Overcoming Drug Resistance in Advanced Prostate Cancer by Drug Repurposing
Abstract
1. Introduction
1.1. Prostate Cancer
1.2. Therapy Resistance in Prostate Cancer
1.3. Molecular Landscape in Prostate Cancer
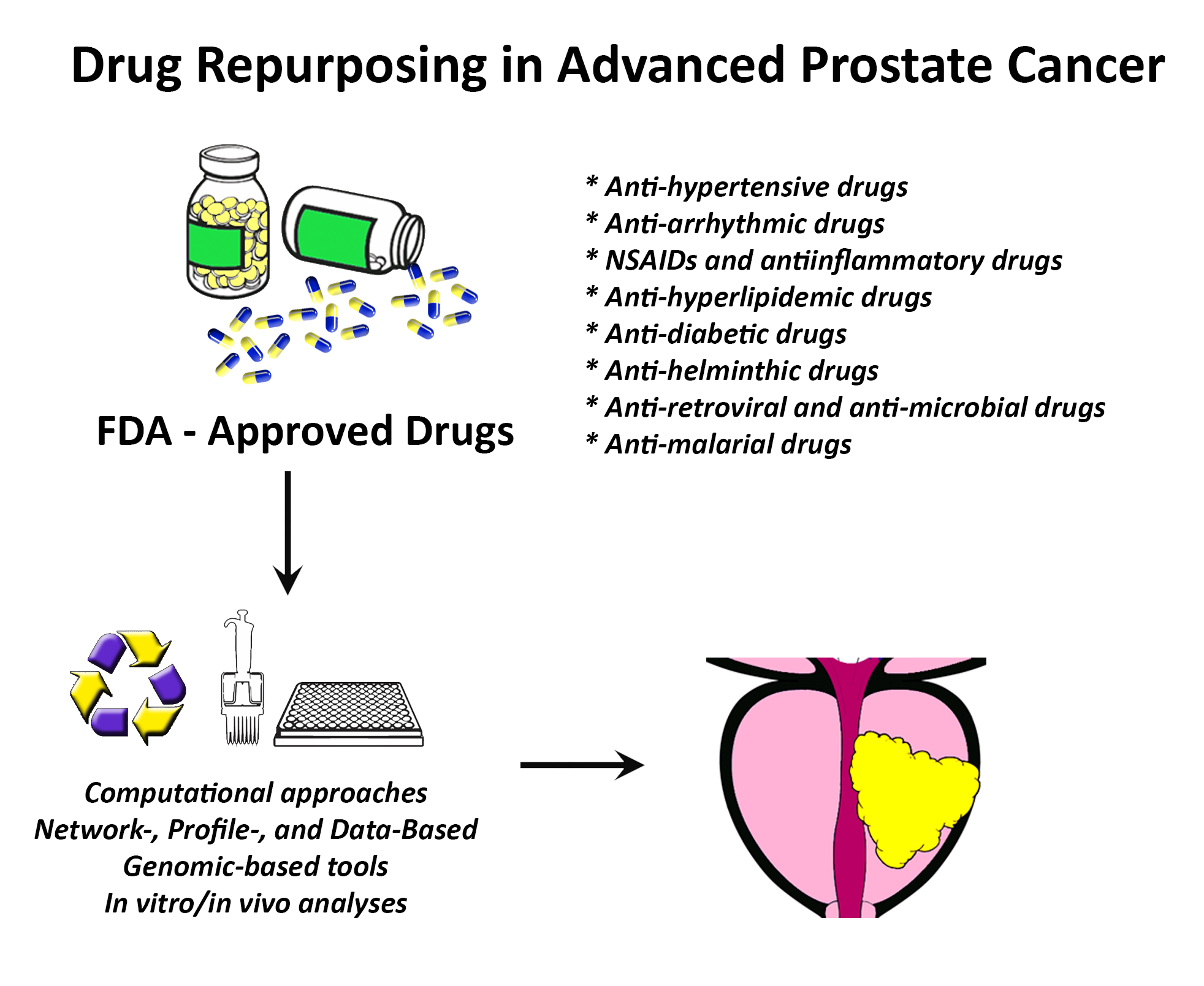
2. Repurposing Approved Drugs in Cancer
2.1. Introduction to Drug Repurposing
2.2. A Computational Approach for Drug Repurposing
2.2.1. Network-Based Drug Repurposing
2.2.2. Profile-Based Drug Repurposing
2.2.3. Data-Based Drug Repurposing
2.3. Drug Repurposing Approach in Cancer
3. Drug Repurposing in Prostate Cancer
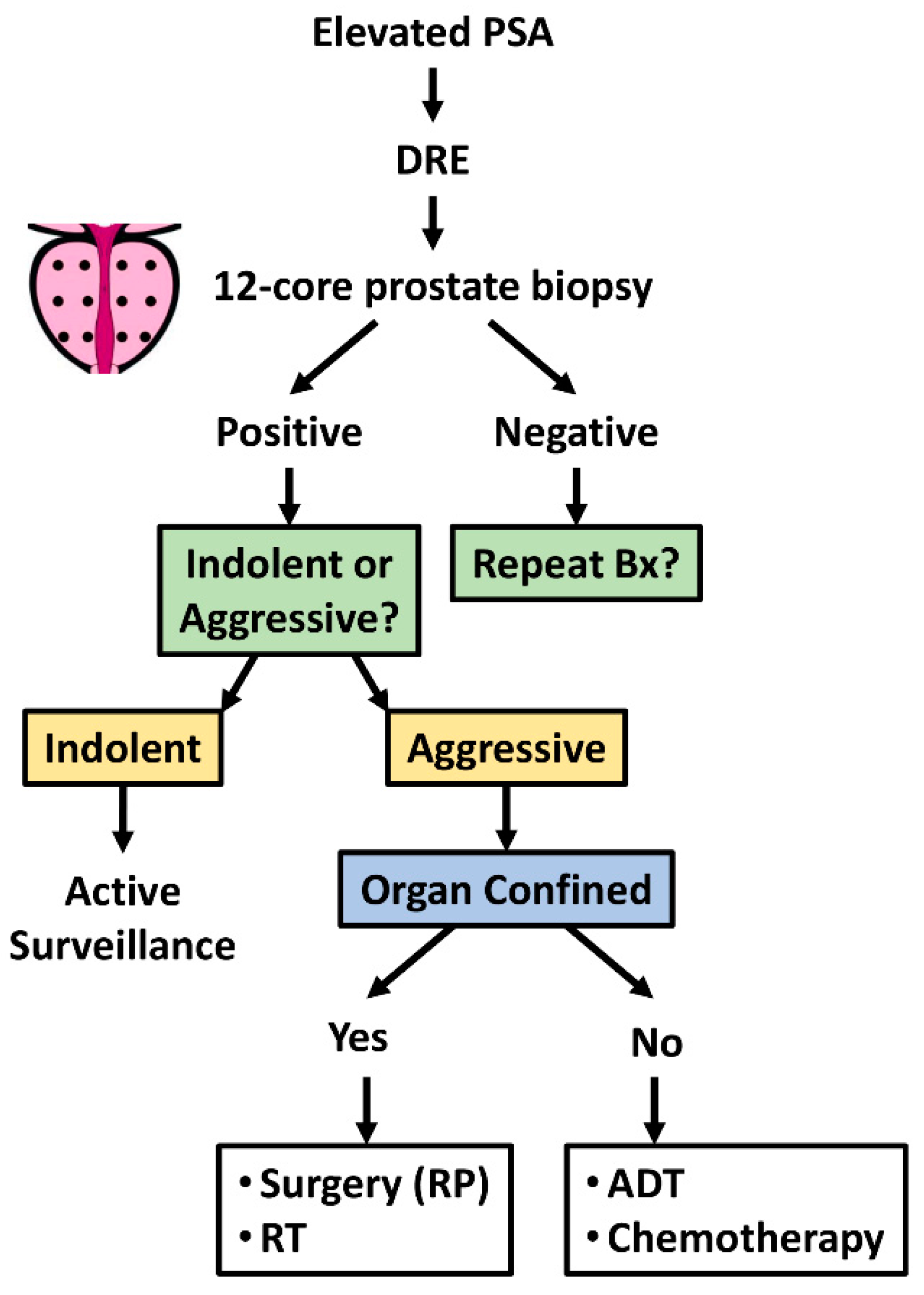
3.1. Clinical Challenges in Prostate Cancer
3.2. Drugs Repurposed in Prostate Cancer: From Benchside to Bedside
| Ref. | Drug | Original Indication | PCa Cell Lines Targeted | In Vivo Studies | Mode(s) of Action | Effect(s) |
|---|---|---|---|---|---|---|
| [137] | Propranolol | Anti-hypertensive | - | - |
|
|
| [138] | Digoxin | Anti-arrhythmic |
| |||
| [138,139] | Ouabain | Anti-arrhythmic | PPC-1 | Male SCID |
| |
| [140] | Aspirin | Anti-inflammatory | LNCaP |
|
| |
| [141] | Celecoxib | Anti-inflammatory | LNCaP & androgen-nonresponsive PC-3 | LNCaP in bovine brain extracts |
|
|
| [142] | dexamethasone | Anti-inflammatory | DU145 PCa cells |
|
| |
| [143,144,145] | Simvastatin | Anti-hyperlipidimic | PC3, 22Rv1, DU145, DU145R80, LNCaP prostate cancer cell lines and EPN normal prostate epithelial cells | DU145R80, 22Rv1 parental and docetaxel resistant cells in xenografts |
|
|
| [146,147,148] | Metformin | Anti-diabetic |
|
| ||
| [149] | Glipizide | Anti-diabetic | PC-3, 22Rv1 and DU145 PC | TRAMP transgenic mouse model |
|
|
| [150] | Mebendazole | Anti-helminthic | LNCaP |
|
| |
| [151] | Niclosamide | Anti-helminthic | LNCaP, VCaP, CWR22Rv1, PC3 and HEK293 | CWR22Rv1 cells in SCID mice |
|
|
| [152,153] | Nelfinavir | Anti-retroviral | DU145 and PC3 cell lines |
|
| |
| [154] | CMT-3 | Anti-microbial | Many lines | xenografts of PC-3 tumors |
|
|
| [155] | Zoledronic acid | Bisphosphonate | LuCaP 23.1, a PSA-producing human CaP xenograft | LuCaP 23.1, a PSA-producing human CaP xenograft |
|
|
| [156] | Valproic acid | Anti-epileptic | AR-positive (LNCaP and C4-2) and AR-negative (DU145 and PC3) | LNCaP, C4-2, and DU145 Xenograft models |
|
|
| [157] | Mifepristone | Anti-progestational steroid |
|
|
3.2.1. Anti-Hypertensives and Anti-Arrhythmic Drugs
3.2.2. NSAIDS, Anti-Inflammatory Drugs and Aspirin
3.2.3. Anti-Hyperlipidemic Drugs
3.2.4. Anti-Diabetic Drugs
3.2.5. Anti-Helminthic Drugs
3.2.6. Anti-Retroviral Drugs
3.2.7. Anti-Microbial Drugs
3.2.8. Anti-Malarial Drugs
3.2.9. Others
4. Clinical Trials of Drugs Repurposed in PCa
5. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics, 2022. CA Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Stamey, T.A.; Donaldson, A.N.; Yemoto, C.E.; McNeal, J.E.; Sözen, S.; Gill, H. Histological and clinical findings in 896 consecutive prostates treated only with radical retropubic prostatectomy: Epidemiologic significance of annual changes. J. Urol. 1998, 160, 2412–2417. [Google Scholar] [CrossRef]
- Al-Hussain, T.; Carter, H.B.; Epstein, J.I. Significance of prostate adenocarcinoma perineural invasion on biopsy in patients who are otherwise candidates for active surveillance. J. Urol. 2011, 186, 470–473. [Google Scholar] [CrossRef] [PubMed]
- Rakic, N.; Jamil, M.; Keeley, J.; Sood, A.; Vetterlein, M.; Dalela, D.; Arora, S.; Modonutti, D.; Bronkema, C.; Novara, G.; et al. Evaluation of lymphovascular invasion as a prognostic predictor of overall survival after radical prostatectomy. Urol. Oncol. 2021, 39, 495.e1–495.e6. [Google Scholar] [CrossRef] [PubMed]
- Harada, M.; Iida, M.; Yamaguchi, M.; Shida, K. Analysis of bone metastasis of prostatic adenocarcinoma in 137 autopsy cases. Adv. Exp. Med. Biol. 1992, 324, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Kasivisvanathan, V.; Rannikko, A.S.; Borghi, M.; Panebianco, V.; Mynderse, L.A.; Vaarala, M.H.; Briganti, A.; Budäus, L.; Hellawell, G.; Hindley, R.G.; et al. MRI-Targeted or Standard Biopsy for Prostate-Cancer Diagnosis. N. Engl. J. Med. 2018, 378, 1767–1777. [Google Scholar] [CrossRef]
- Karakiewicz, P.I.; Benayoun, S.; Kattan, M.W.; Perrotte, P.; Valiquette, L.; Scardino, P.T.; Cagiannos, I.; Heinzer, H.; Tanguay, S.; Aprikian, A.G.; et al. Development and validation of a nomogram predicting the outcome of prostate biopsy based on patient age, digital rectal examination and serum prostate specific antigen. J. Urol. 2005, 173, 1930–1934. [Google Scholar] [CrossRef]
- Bjurlin, M.A.; Taneja, S.S. Standards for prostate biopsy. Curr. Opin. Urol. 2014, 24, 155–161. [Google Scholar] [CrossRef]
- Pound, C.R.; Partin, A.W.; Epstein, J.I.; Walsh, P.C. Prostate-specific antigen after anatomic radical retropubic prostatectomy. Patterns of recurrence and cancer control. Urol. Clin. N. Am. 1997, 24, 395–406. [Google Scholar] [CrossRef]
- Chen, R.C.; Rumble, R.B.; Loblaw, D.A.; Finelli, A.; Ehdaie, B.; Cooperberg, M.R.; Morgan, S.C.; Tyldesley, S.; Haluschak, J.J.; Tan, W.; et al. Active Surveillance for the Management of Localized Prostate Cancer (Cancer Care Ontario Guideline): American Society of Clinical Oncology Clinical Practice Guideline Endorsement. J. Clin. Oncol. 2016, 34, 2182–2190. [Google Scholar] [CrossRef]
- Sanda, M.G.; Cadeddu, J.A.; Kirkby, E.; Chen, R.C.; Crispino, T.; Fontanarosa, J.; Freedland, S.J.; Greene, K.; Klotz, L.H.; Makarov, D.V.; et al. Clinically Localized Prostate Cancer: AUA/ASTRO/SUO Guideline. Part I: Risk Stratification, Shared Decision Making, and Care Options. J. Urol. 2018, 199, 683–690. [Google Scholar] [CrossRef]
- Lowrance, W.T.; Breau, R.H.; Chou, R.; Chapin, B.F.; Crispino, T.; Dreicer, R.; Jarrard, D.F.; Kibel, A.S.; Morgan, T.M.; Morgans, A.K.; et al. Advanced Prostate Cancer: AUA/ASTRO/SUO Guideline PART I. J. Urol. 2021, 205, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Kolodziej, M. Management of biochemically recurrent prostate cancer following local therapy. Am. J. Manag. Care 2014, 20, S273–S281. [Google Scholar] [PubMed]
- Han, M.; Partin, A.W.; Zahurak, M.; Piantadosi, S.; Epstein, J.I.; Walsh, P.C. Biochemical (prostate specific antigen) recurrence probability following radical prostatectomy for clinically localized prostate cancer. J. Urol. 2003, 169, 517–523. [Google Scholar] [CrossRef]
- Kirby, M.; Hirst, C.; Crawford, E.D. Characterising the castration-resistant prostate cancer population: A systematic review. Int. J. Clin. Pract. 2011, 65, 1180–1192. [Google Scholar] [CrossRef]
- Karantanos, T.; Corn, P.G.; Thompson, T.C. Prostate cancer progression after androgen deprivation therapy: Mechanisms of castrate resistance and novel therapeutic approaches. Oncogene 2013, 32, 5501–5511. [Google Scholar] [CrossRef] [PubMed]
- Thigpen, A.E.; Davis, D.L.; Milatovich, A.; Mendonca, B.B.; Imperato-McGinley, J.; Griffin, J.E.; Francke, U.; Wilson, J.D.; Russell, D.W. Molecular genetics of steroid 5 alpha-reductase 2 deficiency. J. Clin. Investig. 1992, 90, 799–809. [Google Scholar] [CrossRef] [PubMed]
- HUGGINS, C.; JOHNSON, M.A. Cancer of the bladder and prostate. J. Am. Med. Assoc. 1947, 135, 1146–1152. [Google Scholar] [CrossRef] [PubMed]
- Nakazawa, M.; Paller, C.; Kyprianou, N. Mechanisms of Therapeutic Resistance in Prostate Cancer. Curr. Oncol. Rep. 2017, 19, 13. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.C.; Chen, W.Y.; Abou-Kheir, W.; Zeng, T.; Yin, J.J.; Bahmad, H.; Lee, Y.C.; Liu, Y.N. Androgen deprivation therapy-induced epithelial-mesenchymal transition of prostate cancer through downregulating SPDEF and activating CCL2. Biochim. Biophys. Acta Mol. Basis. Dis. 2018, 1864, 1717–1727. [Google Scholar] [CrossRef]
- Cheaito, K.A.; Bahmad, H.F.; Hadadeh, O.; Saleh, E.; Dagher, C.; Hammoud, M.S.; Shahait, M.; Mrad, Z.A.; Nassif, S.; Tawil, A.; et al. EMT Markers in Locally-Advanced Prostate Cancer: Predicting Recurrence? Front. Oncol. 2019, 9, 131. [Google Scholar] [CrossRef] [PubMed]
- Weng, H.; Li, S.; Huang, J.Y.; He, Z.Q.; Meng, X.Y.; Cao, Y.; Fang, C.; Zeng, X.T. Androgen receptor gene polymorphisms and risk of prostate cancer: A meta-analysis. Sci. Rep. 2017, 7, 40554. [Google Scholar] [CrossRef] [PubMed]
- Maitland, N.J. Resistance to Antiandrogens in Prostate Cancer: Is It Inevitable, Intrinsic or Induced? Cancers 2021, 13, 327. [Google Scholar] [CrossRef] [PubMed]
- Pearson, H.B.; Li, J.; Meniel, V.S.; Fennell, C.M.; Waring, P.; Montgomery, K.G.; Rebello, R.J.; Macpherson, A.A.; Koushyar, S.; Furic, L.; et al. Identification of Pik3ca Mutation as a Genetic Driver of Prostate Cancer That Cooperates with Pten Loss to Accelerate Progression and Castration-Resistant Growth. Cancer Discov. 2018, 8, 764–779. [Google Scholar] [CrossRef]
- Li, S.; Fong, K.W.; Gritsina, G.; Zhang, A.; Zhao, J.C.; Kim, J.; Sharp, A.; Yuan, W.; Aversa, C.; Yang, X.J.; et al. Activation of MAPK Signaling by CXCR7 Leads to Enzalutamide Resistance in Prostate Cancer. Cancer Res. 2019, 79, 2580–2592. [Google Scholar] [CrossRef]
- Yamamoto, H.; Oue, N.; Sato, A.; Hasegawa, Y.; Matsubara, A.; Yasui, W.; Kikuchi, A. Wnt5a signaling is involved in the aggressiveness of prostate cancer and expression of metalloproteinase. Oncogene 2010, 29, 2036–2046. [Google Scholar] [CrossRef]
- Mulholland, D.J.; Kobayashi, N.; Ruscetti, M.; Zhi, A.; Tran, L.M.; Huang, J.; Gleave, M.; Wu, H. Pten loss and RAS/MAPK activation cooperate to promote EMT and metastasis initiated from prostate cancer stem/progenitor cells. Cancer Res. 2012, 72, 1878–1889. [Google Scholar] [CrossRef]
- Carracedo, A.; Ma, L.; Teruya-Feldstein, J.; Rojo, F.; Salmena, L.; Alimonti, A.; Egia, A.; Sasaki, A.T.; Thomas, G.; Kozma, S.C.; et al. Inhibition of mTORC1 leads to MAPK pathway activation through a PI3K-dependent feedback loop in human cancer. J. Clin. Investig. 2008, 118, 3065–3074. [Google Scholar] [CrossRef]
- Kinkade, C.W.; Castillo-Martin, M.; Puzio-Kuter, A.; Yan, J.; Foster, T.H.; Gao, H.; Sun, Y.; Ouyang, X.; Gerald, W.L.; Cordon-Cardo, C.; et al. Targeting AKT/mTOR and ERK MAPK signaling inhibits hormone-refractory prostate cancer in a preclinical mouse model. J. Clin. Investig. 2008, 118, 3051–3064. [Google Scholar] [CrossRef]
- Tomlins, S.A.; Rhodes, D.R.; Perner, S.; Dhanasekaran, S.M.; Mehra, R.; Sun, X.W.; Varambally, S.; Cao, X.; Tchinda, J.; Kuefer, R.; et al. Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science 2005, 310, 644–648. [Google Scholar] [CrossRef]
- Kim, J.; Wu, L.; Zhao, J.C.; Jin, H.J.; Yu, J. TMPRSS2-ERG gene fusions induce prostate tumorigenesis by modulating microRNA miR-200c. Oncogene 2014, 33, 5183–5192. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Blee, A.M.; Wang, D.; An, J.; Pan, Y.; Yan, Y.; Ma, T.; He, Y.; Dugdale, J.; Hou, X.; et al. Loss of FOXO1 Cooperates with TMPRSS2-ERG Overexpression to Promote Prostate Tumorigenesis and Cell Invasion. Cancer Res. 2017, 77, 6524–6537. [Google Scholar] [CrossRef] [PubMed]
- Rybak, A.P.; Bristow, R.G.; Kapoor, A. Prostate cancer stem cells: Deciphering the origins and pathways involved in prostate tumorigenesis and aggression. Oncotarget 2015, 6, 1900–1919. [Google Scholar] [CrossRef]
- Vadde, R.; Vemula, S.; Jinka, R.; Merchant, N.; Bramhachari, P.V.; Nagaraju, G.P. Role of hypoxia-inducible factors (HIF) in the maintenance of stemness and malignancy of colorectal cancer. Crit. Rev. Oncol. Hematol. 2017, 113, 22–27. [Google Scholar] [CrossRef]
- O’Reilly, D.; Johnson, P.; Buchanan, P.J. Hypoxia induced cancer stem cell enrichment promotes resistance to androgen deprivation therapy in prostate cancer. Steroids 2019, 152, 108497. [Google Scholar] [CrossRef]
- Swinney, D.C. Phenotypic vs. target-based drug discovery for first-in-class medicines. Clin. Pharmacol. Ther. 2013, 93, 299–301. [Google Scholar] [CrossRef]
- Eder, J.; Sedrani, R.; Wiesmann, C. The discovery of first-in-class drugs: Origins and evolution. Nat. Rev. Drug Discov. 2014, 13, 577–587. [Google Scholar] [CrossRef]
- Pushpakom, S.; Iorio, F.; Eyers, P.A.; Escott, K.J.; Hopper, S.; Wells, A.; Doig, A.; Guilliams, T.; Latimer, J.; McNamee, C.; et al. Drug repurposing: Progress, challenges and recommendations. Nat. Rev. Drug Discov. 2019, 18, 41–58. [Google Scholar] [CrossRef]
- Phillips, D.J. Pfizer’s Expiring Viagra Patent Adversely Affects Other Drugmakers Too. Available online: https://www.forbes.com/sites/investor/2013/12/20/pfizers-expiring-viagra-patent-adversely-affects-other-drugmakers-too/?sh=5349531468d4 (accessed on 11 January 2021).
- Kim, J.H.; Scialli, A.R. Thalidomide: The tragedy of birth defects and the effective treatment of disease. Toxicol. Sci. 2011, 122, 1–6. [Google Scholar] [CrossRef]
- Ashburn, T.T.; Thor, K.B. Drug repositioning: Identifying and developing new uses for existing drugs. Nat. Rev. Drug Discov. 2004, 3, 673–683. [Google Scholar] [CrossRef]
- Singhal, S.; Mehta, J.; Desikan, R.; Ayers, D.; Roberson, P.; Eddlemon, P.; Munshi, N.; Anaissie, E.; Wilson, C.; Dhodapkar, M.; et al. Antitumor activity of thalidomide in refractory multiple myeloma. N. Engl. J. Med. 1999, 341, 1565–1571. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, Y.; Ye, D.; Liu, Q. Review of the 2019 novel coronavirus (SARS-CoV-2) based on current evidence. Int. J. Antimicrob. Agents 2020, 55, 105948. [Google Scholar] [CrossRef] [PubMed]
- Guy, R.K.; DiPaola, R.S.; Romanelli, F.; Dutch, R.E. Rapid repurposing of drugs for COVID-19. Science 2020, 368, 829–830. [Google Scholar] [CrossRef] [PubMed]
- Serafin, M.B.; Bottega, A.; Foletto, V.S.; da Rosa, T.F.; Horner, A.; Horner, R. Drug repositioning is an alternative for the treatment of coronavirus COVID-19. Int. J. Antimicrob. Agents 2020, 55, 105969. [Google Scholar] [CrossRef]
- Bahmad, H.F.; Abou-Kheir, W. Crosstalk between COVID-19 and prostate cancer. Prostate Cancer Prostatic Dis. 2020, 23, 561–563. [Google Scholar] [CrossRef]
- Wang, M.; Cao, R.; Zhang, L.; Yang, X.; Liu, J.; Xu, M.; Shi, Z.; Hu, Z.; Zhong, W.; Xiao, G. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020, 30, 269–271. [Google Scholar] [CrossRef]
- Liu, J.; Cao, R.; Xu, M.; Wang, X.; Zhang, H.; Hu, H.; Li, Y.; Hu, Z.; Zhong, W.; Wang, M. Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro. Cell Discov. 2020, 6, 16. [Google Scholar] [CrossRef]
- Sun, M.L.; Yang, J.M.; Sun, Y.P.; Su, G.H. Inhibitors of RAS Might Be a Good Choice for the Therapy of COVID-19 Pneumonia. Chin. J. Tuberc. Respir. Dis. 2020, 43, 219–222. [Google Scholar] [CrossRef]
- Breckenridge, A.; Jacob, R. Overcoming the legal and regulatory barriers to drug repurposing. Nat. Rev. Drug Discov. 2019, 18, 1–2. [Google Scholar] [CrossRef]
- Shim, J.S.; Liu, J.O. Recent advances in drug repositioning for the discovery of new anticancer drugs. Int. J. Biol. Sci. 2014, 10, 654–663. [Google Scholar] [CrossRef]
- Wishart, D.S.; Feunang, Y.D.; Guo, A.C.; Lo, E.J.; Marcu, A.; Grant, J.R.; Sajed, T.; Johnson, D.; Li, C.; Sayeeda, Z.; et al. DrugBank 5.0: A major update to the DrugBank database for 2018. Nucleic Acids Res. 2018, 46, D1074–D1082. [Google Scholar] [CrossRef] [PubMed]
- Seiler, K.P.; George, G.A.; Happ, M.P.; Bodycombe, N.E.; Carrinski, H.A.; Norton, S.; Brudz, S.; Sullivan, J.P.; Muhlich, J.; Serrano, M.; et al. ChemBank: A small-molecule screening and cheminformatics resource database. Nucleic Acids Res. 2008, 36, D351–D359. [Google Scholar] [CrossRef] [PubMed]
- Stelzer, G.; Rosen, N.; Plaschkes, I.; Zimmerman, S.; Twik, M.; Fishilevich, S.; Stein, T.I.; Nudel, R.; Lieder, I.; Mazor, Y.; et al. The GeneCards Suite: From Gene Data Mining to Disease Genome Sequence Analyses. Curr. Protoc. Bioinform. 2016, 54, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Amberger, J.S.; Bocchini, C.A.; Schiettecatte, F.; Scott, A.F.; Hamosh, A. OMIM.org: Online Mendelian Inheritance in Man (OMIM(R)), an online catalog of human genes and genetic disorders. Nucleic Acids Res. 2015, 43, D789–D798. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Thiessen, P.A.; Bolton, E.E.; Chen, J.; Fu, G.; Gindulyte, A.; Han, L.; He, J.; He, S.; Shoemaker, B.A.; et al. PubChem Substance and Compound databases. Nucleic Acids Res. 2016, 44, D1202–D1213. [Google Scholar] [CrossRef]
- Hurle, M.R.; Yang, L.; Xie, Q.; Rajpal, D.K.; Sanseau, P.; Agarwal, P. Computational drug repositioning: From data to therapeutics. Clin. Pharmacol. Ther. 2013, 93, 335–341. [Google Scholar] [CrossRef]
- Sanseau, P.; Agarwal, P.; Barnes, M.R.; Pastinen, T.; Richards, J.B.; Cardon, L.R.; Mooser, V. Use of genome-wide association studies for drug repositioning. Nat. Biotechnol. 2012, 30, 317–320. [Google Scholar] [CrossRef]
- Willyard, C. New human gene tally reignites debate. Nature 2018, 558, 354–355. [Google Scholar] [CrossRef]
- Grover, M.P.; Ballouz, S.; Mohanasundaram, K.A.; George, R.A.; Goscinski, A.; Crowley, T.M.; Sherman, C.D.; Wouters, M.A. Novel therapeutics for coronary artery disease from genome-wide association study data. BMC Med. Genom. 2015, 8 (Suppl. S2), S1. [Google Scholar] [CrossRef]
- Wang, Z.Y.; Zhang, H.Y. Rational drug repositioning by medical genetics. Nat. Biotechnol. 2013, 31, 1080–1082. [Google Scholar] [CrossRef]
- Greene, C.S.; Voight, B.F. Pathway and network-based strategies to translate genetic discoveries into effective therapies. Hum. Mol. Genet. 2016, 25, R94–R98. [Google Scholar] [CrossRef] [PubMed]
- Franke, A.; McGovern, D.P.; Barrett, J.C.; Wang, K.; Radford-Smith, G.L.; Ahmad, T.; Lees, C.W.; Balschun, T.; Lee, J.; Roberts, R.; et al. Genome-wide meta-analysis increases to 71 the number of confirmed Crohn’s disease susceptibility loci. Nat. Genet. 2010, 42, 1118–1125. [Google Scholar] [CrossRef]
- Wu, H.; Gao, L.; Dong, J.; Yang, X. Detecting overlapping protein complexes by rough-fuzzy clustering in protein-protein interaction networks. PLoS ONE 2014, 9, e91856. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Huang, J.; Ma, Z.; Zhang, J.; Zou, Y.; Gao, L. Inferring drug-disease associations based on known protein complexes. BMC Med. Genom. 2015, 8 (Suppl. S2), S2. [Google Scholar] [CrossRef]
- Lu, J.; Chen, L.; Yin, J.; Huang, T.; Bi, Y.; Kong, X.; Zheng, M.; Cai, Y.D. Identification of new candidate drugs for lung cancer using chemical-chemical interactions, chemical-protein interactions and a K-means clustering algorithm. J. Biomol. Struct. Dyn. 2016, 34, 906–917. [Google Scholar] [CrossRef] [PubMed]
- Subelj, L.; Bajec, M. Unfolding communities in large complex networks: Combining defensive and offensive label propagation for core extraction. Phys. Rev. E Stat. Nonlinear Soft Matter Phys. 2011, 83, 036103. [Google Scholar] [CrossRef] [PubMed]
- Martinez, V.; Navarro, C.; Cano, C.; Fajardo, W.; Blanco, A. DrugNet: Network-based drug-disease prioritization by integrating heterogeneous data. Artif. Intell. Med. 2015, 63, 41–49. [Google Scholar] [CrossRef]
- Fei, T.; Chen, Y.; Xiao, T.; Li, W.; Cato, L.; Zhang, P.; Cotter, M.B.; Bowden, M.; Lis, R.T.; Zhao, S.G.; et al. Genome-wide CRISPR screen identifies HNRNPL as a prostate cancer dependency regulating RNA splicing. Proc. Natl. Acad. Sci. USA 2017, 114, E5207–E5215. [Google Scholar] [CrossRef]
- Lei, H.; Wang, Z.; Jiang, D.; Liu, F.; Liu, M.; Lei, X.; Yang, Y.; He, B.; Yan, M.; Huang, H.; et al. CRISPR screening identifies CDK12 as a conservative vulnerability of prostate cancer. Cell Death Dis. 2021, 12, 740. [Google Scholar] [CrossRef]
- Tsujino, T.; Komura, K.; Inamoto, T.; Azuma, H. CRISPR Screen Contributes to Novel Target Discovery in Prostate Cancer. Int. J. Mol. Sci. 2021, 22, 12777. [Google Scholar] [CrossRef]
- Blackman, J.S.; Knighton, L.E.; Takakuwa, J.E.; Calderwood, S.K.; Truman, A.W. Chemogenomic screening identifies the Hsp70 co-chaperone DNAJA1 as a hub for anticancer drug resistance. Sci. Rep. 2020, 10, 13831. [Google Scholar] [CrossRef]
- Keiser, M.J.; Setola, V.; Irwin, J.J.; Laggner, C.; Abbas, A.I.; Hufeisen, S.J.; Jensen, N.H.; Kuijer, M.B.; Matos, R.C.; Tran, T.B.; et al. Predicting new molecular targets for known drugs. Nature 2009, 462, 175–181. [Google Scholar] [CrossRef]
- Hieronymus, H.; Lamb, J.; Ross, K.N.; Peng, X.P.; Clement, C.; Rodina, A.; Nieto, M.; Du, J.; Stegmaier, K.; Raj, S.M.; et al. Gene expression signature-based chemical genomic prediction identifies a novel class of HSP90 pathway modulators. Cancer Cell 2006, 10, 321–330. [Google Scholar] [CrossRef]
- Iorio, F.; Rittman, T.; Ge, H.; Menden, M.; Saez-Rodriguez, J. Transcriptional data: A new gateway to drug repositioning? Drug Discov. Today 2013, 18, 350–357. [Google Scholar] [CrossRef]
- Dudley, J.T.; Deshpande, T.; Butte, A.J. Exploiting drug-disease relationships for computational drug repositioning. Brief. Bioinform. 2011, 12, 303–311. [Google Scholar] [CrossRef]
- Sirota, M.; Dudley, J.T.; Kim, J.; Chiang, A.P.; Morgan, A.A.; Sweet-Cordero, A.; Sage, J.; Butte, A.J. Discovery and preclinical validation of drug indications using compendia of public gene expression data. Sci. Transl. Med. 2011, 3, 96ra77. [Google Scholar] [CrossRef]
- Dudley, J.T.; Sirota, M.; Shenoy, M.; Pai, R.K.; Roedder, S.; Chiang, A.P.; Morgan, A.A.; Sarwal, M.M.; Pasricha, P.J.; Butte, A.J. Computational repositioning of the anticonvulsant topiramate for inflammatory bowel disease. Sci. Transl. Med. 2011, 3, 96ra76. [Google Scholar] [CrossRef]
- Mirza, N.; Sills, G.J.; Pirmohamed, M.; Marson, A.G. Identifying new antiepileptic drugs through genomics-based drug repurposing. Hum. Mol. Genet. 2017, 26, 527–537. [Google Scholar] [CrossRef]
- Shin, E.; Lee, Y.C.; Kim, S.R.; Kim, S.H.; Park, J. Drug Signature-based Finding of Additional Clinical Use of LC28-0126 for Neutrophilic Bronchial Asthma. Sci. Rep. 2015, 5, 17784. [Google Scholar] [CrossRef]
- Malcomson, B.; Wilson, H.; Veglia, E.; Thillaiyampalam, G.; Barsden, R.; Donegan, S.; El Banna, A.; Elborn, J.S.; Ennis, M.; Kelly, C.; et al. Connectivity mapping (ssCMap) to predict A20-inducing drugs and their antiinflammatory action in cystic fibrosis. Proc. Natl. Acad. Sci. USA 2016, 113, E3725–E3734. [Google Scholar] [CrossRef]
- Wagner, A.; Cohen, N.; Kelder, T.; Amit, U.; Liebman, E.; Steinberg, D.M.; Radonjic, M.; Ruppin, E. Drugs that reverse disease transcriptomic signatures are more effective in a mouse model of dyslipidemia. Mol. Syst. Biol. 2015, 11, 791. [Google Scholar] [CrossRef]
- Wei, G.; Twomey, D.; Lamb, J.; Schlis, K.; Agarwal, J.; Stam, R.W.; Opferman, J.T.; Sallan, S.E.; den Boer, M.L.; Pieters, R.; et al. Gene expression-based chemical genomics identifies rapamycin as a modulator of MCL1 and glucocorticoid resistance. Cancer Cell 2006, 10, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Lamb, J.; Crawford, E.D.; Peck, D.; Modell, J.W.; Blat, I.C.; Wrobel, M.J.; Lerner, J.; Brunet, J.P.; Subramanian, A.; Ross, K.N.; et al. The Connectivity Map: Using gene-expression signatures to connect small molecules, genes, and disease. Science 2006, 313, 1929–1935. [Google Scholar] [CrossRef] [PubMed]
- Oprea, T.I.; Tropsha, A.; Faulon, J.L.; Rintoul, M.D. Systems chemical biology. Nat. Chem. Biol. 2007, 3, 447–450. [Google Scholar] [CrossRef] [PubMed]
- Iorio, F.; Isacchi, A.; di Bernardo, D.; Brunetti-Pierri, N. Identification of small molecules enhancing autophagic function from drug network analysis. Autophagy 2010, 6, 1204–1205. [Google Scholar] [CrossRef]
- Kitchen, D.B.; Decornez, H.; Furr, J.R.; Bajorath, J. Docking and scoring in virtual screening for drug discovery: Methods and applications. Nat. Rev. Drug Discov. 2004, 3, 935–949. [Google Scholar] [CrossRef]
- Lee, A.; Lee, K.; Kim, D. Using reverse docking for target identification and its applications for drug discovery. Expert Opin. Drug Discov. 2016, 11, 707–715. [Google Scholar] [CrossRef] [PubMed]
- Dakshanamurthy, S.; Issa, N.T.; Assefnia, S.; Seshasayee, A.; Peters, O.J.; Madhavan, S.; Uren, A.; Brown, M.L.; Byers, S.W. Predicting new indications for approved drugs using a proteochemometric method. J. Med. Chem. 2012, 55, 6832–6848. [Google Scholar] [CrossRef]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Žídek, A.; Potapenko, A.; et al. Highly accurate protein structure prediction with AlphaFold. Nature 2021, 596, 583–589. [Google Scholar] [CrossRef]
- Campillos, M.; Kuhn, M.; Gavin, A.C.; Jensen, L.J.; Bork, P. Drug target identification using side-effect similarity. Science 2008, 321, 263–266. [Google Scholar] [CrossRef]
- Yang, L.; Agarwal, P. Systematic drug repositioning based on clinical side-effects. PLoS ONE 2011, 6, e28025. [Google Scholar] [CrossRef] [PubMed]
- Jang, G.; Lee, T.; Hwang, S.; Park, C.; Ahn, J.; Seo, S.; Hwang, Y.; Yoon, Y. PISTON: Predicting drug indications and side effects using topic modeling and natural language processing. J. Biomed. Inform. 2018, 87, 96–107. [Google Scholar] [CrossRef] [PubMed]
- Bibbins-Domingo, K.; Force, U.S.P.S.T. Aspirin Use for the Primary Prevention of Cardiovascular Disease and Colorectal Cancer: U.S. Preventive Services Task Force Recommendation Statement. Ann. Intern. Med. 2016, 164, 836–845. [Google Scholar] [CrossRef]
- Cavalla, D.; Singal, C. Retrospective clinical analysis for drug rescue: For new indications or stratified patient groups. Drug Discov. Today 2012, 17, 104–109. [Google Scholar] [CrossRef]
- Jensen, P.B.; Jensen, L.J.; Brunak, S. Mining electronic health records: Towards better research applications and clinical care. Nat. Rev. Genet. 2012, 13, 395–405. [Google Scholar] [CrossRef]
- Paik, H.; Chung, A.Y.; Park, H.C.; Park, R.W.; Suk, K.; Kim, J.; Kim, H.; Lee, K.; Butte, A.J. Repurpose terbutaline sulfate for amyotrophic lateral sclerosis using electronic medical records. Sci. Rep. 2015, 5, 8580. [Google Scholar] [CrossRef]
- Kuusisto, F.; Steill, J.; Kuang, Z.; Thomson, J.; Page, D.; Stewart, R. A Simple Text Mining Approach for Ranking Pairwise Associations in Biomedical Applications. AMIA Summits Transl. Sci. Proc. 2017, 2017, 166–174. [Google Scholar]
- Li, J.; Zhu, X.; Chen, J.Y. Building disease-specific drug-protein connectivity maps from molecular interaction networks and PubMed abstracts. PLoS Comput. Biol. 2009, 5, e1000450. [Google Scholar] [CrossRef]
- Gramatica, R.; Di Matteo, T.; Giorgetti, S.; Barbiani, M.; Bevec, D.; Aste, T. Graph theory enables drug repurposing--how a mathematical model can drive the discovery of hidden mechanisms of action. PLoS ONE 2014, 9, e84912. [Google Scholar] [CrossRef]
- Zhang, M.; Schmitt-Ulms, G.; Sato, C.; Xi, Z.; Zhang, Y.; Zhou, Y.; St George-Hyslop, P.; Rogaeva, E. Drug Repositioning for Alzheimer’s Disease Based on Systematic ’omics’ Data Mining. PLoS ONE 2016, 11, e0168812. [Google Scholar] [CrossRef]
- Hammoud, H.; Saker, Z.; Harati, H.; Fares, Y.; Bahmad, H.F.; Nabha, S. Drug Repurposing in Medulloblastoma: Challenges and Recommendations. Curr. Treat. Options Oncol. 2020, 22, 6. [Google Scholar] [CrossRef]
- Bahmad, H.F.; Elajami, M.K.; El Zarif, T.; Bou-Gharios, J.; Abou-Antoun, T.; Abou-Kheir, W. Drug repurposing towards targeting cancer stem cells in pediatric brain tumors. Cancer Metastasis Rev. 2020, 39, 127–148. [Google Scholar] [CrossRef] [PubMed]
- Bahmad, H.F.; Daher, D.; Aljamal, A.A.; Elajami, M.K.; Oh, K.S.; Alvarez Moreno, J.C.; Delgado, R.; Suarez, R.; Zaldivar, A.; Azimi, R.; et al. Repurposing of Anticancer Stem Cell Drugs in Brain Tumors. J. Histochem. Cytochem. 2021, 69, 749–773. [Google Scholar] [CrossRef] [PubMed]
- Kaitin, K.I.; DiMasi, J.A. Pharmaceutical innovation in the 21st century: New drug approvals in the first decade, 2000–2009. Clin. Pharmacol. Ther. 2011, 89, 183–188. [Google Scholar] [CrossRef]
- Nosengo, N. Can you teach old drugs new tricks? Nature 2016, 534, 314–316. [Google Scholar] [CrossRef]
- Hamilton, A.S.; Albertsen, P.C.; Johnson, T.K.; Hoffman, R.; Morrell, D.; Deapen, D.; Penson, D.F. Trends in the treatment of localized prostate cancer using supplemented cancer registry data. BJU Int. 2011, 107, 576–584. [Google Scholar] [CrossRef]
- Jacobs, B.L.; Zhang, Y.; Schroeck, F.R.; Skolarus, T.A.; Wei, J.T.; Montie, J.E.; Gilbert, S.M.; Strope, S.A.; Dunn, R.L.; Miller, D.C.; et al. Use of advanced treatment technologies among men at low risk of dying from prostate cancer. JAMA 2013, 309, 2587–2595. [Google Scholar] [CrossRef]
- Walz, J.; Joniau, S.; Chun, F.K.; Isbarn, H.; Jeldres, C.; Yossepowitch, O.; Chao-Yu, H.; Klein, E.A.; Scardino, P.T.; Reuther, A.; et al. Pathological results and rates of treatment failure in high-risk prostate cancer patients after radical prostatectomy. BJU Int. 2011, 107, 765–770. [Google Scholar] [CrossRef]
- Yossepowitch, O.; Eggener, S.E.; Bianco, F.J., Jr.; Carver, B.S.; Serio, A.; Scardino, P.T.; Eastham, J.A. Radical prostatectomy for clinically localized, high risk prostate cancer: Critical analysis of risk assessment methods. J. Urol. 2007, 178, 493–499. [Google Scholar] [CrossRef]
- Bill-Axelson, A.; Holmberg, L.; Ruutu, M.; Garmo, H.; Stark, J.R.; Busch, C.; Nordling, S.; Haggman, M.; Andersson, S.O.; Bratell, S.; et al. Radical prostatectomy versus watchful waiting in early prostate cancer. N. Engl. J. Med. 2011, 364, 1708–1717. [Google Scholar] [CrossRef]
- Wilt, T.J.; Brawer, M.K.; Jones, K.M.; Barry, M.J.; Aronson, W.J.; Fox, S.; Gingrich, J.R.; Wei, J.T.; Gilhooly, P.; Grob, B.M.; et al. Radical prostatectomy versus observation for localized prostate cancer. N. Engl. J. Med. 2012, 367, 203–213. [Google Scholar] [CrossRef]
- Kelloff, G.J.; Choyke, P.; Coffey, D.S.; Prostate Cancer Imaging Working, G. Challenges in clinical prostate cancer: Role of imaging. AJR Am. J. Roentgenol. 2009, 192, 1455–1470. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.C.; Choueiri, M.; Tu, S.M.; Lin, S.H. Biology and clinical management of prostate cancer bone metastasis. Front. Biosci. 2007, 12, 3273–3286. [Google Scholar] [CrossRef] [PubMed]
- Mollica, V.; Rizzo, A.; Rosellini, M.; Marchetti, A.; Ricci, A.D.; Cimadamore, A.; Scarpelli, M.; Bonucci, C.; Andrini, E.; Errani, C.; et al. Bone Targeting Agents in Patients with Metastatic Prostate Cancer: State of the Art. Cancers 2021, 13, 546. [Google Scholar] [CrossRef] [PubMed]
- Suzman, D.L.; Boikos, S.A.; Carducci, M.A. Bone-targeting agents in prostate cancer. Cancer Metastasis Rev. 2014, 33, 619–628. [Google Scholar] [CrossRef] [PubMed]
- Markowski, M.C.; Carducci, M.A. Early use of chemotherapy in metastatic prostate cancer. Cancer Treat. Rev. 2017, 55, 218–224. [Google Scholar] [CrossRef]
- Silberstein, J.L.; Pal, S.K.; Lewis, B.; Sartor, O. Current clinical challenges in prostate cancer. Transl. Androl. Urol. 2013, 2, 122–136. [Google Scholar] [CrossRef]
- Bostwick, D.G. Prostate-specific antigen. Current role in diagnostic pathology of prostate cancer. Am. J. Clin. Pathol. 1994, 102, S31–S37. [Google Scholar]
- Nogueira, L.; Corradi, R.; Eastham, J.A. Prostatic specific antigen for prostate cancer detection. Int. Braz. J. Urol. 2009, 35, 521–529; discussion 522–530. [Google Scholar] [CrossRef]
- Wright, J.L.; Lange, P.H. Newer potential biomarkers in prostate cancer. Rev. Urol. 2007, 9, 207–213. [Google Scholar]
- Gretzer, M.B.; Partin, A.W. PSA markers in prostate cancer detection. Urol. Clin. N. Am. 2003, 30, 677–686. [Google Scholar] [CrossRef]
- Dall’Era, M.A.; Cooperberg, M.R.; Chan, J.M.; Davies, B.J.; Albertsen, P.C.; Klotz, L.H.; Warlick, C.A.; Holmberg, L.; Bailey, D.E., Jr.; Wallace, M.E.; et al. Active surveillance for early-stage prostate cancer: Review of the current literature. Cancer 2008, 112, 1650–1659. [Google Scholar] [CrossRef]
- Hughes, C.; Murphy, A.; Martin, C.; Sheils, O.; O’Leary, J. Molecular pathology of prostate cancer. J. Clin. Pathol. 2005, 58, 673–684. [Google Scholar] [CrossRef] [PubMed]
- Kastner, C.; Armitage, J.; Kimble, A.; Rawal, J.; Carter, P.G.; Venn, S. The Charlson comorbidity score: A superior comorbidity assessment tool for the prostate cancer multidisciplinary meeting. Prostate Cancer Prostatic Dis. 2006, 9, 270–274. [Google Scholar] [CrossRef] [PubMed]
- Cowen, M.E.; Halasyamani, L.K.; Kattan, M.W. Predicting life expectancy in men with clinically localized prostate cancer. J. Urol. 2006, 175, 99–103. [Google Scholar] [CrossRef]
- Tewari, A.; Johnson, C.C.; Divine, G.; Crawford, E.D.; Gamito, E.J.; Demers, R.; Menon, M. Long-term survival probability in men with clinically localized prostate cancer: A case-control, propensity modeling study stratified by race, age, treatment and comorbidities. J. Urol. 2004, 171, 1513–1519. [Google Scholar] [CrossRef]
- Walz, J.; Gallina, A.; Saad, F.; Montorsi, F.; Perrotte, P.; Shariat, S.F.; Jeldres, C.; Graefen, M.; Benard, F.; McCormack, M.; et al. A nomogram predicting 10-year life expectancy in candidates for radical prostatectomy or radiotherapy for prostate cancer. J. Clin. Oncol. 2007, 25, 3576–3581. [Google Scholar] [CrossRef]
- Shariat, S.F.; Scardino, P.T.; Lilja, H. Screening for prostate cancer: An update. Can. J. Urol. 2008, 15, 4363–4374. [Google Scholar]
- Eichelberger, L.E.; Cheng, L. Does pT2b prostate carcinoma exist? Critical appraisal of the 2002 TNM classification of prostate carcinoma. Cancer 2004, 100, 2573–2576. [Google Scholar] [CrossRef]
- Rajinikanth, A.; Manoharan, M.; Soloway, C.T.; Civantos, F.J.; Soloway, M.S. Trends in Gleason score: Concordance between biopsy and prostatectomy over 15 years. Urology 2008, 72, 177–182. [Google Scholar] [CrossRef]
- Linden, R.A.; Halpern, E.J. Advances in transrectal ultrasound imaging of the prostate. Semin. Ultrasound CT MRI 2007, 28, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Nurgali, K.; Jagoe, R.T.; Abalo, R. Editorial: Adverse Effects of Cancer Chemotherapy: Anything New to Improve Tolerance and Reduce Sequelae? Front. Pharmacol. 2018, 9, 245. [Google Scholar] [CrossRef] [PubMed]
- Hafeez, B.B.; Ganju, A.; Sikander, M.; Kashyap, V.K.; Hafeez, Z.B.; Chauhan, N.; Malik, S.; Massey, A.E.; Tripathi, M.K.; Halaweish, F.T.; et al. Ormeloxifene Suppresses Prostate Tumor Growth and Metastatic Phenotypes via Inhibition of Oncogenic beta-catenin Signaling and EMT Progression. Mol. Cancer Ther. 2017, 16, 2267–2280. [Google Scholar] [CrossRef] [PubMed]
- Masumori, N. Naftopidil for the treatment of urinary symptoms in patients with benign prostatic hyperplasia. Ther. Clin. Risk Manag. 2011, 7, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, Y.; Ishii, K.; Kanda, H.; Kato, M.; Miki, M.; Kajiwara, S.; Arima, K.; Shiraishi, T.; Sugimura, Y. Combination treatment with naftopidil increases the efficacy of radiotherapy in PC-3 human prostate cancer cells. J. Cancer Res. Clin. Oncol. 2017, 143, 933–939. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.-Y.; Huang, W.-Y.; Lin, C.-L.; Huang, T.-C.; Wu, Y.-Y.; Chen, J.-H.; Kao, C.-H. Propranolol Reduces Cancer Risk: A Population-Based Cohort Study. Medicine 2015, 94, e1097. [Google Scholar] [CrossRef] [PubMed]
- Simpson, C.D.; Mawji, I.A.; Anyiwe, K.; Williams, M.A.; Wang, X.; Venugopal, A.L.; Gronda, M.; Hurren, R.; Cheng, S.; Serra, S.; et al. Inhibition of the Sodium Potassium Adenosine Triphosphatase Pump Sensitizes Cancer Cells to Anoikis and Prevents Distant Tumor Formation. Cancer Res. 2009, 69, 2739. [Google Scholar] [CrossRef]
- Brohée, L.; Peulen, O.; Nusgens, B.; Castronovo, V.; Thiry, M.; Colige, A.C.; Deroanne, C.F. Propranolol sensitizes prostate cancer cells to glucose metabolism inhibition and prevents cancer progression. Sci. Rep. 2018, 8, 7050. [Google Scholar] [CrossRef]
- Olivan, M.; Rigau, M.; Colás, E.; Garcia, M.; Montes, M.; Sequeiros, T.; Regis, L.; Celma, A.; Planas, J.; Placer, J.; et al. Simultaneous treatment with statins and aspirin reduces the risk of prostate cancer detection and tumorigenic properties in prostate cancer cell lines. BioMed Res. Int. 2015, 2015, 762178. [Google Scholar] [CrossRef]
- Hsu, A.L.; Ching, T.T.; Wang, D.S.; Song, X.; Rangnekar, V.M.; Chen, C.S. The cyclooxygenase-2 inhibitor celecoxib induces apoptosis by blocking Akt activation in human prostate cancer cells independently of Bcl-2. J. Biol. Chem. 2000, 275, 11397–11403. [Google Scholar] [CrossRef]
- Gayvert, K.M.; Dardenne, E.; Cheung, C.; Boland, M.R.; Lorberbaum, T.; Wanjala, J.; Chen, Y.; Rubin, M.A.; Tatonetti, N.P.; Rickman, D.S.; et al. A Computational Drug Repositioning Approach for Targeting Oncogenic Transcription Factors. Cell Rep. 2016, 15, 2348–2356. [Google Scholar] [CrossRef] [PubMed]
- Iannelli, F.; Roca, M.S.; Lombardi, R.; Ciardiello, C.; Grumetti, L.; De Rienzo, S.; Moccia, T.; Vitagliano, C.; Sorice, A.; Costantini, S.; et al. Synergistic antitumor interaction of valproic acid and simvastatin sensitizes prostate cancer to docetaxel by targeting CSCs compartment via YAP inhibition. J. Exp. Clin. Cancer Res. 2020, 39, 213. [Google Scholar] [CrossRef]
- Salem, O.; Hansen, C.G. The Hippo Pathway in Prostate Cancer. Cells 2019, 8, 370. [Google Scholar] [CrossRef] [PubMed]
- Kong, Y.; Cheng, L.; Mao, F.; Zhang, Z.; Zhang, Y.; Farah, E.; Bosler, J.; Bai, Y.; Ahmad, N.; Kuang, S.; et al. Inhibition of cholesterol biosynthesis overcomes enzalutamide resistance in castration-resistant prostate cancer (CRPC). J. Biol. Chem. 2018, 293, 14328–14341. [Google Scholar] [CrossRef] [PubMed]
- Zingales, V.; Distefano, A.; Raffaele, M.; Zanghi, A.; Barbagallo, I.; Vanella, L. Metformin: A Bridge between Diabetes and Prostate Cancer. Front. Oncol. 2017, 7, 243. [Google Scholar] [CrossRef]
- Zaidi, S.; Gandhi, J.; Joshi, G.; Smith, N.L.; Khan, S.A. The anticancer potential of metformin on prostate cancer. Prostate Cancer Prostatic Dis. 2019, 22, 351–361. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, G.; Tong, D.; Parmar, H.; Hasenmayer, D.; Yuan, W.; Zhang, D.; Jiang, J. Metformin represses androgen-dependent and androgen-independent prostate cancers by targeting androgen receptor. Prostate 2015, 75, 1187–1196. [Google Scholar] [CrossRef]
- Qi, C.; Bin, L.; Yang, Y.; Yang, Y.; Li, J.; Zhou, Q.; Wen, Y.; Zeng, C.; Zheng, L.; Zhang, Q.; et al. Glipizide suppresses prostate cancer progression in the TRAMP model by inhibiting angiogenesis. Sci. Rep. 2016, 6, 27819. [Google Scholar] [CrossRef]
- Rushworth, L.K.; Hewit, K.; Munnings-Tomes, S.; Somani, S.; James, D.; Shanks, E.; Dufès, C.; Straube, A.; Patel, R.; Leung, H.Y. Repurposing screen identifies mebendazole as a clinical candidate to synergise with docetaxel for prostate cancer treatment. Br. J. Cancer 2020, 122, 517–527. [Google Scholar] [CrossRef]
- Liu, C.; Lou, W.; Zhu, Y.; Nadiminty, N.; Schwartz, C.T.; Evans, C.P.; Gao, A.C. Niclosamide inhibits androgen receptor variants expression and overcomes enzalutamide resistance in castration-resistant prostate cancer. Clin. Cancer Res. 2014, 20, 3198–3210. [Google Scholar] [CrossRef]
- Chow, W.A.; Jiang, C.; Guan, M. Anti-HIV drugs for cancer therapeutics: Back to the future? Lancet Oncol. 2009, 10, 61–71. [Google Scholar] [CrossRef]
- Guan, M.; Su, L.; Yuan, Y.C.; Li, H.; Chow, W.A. Nelfinavir and nelfinavir analogs block site-2 protease cleavage to inhibit castration-resistant prostate cancer. Sci. Rep. 2015, 5, 9698. [Google Scholar] [CrossRef]
- Lokeshwar, B.L. Chemically modified non-antimicrobial tetracyclines are multifunctional drugs against advanced cancers. Pharmacol. Res. 2011, 63, 146–150. [Google Scholar] [CrossRef]
- Corey, E.; Brown, L.G.; Quinn, J.E.; Poot, M.; Roudier, M.P.; Higano, C.S.; Vessella, R.L. Zoledronic acid exhibits inhibitory effects on osteoblastic and osteolytic metastases of prostate cancer. Clin. Cancer Res. 2003, 9, 295–306. [Google Scholar] [PubMed]
- Xia, Q.; Sung, J.; Chowdhury, W.; Chen, C.L.; Höti, N.; Shabbeer, S.; Carducci, M.; Rodriguez, R. Chronic administration of valproic acid inhibits prostate cancer cell growth in vitro and in vivo. Cancer Res. 2006, 66, 7237–7244. [Google Scholar] [CrossRef]
- El Etreby, M.F.; Liang, Y.; Lewis, R.W. Induction of apoptosis by mifepristone and tamoxifen in human LNCaP prostate cancer cells in culture. Prostate 2000, 43, 31–42. [Google Scholar] [CrossRef]
- Sosic, I.; Mirkovic, B.; Arenz, K.; Stefane, B.; Kos, J.; Gobec, S. Development of new cathepsin B inhibitors: Combining bioisosteric replacements and structure-based design to explore the structure-activity relationships of nitroxoline derivatives. J. Med. Chem. 2013, 56, 521–533. [Google Scholar] [CrossRef] [PubMed]
- Shim, J.S.; Matsui, Y.; Bhat, S.; Nacev, B.A.; Xu, J.; Bhang, H.E.; Dhara, S.; Han, K.C.; Chong, C.R.; Pomper, M.G.; et al. Effect of nitroxoline on angiogenesis and growth of human bladder cancer. J. Natl. Cancer Inst. 2010, 102, 1855–1873. [Google Scholar] [CrossRef]
- Chang, W.L.; Hsu, L.C.; Leu, W.J.; Chen, C.S.; Guh, J.H. Repurposing of nitroxoline as a potential anticancer agent against human prostate cancer: A crucial role on AMPK/mTOR signaling pathway and the interplay with Chk2 activation. Oncotarget 2015, 6, 39806–39820. [Google Scholar] [CrossRef]
- Lv, Z.; Chu, Y.; Wang, Y. HIV protease inhibitors: A review of molecular selectivity and toxicity. HIV AIDS 2015, 7, 95–104. [Google Scholar] [CrossRef]
- Gantt, S.; Casper, C.; Ambinder, R.F. Insights into the broad cellular effects of nelfinavir and the HIV protease inhibitors supporting their role in cancer treatment and prevention. Curr. Opin. Oncol. 2013, 25, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Qian, D.Z.; Tan, Y.S.; Lee, K.; Gao, P.; Ren, Y.R.; Rey, S.; Hammers, H.; Chang, D.; Pili, R.; et al. Digoxin and other cardiac glycosides inhibit HIF-1alpha synthesis and block tumor growth. Proc. Natl. Acad. Sci. USA 2008, 105, 19579–19586. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Zhan, T.; Duffy, D.; Hoffman-Censits, J.; Kilpatrick, D.; Trabulsi, E.J.; Lallas, C.D.; Chervoneva, I.; Limentani, K.; Kennedy, B.; et al. A pilot phase II Study of digoxin in patients with recurrent prostate cancer as evident by a rising PSA. Am. J. Cancer Ther. Pharmacol. 2014, 2, 21–32. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, M.; Shim, J.S.; Li, R.J.; Dang, Y.; He, Q.; Das, M.; Liu, J.O. Identification of an old antibiotic clofoctol as a novel activator of unfolded protein response pathways and an inhibitor of prostate cancer. Br. J. Pharmacol. 2014, 171, 4478–4489. [Google Scholar] [CrossRef] [PubMed]
- Lupu, R.; Menendez, J.A. Pharmacological inhibitors of Fatty Acid Synthase (FASN)--catalyzed endogenous fatty acid biogenesis: A new family of anti-cancer agents? Curr. Pharm. Biotechnol. 2006, 7, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Sadowski, M.C.; Pouwer, R.H.; Gunter, J.H.; Lubik, A.A.; Quinn, R.J.; Nelson, C.C. The fatty acid synthase inhibitor triclosan: Repurposing an anti-microbial agent for targeting prostate cancer. Oncotarget 2014, 5, 9362–9381. [Google Scholar] [CrossRef] [PubMed]
- Rodricks, J.V.; Swenberg, J.A.; Borzelleca, J.F.; Maronpot, R.R.; Shipp, A.M. Triclosan: A critical review of the experimental data and development of margins of safety for consumer products. Crit. Rev. Toxicol. 2010, 40, 422–484. [Google Scholar] [CrossRef]
- Abou El-Magd, R.M.; Park, H.K.; Kawazoe, T.; Iwana, S.; Ono, K.; Chung, S.P.; Miyano, M.; Yorita, K.; Sakai, T.; Fukui, K. The effect of risperidone on D-amino acid oxidase activity as a hypothesis for a novel mechanism of action in the treatment of schizophrenia. J. Psychopharmacol. 2010, 24, 1055–1067. [Google Scholar] [CrossRef]
- Ayan, D.; Maltais, R.; Poirier, D. Identification of a 17beta-hydroxysteroid dehydrogenase type 10 steroidal inhibitor: A tool to investigate the role of type 10 in Alzheimer’s disease and prostate cancer. ChemMedChem 2012, 7, 1181–1184. [Google Scholar] [CrossRef]
- Tammali, R.; Srivastava, S.K.; Ramana, K.V. Targeting aldose reductase for the treatment of cancer. Curr. Cancer Drug Targets 2011, 11, 560–571. [Google Scholar] [CrossRef]
- Turanli, B.; Gulfidan, G.; Arga, K.Y. Transcriptomic-Guided Drug Repositioning Supported by a New Bioinformatics Search Tool: GeneXpharma. OMICS 2017, 21, 584–591. [Google Scholar] [CrossRef] [PubMed]
- Frishman, W.H. Beta-Adrenergic Receptor Blockers in Hypertension: Alive and Well. Prog. Cardiovasc. Dis. 2016, 59, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Albert, D.; Pergola, C.; Koeberle, A.; Dodt, G.; Steinhilber, D.; Werz, O. The role of diacylglyceride generation by phospholipase D and phosphatidic acid phosphatase in the activation of 5-lipoxygenase in polymorphonuclear leukocytes. J. Leukoc. Biol. 2008, 83, 1019–1027. [Google Scholar] [CrossRef] [PubMed]
- Kirkendall, W.M.; Hammond, J.J.; Thomas, J.C.; Overturf, M.L.; Zama, A. Prazosin and clonidine for moderately severe hypertension. JAMA 1978, 240, 2553–2556. [Google Scholar] [CrossRef]
- Clifford, G.M.; Farmer, R.D. Medical therapy for benign prostatic hyperplasia: A review of the literature. Eur. Urol. 2000, 38, 2–19. [Google Scholar] [CrossRef]
- Waldo, R. Prazosin relieves Raynaud’s vasospasm. JAMA 1979, 241, 1037. [Google Scholar] [CrossRef]
- Assad Kahn, S.; Costa, S.L.; Gholamin, S.; Nitta, R.T.; Dubois, L.G.; Feve, M.; Zeniou, M.; Coelho, P.L.; El-Habr, E.; Cadusseau, J.; et al. The anti-hypertensive drug prazosin inhibits glioblastoma growth via the PKCdelta-dependent inhibition of the AKT pathway. EMBO Mol. Med. 2016, 8, 511–526. [Google Scholar] [CrossRef]
- Lin, S.-C.; Chueh, S.-C.; Hsiao, C.-J.; Li, T.-K.; Chen, T.-H.; Liao, C.-H.; Lyu, P.-C.; Guh, J.-H. Prazosin displays anticancer activity against human prostate cancers: Targeting DNA and cell cycle. Neoplasia 2007, 9, 830–839. [Google Scholar] [CrossRef]
- Bacchi, S.; Palumbo, P.; Sponta, A.; Coppolino, M.F. Clinical Pharmacology of Non-Steroidal Anti-Inflammatory Drugs: A Review. Anti-Inflamm. Anti-Allergy Agents Med. Chem. 2012, 11, 52–64. [Google Scholar] [CrossRef]
- Bilani, N.; Bahmad, H.; Abou-Kheir, W. Prostate Cancer and Aspirin Use: Synopsis of the Proposed Molecular Mechanisms. Front. Pharmacol. 2017, 8, 145. [Google Scholar] [CrossRef]
- Janik, J.E.; Miller, L.L.; Longo, D.L.; Powers, G.C.; Urba, W.J.; Kopp, W.C.; Gause, B.L.; Curti, B.D.; Fenton, R.G.; Oppenheim, J.J.; et al. Phase II trial of interleukin 1 alpha and indomethacin in treatment of metastatic melanoma. J. Natl. Cancer Inst. 1996, 88, 44–49. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Timar, J.; Ladanyi, A.; Forster-Horvath, C.; Lukits, J.; Dome, B.; Remenar, E.; Godeny, M.; Kasler, M.; Bencsik, B.; Repassy, G.; et al. Neoadjuvant immunotherapy of oral squamous cell carcinoma modulates intratumoral CD4/CD8 ratio and tumor microenvironment: A multicenter phase II clinical trial. J. Clin. Oncol. 2005, 23, 3421–3432. [Google Scholar] [CrossRef] [PubMed]
- Guieu, R.; Blin, O.; Pouget, J.; Serratrice, G. Analgesic effect of indomethacin shown using the nociceptive flexion reflex in humans. Ann. Rheum. Dis. 1992, 51, 391–393. [Google Scholar] [CrossRef] [PubMed]
- Percy, J.S.; Stephenson, P.; Thompson, M. Indomethacin in the Treatment of Rheumatic Diseases. Ann. Rheum. Dis. 1964, 23, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Chennamaneni, S.; Zhong, B.; Lama, R.; Su, B. COX inhibitors Indomethacin and Sulindac derivatives as antiproliferative agents: Synthesis, biological evaluation, and mechanism investigation. Eur. J. Med. Chem. 2012, 56, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Touhey, S.; O’Connor, R.; Plunkett, S.; Maguire, A.; Clynes, M. Structure-activity relationship of indomethacin analogues for MRP-1, COX-1 and COX-2 inhibition. identification of novel chemotherapeutic drug resistance modulators. Eur. J. Cancer 2002, 38, 1661–1670. [Google Scholar] [CrossRef]
- Lin, C.C.; Suen, K.M.; Stainthorp, A.; Wieteska, L.; Biggs, G.S.; Leitao, A.; Montanari, C.A.; Ladbury, J.E. Targeting the Shc-EGFR interaction with indomethacin inhibits MAP kinase pathway signalling. Cancer Lett. 2019, 457, 86–97. [Google Scholar] [CrossRef]
- Mazumder, S.; De, R.; Debsharma, S.; Bindu, S.; Maity, P.; Sarkar, S.; Saha, S.J.; Siddiqui, A.A.; Banerjee, C.; Nag, S.; et al. Indomethacin impairs mitochondrial dynamics by activating the PKCzeta-p38-DRP1 pathway and inducing apoptosis in gastric cancer and normal mucosal cells. J. Biol. Chem. 2019, 294, 8238–8258. [Google Scholar] [CrossRef]
- Federica, I.; Rita, L.; Maria, R.M.; Biagio, P.; Simona De, R.; Alfredo, B.; Francesca, B. Targeting Mevalonate Pathway in Cancer Treatment: Repurposing of Statins. Recent Pat. Anti-Cancer Drug Discov. 2018, 13, 184–200. [Google Scholar] [CrossRef]
- Hindler, K.; Cleeland, C.S.; Rivera, E.; Collard, C.D. The role of statins in cancer therapy. Oncologist 2006, 11, 306–315. [Google Scholar] [CrossRef]
- Chan, K.K.; Oza, A.M.; Siu, L.L. The statins as anticancer agents. Clin. Cancer Res. 2003, 9, 10–19. [Google Scholar] [PubMed]
- Zhuang, L.; Kim, J.; Adam, R.M.; Solomon, K.R.; Freeman, M.R. Cholesterol targeting alters lipid raft composition and cell survival in prostate cancer cells and xenografts. J. Clin. Investig. 2005, 115, 959–968. [Google Scholar] [CrossRef] [PubMed]
- Tan, N.; Klein, E.A.; Li, J.; Moussa, A.S.; Jones, J.S. Statin use and risk of prostate cancer in a population of men who underwent biopsy. J. Urol. 2011, 186, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Katz, M.S.; Carroll, P.R.; Cowan, J.E.; Chan, J.M.; D’Amico, A.V. Association of statin and nonsteroidal anti-inflammatory drug use with prostate cancer outcomes: Results from CaPSURE. BJU Int. 2010, 106, 627–632. [Google Scholar] [CrossRef] [PubMed]
- Marcella, S.W.; David, A.; Ohman-Strickland, P.A.; Carson, J.; Rhoads, G.G. Statin use and fatal prostate cancer: A matched case-control study. Cancer 2012, 118, 4046–4052. [Google Scholar] [CrossRef]
- Hutchinson, J.; Marignol, L. Clinical Potential of Statins in Prostate Cancer Radiation Therapy. Anticancer Res. 2017, 37, 5363–5372. [Google Scholar] [CrossRef][Green Version]
- Rena, G.; Hardie, D.G.; Pearson, E.R. The mechanisms of action of metformin. Diabetologia 2017, 60, 1577–1585. [Google Scholar] [CrossRef]
- Evans, J.M.; Donnelly, L.A.; Emslie-Smith, A.M.; Alessi, D.R.; Morris, A.D. Metformin and reduced risk of cancer in diabetic patients. BMJ 2005, 330, 1304–1305. [Google Scholar] [CrossRef]
- Whitburn, J.; Edwards, C.M.; Sooriakumaran, P. Metformin and Prostate Cancer: A New Role for an Old Drug. Curr. Urol. Rep. 2017, 18, 46. [Google Scholar] [CrossRef]
- Mouhieddine, T.H.; Nokkari, A.; Itani, M.M.; Chamaa, F.; Bahmad, H.; Monzer, A.; El-Merahbi, R.; Daoud, G.; Eid, A.; Kobeissy, F.H.; et al. Metformin and Ara-a Effectively Suppress Brain Cancer by Targeting Cancer Stem/Progenitor Cells. Front. Neurosci. 2015, 9, 442. [Google Scholar] [CrossRef]
- Jalving, M.; Gietema, J.A.; Lefrandt, J.D.; de Jong, S.; Reyners, A.K.; Gans, R.O.; de Vries, E.G. Metformin: Taking away the candy for cancer? Eur. J. Cancer 2010, 46, 2369–2380. [Google Scholar] [CrossRef] [PubMed]
- Preston, M.A.; Riis, A.H.; Ehrenstein, V.; Breau, R.H.; Batista, J.L.; Olumi, A.F.; Mucci, L.A.; Adami, H.O.; Sorensen, H.T. Metformin use and prostate cancer risk. Eur. Urol. 2014, 66, 1012–1020. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, H.A.; Iliopoulos, D.; Struhl, K. Metformin inhibits the inflammatory response associated with cellular transformation and cancer stem cell growth. Proc. Natl. Acad. Sci. USA 2013, 110, 972–977. [Google Scholar] [CrossRef] [PubMed]
- Tran, L.N.K.; Kichenadasse, G.; Butler, L.M.; Centenera, M.M.; Morel, K.L.; Ormsby, R.J.; Michael, M.Z.; Lower, K.M.; Sykes, P.J. The Combination of Metformin and Valproic Acid Induces Synergistic Apoptosis in the Presence of p53 and Androgen Signaling in Prostate Cancer. Mol. Cancer Ther. 2017, 16, 2689–2700. [Google Scholar] [CrossRef]
- Gallagher, E.J.; LeRoith, D. Obesity and Diabetes: The Increased Risk of Cancer and Cancer-Related Mortality. Physiol. Rev. 2015, 95, 727–748. [Google Scholar] [CrossRef] [PubMed]
- Bronsveld, H.K.; De Bruin, M.L.; Wesseling, J.; Sanders, J.; Hofland, I.; Jensen, V.; Bazelier, M.T.; Ter Braak, B.; de Boer, A.; Vestergaard, P.; et al. The association of diabetes mellitus and insulin treatment with expression of insulin-related proteins in breast tumors. BMC Cancer 2018, 18, 224. [Google Scholar] [CrossRef]
- Pollak, M. Overcoming Drug Development Bottlenecks With Repurposing: Repurposing biguanides to target energy metabolism for cancer treatment. Nat. Med. 2014, 20, 591–593. [Google Scholar] [CrossRef]
- Howell, J.J.; Hellberg, K.; Turner, M.; Talbott, G.; Kolar, M.J.; Ross, D.S.; Hoxhaj, G.; Saghatelian, A.; Shaw, R.J.; Manning, B.D. Metformin Inhibits Hepatic mTORC1 Signaling via Dose-Dependent Mechanisms Involving AMPK and the TSC Complex. Cell Metab. 2017, 25, 463–471. [Google Scholar] [CrossRef]
- James, N.D.; Sydes, M.R.; Clarke, N.W.; Mason, M.D.; Dearnaley, D.P.; Spears, M.R.; Ritchie, A.W.; Parker, C.C.; Russell, J.M.; Attard, G.; et al. Addition of docetaxel, zoledronic acid, or both to first-line long-term hormone therapy in prostate cancer (STAMPEDE): Survival results from an adaptive, multiarm, multistage, platform randomised controlled trial. Lancet 2016, 387, 1163–1177. [Google Scholar] [CrossRef]
- Circu, M.L.; Dykes, S.S.; Carroll, J.; Kelly, K.; Galiano, F.; Greer, A.; Cardelli, J.; El-Osta, H. A Novel High Content Imaging-Based Screen Identifies the Anti-Helminthic Niclosamide as an Inhibitor of Lysosome Anterograde Trafficking and Prostate Cancer Cell Invasion. PLoS ONE 2016, 11, e0146931. [Google Scholar] [CrossRef]
- Chen, W.; Mook, R.A., Jr.; Premont, R.T.; Wang, J. Niclosamide: Beyond an antihelminthic drug. Cell. Signal. 2018, 41, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Sack, U.; Walther, W.; Scudiero, D.; Selby, M.; Kobelt, D.; Lemm, M.; Fichtner, I.; Schlag, P.M.; Shoemaker, R.H.; Stein, U. Novel effect of antihelminthic Niclosamide on S100A4-mediated metastatic progression in colon cancer. J. Natl. Cancer Inst. 2011, 103, 1018–1036. [Google Scholar] [CrossRef] [PubMed]
- Dahlmann, M.; Kobelt, D.; Walther, W.; Mudduluru, G.; Stein, U. S100A4 in Cancer Metastasis: Wnt Signaling-Driven Interventions for Metastasis Restriction. Cancers 2016, 8, 59. [Google Scholar] [CrossRef] [PubMed]
- Ye, T.; Xiong, Y.; Yan, Y.; Xia, Y.; Song, X.; Liu, L.; Li, D.; Wang, N.; Zhang, L.; Zhu, Y.; et al. The anthelmintic drug niclosamide induces apoptosis, impairs metastasis and reduces immunosuppressive cells in breast cancer model. PLoS ONE 2014, 9, e85887. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chen, X.; Ward, T.; Mao, Y.; Bockhorn, J.; Liu, X.; Wang, G.; Pegram, M.; Shen, K. Niclosamide inhibits epithelial-mesenchymal transition and tumor growth in lapatinib-resistant human epidermal growth factor receptor 2-positive breast cancer. Int. J. Biochem. Cell Biol. 2016, 71, 12–23. [Google Scholar] [CrossRef]
- Guo, C.; Yeh, S.; Niu, Y.; Li, G.; Zheng, J.; Li, L.; Chang, C. Targeting androgen receptor versus targeting androgens to suppress castration resistant prostate cancer. Cancer Lett. 2017, 397, 133–143. [Google Scholar] [CrossRef]
- Garrido-Mesa, N.; Zarzuelo, A.; Gálvez, J. Minocycline: Far beyond an antibiotic. Br. J. Pharmacol. 2013, 169, 337–352. [Google Scholar] [CrossRef]
- Antonarakis, E.S.; Heath, E.I.; Smith, D.C.; Rathkopf, D.; Blackford, A.L.; Danila, D.C.; King, S.; Frost, A.; Ajiboye, A.S.; Zhao, M.; et al. Repurposing itraconazole as a treatment for advanced prostate cancer: A noncomparative randomized phase II trial in men with metastatic castration-resistant prostate cancer. Oncologist 2013, 18, 163–173. [Google Scholar] [CrossRef]
- Karhadkar, S.S.; Bova, G.S.; Abdallah, N.; Dhara, S.; Gardner, D.; Maitra, A.; Isaacs, J.T.; Berman, D.M.; Beachy, P.A. Hedgehog signalling in prostate regeneration, neoplasia and metastasis. Nature 2004, 431, 707–712. [Google Scholar] [CrossRef]
- Nacev, B.A.; Grassi, P.; Dell, A.; Haslam, S.M.; Liu, J.O. The antifungal drug itraconazole inhibits vascular endothelial growth factor receptor 2 (VEGFR2) glycosylation, trafficking, and signaling in endothelial cells. J. Biol. Chem. 2011, 286, 44045–44056. [Google Scholar] [CrossRef]
- Head, S.A.; Shi, W.Q.; Yang, E.J.; Nacev, B.A.; Hong, S.Y.; Pasunooti, K.K.; Li, R.J.; Shim, J.S.; Liu, J.O. Simultaneous Targeting of NPC1 and VDAC1 by Itraconazole Leads to Synergistic Inhibition of mTOR Signaling and Angiogenesis. ACS Chem. Biol. 2017, 12, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhuang, K.; Sun, K.; Yang, Q.; Ran, X.; Xu, X.; Mu, C.; Zheng, B.; Lu, Y.; Zeng, J.; et al. Itraconazole Induces Regression of Infantile Hemangioma via Downregulation of the Platelet-Derived Growth Factor-D/PI3K/Akt/mTOR Pathway. J. Investig. Dermatol. 2019, 139, 1574–1582. [Google Scholar] [CrossRef] [PubMed]
- Smithuis, F.; Kyaw, M.K.; Phe, O.; Win, T.; Aung, P.P.; Oo, A.P.; Naing, A.L.; Nyo, M.Y.; Myint, N.Z.; Imwong, M.; et al. Effectiveness of five artemisinin combination regimens with or without primaquine in uncomplicated falciparum malaria: An open-label randomised trial. Lancet Infect. Dis. 2010, 10, 673–681. [Google Scholar] [CrossRef]
- Wong, Y.K.; Xu, C.; Kalesh, K.A.; He, Y.; Lin, Q.; Wong, W.S.F.; Shen, H.M.; Wang, J. Artemisinin as an anticancer drug: Recent advances in target profiling and mechanisms of action. Med. Res. Rev. 2017, 37, 1492–1517. [Google Scholar] [CrossRef]
- Zhu, S.; Yu, Q.; Huo, C.; Li, Y.; He, L.; Ran, B.; Chen, J.; Li, Y.; Liu, W. Ferroptosis: A Novel Mechanism of Artemisinin and its Derivatives in Cancer Therapy. Curr. Med. Chem. 2021, 28, 329–345. [Google Scholar] [CrossRef] [PubMed]
- Efferth, T. From ancient herb to modern drug: Artemisia annua and artemisinin for cancer therapy. Semin. Cancer Biol. 2017, 46, 65–83. [Google Scholar] [CrossRef]
- Yang, N.D.; Tan, S.H.; Ng, S.; Shi, Y.; Zhou, J.; Tan, K.S.; Wong, W.S.; Shen, H.M. Artesunate induces cell death in human cancer cells via enhancing lysosomal function and lysosomal degradation of ferritin. J. Biol. Chem. 2014, 289, 33425–33441. [Google Scholar] [CrossRef]
- Abarientos, C.; Sperber, K.; Shapiro, D.L.; Aronow, W.S.; Chao, C.P.; Ash, J.Y. Hydroxychloroquine in systemic lupus erythematosus and rheumatoid arthritis and its safety in pregnancy. Expert Opin. Drug Saf. 2011, 10, 705–714. [Google Scholar] [CrossRef]
- Al-Rawi, H.; Meggitt, S.J.; Williams, F.M.; Wahie, S. Steady-state pharmacokinetics of hydroxychloroquine in patients with cutaneous lupus erythematosus. Lupus 2018, 27, 847–852. [Google Scholar] [CrossRef]
- Janku, F.; McConkey, D.J.; Hong, D.S.; Kurzrock, R. Autophagy as a target for anticancer therapy. Nat. Rev. Clin. Oncol. 2011, 8, 528–539. [Google Scholar] [CrossRef]
- Levy, J.M.M.; Towers, C.G.; Thorburn, A. Targeting autophagy in cancer. Nat. Rev. Cancer 2017, 17, 528–542. [Google Scholar] [CrossRef] [PubMed]
- Gurova, K.V.; Hill, J.E.; Guo, C.; Prokvolit, A.; Burdelya, L.G.; Samoylova, E.; Khodyakova, A.V.; Ganapathi, R.; Ganapathi, M.; Tararova, N.D.; et al. Small molecules that reactivate p53 in renal cell carcinoma reveal a NF-kappaB-dependent mechanism of p53 suppression in tumors. Proc. Natl. Acad. Sci. USA 2005, 102, 17448–17453. [Google Scholar] [CrossRef] [PubMed]
- Gasparian, A.V.; Burkhart, C.A.; Purmal, A.A.; Brodsky, L.; Pal, M.; Saranadasa, M.; Bosykh, D.A.; Commane, M.; Guryanova, O.A.; Pal, S.; et al. Curaxins: Anticancer compounds that simultaneously suppress NF-kappaB and activate p53 by targeting FACT. Sci. Transl. Med. 2011, 3, 95ra74. [Google Scholar] [CrossRef] [PubMed]
- Nesher, E.; Safina, A.; Aljahdali, I.; Portwood, S.; Wang, E.S.; Koman, I.; Wang, J.; Gurova, K.V. Role of Chromatin Damage and Chromatin Trapping of FACT in Mediating the Anticancer Cytotoxicity of DNA-Binding Small-Molecule Drugs. Cancer Res. 2018, 78, 1431–1443. [Google Scholar] [CrossRef]
- Lewiecki, E.M. Intravenous zoledronic acid for the treatment of osteoporosis: The evidence of its therapeutic effect. Core Evid. 2010, 4, 13–23. [Google Scholar] [CrossRef][Green Version]
- Saad, F.; Gleason, D.M.; Murray, R.; Tchekmedyian, S.; Venner, P.; Lacombe, L.; Chin, J.L.; Vinholes, J.J.; Goas, J.A.; Chen, B.; et al. A randomized, placebo-controlled trial of zoledronic acid in patients with hormone-refractory metastatic prostate carcinoma. J. Natl. Cancer Inst. 2002, 94, 1458–1468. [Google Scholar] [CrossRef]
- Mason, M.D.; Clarke, N.W.; James, N.D.; Dearnaley, D.P.; Spears, M.R.; Ritchie, A.W.S.; Attard, G.; Cross, W.; Jones, R.J.; Parker, C.C.; et al. Adding Celecoxib With or Without Zoledronic Acid for Hormone-Naive Prostate Cancer: Long-Term Survival Results From an Adaptive, Multiarm, Multistage, Platform, Randomized Controlled Trial. J. Clin. Oncol. 2017, 35, 1530–1541. [Google Scholar] [CrossRef]
- Niraula, S.; Templeton, A.J.; Vera-Badillo, F.; Dodd, A.; Nugent, Z.; Joshua, A.M.; Tannock, I.F. Duration of suppression of bone turnover following treatment with zoledronic acid in men with metastatic castration-resistant prostate cancer. Future Sci. OA 2018, 4, FSO253. [Google Scholar] [CrossRef]
- Ghodke-Puranik, Y.; Thorn, C.F.; Lamba, J.K.; Leeder, J.S.; Song, W.; Birnbaum, A.K.; Altman, R.B.; Klein, T.E. Valproic acid pathway: Pharmacokinetics and pharmacodynamics. Pharm. Genom. 2013, 23, 236–241. [Google Scholar] [CrossRef]
- Yu, S.; Yang, X.; Zhu, Y.; Xie, F.; Lu, Y.; Yu, T.; Yan, C.; Shao, J.; Gao, Y.; Mo, F.; et al. Systems pharmacology of mifepristone (RU486) reveals its 47 hub targets and network: Comprehensive analysis and pharmacological focus on FAK-Src-Paxillin complex. Sci. Rep. 2015, 5, 7830. [Google Scholar] [CrossRef]
- Mobbs, B.G.; Johnson, I.E. Suppression of the growth of the androgen-insensitive R3327 HI rat prostatic carcinoma by combined estrogen and antiprogestin treatment. J. Steroid Biochem. Mol. Biol. 1991, 39, 713–722. [Google Scholar] [CrossRef]
- Tieszen, C.R.; Goyeneche, A.A.; Brandhagen, B.N.; Ortbahn, C.T.; Telleria, C.M. Antiprogestin mifepristone inhibits the growth of cancer cells of reproductive and non-reproductive origin regardless of progesterone receptor expression. BMC Cancer 2011, 11, 207. [Google Scholar] [CrossRef] [PubMed]
- Taplin, M.E.; Manola, J.; Oh, W.K.; Kantoff, P.W.; Bubley, G.J.; Smith, M.; Barb, D.; Mantzoros, C.; Gelmann, E.P.; Balk, S.P. A phase II study of mifepristone (RU-486) in castration-resistant prostate cancer, with a correlative assessment of androgen-related hormones. BJU Int. 2008, 101, 1084–1089. [Google Scholar] [CrossRef] [PubMed]
- Suh, J.J.; Pettinati, H.M.; Kampman, K.M.; O’Brien, C.P. The status of disulfiram: A half of a century later. J. Clin. Psychopharmacol. 2006, 26, 290–302. [Google Scholar] [CrossRef] [PubMed]
- Hald, J.; Jacobsen, E. A drug sensitizing the organism to ethyl alcohol. Lancet 1948, 2, 1001–1004. [Google Scholar] [CrossRef]
- Tacconi, E.M.; Lai, X.; Folio, C.; Porru, M.; Zonderland, G.; Badie, S.; Michl, J.; Sechi, I.; Rogier, M.; Matia Garcia, V.; et al. BRCA1 and BRCA2 tumor suppressors protect against endogenous acetaldehyde toxicity. EMBO Mol. Med. 2017, 9, 1398–1414. [Google Scholar] [CrossRef] [PubMed]
- Dorokhov, Y.L.; Sheshukova, E.V.; Bialik, T.E.; Komarova, T.V. Human Endogenous Formaldehyde as an Anticancer Metabolite: Its Oxidation Downregulation May Be a Means of Improving Therapy. Bioessays 2018, 40, e1800136. [Google Scholar] [CrossRef] [PubMed]
- Pontel, L.B.; Rosado, I.V.; Burgos-Barragan, G.; Garaycoechea, J.I.; Yu, R.; Arends, M.J.; Chandrasekaran, G.; Broecker, V.; Wei, W.; Liu, L.; et al. Endogenous Formaldehyde Is a Hematopoietic Stem Cell Genotoxin and Metabolic Carcinogen. Mol. Cell 2015, 60, 177–188. [Google Scholar] [CrossRef]
- Liu, P.; Brown, S.; Goktug, T.; Channathodiyil, P.; Kannappan, V.; Hugnot, J.P.; Guichet, P.O.; Bian, X.; Armesilla, A.L.; Darling, J.L.; et al. Cytotoxic effect of disulfiram/copper on human glioblastoma cell lines and ALDH-positive cancer-stem-like cells. Br. J. Cancer 2012, 107, 1488–1497. [Google Scholar] [CrossRef]
- Guo, F.; Yang, Z.; Kulbe, H.; Albers, A.E.; Sehouli, J.; Kaufmann, A.M. Inhibitory effect on ovarian cancer ALDH+ stem-like cells by Disulfiram and Copper treatment through ALDH and ROS modulation. Biomed. Pharmacother. 2019, 118, 109371. [Google Scholar] [CrossRef]
- Lin, J.; Haffner, M.C.; Zhang, Y.; Lee, B.H.; Brennen, W.N.; Britton, J.; Kachhap, S.K.; Shim, J.S.; Liu, J.O.; Nelson, W.G.; et al. Disulfiram is a DNA demethylating agent and inhibits prostate cancer cell growth. Prostate 2011, 71, 333–343. [Google Scholar] [CrossRef] [PubMed]
- Schweizer, M.T.; Lin, J.; Blackford, A.; Bardia, A.; King, S.; Armstrong, A.J.; Rudek, M.A.; Yegnasubramanian, S.; Carducci, M.A. Pharmacodynamic study of disulfiram in men with non-metastatic recurrent prostate cancer. Prostate Cancer Prostatic Dis. 2013, 16, 357–361. [Google Scholar] [CrossRef] [PubMed]
- Safi, R.; Nelson, E.R.; Chitneni, S.K.; Franz, K.J.; George, D.J.; Zalutsky, M.R.; McDonnell, D.P. Copper signaling axis as a target for prostate cancer therapeutics. Cancer Res. 2014, 74, 5819–5831. [Google Scholar] [CrossRef] [PubMed]
- Skrott, Z.; Mistrik, M.; Andersen, K.K.; Friis, S.; Majera, D.; Gursky, J.; Ozdian, T.; Bartkova, J.; Turi, Z.; Moudry, P.; et al. Alcohol-abuse drug disulfiram targets cancer via p97 segregase adaptor NPL4. Nature 2017, 552, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Sharif, A. Sirolimus after kidney transplantation. BMJ 2014, 349, g6808. [Google Scholar] [CrossRef]
- Farb, A.; John, M.; Acampado, E.; Kolodgie, F.D.; Prescott, M.F.; Virmani, R. Oral everolimus inhibits in-stent neointimal growth. Circulation 2002, 106, 2379–2384. [Google Scholar] [CrossRef][Green Version]
- Fattori, R.; Piva, T. Drug-eluting stents in vascular intervention. Lancet 2003, 361, 247–249. [Google Scholar] [CrossRef]
- Benjamin, D.; Colombi, M.; Moroni, C.; Hall, M.N. Rapamycin passes the torch: A new generation of mTOR inhibitors. Nat. Rev. Drug Discov. 2011, 10, 868–880. [Google Scholar] [CrossRef]
- Brown, V.I.; Fang, J.; Alcorn, K.; Barr, R.; Kim, J.M.; Wasserman, R.; Grupp, S.A. Rapamycin is active against B-precursor leukemia in vitro and in vivo, an effect that is modulated by IL-7-mediated signaling. Proc. Natl. Acad. Sci. USA 2003, 100, 15113–15118. [Google Scholar] [CrossRef]
- Altman, J.K.; Sassano, A.; Kaur, S.; Glaser, H.; Kroczynska, B.; Redig, A.J.; Russo, S.; Barr, S.; Platanias, L.C. Dual mTORC2/mTORC1 targeting results in potent suppressive effects on acute myeloid leukemia (AML) progenitors. Clin. Cancer Res. 2011, 17, 4378–4388. [Google Scholar] [CrossRef]
- Mancini, M.; Petta, S.; Martinelli, G.; Barbieri, E.; Santucci, M.A. RAD 001 (everolimus) prevents mTOR and Akt late re-activation in response to imatinib in chronic myeloid leukemia. J. Cell. Biochem. 2010, 109, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Sillaber, C.; Mayerhofer, M.; Bohm, A.; Vales, A.; Gruze, A.; Aichberger, K.J.; Esterbauer, H.; Pfeilstocker, M.; Sperr, W.R.; Pickl, W.F.; et al. Evaluation of antileukaemic effects of rapamycin in patients with imatinib-resistant chronic myeloid leukaemia. Eur. J. Clin. Investig. 2008, 38, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Bahmad, H.F.; Mouhieddine, T.H.; Chalhoub, R.M.; Assi, S.; Araji, T.; Chamaa, F.; Itani, M.M.; Nokkari, A.; Kobeissy, F.; Daoud, G.; et al. The Akt/mTOR pathway in cancer stem/progenitor cells is a potential therapeutic target for glioblastoma and neuroblastoma. Oncotarget 2018, 9, 33549–33561. [Google Scholar] [CrossRef] [PubMed]
- Richardson, P.; Anderson, K. Thalidomide and dexamethasone: A new standard of care for initial therapy in multiple myeloma. J. Clin. Oncol. 2006, 24, 334–336. [Google Scholar] [CrossRef][Green Version]
- Eleutherakis-Papaiakovou, V.; Bamias, A.; Dimopoulos, M.A. Thalidomide in cancer medicine. Ann. Oncol. 2004, 15, 1151–1160. [Google Scholar] [CrossRef]
- Bahmad, H.F.; Samman, H.; Monzer, A.; Hadadeh, O.; Cheaito, K.; Abdel-Samad, R.; Hayar, B.; Pisano, C.; Msheik, H.; Liu, Y.N.; et al. The synthetic retinoid ST1926 attenuates prostate cancer growth and potentially targets prostate cancer stem-like cells. Mol. Carcinog. 2019, 58, 1208–1220. [Google Scholar] [CrossRef]
| Drug Category | Clinical Trial Title | Number ClinicalTrials.gov ID | Phase | Study Start Date | Estimated Enrollment | Status | Intervention |
|---|---|---|---|---|---|---|---|
| Anti-diabetic | Metformin Hydrochloride as First-Line Therapy in Treating Patients with Locally Advanced or Metastatic PCa | NCT01243385 | II | 1 December 2010 | 44 | Completed | Metformin |
| Anti-diabetic | Castration Compared to Castration Plus Metformin as First Line Treatment for Patients with Advanced PCa | NCT01620593 | II | 1 April 2011 | 41 | Completed | Metformin |
| Anti-diabetic | Prevention of Metabolic Syndrome and Increased Weight Using Metformin Concurrent to ADT and Radiotherapy for Locally Advanced PCa | NCT01996696 | II | 1 September 2014 | 104 | Recruiting | Metformin |
| Anti-diabetic | Repurposing Metformin as Anticancer Drug in Advanced PCa (Mansmed) | NCT03137186 | II | 1 January 2017 | 120 | Unknown | Metformin |
| Anti-diabetic | Study of Metformin Plus Oligomeric Procyanidin Complex for Pharmacologic Manipulation of AGE Levels in PCa Patients | NCT03465345 | Ib | 1 July 2018 | 0 | Withdrawn | Metformin + oligomeric procyanidin complex |
| Anti-diabetic | Drug-Drug Interaction of SHR3680 with Digoxin, Rosuvastatin Calcium and Metformin Hydrochloride | NCT04621669 | I | 1 November 2020 | 18 | Not yet recruiting | Metformin hydrochloride + SHR3680 + digoxin + rosuvastatin |
| Anti-hyperlipidemic | Impact of Adjuvant Statin Therapy in Patients who Underwent Radical Prostatectomy for Locally Advanced PCa | NCT01759836 | II | 1 October 2012 | 354 | Unknown | Atorvastatin vs. placebo |
| Anti-parasitic | Low, Intermediate, or High Dose Suramin in Treating Patients with Hormone-Refractory PCa | NCT00002723 | III | 1 January 1996 | 390 | Completed | Suramin |
| Anti-parasitic | Combination Chemotherapy with Suramin Plus Doxorubicin in Treating Patients with Advanced Solid Tumors | NCT00003038 | I | 1 October 1997 | 20 | Completed | Doxorubicin + suramin |
| Anti-parasitic | Akt Inhibitor MK2206 and Hydroxychloroquine in Treating Patients with Advanced Solid Tumors, Melanoma, Prostate or Kidney Cancer | NCT01480154 | I | 1 November 2011 | 62 | Active, not recruiting | Hydroxycloroquine + Akt inhibitor MK2206 |
| Anti-helminthic | Enzalutamide and Niclosamide in Treating Patients with Recurrent or Metastatic CRPC | NCT03123978 | I | 1 January 2017 | 12 | Recruiting | Enzalutamide + niclosamide |
| Anti-helminthic | Niclosamide and Enzalutamide in Treating Patients With CRPC | NCT02532114 | I | 1 December 2015 | 5 | Completed | Niclosamide + enzalutamide |
| Anti-fungal | Hormonal Therapy and Chemotherapy Followed by Prostatectomy in Patients with PCa | NCT02494713 | II | 1 October 2015 | 4 | Terminated | Degarelix + Doxorubicin + Ketoconazole + Docetaxel + Estramusine |
| Anti-fungal | Hormone Therapy Plus Chemotherapy as Initial Treatment for Local Failures or Advanced PCa | NCT02560051 | II | 1 November 2015 | 19 | Terminated | Doxorubicin + ketoconazole + docetaxel + estramustine |
| Antiviral | A Study of Aplidin (Plitidepsin) in Subjects with Advanced PCa | NCT00780975 | II | 1 February 2005 | 8 | Terminated | Plitidepsin |
| NSAID | Dexamethasone, Aspirin, and Diethylstilbestrol in Treating Patients with Locally Advanced or Metastatic PCa | NCT00316927 | III | 1 December 2002 | 260 | Completed | Dexamethasone + aspirin vs. dexamethasone + diethylstilbestrol + aspirin |
| Steroid | Combination of Docetaxel + Estramustine + Hydrocortisone Versus Docetaxel + Prednisone in Patients with Advanced PCa (PROSTATA) | NCT00705822 | III | 1 August 2006 | 54 | Terminated | Docetaxel + prednisone vs. docetaxel + estramusine + hydrocortisone |
| Steroid | Docetaxel, Prednisone, and Vatalanib in Treating Patients with Advanced PCa | NCT00293371 | I/II | 1 September 2006 | 6 | Terminated | Docetaxel + prednisone + vatalanib |
| Steroid | A Safety and Efficacy Study of Abiraterone Acetate in Participants with Advanced PCa who Failed ADT and Docetaxel-Based Chemotherapy | NCT00474383 | II | 1 November 2006 | 47 | Completed | Abiraterone acetate + glucocorticoid |
| Steroid | An Open-Label Study of YM155 + Docetaxel in Subjects with Advanced Hormone Refractory PCa and Other Solid Tumors | NCT00514267 | I/II | 1 May 2007 | 32 | Completed | YM 155 + docetaxel + prednisone vs. YM 155 + docetaxel |
| Steroid | An Efficacy and Safety Study of Abiraterone Acetate and Prednisone in Participants with PCa Who Failed ADT and Docetaxel-Based Chemotherapy | NCT00485303 | II | 1 June 2007 | 58 | Completed | Abiraterone acetate + prednisone |
| Steroid | AMG 386 and Abiraterone for Advanced PCa | NCT01553188 | II | 1 February 2012 | 36 | Completed | Abiraterone + prednisone vs. abiraterone + prednisone + AMG |
| Steroid | Cabozantinib Plus Docetaxel and Prednisone for Advanced PCa | NCT01683994 | I | 1 September 2012 | 49 | Completed | Carbozantinib + docetaxel + prednisone |
| Steroid | Reducing Dexamethasone Around Docetaxel Infusion (REDEX) | NCT02776436 | I | 1 January 2016 | 46 | Active, not recruiting | Dexamethasone |
| Steroid | Cognitive Effects of AR Directed Therapies for Advanced PCa | NCT03016741 | IV | 1 March 2017 | 100 | Recruiting | GnRH agonist/antagonist + prednisone + abiraterone acetate vs. GnRH agonist/antagonist + enzalutamide |
| Steroid | Intermittent ADT for Stage IV Castration Sensitive PCa | NCT03511196 | 1b | 1 September 2018 | 17 | Active, not recruiting | ADT + abiraterone + prednisone |
| Steroid | A Study of Nivolumab or Placebo in Combination with Docetaxel in Men with Advanced CRPC (CheckMate 7DX) | NCT04100018 | III | 1 February 2020 | 984 | Recruiting | Nivolumab + prednisone + docetaxel vs. placebo |
| Immunosuppressive drug | Sirolimus Before Surgery in Treating Patients with Advanced Localized PCa | NCT00311623 | I/II | 1 August 2006 | 32 | Completed | No intervention vs. low dose rapamycin vs high dose rapamycin |
| Synthetic vitamin D analogue | Paricalcitol in Treating Patients with Advanced PCa and Bone Metastases | NCT00634582 | II | 1 January 2009 | 2 | Terminated | Paricalcitol |
| Vitamin D analogue | Effect of CTAP101 Capsules on Ca/iPTH in Advanced Breast/PCa Patients Treated with Denosumab/Zoledronic Acid | NCT02274623 | I | 1 December 2014 | 33 | Completed | CTAP101 capsules (vitamin D analogue) |
| Vitamin D | Changes in Bone Mineral Density and Fracture Risk in Patients Receiving ADT for PCa | NCT00536653 | N/A * | 1 October 1999 | 618 | Completed | Bicalutamide + calcium/vitamin D supplementation vs. LHRH agonists + calcium/vitamin D supplementation |
| Bisphosphonate | Study of Zoledronic Acid for Patients with Hormone-sensitive Bone Metastases from PCa | NCT00242567 | III | 1 December 2005 | 522 | Completed | Zoledronic acid |
| Bisphosphonate | Zoledronate in Preventing Osteoporosis and Bone Fractures in Patients with Locally Advanced Nonmetastatic PCa Undergoing Radiation Therapy and Hormone Therapy | NCT00329797 | III | 1 March 2006 | 109 | Completed | Calcium + zoledronic acid + radiation therapy + LHRH + vitamin D vs. calcium + radiation therapy + LHRH + vitamin D |
| Bisphosphonate | Study of Denosumab vs. Zoledronic Acid to Treat Bone Metastases in Subjects with Advanced Cancer or Multiple Myeloma. | NCT00330759 | III | 1 June 2006 | 1779 | Completed | Denosumab + zoledronic acid |
| Somatostatin analogue | Effects of Octreotide Acetate on Circulating Levels of Chromogranin A in Advanced PCa Patients | NCT00166725 | II | 1 February 2004 | 40 | Completed | Octreotide acetate |
| Anticoagulant | Standard Therapy with or Without Dalteparin in Treating Patients with Advanced Breast, Lung, Colorectal, or PCa | NCT00003674 | III | 1 December 1998 | 141 | Completed | Dalteparin |
| Anticoagulant | Effects of Nadroparin in Patients with Lung, Pancreas or PCa | NCT00312013 | III | 1 May 2006 | 503 | Completed | Nadroparin |
| Synthetic retinoid | Fenretinide In Treating Patients with Advanced or Metastatic Hormone-Refractory Prostate Cancer | NCT00077402 | II | 1 February 2004 | 50 | Completed | Fenretinide |
| Other | Preoperative Thalidomide Followed by Radical Retropubic Prostatectomy in Select Patients with Locally Advanced PCa | NCT00038181 | II | 5 October 2000 | 18 | Completed | Thalidomide preoperatively |
| Other | Trial of Docetaxel-Samarium in Patients with Hormone-Refractory Advanced PCa | NCT00126230 | II | 1 January 2004 | 55 | Terminated | Docetaxel-samarium |
| Other | Samarium-153 With Neoadjuvant Hormonal and Radiation Therapy for Locally Advanced PCa | NCT00328614 | I | 1 March 2003 | 32 | Completed | Samarium + hormonal and radiation therapy |
| Other | Study of Chitosan for Pharmacologic Manipulation of AGE Levels in PCa Patients | NCT03712371 | Ib/2 | 1 January 2019 | 45 | Recruiting | Chitosan |
| Other | Study of Pharmacologic Manipulation of AGE Levels in PCa Patients Receiving ADT | NCT02946996 | II | 1 December 2016 | 45 | Recruiting | AGE |
| Other | A Phase I/II Trial of 2-Deoxyglucose (2DG) for the Treatment of Advanced and Hormone Refractory PCa | NCT00633087 | I/II | 1 March 2011 | 12 | Terminated | 2-deoxyglucose |
| Other | Omega-3 Fatty Acids in Treating Patients with Advanced PCa | NCT00996749 | 1 May 2011 | 0 | Withdrawn | Omega-3 PUFA supplementation | |
| Other | Hyperpolarized C-13 Pyruvate as a Biomarker in Patients with Advanced Solid Tumor Malignancies | NCT02913131 | I/II | 1 October 2016 | 20 | Terminated | Pyruvate |
| Other | Nanoparticle Albumin-Bound Rapamycin in Treating Patients with Advanced Cancer with mTOR Mutations | NCT02646319 | I | 1 January 2016 | 2 | completed | Rapamycin |
| Other | Enzalutamide/Leuprolide ± Abiraterone/Pred in PCa | NCT02268175 | II | 1 October 2014 | 75 | Active, not recruiting | Enzalutamide + leuprolife + abiraterone acetate + prednisone vs. enzalutamide + leuprolide |
| Other | Study to Compare the Effects of Repeated Doses of an Investigational New Drug and a Placebo on Appetite in Advanced PCa and Anorexia | NCT04803305 | I | 1 May 2021 | 40 | Recruiting | PF-06946860 |
| Other | Study of XL102 as Single-Agent and Combination Therapy in Subjects with Solid Tumors | NCT04726332 | I | 1 February 2021 | 298 | Recruiting | XL102 + fulvestrant + abiraterone + prednisone |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bahmad, H.F.; Demus, T.; Moubarak, M.M.; Daher, D.; Alvarez Moreno, J.C.; Polit, F.; Lopez, O.; Merhe, A.; Abou-Kheir, W.; Nieder, A.M.; et al. Overcoming Drug Resistance in Advanced Prostate Cancer by Drug Repurposing. Med. Sci. 2022, 10, 15. https://doi.org/10.3390/medsci10010015
Bahmad HF, Demus T, Moubarak MM, Daher D, Alvarez Moreno JC, Polit F, Lopez O, Merhe A, Abou-Kheir W, Nieder AM, et al. Overcoming Drug Resistance in Advanced Prostate Cancer by Drug Repurposing. Medical Sciences. 2022; 10(1):15. https://doi.org/10.3390/medsci10010015
Chicago/Turabian StyleBahmad, Hisham F., Timothy Demus, Maya M. Moubarak, Darine Daher, Juan Carlos Alvarez Moreno, Francesca Polit, Olga Lopez, Ali Merhe, Wassim Abou-Kheir, Alan M. Nieder, and et al. 2022. "Overcoming Drug Resistance in Advanced Prostate Cancer by Drug Repurposing" Medical Sciences 10, no. 1: 15. https://doi.org/10.3390/medsci10010015
APA StyleBahmad, H. F., Demus, T., Moubarak, M. M., Daher, D., Alvarez Moreno, J. C., Polit, F., Lopez, O., Merhe, A., Abou-Kheir, W., Nieder, A. M., Poppiti, R., & Omarzai, Y. (2022). Overcoming Drug Resistance in Advanced Prostate Cancer by Drug Repurposing. Medical Sciences, 10(1), 15. https://doi.org/10.3390/medsci10010015








