Inhibitor of DNA-Binding/Differentiation Proteins and Environmental Toxicants: Genomic Impact on the Onset of Depressive Dysfunction
Abstract
:1. Introduction
2. Inhibitor of DNA Binding/Differentiation
2.1. Background
2.2. Inhibitor of DNA Binding/Differentiation and Depressive Disorders
2.3. Inhibitor of DNA Binding/Differentiation and Environmental Pollutants
3. Relationship between Environmental Toxicants and Depressive Disorders
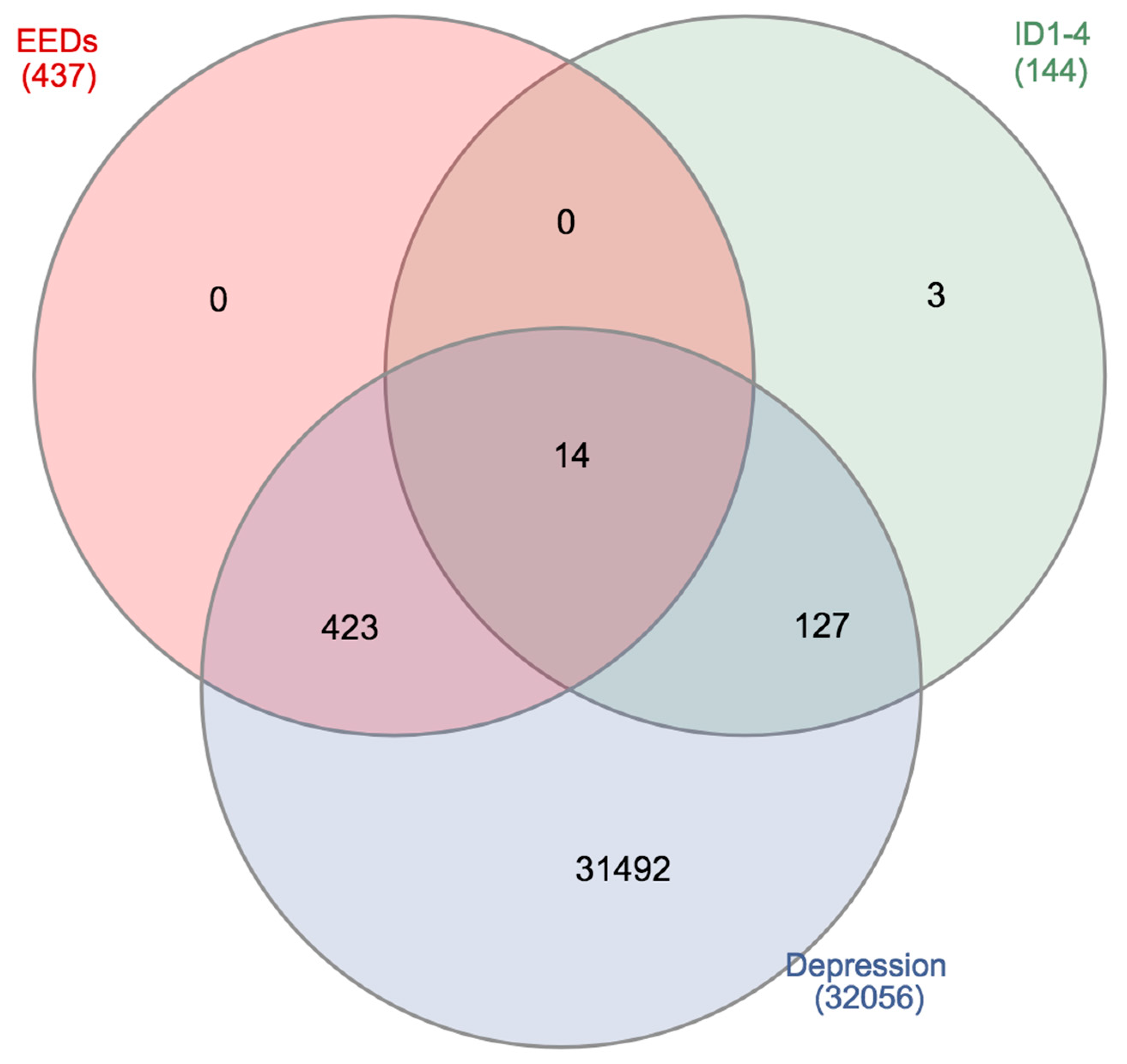
4. Genomic Interactions between the Inhibitor of DNA Binding/Differentiation, Environmental Toxicants, and Depression
5. Conclusions
Funding
Conflicts of Interest
References
- Depression. NIH National Institute of Mental Health. Available online: https://www.nimh.nih.gov/health/topics/depression/index.shtml (accessed on 5 September 2018).
- Mental Health Information, Statistics of Depression. NIH National Institute of Mental Health. Available online: https://www.nimh.nih.gov/health/statistics/major-depression.shtml (accessed on 5 September 2018).
- Zhang, M.R.; Qu, C.; Sun, J.; Wang, C.; Li, H.Y.; Zhang, Y.J.; Zhang, B.Q.; Zou, W. Different subtypes of estrogen receptor α and related signal molecules in the hippocampus are associated with spatial cognitive impairment of diabetic mice. Acta Physiol. Sin. 2017, 69, 252–260. [Google Scholar]
- Richard, A.; Rohrmann, S.; Mohler-Kuo, M.; Rodgers, S.; Moffat, R.; Güth, U.; Eichholzer, M. Urinary phytoestrogens and depression in perimenopausal US women: NHANES 2005–2008. J. Affect Disord. 2014, 156, 200–205. [Google Scholar] [CrossRef]
- Newhouse, P.; Albert, K. Estrogen, Stress, and Depression: A Neurocognitive Model. JAMA Psychiatry 2015, 72, 727–729. [Google Scholar] [CrossRef]
- Najjar, F.; Ahmad, M.; Lagace, D.; Leenen, F.H.H. Sex Differences in Depression-Like Behavior and Neuroinflammation in Rats Post MI: Role of Estrogens. Am. J. Physiol. Heart Circ. Physiol. 2018, 315, H1159–H1173. [Google Scholar] [CrossRef]
- Nadal, A.; Ropero, A.B.; Laribi, O.; Maillet, M.; Fuentes, E.; Soria, B. Nongenomic actions of estrogens and xenoestrogens by binding at a plasma membrane receptor unrelated to estrogen receptor α and estrogen receptor β. Proc. Natl. Acad. Sci. USA 2000, 97, 11603–11608. [Google Scholar] [CrossRef]
- Watson, C.S.; Bulayeva, N.N.; Wozniak, A.L.; Finnerty, C.C. Signaling from the membrane via membrane estrogen receptor-α: Estrogens, xenoestrogens, and phytoestrogens. Steroids 2005, 70, 364–371. [Google Scholar] [CrossRef]
- Thomas, P.; Dong, J. Binding and activation of the seven transmembrane estrogen receptor GPR30 by environmental estrogens: A potential novel mechanism of endocrine disruption. J. Steroid Biochem. Mol. Biol. 2006, 102, 175–179. [Google Scholar] [CrossRef]
- Korach, K.S.; Sarver, P.; Chae, K.; McLachlan, J.A.; McKinney, J.D. Estrogen receptor-binding activity of polychlorinated hydroxybiphenyls: Conformationally restricted structural probes. Mol. Pharmacol. 1988, 33, 120–126. [Google Scholar]
- Bonefeld-Jorgensen, E.C.; Andersen, H.R.; Rasmussen, T.H.; Vinggaard, A.M. Effect of highly bioaccumulated polychlorinated biphenyl congeners on estrogen and androgen receptor activity. Toxicology 2001, 158, 141–153. [Google Scholar] [CrossRef]
- Alboni, S.; van Dijk, R.M.; Poggini, S.; Milior, G.; Perrotta, M.; Drenth, T.; Brunello, N.; Wolfer, D.P.; Limatola, C.; Amrein, I.; et al. Fluoxetine effects on molecular, cellular and behavioral endophenotypes of depression are driven by the living environment. Mol. Psychiatry 2017, 22, 552–561. [Google Scholar] [CrossRef]
- Strøm, M.; Hansen, S.; Olsen, S.F.; Haug, L.S.; Rantakokko, P.; Kiviranta, H.; Halldorsson, T.I. Persistent organic pollutants measured in maternal serum and offspring neurodevelopmental outcomes—A prospective study with long-term follow-up. Environ. Int. 2014, 68, 41–48. [Google Scholar] [CrossRef]
- Kajta, M.; Wójtowicz, A.K. Impact of endocrine-disrupting chemicals on neural development and the onset of neurological disorders. Pharmacol. Rep. 2013, 65, 1632–1639. [Google Scholar] [CrossRef]
- Xu, X.; Dong, F.; Yang, Y.; Wang, Y.; Wang, R.; Shen, X. Sex-specific effects of long-term exposure to bisphenol-A on anxiety- and depression-like behaviors in adult mice. Chemosphere 2015, 120, 258–266. [Google Scholar] [CrossRef]
- Harley, K.G.; Gunier, R.B.; Kogut, K.; Johnson, C.; Bradman, A.; Calafat, A.M.; Eskenazi, B. Prenatal and early childhood bisphenol A concentrations and behavior in school-aged children. Environ. Res. 2013, 126, 43–50. [Google Scholar] [CrossRef] [Green Version]
- Mustieles, V.; Pérez-Lobato, R.; Olea, N.; Fernández, M.F. Bisphenol A: Human exposure and neurobehavior. Neurotoxicology 2015, 49, 174–184. [Google Scholar] [CrossRef]
- Avecilla, V.; Doke, M.; Felty, Q. Contribution of Inhibitor of DNA Binding/Differentiation-3 and Endocrine Disrupting Chemicals to Pathophysiological Aspects of Chronic Disease. Biomed. Res. Int. 2017, 2017, 6307109. [Google Scholar] [CrossRef]
- Avecilla, A.; Doke, M.; Jovellanos, J.; Avecilla, V. Contribution of Inhibitor of Differentiation and Estrogenic Endocrine Disruptors to Neurocognitive Disorders. Med. Sci. 2018, 6, 61. [Google Scholar] [CrossRef]
- Savalli, G.; Diao, W.; Schulz, S.; Todtova, K.; Pollak, D.D. Diurnal oscillation of amygdala clock gene expression and loss of synchrony in a mouse model of depression. Int. J. Neuropsychopharmacol. 2014, 18, pyu095. [Google Scholar] [CrossRef]
- Weder, N.; Zhang, H.; Jensen, K.; Yang, B.Z.; Simen, A.; Jackowski, A.; Lipschitz, D.; Douglas-Palumberi, H.; Ge, M.; Perepletchikova, F.; et al. Child abuse, depression, and methylation in genes involved with stress, neural plasticity, and brain circuitry. J. Am. Acad. Child. Adolesc. Psychiatry 2014, 53, 417–424. [Google Scholar] [CrossRef]
- Montalvo-Ortiz, J.L.; Bordner, K.A.; Carlyle, B.C.; Gelernter, J.; Simen, A.A.; Kaufman, J. The role of genes involved in stress, neural plasticity, and brain circuitry in depressive phenotypes: Convergent findings in a mouse model of neglect. Behav. Brain Res. 2016, 315, 71–74. [Google Scholar] [CrossRef] [Green Version]
- Lyden, D.; Young, A.Z.; Zagzag, D.; Yan, W.; Gerald, W.; O’Reilly, R.; Bader, B.L.; Hynes, R.O.; Zhuang, Y.; Manova, K.; et al. Id1 and Id3 are required for neurogenesis, angiogenesis and vascularization of tumour xenografts. Nature 1999, 401, 670–677. [Google Scholar] [CrossRef]
- Yang, J.; Li, X.; Morrell, N.W. Id proteins in the vasculature: From molecular biology to cardiopulmonary medicine. Cardiovasc. Res. 2014, 104, 388–398. [Google Scholar] [CrossRef]
- Benezra, R.; Davis, R.L.; Lockshon, D.; Turner, D.L.; Weintraub, H. The protein Id: A negative regulator of helix-loop-helix DNA binding proteins. Cell 1990, 61, 49–59. [Google Scholar] [CrossRef]
- Norton, J.D. ID helix-loop-helix proteins in cell growth, differentiation and tumorigenesis. J. Cell Sci. 2000, 113, 3897–3905. [Google Scholar]
- Ruzinova, M.B.; Benezra, R. ID proteins in development, cell cycle and cancer. Trends Cell Biol. 2003, 13, 410–418. [Google Scholar] [CrossRef]
- Fouad, Y.A.; Aanei, C. Revisiting the hallmarks of cancer. Am. J. Cancer Res. 2017, 7, 1016–1036. [Google Scholar]
- Tzeng, S.F.; de Vellis, J. Id1, Id2, and Id3 gene expression in neural cells during development. Glia 1998, 24, 372–381. [Google Scholar] [CrossRef]
- Neuman, T.; Keen, A.; Zuber, M.X.; Kristjansson, G.I.; Gruss, P.; Nornes, H.O. Neuronal Expression of Regulatory Helix-Loop-Helix Factor Id2 Gene in Mouse. Dev. Biol. 1993, 160, 186–195. [Google Scholar] [CrossRef]
- Roschger, C.; Cabrele, C. The Id-protein family in developmental and cancer-associated pathways. Cell Commun. Signal. 2017, 15, 7. [Google Scholar] [CrossRef] [Green Version]
- Shibata, M.; Suzuki, N. Exploring the role of microglia in cortical spreading depression in neurological disease. J. Cereb. Blood Flow Metab. 2017, 37, 1182–1191. [Google Scholar] [CrossRef] [Green Version]
- Tohyama, M.; Miyata, S.; Hattori, T.; Shimizu, S.; Matsuzaki, S. Molecular basis of major psychiatric diseases such as schizophrenia and depression. Anat. Sci. Int. 2015, 90, 137–143. [Google Scholar] [CrossRef]
- Akeel, S.; El-awady, A.; Hussein, K.; El-Refaey, M.; Elsalanty, M.; Sharawy, M.; Al-Shabrawey, M. Recombinant bone morphogenetic protein-2 induces up-regulation of vascular endothelial growth factor and interleukin 6 in human pre-osteoblasts: Role of reactive oxygen species. Arch. Oral Biol. 2012, 57, 445–452. [Google Scholar] [CrossRef]
- Mueller, C.; Baudler, S.; Welzel, H.; Böhm, M.; Nickenig, G. Identification of a novel redox-sensitive gene, Id3, which mediates angiotensin II-induced cell growth. Circulation 2002, 105, 2423–2428. [Google Scholar] [CrossRef]
- Nickenig, G.; Baudler, S.; Müller, C.; Werner, C.; Werner, N.; Welzel, H.; Strehlow, K.; Böhm, M. Redox-sensitive vascular smooth muscle cell proliferation is mediated by GKLF and Id3 in vitro and in vivo. FASEB J. 2002, 16, 1077–1086. [Google Scholar] [CrossRef]
- Das, J.K.; Felty, Q. PCB153-induced overexpression of ID3 contributes to the development of microvascular lesions. PLoS ONE 2014, 9, e104159. [Google Scholar] [CrossRef]
- Das, J.K.; Felty, Q. Microvascular Lesions by Estrogen-Induced ID3: Its Implications in Cerebral and Cardiorenal Vascular Disease. J. Mol. Neurosci. 2014, 55, 618–631. [Google Scholar] [CrossRef]
- Felty, Q. Proteomic 2D DIGE profiling of human vascular endothelial cells exposed to environmentally relevant concentration of endocrine disruptor PCB153 and physiological concentration of 17β-estradiol. Cell Biol. Toxicol. 2011, 27, 49–68. [Google Scholar] [CrossRef]
- Morris, G.; Walder, K.; McGee, S.L.; Dean, O.M.; Tye, S.J.; Maes, M.; Berk, M. A model of the mitochondrial basis of bipolar disorder. Neurosci. Biobehav. Rev. 2017, 74, 1–20. [Google Scholar] [CrossRef]
- Sperner-Unterweger, B.; Kohl, C.; Fuchs, D. Immune changes and neurotransmitters: Possible interactions in depression? Prog. Neuropsychopharmacol. Biol. Psychiatry 2014, 48, 268–276. [Google Scholar] [CrossRef]
- Yasunari, K.; Matsui, T.; Maeda, K.; Nakamura, M.; Watanabe, T.; Kiriike, N. Anxiety-induced plasma norepinephrine augmentation increases reactive oxygen species formation by monocytes in essential hypertension. Am. J. Hypertens. 2006, 19, 573–578. [Google Scholar] [CrossRef]
- Then, C.K.; Liu, K.H.; Liao, M.H.; Chung, K.H.; Wang, J.Y.; Shen, S.C. Antidepressants, sertraline and paroxetine, increase calcium influx and induce mitochondrial damage-mediated apoptosis of astrocytes. Oncotarget 2017, 8, 115490–115502. [Google Scholar] [CrossRef]
- Doke, M.; Avecilla, V.; Felty, Q. Inhibitor of Differentiation-3 and Estrogenic Endocrine Disruptors: Implications for Susceptibility to Obesity and Metabolic Disorders. BioMed Res. Int. 2018, 2018, 6821601. [Google Scholar] [CrossRef] [PubMed]
- LaRocca, J.; Boyajian, A.; Brown, C.; Smith, S.D.; Hixon, M. Effects of in utero exposure to Bisphenol A or diethylstilbestrol on the adult male reproductive system. Birth Defects Res. B Dev. Reprod. Toxicol. 2011, 92, 526–533. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.H.; Yang, M.H.; Hung, A.C.; Wu, S.C.; Chiu, W.C.; Hou, M.F.; Tyan, Y.C.; Wang, Y.M.; Yuan, S.F. Identification of Id1 as a downstream effector for arsenic-promoted angiogenesis via PI3K/Akt, NF-κB and NOS signaling. Toxicol. Res. 2015, 5, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Kurooka, H.; Sugai, M.; Mori, K.; Yokota, Y. The metalloid arsenite induces nuclear export of Id3 possibly via binding to the N-terminal cysteine residues. Biochem. Biophys. Res. Commun. 2013, 433, 579–585. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Lv, C.; Du, B.; Ma, X.; Guan, J.; Gao, S.; Li, X.; Zheng, L.; Lei, L. Upregulation of ID2 antagonizes arsenic trioxide-induced antitumor effects in cancer cells. Tumori 2014, 100, 352–357. [Google Scholar] [CrossRef]
- Yao, P.L.; Lin, Y.C.; Richburg, J.H. Mono-(2-ethylhexyl) phthalate (MEHP) promotes invasion and migration of human testicular embryonal carcinoma cells. Biol. Reprod. 2012, 86, 1–10. [Google Scholar] [CrossRef]
- Gaum, P.M.; Gube, M.; Schettgen, T.; Putschögl, F.M.; Kraus, T.; Fimm, B.; Lang, J. Polychlorinated biphenyls and depression: Cross-sectional and longitudinal investigation of a dopamine-related Neurochemical path in the German HELPcB surveillance program. Environ. Health 2017, 16, 106. [Google Scholar] [CrossRef]
- Perera, F.; Nolte, E.L.R.; Wang, Y.; Margolis, A.E.; Calafat, A.M.; Wang Garcia, W.; Hoepner, L.A.; Peterson Rauh, V.; Herbstman, J. Bisphenol A exposure and symptoms of anxiety and depression among inner city children at 10–12 years of age. Environ. Res. 2016, 151, 195–202. [Google Scholar] [CrossRef] [Green Version]
- Valdés, M.; Hanchey, A.; Muñoz, M.P.; Baumert, B.; Iglesias, V. Low-level arsenic exposure during pregnancy and its association with postpartum depression: A cohort study of women from Arica, Chile. Rev. Epidemiol. 2017, 65, 427–435. [Google Scholar] [CrossRef]
- Chang, C.Y.; Guo, H.R.; Tsai, W.C.; Yang, K.L.; Lin, L.C.; Cheng, T.J.; Chuu, J.J. Subchronic Arsenic Exposure Induces Anxiety-Like Behaviors in Normal Mice and Enhances Depression-Like Behaviors in the Chemically Induced Mouse Model of Depression. Biomed. Res. Int. 2015, 2015, 159015. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.P.; Grondin, C.J.; Johnson, R.J.; Sciaky, D.; King, B.L.; McMorran, R.; Wiegers, J.; Wiegers, T.C.; Mattingly, C.J. The Comparative Toxicogenomics Database: Update 2017. Nucleic Acids Res. 2017, 45, 972–978. [Google Scholar] [CrossRef] [PubMed]
- Heberle, H.; Meirelles, G. V.; da Silva, F. R.; Telles, G. P.; Minghim, R. InteractiVenn: A web-based tool for the analysis of sets through Venn diagrams. BMC Bioinform. 2015, 16, 169. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 2000, 28, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Caraci, F.; Spampinato, S.F.; Morgese, M.G.; Tascedda, F.; Salluzzo, M.G.; Giambirtone, M.C.; Caruso, G.; Munafò, A.; Torrisi, S.A.; Leggio, G.M.; et al. Neurobiological links between depression and AD: The role of TGF-β1 signaling as a new pharmacological target. Pharmacol. Res. 2018, 130, 374–384. [Google Scholar] [CrossRef] [PubMed]
- Trojan, E.; Ślusarczyk, J.; Chamera, K.; Kotarska, K.; Głombik, K.; Kubera, M.; Basta-Kaim, A. The Modulatory Properties of Chronic Antidepressant Drugs Treatment on the Brain Chemokine—Chemokine Receptor Network: A Molecular Study in an Animal Model of Depression. Front. Pharmacol. 2017, 8, 779. [Google Scholar] [CrossRef]
- Chhibber, A.; Woody, S.K.; Karim Rumi, M.A.; Soares, M.J.; Zhao, L. Estrogen receptor β deficiency impairs BDNF-5-HT2A signaling in the hippocampus of female brain: A possible mechanism for menopausal depression. Psychoneuroendocrinology 2017, 82, 107–116. [Google Scholar] [CrossRef]
- Heldring, N.; Pike, A.; Andersson, S.; Matthews, J.; Cheng, G.; Hartman, J.; Tujague, M.; Ström, A.; Treuter, E.; Warner, M.; Gustafsson, J.A. Estrogen receptors: How do they signal and what are their targets. Physiol. Rev. 2007, 87, 905–931. [Google Scholar] [CrossRef]
- Borsini, A.; Zunszain, P.A. Advances in Stem Cells Biology: New Approaches to Understand Depression. In Stem Cells in Neuroendocrinology; Springer: Cham, Switzerland, 2016. [Google Scholar]
- Szklarczyk, D.; Morris, J.H.; Cook, H.; Kuhn, M.; Wyder, S.; Simonovic, M.; Santos, A.; Doncheva, N.T.; Roth, A.; Bork, P.; et al. The STRING database in 2017: Quality-controlled protein-protein association networks, made broadly accessible. Nucleic Acids Res. 2017, 45, 362–368. [Google Scholar] [CrossRef]
- Szklarczyk, D.; Franceschini, A.; Wyder, S.; Forslund, K.; Heller, D.; Huerta-Cepas, J.; Simonovic, M.; Roth, A.; Santos, A.; Tsafou, K.P.; et al. STRING v10: Protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res. 2015, 43, 447–452. [Google Scholar] [CrossRef]


| Gene Symbol | Gene Name |
|---|---|
| ATF3 | Activating transcription factor 3 |
| CDK2 | Cyclin-dependent kinase 2 |
| ELOC | Elongin C |
| GATA4 | GATA binding protein 4 |
| HSPA1A | Heat shock protein family A (Hsp70) member 1A |
| HSPA5 | Heat shock protein family A (Hsp70) member 5 |
| HSPA8 | Heat shock protein family A (Hsp70) member 8 |
| HSPA9 | Heat shock protein family A (Hsp70) member 9 |
| ID1 | Inhibitor of DNA binding 1, HLH protein |
| ID2 | Inhibitor of DNA binding 2, HLH protein |
| ID3 | Inhibitor of DNA binding 3, HLH protein |
| MAPK1 | Mitogen-activated protein kinase 1 |
| MAPK3 | Mitogen-activated protein kinase 3 |
| SREBF1 | Sterol regulatory element binding transcription factor 1 |
| Pathway Name | Gene Count | p-Value | Genes |
|---|---|---|---|
| TGF-beta signaling pathway | 5 | 9.34 × 10−6 | MAPK1, ID2, ID1, MAPK3, ID3 |
| Signaling pathways regulating pluripotency of stem cells | 5 | 7.04 × 10−5 | MAPK1, ID2, ID1, MAPK3, ID3 |
| Estrogen signaling pathway | 4 | 5.71 × 10−4 | MAPK1, MAPK3, HSPA1A, HSPA8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Avecilla, V.; Avecilla, A. Inhibitor of DNA-Binding/Differentiation Proteins and Environmental Toxicants: Genomic Impact on the Onset of Depressive Dysfunction. Med. Sci. 2019, 7, 7. https://doi.org/10.3390/medsci7010007
Avecilla V, Avecilla A. Inhibitor of DNA-Binding/Differentiation Proteins and Environmental Toxicants: Genomic Impact on the Onset of Depressive Dysfunction. Medical Sciences. 2019; 7(1):7. https://doi.org/10.3390/medsci7010007
Chicago/Turabian StyleAvecilla, Vincent, and Andrea Avecilla. 2019. "Inhibitor of DNA-Binding/Differentiation Proteins and Environmental Toxicants: Genomic Impact on the Onset of Depressive Dysfunction" Medical Sciences 7, no. 1: 7. https://doi.org/10.3390/medsci7010007





