Preliminary Investigation into Plasmodium-like Piroplasms (Babesia/Theileria) among Cattle, Dogs and Humans in A Malaria-Endemic, Resource-Limited Sub-Saharan African City
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Sites and Blood Samples Collection
2.2. Laboratory Procedures
2.3. Statistical Analysis
3. Results
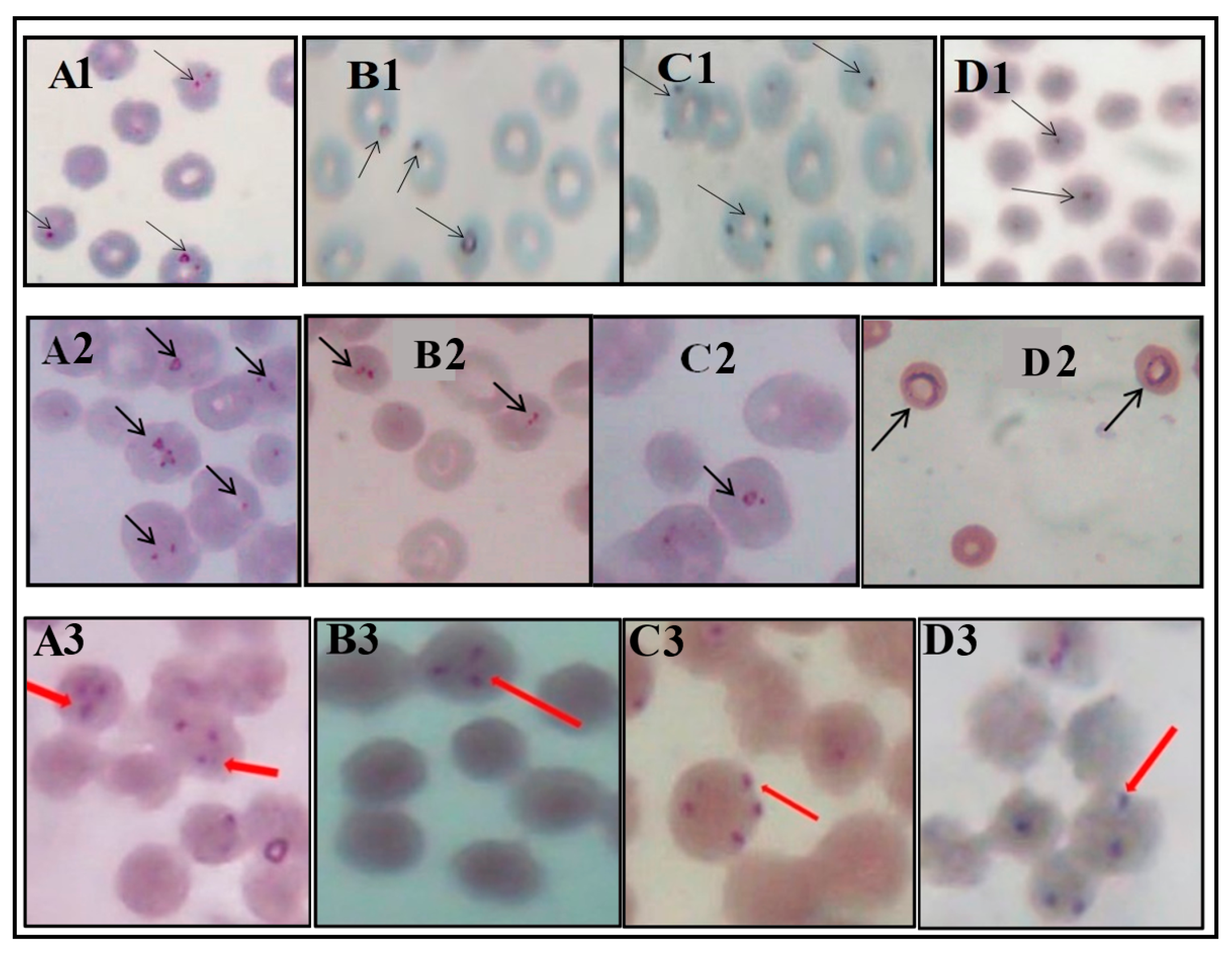
3.1. Microscopic Suspicion of Babesia/Theileria in Cattle, Dogs and Humans
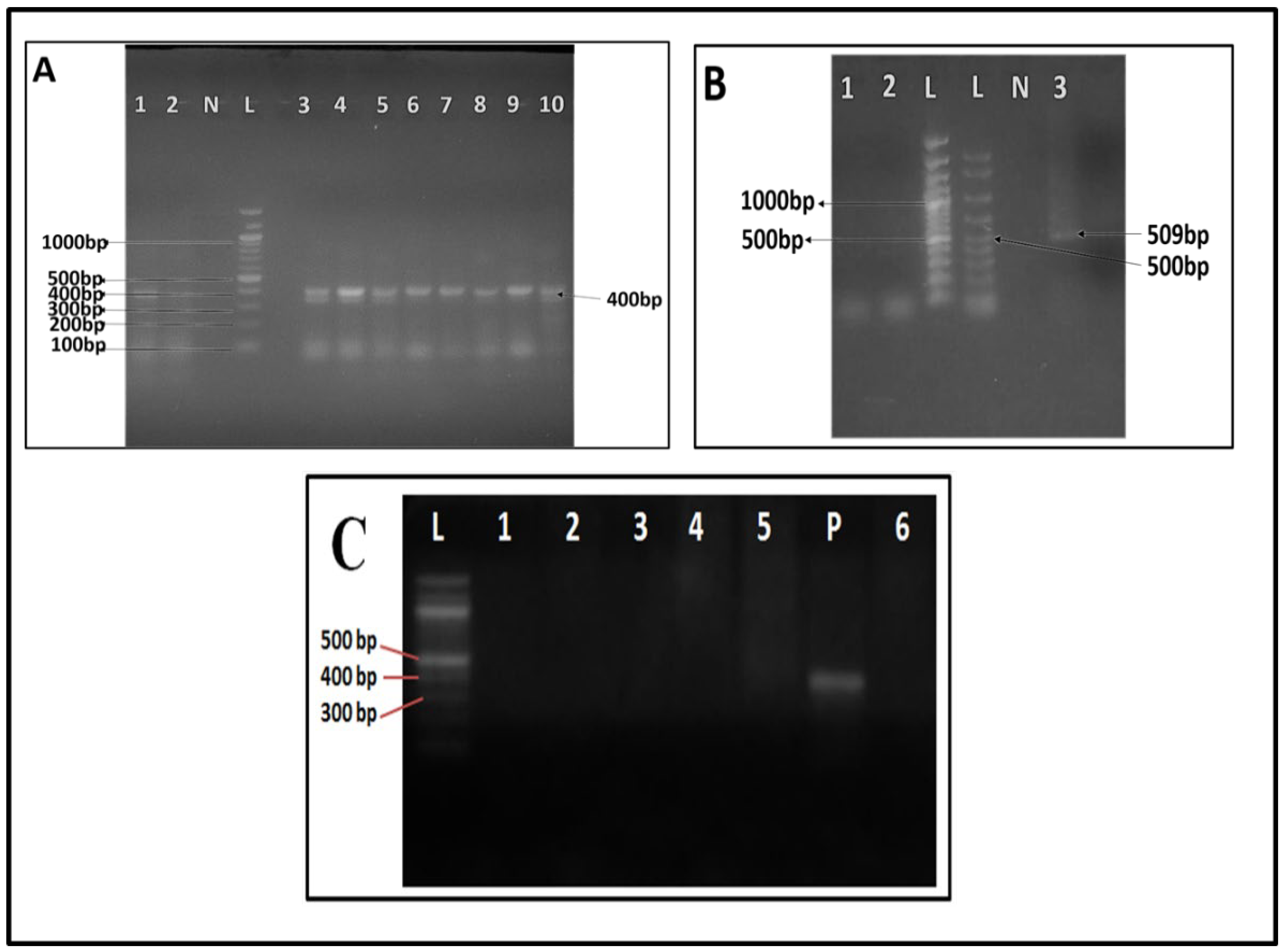
3.2. Molecular Detection of Babesia/Theileria in Cattle, Dogs and Humans
4. Discussion
5. Conclusions
6. Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
Abbreviations
References
- Vannier, E.; Krause, P.J. Human babesiosis. N. Engl. J. Med. 2012, 366, 2397–2407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Skrabalo, Z.; Deanovic, Z. Piroplasmosis in man; report of a case. Doc. de Med. Geogr. Trop. 1957, 9, 11–16. [Google Scholar]
- Kim, J.; Cho, S.; Joo, H.; Tsuji, M.; Cho, S.-R.; Park, J., II; Chung, G.-T.; Ju, J.-W.; Cheun, H., II; Lee, H.-W.; et al. First Case of Human Babesiosis in Korea”: Detection and Characterization of a Novel Type of Babesia sp. (KO1) Similar to Ovine Babesia. J. Clin. Microbiol. 2007, 45, 2084–2087. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leiby, D.A. Transfusion-associated babesiosis: Shouldn’t we be ticked off? Ann. Intern. Med. 2011, 155, 556–557. [Google Scholar] [CrossRef] [Green Version]
- Krause, P.J.; Lepore, T.; Sikand, V.K.; Gadbaw, J.; Burke, G.; Telford, S.R.; Brassard, P.; Pearl, D.; Azlanzadeh, J.; Christianson, D.; et al. Atovaquone and Azithromycin for the Treatment of Babesiosis. N. Engl. J. Med. 2000, 343, 1454–1458. [Google Scholar] [CrossRef] [Green Version]
- Hunfeld, K.-P.; Hildebrandt, A.; Gray, J.S. Babesiosis: Recent insights into an ancient disease. Int. J. Parasitol. 2008, 38, 1219–1237. [Google Scholar] [CrossRef]
- Gray, J.; Zintl, A.; Hildebrandt, A.; Hunfeld, K.-P.; Weiss, L. Zoonotic babesiosis: Overview of the disease and novel aspects of pathogen identity. Ticks Tick-Borne Dis. 2010, 1, 3–10. [Google Scholar] [CrossRef]
- Vannier, E.; Krause, P.J. Update on Babesiosis. Interdiscip. Perspect. Infect. Dis. 2009, 2009, 984568. [Google Scholar] [CrossRef] [Green Version]
- Bell-Sakyi, L.; Koney, E.; Dogbey, O.; Walker, A. Incidence and prevalence of tick-borne haemoparasites in domestic ruminants in Ghana. Vet. Parasitol. 2004, 124, 25–42. [Google Scholar] [CrossRef]
- El-Ashker, M.; Hotzel, H.; Gwida, M.; El-Beskawy, M.; Silaghi, C.; Tomaso, H. Molecular biological identification of Babesia, Theileria, and Anaplasma species in cattle in Egypt using PCR assays, gene sequence analysis and a novel DNA microarray. Vet. Parasitol. 2015, 207, 329–334. [Google Scholar] [CrossRef] [Green Version]
- Zahler, M.; Rinder, H.; Schein, E.; Gothe, R. Detection of a new pathogenic Babesia microti-like species in dogs. Vet. Parasitol. 2000, 89, 241–248. [Google Scholar] [CrossRef]
- Nagano, D.; Sivakumar, T.; De De Macedo, A.C.C.; Inpankaew, T.; Alhassan, A.; Igarashi, I.; Yokoyama, N. The Genetic Diversity of Merozoite Surface Antigen 1 (MSA-1) among Babesia bovis Detected from Cattle Populations in Thailand, Brazil and Ghana. J. Vet. Med. Sci. 2013, 75, 1463–1470. [Google Scholar] [CrossRef] [Green Version]
- Quick, R.E.; Herwaldt, B.L.; Thomford, J.W.; Garnett, M.E.; Eberhard, M.L.; Wilson, M.; Spach, D.H.; Dickerson, J.W.; Telford, S.R.; Steingart, K.R.; et al. Babesisosis in Washington State: A New Species of Babesia? Ann. Intern. Med. 1993, 119, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Persing, D.H.; Herwaldt, B.L.; Glaser, C.; Lane, R.S.; Thomford, J.W.; Mathiesen, D.; Krause, P.J.; Phillip, D.F.; Conrad, P.A. Infection with a Babesia-Like Organism in Northern California. N. Engl. J. Med. 1995, 332, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Kunimoto, D.; Krause, K.; Morrison, D. First case of (imported) babesiosis diagnosed in Canada. Can. J. Infect. Dis. Med. Microbiol. 1998, 9, 387–389. [Google Scholar] [CrossRef] [Green Version]
- Hildebrandt, A.; Hunfeld, K.-P.; Baier, M.; Krumbholz, A.; Sachse, S.; Lorenzen, T.; Kiehntopf, M.; Fricke, H.-J.; Straube, E. First confirmed autochthonous case of human Babesia microti infection in Europe. Eur. J. Clin. Microbiol. 2007, 26, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Centeno-Lima, S.; do Rosa’rio, V.; Parreira, R.; Maia, A.J.; Freudenthal, A.M.; Nijhof, A.M.; Jongejan, F. Fatal case of human babesiosis in Portugal: Molecular and phylogenetic analysis. Trop. Med. Int. Health 2003, 8, 760–764. [Google Scholar] [CrossRef]
- Williams, J.E.; Cairns, M.; Njie, F.; Quaye, S.L.; Awine, T.; Oduro, A.; Tagbor, H.; Bojang, K.; Magnussen, P.; ter Kuile, F.; et al. The Performance of a Rapid Diagnostic Test in Detecting Malaria Infection in Pregnant Women and the Impact of Missed Infections. Clin. Infect. Dis. 2016, 62, 837–844. [Google Scholar] [CrossRef]
- Cheng, Q.; Gatton, M.L.; Barnwell, J.; Chiodini, P.; McCarthy, J.; Bell, D.; Cunningham, J. Plasmodium falciparum parasites lacking histidine-rich protein 2 and 3: A review and recommendations for accurate reporting. Malar. J. 2014, 13, 283. [Google Scholar] [CrossRef] [Green Version]
- Marathe, A.; Tripathi, J.; Handa, V.; Date, V. Human Babesiosis—A Case Report. Indian J. Med. Microbiol. 2005, 23, 267–269. [Google Scholar] [CrossRef]
- Ramharter, M.; Walochnik, J.; Lagler, H.; Winkler, S.; Wernsdorfer, W.H.; Stoiser, B.; Graninger, W. Clinical and Molecular Characterization of a Near Fatal Case of Human Babesiosis in Austria. J. Travel Med. 2010, 17, 416–418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martínez-García, G.; Santamaría-Espinosa, R.; Lira-Amaya, J.; Figueroa, J. Challenges in Tick-Borne Pathogen Detection: The Case for Babesia spp. Identification in the Tick Vector. Pathogens 2021, 10, 92. [Google Scholar] [CrossRef]
- Hilpertshauser, H.; Deplazes, P.; Schnyder, M.; Gern, L.; Mathis, A. Babesia spp. Identified by PCR in Ticks Collected from Domestic and Wild Ruminants in Southern Switzerland. Appl. Environ. Microbiol. 2006, 72, 6503–6507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zygner, W.; Jaros, S.; Wedrychowicz, H. Prevalence of Babesia canis, Borrelia afzelii, and Anaplasma phagocytophilum infection in hard ticks removed from dogs in Warsaw (central Poland). Vet. Parasitol. 2008, 153, 139–142. [Google Scholar] [CrossRef]
- Hussein, H.E.; Johnson, W.C.; Taus, N.S.; Suarez, C.E.; Scoles, G.A.; Ueti, M.W. Silencing expression of the Rhipicephalus microplus vitellogenin receptor gene blocks Babesia bovis transmission and interferes with oocyte maturation. Parasites Vectors 2019, 12, 7. [Google Scholar] [CrossRef] [PubMed]
- Brasseur, P.; Gorrenflot, A. Human Babesiosis in Europe. Mem. Do Institudo Oswaldo Cruz Rio De Jan. 1992, 87 (Suppl. S3), 131–132. [Google Scholar] [CrossRef] [Green Version]
- Wei, Q.; Tsuji, M.; Zamoto, A.; Kohsaki, M.; Matsui, T.; Shiota, T.; Telford, S.R., III; Ishihara, C. Human Babesiosis in Japan: Isolation of Babesia microti -Like Parasites from an Asymptomatic Transfusion Donor and from a Rodent from an Area Where Babesiosis Is Endemic. J. Clin. Microbiol. 2001, 39, 2178–2183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Osorno, B.M.; Vega, C.; Ristic, M.; Robbles, C.; Ibarra, S. Isolation of Babesia sp. from asymptomatic human beings. Vet. Parasitol. 1976, 2, 111–120. [Google Scholar] [CrossRef]
- Lorelei, L.C.; Lora, R.B.; Kelly, A.; Susan, E.L.; Michael, R.L. Prevalence of select vector-borne disease agents in owned dogs of Ghana. J. S. Afr. Vet. Assoc. 2014, 85, 996. [Google Scholar]
- Kuleš, J.; Mrljak, V.; Rafaj, R.B.; Selanec, J.; Burchmore, R.; Eckersall, P.D. Identification of serum biomarkers in dogs naturally infected with Babesia canis canis using a proteomic approach. BMC Vet. Res. 2014, 10, 111. [Google Scholar] [CrossRef] [Green Version]
- El-Bahnasawy, M.M.; Khalil, H.H.; Morsy, T.A. Babesiosis in an Egyptian boy acquired from pet dog, and a general review. J. Egypt. Soc. Parasitol. 2011, 41, 99–108. [Google Scholar] [PubMed]
| Primer Code (Name) | Oligonucleotide Sequence | Used for Detection of (Piroplasms) | Target Gene or Region | Size | Ref. |
|---|---|---|---|---|---|
| Bab5/ Bab8 | AATTACCCAATCCTGACACAGG TTTCGCAGTAGTTCGTCTTTAACA | Most Babesia/Theleria spp. (including B. microti, B. divergens and T. annae) | 18S rRNA | about 400 bp | [3,22] |
| Bab6/ Bab7 | GACACAGGGAGGTAGTGACAAGA CCCAACTGCTCCTATTAACCATTAC | ||||
| BoF/ BoR | CACGAGGAAGGAACTACCGATGTTGA CCAAGGAGCTTCAACGTACGAGGTCA | B. bovis | rap-1 | 354 bp | [25] |
| ND | GTTTCTGMCCCATCAGCTTGAC CAATATTAACACCACGCAAAAATTC | Bovine Babesia spp. (including B. divergens) | 18S rRNA | 422–440 bp | [23] |
| BcW-A/ BcW-B | CATCTAAGGAAGGCAGCAGG TTAATGGAAACGTCCTTGGC | B. canis | 18S rRNA | 500 bp | [24] |
| Primers | Denaturation | Annealing | Extension | Cycles | Fragment Size |
|---|---|---|---|---|---|
| B. divergens | 94 °C (30 s) | 61 °C (30 s) | 72 °C (45 s) | 35 | 353 bp |
| B canis | 94 °C (2 min) | 60 °C (30 s) | 72 °C (30 s) | 35 | 509 bp |
| B. bovis | 94 °C (30 s) | 63 °C (30 s) | 72 °C (1 min) | 35 | 350 bp |
| Host | N | “Suspected” Based on Microscopy n (%) | Amplification of “Suspected” by PCR, n (%) | |||
|---|---|---|---|---|---|---|
| Babesia/Theileria# N * n * | B. bovis # N * n * | B. divergens # N * n * | B. canis # N * n * | |||
| Cattle | 30 | 10 (33) | 9 (30) 9 (90) | 0 (0) 0 (0) | 0(0) 0 (0) | NS NS |
| Dogs | 33 | 3 (9) | 0 (0) 0 (0) | NS NS | NS NS | 1 (3) 1 (33) |
| Humans | 150 | 6 (4) | 0 (0) 0 (0) | 0 (0) 0 (0) | 0 (0) 0(0) | 0 (0) 0 (0) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ayeh-Kumi, P.F.; Owusu, I.A.; Tetteh-Quarcoo, P.B.; Dayie, N.T.K.D.; Adutwum-Ofosu, K.K.; Amponsah, S.K.; Udofia, E.A.; Afutu, E.; Attah, S.K.; Armah, R.; et al. Preliminary Investigation into Plasmodium-like Piroplasms (Babesia/Theileria) among Cattle, Dogs and Humans in A Malaria-Endemic, Resource-Limited Sub-Saharan African City. Med. Sci. 2022, 10, 10. https://doi.org/10.3390/medsci10010010
Ayeh-Kumi PF, Owusu IA, Tetteh-Quarcoo PB, Dayie NTKD, Adutwum-Ofosu KK, Amponsah SK, Udofia EA, Afutu E, Attah SK, Armah R, et al. Preliminary Investigation into Plasmodium-like Piroplasms (Babesia/Theileria) among Cattle, Dogs and Humans in A Malaria-Endemic, Resource-Limited Sub-Saharan African City. Medical Sciences. 2022; 10(1):10. https://doi.org/10.3390/medsci10010010
Chicago/Turabian StyleAyeh-Kumi, Patrick F., Irene A. Owusu, Patience B. Tetteh-Quarcoo, Nicholas T. K. D. Dayie, Kevin Kofi Adutwum-Ofosu, Seth K. Amponsah, Emilia A. Udofia, Emmanuel Afutu, Simon K. Attah, Robert Armah, and et al. 2022. "Preliminary Investigation into Plasmodium-like Piroplasms (Babesia/Theileria) among Cattle, Dogs and Humans in A Malaria-Endemic, Resource-Limited Sub-Saharan African City" Medical Sciences 10, no. 1: 10. https://doi.org/10.3390/medsci10010010







