Prediction of Holocene Mercury Accumulation Trends by Combining Palynological and Geochemical Records of Lake Sediments (Black Forest, Germany)
Abstract
1. Introduction
2. Materials and Methods
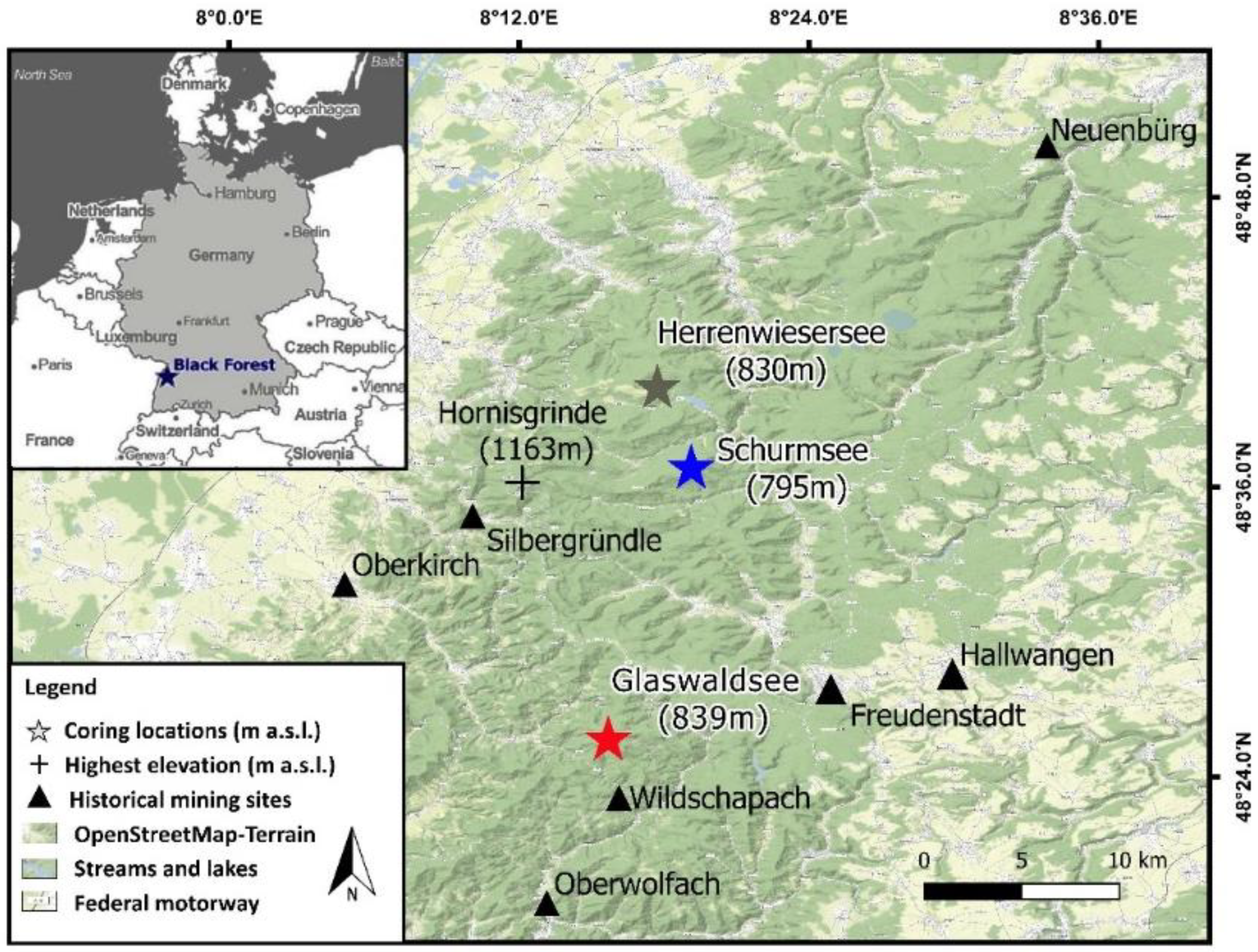
2.1. Site Description
2.1.1. Coring
2.1.2. Chronology
2.2. Chemical Analysis
2.2.1. Carbon, Nitrogen and Sulphur
2.2.2. Major and Trace Elements
2.2.3. Mercury
2.3. Sedimentation Rates
2.4. Statistical Treatment
3. Results
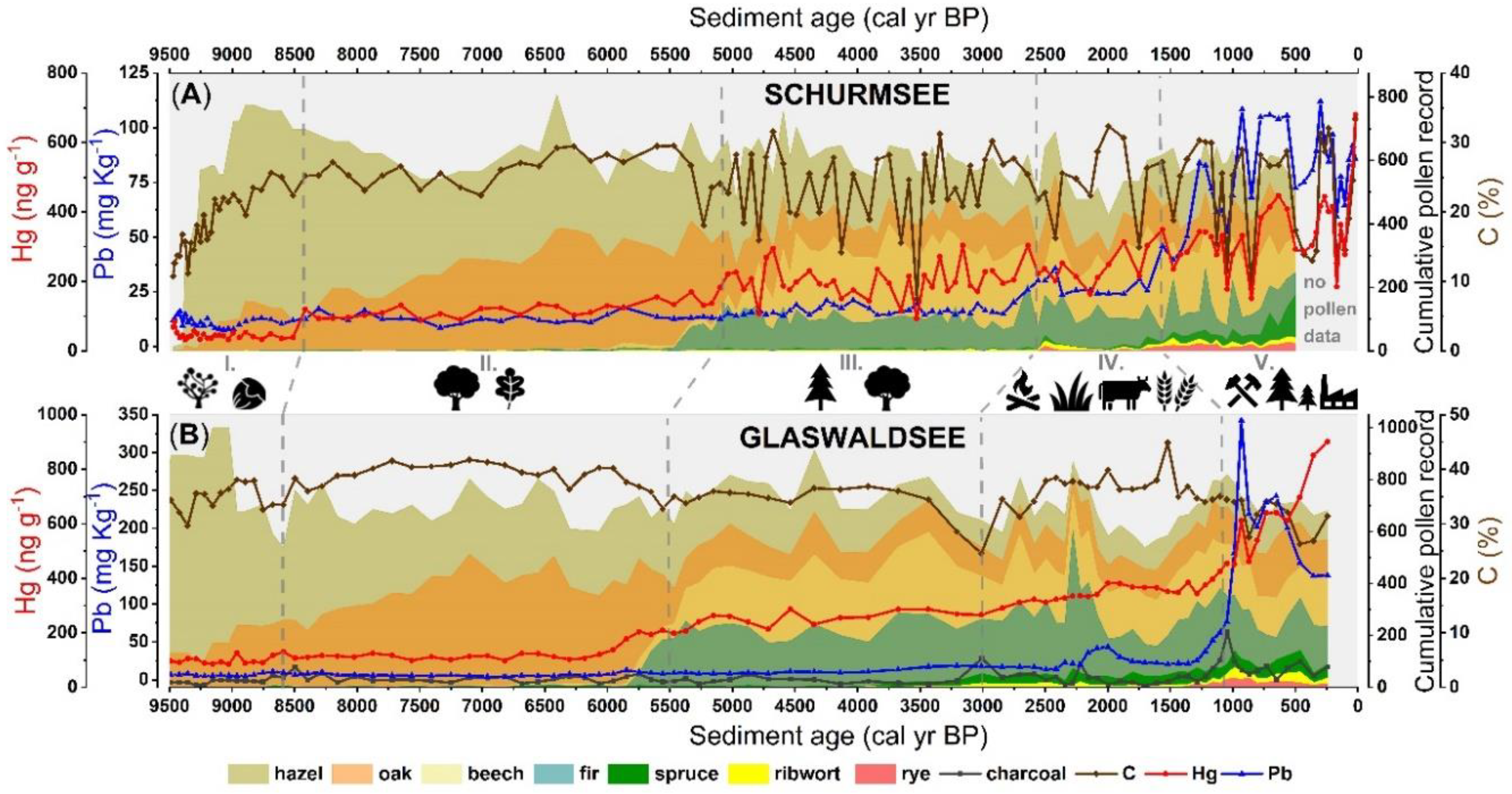
3.1. Vegetation Changes and Land Use Practices
3.2. Holocene Records of Mercury and Lead Concentrations
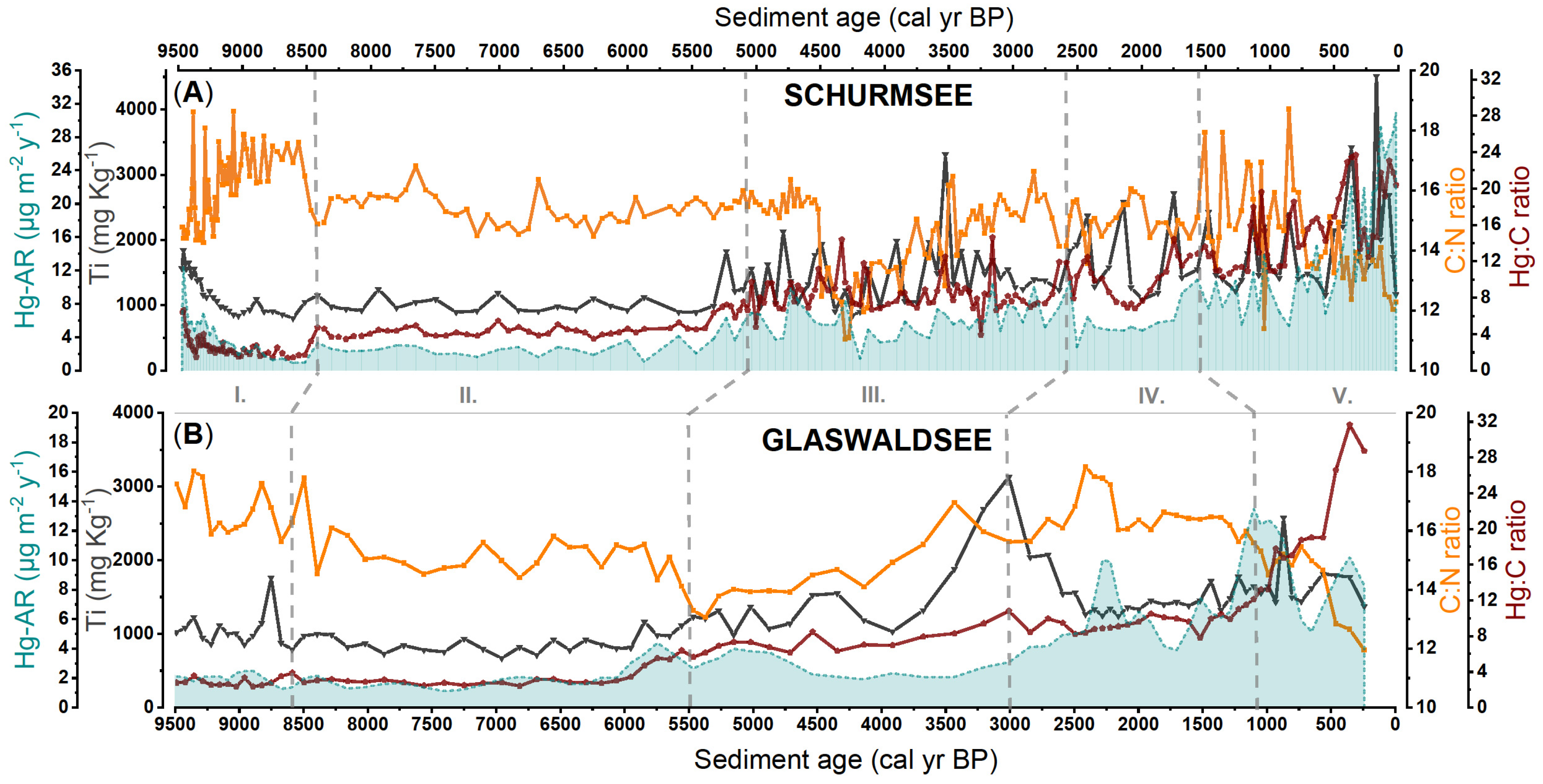
3.3. Variation in Sediment Geochemistry Revealed from Principal Component Analysis
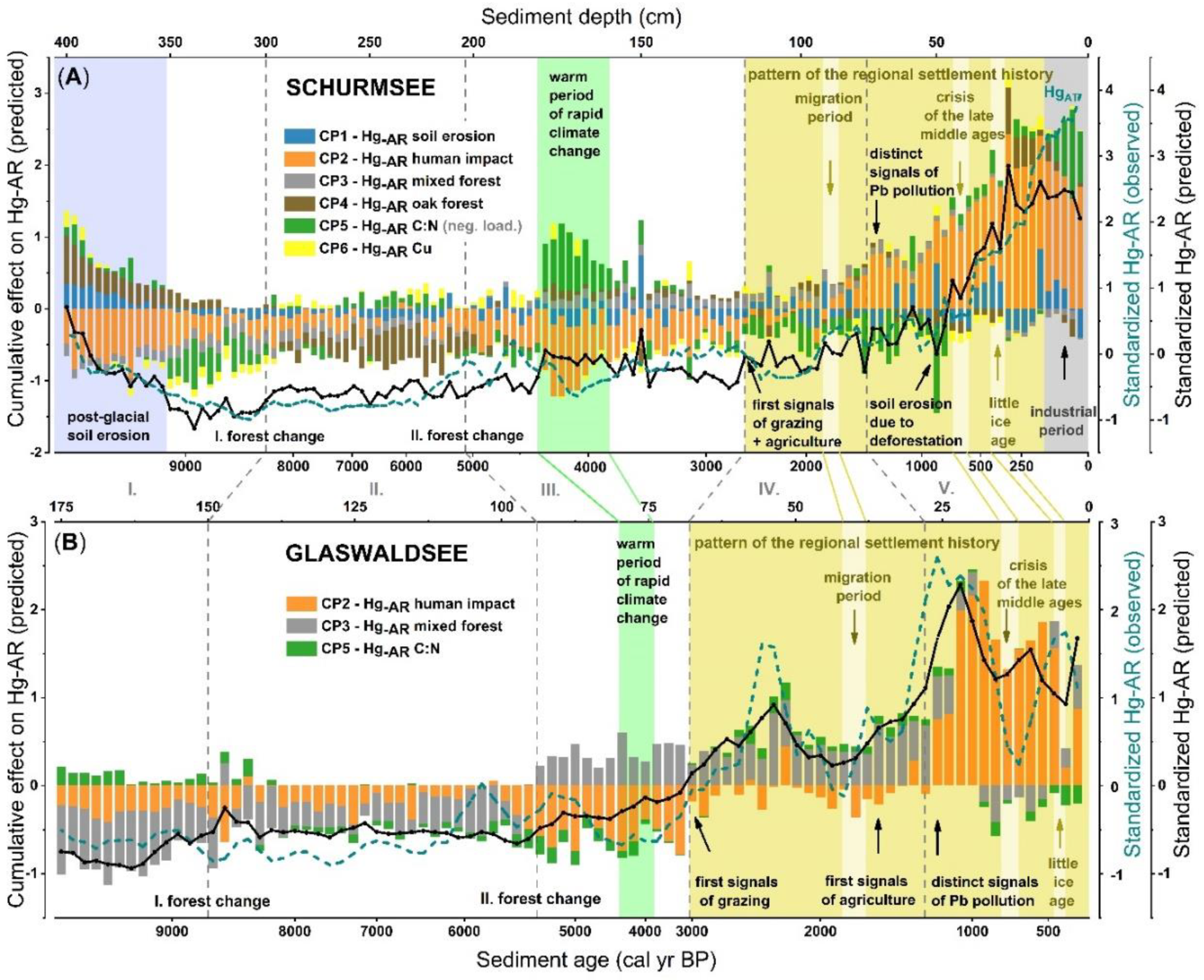
3.4. Prediction of Factors Controlling Mercury Accumulation Using Stepwise Regression Analysis
4. Discussion
4.1. Evolution of Mercury Concentration and Accumulation
4.2. Environmental Processes Controlling Sediment Geochemistry
4.3. Chronology of Factors Controlling Mercury Accumulation
4.3.1. Declining Soil Erosion in an Establishing Hazel-Dominated Shrubland (Section I)
4.3.2. Steady Fluxes of Soil Organic Matter in an Oak-Dominated Forest (Section II)
4.3.3. Warm Period Caused Enhanced Mercury Uptake in Lakes Surrounded by a Mixed Forest (Section III)
4.3.4. Distinct Traces of Grazing and Agriculture in a Mixed Forest (Section IV)
4.3.5. Pollution from Mining and Industrial Activities in a Mixed Forest (Section V)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sheehan, K.D.; Fernandez, I.J.; Kahl, J.S.; Amirbahman, A. Litterfall Mercury in Two Forested Watersheds at Acadia National Park, Maine, USA. Water Air Soil Pollut. 2006, 170, 249–265. [Google Scholar] [CrossRef]
- Xia, K.; Skyllberg, U.L.; Bleam, W.F.; Bloom, P.R.; Nater, E.A.; Helmke, P.A. X-ray absorption spectroscopic evidence for the complexation of HG(II) by reduced sulfur in soil humic substances. Sci. Technol. 1999, 33, 257–261. [Google Scholar] [CrossRef]
- Biester, H.; Bindler, R.; Martinez Cortizas, A.; Engstrom, D.R. Modeling the past atmospheric deposition of mercury using natural archives. Environ. Sci. Technol. 2007, 41, 4851–4860. [Google Scholar] [CrossRef] [PubMed]
- Swain, E.B.; Engstrom, D.R.; Brigham, M.E.; Henning, T.A.; Brezonik, P.L. Increasing Rates of Atmospheric Mercury Deposition in Midcontinental North America. Science 1992, 257, 784–787. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Engstrom, D.R.; Rose, N.L. Recent changes in atmospheric mercury deposition recorded in the sediments of remote equatorial lakes in the Rwenzori Mountains, Uganda. Environ. Sci. Technol. 2010, 44, 6570–6575. [Google Scholar] [CrossRef] [PubMed]
- Rydberg, J.; Klaminder, J.; Rosén, P.; Bindler, R. Climate driven release of carbon and mercury from permafrost mires increases mercury loading to sub-arctic lakes. Sci. Total Environ. 2010, 408, 4778–4783. [Google Scholar] [CrossRef] [PubMed]
- Ravichandran, M. Interactions between mercury and dissolved organic matter—A review. Chemosphere 2004, 55, 319–331. [Google Scholar] [CrossRef] [PubMed]
- Hart, B.T. Uptake of trace metals by sediments and suspended particulates: A review. In Sediment/Freshwater Interaction; Springer: Dordrecht, The Netherlands, 1982; pp. 299–313. [Google Scholar]
- Brännvall, M.-L.; Bindler, R.; Emteryd, O.; Nilsson, M.; Renberg, I. Stable isotope and concentration records of atmospheric lead pollution in peat and lake sediments in Sweden. Water Air Soil Pollut. 1997, 100, 243–252. [Google Scholar] [CrossRef]
- Engstrom, D.R. Lake Development in the Boreal Peatlands of Southeastern Labrador, Canada. Arct. Alp. Res. 1984, 16, 447–452. [Google Scholar] [CrossRef]
- Hermanns, Y.M.; Biester, H. A 17,300-year record of mercury accumulation in a pristine lake in southern Chile. J. Paleolimnol. 2013, 49, 547–561. [Google Scholar] [CrossRef]
- Guevara, S.R.; Meili, M.; Rizzo, A.; Daga, R.; Arribere, M. Sediment records of highly variable mercury inputs to mountain lakes in Patagonia during the past millennium. Atmos. Chem. Phys. 2010, 10, 3443–3453. [Google Scholar] [CrossRef]
- Bindler, R.; Olofsson, C.; Renberg, I.; Frech, W. Temporal Trends in Mercury Accumulation in Lake Sediments in Sweden. Water Air Soil Pollut. 2001, 1, 343–355. [Google Scholar] [CrossRef]
- Koinig, K.A.; Shotyk, W.; Lotter, A.F.; Ohlendorf, C.; Sturm, M. 9000 years of geochemical evolution of lithogenic major and trace elements in the sediment of an alpine lake—The role of climate, vegetation, and land-use history. J. Paleolimnol. 2003, 30, 307–320. [Google Scholar] [CrossRef]
- Armstrong, A.; Holden, J.; Luxton, K.; Quinton, J.N. Multi-scale relationship between peatland vegetation type and dissolved organic carbon concentration. Ecol. Eng. 2012, 47, 182–188. [Google Scholar] [CrossRef]
- Smolander, A.; Loponen, J.; Suominen, K.; Kitunen, V. Organic matter characteristics and C and N transformations in the humus layer under two tree species, Betula pendula and Picea abies. Soil Biol. Biochem. 2005, 37, 1309–1318. [Google Scholar] [CrossRef]
- Hansson, K.; Olsson, B.A.; Olsson, M.; Johansson, U.; Kleja, D.B. Differences in soil properties in adjacent stands of Scots pine, Norway spruce and silver birch in SW Sweden. For. Ecol. Manag. 2011, 262, 522–530. [Google Scholar] [CrossRef]
- Berg, B. Litter decomposition and organic matter turnover in northern forest soils. For. Ecol. Manag. 2000, 133, 13–22. [Google Scholar] [CrossRef]
- Koorem, K.; Moora, M. Positive association between understory species richness and a dominant shrub species (Corylus avellana) in a boreonemoral spruce forest. For. Ecol. Manag. 2010, 260, 1407–1413. [Google Scholar] [CrossRef]
- Van Nevel, L.; Mertens, J.; De Schrijver, A.; Baeten, L.; De Neve, S.; Tack, F.M.G.; Meers, E.; Verheyen, K. Forest floor leachate fluxes under six different tree species on a metal contaminated site. Sci. Total Environ. 2013, 447, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Obrist, D.; Johnson, D.W.; Edmonds, R.L. Effects of vegetation type on mercury concentrations and pools in two adjacent coniferous and deciduous forests. J. Plant Nutr. Soil Sci. 2012, 175, 68–77. [Google Scholar] [CrossRef]
- Schwesig, D.; Matzner, E. Pools and fluxes of mercury and methylmercury in two forested catchments in Germany. Sci. Total Environ. 2000, 260, 213–223. [Google Scholar] [CrossRef]
- Demers, J.D.; Driscoll, C.T.; Fahey, T.J.; Yavitt, J.B. Mercury cycling in litter and soil in different forest types in the Adirondack region, New York, USA. Ecol. Appl. 2007, 17, 1341–1351. [Google Scholar] [CrossRef] [PubMed]
- Navrátil, T.; Shanley, J.B.; Rohovec, J.; Oulehle, F.; Šimeček, M.; Houška, J.; Cudlín, P. Soil mercury distribution in adjacent coniferous and deciduous stands highly impacted by acid rain in the Ore Mountains, Czech Republic. Appl. Geochem. 2016, 75, 63–75. [Google Scholar] [CrossRef]
- Jiskra, M.; Wiederhold, J.G.; Skyllberg, U.; Kronberg, R.-M.; Hajdas, I.; Kretzschmar, R. Mercury Deposition and Re-emission Pathways in Boreal Forest Soils Investigated with Hg Isotope Signatures. Environ. Sci. Technol. 2015, 49, 7188–7196. [Google Scholar] [CrossRef] [PubMed]
- Navrátil, T.; Shanley, J.; Rohovec, J.; Hojdová, M.; Penížek, V.; Buchtová, J. Distribution and Pools of Mercury in Czech Forest Soils. Water Air Soil Pollut. 2014, 225, 1829. [Google Scholar] [CrossRef]
- Drenner, R.W.; Chumchal, M.M.; Jones, C.M.; Lehmann, C.M.B.; Gay, D.A.; Donato, D.I. Effects of Mercury Deposition and Coniferous Forests on the Mercury Contamination of Fish in the South Central United States. Environ. Sci. Technol. 2013, 47, 1274–1279. [Google Scholar] [CrossRef] [PubMed]
- Hare, A.A.; Stern, G.A.; Kuzyk, Z.Z.A.; MacDonald, R.W.; Johannessen, S.C.; Wang, F. Natural and anthropogenic mercury distribution in marine sediments from Hudson Bay, Canada. Environ. Sci. Technol. 2010, 44, 5805–5811. [Google Scholar] [CrossRef] [PubMed]
- Renberg, I.; Persson, M.W.; Emteryd, O. Pre-industrial atmospheric lead contamination detected in Swedish lake sediments. Nature 1994, 368, 323–326. [Google Scholar] [CrossRef]
- Rose, N.; Yang, H.; Turner, S.D.; Simpson, G. An assessment of the mechanisms for the transfer of lead and mercury from atmospherically contaminated organic soils to lake sediments with particular reference to Scotland, UK. Geochim. Cosmochim. Acta 2012, 82, 113–135. [Google Scholar] [CrossRef]
- Rydberg, J.; Karlsson, J.; Nyman, R.; Wanhatalo, I.; Näthe, K.; Bindler, R. Importance of vegetation type for mercury sequestration in the northern Swedish mire, Rödmossamyran. Geochim. Cosmochim. Acta 2010, 74, 7116–7126. [Google Scholar] [CrossRef]
- Pompeani, D.P.; Cooke, C.A.; Abbott, M.B.; Drevnick, P.E. Climate, Fire, and Vegetation Mediate Mercury Delivery to Midlatitude Lakes over the Holocene. Environ. Sci. Technol. 2018, 52, 8157–8164. [Google Scholar] [CrossRef] [PubMed]
- Rydberg, J.; Rösch, M.; Heinz, E.; Biester, H. Influence of catchment vegetation on mercury accumulation in lake sediments from a long-term perspective. Sci. Total Environ. 2015, 538, 896–904. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Cortizas, A.; Pontevedra-Pombal, X.; García-Rodeja, E.; Nóvoa-Muñoz, J.C.; Shotyk, W. Mercury in a spanish peat bog: Archive of climate change and atmospheric metal deposition. Science 1999, 284, 939–942. [Google Scholar] [CrossRef] [PubMed]
- Thevenon, F.; Guédron, S.; Chiaradia, M.; Loizeau, J.-L.; Poté, J. (Pre-)historic changes in natural and anthropogenic heavy metals deposition inferred from two contrasting Swiss Alpine lakes. Quat. Sci. Rev. 2011, 30, 224–233. [Google Scholar] [CrossRef]
- Geological Survey of Baden-Württemberg LGRB-Kartenviewer. Available online: http://maps.lgrb-bw.de/ (accessed on 6 March 2018).
- German Meteorological Service Wetter und Klima—Deutscher Wetterdienst—Fachinformationsdienst Meteorologie. Available online: https://www.dwd.de/DE/derdwd/bibliothek/fachinfodienst/recherchetipps.htm (accessed on 5 March 2018).
- Hinderer, M.; Jüttner, I.; Winkler, R.; Steinberg, C.E.; Kettrup, A. Comparing trends in lake acidification using hydrochemical modelling and paleolimnology: The case of the Herrenwieser See, Black Forest, Germany. Sci. Total Environ. 1998, 218, 113–121. [Google Scholar] [CrossRef]
- Berglund, B.E.; Birks, H.J.B.; Ralska-Jasiewiczowa, M.; Wright, H.E. Palaeoecological Events During the Last 15,000 Years: Regional Synthesis of Palaeoecological Studies of Lakes and Mires in Europe; John Wiley & Sons: Chichester, UK, 1996. [Google Scholar]
- Rösch, M. Botanical evidence for prehistoric and medieval land use in Black Forest. Medieval Rural Settlement in Marginal Landscapes. Ruralia 2007, VII, 335–343. [Google Scholar]
- Livingstone, D.A. A Lightweight Piston Sampler for Lake Deposits. Ecology 1955, 36, 137–139. [Google Scholar] [CrossRef]
- Rösch, M. Nationalpark–Natur–Weißtanne–Fichte. Sechs Jahrtausende Wald und Mensch im Nordschwarzwald. Denkmalpfl. Baden-Württ. Landesdenkmalpflege 2015, 44, 154–159. [Google Scholar]
- Rösch, M.; Lechterbeck, J. Seven Millennia of human impact as reflected in a high resolution pollen profile from the profundal sediments of Litzelsee, Lake Constance region, Germany. Veg. Hist. Archaeobotany 2016, 25, 339–358. [Google Scholar] [CrossRef]
- Reimer, P.J.; Bard, E.; Bayliss, A.; Beck, J.W.; Blackwell, P.G.; Ramsey, C.B.; Buck, C.E.; Cheng, H.; Edwards, R.L.; van der Plicht, J.; et al. IntCal13 and Marine13 Radiocarbon Age Calibration Curves 0–50,000 Years cal BP. Radiocarbon 2013, 55, 1869–1887. [Google Scholar] [CrossRef]
- Blaauw, M. Methods and code for classical age-modelling of radiocarbon sequences. Quat. Geochronol. 2010, 5, 512–518. [Google Scholar] [CrossRef]
- Meyers, P.A.; Ishiwatari, R. Lacustrine organic geochemistry—An overview of indicators of organic matter sources and diagenesis in lake sediments. Org. Geochem. 1993, 20, 867–900. [Google Scholar] [CrossRef]
- Müller, G.; Gastner, M. The Karbonat-Bombe, a simple device for the determination of carbonate content in sediments, soils and other materials. Neues Jahrbuch für Mineralogie-Monatshefte 1971, 10, 466–469. [Google Scholar]
- Cheburkin, A.K.; Shotyk, W. An energy-dispersive miniprobe multielement analyzer (EMMA) for direct analysis of Pb and other trace elements in peats. Fresenius J. Anal. Chem. 1996, 354, 688–691. [Google Scholar] [CrossRef] [PubMed]
- US EPA. US EPA Method 7473 US EPA Method 7473 (SW-846): Mercury in Solids and Solutions by Thermal Decomposition, Amalgamation, and Atomic Absorption Spectrophotometry; US EPA: Washington, DC, USA, 1998.
- Eriksson, L.; Johansson, E.; Kettaneh-Wold, N.W.S. Introduction to Multi and Megavariate Data Analysis Using Projection Methods (PCS & PLS); Umetrics: San Jose, CA, USA, 1999. [Google Scholar]
- Pérez-Rodríguez, M.; Horák-Terra, I.; Rodríguez-Lado, L.; Aboal, J.R.; Martínez Cortizas, A. Long-Term (∼57 ka) Controls on Mercury Accumulation in the Souther Hemisphere Reconstructed Using a Peat Record from Pinheiro Mire (Minas Gerais, Brazil). Environ. Sci. Technol. 2015, 49, 1356–1364. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation: Vienna, Austria, 2016. [Google Scholar]
- Rösch, M. Abies alba and Homo sapiens in the Schwarzwald–A Difficult Story. Interdiscip. Archaeol. 2015, 6, 47–62. [Google Scholar] [CrossRef]
- Munthe, J.; Hultberg, H.; Lee, Y.-H.; Parkman, H.; Iverfeldt, A.; Renberg, I. Trends of mercury and methylmercury in deposition, run-off water and sediments in relation to experimental manipulations and acidification. Water Air Soil Pollut. 1995, 85, 743–748. [Google Scholar] [CrossRef]
- Yang, H.; Rose, N.L. Distribution of mercury in six lake sediment cores across the UK. Sci. Total Environ. 2003, 304, 391–404. [Google Scholar] [CrossRef]
- Corella, J.P.; Saiz-Lopez, A.; Sierra, M.J.; Mata, M.P.; Millán, R.; Morellón, M.; Cuevas, C.A.; Moreno, A.; Valero-Garcés, B.L. Trace metal enrichment during the Industrial Period recorded across an altitudinal transect in the Southern Central Pyrenees. Sci. Total Environ. 2018, 645, 761–772. [Google Scholar] [CrossRef] [PubMed]
- Cannon, W.F.; Dean, W.E.; Bullock, J.H. Effects of Holocene climate change on mercury deposition in Elk Lake, Minnesota: The importance of eolian transport in the mercury cycle. Geology 2003, 31, 187–190. [Google Scholar] [CrossRef]
- Hermanns, Y.M.; Biester, H. Anthropogenic mercury signals in lake sediments from southernmost Patagonia, Chile. Sci. Total Environ. 2013, 445–446, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Vandal, G.M.; Fitzgerald, W.F.; Boutron, C.F.; Candelone, J.-P. Variations in mercury deposition to Antarctica over the past 34,000 years. Nature 1993, 362, 621–623. [Google Scholar] [CrossRef]
- Fitzgerald, W.F.; Engstrom, D.R.; Lamborg, C.H.; Tseng, C.-M.; Balcom, P.H.; Hammerschmidt, C.R. Modern and Historic Atmospheric Mercury Fluxes in Northern Alaska: Global Sources and Arctic Depletion. Environ. Sci. Technol. 2005, 39, 557–568. [Google Scholar] [CrossRef] [PubMed]
- Driscoll, C.T.; Blette, V.; Yan, C.; Schofield, C.L.; Munson, R.; Holsapple, J. The role of dissolved organic carbon in the chemistry and bioavailability of mercury in remote Adirondack lakes. Water Air Soil Pollut. 1995, 80, 499–508. [Google Scholar] [CrossRef]
- Kolka, R.K.; Nater, E.A.; Grigal, D.F.; Verry, E.S. Atmospheric Inputs of Mercury and Organic Carbon into a Forested Upland/Bog Watershed. Water Air. Soil Pollut. 1999, 113, 273–294. [Google Scholar] [CrossRef]
- Ouellet, J.-F.; Lucotte, M.; Teisserenc, R.; Paquet, S.; Canuel, R. Lignin biomarker as tracers of mercury in lakes water column. Biogeochemistry 2009, 94, 123–140. [Google Scholar] [CrossRef]
- Teisserenc, R.; Lucotte, M.; Houel, S.; Carreau, J. Integrated transfers of terrigenous organic matter to lakes at their watershed level: A combined biomarker and GIS analysis. Geochim. Cosmochim. Acta 2010, 74, 6375–6386. [Google Scholar] [CrossRef]
- Rydberg, J.; Rosén, P.; Lambertsson, L.; De Vleeschouwer, F.; Tomasdotter, S.; Bindler, R. Assessment of the spatial distributions of total- and methyl-mercury and their relationship to sediment geochemistry from a whole-lake perspective. J. Geophys. Res. Biogeosci. 2012, 117, 1–13. [Google Scholar] [CrossRef]
- Blais, J.M.; Kalff, J.; Cornett, R.J.; Evans, R.D. Evaluation of U210Pb dating in lake sediments using stable Pb, Ambrosia pollen, and 137Cs. J. Paleolimnol. 1995, 13, 169–178. [Google Scholar]
- MSC-E. Data of HMs and POPs for the EMEP Region; Meteorological Synthesizing Centre-East: Moscow, Russian, 2017. [Google Scholar]
- Kainz, M.; Lucotte, M. Mercury Concentrations in Lake Sediments—Revisiting the Predictive Power of Catchment Morphometry and Organic Matter Composition. Water Air Soil Pollut. 2006, 170, 173–189. [Google Scholar] [CrossRef]
- Grassmann, G.; Wieland, G.; Rösch, M. Das Neuenbürger Erzevier im Nordschwarzwald als Wirtschaftsraum während der Späthallstatt- und dem Frühlatènezeit. Ger. Anz. Röm.-Ger. Kom. Dtsch. Archäol. Inst. 2006, 84, 273–306. [Google Scholar]
- Rösch, M.; Tserendorj, G. Florengeschichtliche Beobachtungen im Nordschwarzwald (Südwestdeutschland). Hercynia-Ökol. Umw. Mitteleur. 2011, 44, 53–71. [Google Scholar]
- Corella, J.P.; Valero-Garcés, B.L.; Wang, F.; Martínez-Cortizas, A.; Cuevas, C.A.; Saiz-Lopez, A. 700 years reconstruction of mercury and lead atmospheric deposition in the Pyrenees (NE Spain). Atmos. Environ. 2017, 155, 97–107. [Google Scholar] [CrossRef]
- Brooks, M.L.; Meyer, J.S.; McKnight, D.M. Photooxidation of wetland and riverine dissolved organic matter: altered copper complexation and organic composition. Hydrobiologia 2007, 579, 95–113. [Google Scholar] [CrossRef]
- Biester, H.; Pérez-Rodríguez, M.; Gilfedder, B.-S.; Martínez Cortizas, A.; Hermanns, Y.-M. Solar irradiance and primary productivity controlled mercury accumulation in sediments of a remote lake in the Southern Hemisphere during the past 4000 years: Primary productivity and mercury accumulation. Limnol. Oceanogr. 2018, 63, 540–549. [Google Scholar] [CrossRef]
- Outridge, P.M.; Sanei, H.; Stern, G.A.; Hamilton, P.B.; Goodarzi, F. Evidence for control of mercury accumulation rates in Canadian High Arctic Lake sediments by variations of aquatic primary productivity. Environ. Sci. Technol. 2007, 41, 5259–5265. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Rodríguez, M.; Gilfedder, B.-S.; Hermanns, Y.-M.; Biester, H. Solar Output Controls Periodicity in Lake Productivity and Wetness at Southernmost South America. Sci. Rep. 2016, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Gustin, M.S.; Engle, M.; Ericksen, J.; Lyman, S.; Stamenkovic, J.; Xin, M. Mercury exchange between the atmosphere and low mercury containing substrates. Appl. Geochem. 2006, 21, 1913–1923. [Google Scholar] [CrossRef]
- Lindberg, S.E. Forests and the global biogeochemical cycle of mercury: the importance of understanding air/vegetation exchange processes. In Global and Regional Mercury Cycles: Sources, Fluxes and Mass Balances; Springer: Dordrecht, The Netherlands, 1996; pp. 359–380. [Google Scholar]
- Witt, E.L.; Kolka, R.K.; Nater, E.A.; Wickman, T.R. Influence of the Forest Canopy on Total and Methyl Mercury Deposition in the Boreal Forest. Water Air Soil Pollut. 2009, 199, 3–11. [Google Scholar] [CrossRef]
- Kalis, A.J.; Merkt, J.; Wunderlich, J. Environmental changes during the Holocene climatic optimum in central Europe-human impact and natural causes. Quat. Sci. Rev. 2003, 22, 33–79. [Google Scholar] [CrossRef]
- Mayer, B.; Schwark, L. A 15,000-year stable isotope record from sediments of Lake Steisslingen, Southwest Germany. Chem. Geol. 1999, 161, 315–337. [Google Scholar] [CrossRef]
- Mayewski, P.A.; Rohling, E.E.; Stager, J.C.; Karlén, W.; Maasch, K.A.; Meeker, L.D.; Meyerson, E.A.; Gasse, F.; van Kreveld, S.; Steig, E.J.; et al. Holocene climate variability. Quat. Res. 2004, 62, 243–255. [Google Scholar] [CrossRef]
- Schönfelder, I.; Giesecke, T.H.; Gelbrecht, J.; Steinberg, C. Paleolimnological investigations on Late Glacial and Holocene sediments of lake Großer Treppelsee (Brandenburg). Berichte IGB 2000, 10, 171–184. [Google Scholar]
- Schönfelder, I.; Steinberg, C. Paläolimnologische Analyse Fluviatiler Sedimente der Spree; Final Report for DFG: Berlin, Germany, 1998. [Google Scholar]
- Schönfelder, I.; Steinberg, C.E.W. How did the nutrient concentrations change in northeastern german lowland rivers during the last four millennia?—A paleolimnological study of floodplain sediments. Stud. Quat. 2004, 129–138. [Google Scholar]
- Rösch, M. Land use and food production in central Europe from the Neolithic to the Medieval period: change of landscape, soils and agricultural systems according to archaeobotanical data. Econ. Archaeol. Struct. Perform. Eur. Archaeol. 2013, 109–127. [Google Scholar]
- Mariet, A.-L.; Monna, F.; Gimbert, F.; Walter-Simonnet, A.-V. Tracking past mining activity using trace metals, lead isotopes and compositional data analysis of a sediment core from Longemer Lake, Vosges Mountains, France. J. Paleolimnol. 2018, 60, 399–412. [Google Scholar] [CrossRef]
- Martín-Puertas, C.; Jiménez-Espejo, F.; Martínez-Ruiz, F.; Nieto-Moreno, V.; Rodrigo, M.; Mata, M.P.; Valero-Garcés, B.L. Late Holocene climate variability in the southwestern Mediterranean region: An integrated marine and terrestrial geochemical approach. Clim. Past 2010, 6, 807–816. [Google Scholar] [CrossRef]
- Shotyk, W.; Weiss, D.; Appleby, P.G.; Cheburkin, A.K.; Frei, R.; Gloor, M.; Kramers, J.D.; Reese, S.; Van Der Knaap, W.O. History of Atmospheric Lead Deposition Since 12,370 14C yr BP from a Peat Bog, jura Mountains, Switzerland; American Association of the of the Advancement of Sciences: Washington, DC, USA, 1998; pp. 1635–1640. [Google Scholar]
- Amos, H.M.; Sonke, J.E.; Obrist, D.; Robins, N.; Hagan, N.; Horowitz, H.M.; Mason, R.P.; Witt, M.; Hedgecock, I.M.; Corbitt, E.S.; et al. Observational and Modeling Constraints on Global Anthropogenic Enrichment of Mercury. Environ. Sci. Technol. 2015, 49, 4036–4047. [Google Scholar] [CrossRef] [PubMed]
- Engstrom, D.R.; Fitzgerald, W.F.; Cooke, C.A.; Lamborg, C.H.; Drevnick, P.E.; Swain, E.B.; Balogh, S.J.; Balcom, P.H. Atmospheric Hg Emissions from Preindustrial Gold and Silver Extraction in the Americas: A Reevaluation from Lake-Sediment Archives. Environ. Sci. Technol. 2014, 48, 6533–6543. [Google Scholar] [CrossRef] [PubMed]
- Bliedtner, M.; Martin, M. Erz-und Minerallagerstätten des Mittleren Schwarzwald; LRGB: Freiburg, Germany, 1986. [Google Scholar]
- Metz, R.; Richter, M.; Schürenberg, H. Die Blei-Zink-Erzgänge des Schwarzwaldes; Beihefte zum Geologischen Jahrbuch; Amt für Bodenforschung: Hannover, Germany, 1957. [Google Scholar]
- Breitenlechner, E.; Hilber, M.; Lutz, J.; Kathrein, Y.; Unterkircher, A.; Oeggl, K. The impact of mining activities on the environment reflected by pollen, charcoal and geochemical analyses. J. Archaeol. Sci. 2010, 37, 1458–1467. [Google Scholar] [CrossRef]
- Schwartz, M.O. Mercury in Zinc Deposits: Economic Geology of a Polluting Element. Int. Geol. Rev. 1997, 39, 905–923. [Google Scholar] [CrossRef]
- Lacerda, L.D. Global mercury emissions from gold and silver mining. Water Air Soil Pollut. 1997, 97, 209–221. [Google Scholar] [CrossRef]
- Rytuba, J.J. Mercury from mineral deposits and potential environmental impact. Environ. Geol. 2003, 326–338. [Google Scholar] [CrossRef]
- Brunke, E.-G.; Labuschagne, C.; Slemr, F. Gaseous mercury emissions from a fire in the Cape Peninsula, South Africa, during January 2000. Geophys. Res. Lett. 2001, 28, 1483–1486. [Google Scholar] [CrossRef]
- Friedli, H.R.; Radke, L.F.; Lu, J.Y. Mercury in smoke from biomass fires. Geophys. Res. Lett. 2001, 28, 3223–3226. [Google Scholar] [CrossRef]
- Nelson, S.J.; Johnson, K.B.; Kahl, J.S.; Haines, T.A.; Fernandez, I.J. Mass Balances of Mercury and Nitrogen in Burned and Unburned Forested Watersheds at Acadia National Park, Maine, USA. Environ. Monit. Assess. 2007, 126, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Ko, D.W.; Sparrow, A.D.; Weisberg, P.J. Land-use legacy of historical tree harvesting for charcoal production in a semi-arid woodland. For. Ecol. Manag. 2011, 261, 1283–1292. [Google Scholar] [CrossRef]
- Buntgen, U.; Tegel, W.; Nicolussi, K.; McCormick, M.; Frank, D.; Trouet, V.; Kaplan, J.O.; Herzig, F.; Heussner, K.-U.; Wanner, H.; et al. 2500 Years of European Climate Variability and Human Susceptibility. Science 2011, 331, 578–582. [Google Scholar] [CrossRef] [PubMed]
- Schmid, B.V.; Büntgen, U.; Easterday, W.R.; Ginzler, C.; Walløe, L.; Bramanti, B.; Stenseth, N.C. Climate-driven introduction of the Black Death and successive plague reintroductions into Europe. Proc. Natl. Acad. Sci. USA 2015, 112, 3020–3025. [Google Scholar] [CrossRef] [PubMed]
- Rabb, T.K. The Effects of the Thirty Years’ War on the German Economy. J. Mod. Hist. 1962, 34, 40–51. [Google Scholar] [CrossRef]
- Rumsby, B.T.; Macklin, M. River response to the last neoglacial (the ‘Little Ice Age’) in northern, western and central Europe. Geol. Soc. Lond. Spec. Publ. 1996, 115, 217–233. [Google Scholar] [CrossRef]
- Pierenkemper, T. Wirtschaftsgeschichte: Eine Einführung-Oder: Wie wir Reich Wurden; Oldenbourg: Leipzig, Germany, 2005. [Google Scholar]
- Fremdling, R. Modernisierung und Wachstum der Schwerindustrie in Deutschland, 1830–1860. Gesch. Ges. 1979, 5, 201–227. [Google Scholar]
- Joint Center for History and Economics Long-Term Energy Transitions, Germany. Available online: https://ourworldindata.org/energy-production-and-changing-energy-sources (accessed on 11 June 2018).
- Kander, A.; Malanima, P.; Warde, P. Power to the People: Energy in Europe over the Last Five Centuries; Princeton University Press: Princeton, NJ, USA, 2014. [Google Scholar]
- Fitzgerald, W.F.; Engstrom, D.R.; Hammerschmidt, C.R.; Lamborg, C.H.; Balcom, P.H.; Lima-Braun, A.L.; Bothner, M.H.; Reddy, C.M. Global and Local Sources of Mercury Deposition in Coastal New England Reconstructed from a Multiproxy, High-Resolution, Estuarine Sediment Record. Environ. Sci. Technol. 2018, 52, 7614–7620. [Google Scholar] [CrossRef] [PubMed]
- Pirrone, N.; Keeler, G.J.; Nriagu, J.O. Regional differences in worldwide emissions of mercury to the atmosphere. Atmos. Environ. 1996, 30, 2981–2987. [Google Scholar] [CrossRef]
| Schurmsee | Glaswaldsee | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Proxies | CP1 | CP2 | CP3 | CP4 | CP5 | CP6 | Proxies | CP1 | CP2 | CP3 | CP4 | CP5 | CP6 |
| C | 0.91 | 0.15 | 0.11 | 0.17 | −0.09 | 0.18 | C | −0.83 | −0.26 | 0.00 | 0.32 | 0.04 | −0.23 |
| N | 0.87 | 0.25 | 0.20 | 0.11 | 0.26 | 0.17 | N | −0.74 | −0.14 | 0.07 | 0.28 | −0.55 | −0.03 |
| Zr | −0.83 | 0.41 | −0.05 | −0.18 | 0.08 | −0.09 | Zr | 0.86 | 0.19 | 0.02 | 0.06 | 0.10 | 0.25 |
| Al | −0.89 | 0.27 | 0.24 | 0.01 | 0.08 | −0.06 | Al | 0.83 | 0.08 | 0.31 | −0.10 | −0.08 | −0.32 |
| Ti | −0.79 | 0.42 | 0.33 | −0.02 | 0.09 | 0.01 | Ti | 0.80 | 0.30 | 0.46 | 0.03 | 0.01 | −0.13 |
| Mn | 0.59 | 0.01 | 0.55 | 0.19 | 0.27 | 0.07 | |||||||
| rye | −0.13 | 0.95 | 0.10 | −0.09 | 0.04 | −0.02 | rye | 0.12 | 0.92 | 0.08 | −0.07 | −0.10 | 0.01 |
| ribwort | −0.14 | 0.89 | 0.25 | −0.07 | 0.11 | −0.04 | ribwort | 0.21 | 0.86 | 0.35 | 0.00 | 0.02 | 0.02 |
| Pb | 0.00 | 0.91 | 0.18 | −0.10 | 0.05 | −0.02 | Pb | 0.13 | 0.88 | 0.05 | −0.20 | −0.20 | 0.06 |
| spruce | −0.13 | 0.90 | −0.03 | −0.17 | 0.27 | 0.02 | spruce | 0.15 | 0.67 | 0.59 | −0.05 | 0.14 | 0.00 |
| Mn | 0.05 | −0.82 | −0.35 | −0.19 | 0.12 | 0.12 | charcoal | 0.17 | 0.67 | 0.14 | 0.31 | 0.10 | 0.17 |
| beech | 0.03 | 0.12 | 0.91 | −0.08 | 0.07 | −0.06 | beech | 0.22 | 0.17 | 0.85 | −0.24 | −0.11 | −0.18 |
| fir | 0.02 | 0.22 | 0.83 | 0.03 | 0.03 | 0.06 | fir | 0.17 | 0.20 | 0.84 | −0.21 | −0.01 | −0.22 |
| hazel | 0.16 | −0.36 | −0.65 | 0.13 | −0.21 | −0.30 | hazel | 0.03 | −0.29 | −0.80 | −0.23 | 0.25 | 0.16 |
| oak | 0.25 | −0.03 | −0.21 | 0.83 | 0.04 | 0.21 | oak | −0.42 | −0.08 | −0.45 | 0.68 | −0.27 | 0.05 |
| Fe | −0.02 | 0.39 | −0.39 | −0.60 | 0.25 | 0.33 | |||||||
| C:N | 0.13 | −0.27 | −0.33 | 0.11 | −0.84 | 0.00 | C:N | 0.08 | −0.16 | −0.14 | −0.05 | 0.91 | −0.25 |
| Zn | 0.21 | −0.12 | −0.35 | 0.43 | 0.55 | 0.40 | |||||||
| Cu | 0.37 | −0.16 | 0.17 | 0.13 | 0.05 | 0.80 | Cu | −0.13 | 0.04 | −0.16 | 0.00 | −0.22 | 0.90 |
| Zn | 0.04 | 0.01 | −0.24 | 0.16 | −0.19 | 0.86 | |||||||
| Fe | 0.38 | 0.20 | −0.03 | −0.17 | 0.23 | 0.79 | |||||||
| eigv. | 3.15 | 4.04 | 2.87 | 1.50 | 1.29 | 1.12 | eigv. | 4.19 | 3.70 | 3.48 | 1.05 | 1.59 | 2.62 |
| var. | 20 | 25 | 18 | 9 | 8 | 7 | var. | 22 | 19 | 18 | 6 | 8 | 14 |
| CP1 | CP2 | CP3 | CP4 | CP5 | CP6 | R | Error | |
|---|---|---|---|---|---|---|---|---|
| model 1a SCHURMSEE | ||||||||
| reg. coef. | −0.20 | 0.73 | 0.11 | −0.26 | 0.33 | 0.08 | 0.88 | 0.49 |
| t-value | −4.55 | 16.83 | 2.47 | −6.02 | 7.50 | 1.90 | ||
| p-value | <0.001 | <0.001 | <0.05 | <0.001 | <0.001 | <0.1 | ||
| model 1b SCHURMSEE | ||||||||
| reg. coef. | −0.21 | 0.67 | 0.36 | −0.07 | 0.37 | − | 0.87 | 0.50 |
| t-value | −4.59 | 14.90 | 8.16 | −1.62 | 8.25 | − | ||
| p-value | <0.001 | <0.001 | <0.001 | <0.1 | <0.001 | − | ||
| model 2a GLASWALDSEE | ||||||||
| reg. coef. | − | 0.68 | 0.44 | − | 0.11 | − | 0.82 | 0.59 |
| t-value | − | 10.81 | 6.92 | − | 1.74 | − | ||
| p-value | − | <0.001 | <0.001 | − | <0.1 | − | ||
| model 2b GLASWALDSEE | ||||||||
| reg. coef. | − | 0.70 | 0.09 | −0.17 | − | − | 0.80 | 0.62 |
| t-value | − | 10.50 | 5.06 | −2.55 | − | − | ||
| p-value | − | <0.001 | <0.001 | <0.05 | − | − | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schütze, M.; Tserendorj, G.; Pérez-Rodríguez, M.; Rösch, M.; Biester, H. Prediction of Holocene Mercury Accumulation Trends by Combining Palynological and Geochemical Records of Lake Sediments (Black Forest, Germany). Geosciences 2018, 8, 358. https://doi.org/10.3390/geosciences8100358
Schütze M, Tserendorj G, Pérez-Rodríguez M, Rösch M, Biester H. Prediction of Holocene Mercury Accumulation Trends by Combining Palynological and Geochemical Records of Lake Sediments (Black Forest, Germany). Geosciences. 2018; 8(10):358. https://doi.org/10.3390/geosciences8100358
Chicago/Turabian StyleSchütze, Martin, Gegeensuvd Tserendorj, Marta Pérez-Rodríguez, Manfred Rösch, and Harald Biester. 2018. "Prediction of Holocene Mercury Accumulation Trends by Combining Palynological and Geochemical Records of Lake Sediments (Black Forest, Germany)" Geosciences 8, no. 10: 358. https://doi.org/10.3390/geosciences8100358
APA StyleSchütze, M., Tserendorj, G., Pérez-Rodríguez, M., Rösch, M., & Biester, H. (2018). Prediction of Holocene Mercury Accumulation Trends by Combining Palynological and Geochemical Records of Lake Sediments (Black Forest, Germany). Geosciences, 8(10), 358. https://doi.org/10.3390/geosciences8100358







