Cell Therapy in Idiopathic Pulmonary Fibrosis†
Abstract
1. Introduction
2. Stem Cells
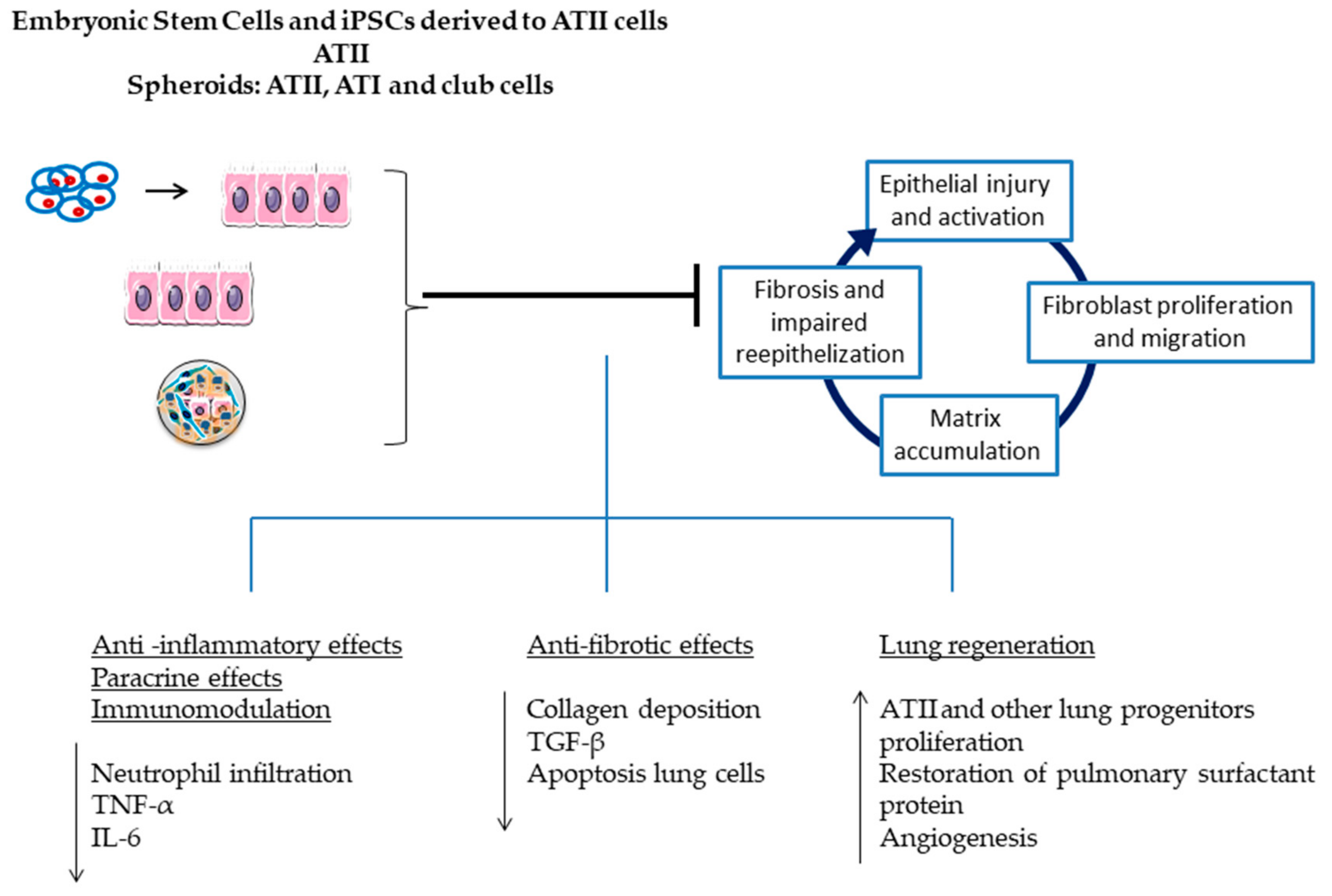
2.1. Embryonic Stem Cells
2.2. Adult Stem Cells
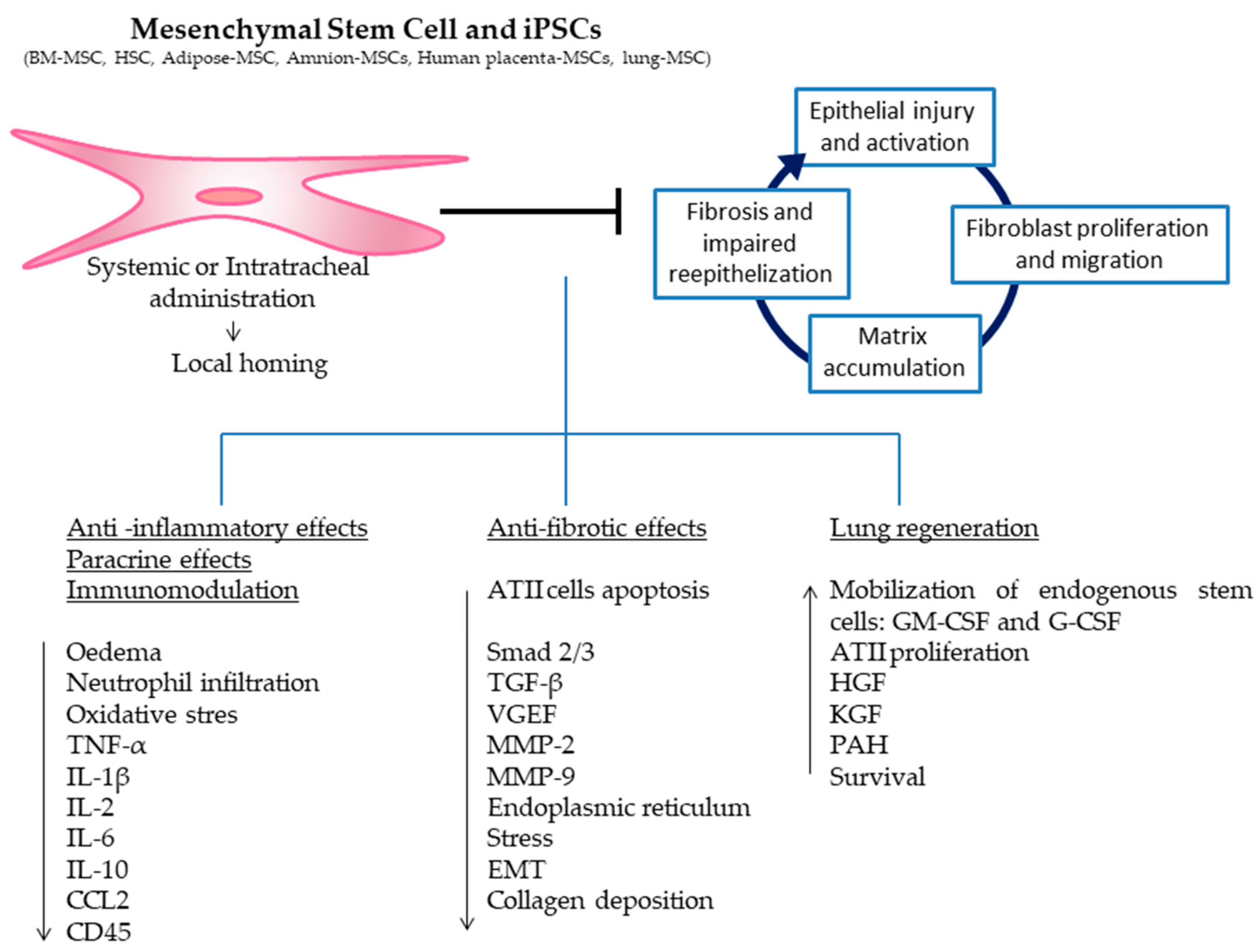
2.2.1. Bone Marrow Stem Cells
- Hematopoietic stem cells
- Mesenchymal stromal stem cells
2.2.2. Induced Pluripotent Stem Cells
2.2.3. Lung Stem/Progenitor Cells
3. Preclinical Experiences
4. Clinical Experiences
5. Conclusions and Future Perspectives
Funding
Conflicts of Interest
References
- Katzenstein, A.L.; Myers, J.L. Idiopathic pulmonary fibrosis: Clinical relevance of pathologic classification. Am. J. Respir. Crit. Care Med. 1998, 157, 1301–1315. [Google Scholar] [CrossRef] [PubMed]
- Phan, S.H. Biology of fibroblasts and myofibroblasts. Proc. Am. Thorac. Soc. 2008, 15, 334–337. [Google Scholar] [CrossRef] [PubMed]
- Wynes, M.W.; Edelman, B.L.; Kostyk, A.G.; Edwards, M.G.; Coldren, C.; Groshong, S.D.; Cosgrove, G.P.; Redente, E.F.; Bamberg, A.; Brown, K.K.; et al. Increased cell surface Fas expression is necessary and sufficient to sensitize lung fibroblasts to Fas ligation-induced apoptosis: Implications for fibroblast accumulation in idiopathic pulmonary fibrosis. J. Immunol. 2011, 187, 527–537. [Google Scholar] [CrossRef] [PubMed]
- King, T.E., Jr.; Bradford, W.Z.; Castro-Bernardini, S.; Fagan, E.A.; Glaspole, I.; Glassberg, M.K.; Gorina, E.; Hopkins, P.M.; Kardatzke, D.; Lancaster, L.; et al. ASCEND Study Group. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N. Engl. J. Med. 2014, 370, 2083–2092. [Google Scholar] [CrossRef] [PubMed]
- Richeldi, L.; du Bois, R.M.; Raghu, G.; Azuma, A.; Brown, K.K.; Costabel, U.; Cottin, V.; Flaherty, K.R.; Hansell, D.M.; Inoue, Y.; et al. Efficacy and safety of nintedanib in diopathic pulmonary fibrosis. N. Engl. J. Med. 2014, 370, 2071–2082. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, L.A.; Gambelli, F.; McBride, C.; Gaupp, D.; Baddoo, M.; Kaminski, N.; Phinney, D.G. Mesenchymal stem cell engraftment in lung is enhanced in response to bleomycin exposure and ameliorates its fibrotic effects. Proc. Natl. Acad. Sci. USA 2003, 100, 8407–8411. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.; Kang, X.; Wang, X.; Wu, S.; Xiao, J.; Li, Z.; Wu, X.; Zhang, W. Conversion of bone marrow mesenchymal stem cells into type II alveolar epithelial cells reduces pulmonary fibrosis by decreasing oxidative stress in rats. Mol. Med. Rep. 2015, 11, 1685–1692. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, L.A.; Dutreil, M.; Fattman, C.; Pandey, A.C.; Torres, G.; Go, K.; Phinney, D.G. Interleukin 1 receptor antagonist mediates the antiinflammatory and antifibrotic effect of mesenchymal stem cells during lung injury. Proc. Natl. Acad. Sci. USA 2007, 104, 11002–11007. [Google Scholar] [CrossRef] [PubMed]
- Rojas, M.; Xu, J.; Woods, C.R.; Mora, A.L.; Spears, W.; Roman, J.; Brigham, K.L. Bone marrow-derived mesenchymal stem cells in repair of the injured lung. Am. J. Respir. Cell Mol. Biol. 2005, 33, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Cahill, E.F.; Kennelly, H.; Carty, F.; Mahon, B.P.; English, K. Hepatocyte growth factor is required for mesenchymal stromal cell protection against bleomycin-induced pulmonary fibrosis. Stem Cells Transl. Med. 2016, 5, 1307–1318. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, S.; Scotton, C.J.; McNulty, K.; Nye, E.; Stamp, G.; Laurent, G.; Bonnet, D.; Janes, S.M. Bone marrow stem cells expressing keratinocyte growth factor via an inducible lentivirus protects against bleomycin-induced pulmonary fibrosis. PLoS ONE 2009, 4, e8013. [Google Scholar] [CrossRef] [PubMed]
- Ono, M.; Ohkouchi, S.; Kanehira, M.; Tode, N.; Kobayashi, M.; Ebina, M.; Nukiwa, T.; Irokawa, T.; Ogawa, H.; Akaike, T.; et al. Mesenchymal stem cells correct inappropriate epithelial-mesenchyme relation in pulmonary fibrosis using stanniocalcin-1. Mol. Ther. 2015, 23, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Liebler, J.M.; Lutzko, C.; Banfalvi, A.; Senadheera, D.; Aghamohammadi, N.; Crandall, E.D.; Borok, Z. Retention of human bone marrow-derived cells in murine lungs following bleomycin-induced lung injury. Am. J. Physiol. Lung Cell. Mol. Physiol. 2008, 295, L285–L292. [Google Scholar] [CrossRef] [PubMed]
- Lan, Y.W.; Choo, K.B.; Chen, C.M.; Hung, T.H.; Chen, Y.B.; Hsieh, C.H.; Kuo, H.P.; Chong, K.Y. Hypoxia-preconditioned mesenchymal stem cells attenuate bleomycin-induced pulmonary fibrosis. Stem Cell Res. Ther. 2015, 6, 97. [Google Scholar] [CrossRef] [PubMed]
- Lan, Y.W.; Theng, S.M.; Huang, T.T.; Choo, K.B.; Chen, C.M.; Kuo, H.P.; Chong, K.Y. Oncostatin M-preconditioned mesenchymal stem cells alleviate bleomycin-induced pulmonary fibrosis through paracrine effects of the hepatocyte growth factor. Stem Cells Transl. Med. 2017, 6, 1006–1017. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Jang, A.S.; Kim, Y.E.; Cha, J.Y.; Kim, T.H.; Jung, S.; Park, S.K.; Lee, Y.K.; Won, J.H.; Kim, Y.H.; Park, C.S. Modulation of cytokine and nitric oxide by mesenchymal stem cell transfer in lung injury/fibrosis. Respir. Res. 2010, 11, 16. [Google Scholar] [CrossRef] [PubMed]
- Moodley, Y.; Vaghjiani, V.; Chan, J.; Baltic, S.; Ryan, M.; Tchongue, J.; Samuel, C.S.; Murthi, P.; Parolini, O.; Manuelpillai, U. Anti-inflammatory effects of adult stem cells in sustained lung injury: A comparative study. PLoS ONE 2013, 8, e69299. [Google Scholar] [CrossRef] [PubMed]
- Gazdhar, A.; Susuri, N.; Hostettler, K.; Gugger, M.; Knudsen, L.; Roth, M.; Ochs, M.; Geiser, T. HGF expressing stem cells in usual interstitial pneumonia originate from the bone marrow and are antifibrotic. PLoS ONE 2013, 8, e65453. [Google Scholar] [CrossRef] [PubMed]
- Huleihel, L.; Sellares, J.; Cardenes, N.; Álvarez, D.; Faner, R.; Sakamoto, K.; Yu, G.; Kapetanaki, M.G.; Kaminski, N.; Rojas, M. Modified mesenchymal stem cells using miRNA transduction alter lung injury in a bleomycin model. Am. J. Physiol. Lung Cell. Mol. Physiol. 2017, 313, L92–L103. [Google Scholar] [CrossRef] [PubMed]
- Tashiro, J.; Elliot, S.J.; Gerth, D.J.; Xia, X.; Pereira-Simon, S.; Choi, R.; Catanuto, P.; Shahzeidi, S.; Toonkel, R.L.; Shah, R.H.; et al. Therapeutic benefits of young, but not old, adipose-derived mesenchymal stem cells in a chronic mouse model of bleomycin-induced pulmonary fibrosis. Transl. Res. 2015, 166, 554–567. [Google Scholar] [CrossRef] [PubMed]
- Kotani, T.; Masutani, R.; Suzuka, T.; Oda, K.; Makino, S.; Ii, M. Anti-inflammatory and anti-fibrotic effects of intravenous adipose-derived stem cell transplantation in a mouse model of bleomycin-induced interstitial pneumonia. Sci. Rep. 2017, 7, 14608. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Lee, E.J.; Lee, S.Y.; Kim, J.H.; Shim, J.J.; Shin, C.; In, K.H.; Kang, K.H.; Uhm, C.S.; Kim, H.K.; et al. The effect of adipose stem cell therapy on pulmonary fibrosis induced by repetitive intratracheal bleomycin in mice. Exp. Lung Res. 2014, 40, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Cargnoni, A.; Gibelli, L.; Tosini, A.; Signoroni, P.B.; Nassuato, C.; Arienti, D.; Lombardi, G.; Albertini, A.; Wengler, G.S.; Parolini, O. Transplantation of allogeneic and xenogeneic placenta-derived cells reduces bleomycin-induced lung fibrosis. Cell Transplant. 2009, 18, 405–422. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Han, F.; Li, H.; Zhang, J.; Qiao, X.; Shi, J.; Yang, L.; Dong, J.; Luo, M.; Wei, J.; et al. Human placental mesenchymal stem cells of fetal origins-alleviated inflammation and fibrosis by attenuating MyD88 signaling in bleomycin-induced pulmonary fibrosis mice. Mol. Immunol. 2017, 90, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Moodley, Y.; Atienza, D.; Manuelpillai, U.; Samuel, C.S.; Tchongue, J.; Ilancheran, S.; Boyd, R.; Trounson, A. Human umbilical cord mesenchymal stem cells reduce fibrosis of bleomycin-induced lung injury. Am. J. Pathol. 2009, 175, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Garcia, O.; Carraro, G.; Turcatel, G.; Hall, M.; Sedrakyan, S.; Roche, T.; Buckley, S.; Driscoll, B.; Perin, L.; Warburton, D. Amniotic fluid stem cells inhibit the progression of bleomycin-induced pulmonary fibrosis via CCL2 modulation in bronchoalveolar lavage. PLoS ONE 2013, 8, e71679. [Google Scholar] [CrossRef] [PubMed]
- How, C.K.; Chien, Y.; Yang, K.Y.; Shih, H.C.; Juan, C.C.; Yang, Y.P.; Chiou, G.Y.; Huang, P.I.; Chang, Y.L.; Chen, L.K.; et al. Induced pluripotent stem cells mediate the release of interferon gamma-induced protein 10 and alleviate bleomycin-induced lung inflammation and fibrosis. Shock 2013, 39, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; He, Z.; Gao, Y.; Zheng, R.; Zhang, X.; Zhao, L.; Tan, M. Induced pluripotent stem cells inhibit bleomycin-induced pulmonary fibrosis in mice through suppressing TGF-β1/Smad-mediated epithelial to mesenchymal transition. Front. Pharmacol. 2016, 7, 430. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Ye, X.; Sun, R.; Matsumoto, Y.; Moriyama, M.; Asano, Y.; Ajioka, Y.; Saijo, Y. Differentiation of mouse induced pluripotent stem cells into alveolar epithelial cells in vitro for use in vivo. Stem Cells Transl. Med. 2014, 3, 675–685. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, E.R.; Laflamme, M.A.; Papayannopoulou, T.; Kahn, M.; Murry, C.E.; Henderson, W.R., Jr. Human embryonic stem cells differentiated to lung lineage-specific cells ameliorate pulmonary fibrosis in a xenograft transplant mouse model. PLoS ONE 2012, 7, e33165. [Google Scholar] [CrossRef] [PubMed]
- Jun, D.; Garat, C.; West, J.; Thorn, N.; Chow, K.; Cleaver, T.; Sullivan, T.; Torchia, E.C.; Childs, C.; Shade, T.; et al. The pathology of bleomycin-induced fibrosis is associated with loss of resident lung mesenchymal stem cells that regulate effector T.-cell proliferation. Stem Cells 2011, 29, 725–735. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Mollar, A.; Nacher, M.; Gay-Jordi, G.; Closa, D.; Xaubet, A.; Bulbena, O. Intratracheal transplantation of alveolar type II cells reverses bleomycin-induced lung fibrosis. Am. J. Respir. Crit. Care Med. 2007, 176, 1261–1268. [Google Scholar] [CrossRef] [PubMed]
- Guillamat-Prats, R.; Gay-Jordi, G.; Xaubet, A.; Peinado, V.I.; Serrano-Mollar, A. Alveolar type II cell transplantation restores pulmonary surfactant protein levels in lung fibrosis. J. Heart Lung Transplant. 2014, 33, 758–765. [Google Scholar] [CrossRef] [PubMed]
- Cores, J.; Hensley, M.T.; Kinlaw, K.; Rikard, S.M.; Dinh, P.U.; Paudel, D.; Tang, J.; Vandergriff, A.C.; Allen, T.A.; Li, Y.; et al. Safety and efficacy of allogeneic lung spheroid cells in a mismatched rat model of pulmonary fibrosis. Stem Cells Transl. Med. 2017, 6, 1905–1916. [Google Scholar] [CrossRef] [PubMed]
- Tzouvelekis, A.; Paspaliaris, V.; Koliakos, G.; Ntolios, P.; Bouros, E.; Oikonomou, A.; Zissimopoulos, A.; Boussios, N.; Dardzinski, B.; Gritzalis, D.; et al. A prospective, non-randomized, no placebo-controlled, phase Ib clinical trial to study the safety of the adipose derived stromal cells-stromal vascular fraction in idiopathic pulmonary fibrosis. J. Transl Med. 2013, 11, 171. [Google Scholar] [CrossRef] [PubMed]
- Chambers, D.C.; Enever, D.; Ilic, N.; Sparks, L.; Whitelaw, K.; Ayres, J.; Yerkovich, S.T.; Khalil, D.; Atkinson, K.M.; Hopkins, P.M. A phase 1b study of placenta-derived mesenchymal stromal cells in patients with idiopathic pulmonary fibrosis. Respirology 2014, 19, 1013–1018. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Mollar, A.; Gay-Jordi, G.; Guillamat-Prats, R.; Closa, D.; Hernandez-Gonzalez, F.; Marin, P.; Burgos, F.; Martorell, J.; Sánchez, M.; Arguis, P.; et al. Safety and tolerability of alveolar type II cell transplantation in idiopathic pulmonary fibrosis. Chest 2016, 150, 533–543. [Google Scholar] [CrossRef] [PubMed]
- Glassberg, M.K.; Minkiewicz, J.; Toonkel, R.L.; Simonet, E.S.; Rubio, G.A.; DiFede, D.; Shafazand, S.; Khan, A.; Pujol, M.V.; LaRussa, V.F.; et al. Allogeneic human mesenchymal stem cells in patients with idiopathic pulmonary fibrosis via intravenous delivery (AETHER): A phase I safety clinical trial. Chest 2017, 151, 971–981. [Google Scholar] [CrossRef] [PubMed]
- Smith, A. A glossary for stem-cell biology. Nature 2006, 441, 1060. [Google Scholar] [CrossRef]
- Hawkins, F.; Kotton, D.N. Embryonic and induced pluripotent stem cells for lung regeneration. Ann. Am. Thorac. Soc. 2015, 12, 50–53. [Google Scholar] [CrossRef] [PubMed]
- Coraux, C.; Nawrocki-Raby, B.; Hinnrasky, J.; Kileztky, C.; Gaillard, D.; Dani, C.; Puchelle, E. Embryonic stem cells generate airway epithelial tissue. Am. J. Respir. Cell Mol. Biol. 2005, 32, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Asahara, T.; Murohara, T.; Sullivan, A.; Silver, M.; van der Zee, R.; Li, T.; Witzenbichler, B.; Schatteman, G.; Isner, J.M. Isolation of putative progenitor endothelial cells for angiogenesis. Science 1997, 275, 964–967. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, R.; Yajima, T.; Yamazaki, M.; Kanai, T.; Mukai, M.; Okamoto, S.; Ikeda, Y.; Hibi, T.; Inazawa, J.; Watanabe, M. Damaged epithelia regenerated by bone marrow-derived cells in the human gastrointestinal tract. Nat. Med. 2002, 8, 1011–1017. [Google Scholar] [CrossRef] [PubMed]
- Grove, J.E.; Lutzko, C.; Priller, J.; Henegariu, O.; Theise, N.D.; Kohn, D.B.; Krause, D.S. Marrow-derived cells as vehicles for delivery of gene therapy to pulmonary epithelium. Am. J. Respir. Cell Mol. Biol. 2002, 27, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Krause, D.S.; Theise, N.D.; Collector, M.I.; Henegariu, O.; Hwang, S.; Gardner, R.; Neutzel, S.; Sharkis, S.J. Multi-organ, multi-lineage engraftment by a single bone marrow-derived stem cell. Cell 2001, 105, 369–377. [Google Scholar] [CrossRef]
- Koç, O.N.; Day, J.; Nieder, M.; Gerson, S.L.; Lazarus, H.M.; Krivit, W. Allogeneic mesenchymal stem cell infusion for treatment of metachromatic leukodystrophy (MLD) and Hurler syndrome (MPS-IH). Bone Marrow Transplant. 2002, 30, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Friedenstein, A.J.; Petrakova, K.V.; Kurolesova, A.I.; Frolova, G.P. Heterotopic of bone marrow. Analysis of precursor cells for osteogenic and hematopoietic tissues. Transplantation 1968, 6, 230–247. [Google Scholar] [CrossRef] [PubMed]
- Keating, A. Mesenchymal Stromal Cells: New Directions. Cell Stem Cell 2012, 10, 709–716. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Liu, Y.; Han, Q.; Jia, M.; Liao, L.; Qi, M.; Zhao, R.C. Injured microenvironment directly guides the differentiation of engrafted Flk-1(+) mesenchymal stem cell in lung. Exp. Hematol. 2007, 35, 1466–1475. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, A. Mesenchymal stem cell delivery routes and fate. Int. J. Stem Cells 2008, 1, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Le Blanc, K.; Frassoni, F.; Ball, L.; Locatelli, F.; Roelofs, H.; Lewis, I.; Ichikado, K.; Ando, M.; Nakamura, H. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: A phase II study. Lancet 2008, 371, 1579–1586. [Google Scholar] [CrossRef]
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef] [PubMed]
- Geoghegan, E.; Byrnes, L. Mouse induced pluripotent stem cells. Int. J. Dev. Biol. 2008, 52, 1015–1022. [Google Scholar] [CrossRef] [PubMed]
- Jacob, A.; Morley, M.; Hawkins, F.; McCauley, K.B.; Jean, J.C.; Heins, H.; Na, C.L.; Weaver, T.E.; Vedaie, M.; Hurley, K.; et al. Differentiation of human pluripotent stem cells into functional lung alveolar epithelial cells. Cell Stem Cell 2017, 21, 472–488. [Google Scholar] [CrossRef] [PubMed]
- Collins, J.J.P.; Thebaud, B. Lung mesenchymal stromal cells in development and disease: To serve and protect? Antioxid. Redox. Signal 2014, 21, 1849–1862. [Google Scholar] [CrossRef] [PubMed]
- Smirnova, N.F.; Schamberger, A.C.; Nayakanti, S.; Hatz, R.; Behr, J.; Eickelberg, O. Detection and quantification of epithelial progenitor cell populations in human healthy and IPF lungs. Respir. Res. 2016, 16, 83. [Google Scholar] [CrossRef] [PubMed]
- Rock, J.R.; Onaitis, M.W.; Rawlins, E.L.; Lu, Y.; Clark, C.P.; Xue, Y.; Randell, S.H.; Hogan, B.L. Basal cells as stem cells of the mouse trachea and human airway epithelium. Proc. Natl. Acad. Sci. USA 2009, 106, 12771–12775. [Google Scholar] [CrossRef] [PubMed]
- Tata, P.R.; Mou, H.; Pardo-Saganta, A.; Zhao, R.; Prabhu, M.; Law, B.M.; Vinarsky, V.; Cho, J.L.; Breton, S.; Sahay, A.; et al. Dedifferentiation of committed epithelial cells into stem cells in vivo. Nature 2013, 503, 218–223. [Google Scholar] [CrossRef] [PubMed]
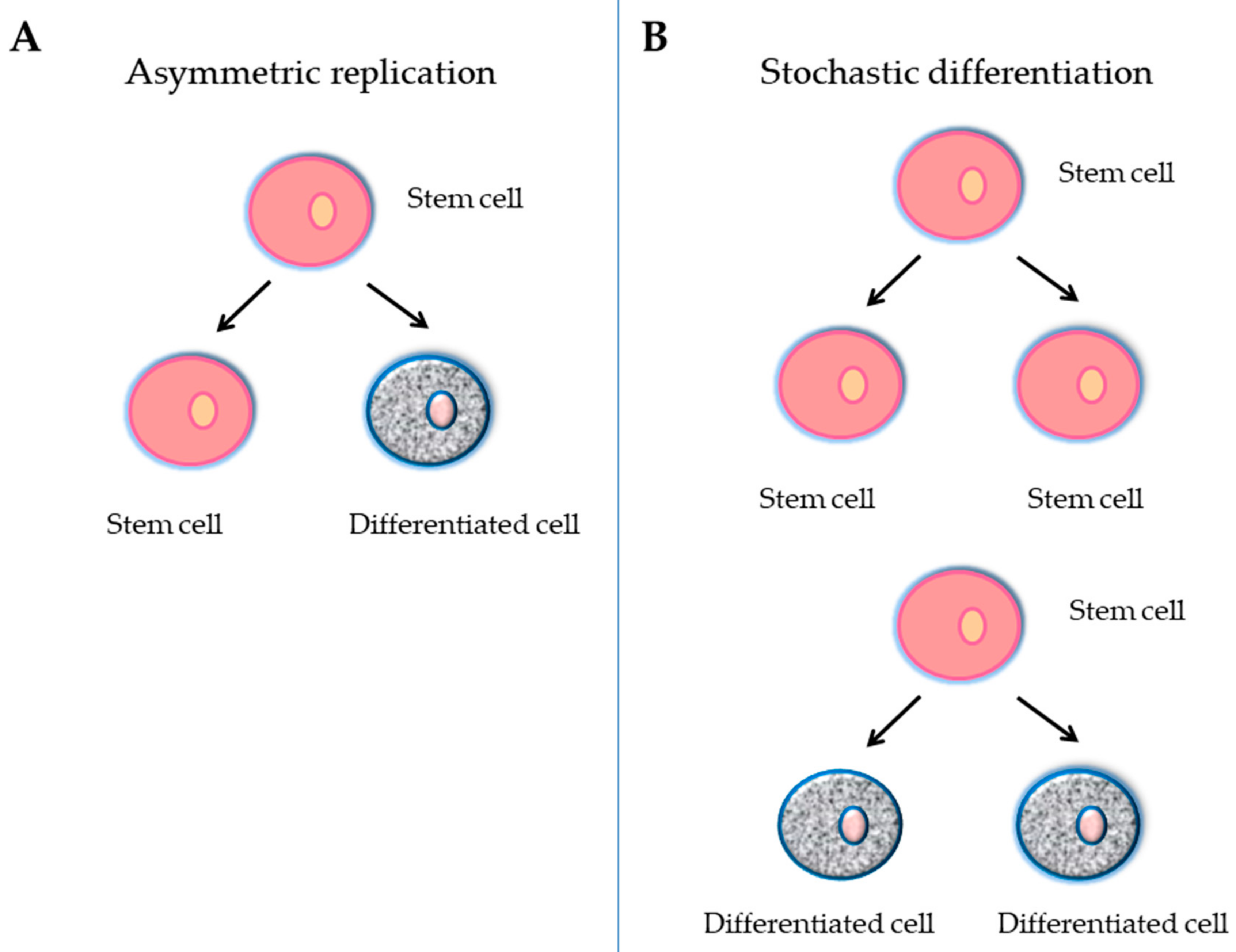
- Teixeira, V.H.; Nadarajan, P.; Graham, T.A.; Pipinikas, C.P.; Brown, J.M.; Falzon, M.; Nye, E.; Poulsom, R.; Lawrence, D.; Wright, N.A.; et al. Stochastic homeostasis in human airway epithelium is achieved by neutral competition of basal cell progenitors. eLife 2013, 22, e00966. [Google Scholar] [CrossRef] [PubMed]
- O’Koren, E.G.; Hogan, B.L.; Gunn, M.D. Loss of basal cells precedes bronchiolitis obliterans-like pathological changes in a murine model of chlorine gas inhalation. Am. J. Respir. Cell Mol. Biol. 2013, 49, 788–797. [Google Scholar] [CrossRef] [PubMed]
- Rawlins, E.L.; Okubo, T.; Xue, Y.; Brass, D.M.; Auten, R.L.; Hasegawa, H.; Wang, F.; Hogan, B.L. The role of Scgb1a1+ Clara cells in the long-term maintenance and repair of lung airway, but not alveolar, epithelium. Cell Stem Cell 2009, 4, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Giangreco, A.; Reynolds, S.D.; Stripp, B.R. Terminal bronchioles harbor a unique airway stem cell population that localizes to the bronchoalveolar duct junction. Am. J. Pathol. 2002, 161, 173–182. [Google Scholar] [CrossRef]
- Reynolds, S.D.; Giangreco, A.; Hong, K.U.; McGrath, K.E.; Ortiz, L.A.; Stripp, B.R. Airway injury in lung disease pathophysiology: Selective depletion of airway stem and progenitor cell pools potentiates lung inflammation and alveolar dysfunction. Am. J. Physiol. Lung Cell. Mol. Physiol. 2004, 287, L1256–L1265. [Google Scholar] [CrossRef] [PubMed]
- Hong, K.U.; Reynolds, S.D.; Giangreco, A.; Hurley, C.M.; Stripp, B.R. Clara cell secretory protein-expressing cells of the airway neuroepithelial body microenvironment include a label-retaining subset and are critical for epithelial renewal after progenitor cell depletion. Am. J. Respir. Cell. Mol. Biol. 2001, 24, 671–681. [Google Scholar] [CrossRef] [PubMed]
- Barkauskas, C.E.; Cronce, M.J.; Rackley, C.R.; Bowie, E.J.; Keene, D.R.; Stripp, B.R.; Randell, S.H.; Noble, P.W.; Hogan, B.L. Type 2 alveolar cells are stem cells in adult lung. J. Clin. Investig. 2013, 123, 3025–3036. [Google Scholar] [CrossRef] [PubMed]
- Desai, T.J.; Brownfield, D.G.; Krasnow, M.A. Alveolar progenitor and stem cells in lung development, renewal and cancer. Nature 2014, 507, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Rock, J.R.; Barkauskas, C.E.; Cronce, M.J.; Xue, Y.; Harris, J.R.; Liang, J.; Noble, P.W.; Hogan, B.L. Multiple stromal populations contribute to pulmonary fibrosis without evidence for epithelial to mesenchymal transition. Proc. Natl. Acad. Sci. USA 2011, 27, E1475–E1483. [Google Scholar] [CrossRef] [PubMed]
- Treutlein, B.; Brownfield, D.G.; Wu, A.R.; Neff, N.F.; Mantalas, G.L.; Espinoza, F.H.; Desai, T.J.; Krasnow, M.A.; Quake, S.R. Reconstructing lineage hierarchies of the distal lung epithelium using single-cell RNA-seq. Nature 2014, 15, 371–375. [Google Scholar] [CrossRef] [PubMed]
- Frank, D.B.; Peng, T.; Zepp, J.A.; Snitow, M.; Vincent, T.L.; Penkala, I.J.; Cui, Z.; Herriges, M.J.; Morley, M.P.; Zhou, S.; et al. Emergence of a wave of wnt signaling that regulates lung alveologenesis by controlling epithelial self-renewal and differentiation. Cell Rep. 2016, 22, 2312–2325. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.; Soh, B.S.; Yin, L.; Hu, G.; Chen, Q.; Choi, H.; Han, J.; Chow, V.T.; Chen, J. Differentiation of club cells to alveolar epithelial cells in vitro. Sci Rep. 2017, 7, 41661. [Google Scholar] [CrossRef] [PubMed]
- McQualter, J.L.; Brouard, N.; Williams, B.N.; Baird, B.N.; Sims-Lucas, S.; Yuen, K.; Nilsson, S.K.; Simmons, P.J.; Bertoncello, I. Endogenous fibroblastic progenitor cells in the adult mouse lung are highly enriched in the SCA-1 positive cell fraction. Stem Cells 2009, 27, 623–633. [Google Scholar] [CrossRef] [PubMed]
- Summer, R.; Fitzsimmons, K.; Dwyer, D.; Murphy, J.; Fine, A. Isolation of an adult mouse lung mesenchymalprogenitor cell population. Am. J. Respir. Cell Mol. Biol. 2007, 37, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Hegab, A.E.; Kubo, H.; Fujino, N.; Suzuki, T.; He, M.; Kato, H.; Yamaya, M. Isolation and characterization of murine multipotent lung stem cells. Stem Cells Dev. 2010, 19, 523–536. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, S.D.; Shen, H.; Reynolds, P.R.; Betsuyaku, T.; Pilewski, J.M.; Gambelli, F.; di Giuseppe, M.; Ortiz, L.A.; Stripp, B.R. Molecular and functional properties of lung SP cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 2007, 292, L972–L983. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.; Helm, K.; Ruegg, P.; Varella-Garcia, M.; Burnham, E.; Majka, S. Adult lung side population cells have mesenchymal stem cell potential. Cytotherapy 2008, 10, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Jarvinen, L.; Badri, L.; Wettlaufer, S.; Ohtsuka, T.; Standiford, T.J.; Toews, G.B.; Pinsky, D.J.; Peters-Golden, M.; Lama, V.N. Lung resident mesenchymal stem cells isolated from human lung allografts inhibit T cell proliferation via a soluble mediator. J. Immunol. 2008, 181, 4389–4396. [Google Scholar] [CrossRef] [PubMed]
- Lama, V.N.; Smith, L.; Badri, L.; Flint, A.; Andrei, A.C.; Murray, S.; Wang, Z.; Liao, H.; Toews, G.B.; Krebsbach, P.H.; et al. Evidence for tissueresident mesenchymal stem cells in human adult lung from studies of transplanted allografts. J. Clin. Investig. 2007, 117, 989–996. [Google Scholar] [CrossRef] [PubMed]
- Hennrick, K.T.; Keeton, A.G.; Nanua, S.; Kijek, T.G.; Goldsmith, A.M.; Sajjan, U.S.; Bentley, J.K.; Lama, V.N.; Moore, B.B.; Schumacher, R.E.; et al. Lung cells from neonates show a mesenchymal stem cell phenotype. Am. J. Respir. Crit. Care Med. 2007, 175, 1158–1164. [Google Scholar] [CrossRef] [PubMed]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- McQualter, J.; Yuen, K.; Williams, B.; Bertoncello, I. Evidence of an epithelial stem/progenitor cell hierarchy in the adult mouse lung. Proc. Natl. Acad. Sci. USA 2010, 107, 1414–1419. [Google Scholar] [CrossRef] [PubMed]
- Chua, F.; Gauldie, J.; Laurent, G.J. Pulmonary fibrosis: Searching for model answers. Am. J. Respir. Cell Mol. Biol. 2005, 33, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Moeller, A.; Ask, K.; Warburton, D.; Gauldie, J.; Kolb, M. The bleomycin animal model: A useful tool to investigate treatment options for idiopathic pulmonary fibrosis? Int. J. Biochem. Cell Biol. 2008, 40, 362–382. [Google Scholar] [CrossRef] [PubMed]
- Kropski, J.A.; Blackwell, T.S.; Loyd, J.E. The genetic basis of idiopathic pulmonary fibrosis. Eur. Respir. J. 2015, 45, 1717–1727. [Google Scholar] [CrossRef] [PubMed]

| Study | Cell Source | Dose and Route of Administration | Time of Cell Transplantation after Injury | Study Design | Results | Ref. |
|---|---|---|---|---|---|---|
| Preclinical | BM-MSCs | 5 × 106/mouse, IV | Immediately or 7 days after BLM instillation | BLM-induced fibrosis in mice | Reduced inflammation and collagen deposition | [6] |
| Preclinical | BM-MSCs | 2.5 × 106/rat, IV | Immediately or 7 days after BLM instillation | BLM-induced fibrosis in rats | Reduced collagen deposition and reduced oxidative stress | [7] |
| Preclinical | BM-MSCs | 5 × 105/mouse, IV | Immediately after BLM instillation | BLM-induced fibrosis in mice | MSCs protect lung tissue, blocking the pro-inflammatory cytokines TNF-α and IL-1 | [8] |
| Preclinical | BM-MSCs | 5 × 106/mouse, IV | 6 h after BLM instillation | Myelosuppression and bone-marrow MSC administration. BLM induction in mice | Suppression of inflammation and production of reparative growth factors | [9] |
| Preclinical | BM-MSCs HGF knockdown BM-MSCs | 5 × 104/g body weight, IV | 6–8 h after BLM administration or 9 days later | BLM-induced fibrosis in mice | Reduced fibrosis, reduced levels of interleukin-1b and apoptosis, increased levels of HGF; these effects were mediated in part by the HGF | [10] |
| Preclinical | BM-HSCs MSCs MSCs: transduced (MSCs-KGF) MSCs: non-transduced (MSCs-GFP) HSCs: transduced HSCs with HSCs-KGF HSCs: non-transduced (HSCs-GFP) | 0.5 × 106/mouse, IV for MSCs and 0.6 × 106, IV for BM-HSC transplantation | MSCs: 8 h after BLM instillation, second dose 3 days after HSCs: 7 weeks after BM-HSC transplantation BLM instillation + doxycycline | BLM-induced fibrosis in mice and BM-MSC administration. BM-HSC transplantation and BLM-induced fibrosis | MSCs and HSCs reduced collagen disposition. Transduced HSCs attenuated histological damage through endogenous ATII cell proliferation induced by KGF. | [11] |
| Preclinical | Human BM-MSCs | 5 × 105/mouse, IV | 24 h after BLM instillation | BLM-induced fibrosis in mice | Reduction of oxidative stress, endoplasmic reticulum stress, and TGF-β1 produced by alveolar cells | [12] |
| Preclinical | Human BM-MSCs | 5 × 106/mouse, IV | 1, 2, 3, or 4 days after BLM instillation | BLM-induced fibrosis in immunodeficient NOD/SCID and NOD/SCID/β2 microglobulin (β2M) null mice | Low levels of BM-MSCs engraft | [13] |
| Preclinical | Hypoxia-preconditioned BM-MSCs | 5 × 105/mouse, intratracheal | 3 days after BLM instillation | BLM-induced fibrosis in mice | Reduction of inflammation and fibrosis and improved pulmonary function | [14] |
| Preclinical | Oncostatin M preconditioned BM-MSCs Oncostatin M preconditioned HGF knockdown BM-MSCs | 2 × 105/mouse, intratracheal | 3 days after BLM instillation | BLM-induced fibrosis in mice | Reduction of inflammation and fibrosis factors and improved pulmonary function | [15] |
| Preclinical | BM-MSCs | 106/rat, IV | 4 days after BLM instillation | BLM-induced fibrosis in rats | Reduction of inflammation and fibrosis factors (IL-1β, TGF-β, VEGF, IL-6, TNF-α, and NOS) | [16] |
| Preclinical | BM-MSCs Amnion-MSCs Human amniotic epithelial cells (hAECs) | 1 × 106/mouse, IV | 10 days after BLM (72 h after the second BLM dose) | BLM-induced fibrosis in mice (2 repeated doses at 0 days and 7 days) | Amnion-MSCs, BM-MSCs, and hAECs exert a wide range of anti-inflammatory effects. Among all cells, amnion-MSCs were more effective, reducing fibrosis and TGF-β, and increasing MMP-9 activity, GM-CSF secretion and induction of IL-1RA. | [17] |
| Preclinical | BM-MSCs transfected with HGF | 3 × 106/rat, intratracheal | 7 days after BLM instillation | BLM-induced fibrosis in rats | Reduced collagen deposition and reduced fibrosis in Ashcroft score | [18] |
| Preclinical | Human BM-MSCs overexpressing microRNAs let-7d or miR-154 | 5 × 104/mouse, IV | 7 days after BLM instillation | BLM-induced fibrosis in mice | B-MSCs overexpressing let-7d revealed shifts in animal weight loss as well as reduced collagen deposition and decreased CD45-positive cells | [19] |
| Preclinical | Young-donor adipose-MSCs Old-donor adipose-MSCs | 5 × 105/mouse, IV | 24 h after BLM instillation | BLM-induced fibrosis in old mice >22 weeks old | Young adipose MSCs showed greater effect on decreased fibrosis, (MMP)-2 activity, oxidative stress, and markers of apoptosis | [20] |
| Preclinical | Adipose-MSCs | 2.5 × 104 or 2.5 × 105/mouse, IV | 7 days after BLM instillation | BLM-induced fibrosis in mice | Inhibition of both pulmonary inflammation and fibrosis in a dose-dependent manner | [21] |
| Preclinical | Human adipose-MSCs | 3 × 105/mouse, IP | Were simultaneously administered in the latter 2 months of the 4-month BLM regimen at the same time of BLM | Biweekly administration of a total of 8 doses of BLM during 4 months in mice | Reduced epithelial cell hyperplasia and reduced inflammatory cell infiltration and fibrosis. Inhibition of apoptosis in epithelial cells and in the expression of TGF-β) | [22] |
| Preclinical | Human placenta-MSCs Murine placenta-MSCs | 4 × 106/mouse, IP 1 × 106/mouse, IV or intratracheal | 15 min after intratracheal BLM instillation | BLM-induced fibrosis in mice | Reduction in neutrophil infiltration and in the severity of BLM-induced lung fibrosis | [23] |
| Preclinical | Human placenta-MSCs | 1 × 105/mouse, IV | 3 days after BLM instillation | BLM-induced fibrosis in MyD88-deficient mice | Reduced collagen deposition, MyD88 and TGF-β signalling activation, and production of pro-fibrotic cytokines | [24] |
| Preclinical | Human umbilical-MSCs | 1 × 106/mouse, IV | 24 h after BLM instillation | BLM-induced fibrosis in mice | Inhibition of inflammation and fibrosis and down-regulation of lung cytokine and TIMP expression while up-regulating MMPs | [25] |
| Preclinical | Amnion stem cells (ASCs) | 5 × 106/mouse, IV | 2 h after BLM instillation, 0 or 14 days after BLM instillation | BLM-induced fibrosis in mice | Inhibition of collagen deposition, preservation of pulmonary function, and decreased CCL2 expression on either day 0 or day 14 | [26] |
| Preclinical | iPSCs iPSC conditioned medium | 2 × 106/mouse, IV | 24 h after BLM instillation. | BLM-induced fibrosis in mice | Decreased myeloperoxidase activity and neutrophil infiltration. Rescue of pulmonary function. Reduced collagen deposition | [27] |
| Preclinical | iPSCs | 2 × 105/mouse, IV | 24 h after BLM instillation | BLM-induced fibrosis in mice | Suppression of inflammatory responses, the TGF-b1/Smad2/3 pathway, and EMT | [28] |
| Preclinical | iPSCs derived to ATII cells | 5 × 105/mouse, intratracheal | 24 h after BLM instillation. | BLM-induced fibrosis in mice | Reduced lung inflammation and collagen deposition | [29] |
| Preclinical | Human- ESCs derived to epithelial lineage-specific cells (ATII, ATI and club cells) | 105 Human-ESC, intratracheal | 7 days after BLM instillation | BLM-induced fibrosis in mice | Reduced collagen and increased levels of ATI and ATII and progenitors in the lungs | [30] |
| Preclinical | Lung resident-MSCs | 0.15 × 106 or 0.25 × 106/mouse, IV | Immediately after BLM instillation | BLM-induced fibrosis in mice | Decreased pulmonary damage and mitigation of the development of PAH. Decreased lymphocyte and granulocyte infiltration | [31] |
| Preclinical | ATII cells | 2.5 × 106/rat, intratracheal | 3, 7 or 14 days after BLM | BLM-induced fibrosis in rats | Reduced collagen deposition and reduced severity of pulmonary fibrosis | [32] |
| Preclinical | ATII cells | 2.5 × 106/rat, 14 days after BLM | 14 days after BLM | BLM-induced fibrosis in rats | Restoration of lung surfactant protein levels | [33] |
| Preclinical | LSCs formed basically by ATI, ATI and club cells | 5 × 106 LSC/rat, IV | At the same moment of intratracheal BLM instillation | BLM-induced fibrosis in rats | LSCs attenuated the progression and severity of fibrosis, decreased apoptosis, protected alveolar structures, and increased angiogenesis | [34] |
| Clinical | Autologous adipose-MSCs | 0.5 × 106 cells/kg of body weight, intratracheal | Mild to moderate IPF patients | Phase1b, prospective, non-randomized, non-placebo (n = 14) | Adipose-MSCs were safe and no deterioration of functional parameters and indicators of quality of life were observed | [35] |
| Clinical | Heterologous placenta-MSCs | 1 × 106 or 2 × 10 6 cells/kg of body weight, IV | Mild to moderate IPF patients | Phase 1b, non-randomized, non-placebo, dose escalation study (n = 8) | Placenta-MSCs were safe, with no evidence of worsening fibrosis | [36] |
| Clinical | Heterologous ATII cells | 1000 to 1200 × 106 cells/patient, intratracheal | Mild to moderate IPF patients | Clinical study, non-randomized, non-placebo (n = 16) | ATII cells were safe and well tolerated, and halted disease progression | [37] |
| Clinical | Heterologous BM-MSCs | 20, 100, or 200 × 106 cells/patient, IV | Mild to moderate IPF patients | phase 1b, non-randomized, non-placebo, dose escalation study (n = 9) | BM-MSCs were safe, no evidence of worsening fibrosis | [38] |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serrano-Mollar, A. Cell Therapy in Idiopathic Pulmonary Fibrosis†. Med. Sci. 2018, 6, 64. https://doi.org/10.3390/medsci6030064
Serrano-Mollar A. Cell Therapy in Idiopathic Pulmonary Fibrosis†. Medical Sciences. 2018; 6(3):64. https://doi.org/10.3390/medsci6030064
Chicago/Turabian StyleSerrano-Mollar, Anna. 2018. "Cell Therapy in Idiopathic Pulmonary Fibrosis†" Medical Sciences 6, no. 3: 64. https://doi.org/10.3390/medsci6030064
APA StyleSerrano-Mollar, A. (2018). Cell Therapy in Idiopathic Pulmonary Fibrosis†. Medical Sciences, 6(3), 64. https://doi.org/10.3390/medsci6030064




