Environmental and Health: The Importance of Tremolite Occurence in the Pollino Geopark (Southern Italy)
Abstract
:1. Introduction
2. Environmental and Toxicological Relevance
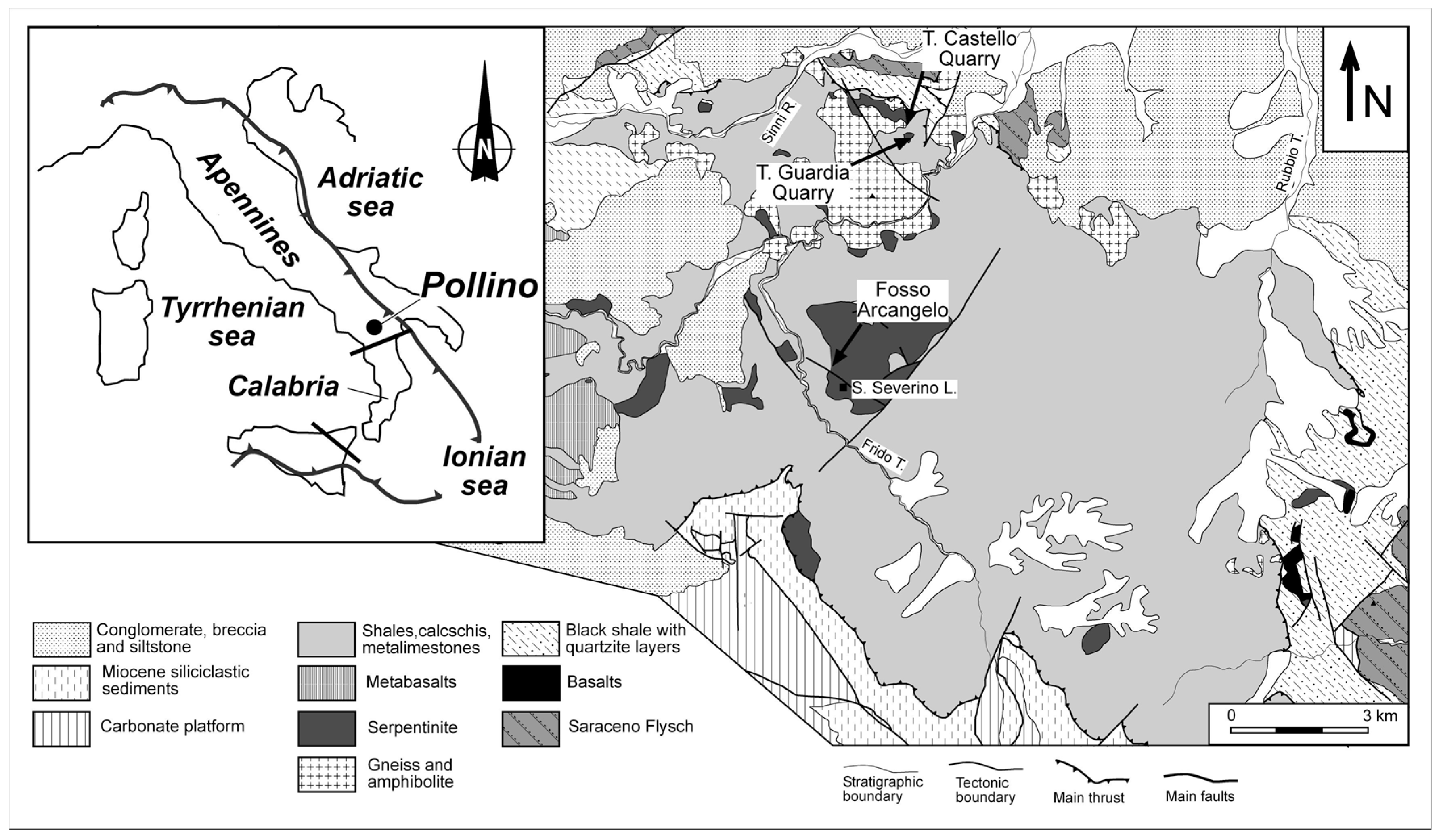
3. Geological Outline
4. Sampling
5. Analytical Methods
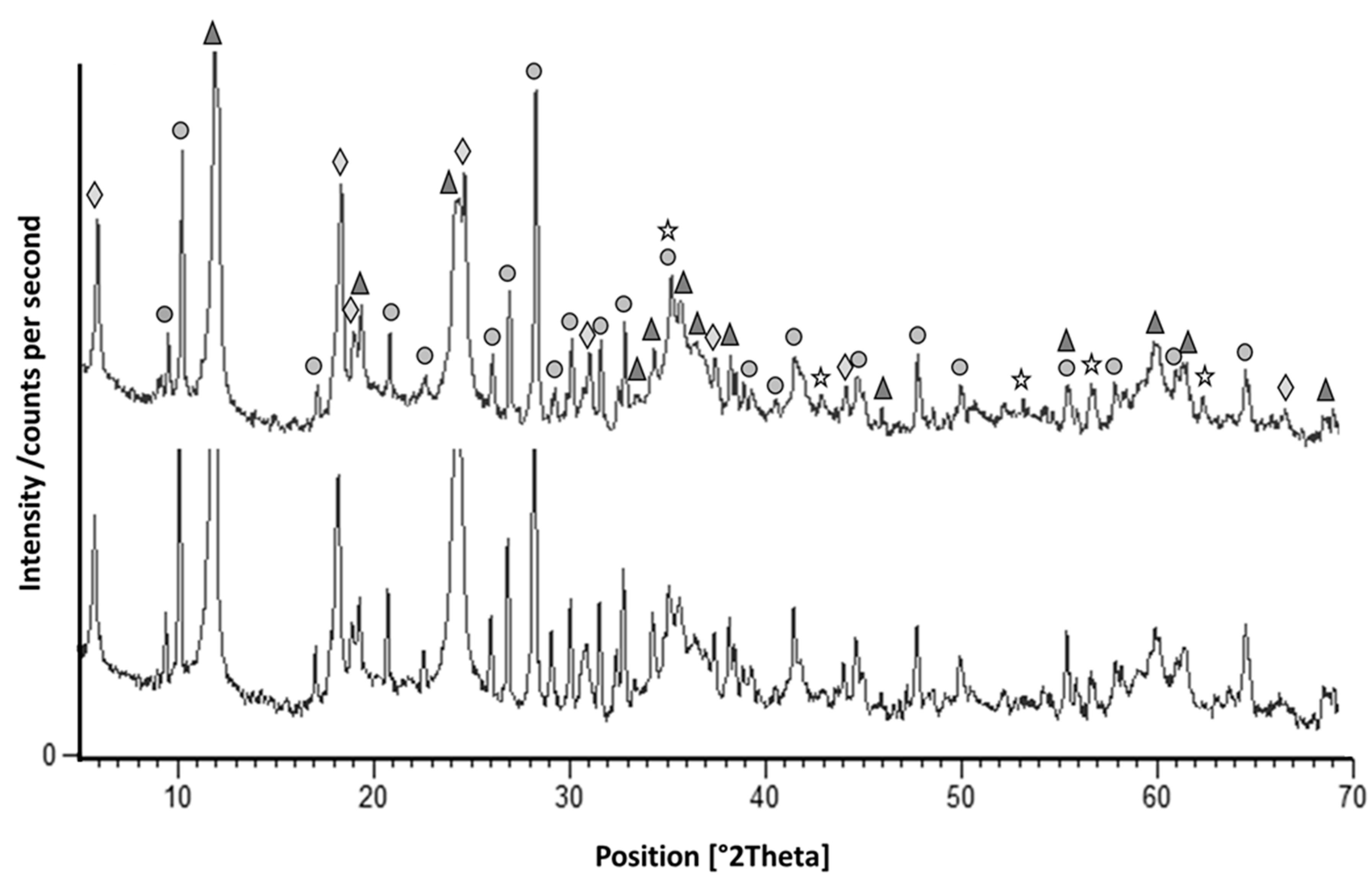
5.1. X-Ray Powder Diffraction
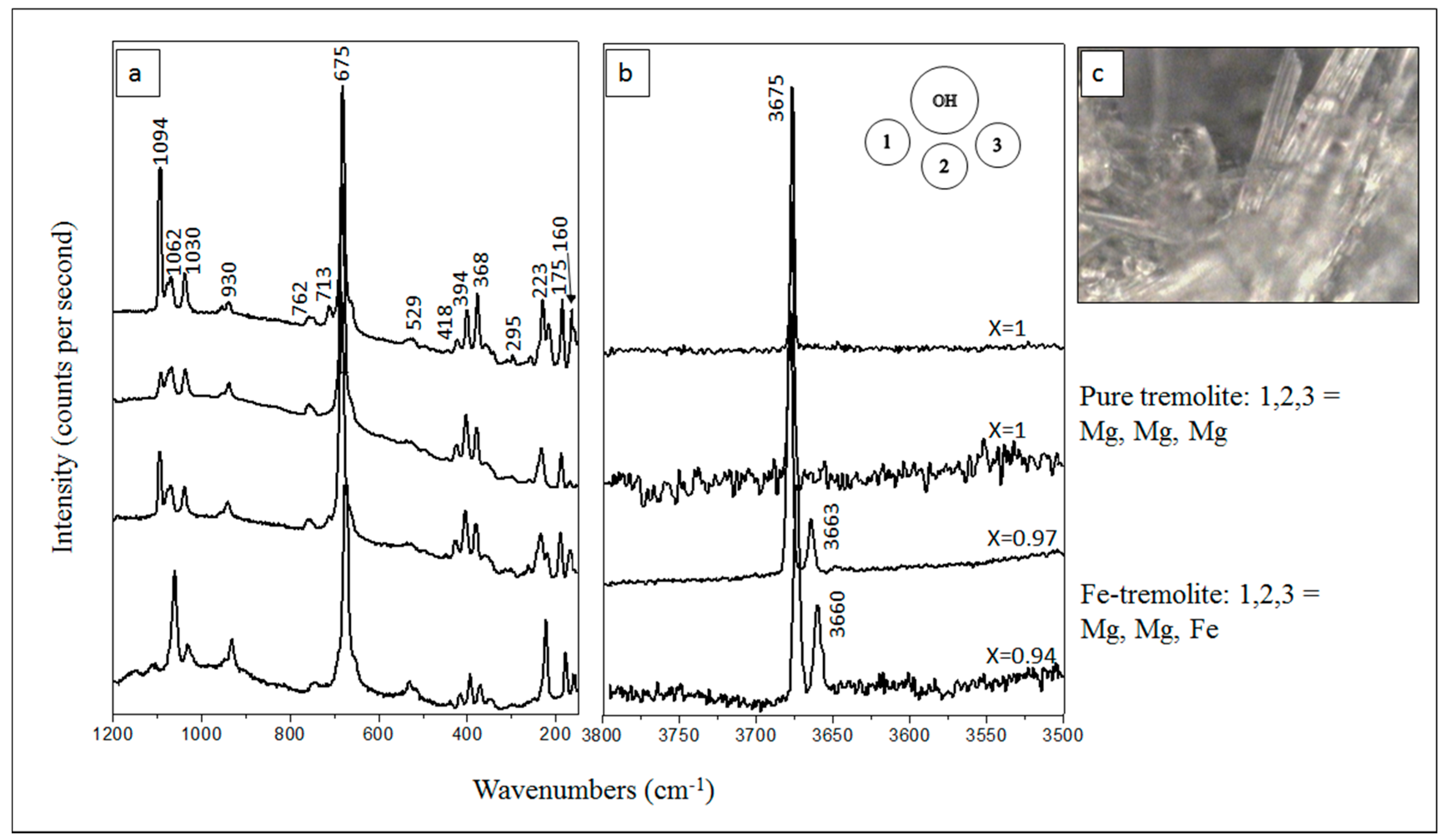
5.2. µ-Raman Spectroscopy
5.3. Electron Microprobe Analyses
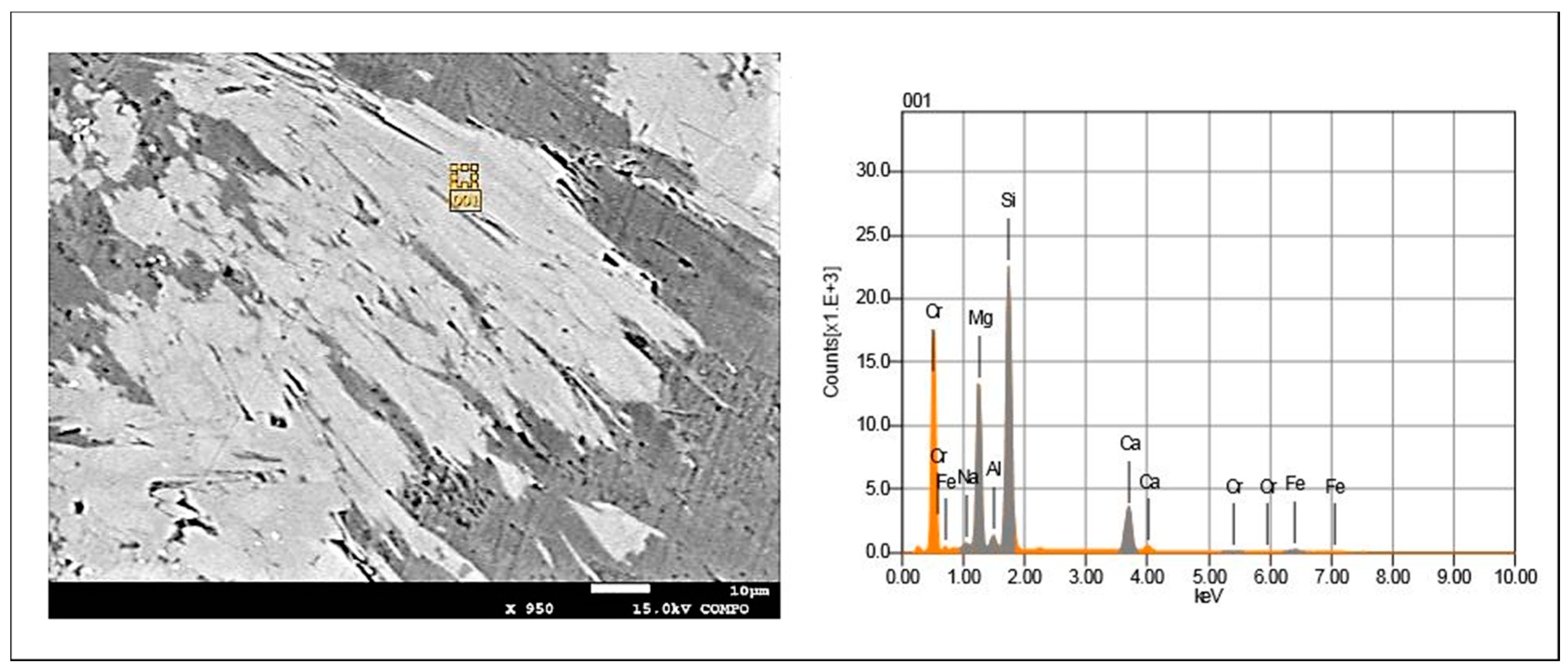
5.4. Scanning Electron Microscopy and Microanalysis EDS
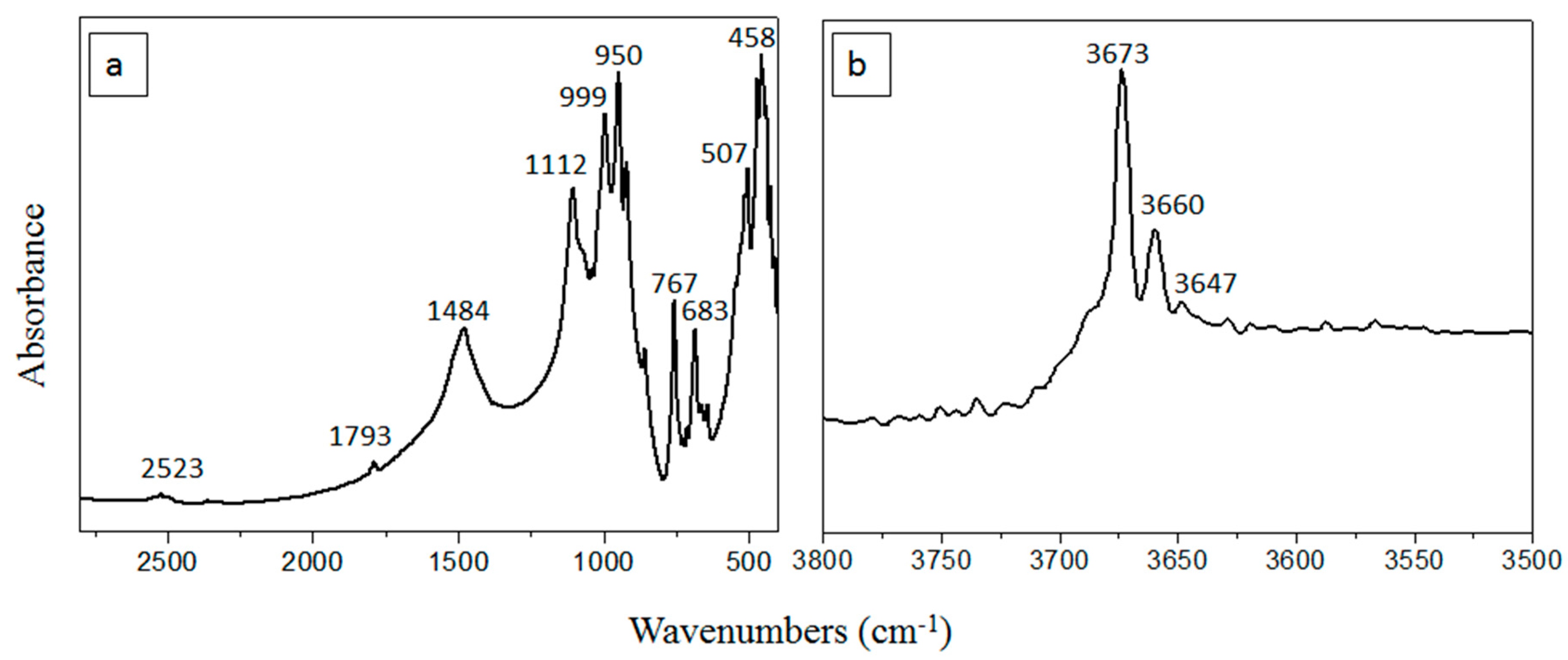
5.5. Fourier Transform InfraRed Spectroscopy
6. Results and Discussion
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bloise, A.; Catalano, M.; Critelli, T.; Apollaro, C.; Miriello, D. Naturally occurring asbestos: Potential for human exposure, San Severino Lucano (Basilicata, Southern Italy). Environ. Earth Sci. 2017, 76, 648. [Google Scholar] [CrossRef]
- Bloise, A.; Barca, D.; Gualtieri, A.F.; Pollastri, S.; Belluso, E. Trace elements in hazardous mineral fibres. Environ. Pollut. 2016, 216, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Margiotta, S.; Mongelli, G.; Paternoster, M.; Summa, V.; Fiore, S. Trace element distribution and Cr(VI) speciation in Ca-HCO3 and Mg-HCO3 spring waters from the northern sector of the Pollino massif, southern Italy. J. Geochem. Explor. 2012, 115, 1–12. [Google Scholar] [CrossRef]
- Mossman, B.T.; Lippmann, M.; Hesterberg, T.W.; Kelsey, K.T.; Barchowsky, A.; Bonner, J.C. Pulmonary Endpoints (Lung Carcinomas and Asbestosis) Following Inhalation Exposure to Asbestos. J. Toxicol. Environ. Health Part B 2011, 14, 76–121. [Google Scholar] [CrossRef] [PubMed]
- Brody, A.R. Asbestos and lung disease. Am. J. Respir. Cell Mol. Biol. 2010, 42, 131–132. [Google Scholar] [CrossRef] [PubMed]
- Jamrozik, E.; de Klerk, N.; Musk, A.W. Asbestos-related disease. Internal Med. J. 2011, 41, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Cheresh, P.; Kamp, D.W. Molecular basis of asbestos-induced lung disease. Annu. Rev. Pathol. 2013, 8, 161–187. [Google Scholar] [CrossRef] [PubMed]
- Stayner, L.; Welch, L.S.; Lemen, R. The worldwide pandemic of asbestos-related diseases. Annu. Rev. Public Health 2013, 34, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Gunter, M.E. Asbestos as a metaphor for teaching risk perception. J. Geol. Educ. 1994, 42, 17–24. [Google Scholar] [CrossRef]
- Virta, R.L. Mineral Commodity Profiles: Asbestos; USGS Circular 1255-KK; US Geological Survey (USGS): Reston, Virginia, USA, 2005; p. 56.
- Hawthorne, F.C.; Oberti, R. Amphiboles: Crystal Chemistry. Rev. Mineral. Geochem. 2007, 67, 1–54. [Google Scholar] [CrossRef]
- Roth, P. Minerals First Discovered in Switzerland and Minerals Named after Swiss Individual; Excalibur Mineral Corp.: Charlottesville, VA, USA, 2007. [Google Scholar]
- Leake, B.E.; Woolley, A.R.; Arps, C.E.S.; Birch, W.D.; Gilbert, M.C.; Grice, J.D.; Hawthorne, F.C.; Kato, A.; Kisch, H.J.; Krivovichev, V.G.; et al. Report. Nomenclature of Amphiboles: Report of Amphiboles: Report of the subcommittee on Amphiboles of the International Mineralogical Association Commission on New Minerals and Mineral Names. Mineral. Mag. 1997, 61, 295–321. [Google Scholar] [CrossRef]
- Baumann, F.; Buck, B.J.; Metcalf, R.V.; McLaurin, B.T.; Merkler, D.J.; Carbone, M. The Presence of Asbestos in the Natural Environment is Likely Related to Mesothelioma in Young Individuals and Women from Southern Nevada. J. Thorac. Oncol. 2015, 10, 731–737. [Google Scholar] [CrossRef] [PubMed]
- Pasetto, R.; Bruni, B.; Bruno, C.; D’Antona, C.; De Nardo, P.; Di Maria, G.; Di Stefano, R.; Fiorentini, C.; Gianfagna, A.; Marconi, A.; et al. Problematiche sanitarie della fibre anfibolica di Biancavilla. Aspetti epidemiologici, clinici e sperimentali. Notiziario dell’Istituto Superiore di Sanità 2004, 17, 8–12. [Google Scholar]
- Massaro, T.; Fiore, S.; Dragonieri, S.; Grimaldi, M.C.; Pistillo, S.; Cauzzillo, G.; Musti, M. Pleura malignant mesothelioma among resident population in areas with natural occurring asbestos on Calabria-lucania border. In Proceedings of the GeoMed—4th International Conference on Medical Geology, Bari, Italy, 20–25 September 2011. [Google Scholar]
- Ripabelli, G.; Tamburro, M.; Di Tella, D.; Carrozza, F.; Sammarco, M.L. Asbestos Exposures, Mesothelioma Incidence and Mortality, and Awareness by General Practitioners in the Molise Region, Central Italy. J. Occup. Environ. Med. 2018, 60, e90–e97. [Google Scholar] [CrossRef] [PubMed]
- Baris, Y.I.; Sahin, A.A.; Ozesmi, M.; Kerse, I.; Ozen, E.; Kolacan, B. An outbreak of pleural mesothelioma and chronic fibrosing pleurisy in the village of Krain/Urgüp in Anatolia. Thorax 1978, 33, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Bloise, A.; Barrese, E.; Apollaro, C. Hydrothermal alteration of Ti-doped forsterite to chrysotile and characterization of the resulting chrysotile fibres. Neues Jahrb. Miner. Abh. 2009, 185, 297–304. [Google Scholar] [CrossRef]
- Bloise, A.; Belluso, E.; Barrese, E.; Miriello, D.; Apollaro, C. Synthesis of Fe-doped chrysotile and characterization of the resulting chrysotile fibers. Cryst. Res. Technol. 2009, 44, 590–596. [Google Scholar] [CrossRef]
- Bloise, A.; Belluso, E.; Fornero, E.; Rinaudo, C.; Barrese, E.; Capella, S. Influence of synthesis condition on growth of Ni-doped chrysotile. Microporous Mesoporous Mater. 2010, 132, 239–245. [Google Scholar] [CrossRef]
- Spadea, P. Calabria-Lucania ophiolites. B Geofis. Teori. Appl. 1994, 36, 271–281. [Google Scholar]
- Fubini, B.; Otero-Arean, C. Chemical aspects of the toxicity of invale mineral dusts. Chem. Soc. Rev. 1999, 28, 373–381. [Google Scholar] [CrossRef]
- Loreto, C.; Carnazza, M.L.; Cardile, V.; Libra, M.; Lombardo, L.; Malaponte, G.; Martinez, G.; Musumeci, G.; Papa, V.; Cocco, L. Mineral fiber-mediated activation of phosphoinositide-specific phospho-lipasec in human bronchoalveolar carcinoma-derived alveolar epithelial A549 cells. Int. J. Oncol. 2009, 34, 371–376. [Google Scholar] [PubMed]
- Pugnaloni, A.; Giantomassi, F.; Lucarini, G.; Capella, S.; Bloise, A.; Di Primio, R.; Belluso, E. Cytotoxicity induced by exposure to natural and synthetic tremolite asbestos: An in vitro pilot study. Acta Histochem. 2013, 115, 100–112. [Google Scholar] [CrossRef] [PubMed]
- Censi, P.; Tamburo, E.; Speziale, S.; Zuddas, P.; Randazzo, L.A.; Punturo, R.; Cuttitta, A.; Aricò, P. Yttrium and lanthanides in human lung fluids, probing the exposure to atmospheric fallout. J. Hazard. Mater. 2011, 186, 1103–1110. [Google Scholar] [CrossRef] [PubMed]
- Censi, P.; Zuddas, P.; Randazzo, L.A.; Tamburo, E.; Spezial, S.; Cuttitta, A.; Punturo, R.; Aricò, P.; Santagata, R. Source and nature of inhaled atmospheric dust from trace element analyses of human bronchial fluids. Environ. Sci. Technol. 2011, 45, 6262–6267. [Google Scholar] [CrossRef] [PubMed]
- Nordberg, G.F.; Nogawa, K.; Nordberg, M.; Friberg, L.T. Handbook on the Toxicology of Metals, 3rd ed.; Academic Press/Elsevier: London, UK, 2007; Volume 23, pp. 446–486. [Google Scholar]
- Langer, A.M.; Nolan, R.P.; Constantopoulos, S.H.; Moutsopoulos, H.M. Association of Metsovo lung and pleural mesothelioma with exposure to tremolite containing whitewash. Lancet 1987, 1, 965–967. [Google Scholar] [CrossRef]
- McConnochie, K.; Simonato, L.; Mavrides, P.; Christofides, P.; Pooley, F.D.; Wagner, J.C. Mesothelioma in Cyprus: The role of tremolite. Thorax 1987, 42, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Liu, X.; Mu, S.; Tsai, S.P.; Wen, C.P. Asbestos related diseases from environmental exposure to crocidolite in Da-yao, China I. Review of exposure and epidemiological data. Occup. Environ. Med. 2003, 60, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Verkounteren, J.R.; Wylie, A.G. The tremolite-actinolite-ferro–actinolite series: Systematic relationships among cell parameters, composition, optical properties, and habit, and evidence of discontinuities. Am. Miner. 2000, 85, 1239–1254. [Google Scholar] [CrossRef]
- Skogby, H.; Rossman, G.R. The intensity of amphibole OH bands in the Infrared absorption spectrum. Phys. Chem. Miner. 1991, 18, 64–68. [Google Scholar] [CrossRef]
- Spadea, P. Continental crust rocks associated with ophiolites in Lucanian Apennine (southern Italy). Ofioliti 1982, 7, 501–522. [Google Scholar]
- Mongelli, G.; Dinelli, E. The geochemistry of shales from the “Frido Unit”, Liguride complex, lucanian Apennines, Italy: Implications for provenance and tectonic setting. Ofioliti 2001, 26, 457–466. [Google Scholar]
- Laurita, S.; Prosser, G.; Rizzo, G.; Langone, A.; Tiepolo, M.; Laurita, A. Geochronological study of zircons from continental crust rocks in the Frido Unit (southern Apennines). Int. J. Earth Sci. 2014, 104, 179–203. [Google Scholar] [CrossRef]
- Rizzo, G.; Sansone, M.T.C.; Perri, F.; Laurita, S. Mineralogy and petrology of the metasedimentary rocks from the Frido Unit (southern Apennines, Italy). Period. Mineral. 2016, 85, 153–168. [Google Scholar]
- Knott, S.D. The Liguride Complex of southern Italy a Cretaceous to Paleogene accretionary wedge. Tectonophysics 1987, 142, 217–243. [Google Scholar] [CrossRef]
- Knott, S.D. Structure, kinematics and metamorphism in the Liguride Complex, Southern Apennine, Italy. J. Struct. Geol. 1994, 16, 1107–1120. [Google Scholar] [CrossRef]
- Monaco, C.; Tortorici, L.; Morten, L.; Critelli, S.; Tansi, C. Geologia del versante Nord-orientale del Massiccio del Pollino (Confine calabro lucano). Nota illustrativa sintetica alla scala 1:50.000. Boll. Soc. Geol. Ital. 1995, 114, 277–291. [Google Scholar]
- Monaco, C.; Tortorici, L. Tectonic role of ophiolite-bearing terranes in building of the Southern Apennines orogenic belt. Terra Nova 1995, 7, 153–160. [Google Scholar] [CrossRef]
- Rizzo, G.; Laurita, S.; Altenberger, U. The Timpa delle Murge ophiolitic gabbros, southern Apennines: Insights from petrology and geochemistry and consequences to the geodynamic setting. Period. Mineral. 2017, 86. [Google Scholar] [CrossRef]
- Sansone, M.T.C.; Prosser, G.; Rizzo, G.; Tartarotti, P. Spinel-peridotites of the Frido Unit ophiolites (southern ApenninesH Italy): Evidence for oceanic evolution. Period. Mineral. 2012, 81, 35–59. [Google Scholar]
- Sansone, M.T.C.; Tartarotti, P.; Prosser, G.; Rizzo, G. From ocean to subduction: The polyphase metamorphic evolution of the Frido Unit metadolerite dykes (Southern Apennine, Italy). J. Virtual Explor. 2012, 41. [Google Scholar] [CrossRef]
- Gueguen, E.; Doglioni, C.; Fernandez, M. On the post-25 Ma geodynamic evolution of the western Mediterranean. Tectonophysics 1998, 298, 259–269. [Google Scholar] [CrossRef]
- Evaus, B.W.; Yaug, H. Fe-Mg order-disorder in tremolite-actinoliteferro-actinolite at ambient and high temperature. Am. Mineral. 1998, 83, 458–475. [Google Scholar]
- Patacca, E.; Scandone, P. Geology of the southern Apennines. In Results of the CROP Project, Sub-Project CROP-04 Southern Apennines (Italy); Mazzotti, A., Patacca, E., Scandone, P., Eds.; Società Geologica Italiana: Bologna, Italy, 2007; pp. 75–119. [Google Scholar]
- Sansone, M.T.C.; Rizzo, G. Pumpellyite veins in the metadolerite of the Frido Unit (southern Apennines-Italy). Period. Mineral. 2012, 81, 75–92. [Google Scholar]
- Dichicco, M.C.; De Bonis, A.; Mongelli, G.; Rizzo, G.; Sinisi, R. Naturally occurring asbestos in the southern Apennines: Quick μ-Raman Spectroscopy identification as a tool of environmental control. In Proceedings of the Book of Abstracts of the 13th International Conference on Protection and Restoration of the Environment, Mykonos Island, Greece, 3–8 July 2016. [Google Scholar]
- Dichicco, M.C.; Laurita, S.; Paternoster, M.; Rizzo, G.; Sinisi, R.; Mongelli, G. Serpentinite Carbonation for CO2 Sequestration in the Southern Apennines: Preliminary Study. Energy Procedia 2015, 76, 477–486. [Google Scholar] [CrossRef]
- Dichicco, M.C.; De Bonis, A.; Mongelli, G.; Rizzo, G.; Sinisi, R. μ-Raman spectroscopy and X-ray diffraction of asbestos’ minerals for geo-environmental monitoring: The case of the southern Apennines natural sources. Appl. Clay Sci. 2017, 141, 292–299. [Google Scholar] [CrossRef]
- Bersani, D.; Andò, S.; Scrocco, L.; Gentile, P.; Salvioli-Mariani, E.; Lottici, P.P. Study of composition of amphiboles in the tremolite—Ferro-actinolite series by micro-Raman and SEM-EDXS. In Proceedings of the 11th International Geo Raman Conference, Louis, MI, USA, 15–19 June 2014; Volume 1783. [Google Scholar]
- Siivola, J.; Schmid, R. List of Mineral Abbrevations—Recommendations by the IUGS Subcommission on the Systematics of Metamorphic Rocks. Web Version 01.02.2007. Available online: https://www.bgs.ac.uk/scmr/products.html (accessed on 8 March 2018).
- Chen, T.H.; Calligaro, T.; Pagès-Camagna, S.; Menu, M. Investigation of Chinese archaic jade by PIXE and μ Raman spectrometry. Appl. Phys. A 2004, 79, 177–180. [Google Scholar] [CrossRef]
- Ballirano, P.; Andreozzi, G.B.; Belardi, G. Crystal chemical and structural characterization of fibrous tremolite from Susa Valley, Italy, with comments on potential harmful effects on human health. Am. Mineral. 2008, 93, 1349–1355. [Google Scholar] [CrossRef]
- Shukla, A.; Gulumian, M.; Hei, T.K.; Kamp, D.; Rahman, Q.; Mossman, B.T. Multiple roles of oxidants in the pathogenesis of asbestos induced diseases. Free Radic. Biol. Med. 2003, 34, 1117–1129. [Google Scholar] [CrossRef]
- Raudsepp, M.; Turnock, A.C.; Hawthorne, F.C.; Sherriff, B.K.; Hartmau, J.S. Characterization of synthetic pargasitic amphiboles (NaCa2Mg4M3+Si6Al2,O22(OH,F)2; M3+=Al, Cr, Ga, Sc, In) by infrared spectroscopy, Rietveld structurerefmement, and 27AI,29Si, and 19p: MASNMR spectroscopy. Am. Mineral. 1987, 72, 580–593. [Google Scholar]
- Pacella, A.; Ballirano, P. Chemical and structural characterization of fibrous richterite with high environmental and health relevance from Libby, Montana (USA). Period. Mineral. 2016, 85, 169–177. [Google Scholar]
- Barlow, C.A.; Barrett, T.F.; Shukla, A.; Mossman, B.T.; Lounsbury, K.M. Asbestos-mediated CREB phosphorylation is regulated by protein kinase A and extracellular signal-regulated kinases ½. Am. J. Physiol. Lung Cell. Mol. Physiol. 2007, 292, L1361–L1369. [Google Scholar] [CrossRef] [PubMed]
- Shukla, A.; Jung, M.; Stern, M.; Fukagawa, N.K.; Taatjes, D.J.; Sawyer, D.; Van Houten, B.; Mossman, B.T. Asbestos induces mitochondrial DNA damage and dysfunction linked to the development of apoptosis. Am. J. Physiol. Lung Cell. Mol. Physiol. 2003, 285, L1018–L1025. [Google Scholar] [CrossRef] [PubMed]
- Stanton, M.F.; Layard, M.; Tegeris, A.; Miller, E.; May, M.; Morgan, E.; Smith, A. Relation of particle dimension to carcinogenicity in amphibole asbestoses and other fibrous minerals. J. Natl. Cancer Inst. 1981, 67, 965–975. [Google Scholar] [PubMed]
- Berman, D.W.; Crump, K.S.; Chatfield, E.J.; Davis, J.M.G.; Jones, A.D. The sizes, shapes, and mineralogy of asbestos structures that induce lung tumors or mesothelioma in AF/HAN rats following inhalation. Risk Anal. 1995, 15, 181–195. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, D.; Castranova, V.; Donaldson, K.; Fubini, B.; Hadley, J.; Hesterberg, T.; Kane, A.; Lai, D.; McConnell, E.E.; Muhle, H.; et al. Testing of fibrous particles: Short-term assays and strategies. Inhal. Toxicol. 2005, 17, 497–537. [Google Scholar] [CrossRef] [PubMed]
- Dodson, R.F.; Atkinson, M.A.; Levin, J.L. Asbestos fiber length as related to potential pathogenicity: A critical review. Am. J. Ind. Med. 2003, 44, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Loomis, D.; Dement, J.; Richardson, D.; Wolf, S. Asbestos fibre dimensions and lung cancer mortality among workers exposed to chrysotile. Occup. Environ. Med. 2010, 67, 580–584. [Google Scholar] [CrossRef] [PubMed]
- Fantauzzi, M.; Pacella, A.; Atzei, D.; Gianfagna, A.; Andreozzi, G.B.; Rossi, A. Combined use of X-ray photoelectron and Mössbauer spectroscopic techniques in the analytical characterization of iron oxidation state in amphibole asbestos. Anal. Bioanal. Chem. 2010, 396, 2889–2898. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, R.K.; Lohani, M.; Pant, A.B.; Rahman, Q. Cyto-genotoxicity of amphibole asbestos fibers in cultured human lung epithelial cell line: Role of surface iron. Toxicol. Ind. Health 2010, 26, 575–582. [Google Scholar] [CrossRef] [PubMed]
| Samples | GPS Coordinates | Field Characteristic | Texture | Mineral Assemblages |
|---|---|---|---|---|
| S1 | 40°01′40.02″ N–16°08′07.9″ E | Cataclastic serpentinite | Pseudomorphic and vein | Srp–Spl–Chl–relics Ol–relics Cpx and Opx–Op–Am–Prh * |
| S4 | 40° 01′39.2″ N–16°08′09.6″ E | Cataclastic serpentinite | Pseudomorphic and vein | Srp–Spl–Chl–relics Ol–relics Opx and Cpx–Am |
| S5 | 40°01′39.02″ N–16°08′09.6″ E | Cataclastic serpentinite cut by carbonate veins | Pseudomorphic and brecciated-vein | Srp–Chl–relics Cpx and Opx–Am–Cal–Op |
| S6.2 | 40°01′38.3″ N–16°08′10.4″ E | Cataclastic serpentinite | Pseudomorphic | Srp–Spl–relics Ol–relics Cpx–Am |
| S7 | 40°01′37.6″ N–16° 08′10.7″ E | Cataclastic serpentinite | Pseudomorphic | Srp–Spl–relics Ol–relics Cpx–Am |
| S27 | 40°01′39.2″ N–16°0.8′25.5″ E | Cataclastic serpentinite | Pseudomorphic | Srp–Spl–relics Ol–relics Cpx–Am |
| S30 | 40°01′45.3″ N–16°0.8′27.3″ E | Cataclastic serpentinite | Pseudomorphic | Srp–Spl–Chl–relics Ol–relics Opx and Cpx–Am |
| Oxides | wt % | Range |
|---|---|---|
| SiO2 | 52.25 | 45.13–59.36 |
| TiO2 | 1.25 | 0.50–2.00 |
| Al2O3 | 8.65 | 1.84–15.46 |
| Cr2O3 | 0.77 | −1.41 |
| MgO | 20.81 | 14.97–26.66 |
| CaO | 16.18 | 7.97–24.39 |
| MnO | 0.10 | 0.00–0.21 |
| FeOtot | 3.76 | 2.47–5.06 |
| Fe2O3 | n.d. | n.d. |
| Na2O | 0.98 | 0.05–1.91 |
| K2O | 0.60 | 0.56–0.64 |
| Oxides | (wt %) | Rim (wt %) | Core (wt %) | Apfu |
|---|---|---|---|---|
| SiO2 | 56.87 | 57.89 | 57.66 | 8.09 |
| Al2O3 | 3.47 | 5.05 | 3.86 | 0.28 |
| Cr2O3 | 0.23 | n.d. | n.d. | 0.05 |
| MgO | 25.56 | 24.89 | 21.77 | 5.14 |
| CaO | 15.11 | 12.69 | 14.54 | 1.85 |
| Fe2O3 | 2.93 | n.d. | n.d. | n.d. |
| Na2O | 1.50 | 3.21 | 2.83 | 0.30 |
| K2O | n.d. | 2.15 | 0.39 | 0.33 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dichicco, M.C.; Laurita, S.; Sinisi, R.; Battiloro, R.; Rizzo, G. Environmental and Health: The Importance of Tremolite Occurence in the Pollino Geopark (Southern Italy). Geosciences 2018, 8, 98. https://doi.org/10.3390/geosciences8030098
Dichicco MC, Laurita S, Sinisi R, Battiloro R, Rizzo G. Environmental and Health: The Importance of Tremolite Occurence in the Pollino Geopark (Southern Italy). Geosciences. 2018; 8(3):98. https://doi.org/10.3390/geosciences8030098
Chicago/Turabian StyleDichicco, Maria Carmela, Salvatore Laurita, Rosa Sinisi, Raimondo Battiloro, and Giovanna Rizzo. 2018. "Environmental and Health: The Importance of Tremolite Occurence in the Pollino Geopark (Southern Italy)" Geosciences 8, no. 3: 98. https://doi.org/10.3390/geosciences8030098
APA StyleDichicco, M. C., Laurita, S., Sinisi, R., Battiloro, R., & Rizzo, G. (2018). Environmental and Health: The Importance of Tremolite Occurence in the Pollino Geopark (Southern Italy). Geosciences, 8(3), 98. https://doi.org/10.3390/geosciences8030098





