Nitric Oxide: The Missing Factor in COVID-19 Severity? †
Abstract
1. Prologue
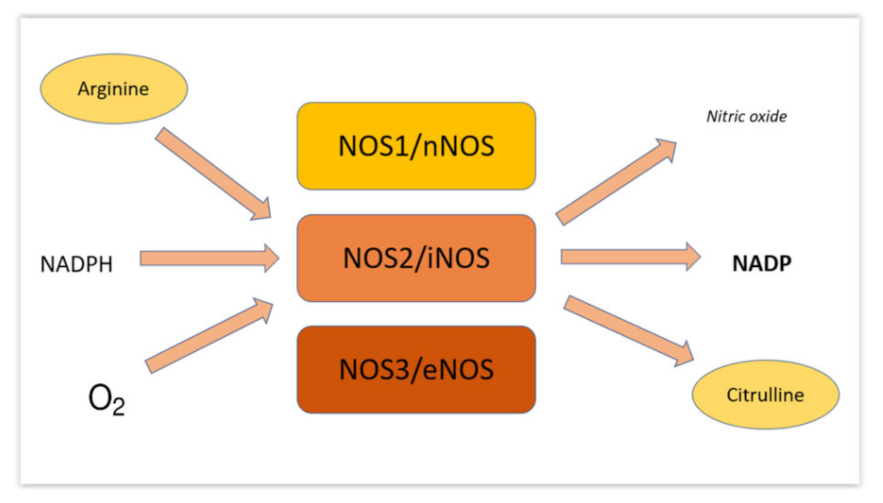
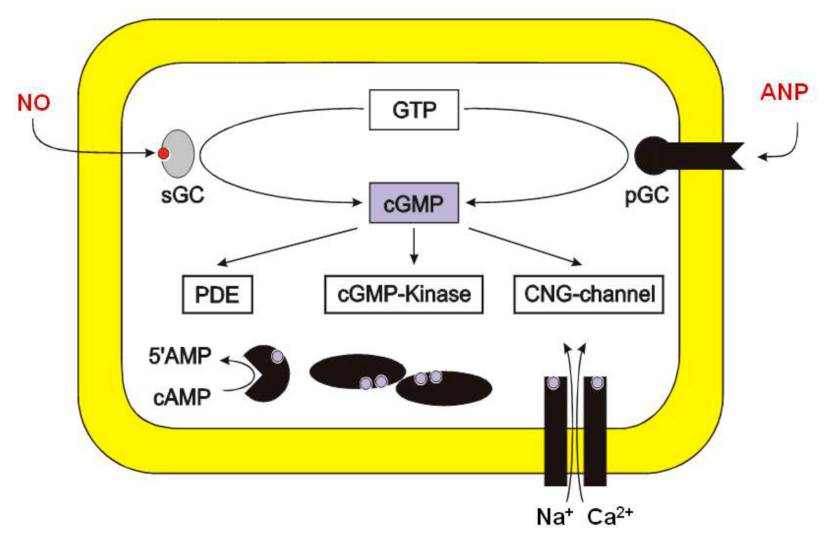
2. About Nitric Oxide
2.1. Nitric Oxide and COVID-19
2.2. Nitric Oxide and Age
2.3. Comorbidities of COVID-19 and Nitric Oxide
2.3.1. Nitric Oxide and Hypertension/Cardiovascular Disease
2.3.2. Nitric Oxide and Diabetes
2.3.3. Nitric Oxide and Metabolic Disease/Obesity
2.3.4. Nitric Oxide and Pregnancy
2.3.5. Nitric Oxide and Immune Suppression
2.3.6. Nitric Oxide and COPD
2.3.7. Nitric Oxide and Asthma
2.3.8. Nitric Oxide and Smoking
3. Severe COVID-19 Clinical Outcomes
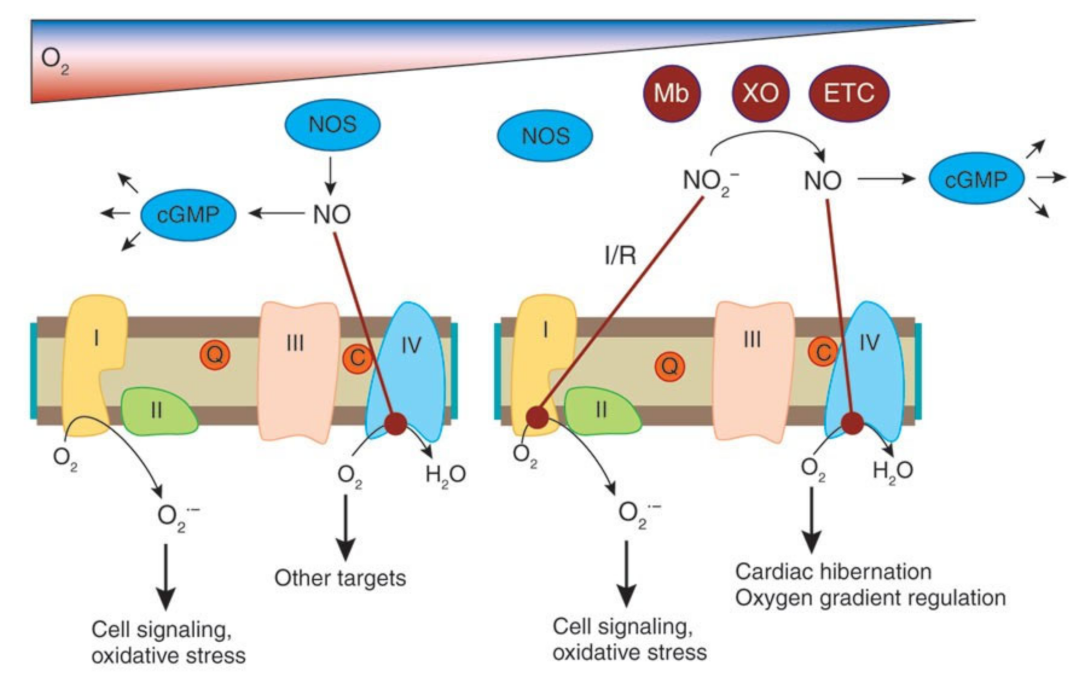
3.1. Is COVID-19 Simply a Case of Systematic NO Depletion?
3.2. Nitric Oxide and COVID-19 Patient Outcomes: What Does the Evidence Suggest?
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Chhetri, B.; Vamsi, D.K.K.; Sanjeevi, C.B. Optimal Control Studies on Age Structural Modeling of COVID-19 in Presence of Saturated Medical Treatment of Holling Type III. arXiv 2020, arXiv:2012.06719. Available online: https://arxiv.org/abs/2012.06719 (accessed on 5 June 2021).
- Zaim, S.; Chong, J.H.; Sankaranarayanan, V.; Harky, A. COVID-19 and Multiorgan Response. Curr. Probl. Cardiol. 2020, 45, 100618. [Google Scholar] [CrossRef]
- Perico, L.; Benigni, A.; Casiraghi, F.; Ng, L.F.P.; Renia, L.; Remuzzi, G. Immunity, endothelial injury and complement-induced coagulopathy in COVID-Nat. Rev. Nephrol. 2021, 17, 46–64. [Google Scholar] [CrossRef] [PubMed]
- Leung, T.Y.M.; Chan, A.Y.L.; Chan, E.W.; Chan, V.K.Y.; Chui, C.S.L.; Cowling, B.J. Short and potential long-term adverse health outcomes of COVID-19: A rapid review. Emerg Microbes Infect. 2020, 9, 2190–2199. [Google Scholar] [CrossRef]
- Post-COVID Conditions. Available online: https://www.cdc.gov/coronavirus/2019-ncov/long-term-effects.html (accessed on 5 June 2021).
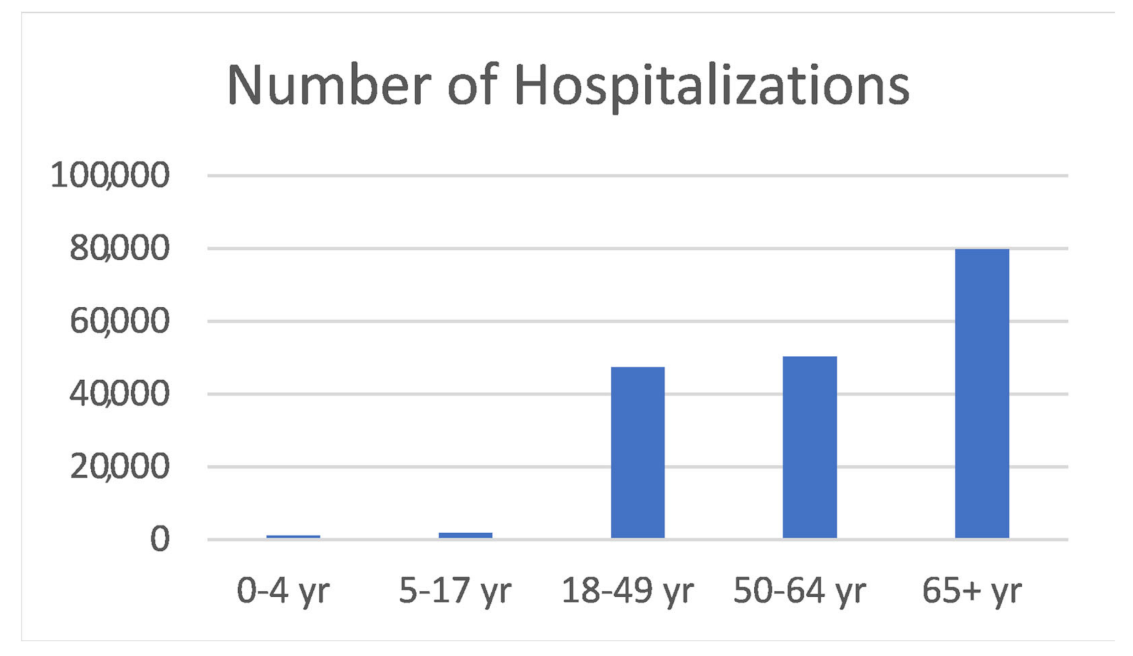
- Laboratory-Confirmed COVID-19-Associated Hospitalizations. Available online: https://gis.cdc.gov/grasp/covidnet/COVID19_5.html (accessed on 5 June 2021).
- Akil, L.; Ahmad, H.A. Relationships between Obesity and Cardiovascular Diseases in Four Southern States and Colorado. JHCPU 2011, 22, 61–72. [Google Scholar] [CrossRef]
- Jiang, S.-Z.; Lu, W.; Zong, X.-F.; Ruan, H.-Y.; Liu, Y. Obesity and hypertension. Exp. Med. 2016, 12, 2395–2399. [Google Scholar] [CrossRef] [PubMed]
- Barnes, A.S. The epidemic of obesity and diabetes: Trends and treatments. Tex. Heart Inst. J. 2011, 38, 142–144. [Google Scholar] [PubMed]
- AlGhatrif, M.; Cingolani, O.; Lakatta, E.G. The Dilemma of Coronavirus Disease 2019, Aging, and Cardiovascular Disease. JAMA Cardiol. 2020, 5, 747. [Google Scholar] [CrossRef] [PubMed]
- COVID-19-related deaths not increased with ACE inhibitor or ARB use. React. Wkly. 2020, 1835, 5. [CrossRef]
- Ostojic, S.M.; Milovancev, A.; Drid, P.; Nikolaidis, A. Oxygen saturation improved with nitrate-based nutritional formula in patients with COVID. J. Int. Med. Res. 2021, 49, 3000605211012380. [Google Scholar] [CrossRef] [PubMed]
- A Gruetter, C.; Barry, B.K.; McNamara, D.B.; Gruetter, D.Y.; Kadowitz, P.J.; Ignarro, L. Relaxation of bovine coronary artery and activation of coronary arterial guanylate cyclase by nitric oxide, nitroprusside and a carcinogenic nitrosoamine. J. Cycl. Nucleotide Res. 1979, 5, 211–224. [Google Scholar]
- Förstermann, U.; Sessa, W.C. Nitric oxide synthases: Regulation and function. Eur. Heart J. 2012, 33, 829–837. [Google Scholar] [CrossRef]
- Martin, M.D.J.A.V.M.M.J.; Jimenez, M.D.; Motilva, V. New Issues about Nitric Oxide and its Effects on the Gastrointestinal Tract. Curr. Pharm. Des. 2001, 7, 881–908. [Google Scholar] [CrossRef]
- Ho, J.J.D.; Man, H.S.J.; Marsden, P.A. Nitric oxide signaling in hypoxia. J. Mol. Med. 2012, 90, 217–231. [Google Scholar] [CrossRef] [PubMed]
- Larsen, F.J.; Ekblom, B.; Sahlin, K.; Lundberg, J.O.; Weitzberg, E. Effects of Dietary Nitrate on Blood Pressure in Healthy Volunteers. N. Engl. J. Med. 2006, 355, 2792–2793. [Google Scholar] [CrossRef]
- Larsen, F.; Weitzberg, E.; Lundberg, J.; Ekblom, B. Effects of dietary nitrate on oxygen cost during exercise. Acta Physiol. 2007, 191, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Kovačević, P.; Dragić, S.; Vidović, J.; Zeljković, S.; Momčičević, D.; Rajkovača, Z.; Kovačević, T. Serum levels of nitric oxide as a predictor of survival in acute respiratory distress syndrome caused by H1N1 pneumonia? Signa Vitae A J. Intensiv. Care Emerg. Med. 2015, 10, 63. [Google Scholar] [CrossRef][Green Version]
- Murphy, A.W.; Platts-Mills, T.A.; Lobo, M.; Hayden, F. Respiratory nitric oxide levels in experimental human influenza. Chest 1998, 114, 452–456. [Google Scholar] [CrossRef]
- Akerström, S.; Mousavi-Jazi, M.; Klingström, J.; Leijon, M.; Lundkvist, A.; Mirazimi, A. Nitric Oxide Inhibits the Replication Cycle of Severe Acute Respiratory Syndrome Coronavirus. J. Virol. 2005, 79, 1966–1969. [Google Scholar] [CrossRef]
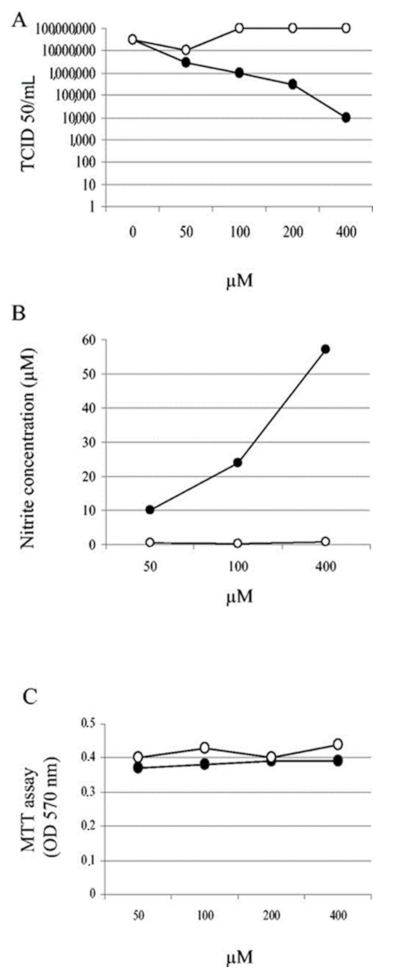
- Akaberi, D.; Krambrich, J.; Ling, J.; Luni, C.; Hedenstierna, G.; Järhult, J.D.; Lennerstrand, J.; Lundkvist, A. Mitigation of the replication of SARS-CoV-2 by nitric oxide in vitro. Redox Biol. 2020, 37, 101734. [Google Scholar] [CrossRef]
- Alvarez, R.A.; Berra, L.; Gladwin, M.T. Home Nitric Oxide Therapy for COVID. Am. J. Respir. Crit. Care Med. 2020, 202, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Ichinose, F.; Roberts, J.D., Jr.; Zapol, W.M. Inhaled Nitric Oxide a Selective Pulmonary Vasodilator: Current Uses and Therapeutic Potential. Circulation 2004, 109, 3106–3111. [Google Scholar] [CrossRef]
- Weinberger, B.; Djerad, A.; Monier, C.; Houzé, P.; Borron, S.W.; Lefauconnier, J.-M.; Baud, F.J. The Toxicology of Inhaled Nitric Oxide. Toxicol. Sci. 2001, 59, 5–16. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, M.; Hess, D.; Kacmarek, R.M.; Ritz, R.; Hurford, W.E. Nitrogen Dioxide Production during Mechanical Ventilation with Nitric Oxide in Adults. Anesthesiology 1995, 82, 1246–1254. [Google Scholar] [CrossRef]
- Petit, P.C.; Fine, D.H.; Vásquez, G.B.; Gamero, L.; Slaughter, M.S.; Dasse, K.A. The Pathophysiology of Nitrogen Dioxide During Inhaled Nitric Oxide Therapy. ASAIO J. 2017, 63, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Prakash, A.; Kaur, S.; Kaur, C.; Prabha, P.K.; Bhatacharya, A.; Sarma, P.; Medhi, B. Efficacy and safety of inhaled nitric oxide in the treatment of severe/critical COVID-19 patients: A systematic review. Indian J. Pharm. 2021, 53, 236–243. [Google Scholar]
- Smith, A.R.; Visioli, F.; Frei, B.; Hagen, T.M. Age-related changes in endothelial nitric oxide synthase phosphorylation and nitric oxide dependent vasodilation: Evidence for a novel mechanism involving sphingomyelinase and ceramide-activated phosphatase 2A. Aging Cell. 2006, 5, 391–400. [Google Scholar] [CrossRef]
- Cau, S.B.D.A.; Carneiro, F.S.; Tostes, R.C. Differential Modulation of Nitric Oxide Synthases in Aging: Therapeutic Opportunities. Front. Physiol. 2012, 3, 218. [Google Scholar] [CrossRef]
- Mccarty, M.F. Optimizing endothelial nitric oxide activity may slow endothelial aging. Med. Hypotheses 2004, 63, 719–723. [Google Scholar] [CrossRef]
- Glynn, J.R.; Moss, P.A.H. Systematic analysis of infectious disease outcomes by age shows lowest severity in school-age children. Sci. Data 2020, 7, 329. [Google Scholar] [CrossRef]
- Bryan, N.S.; Grisham, M.B. Methods to detect nitric oxide and its metabolites in biological samples. Free Radic. Biol. Med. 2007, 43, 645–657. [Google Scholar] [CrossRef]
- Lauer, T.; Preik, M.; Rassaf, T.; Strauer, B.E.; Deussen, A.; Feelisch, M.; Kelm, M. Plasma nitrite rather than nitrate reflects regional endothelial nitric oxide synthase activity but lacks intrinsic vasodilator action. Proc. Natl. Acad. Sci. USA 2001, 98, 12814–12819. [Google Scholar] [CrossRef]
- Ghasemi, A.; Zahediasl, S.; Syedmoradi, L.; Azizi, F. Association between serum nitric oxide metabolites and hypertension in a general population. Int. Angiol. 2011, 4, 380–387. [Google Scholar]
- Li, D.; Nishi, S.K.; Jovanovski, E.; Zurbau, A.; Komishon, A.; Mejia, S.B.; Khan, T.A.; Sievenpiper, J.L.; Milicic, D.; Jenkins, A.; et al. Repeated administration of inorganic nitrate on blood pressure and arterial stiffness. J. Hypertens. 2020, 38, 2122–2140. [Google Scholar] [CrossRef] [PubMed]
- Leuenberger, U.A.; Brubaker, D.; Quraishi, S.; Hogeman, C.S.; Imadojemu, V.A.; Gray, K.S. Effects of intermittent hypoxia on sympathetic activity and blood pressure in humans. Auton. Neurosci. 2005, 121, 87–93, Erratum in: Auton. Neurosci. 2014, 183, 120. [Google Scholar] [CrossRef]
- Kulkarni, S.; O’Farrell, I.; Erasi, M.; Kochar, M.S. Stress and hypertension. WMJ 1998, 11, 34. [Google Scholar]
- Naseem, K.M. The role of nitric oxide in cardiovascular diseases. Mol. Asp. Med. 2005, 26, 33–65. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, F.D.; Whelton, P.K. High Blood Pressure and Cardiovascular Disease. Hypertension 2020, 75, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Green, S.J. Covid-19 accelerates endothelial dysfunction and nitric oxide deficiency. Microbes Infect. 2020, 22, 149–150. [Google Scholar] [CrossRef]
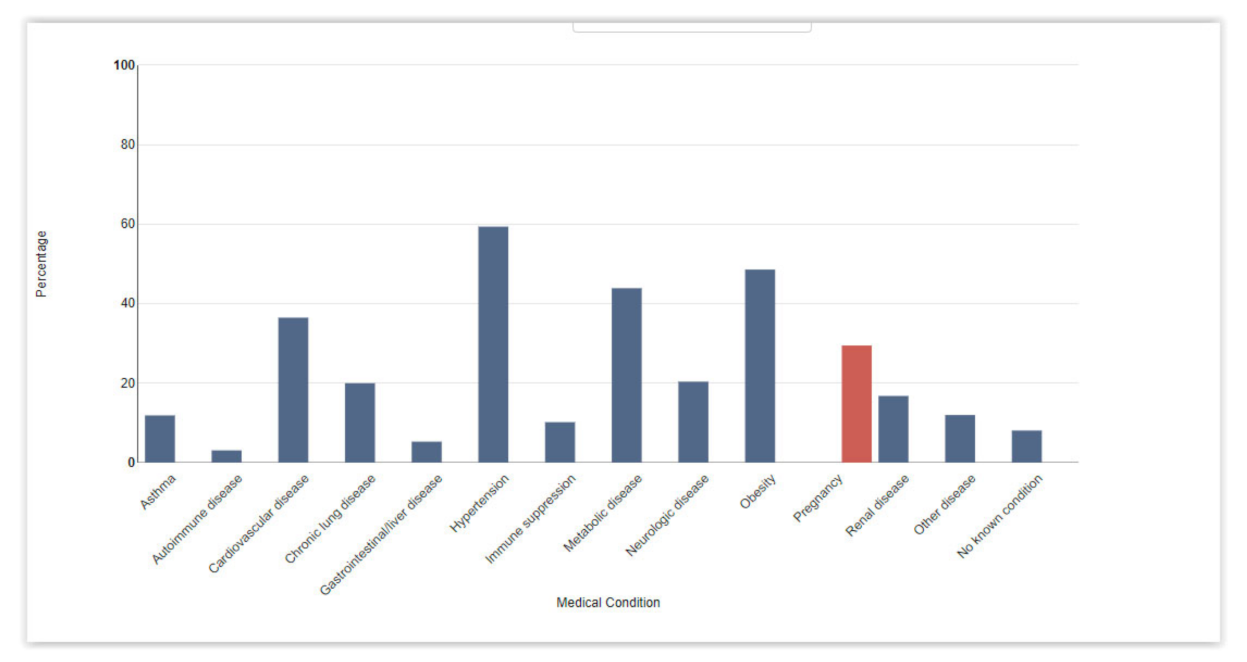
- Centers for Disease Control and Prevention. People with Certain Medical Conditions. Available online: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html (accessed on 12 July 2021).
- Eckel, R.H.; Kahn, S.E.; Ferrannini, E.; Goldfine, A.B.; Nathan, D.M.; Schwartz, M.W.; Smith, R.J.; Smith, S.R. Obesity and Type 2 Diabetes: What Can Be Unified and What Needs to Be Individualized? J. Clin. Endocrinol. Metab. 2011, 96, 1654–1663. [Google Scholar] [CrossRef]
- Tessari, P.; Cecchet, D.; Cosma, A.; Vettore, M.; Coracina, A.; Millioni, R.; Iori, E.; Puricelli, L.; Avogaro, A.; Vedovato, M. Nitric Oxide Synthesis Is Reduced in Subjects with Type 2 Diabetes and Nephropathy. Diabetes 2010, 59, 2152–2159. [Google Scholar] [CrossRef]
- Takahashi, T.; Harris, R.C. Role of Endothelial Nitric Oxide Synthase in Diabetic Nephropathy: Lessons from Diabetic eNOS Knockout Mice. J. Diabetes Res. 2014, 2014, 590541. [Google Scholar] [CrossRef]
- Srinivasan, S.; Hatley, M.E.; Bolick, D.T.; Palmer, L.A.; Edelstein, D.; Brownlee, M.; Hedrick, C.C. Hyperglycaemia-induced superoxide production decreases eNOS expression via AP-1 activation in aortic endothelial cells. Diabetologia 2004, 47, 1727–1734. [Google Scholar] [CrossRef] [PubMed]
- Sansbury, B.E.; Hill, B.G. Regulation of obesity and insulin resistance by nitric oxide. Free Radic. Biol. Med. 2014, 73, 383–399. [Google Scholar] [CrossRef]
- Bahadoran, Z.; Ghasemi, A.; Mirmiran, P.; Azizi, F.; Hadaegh, F. Beneficial effects of inorganic nitrate/nitrite in type 2 diabetes and its complications. Nutr. Metab. 2015, 12, 16. [Google Scholar] [CrossRef] [PubMed]
- Ashor, A.W.; Chowdhury, S.; Oggioni, C.; Qadir, O.; Brandt, K.; Ishaq, A.; Mathers, J.C.; Saretzki, G.; Siervo, M. Inorganic Nitrate Supplementation in Young and Old Obese Adults Does Not Affect Acute Glucose and Insulin Responses but Lowers Oxidative Stress. J. Nutr. 2016, 146, 2224–2232. [Google Scholar] [CrossRef]
- Han, T.S.; Lean, M.E. A clinical perspective of obesity, metabolic syndrome and cardiovascular disease. JRSM Cardiovasc. Dis. 2016, 5, 2048004016633371. [Google Scholar] [CrossRef]
- Schiffer, T.A.; Lundberg, J.O.; Weitzberg, E.; Carlström, M. Modulation of mitochondria and NADPH oxidase function by the nitrate-nitrite-NO pathway in metabolic disease with focus on type 2 diabetes. Biochim. et Biophys. Acta (BBA) Mol. Basis Dis. 2020, 1866, 165811. [Google Scholar] [CrossRef]
- Phoswa, W.N.; Khaliq, O.P. Is pregnancy a risk factor of COVID-19? Eur. J. Obs. Gynecol. Reprod. Biol. 2020, 252, 605–609. [Google Scholar] [CrossRef]
- Darkwa, E.O.; Djagbletey, R.; Sottie, D.; Owoo, C.; Vanderpuye, N.M.; Essuman, R.; Arye, G. Serum nitric oxide levels in healthy pregnant women: A case- control study in a tertiary facility in Ghana. Matern. Health Neonatol. Perinatol. 2018, 3, 3. [Google Scholar] [CrossRef]
- Bryson, C.L.; Ioannou, G.N.; Rulyak, S.J.; Critchlow, C. Association between Gestational Diabetes and Pregnancy-induced Hypertension. Am. J. Epidemiol. 2003, 158, 1148–1153. [Google Scholar] [CrossRef]
- Haworth, S.M.M.; Zhuge, Z.; Nihlén, C.; Von Rosen, M.F.; Weitzberg, E.; Lundberg, J.O.; Krmar, R.T.; Nasiell, J.; Carlström, M. Red blood cells from patients with pre-eclampsia induce endothelial dysfunction. J. Hypertens. 2021, 39, 1628–1641. [Google Scholar] [CrossRef]
- Bogdan, C.; Röllinghoff, M.; Diefenbach, A. The role of nitric oxide in innate immunity. Immunol. Rev. 2000, 173, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Taylor, E.L.; Megson, I.L.; Haslett, C.; Rossi, A.G. Nitric oxide: A key regulator of myeloid inflammatory cell apoptosis. Cell Death Differ 2003, 10, 418–430. [Google Scholar] [CrossRef] [PubMed]
- Palmieri, E.M.; McGINITY, C.; Wink, D.A.; McVicar, D.W. Nitric Oxide in Macrophage Immunometabolism: Hiding in Plain Sight. Metabolites 2020, 10, 429. [Google Scholar] [CrossRef] [PubMed]
- Xiao, N.; Nie, M.; Pang, H.; Wang, B.; Hu, J.; Meng, X.; Li, K.; Ran, X.; Long, Q.; Deng, H.; et al. Integrated cytokine and metabolite analysis reveals immunometabolic reprogramming in COVID-19 patients with therapeutic implications. Nat. Commun. 2021, 12, 1618. [Google Scholar] [CrossRef] [PubMed]
- Olwal, C.O.; Nganyewo, N.N.; Tapela, K.; Zune, A.L.D.; Owoicho, O.; Bediako, Y.; Duodu, S. Parallels in Sepsis and COVID-19 Conditions: Implications for Managing Severe COVID-Front. Immunol. 2021, 12, 602848. [Google Scholar] [CrossRef]
- Lambden, S. Bench to bedside review: Therapeutic modulation of nitric oxide in sepsis—An update. Intensiv. Care Med. Exp. 2019, 7, 64. [Google Scholar] [CrossRef]
- Sharawy, N.; Lehmann, C. Molecular mechanisms by which iNOS uncoupling can induce cardiovascular dysfunction during sepsis: Role of posttranslational modifications (PTMs). Life Sci. 2020, 255, 117821. [Google Scholar] [CrossRef] [PubMed]
- Bogdan, C. Regulation of Lymphocytes by Nitric Oxide. Methods Mol. Biol. 2011, 677, 375–393. [Google Scholar] [CrossRef]
- Surazakov, A.; Klassen, A.; Gizinger, O. The bioenergetics of COVID-19 immunopathology and the therapeutic potential of biophysical radiances. J. Photochem. Photobiol. B Biol. 2020, 213, 112083. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.; Kirschenbaum, L.A.; LaRow, C.; Berna, G.; Griffin, K.; Astiz, M.E. Augmentation of Platelet and Endothelial Cell Enos Activity Decreases Sepsis-Related Neutrophil-Endothelial Cell Interactions. Shock 2010, 33, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Sin, D.D. COVID-19 in COPD: A growing concern. E Clin. Med. 2020, 26, 100546. [Google Scholar] [CrossRef] [PubMed]
- Csoma, B.; Bikov, A.; Nagy, L.; Tóth, B.; Tábi, T.; Szűcs, G.; Komlósi, Z.I.; Müller, V.; Losonczy, G.; Lázár, Z. Dysregulation of the endothelial nitric oxide pathway is associated with airway inflammation in COPD. Respir. Res. 2019, 20, 156. [Google Scholar] [CrossRef]
- Arif, E.; Ahsan, A.; Vibhuti, A.; Rajput, C.; Deepak, D.; Athar, M.; Singh, B.; Pasha, M.Q. Endothelial nitric oxide synthase gene variants contribute to oxidative stress in COPD. Biochem. Biophys. Res. Commun. 2007, 361, 182–188. [Google Scholar] [CrossRef]
- Oldreive, C.; Rice-Evans, C. The mechanisms for nitration and nitrotyrosine formationin vitroandin vivo: Impact of diet. Free Radic. Res. 2001, 35, 215–231. [Google Scholar] [CrossRef]
- Hongjun, J.; Webb-Robertson, B.-J.; Peterson, E.S.; Tan, R.; Bigelow, D.J.; Scholand, M.B.; Hoidal, J.R.; Pounds, J.G.; Zangar, R.C. Smoking, COPD, and 3-Nitrotyrosine Levels of Plasma Proteins. Env. Health Perspect. 2011, 119, 1314–1320. [Google Scholar]
- Tufvesson, E.; Andersson, C.; Weidner, J.; Erjefält, J.S.; Bjermer, L. Inducible nitric oxide synthase expression is increased in the alveolar compartment of asthmatic patients. Allergy 2017, 72, 627–635. [Google Scholar] [CrossRef]
- Tang, S.; Xie, Y.; Yuan, C.; Sun, X.; Cui, Y. Fractional Exhaled Nitric Oxide for the Diagnosis of Childhood Asthma: A Systematic Review and Meta-analysis. Clin Rev Allergy Immunol. 2019, 56, 129–138. [Google Scholar] [CrossRef]
- Broeke, R.T.; De Crom, R.; Van Haperen, R.; Verweij, V.; Leusink-Muis, T.; Van Ark, I.; De Clerck, F.; Nijkamp, F.P.; Folkerts, G. Overexpression of endothelial nitric oxide synthase suppresses features of allergic asthma in mice. Respir. Res. 2006, 7, 58. [Google Scholar] [CrossRef]
- Yildiz, P.; Oflaz, H.; Cine, N.; Genchallac, H.; Erginel-Unaltuna, N.; Yildiz, A.; Yilmaz, V. Endothelial Dysfunction in Patients with Asthma: The Role of Polymorphisms of ACE and Endothelial NOS Genes. J. Asthma 2004, 41, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Fernando, Y.; Wickramasinghe, P.; De Silva, U.; Alahakoon, M.; Anuradha, K.W.D.A.; Handunnetti, S. Differences in serum markers of oxidative stress in well controlled and poorly controlled asthma in Sri Lankan children: A pilot study. Allergy Asthma Clin. Immunol. 2020, 16, 1–9. [Google Scholar] [CrossRef]
- Shimasaki, Y.; Saito, Y.; Yoshimura, M.; Kamitani, S.; Miyamoto, Y.; Masuda, I.; Nakayama, M.; Mizuno, Y.; Ogawa, H.; Yasue, H.; et al. The Effects of Long-term Smoking on Endothelial Nitric Oxide Synthase mRNA Expression in Human Platelets as Detected with Real-time Quantitative RT-PCR. Clin. Appl. Thromb. 2007, 13, 43–51. [Google Scholar] [CrossRef]
- He, Z.; Chen, Y.; Hou, C.; He, W.; Chen, P. Cigarette Smoke Extract Changes Expression of Endothelial Nitric Oxide Synthase (eNOS) and p16(INK4a) and is Related to Endothelial Progenitor Cell Dysfunction. Med. Sci. Monit. 2017, 23, 3224–3231. [Google Scholar] [CrossRef][Green Version]
- Colasanti, M.; Persichini, T.; Venturini, G.; Ascenzi, P. S-Nitrosylation of Viral Proteins: Molecular Bases for Antiviral Effect of Nitric Oxide. IUBMB Life 1999, 48, 25–31. [Google Scholar] [CrossRef]
- Margaritis, M. Endothelial dysfunction in HIV infection: Experimental and clinical evidence on the role of oxidative stress. Ann. Res. Hosp. 2019, 3, 7. [Google Scholar] [CrossRef]
- Kline, E.R.; Kleinhenz, D.J.; Liang, B.; Dikalov, S.; Guidot, D.M.; Hart, C.M.; Jones, D.P.; Sutliff, R.L. Vascular oxidative stress and nitric oxide depletion in HIV-1 transgenic rats are reversed by glutathione restoration. Am. J. Physiol. Heart Circ. Physiol. 2008, 294, 2792–2804. [Google Scholar] [CrossRef]
- Åkerström, S.; Gunalan, V.; Keng, C.T.; Tan, Y.-J.; Mirazimi, A. Dual effect of nitric oxide on SARS-CoV replication: Viral RNA production and palmitoylation of the S protein are affected. Virology 2009, 395, 1–9. [Google Scholar] [CrossRef] [PubMed]
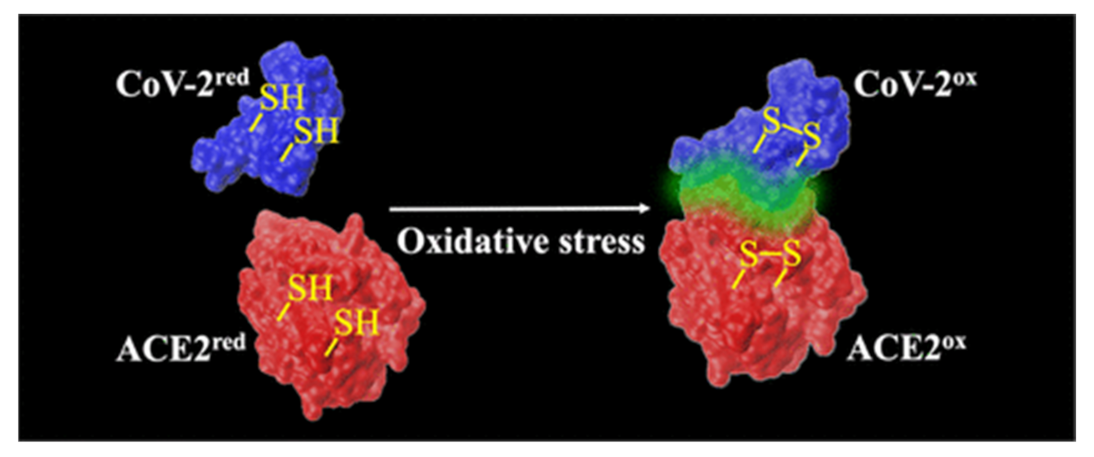
- Hati, S.; Bhattacharyya, S. Impact of Thiol–Disulfide Balance on the Binding of Covid-19 Spike Protein with Angiotensin-Converting Enzyme 2 Receptor. ACS Omega 2020, 5, 16292–16298. [Google Scholar] [CrossRef] [PubMed]
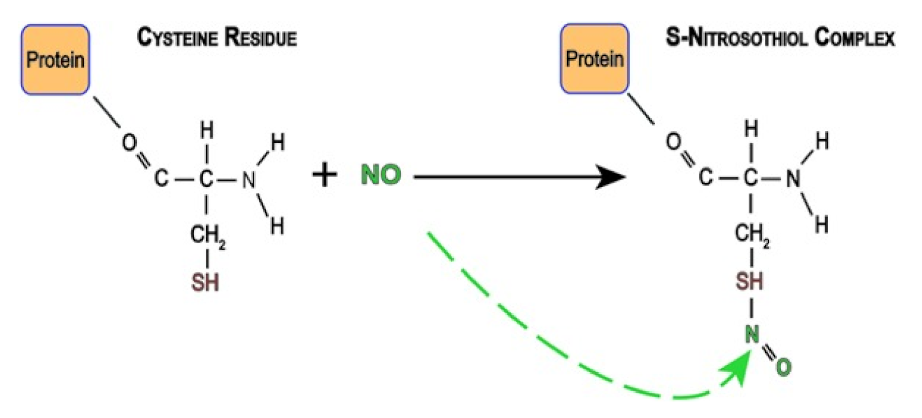
- Gaston, B. Nitric oxide and thiol groups. Biochim. Biophys. Acta 1999, 1411, 323–333. [Google Scholar] [CrossRef]
- Abrams, J. Interactions between organic nitrates and thiol groups. Am. J. Med. 1991, 91, 106–112. [Google Scholar] [CrossRef]
- Da Silva, M.P.; Cedraz-Mercez, P.L.; Varanda, W.A. Effects of nitric oxide on magnocellular neurons of the supraoptic nucleus involve multiple mechanisms. Braz. J. Med. Biol Res. 2014, 47, 90–100. [Google Scholar] [CrossRef] [PubMed]
- Packer, M. What Causes Tolerance to Nitroglycerin? The 100 Year Old Mystery Continues. J. Am. Coll Cardiol. 1990, 16, 16932–16935. [Google Scholar] [CrossRef]
- Tsutsumi, N.; Itoh, T.; Ohsawa, A. Cleavage of S-S Bond by Nitric Oxide (NO) in the Presence of Oxygen. A Disproportionation Reaction of Two Disulfides. Chem. Pharm. Bull. 2000, 48, 1524–1528. [Google Scholar] [CrossRef][Green Version]
- Rouillard, K.; Hill, D.B.; Schoenfisch, M.H. Antibiofilm and mucolytic action of nitric oxide delivered via gas or macromolecular donor using in vitro and ex vivo models. J. Cyst. Fibros. 2020, 19, 1004–1010. [Google Scholar] [CrossRef] [PubMed]
- Sender, R.; Bar-On, Y.M.; Gleizer, S.; Bernshtein, B.; Flamholz, A.; Phillips, R.; Milo, R. The total number and mass of SARS-CoV-2 virions. Proc. Natl. Acad. Sci. USA 2021, 118, 25. [Google Scholar] [CrossRef]
- Liu, X.; Yan, Q.; Baskerville, K.L.; Zweier, J.L. Estimation of Nitric Oxide Concentration in Blood for Different Rates of Generation. J. Biol. Chem. 2007, 282, 8831–8836. [Google Scholar] [CrossRef] [PubMed]
- Rubini, A. The Effect of NG-Nitro-L-Arginine Methyl Ester, a Nitric Oxide Synthase Inhibitor, on Respiratory Mechanics in Rats. Respiration 2011, 82, 468–475. [Google Scholar] [CrossRef]
- Dambisya, Y.M.; Lee, T.-L. A thromboelastography study on the in vitro effects of L-arginine and L-NG-nitro arginine methyl ester on human whole blood coagulation and fibrinolysis. Blood Coagul. Fibrinolysis 1996, 7, 678–683. [Google Scholar] [CrossRef]
- De Angelis, K.; Ogawa, T.; Sanches, I.C.; Rigatto, K.V.; Krieger, E.M.; Irigoyen, M.-C. Impairment on cardiac output and blood flow adjustments to exercise in L-NAME-induced hypertensive rats. J. Cardiovasc. Pharm. 2006, 47, 371–376. [Google Scholar] [CrossRef]
- Landis, B.N.; Lacroix, J.-S. Olfactory function and nasal nitric oxide. Curr. Opin. Otolaryngol. Head Neck Surg. 2009, 17, 18–22. [Google Scholar] [CrossRef] [PubMed]
- Dominic, P.; Ahmad, J.; Bhandari, R.; Pardue, S.; Solorzano, J.; Jaisingh, K.; Watts, M.; Bailey, S.R.; Orr, A.W.; Kevil, C.G.; et al. Decreased availability of nitric oxide and hydrogen sulfide is a hallmark of COVID. Redox Biol. 2021, 43, 101982. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Mei, F.; Bai, L.; Zhou, S.; Liu, D.; Yao, L.; Ahluwalia, A.; Ghiladi, R.A.; Su, L.; Shu, T.; et al. Serum nitrite and nitrate: A potential biomarker for post-covid-19 complications? Free Radic. Biol. Med. 2021, 175, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Oliveira-Paula, G.H.; Lacchini, R.; Tanus-Santos, J.E. Clinical and pharmacogenetic impact of endothelial nitric oxide synthase polymorphisms on cardiovascular diseases. Nitric Oxide 2017, 63, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, J.; Gladwin, M.T.; Ahluwalia, A.; Benjamin, N.; Bryan, N.S.; Butler, A.; Cabrales, P.; Fago, A.; Feelisch, M.; Ford, P.C.; et al. Nitrate and nitrite in biology, nutrition and therapeutics. Nat. Chem. Biol. 2009, 5, 865–869. [Google Scholar] [CrossRef]
- Stroes, E.S.G.; van Faassen, E.E.; Yo, M.; Martasek, P.; Boer, P.; Govers, R.; Rabelink, T.J. Folic Acid Reverts Dysfunction of Endothelial Nitric Oxide Synthase. Circ. Res. 2000, 86, 1129–1134. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nikolaidis, A.; Kramer, R.; Ostojic, S. Nitric Oxide: The Missing Factor in COVID-19 Severity? Med. Sci. 2022, 10, 3. https://doi.org/10.3390/medsci10010003
Nikolaidis A, Kramer R, Ostojic S. Nitric Oxide: The Missing Factor in COVID-19 Severity? Medical Sciences. 2022; 10(1):3. https://doi.org/10.3390/medsci10010003
Chicago/Turabian StyleNikolaidis, Alexandros, Ron Kramer, and Sergej Ostojic. 2022. "Nitric Oxide: The Missing Factor in COVID-19 Severity?" Medical Sciences 10, no. 1: 3. https://doi.org/10.3390/medsci10010003
APA StyleNikolaidis, A., Kramer, R., & Ostojic, S. (2022). Nitric Oxide: The Missing Factor in COVID-19 Severity? Medical Sciences, 10(1), 3. https://doi.org/10.3390/medsci10010003






