 2019, 111. [Google Scholar] [CrossRef]
- Hortobagyi, G.N.; Stemmer, S.M.; Burris, H.A.; Yap, Y.-S.; Sonke, G.S.; Paluch-Shimon, S.; Campone, M.; Blackwell, K.L.; André, F.; Winer, E.P.; et al. Ribociclib as First-Line Therapy for HR-Positive, Advanced Breast Cancer. N. Engl. J. Med. 2016, 375, 1738–1748. [Google Scholar] [CrossRef]
- Slamon, D.J.; Neven, P.; Chia, S.; Fasching, P.A.; De Laurentiis, M.; Im, S.A.; Petrakova, K.; Val Bianchi, G.; Esteva, F.J.; Martín, M.; et al. Phase III randomized study of ribociclib and fulvestrant in hormone receptor-positive, human epidermal growth factor receptor 2-negative advanced breast cancer: MONALEESA-3. J. Clin. Oncol. 2018, 36, 2465–2472. [Google Scholar] [CrossRef] [PubMed]
- Sledge, G.W.; Toi, M.; Neven, P.; Sohn, J.; Inoue, K.; Pivot, X.; Burdaeva, O.; Okera, M.; Masuda, N.; Kaufman, P.A.; et al. MONARCH 2: Abemaciclib in combination with fulvestrant in women with HR+/HER2-advanced breast cancer who had progressed while receiving endocrine therapy. J. Clin. Oncol. 2017, 35, 2875–2884. [Google Scholar] [CrossRef] [PubMed]
- Goetz, M.P.; Toi, M.; Campone, M.; Trédan, O.; Bourayou, N.; Sohn, J.; Park, I.H.; Paluch-Shimon, S.; Huober, J.; Chen, S.C.; et al. MONARCH 3: Abemaciclib as initial therapy for advanced breast cancer. J. Clin. Oncol. 2017, 35, 3638–3646. [Google Scholar] [CrossRef]
- ClinicalTrials.gov. A Study of Nonsteroidal Aromatase Inhibitors Plus Abemaciclib (LY2835219) in Postmenopausal Women with Breast Cancer-Study Results. Available online: https://clinicaltrials.gov/ct2/show/results/NCT02246621?term=NCT02246621&draw=2 (accessed on 14 November 2020).
- ClinicalTrials.gov. A Study of Abemaciclib (LY2835219) in Participants with Breast Cancer-Study Results. Available online: https://clinicaltrials.gov/ct2/show/NCT02763566?term=MONARCH+PLUS&draw=2&rank=1 (accessed on 24 December 2020).
- Scharf, O.; Colevas, A.D. Adverse event reporting in publications compared with sponsor database for cancer clinical trials. J. Clin. Oncol. 2006, 24, 3933–3938. [Google Scholar] [CrossRef] [PubMed]
- Geisler, J.; Haynes, B.; Anker, G.; Dowsett, M.; Lønning, P.E. Influence of Letrozole and Anastrozole on Total Body Aromatization and Plasma Estrogen Levels in Postmenopausal Breast Cancer Patients Evaluated in a Randomized, Cross-Over Study. J. Clin. Oncol. 2002, 20, 751–757. [Google Scholar] [CrossRef]
- Ushiyama, T.; Ueyama, H.; Inoue, K.; Ohkubo, I.; Hukuda, S. Expression of genes for estrogen receptors α and β in human articular chondrocytes. Osteoarthr. Cartil. 1999, 7, 560–566. [Google Scholar] [CrossRef]
- Nevitt, M.C.; Cummings, S.R.; Lane, N.E.; Hochberg, M.C.; Scott, J.C.; Pressman, A.R.; Genant, H.K.; Cauley, J.A. Association of estrogen replacement therapy with the risk of osteoarthritis of the hip in elderly white women. Study of Osteoporotic Fractures Research Group. Arch. Intern. Med. 1996, 156, 2073–2080. [Google Scholar] [CrossRef]
- Spector, T.D.; Nandra, D.; Hart, D.J.; Doyle, D.V. Is hormone replacement therapy protective for hand and knee osteoarthritis in women?: The Chingford Study. Ann. Rheum. Dis. 1997, 56, 432–434. [Google Scholar] [CrossRef]
- McCloskey, E. Effects of third-generation aromatase inhibitors on bone. Eur. J. Cancer 2006, 42, 1044–1051. [Google Scholar] [CrossRef]
- Laroche, M.; Seniow, M.; Roche, H.; Ruyssen-Witrand, A. Arthralgia associated with autoimmune abnormalities under aromatase inhibitor therapy: Outcome after cessation of treatment. J. Rheumatol. 2016, 43, 1945–1946. [Google Scholar] [CrossRef] [PubMed]
- Zarkavelis, G.; Kollas, A.; Kampletsas, E.; Vasiliou, V.; Kaltsonoudis, E.; Drosos, A.; Khaled, H.; Pavlidis, N. Aromatase inhibitors induced autoimmune disorders in patients with breast cancer: A review. J. Adv. Res. 2016, 7, 719–726. [Google Scholar] [CrossRef]
- Morel, B.; Marotte, H.; Miossec, P. Will steroidal aromatase inhibitors induce rheumatoid arthritis? Ann. Rheum. Dis. 2007, 66, 557–558. [Google Scholar] [CrossRef]
- Burstein, H.J. Aromatase inhibitor-associated arthralgia syndrome. Breast 2007, 16, 223–234. [Google Scholar] [CrossRef]
- Shi, Q.; Giordano, S.H.; Lu, H.; Saleeba, A.K.; Malveaux, D.; Cleeland, C.S. Anastrozole-associated joint pain and other symptoms in patients with breast cancer. J. Pain 2013, 14, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Schaal, C.; Pillai, S.; Chellappan, S.P. The Rb-E2F transcriptional regulatory pathway in tumor angiogenesis and metastasis. In Advances in Cancer Research; Academic Press Inc.: Cambridge, MA, USA, 2014; Volume 121, pp. 147–182. [Google Scholar]
- Chen, H.Z.; Tsai, S.Y.; Leone, G. Emerging roles of E2Fs in cancer: An exit from cell cycle control. Nat. Rev. Cancer 2009, 9, 785–797. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Wang, L.; Pan, J.H.; Han, J. A critical role of E2F transcription factor 2 in proinflammatory cytokines-dependent proliferation and invasiveness of fibroblast-like synoviocytes in rheumatoid Arthritis. Sci. Rep. 2018, 8, 2623. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wang, L.; Wu, C.; Sun, S.; Pan, J.H. E2F2 directly regulates the STAT1 and PI3K/AKT/NF-κB pathways to exacerbate the inflammatory phenotype in rheumatoid arthritis synovial fibroblasts and mouse embryonic fibroblasts. Arthritis Res. Ther. 2018, 20, 225. [Google Scholar] [CrossRef]
- Handschick, K.; Beuerlein, K.; Jurida, L.; Bartkuhn, M.; Müller, H.; Soelch, J.; Weber, A.; Dittrich-Breiholz, O.; Schneider, H.; Scharfe, M.; et al. Cyclin-Dependent Kinase 6 Is a Chromatin-Bound Cofactor for NF-κB-Dependent Gene Expression. Mol. Cell 2014, 53, 193–208. [Google Scholar] [CrossRef] [PubMed]
- Buss, H.; Handschick, K.; Jurrmann, N.; Pekkonen, P.; Beuerlein, K.; Müller, H.; Wait, R.; Saklatvala, J.; Ojala, P.M.; Schmitz, M.L.; et al. Cyclin-Dependent Kinase 6 Phosphorylates NF-κB P65 at Serine 536 and Contributes to the Regulation of Inflammatory Gene Expression. PLoS ONE 2012, 7, e51847. [Google Scholar] [CrossRef]
- Hosoya, T.; Iwai, H.; Yamaguchi, Y.; Kawahata, K.; Miyasaka, N.; Kohsaka, H. Cell cycle regulation therapy combined with cytokine blockade enhances antiarthritic effects without increasing immune suppression. Ann. Rheum. Dis. 2016, 75, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Shimamura, T.; Shibata, J.; Kurihara, H.; Mita, T.; Otsuki, S.; Sagara, T.; Hirai, H.; Iwasawa, Y. Identification of potent 5-pyrimidinyl-2-aminothiazole CDK4, 6 inhibitors with significant selectivity over CDK1, 2, 5, 7, and 9. Bioorganic Med. Chem. Lett. 2006, 16, 3751–3754. [Google Scholar] [CrossRef]
- Sekine, C.; Sugihara, T.; Miyake, S.; Hirai, H.; Yoshida, M.; Miyasaka, N.; Kohsaka, H. Successful Treatment of Animal Models of Rheumatoid Arthritis with Small-Molecule Cyclin-Dependent Kinase Inhibitors. J. Immunol. 2008, 180, 1954–1961. [Google Scholar] [CrossRef] [PubMed]
| Overall Toxicity N (%) | Yes | 29 (61.7) |
|---|---|---|
| No | 15 (31.9) | |
| Not reported | 3 (6.4) | |
| Hematological toxicity N (%) | Yes | 24 (51.1) |
| No | 20 (42.6) | |
| Not reported | 3 (6.4) | |
| Type of hematological toxicity N (%) | Neutropenia | 22 (86.8) |
| Anemia | 2 (4.3) | |
| Neutropenia/thrombocytopenia | 1 (2.1) | |
| Neutropenia/leukopenia | 1 (2.1) | |
| Arthralgia N (%) | Yes | 3 (6.4) |
| No | 42 (89.4) | |
| Not reported | 2 (4.3) |
| Patient Characteristics | All Patients (47) | |
|---|---|---|
| Age, median, range | 58 | (29–81) |
| Menopausal status, N (%) | Perimenopausal | 6 (12.8) |
| Premenopausal | 10 (21.3) | |
| Postmenopausal | 31 (66) | |
| Stage at initial diagnosis, N (%) | Non-metastatic | 25 (53.2) |
| Metastatic | 22 (46.8) | |
| Histology, N (%) | IDC | 39 (83) |
| ILC | 5 (10.6) | |
| Mucinous | 1 (2.1) | |
| IDC/ILC | 1 (2.1) | |
| Not reported | 1 (2.1) | |
| ER status, N (%) | Positive | 45 (95.7) |
| Negative | 2 (4.3) | |
| PR status, N (%) | Positive | 43 (91.5) |
| Negative | 4 (8.5) | |
| Grade, N (%) | I | 2 (4.3) |
| II | 24 (51.1) | |
| III | 18 (38.3) | |
| Not reported | 3 (6.4) | |
| Adjuvant hormonotherapy, N (%) | Yes | 20 (80) |
| No | 4 (16) | |
| Not reported | 1 (4) | |
| Adjuvant chemotherapy, N (%) | Yes | 23 (92) |
| No | 1 (4) | |
| Not reported | 1 (4) | |
| Adjuvant radiation therapy, N (%) | Yes | 18 (72) |
| No | 6 (24) | |
| Not reported | 1 (4) | |
| Disease progression, N (%) | 20 (42.6) | |
| CDK4/6 inhibitor, N (%) | Abemaciclib | 6 (12.7) |
| Palbociclib | 30 (63.8) | |
| Ribociclib | 11 (23.4) | |
| Concomitant treatment, N (%) | Fulvestrant | 16 (34) |
| Letrozole | 28 (59.6) | |
| Tamoxifen | 3 (6.4) | |
| Treatment duration, median, range | 8.76 | 0.47–30.13 |
| Dose reduction, N (%) | Yes | 12 (25.5) |
| No | 33 (70.2) | |
| Not reported | 2 (4.3) | |
| Dose interruption, N (%) | Yes | 24 (51.1) |
| No | 20 (42.6) | |
| Not reported | 3 (6.4) | |
| Treatment discontinuation, N (%) | Yes | 18 (38.3) |
| No | 27 (57.4) | |
| Not reported | 2 (4.3) | |
| Reason of discontinuation, N (%) | Hematological toxicity | 2 (4.3) |
| Hyperkalemia | 1 (2.1) | |
| PD | 16 (34) | |
| Disease progression on CDK4/6 inhibitor, N (%) | 16 (34) | |
| Progression-free survival (PFS), median, range | 24.3 | 1.71–46.96 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andrikopoulou, A.; Fiste, O.; Apostolidou, K.; Skafida, E.; Markellos, C.; Liontos, M.; Kyriazoglou, A.; Dimopoulos, M.-A.; Zagouri, F. CDK4/6 Inhibitors and Arthralgia: A Single Institution Experience. Med. Sci. 2021, 9, 42. https://doi.org/10.3390/medsci9020042
Andrikopoulou A, Fiste O, Apostolidou K, Skafida E, Markellos C, Liontos M, Kyriazoglou A, Dimopoulos M-A, Zagouri F. CDK4/6 Inhibitors and Arthralgia: A Single Institution Experience. Medical Sciences. 2021; 9(2):42. https://doi.org/10.3390/medsci9020042
Chicago/Turabian StyleAndrikopoulou, Angeliki, Oraianthi Fiste, Kleoniki Apostolidou, Efthymia Skafida, Christos Markellos, Michalis Liontos, Anastasios Kyriazoglou, Meletios-Athanasios Dimopoulos, and Flora Zagouri. 2021. "CDK4/6 Inhibitors and Arthralgia: A Single Institution Experience" Medical Sciences 9, no. 2: 42. https://doi.org/10.3390/medsci9020042
APA StyleAndrikopoulou, A., Fiste, O., Apostolidou, K., Skafida, E., Markellos, C., Liontos, M., Kyriazoglou, A., Dimopoulos, M.-A., & Zagouri, F. (2021). CDK4/6 Inhibitors and Arthralgia: A Single Institution Experience. Medical Sciences, 9(2), 42. https://doi.org/10.3390/medsci9020042







