Creatine Kinase and Blood Pressure: A Systematic Review
Abstract
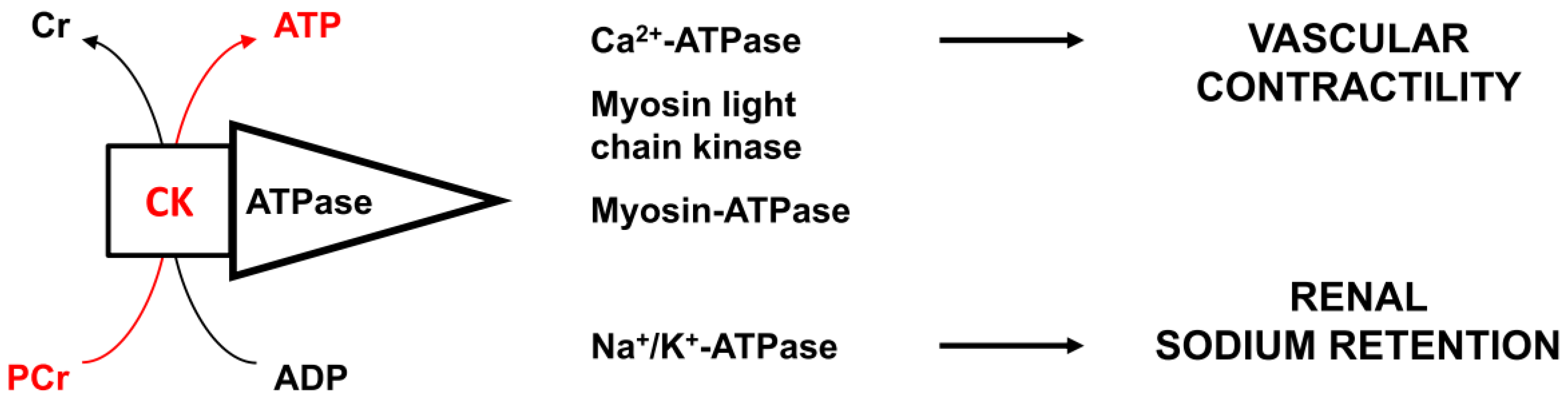
:1. Introduction
2. Methods
3. Results
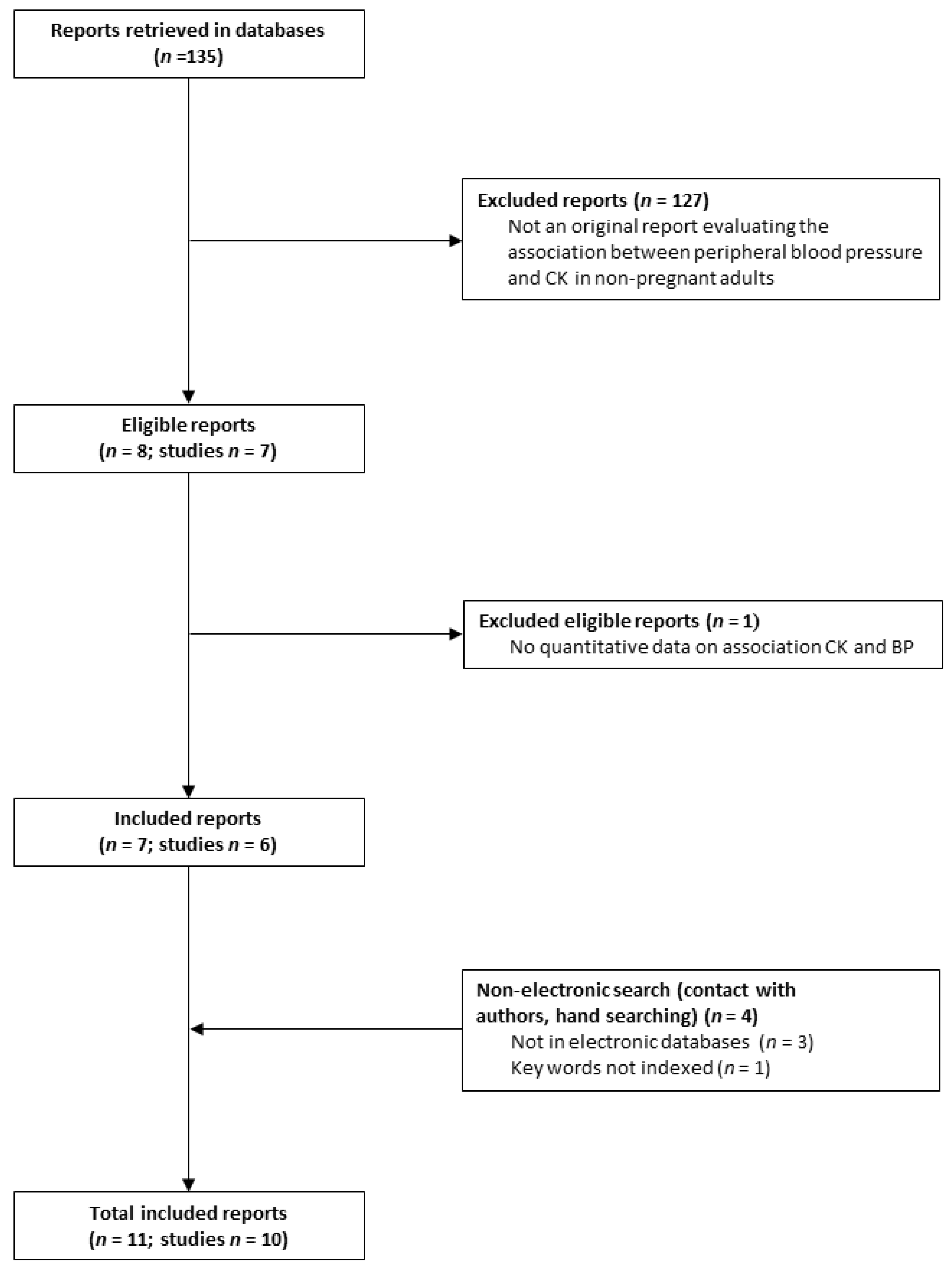
3.1. Systematic Search Yield
3.2. Studies and Participants
3.3. Exposure
3.4. Comparisons and Outcomes
3.5. Subgroup Analysis
4. Discussion
4.1. Strengths and Limitations of This Study
Author Contributions
Funding
Conflicts of Interest
References
- WHO. Raised blood pressure. Available online: https://www.who.int/gho/ncd/risk_factors/blood_pressure_prevalence_text/en/ (accessed on 11 February 2019).
- NCD Risk Factor Collaboration. Worldwide trends in blood pressure from 1975 to 2015: A pooled analysis of 1479 population-based measurement studies with 19·1 million participants. Lancet 2017, 389, 37–55. [Google Scholar] [CrossRef]
- Lim, S.; Vos, A.; Flaxman, A.; Danaei, G.; Shibuya, K.; Adair-Rohani, H.; AlMazroa, M.A.; Amann, M.; Anderson, H.R.; Andrews, K.G. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2224–2260. [Google Scholar] [CrossRef]
- NCD Risk Factor Collaboration. Contributions of mean and shape of blood pressure distribution to worldwide trends and variations in raised blood pressure: A pooled analysis of 1018 population-based measurement studies with 88.6 million participants. Int. J. Epidemiol. 2018, 47, 872–883i. [Google Scholar] [CrossRef]
- Brewster, L.M.; Clark, J.F.; van Montfrans, G.A. Is greater tissue activity of creatine kinase the genetic factor increasing hypertension risk in black people of sub-Saharan African descent? J. Hypertens. 2000, 18, 1537–1544. [Google Scholar] [CrossRef] [PubMed]
- Brewster, L.M.; Mairuhu, G.; Bindraban, N.R.; Koopmans, R.P.; Clark, J.F.; van Montfrans, G.A. Creatine kinase activity is associated with blood pressure. Circulation 2006, 114, 2034–2039. [Google Scholar] [CrossRef]
- Oudman, I.; Kewalbansing, P.; Valkengoed, I.; Zwinderman, A.; Clark, J.F.; van Montfrans, G.A.; Brewster, L.M. Creatine kinase is associated with failure of hypertension treatment. J. Hypertens. 2013, 31, 1025–1031. [Google Scholar] [CrossRef]
- Munn, Z.; Moola, S.; Lisy, K.; Riitano, D.; Tufanaru, C. Chapter 5: Systematic reviews of prevalence and incidence. In Joanna Briggs Institute Reviewer’s Manual; Aromataris, E., Munn, Z., Eds.; The Joanna Briggs Institute: City, Countyry, 2017; Available online: https://reviewersmanual.joannabriggs.org/ (accessed on 11 February 2019).
- Rodgers, M.; Arai, L.; Popay, J.; Britten, N.; Roberts, H.; Petticrew, M.; Sowden, A. Testing methodological guidance on the conduct of narrative synthesis in systematic reviews: Effectiveness of interventions to promote smoke alarm ownership and function. Evaluation 2009, 15, 49–73. [Google Scholar] [CrossRef]
- Brewster, L.M.; van Montfrans, G.A. Distribution of creatine kinase in the general population: Implications for statin therapy. Am. Heart J. 2007, 154, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Johnsen, S.H.; Lilleng, H.; Wilsgaard, T.; Bekkelund, S.I. Creatine kinase activity and blood pressure in a normal population: The Tromsø study. J. Hypertens. 2011, 29, 36–42. [Google Scholar] [CrossRef]
- Sanjay Kumar, H.R. A study to determine the association between creatine kinase and hypertension in a study group of age >40 years. Ph.D. Thesis, Rajiv Gandhi University of Health Sciences, Karnataka, Bangalore, 2013. [Google Scholar]
- Luman, A.; Lubus, A.R. Creatine kinase increases in adults with uncontrolled hypertension. Univ. Med. 2014, 33, 36–42. [Google Scholar]
- George, M.D.; McGill, N.K.; Baker, J.F. Creatine kinase in the U.S. population: Impact of demographics, comorbidities, and body composition on the normal range. Medicine 2016, 95, e4344. [Google Scholar] [CrossRef]
- Mels, C.M.; van Zyl, C.; Huisman, H.W. Cardiovascular function is not associated with creatine kinase activity in a black African population: The SABPA study. BMC Cardiovasc, Disord. 2016, 16, 134. [Google Scholar] [CrossRef]
- Emokpae, M.A.; Nwagbara, G.O.N.A. Serum Creatine Kinase-MB Isoenzyme Activity among Subjects with Uncomplicated Essential Hypertension: Any Sex Differences. Med. Sci. 2017, 27, 5. [Google Scholar] [CrossRef]
- Yen, C.H.; Wang, K.T.; Lee, P.Y.; Liu, C.C.; Hsieh, Y.C.; Kuo, J.Y.; Bulwer, B.E.; Hung, C.L.; Chang, S.C.; Shih, S.C.; et al. Gender-differences in the associations between circulating creatine kinase, blood pressure, body mass and non-alcoholic fatty liver disease in asymptomatic Asians. PLoS ONE 2017, 12, e0179898. [Google Scholar] [CrossRef]
- Sukul, S.; Bahinipati, J.; Patra, S.; Ravichandran, K. Serum Creatine Kinase Activity among Hypertensive Patients and its Role as a Predictor for Failure of Antihypertensive Treatment. J. Clin. Diagn. Res. 2018, 11, BC19–BC22. [Google Scholar]
- Brewster, L.M.; van Bree, S.; Reijneveld, J.C.; Notermans, N.C.; Verschuren, W.M.; Clark, J.F.; van Montfrans, G.A.; de Visser, M. Hypertension risk in idiopathic hyperCKemia. J. Neurol. 2008, 255, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Schumann, G.; Bonora, R.; Ceriotti, F.; Clerc-Renaud, P.; Ferrero, C.A.; Férard, G.; Franck, P.F.; Gella, F.J.; Hoelzel, W.; Jørgensen, P.J.; et al. International Federation of Clinical Chemistry and Laboratory Medicine (IFCC). IFCC primary reference procedures for the measurement of catalytic activity concentrations of enzymes at 37 degrees C. Part 2. Reference procedure for the measurement of catalytic concentration of creatine kinase. Clin. Chem. Lab. Med. 2002, 40, 635–642. [Google Scholar]
- Mikolaenko, I.; Benson, E.; Konrad, R.J.; Chaffin, C.; Robinson, C.A.; Hardy, R.W. Evaluation of the Beckman Coulter LX20 Clinical Chemistry Analyzer. Lab. Med. 2000, 31, 387–393. [Google Scholar] [CrossRef]
- Weykamp, C.; Secchiero, S.; Plebani, M.; Thelen, M.; Cobbaert, C.; Thomas, A.; Jassam, N.; Barth, J.H.; Perich, C.; Ricós, C.; et al. Analytical performance of 17 general chemistry analytes across countries and across manufacturers in the INPUtS project of EQA organizers in Italy, the Netherlands, Portugal, United Kingdom and Spain. Clin. Chem. Lab. Med. 2017, 55, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Stepman, H.C.; Tiikkainen, U.; Stöckl, D.; Vesper, H.W.; Edwards, S.H.; Laitinen, H.; Pelanti, J.; Thienpont, L.M. Participating Laboratories. Measurements for 8 common analytes in native sera identify inadequate standardization among 6 routine laboratory assays. Clin. Chem. 2014, 60, 855–863. [Google Scholar] [CrossRef] [PubMed]
- Jackson, C.M.; Esnouf, M.P.; Winzor, D.J.; Duewer, D.L. Defining and measuring biological activity: Applying the principles of metrology. Accred Qual. Assur. 2007, 12, 283–293. [Google Scholar] [CrossRef]
- Brewster, L.M.; Coronel, C.M.; Sluiter, W.; Clark, J.F.; van Montfrans, G.A. Ethnic differences in tissue creatine kinase activity: An observational study. PLoS ONE 2012, 7, e32471. [Google Scholar] [CrossRef] [PubMed]
- Horjus, D.L.; Nieuwland, R.; Boateng, K.B.; Schaap, M.C.; van Montfrans, G.A.; Clark, J.F.; Sturk, A.; Brewster, L.M. Creatine kinase inhibits ADP-induced platelet aggregation. Sci. Rep. 2014, 4, 6551. [Google Scholar] [CrossRef] [PubMed]
- Brewster, L.M. Creatine kinase, energy reserve, and hypertension: From bench to bedside. Ann. Transl. Med. 2018, 6, 292. [Google Scholar] [CrossRef] [PubMed]
- Taherzadeh, Z.; Karamat, F.A.; Ankum, W.M.; Clark, J.F.; van Montfrans, G.A.; van Bavel, E.; Brewster, L.M. The effect of creatine kinase inhibition on contractile properties of human resistance arteries. Am. J. Hypertens. 2015, 29, 170–177. [Google Scholar] [CrossRef]
- Brewster, L.M.; Oudman, I.; Nannan Panday, R.V.; Khoyska, I.; Haan, Y.C.; Karamat, F.A.; Clark, J.F.; van Montfrans, G.A. Creatine kinase and renal sodium excretion in African and European men on a high sodium diet. J. Clin. Hypertens (Greenwich) 2018, 20, 334–341. [Google Scholar] [CrossRef] [PubMed]
- Karamat, F.A.; Oudman, I.; Ris-Stalpers, C.; Afink, G.B.; Keijser, R.; Clark, J.F.; van Montfrans, G.A.; Brewster, L.M. Resistance artery creatine kinase mRNA and blood pressure in humans. Hypertension 2014, 63, 68–73. [Google Scholar] [CrossRef]
- Haan, Y.C.; Oudman, I.; Diemer, F.S.; Karamat, F.A.; van Valkengoed, I.G.; van Montfrans, G.A.; Brewster, L.M. Creatine kinase as a marker of obesity in a multi-ethnic population. Mol. Cell Endocrinol. 2017, 15, 442:24–442:31. [Google Scholar] [CrossRef]
- Sun, G.; Ukkola, O.; Rankinen, T.; Joanisse, D.R.; Bouchard, C. Skeletal muscle characteristics predict body fat gain in response to overfeeding in never-obese young men. Metabolism 2002, 51, 451–456. [Google Scholar] [CrossRef]
- Johnsen, S.H.; Lilleng, H.; Bekkelund, S.I. Creatine kinase as predictor of blood pressure and hypertension. Is it all about body mass index? A follow-up study of 250 patients. J. Clin. Hypertens. 2014, 16, 820–826. [Google Scholar] [CrossRef]
- Osbakken, M.; Douglas, P.S.; Ivanics, T.; Zhang, D.; Van Winkle, T. Creatine kinase kinetics studied by phosphorus-31 nuclear magnetic resonance in a canine model of chronic hypertension-induced cardiac hypertrophy. J. Am. Coll Cardiol. 1992, 19, 223–228. [Google Scholar] [CrossRef]
- Karamat, F.A.; Oudman, I.; Haan, Y.C.; van Kuilenburg, A.B.; Leen, R.; Danser, J.A.; Leijten, F.P.; Ris-Stalpers, C.; van Montfrans, G.A.; Clark, J.F.; et al. Creatine kinase inhibition lowers systemic arterial blood pressure in spontaneously hypertensive rats: A randomized controlled trial. J. Hypertens. 2016, 34, 2418–2426. [Google Scholar] [CrossRef] [PubMed]
| Author, Year | Population | Ancestry | Country | N | Age * | CK Estimations | Outcome | Effect Size ¶ | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Resting † | Device | IFCC | ||||||||
| BLOOD PRESSURE | ||||||||||
| Brewster 2006 [6] | Random population sample | African Asian European | Netherlands | 1444 | 35–60 | Yes | Roche/Hitachi Systems | Yes ║ | CK associated with SBP and DBP | CK T1 (<88) vs. CK T3 (≥145) SBP 122.5 (1.0) vs. 130.6 (0.9) DBP 79.2 (0.6) vs. 84.8 (0.6) |
| Univariable SBP: + 13.9 [9.6 to 18.3]/log CK DBP: + 9.3 [6.8 to 11.9]/log CK | ||||||||||
| Multivariable SBP + 8.0 [3.3 to 12.7]/log CK DBP + 4.7 [1.9 to 7.0]/log CK | ||||||||||
| Johnsen 2011 [11] | Population sample | European | Norway | 12,776 | 30–87 | No ‡ | Modular P, Roche | Yes | CK associated with SBP and DBP | CK T1 vs. CK T3 SBP 134.4 (0.4) vs. 138.2 (0.4) DBP 76.3 (0.2) vs. 79.8 (0.2) |
| Multivariable SBP + 3.3 [1.4 to 5.2]/log CK DBP + 1.3 [0.3 to 2.3]/log CK | ||||||||||
| Mels 2016 [15] | Teachers | African | South Africa | 405 | 45 (0.5) | No | Beckman UniCel DxC800; Konelab 20i | Yes | Only subgroup analysis | CK only associated with BP in women of European ancestry. Adjusted R2 = 0.46; β = 0.17; p = 0.03 |
| Yen 2017 [17] | Population health survey | Asian | Taiwan | 4562 | 49 (0.2) | Yes | Modular P, Roche | Yes | CK associated with SBP and DBP | CK Q1 (<69) vs. CK Q4 (≥128) SBP 118.6 (0.3) vs. 124.2 (0.3) DBP 73.1 (0.2) vs. 76.6 (0.2) |
| Univariable SBP + 6.5 [5.2 to 7.7] CK/10 mmHg DBP + 10.1 [8.0 to 12.1] CK/10 mmHg | ||||||||||
| Multivariable SBP + 1.68 CK/10 mm Hg | ||||||||||
| HYPERTENSION | ||||||||||
| Brewster 2008 [19] | Cases with hyperCKemia vs. population controls | European | Netherlands | 46 (controls 22,612) | 18–67 | Yes | Modular P, Roche | Yes ║ | High CK associated with hypertension | Odds ratio of hypertension § Crude: 3.9 [2.2 to 6.9] Adjusted: 2.0 [1.1 to 3.8] |
| Johnsen 2011 [11] | Population sample | European | Norway | 12776 | 30–87 | No‡ | Modular P, Roche | Yes | CK higher with HT | CK higher in persons using anti-HT drugs vs. no anti-HT drugs (104 vs. 99) |
| Brewster 2013 [7] | Random population sample | African Asia European | Netherlands | 1444 | 35–60 | Yes | Roche/Hitachi Systems | Yes ║ | CK higher in HT vs. NT | Odds ratio of hypertension CK T1 (<88) vs. CK T3 (≥145) HT prevalence: 26.8 vs. 41.2% Odds ratio 1.9 [1.5 to 2.5] |
| CK in HT vs. controls CK 145.9 (7.0) HT vs. 126.8 (2.5) controls | ||||||||||
| George 2016 [14] | Population study | African Asian European | USA | 10,096 | >20 | No | Beckman UniCel DxC800 | Yes | Only subgroup analysis | Odds ratio of HT (CK dichotomized, ULN) ** Men: 1.2 [0.8 to 1.7] Women: 1.4 [1.0 to 2.1] |
| Yen 2017 [17] | Population health survey | Asian | Taiwan | 4562 | 49 (0.2) | Yes | Modular P, Roche | Yes | CK higher in HT vs. NT | CK in HT vs. controls CK +20.7 [15.8 to 25.6] in HT vs. controls |
| Sukul 2018 [18] | Hypertensives vs. controls | Asian | India | 115 | 25–60 | Yes | Roche diagnostics | Yes ║ | CK higher in HT vs. NT | CK in HT vs. controls CK 199.6 (16.4) HT vs. 72.7 (4.0) controls |
| Sanjay Kumar 2013 [12] | Hypertensives vs. controls | Asian | India | 150 | 40–90 | No | NR | NR | CK MB higher in HT vs. NT | CK MB in HT vs. controls 21.5 (4.0) HT vs. 17.2 (2.4) controls |
| Emokpae 2017 [16] | Hypertensives vs. controls | African | Nigeria | 340 | 28–62 | No | Selectra Pro S | Yes | CK MB higher in HT vs. NT | CK MB in HT vs. controls 51.6 (3.0) HT vs. 15.0 (0.8) controls |
| TREATMENT FAILURE | ||||||||||
| Johnsen 2011 [11] | Population sample | European | Norway | 12776 | 30–87 | No ‡ | Modular P, Roche | Yes | CK not significantly higher in uncontrolled vs. controlled HT | CK in controlled vs. uncontrolled HT 101 vs. 110 †† |
| Brewster 2013 [7] | Random population sample | African Asian European | Netherlands | 1444 | 35–60 | Yes | Roche/Hitachi Systems | Yes║ | CK higher in uncontrolled vs. controlled HT | CK in controlled vs. uncontrolled HT 124.3 (10.9) vs. 157.9 (9.4) |
| Odds ratio of treatment failure CK T1 (<88) vs. CK T3 (≥145) HT treatment failure 46.7% vs. 72.9% Odds ratio 1.6 [1.3 to 1.9] | ||||||||||
| Adjusted odds ratio treatment failure 3.7 [1.2 to 10.9]/log CK | ||||||||||
| Luman 2015 [13] | Hypertensives | Asian | Indonesia | 82 | >18 | No | Roche/Hitachi cobas analyzer | Yes | CK higher in uncontrolled vs. controlled HT | Mean CK in controlled vs. uncontrolled HT 81.8 (3.3) vs. 132.2 (6.2) |
| High CK (T3 CK>109.33 U/L) Controlled hypertension 18.5% Uncontrolled hypertension 81.5% | ||||||||||
| Sukul 2018 [18] | Hypertensives vs. controls | Asian | India | 115 | 25–60 | Yes | Roche diagnostics | Yes ║ | CK higher in uncontrolled vs. controlled HT | CK in controlled vs. uncontrolled HT 99.6 (4.5) vs. 313.9 (22.5) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brewster, L.M.; Karamat, F.A.; van Montfrans, G.A. Creatine Kinase and Blood Pressure: A Systematic Review. Med. Sci. 2019, 7, 58. https://doi.org/10.3390/medsci7040058
Brewster LM, Karamat FA, van Montfrans GA. Creatine Kinase and Blood Pressure: A Systematic Review. Medical Sciences. 2019; 7(4):58. https://doi.org/10.3390/medsci7040058
Chicago/Turabian StyleBrewster, L. M., F. A. Karamat, and G. A. van Montfrans. 2019. "Creatine Kinase and Blood Pressure: A Systematic Review" Medical Sciences 7, no. 4: 58. https://doi.org/10.3390/medsci7040058
APA StyleBrewster, L. M., Karamat, F. A., & van Montfrans, G. A. (2019). Creatine Kinase and Blood Pressure: A Systematic Review. Medical Sciences, 7(4), 58. https://doi.org/10.3390/medsci7040058





