Effects of pH-Induced Changes in Soil Physical Characteristics on the Development of Soil Water Erosion
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Physcial Characteristics of Simulated Soils at Different pH Values
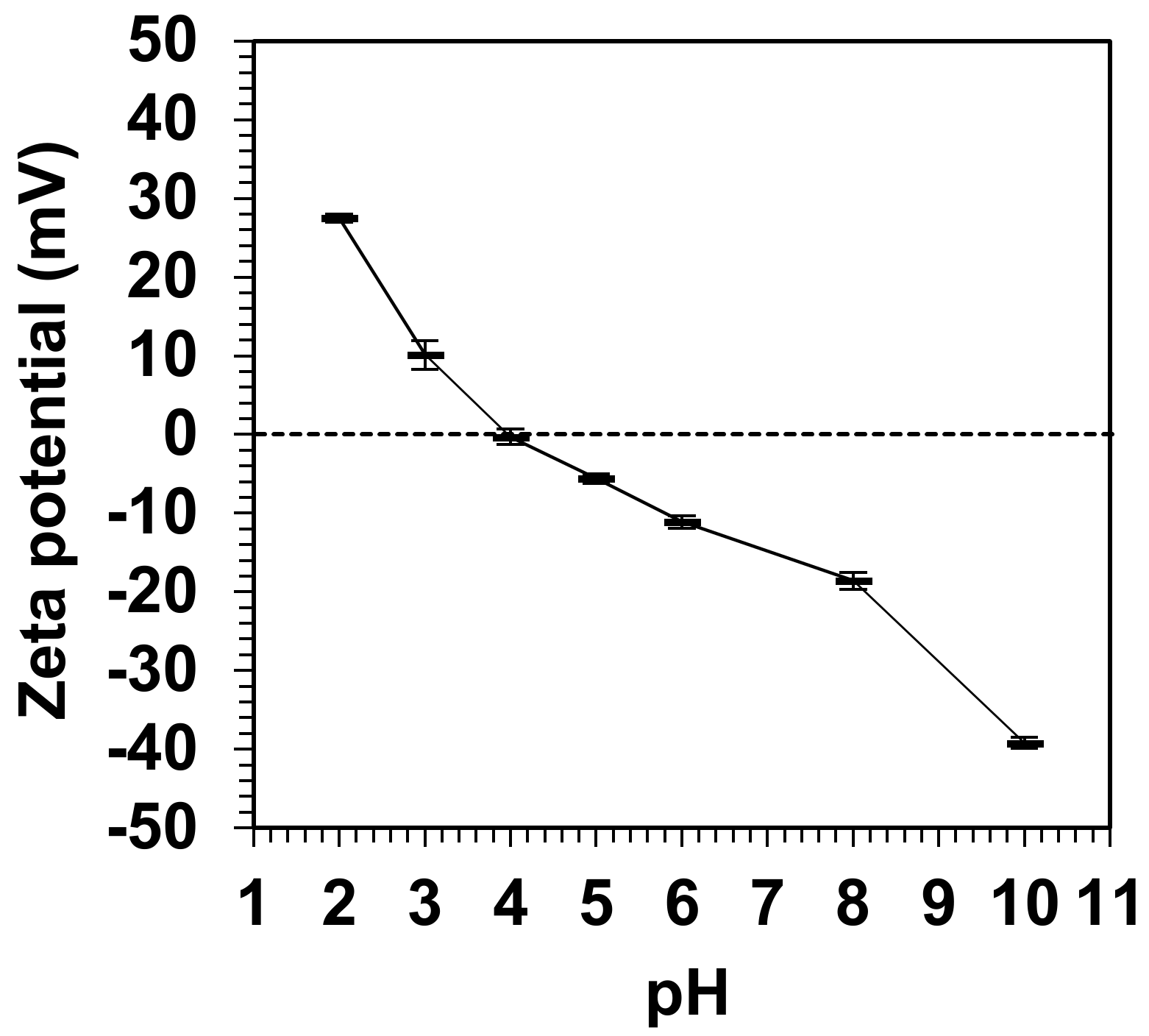
2.3. Zeta-Potential Measurement
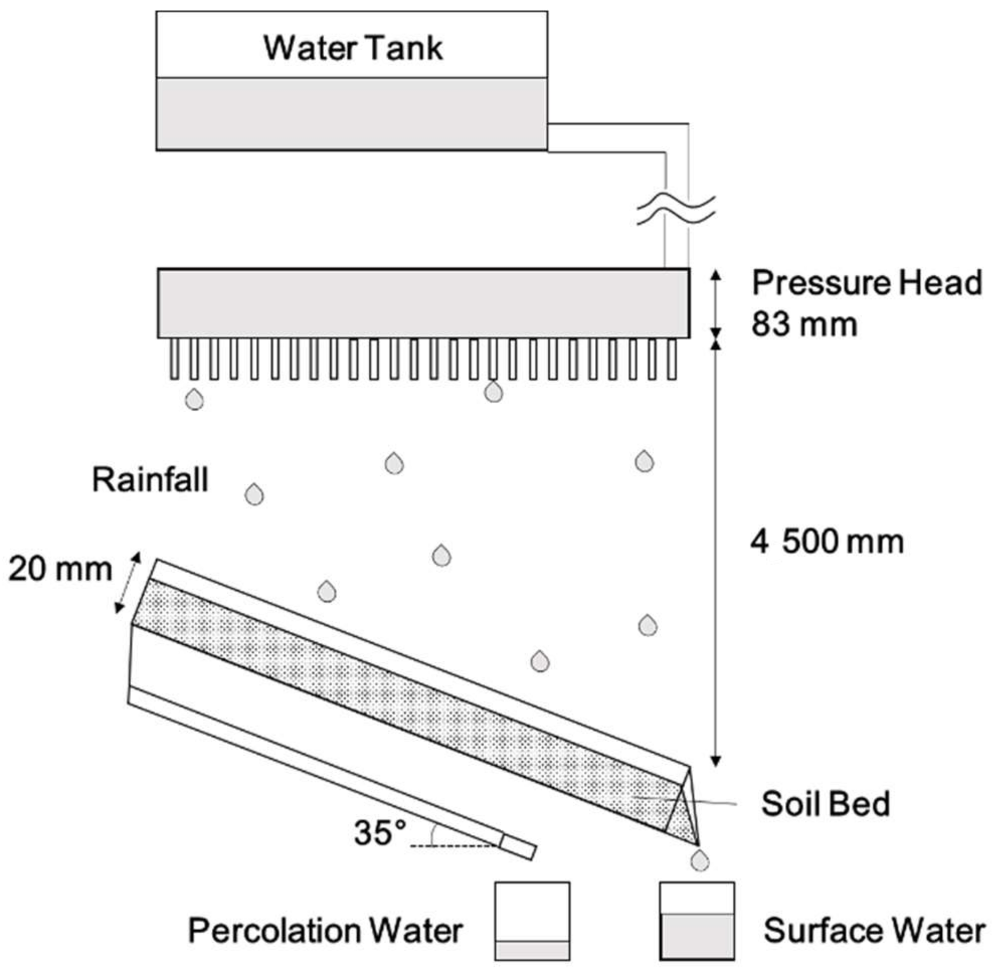
2.4. Artificial Rainfall Test
3. Results
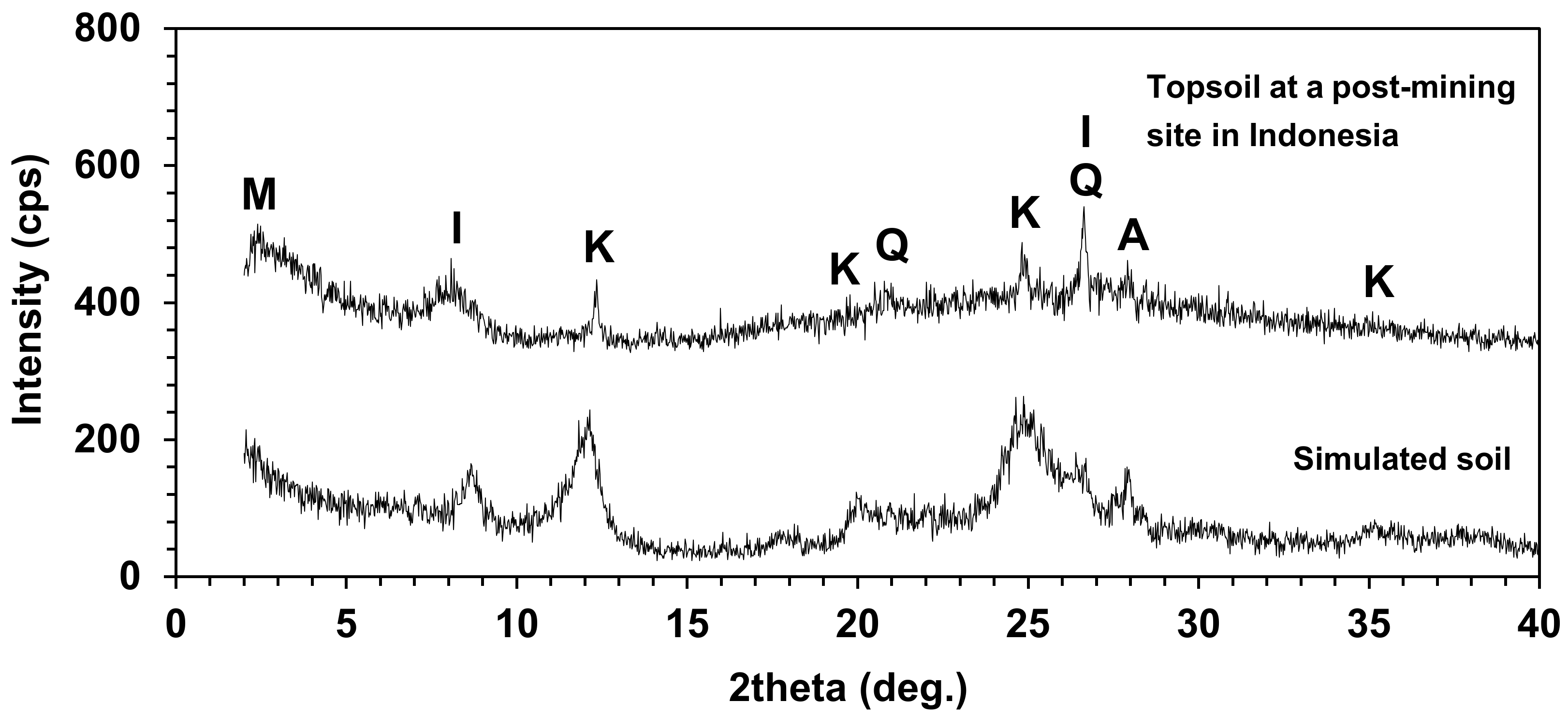
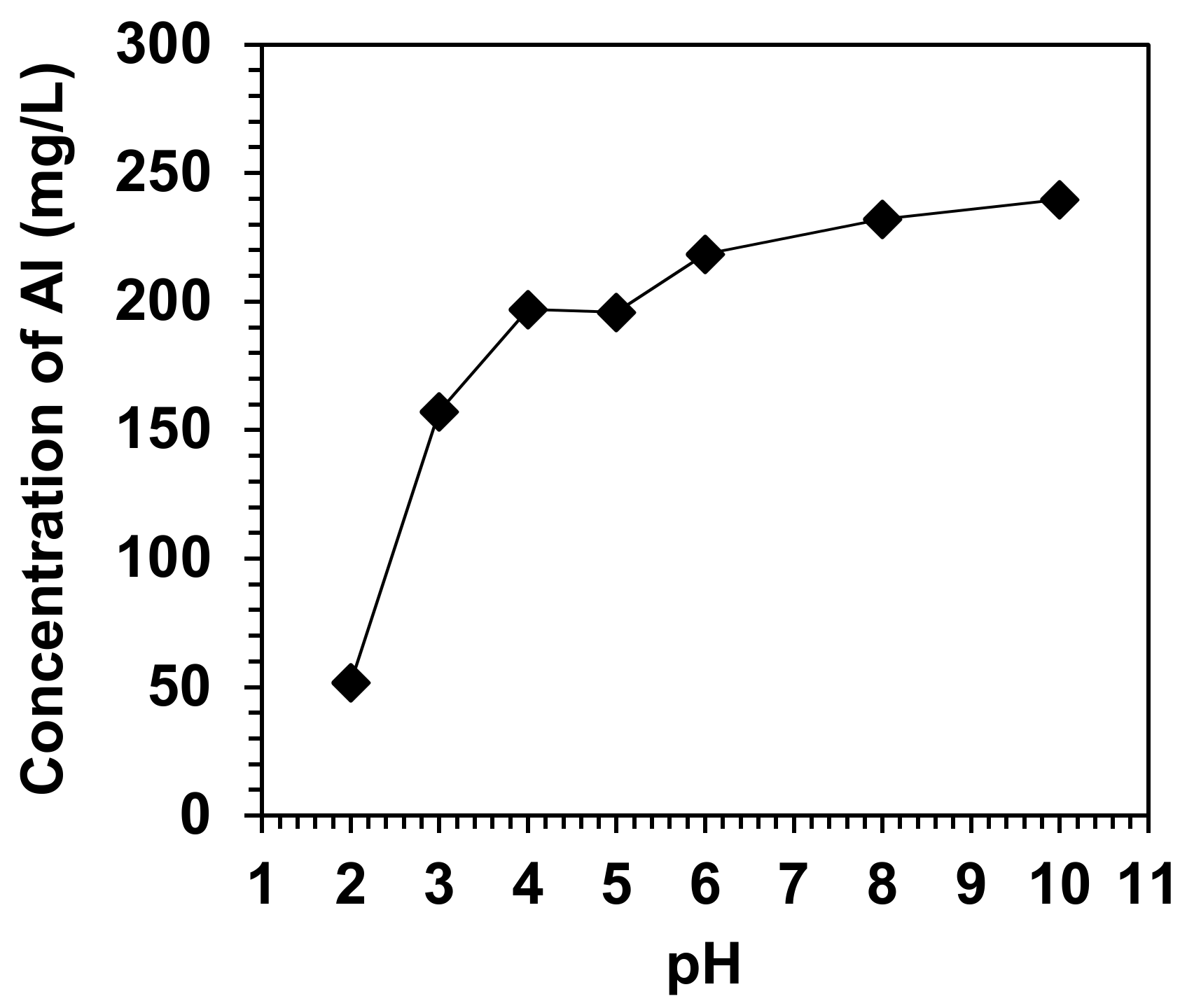
3.1. Chemical and Physical Properties of Simulated Soils
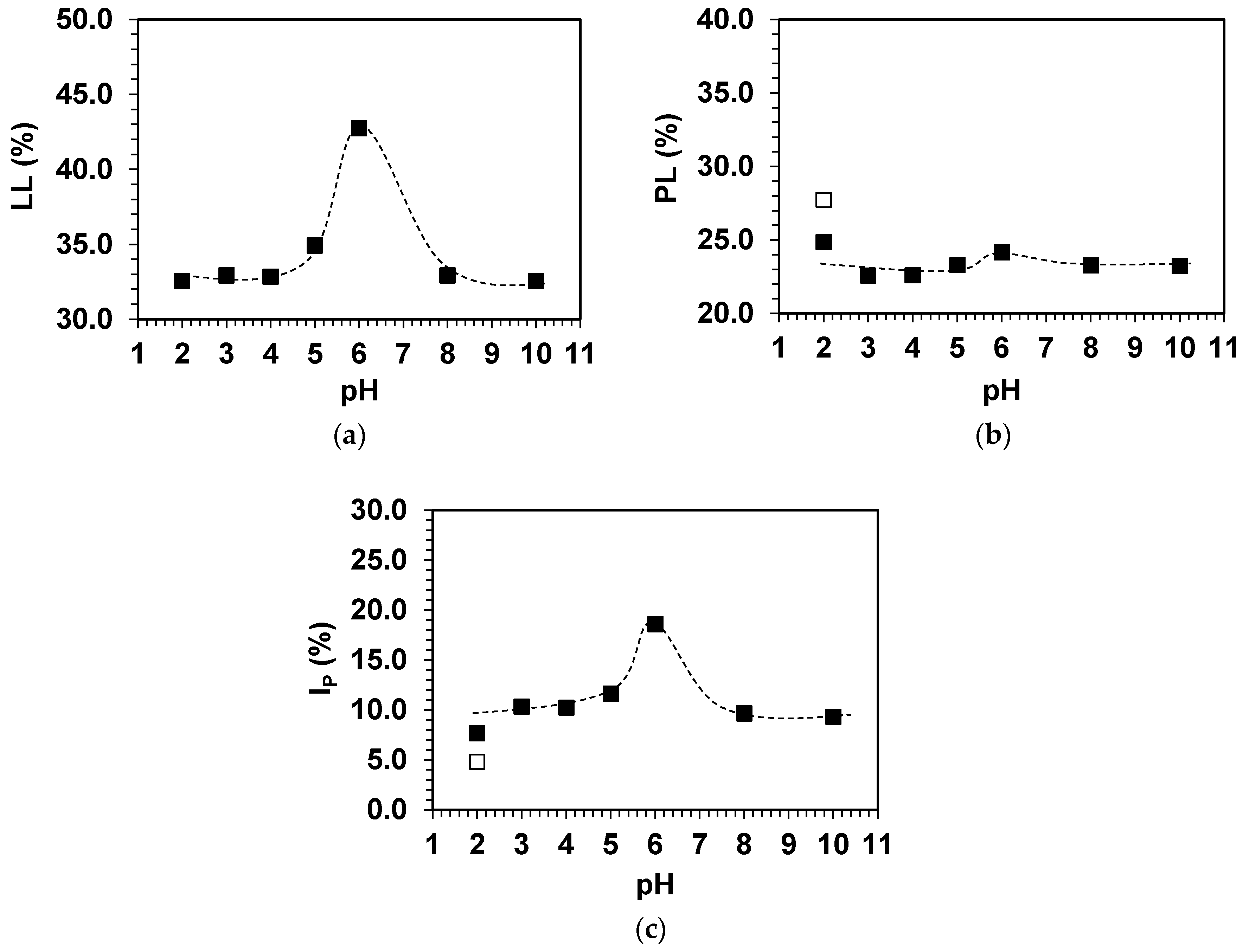
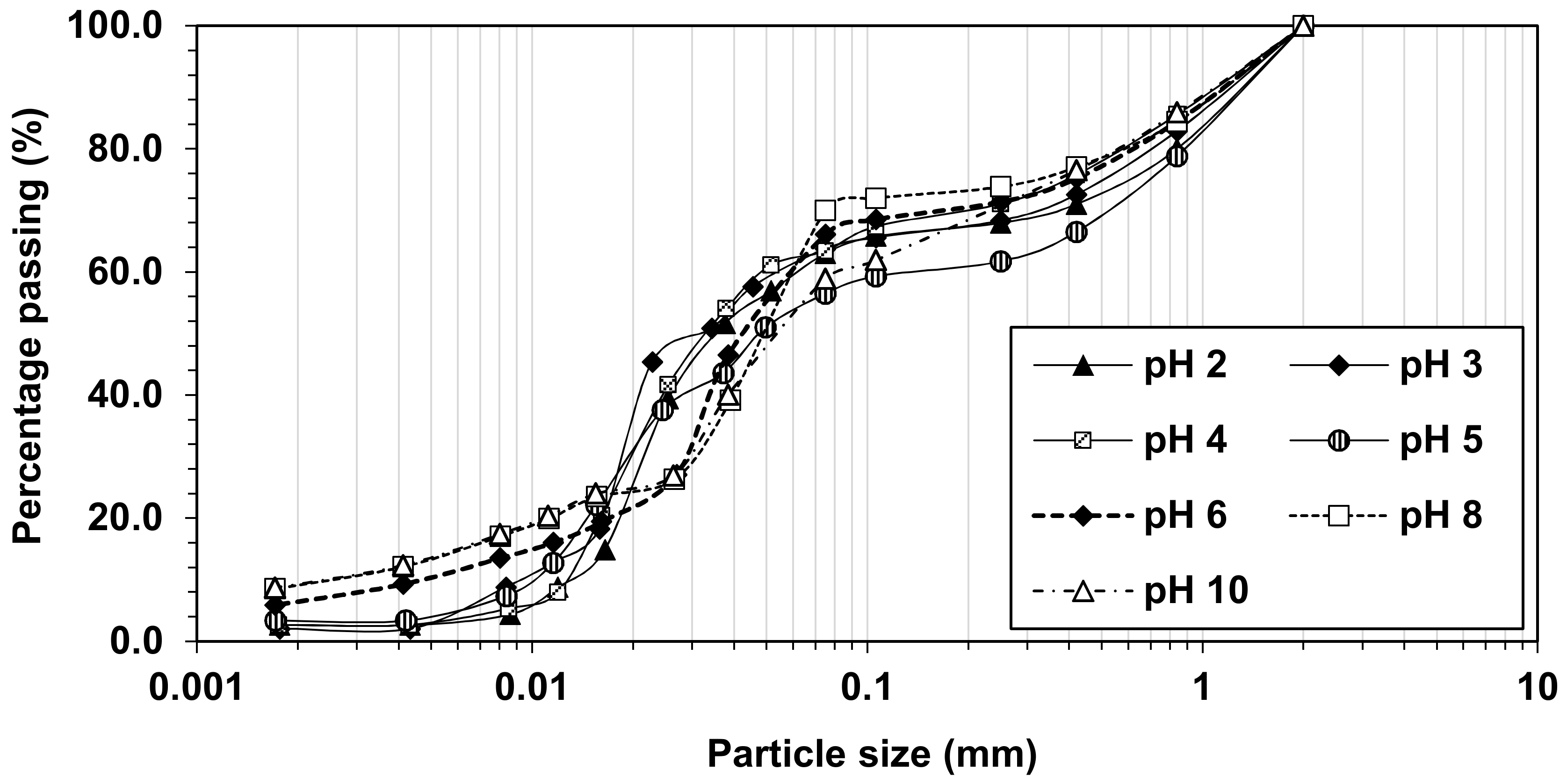
3.2. Effects of pH on the Physical Characteristics of Simulated Soils
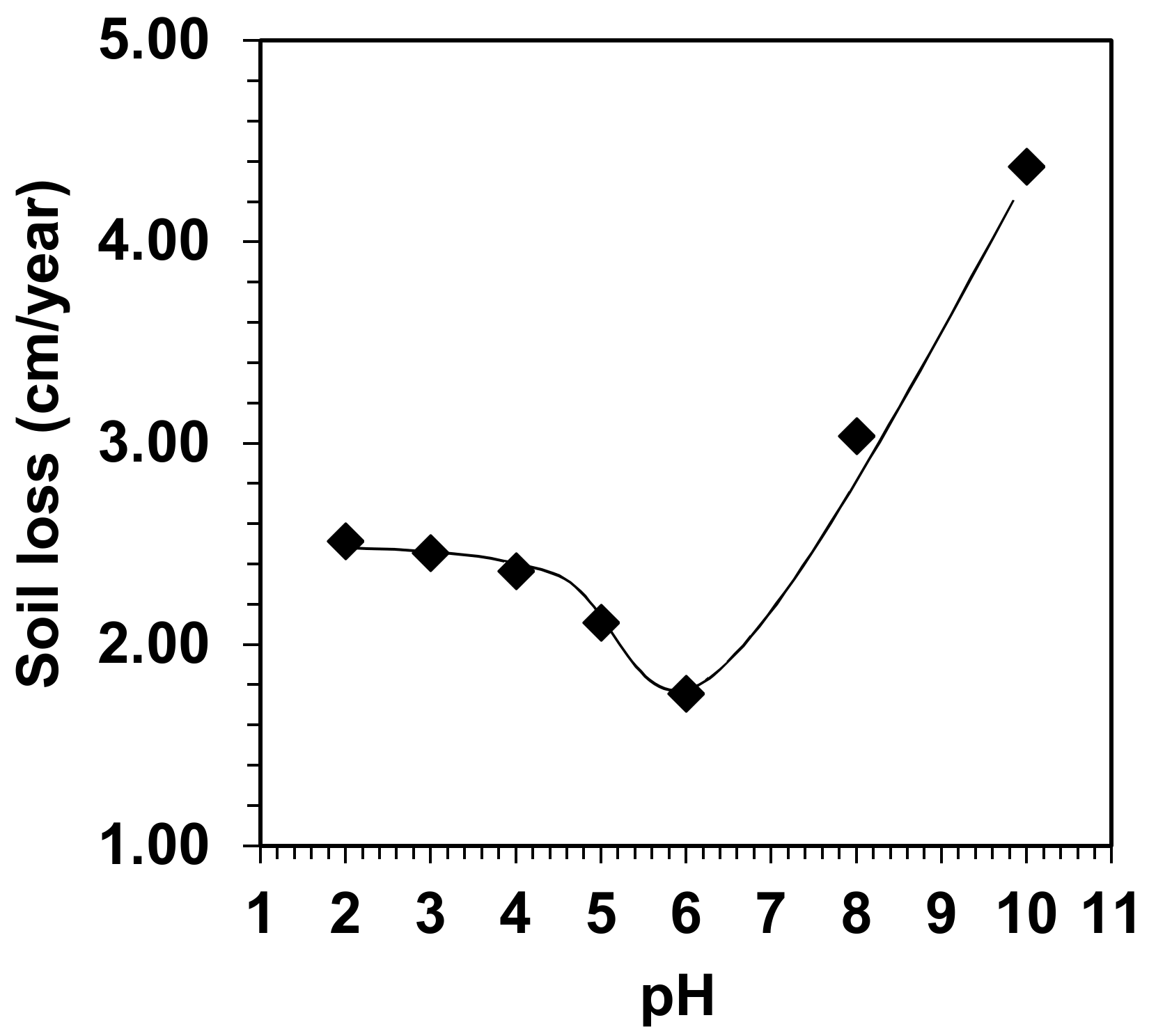
3.3. Development of Soil Water Erosion at Different pH Values
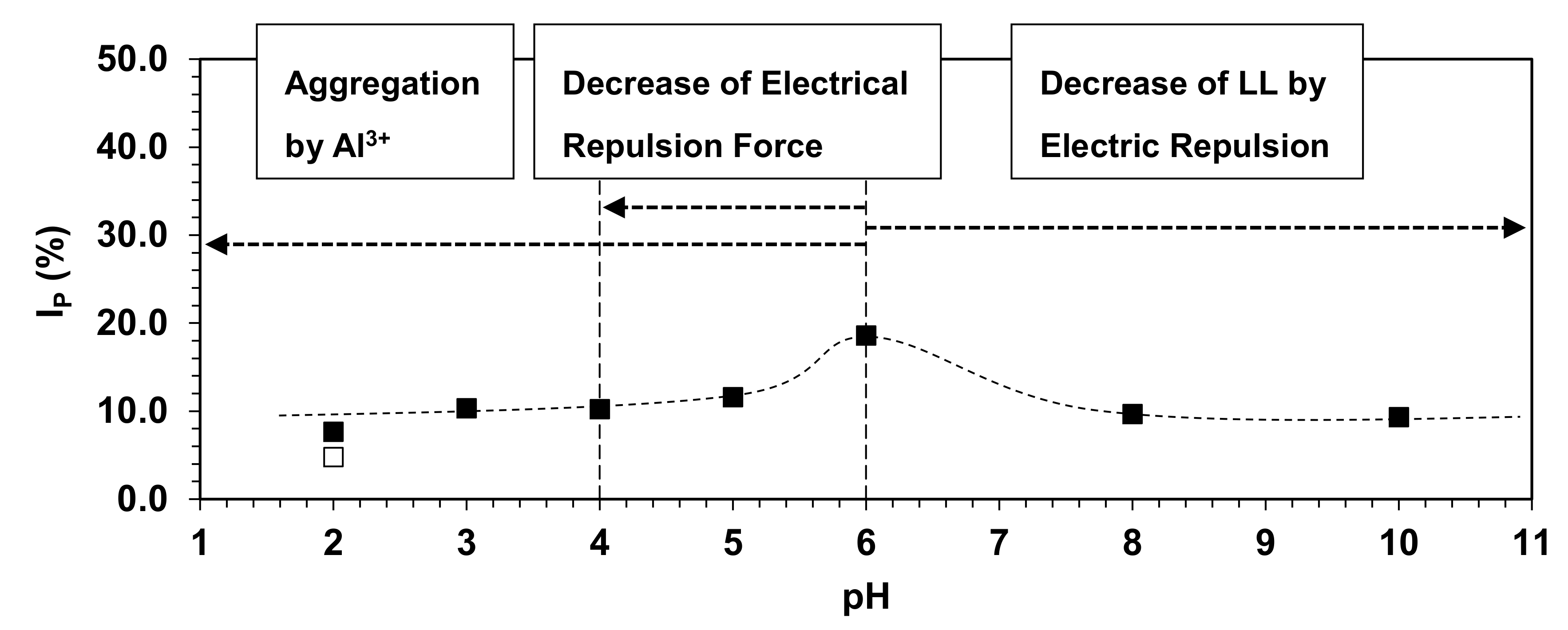
3.4. Implication of pH-Induced Changes in Soil Physical Characteristics with Soil Water Erosion Model
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yusuf, M.; Arisoesilaningsih, E. Exotic plant species attack revegetation plants in post-coal mining areas. AIP Conf. Proc. 2017, 1908, 1–5. Available online: https://doi.org/10.1063/1.5012716 (accessed on 27 January 2018).
- Sheoran, V.; Sheoran, A.S.; Poonia, P. Soil reclamation of abandoned mine land by revegetation: A review. Int. J. Soil Sediment Water 2010, 3, 13. [Google Scholar]
- Kusumandari, A.; Mitchell, B. Soil erosion and sediment yield in forest and agroforestry areas in West Java, Indonesia. J. Soil Water Conserv. 1997, 52, 376–380. [Google Scholar]
- Elsheikh, R.F.A.; Ouerghi, S.; Elhag, A.R. Soil erosion risk map based on geographic information system and universal soil loss equation (case study: Terengganu, Malaysia). Ind. J. Sci. Res. Technol. 2015, 3, 38–43. [Google Scholar]
- Phuong, T.T.; Thong, C.V.T.; Ngoc, N.B.; Chuong, H.V. Modeling soil erosion within small mountainous watershed in central Vietnam using GIS and SWAT. Resour. Environ. 2014, 4, 139–147. [Google Scholar]
- Hamanaka, A.; Inoue, N.; Shimada, H.; Sasaoka, T.; Matsui, K. An Experimental Study for Assessment of Soil Erosion at Rehabilitation Area in Indonesian Coal Mine. In Proceedings of the International Symposium on Land Reclamation and Ecological Restoration (LRER’14), Beijing, China, 16–19 October 2014; pp. 171–175. [Google Scholar]
- Deng, Y.; Cai, C.; Xia, D.; Ding, S.; Chen, J.; Wang, T. Soil atterberg limits of different weathering profiles of the collapsing gullies in the hilly granitic region of southern china. Solid Earth 2017, 8, 499–513. [Google Scholar] [CrossRef]
- Sharmeen, S.; Willgoose, G.R. The interaction between Armouring and particle weathering for eroding landscapes. Earth Surf. Process. Landf. 2006, 31, 1195–1210. [Google Scholar] [CrossRef]
- Marcus, J.J. Mining Environmental Handbook: Effects of Mining on the Environment and American Environmental Controls on Mining; Imperial College Press: London, UK, 1997; pp. 136–138. [Google Scholar]
- El-swaify, S.A.; Dangler, E.W.; Armstrong, C.L. Soil Erosion by Water in the Tropics. Research Extension Series 024; 1982. Available online: http://pdf.usaid.gov/pdf_docs/Pnaar134.pdf (accessed on 13 March 2018).
- Zhang, L.; Wang, J.M.; Bai, Z.K.; Lv, C.J. Effects of vegetation on runoff and soil erosion on reclaimed land in an opencast coal-mine dump in a loess area. Catena 2015, 128, 44–53. [Google Scholar] [CrossRef]
- Dubey, A.; Nath, R. Erosion Problems of Mine Waste Dump Deposited During Opencast Mining Activity in Highland and Headwater Regions. In Proceedings of the 3rd International Symposium on Headwater Control, New Delhi, India, 6–8 October 1995; pp. 189–198. [Google Scholar]
- Matsumoto, S.; Shimada, H.; Sasaoka, T.; Kusuma, G.J.; Gautama, R.S. Construction of dry cover system for prevention of acid mine drainage at mine waste dump in open cast coal mines. J. Environ. Prot. 2016, 7, 160–169. [Google Scholar] [CrossRef]
- Matsumoto, S.; Ishimatsu, H.; Shimada, H.; Sasaoka, T.; Matsui, K.; Kusuma, G.J. Prevention of acid mine drainage (AMD) by using sulfur-bearing rocks for a cover layer in a dry cover system in view of the form of sulfur. J. Pol. Miner. Eng. Soc. 2015, 2, 29–35. [Google Scholar]
- Kusuma, G.J.; Shimada, H.; Sasaoka, T.; Matsui, K.; Nugraha, C.; Gautama, R.S.; Sulistianto, B. Physical and geochemical characteristics of coal mine overburden dump related to acid mine drainage generation. Mem. Fac. Eng. 2012, 72, 23–38. [Google Scholar]
- Gratchev, I.; Towhata, I. Stress-strain characteristics of two natural soils subjected to long-term acidic contamination. Soils Found. 2013, 53, 469–476. [Google Scholar] [CrossRef]
- Hasan, M.R.; Hasan, M.H.; Islam, M.R.; Razi, K.A.A.; Alam, S.; Abdullah, T. Changes in geotechnical properties of soil with pH in household and industrial waste dump site. Int. J. Appl. Sci. Eng. Res. 2013, 2, 119–127. [Google Scholar]
- Dimitrakopoulos, D.; Vasileiou, E.; Stathopoulos, N.; Dimitrakopoulou, S. Estimation of the Qualitative Characteristics of Post Mining Lakes in Different Lignite Fields in Greece. In Proceedings of the International Mine Water Association Symposium (IMWA) 2016, Freiberg, Germany, 11–15 July 2016; pp. 219–225. [Google Scholar]
- Tajnin, R.; Abdullah, T.; Rokonuzzaman, M.D. Study on the salinity and pH and its effect on geotechnical properties of soil in south-west region of Bangladesh. Int. J. Adv. Struct. Geotech. Eng. 2014, 3, 138–147. [Google Scholar]
- Matsumoto, S.; Ogata, S.; Shimada, H.; Sasaoka, T.; Kusuma, G.J.; Gautama, R.S. Application of coal ASH to postmine land for prevention of soil erosion in coal mine in Indonesia: Utilization of fly ash and bottom ASH. Adv. Mater. Sci. Eng. 2016, 2016, 8386598. [Google Scholar] [CrossRef]
- American Society for Testing and Materials (ASTM). Standard Test Methods for Liquid Limit, Plastic Limit, and Plasticity Index of Soils; ASTM D4318-17; ASTM International: West Conshohocken, PA, USA, 2017. [Google Scholar]
- American Society for Testing and Materials (ASTM). Standard Test Method for Particle-Size Analysis of Soils (Withdrawn 2016); ASTM D422-63(2007)e2; ASTM International: West Conshohocken, PA, USA, 2007. [Google Scholar]
- Hamanaka, A.; Inoue, N.; Shimada, H.; Sasaoka, T.; Matsui, K.; Miyajima, I. Study on backfilling of soil for revegetation at the rehabilitation area in Indonesian coal mine. In Mine Planning and Equipment Selection; Drebenstedt, C., Singhal, R., Eds.; Springer International Publishing: Cham, Switzerland, 2013; pp. 803–811. [Google Scholar]
- Annual Weather Review. 2011. Available online: http://www.nea.gov.sg/training-knowledge/publications/annual-weather-review-2011 (accessed on 29 January 2018).
- Hamanaka, A.; Shimada, H.; Sasaoka, T.; Matsui, K.; Miyajima, I.; Ichinose, M. Assessment of Soil Erosion at the Rehabilitation Area in Indonesian Coal Mine. In Proceedings of the Spring Annual Symposium on the Mining and Materials Processing, Tokyo, Japan, 28–30 March 2013; pp. 370–371. [Google Scholar]
- Dina, L. Analysis Finansial Jeruk Keprok di Kabupaten Kutai Timur (Prospect of Keprok Orange in Kutai Timur Regency). EPP 2009, 6, 36–43. Available online: http://agb.faperta.unmul.ac.id/wp-content/uploads/2017/04/jurnal-vol-6-no-1-dina.pdf (accessed on 29 January 2018).
- Shimada, H.; Matsumoto, S.; Hamanaka, A.; Sasaoka, T.; Kusuma, G.J. Effects of Acid Soil Properties on Soil Erosion at Dumping Site in Open-pit Coal Mine, Indonesia. In Proceedings of the 2nd International Symposium on Land Reclamation and Ecological Restoration, Beijing, China, 20–23 October 2017; pp. 239–244. [Google Scholar]
- White, W.A. Atterberg Plastic Limits of Clay Minerals. Report of Investigations—No. 144. 1949. Available online: http://www.minsocam.org/ammin/AM34/AM34_508.pdf (accessed on 29 January 2018).
- Cerqueira, A.A.; Marques, M.R.D.C. Electrolytic treatment of wastewater in the oil industry. In New Technologies in the Oil and Gas Industry; Gomes, J.S., Ed.; InTech Publisher: Rijeka, Croatia, 2012. [Google Scholar]
- Yukselen-aksoy, Y.; Kaya, A. The zeta potential of a mixed mineral clay in the presence of Cations. J. Eng. Earth Sci. 2016, 1, 14–21. [Google Scholar]
- Yong, R.N.; Warkentin, B.P. Introduction to Soil Behavior; The Macmillan Company: New York, NY, USA, 1966. [Google Scholar]
- Ritung, S.; Agus, F.; Hidayat, H. Land Suitability Evaluation with a Case Map of Aceh Barat District; Indonesian Soil Research Institute and World Agroforestry Center: Bogor, Indonesia, 2007. [Google Scholar]
- Singh, A.; Agrawal, M. Acid rain and its ecological consequences. J. Environ. Biol. 2008, 29, 15–24. [Google Scholar] [PubMed]
- Khemani, L.T.; Momin, G.A.; Rao, P.S.P.; Pillai, A.G.; Safai, P.D.; Mohan, K.; Rao, M.G. Atmospheric pollutants and their influence on acidification of rain water at an industrial location on the west coast of India. Atmos. Environ. 1994, 28, 3145–3154. [Google Scholar] [CrossRef]
- Zhang, D.D.; Peart, M.R.; Jim, C.Y. Alkaline rains on the Tibetan Plateau and their implication for the original pH of natural rainfall. J. Geophys. Res. 2002, 107, 9-1–9-6. [Google Scholar] [CrossRef]
- Khan, M.N.; Sarwar, A. Chemical composition of wet precipitation of air pollutants: A case study in Karachi, Pakistan. Atmosfera 2014, 27, 35–46. [Google Scholar] [CrossRef]
- Sumiran, S.G.; Abha, R.; Ranjit, K.; Pal, S.S.; Anita, L.; Maharaj, K.K.; Saran, S.S. Status of Acid Rain in India and Study on Rainwater Composition at Gopalpura (Agra); Bharatiya Vaigyanik evam Audyogik Anusandhan Patrika (BVAAP): New Delhi, India, 2000; pp. 15–22. [Google Scholar]
- Bartnik, A.; Moniewski, P. Changes in water quality of a small urban river triggered by deep drainage of a construction site. J. Water Land Dev. 2016, 31, 11–22. [Google Scholar] [CrossRef][Green Version]
- Gomes, H.I.; Mayes, W.M.; Rogerson, M.; Stewart, D.I.; Burke, I.T. Alkaline residues and the environment: A review of impacts, management practices and opportunities. J. Clean. Prod. 2016, 112, 3571–3582. [Google Scholar] [CrossRef]
- Hull, S.L.; Oty, U.V.; Mayes, W.M. Rapid recovery of benthic invertebrates 8 downstream of hyperalkaline steel slag discharges. Hydrobiologia 2014, 736, 83–97. [Google Scholar] [CrossRef]
- Byerly, D.W. Handling acid-producing material during construction. Environ. Eng. Geosci. 1996, 2, 49–57. [Google Scholar] [CrossRef]
- Flues, M.; Hama, P.; Lemes, M.J.L.; Dantas, E.S.K.; Fornaro, A. Evaluation of the rainwater acidity of a rural region due to a coal-fired power plant in Brazil. Atmos. Environ. 2002, 36, 2397–2404. [Google Scholar] [CrossRef]
- Gasnari, B.P.; Ramesh, H. Assessment of soil erosion by RUSLE model using remote sensing and GIS—A case study of Nethravathi Basin. Geosci. Front. 2016, 7, 953–961. [Google Scholar]
- Sang, D.P.; Kyu, S.L.; Seung, S.S. A statistical soil erosion model for burnt mountain areas in Korea-RUSLE Approach. J. Hydrol. Eng. 2012, 17, 292–304. [Google Scholar]
- Prasannakumar, V.; Vijith, H.; Geetha, N. Estimation of soil erosion risk within a small mountainous sub-watershed in Kerala, India, using revised universal soil loss equation (RUSLE) and geoinformation technology. Geosci. Front. 2011, 3, 209–215. [Google Scholar] [CrossRef]

| Sample | Sand (%) | Silt (%) | Clay (%) | Texture |
|---|---|---|---|---|
| Topsoil in A coal mines in Indonesia | 22.0–75.0 | 8.0–32.1 | 17.0–48.0 | Light Clay Sandy Clay Loam Heavy Clay |
| Simulated soils | 36.7 | 26.7 | 36.6 | Light Clay |
| Elements | (mass %) | Elements | (mass %) |
|---|---|---|---|
| SiO2 | 69.43 | Na2O | 2.16 |
| Al2O3 | 16.37 | K2O | 4.23 |
| Fe2O3 | 3.59 | P2O5 | 0.09 |
| MnO | 0.06 | H2O | 0.40 |
| MgO | 1.46 | SO3 | 0.04 |
| CaO | 1.58 | - | - |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matsumoto, S.; Ogata, S.; Shimada, H.; Sasaoka, T.; Hamanaka, A.; Kusuma, G.J. Effects of pH-Induced Changes in Soil Physical Characteristics on the Development of Soil Water Erosion. Geosciences 2018, 8, 134. https://doi.org/10.3390/geosciences8040134
Matsumoto S, Ogata S, Shimada H, Sasaoka T, Hamanaka A, Kusuma GJ. Effects of pH-Induced Changes in Soil Physical Characteristics on the Development of Soil Water Erosion. Geosciences. 2018; 8(4):134. https://doi.org/10.3390/geosciences8040134
Chicago/Turabian StyleMatsumoto, Shinji, Shunta Ogata, Hideki Shimada, Takashi Sasaoka, Akihiro Hamanaka, and Ginting Jalu Kusuma. 2018. "Effects of pH-Induced Changes in Soil Physical Characteristics on the Development of Soil Water Erosion" Geosciences 8, no. 4: 134. https://doi.org/10.3390/geosciences8040134
APA StyleMatsumoto, S., Ogata, S., Shimada, H., Sasaoka, T., Hamanaka, A., & Kusuma, G. J. (2018). Effects of pH-Induced Changes in Soil Physical Characteristics on the Development of Soil Water Erosion. Geosciences, 8(4), 134. https://doi.org/10.3390/geosciences8040134