The Influence of Cryogenic Mass Exchange on the Composition and Stabilization Rate of Soil Organic Matter in Cryosols of the Kolyma Lowland (North Yakutia, Russia)
Abstract
:1. Introduction
- (1)
- to evaluate the bulk content and elemental composition of humic acids (HAs), extracted from surface horizons, the central parts of soil profile, and the lower active layer;
- (2)
- to assess the molecular composition of the HAs by solid state 13C NMR resonance;
- (3)
- to evaluate the degree of HA molecule stabilization using electron spin resonance.
2. Study Area
3. Materials and Methods
3.1. Routine Soil Analyses and Microscopy
3.2. Extraction of Humic Acids
3.3. Characterization of HAs
3.4. Statistics
4. Results and Discussion
4.1. Genesis, Structure, and Morphology of Organo-Mineral Material
4.1.1. Modern Surface Organic and Organo-Mineral Horizons Undisturbed by Frost Boiling (Samples 1 and 3)
4.1.2. Fragments of Former Surface Organo-Mineral Horizons Buried after Solifluction Processes (Sample 4)
4.1.3. Fragments of Cryoturbated Organo-Mineral Material in the Central Parts of Cryosol Profiles (Sample 5)
4.1.4. Fragments of Cryoturbated Organo-Mineral Material Accumulated in the Lower Active Layer (Samples 2 and 6)
4.2. Total Organic Carbon and Nitrogen Content
4.3. Elemental Composition of HAs
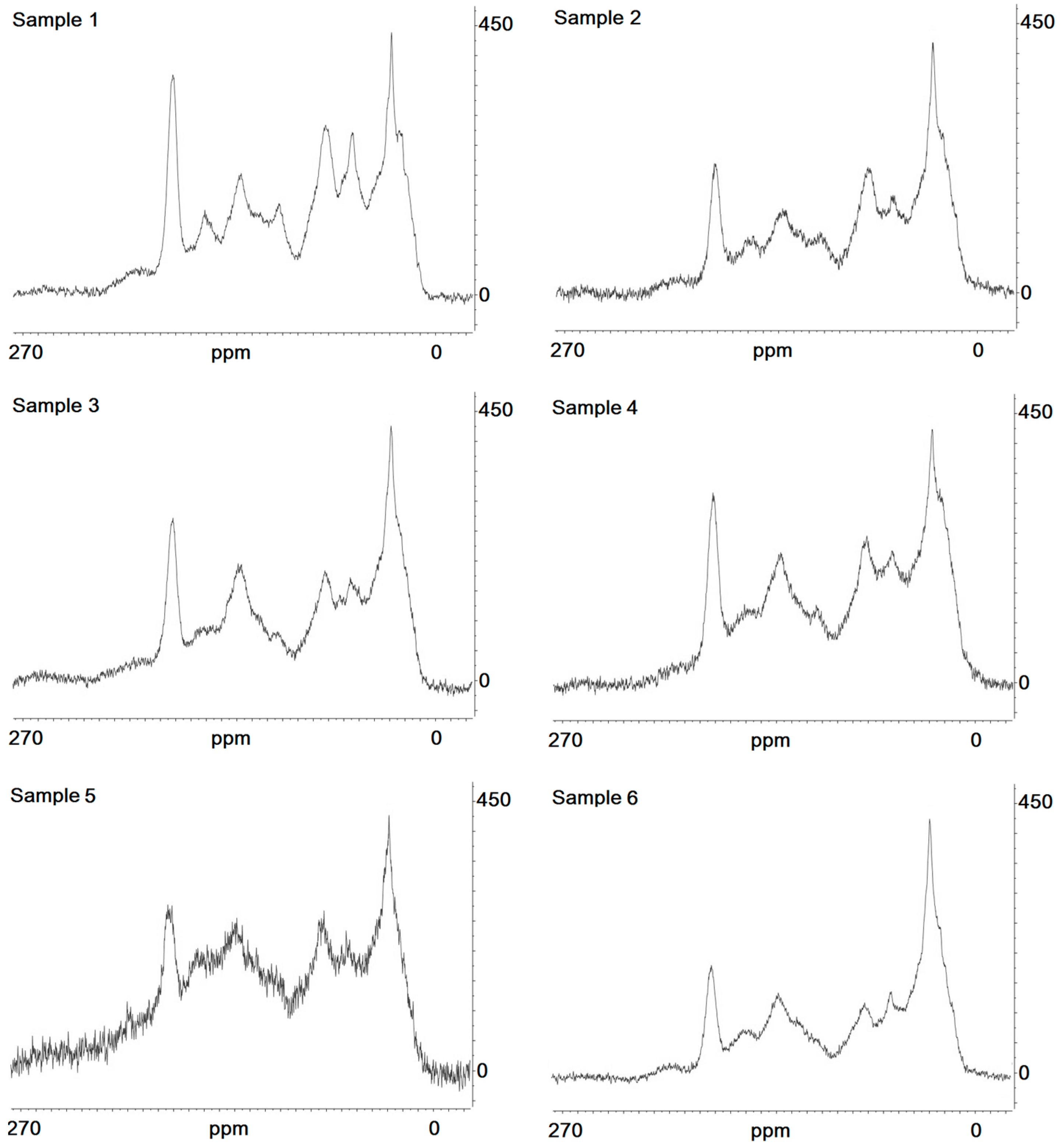
4.4. Structural Composition of the HAs
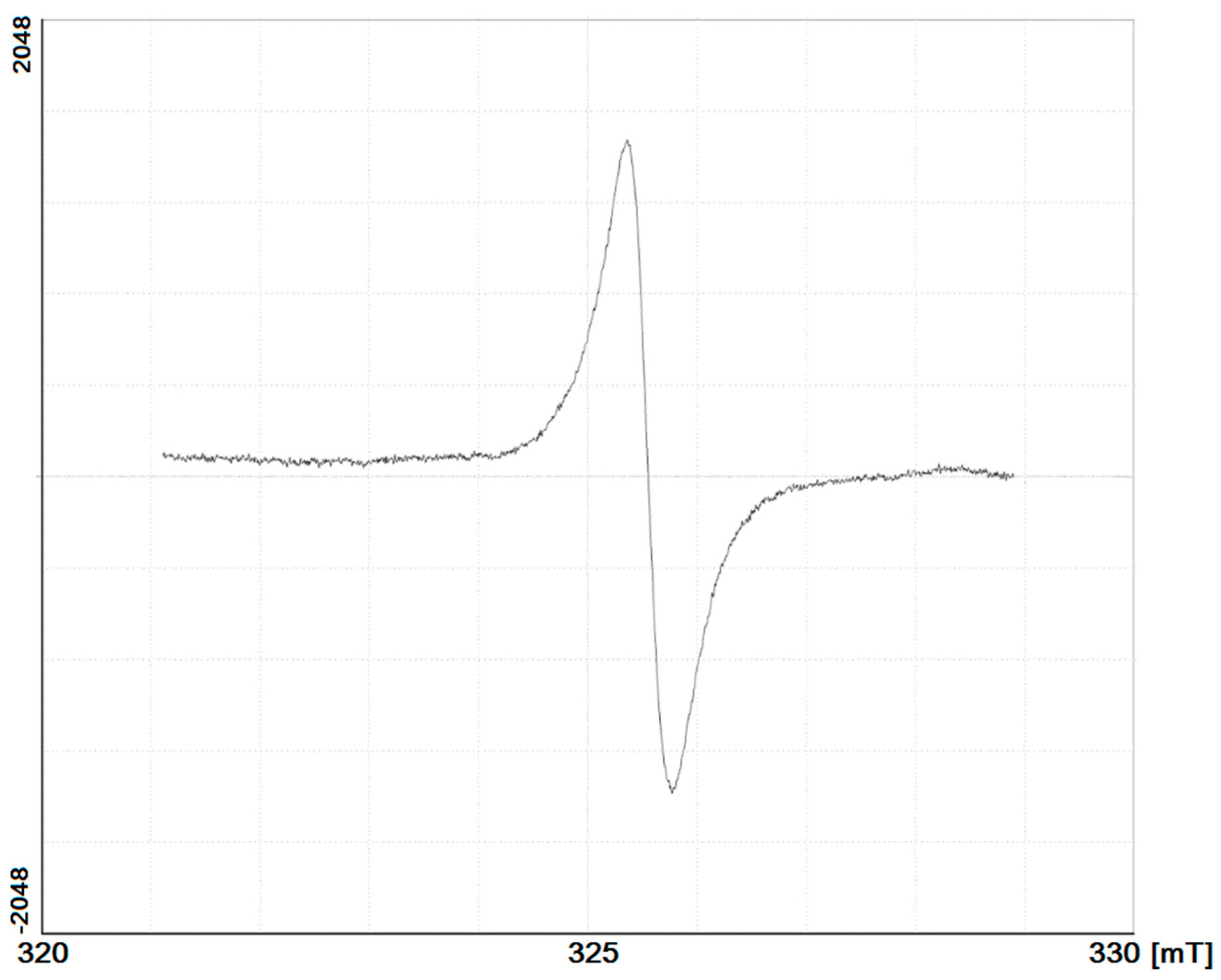
4.5. Biochemical Activity of the HAs: Insights from Spectroscopy of Electron Spin Resonance
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schädel, C.; Schuur, E.A.G.; Bracho, R.; Elberling, B.; Knoblauch, C.; Lee, H.; Luo, Y.; Shaver, G.R.; Turetsky, M.R. Circumpolar assessment of permafrost C quality and its vulnerability over time using long-term incubation data. Glob. Chang. Biol. 2014, 20, 641–652. [Google Scholar] [CrossRef] [PubMed]
- Zubrzycki, S.; Kutzbach, L.; Pfeiffer, E.-M. Permafrost affected soils and their carbon pools with a focus on the Russian Arctic. Solid Earth 2014, 5, 595–609. [Google Scholar] [CrossRef]
- Schuur, E.A.G.; McGuire, A.D.; Grosse, G.; Harden, J.W.; Hayes, D.J.; Hugelius, G.; Koven, C.D.; Kuhry, P. Climate change and the permafrost carbon feedback. Nature 2015, 520, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Roulet, N.T. Peatlands, carbon storage, greenhouse gases and the Kyoto protocol: Prospects and significance for Canada. Wetlands 2000, 20, 605–615. [Google Scholar] [CrossRef]
- Zamolodchikov, D.G.; Utkin, A.I.; Korovin, G.N.; Chestnykh, O.V. Dynamics of carbon pools and fluxes in Russia′s forest lands. Russ. J. Ecol. 2005, 36, 291–301. [Google Scholar] [CrossRef]
- Schädel, C.; Bader, M.K.-F.; Schuur, E.A.G.; Biasi, C.; Bracho, R.; Čapek, P.; De Baets, S.; Diáková, K.; Ernakovich, J.; Estop-Aragones, C. Potential carbon emissions dominated by carbon dioxide from thawed permafrost soils. Nat. Clim. Chang. 2016, 6, 950–953. [Google Scholar] [CrossRef]
- Tarnocai, C.; Canadell, J.G.; Schuur, E.A.G.; Kuhry, P.; Mazhitova, G.; Zimov, S. Soil organic carbon pools in the northern circumpolar permafrost region. Glob. Biogeochem. 2009, 23, 1–11. [Google Scholar] [CrossRef]
- Elberling, B.; Michelsen, A.; Schädel, C.; Schuur, E.A.G.; Christiansen, H.H.; Berg, L.; Tamstorf, M.P.; Sigsgaard, C. Long-term CO2 production following permafrost thaw. Nat. Clim. Chang. 2013, 3, 890–894. [Google Scholar] [CrossRef]
- Dai, X.Y.; Ping, C.L.; Michaelson, G.J. Characterizing soil organic matter in Arctic tundra soils by different analytical approaches. Org. Geochem. 2002, 33, 407–419. [Google Scholar] [CrossRef]
- Bockheim, J.G.; Tarnocai, C. Recognition of cryoturbation for classifying permafrost-affectеd soil. Geoderma 1998, 81, 281–293. [Google Scholar] [CrossRef]
- Lupachev, A.V.; Gubin, S.V. Suprapermafrost organic-accumulative horizons in the tundra Cryozems of Northern Yakutia. Eurasian Soil Sci. 2012, 45, 45–55. [Google Scholar] [CrossRef]
- Gubin, S.V.; Lupachev, A.V.; Shatilovich, A.V.; Mylnikov, A.P.; Ryss, A.Y.; Veremeeva, A.A. The influence of cryogenic mass exchange on the distribution of viable microfauna in Cryozems. Eurasian Soil Sci. 2016, 49, 1400–1413. [Google Scholar] [CrossRef]
- IUSS Working Group WRB. World Reference Base for Soil Resources 2014. In International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; World Soil Resources Reports No. 106; Food and Agriculture Organization (FAO): Rome, Italy, 2014. [Google Scholar]
- Walker, D.A.; Raynolds, M.K.; Daniëls, F.J.A.; Einarsson, E.; Elvebakk, A.; Gould, W.A.; Katenin, A.E.; Kholod, S.S.; Markon, C.J.; Melnikov, E.S.; et al. The Circumpolar Arctic vegetation map. J. Veg. Sci. 2005, 16, 267–282. [Google Scholar] [CrossRef]
- Murton, J.B.; Goslar, T.; Edwards, M.E.; Bateman, M.D.; Danilov, P.P.; Savvinov, G.N.; Gubin, S.V.; Ghaleb, B.; Haile, J.; Kanevskiy, M.; et al. Wolfe Palaeoenvironmental Interpretation of Yedoma Silt (Ice Complex) Deposition as Cold-Climate Loess, Duvanny Yar, Northeast Siberia. Permafr. Periglac. Process. 2015, 26, 208–288. [Google Scholar] [CrossRef]
- Russian Institute of Hydrometeorological Information—World Data Center. Available online: http://meteo.ru/english/index.php (accessed on 15 February 2017).
- Stuiver, M.; Reimer, P.J.; Reimer, R.W. CALIB 7.1. 2017. Available online: http://calib.org (accessed on 15 February 2017).
- Schnitzer, M. Organic matter characterization. In Methods of Soil Analysis; Page, B., Miller, R., Keeney, D., Eds.; Soil Science Society of America: Madison, WI, USA, 1982; pp. 581–594. [Google Scholar]
- Schnitzer, M. Soil organic matter—The next 75 years. Soil Sci. 1991, 151, 41–58. [Google Scholar] [CrossRef]
- Dai, X.Y.; Ping, C.L.; Candler, R.; Haumaier, L.; Zech, W. Characterization of soil organic matter fractions of tundra soils in arctic Alaska by Carbon-13 nuclear magnetic resonance spectroscopy. Soil Sci. Soc. Am. J. 2001, 65, 87–93. [Google Scholar] [CrossRef]
- Chefetz, B.; Simpson, M.J.; Deshmukh, A.P.; Hatcher, P.G. Structural components of humic acids as determined by chemical modifications and carbon-13 NMR, pyrolysis- and thermochemolysis-gas chromatography/mass spectrometry. Soil Sci. Soc. Am. J. 2002, 66, 1159–1171. [Google Scholar] [CrossRef]
- Frund, R.; Guggenberger, G.; Haider, K. Recent advances in the spectroscopic characterization of soil humic substances and their ecological relevance. Pflanzenernahr. Bodenkd. 1994, 157, 175–186. [Google Scholar] [CrossRef]
- Gonzalez-Perez, M.; Martin-Neto, L.; Saab, S.C.; Novotny, E.H.; Milori, D.M.B.P.; Bagnato, V.S.; Colnago, L.A.; Melo, W.J.; Knicker, H. Characterization of humic acids from a Brazilian Oxisol under different tillage systems by EPR, 13C NMR, FTIR and fluorescence spectroscopy. Geoderma 2004, 118, 181–190. [Google Scholar] [CrossRef]
- Chukov, S.N.; Abakumov, E.V.; Tomashunas, V.M. Characterisation of humic acids isolated from Antarctic soils by 13C NMR spectroscopy. Eurasian Soil Sci. 2015, 48, 1207–1211. [Google Scholar] [CrossRef]
- Lodygin, E.D.; Beznosikov, V.A.; Vasilevich, R.S. Molecular composition of humic substances in tundra soils (13C-NMR spectroscopic study). Eurasian Soil Sci. 2014, 47, 400–406. [Google Scholar] [CrossRef]
- Ejarque, E.; Abakumov, E. Stability and biodegradability of organic matter from Arctic soils of Western Siberia: insights from 13C-NMR spectroscopy and elemental analysis. Solid Earth 2016, 7, 153–165. [Google Scholar] [CrossRef]
- Abakumov, E.V.; Tomashunas, V.M. 13C NMR and ESR characterization of humic substances isolated from soils of two Siberian arctic islands. Int. J. Ecol. 2015, 1–7. [Google Scholar] [CrossRef]
- Senesi, N.; D′Orazio, V.; Ricca, G. Humic acids in the first generation of EUROSOILS. Geoderma 2003, 116, 325–344. [Google Scholar] [CrossRef]
- Senesi, N. Molecular and quantitative aspects of the chemistry of fulvic acids and its interactions with mineral ions and organic chemicals. Anal. Chim. Acta 1990, 232, 51–75. [Google Scholar] [CrossRef]
- Chukov, S.; Ejarque, E.; Abakumov, E. Characterisation of the humic acids of soils of tundra zone by electron spin resonance spectroscopy. Eurasian Soil Sci. 2017, 50, 35–39. [Google Scholar] [CrossRef]



| Sample Code | Loss on Ignition (%) | Total С (%) | Total N (%) | С/N | pH | Color (by Munsell System) | Radiocarbon Ages (yr BP) | Calibrated Ages 1σ (yr BP) |
|---|---|---|---|---|---|---|---|---|
| 1 | 11.33 | 3.57 | 0.27 | 13.32 | 5.30 | 7.5YR 4/2 | n/a (modern) | n/a |
| 2 | 14.75 | 6.64 | 0.36 | 18.54 | 5.60 | 5YR 3/2 | 1900 ± 40 | 1813–1897 |
| 3 | 12.43 | 1.64 | 0.15 | 10.74 | 5.93 | 7.5YR 4/2 | n/a (modern) | n/a |
| 4 | 14.67 | 1.42 | 0.13 | 10.64 | 6.78 | 7.5YR 3/2 | 2370 ± 50 | 2299–2351 |
| 5 | 13.76 | 5.23 | 0.34 | 15.36 | 6.05 | 10YR 3/2 | 720 ± 50 | 649–698 |
| 6 | 15.24 | 5.58 | 0.35 | 15.72 | 6.10 | 10YR 6/1 | 1530 ± 50 | 1461–1513 |
| Sample Code | C | H | N | O | C/N | H/C | O/C |
|---|---|---|---|---|---|---|---|
| 1 | 47.20 | 5.71 | 4.25 | 43.01 | 10.97 | 0.12 | 0.91 |
| 2 | 46.17 | 5.52 | 3.48 | 44.99 | 13.12 | 0.12 | 0.98 |
| 3 | 46.82 | 5.29 | 4.04 | 44.02 | 11.46 | 0.11 | 0.94 |
| 4 | 36.08 | 4.36 | 3.27 | 56.37 | 10.92 | 0.12 | 1.57 |
| 5 | 48.26 | 4.29 | 3.93 | 43.7 | 12.14 | 0.09 | 0.91 |
| 6 | 48.24 | 5.59 | 3.52 | 42.83 | 13.56 | 0.12 | 0.89 |
| Sample Code | Chemical Shift (ppm) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0–27 | 27–32 | 32–45 | 45–60 | 60–95 | 95–110 | 110–140 | 140–160 | 160–185 | 185–200 | |
| 1 | 9.59 | 9.72 | 4.78 | 13.07 | 4.01 | 13.86 | 4.41 | 10.50 | 5.80 | 17.22 |
| 2 | 7.59 | 6.60 | 5.49 | 12.26 | 3.72 | 13.73 | 13.41 | 12.47 | 6.36 | 19.33 |
| 3 | 8.92 | 7.97 | 6.32 | 15.10 | 3,05 | 12.28 | 12.62 | 12.66 | 6.99 | 14.05 |
| 4 | 8.09 | 8.44 | 6.06 | 14.70 | 3.18 | 13.25 | 13.59 | 13.63 | 4.13 | 14.93 |
| 5 | 8.55 | 9.82 | 8.86 | 16.34 | 4.49 | 14.63 | 11.79 | 12.79 | 3.20 | 9.49 |
| 6 | 6.45 | 7.20 | 5.58 | 14.78 | 2.69 | 10.27 | 11.18 | 14.74 | 8.02 | 19.03 |
| p One-way ANOVA | <0.04 | <0.04 | <0.04 | <0.05 | <0.05 | <0.08 | <0.03 | <0.05 | <0.05 | <0.07 |
| Sample Code | Aromatic (%) | Aliphatic (%) |
|---|---|---|
| 1 | 27.24 | 72.76 |
| 2 | 25.76 | 74.24 |
| 3 | 28.25 | 71.75 |
| 4 | 27.85 | 72.14 |
| 5 | 34.62 | 65.37 |
| 6 | 26.57 | 73.42 |
| Sample Code | Mass Concentration of Free Radical, 1015 spin·g−1 | g-Factor |
|---|---|---|
| 1 | 1.206 × 1015 | 1.98888 |
| 2 | 4.496 × 1014 | 1.98887 |
| 3 | 2.090 × 1015 | 1.98863 |
| 4 | 9.307 × 1014 | 1.98860 |
| 5 | 5.23 × 1014 | 1.98860 |
| 6 | 1.059 × 1014 | 1.98866 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lupachev, A.; Abakumov, E.; Gubin, S. The Influence of Cryogenic Mass Exchange on the Composition and Stabilization Rate of Soil Organic Matter in Cryosols of the Kolyma Lowland (North Yakutia, Russia). Geosciences 2017, 7, 24. https://doi.org/10.3390/geosciences7020024
Lupachev A, Abakumov E, Gubin S. The Influence of Cryogenic Mass Exchange on the Composition and Stabilization Rate of Soil Organic Matter in Cryosols of the Kolyma Lowland (North Yakutia, Russia). Geosciences. 2017; 7(2):24. https://doi.org/10.3390/geosciences7020024
Chicago/Turabian StyleLupachev, Alexey, Evgeny Abakumov, and Stanislav Gubin. 2017. "The Influence of Cryogenic Mass Exchange on the Composition and Stabilization Rate of Soil Organic Matter in Cryosols of the Kolyma Lowland (North Yakutia, Russia)" Geosciences 7, no. 2: 24. https://doi.org/10.3390/geosciences7020024
APA StyleLupachev, A., Abakumov, E., & Gubin, S. (2017). The Influence of Cryogenic Mass Exchange on the Composition and Stabilization Rate of Soil Organic Matter in Cryosols of the Kolyma Lowland (North Yakutia, Russia). Geosciences, 7(2), 24. https://doi.org/10.3390/geosciences7020024








